Abstract
Laurel (Laurus nobilis L.) has been used in the Mediterranean basin since ancient ages. Nowadays, Turkey, Mexico, Portugal, Italy, Spain, France, Algeria, and Morocco use aromatic leaves for commercial purposes, and Turkey is the largest exporter in the world. In this study, molecular characterization, and genetic relationships of 94 Turkish laurel genotypes were determined by ISSR and SCoT markers. The experiment was conducted with 16 ISSR and 10 SCoT markers. While 348 of 373 bands were polymorphic with a 93.3% polymorphism rate, Nei’s genetic distances ranged between 0.17 and 0.70 with 0.39 mean in ISSR. In SCoT, 175 of 227 bands were polymorphic with 77.1% polymorphism rate, and Nei’s genetic distances varied between 0.12 and 0.51. Sufficient genetic diversity determined with diversity parameters consisting of the average Shannon’s information index (ISSR: 0.46, SCoT:0.35), the overall gene diversity (ISSR:0.19, SCoT:0.18), and the effective number of alleles (ISSR:1.52, SCoT:1.38). AMOVA (Analysis of molecular variance) revealed most of the variation was within genotypes (96%). Neighbor-joining algorithms, principal coordinate analysis (PCoA), and model-based structure resulted in harmony and clustered according to the geographical regions and provinces they collected. Genotypes were divided into two groups in ISSR and SCoT with UPGMA clustering resulting in a similar polymorphism distribution. The correlation coefficient (r) determined by marker systems’ Nei’s genetic distances was 0.25. The results of the study put forward resources for advanced breeding techniques, contribute to the preservation of genetic diversity, and management of genetic resources for the breeders.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Medicinal plants are mainly used in medicines to prevent diseases, maintain healthiness, or heal illnesses, body care, nutrition, cosmetics, incense, and religious ceremonies, while aromatic plants are used to maintain a pleasant smell and taste [1]. The importance of these plants is increasing day by day with people’s tendency towards nature and natural sources in the nutrition, health, and cosmetic sectors [2]. The necessity and significance of medicinal and aromatic plants usage in routine life will never decline, and it will maintain its popularity among the scientific community [1]. Medicinal plants have become increasingly recognized especially in the last decade, particularly in the role of rural people’s economies, scope in medicinal practices, use in cultural matters, and ultimately their contribution to the well-being of people [1, 3].
Laurel is not widely recognized compared to other medicinal plants worldwide despite having a high economic value and popularity in Turkey. Currently, it has many synonyms as ‘laurel’, ‘sweet bay’, ‘bay leaf’, and ‘bay-tree’. The laurel plant originated in Anatolia and the Balkans. It naturally grows up to 600–800 m altitudes through Turkey coastline [4]. Laurel trees are generally grown in America and Europe as an ornamental plant. Besides, it is one of the most popular plants in the Mediterranean region having an enormous variety [1]. Its aromatic leaves are grown for commercial purposes in Turkey, Mexico, Portugal, Italy, Spain, France, Algeria, and Morocco [5, 6]. Both essential oils and fixed oils of laurel are used in food, cosmetics, and medicine [1]. Laurel leaves are also extensively used in folk medicine to treat rheumatism, gastrointestinal, and urinary problems [1, 7]. Turkey is the largest exporter in the world at laurel trade [5]. In addition to its essential oil, some of which are exported, laurel has many uses in Turkey [1].
In 21st-century, crop breeding programs researching natural biodiversity as a source of novel alleles to increase adaptability, yield, health properties, and nutritional value of crops have gained considerable importance. Genetic variations identification in plants was performed by physiological, morphological, and cytological features previously. However, morphological data is inadequate in determining genetic differences due to a gene affects more than one character, and that one character recognized more than one locus. The trend towards molecular methods has increased due to the insufficiency of these data in plant systematics descriptions for taxonomy studies in recent years. Hereby molecular techniques were developed concerning DNA for genotyping [8, 9]. ISSR, one of the PCR-based marker techniques containing multi-allelic loci, can quickly show the differences between closely related individuals very informatively. This is a low-cost technique developed by Zietkiewicz et al. [10] that detecting polymorphisms in microsatellites and requires a small amount of template DNA. ISSR outputs a large number of fragments and reproducibility [11]. Primers in this technique are generally based on a repeated sequence range by degenerate 3′ anchor and amplify the sequence among two microsatellites [12]. Gene targeted markers are currently very popular in developing new markers, and generate polymorphisms from the exon, intron, or promoter regions of the genes that may be directly related to gene functions [13]. SCoT, a member of gene-targeted markers, is a simple and reliable marker system developed by Collard and Mackill [14]. Compared to other marker systems, SCoT markers provide more information on biological properties and universality in plants. They also resemble inter simple sequence repeat or random amplified polymorphic DNA but have the advantage of close linkage with the target [14]. They have many benefits compared to other markers thanks to high polymorphism and reproducibility [15]. In recent studies, SSR and SCoT provided remarkably high polymorphism in the various plants [16,17,18,19]. ISSR and SCoT widely preferred for genetic diversity analysis because of their simple procedures, excellent stability, not affected by environmental factors and high reproducibility [16, 17].
Genetic diversity of laurel plant has been investigated in some studies. In brief, limited number of molecular characterization studies have been performed related to this subject on local laurel genotypes. However, the germplasm size or/and the number of molecular markers used previously are relatively small except for the work of Karik et al. [20]. Therefore, more extensive regional studies were required. There are only two studies that have been conducted with ISSR markers so far [21, 22], but the research area sizes were very small compared to this study. On the other hand, SCoT has not been studied yet. In light of previous studies, the opportunity to examine the genetic diversity in laurel plants in wider areas with different marker systems has emerged for this study. On this occasion, Turkish laurel germplasm and its current variations were identified at the molecular level in this study, and the fundamental information was established for future breeding studies by utilizing these variations.
Materials and methods
Plant material and DNA extraction
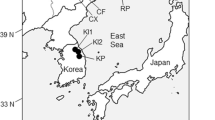
Breeding materials that comprised 94 laurel genotypes collected from 26 Turkish provinces of 4 geographical regions provided from AARI (Aegean Agricultural Research Institute) were tested in this study (Supplementary Table 1, Fig. 1). The Mediterranean and Aegean Regions have hot and dry summers, winters are mild and rainy. Snowfall and frost are rare in the coastal zone. Winters are cold and snowy at higher altitudes. The Black Sea Region has relatively cool summers, warm winters in the coastal areas, and snowy and cold higher areas. It is rainy in all seasons and there is no water shortage. In the Marmara Region, the winters are not as warm as the Mediterranean climate, and the summers are not as rainy as the Black Sea climate. It is not as cold in winter and dry in summer as the continental climate. Because of these features, the Marmara climate is a transitional climate between the continental Black Sea and Mediterranean climates.
Young leaves of the plants were harvested and brought to the laboratory at the optimum time. DNAs isolated following the CTAB protocol [23] with some minor modifications [24]. İsolated DNA samples were separated on 0.8% agarose gel and concentrations were measured using NanoDrop (DeNovix DS-11 FX). The optimum level for PCR amplification was determined as 5 ng µL−1. The stocks were diluted up to this concentration and kept at – 25 ℃ until analyses.
ISSR and SCoT amplification
Some modifications were made on the protocols used by Ali et al. [17] for ISSR and by Collard and Mackill [14] for SCoT. 16 ISSR and 10 SCoT primers were used in this study. PCR amplification for ISSR had 20 μL solution with 3 μL template DNA (5 ng μL−1), 2 mM dNTPs (Thermo Scientific), 1.5 μL MgCl2, 2 μL 10XPCR buffer (Thermo Scientific), 0.8 μL primer, 0.2 μL U Taq DNA polymerase (Thermo Scientific), and 9.5 μL ddH2O. PCR cycle consisted of 3 min denaturation at 94 ℃, 1 min of 30 denaturation cycles at 94 ℃, 1 min of annealing temperature at 48–54 ℃ depending upon the primer, 1 min of extension step at 72 ℃, and 10 min of final extension at 72 ℃. Followings were different for SCoT; 2.5 μL MgCl2, 1.5 μL primer, and 7.8 μL ddH2O. The reaction cycle consisted of 5 min denaturation at 94 ℃, 1 min of 35 denaturation cycles at 94 ℃, 1 min of annealing temperature at 50 ℃, 2 min of extension step at 72 ℃, and 8 min of final extension at 72 ℃, using BIO-RAD T100 Thermal Cycler.
The amplified DNA products obtained were loaded onto a 2% agarose gel and separated in 4 h (for ISSR) and 5 h (for SCoT) at 120 V. After the gels were removed from the electrophoresis system and put into ethidium bromide for 5 min and passed through pure water. At the end of the process, the gels were imaged under UV and recorded for analysis.
Data analysis
Clear, unambiguous, and strong bands were selected for scoring. The binary system is used by scanning as 1 and 0 according to the presence or absence of specific bands, among the markers according to 100 bp+ DNA ladder. Polymorphism information content (PIC) was determined according to the method of Baloch et al. [25] for ISSR and SCoT. PopGene ver. 1.32 [26] was used to predict various important genetic diversity parameters like; gene diversity (H), Shannon's Information Index (I), effective alleles number (NE), overall gene diversity (HT), and Nei's genetic distance (GD). This study’s genotypes were supplied from 4 geographical regions (Aegean, Mediterranean, Black Sea, Marmara) in Turkey. For this reason, some diversity values calculated on a regional basis using PopGene ver. 1.32 [26]. AMOVA (Analysis of molecular variance) and UPGMA (The unweighted pair group method with arithmetic mean) were performed using R software [27] to explore the level of diversity among laurel germplasm. STRUCTURE software (version 2.3.4, Stanford, CA, USA) was used to reveal the population structure of genotypes with the Bayesian clustering model’s help. The cluster numbers (the number of K; the number of subpopulations) were determined following the protocol of Evanno et al. [28] was plotted and followed the K (number of clusters) against logarithm possibility relative to the standard deviation (ΔK). A total of 10 independent runs were set for each K value, and each run, the beginning burn-in period was set to 5000 with 10,000 MCMC (Markov chain Monte Carlo) iterations. Mantel test was performed with GENALEX ver.6.51 [29] to determine linear relationship between Nei's genetic distances of ISSR and SCoT.
Results
ISSR genetic diversity
A total of 373 clean and scorable bands were identified, with an average of 23.31 per primer. Among the scoreable bands, 348 (93.3% of total) were polymorphic, with an average of 21.75 per primer. Primer UBC890 had the highest number of bands (46) and the highest number of polymorphic bands (42). The least scored (13) primers were UBC812 and UBC825. The average PIC (Polymorphism information content) was 0.70, and while the highest PIC was obtained from UBC825 with 0.80, the lowest PIC was obtained from UBC810 with 0.54. The mean, the highest, and the lowest NE (Effective alleles number) were 1.52, 1.73 (UBC814), and 1.42 (UBC888), respectively. The mean genetic diversity (H) was 0.31, the highest was 0.39 (UBC814), and the lowest was 0.25 (UBC888). The mean Shannon Information Index (I) was 0.46, the highest was 0.56 (UBC814), and the lowest was 0.38 (UBC888). Overall gene diversity (HT) values were between 0.06 (UBC815) and 0.29 (UBC810), with an average of 0.19 (Table 1). Nei's genetic distances were calculated to evaluate genetic diversity, with paired genetic distance measurement in 94 laurel genotypes. The mean genetic distance among genotypes was 0.39, while the highest was 0.70 (Antalya1 and Zonguldak5), and the lowest was 0.17 (Sinop5 and Zongudak5) (Supplementary Fig. 1).
SCoT genetic diversity
A total of 227 clean and scorable bands were identified, with an average of 22.7 per primer. Among the 227 scorable bands, 175 (77.1% of total) were polymorphic, with an average of 17.5 polymorphic bands per primer. Primer SCoT-2 had the highest number of bands (34). The least scored (16) primers were SCoT-7, SCoT8, SCoT10, and SCoT11. Primer SCOT-1 had the highest polymorphic bands (28). The average PIC was 0.45, the highest PIC was obtained from SCoT-5 with 0.63, and the lowest PIC was obtained from SCoT-8 with 0.15. The mean, the highest, and the lowest NE were 1.38, 1.52 (SCoT-6/SCoT-10), and 1.13 (SCoT-8), respectively. The mean H was 0.23, the highest was 0.31 (SCoT-10), and the lowest was 0.09 (SCoT-8). The mean I was 0.35, the highest was 0.46 (SCoT-6), and the lowest was 0.11 (SCoT-8). HT values were between 0.08 (SCoT-8) and 0.24 (SCoT-1) with an average value of 0.18 (Table 1). The average genetic distance between genotypes was 0.26, the highest genetic distance (0.51) was found between Antalya5 and Zonguldak2, and the lowest genetic distance (0.12) between Bartin2 and Istanbul2 (Supplementary Fig. 2).
Clustering analysis of ISSR and SCoT results
STRUCTURE software divided 94 laurel genotypes into two main populations by the Bayesian clustering model according to ISSR and SCoT data (Fig. 2). K values were also obtained from the STRUCTURE software were used to detect the true number of laurel subpopulations (Fig. 3). Both results showed that two populations were formed in the Turkish laurel germplasm, supporting each other.
Populations were classified according to their existence in 4 geographical regions (Black Sea Region, Marmara Region, Aegean Region, and Mediterranean Region) to detect better analyze the variation. From this point, to understand the population structure more clearly, laurel germplasm was divided into two populations according to the Bayesian-based clustering algorithm. Population A included mostly genotypes from the Mediterranean region. Population B had mostly genotypes from the Aegean and Marmara regions. A high rate of the Black Sea region’s genotypes was present in population A (Fig. 4). Although few differences were detected between ISSR and SCoT, a high level of consistency was found between population structures (Figs. 2, 4).
As a result of AMOVA, the variation within the population was higher than the variation among populations. The majority of genetic variations occurred as 96% in the population sets in AMOVA for both markers (Table 2).
Genetic diversity was calculated according to the collection regions (Mediterranean, Aegean, Marmara, and Black Sea) of 94 laurel genotypes. Genotypes collected from the Black Sea region showed a higher number of gene diversity (0.28), effective alleles (1.49), expected heterozygosity (0.20), and Shannon’s information index (0.43) according to ISSR analysis. Besides, genotypes of Mediterranean showed higher values except for Ht in SCoT analysis (Table 3).
Neighbor-joining clustering analysis performed to determine the genetic relationships by showing the distribution among 94 laurel genotypes based on their provinces and geographical regions is given in supplementary Fig. 1 and Fig. 2 The biplot graph showing the PCoA analysis performed to separate genotypes according to their geographical regions is also given in supplementary Fig. 3.
The linear relationship of ISSR and SCoT was determined with mantel test. The correlation coefficient was 0.25 according to Nei’s genetic distances of the two marker systems. Mantel test graph is given in Fig. 5.
Discussion
Polymorphism in Turkish laurel germplasm
A total of 16 ISSR and 10 SCoT markers were used to determine the genetic diversity of the Turkish laurel germplasm (Table 1). Total and polymorphic band numbers were higher than Karik et al.'s [16] iPBS-Retrotranspozon (195/164), Sevindik's [22] ISSR (46/22), and Arroyo-Garcia et al.'s [30] AFLP (167/100) studies on laurel. Also, Mohamed et al. [21] worked widely on the Lauraceae family using ISSR markers (209/165), and findings were lesser than ours. The polymorphism mean in this study was 93.3% for ISSR markers and 77.1% for SCoT markers. The rate of ISSR was higher than reported by Karik et al.'s [20] 84.1% and Sevindik's [22] 47.8%. High polymorphism obtained in this study is thought to occur due to differentiation of marker systems, population sizes, and primer numbers. Moreover, the difference between polymorphism ratios of ISSR and SCoT determined in this study supports these circumstances. PIC, which is the value for the exploration of discriminating power marker among genotypes [31], and efficiency of polymorphic loci [32], was the mean of 0.70 in ISSR and 0.45 in SCoT. These values were higher than reported by Karik et al.'s [20] PIC value (0.36) in laurel accessions. When the marker systems were compared, ISSR and SCoT had a higher value than iPBS-Retrotransposon in terms of PIC in Turkish laurel germplasm. Additionally, PIC values in some previous ISSR and SCoT studies, 0.35 in rice with ISSR [33], 0.35 in safflower with SCoT [34], 0.32 in sesame with ISSR [35], in salvia 0.38 and 0.40 with ISSR and SCoT respectively [36], 0.30 in olive with ISSR [16], and 0.45 in safflower with ISSR [17] did not exceed the results of this study. Besides, Agarwal et al.'s [18] PIC value (0.79) of SCoT exceeded the PIC of SCoT in this study. In another study with both marker systems (ISSR and SCoT), Igwe et al.'s [19] PIC values in Vigna unguiculata L. (Walp) accessions were higher than the PIC values of this study. In comparison to previous studies, the findings of this study mostly had higher PIC values. The reason for the low SCoT PIC value in this study is due to the number of polymorphic bands of the markers. It is seen that polymorphic bands in SCoT markers were relatively low compared to ISSR markers. In addition, this difference has increased even more, especially due to a large number of monomorphic bands in the SCoT-8 marker. Markers pair with different regions according to the presence and absence of different base sequences in the genome. The data obtained in this study proved that ISSR markers are able to capture these differences remarkably exhibiting greater variation in the laurel plant compared to both the SCoT (in this study) and the other marker systems in the literature. These facts prove the ISSR's discriminative power between genotypes and the efficiency of polymorphic loci.
Genetic diversity of Turkish laurel germplasm
One of the most crucial steps towards breeding activities is genetic diversity assessment since it provides a new source of variation [37]. Local genotypes underwent genetic changes over time due to biotic and abiotic factors, and they have been the main source of agricultural production for centuries [38]. Therefore, it is important to collect and preserve local genetic resources and to evaluate them appropriately. Diagnosing of laurel genetic sources at the molecular level is important in terms of laurel breeding activities to be carried out in Turkey. Turkish laurel germplasm's genetic diversity identified using ISSR and SCoT markers (Table 1). The maximum number of effective alleles is always an indispensable parameter for detecting genetic variation since it reveals more genetic variation. The average number of effective alleles in this study was 1.52 in ISSR, and 1.38 in SCoT, which was higher than Agarwal et al. [39] (1.36) with ISSR, and Gogoi et al. [40] (1.33 and 1.19) with SCoT and ISSR respectively. In another study, Dilipan et al. [41] obtained a 1.44 Ne value from SCoT and exceeded ours. Mean gene diversity results (ISSR: 0.308; SCoT: 0.230) higher than reported in other studies [42,43,44,45], and the germplasm in this study appears to have higher diversity. Shannon's information index (I) is a crucial parameter for understanding genetic diversity variation by integrating equality and abundance [46]. The mean Shannon information indexes in this study (ISSR:0.46; SCoT: 0.35) were also higher than some previous studies [40, 42]. The relatively high values of the number of effective alleles, gene diversity, and Shannon information index determined in this study show Turkish laurel germplasm has higher genetic diversity compared to other studies in the literature. Thereby, the marker systems used in this study were considered quite convenient in genetic diversity analysis.
In this study, the distances between sampling regions were quite consistent with the genetic distances. These results confirm the fact that geographical distance is one of the main causes of genetic differentiation between populations by barring gene flow [47]. Detection of the highest genetic distances from genotypes of Antalya and Zonguldak provinces in both marker systems shows that more focus should be given to these provinces for sake of breeding studies. The most important information obtained from genetic distances in testing plant breeding methods is selecting genotypes with the furthest distance from each other since the gene pool is desired as large as possible. In this perspective, according to ISSR results, Zonguldak5 and Antalya1 genotypes, according to the SCoT results, Kastamonu2 and Antalya5 genotypes, can be the most recommended genotypes as parents for laurel breeding. Various diversity parameters were also calculated according to the geographical regions where the genotypes were collected to elucidate the genetic variations in more detail (Table 3). According to ISSR results, The Black Sea region and according to the SCoT results, the Mediterranean region showed a higher level of diversity than other regions. These regions' common feature is that they are superior to the other two regions in geographical distances between genotypes. On this occasion, it clearly seems that expanding the geographical boundaries will expand the gene pool gathered.
According to AMOVA, higher variations were identified within the laurel genotypes as 96% (in both analyses). Higher variations within the genotypes may be due to genetic drift, pollination, gene flow, adaptation, and ecotypes variation [48]. Also, environmental transformations and human activities may be responsible for this situation [49]. Neighbor clustering analysis aims to group genotypes according to their geographical regions and provinces, was performed for 94 Turkish laurel germplasm. Apart from the Aegean region, the same regions’ genotypes were generally together. In Turkey, laurel plants are native to Hatay, Mersin, Adana, and Kahramanmaras provinces (Mediterranean Region) [5]. Over time, with the farmers’ attraction to this plant, it spread to Marmara and Black Sea regions. The mixture in this region is very high since it passes from all regions to the Aegean region after these regions. Genotypes were divided into geographical regions where they collected with PCoA (Supplementary Fig. 3). The conflict regarding the Aegean region’s laurel gene pool was proved by this study, which gave results to the degree that supports this hypothesis. Aegean region genotypes appeared as mixture of 3 regions’ genotypes, supporting the gene flow hypothesis. According to the model-based structure algorithm, genotypes were divided into two population groups to their provinces and corresponding geographical regions (Fig. 2). Genetic distances of 94 laurel genotypes in 4 geographical regions were separated as Population A and B. Population A contained mostly genotypes of the Mediterranean region except for three genotypes. Population B contained mainly genotypes from the Aegean and Marmara regions. A high rate of the Black Sea’s genotypes was present in population A (Fig. 3). Gene escapes were observed between these two populations, albeit in small amounts, due to the possibility of seed changes and some crossbreeding.
The comparison of genetic distances by the Mantel test is one of the most popular ways to interpret spatial processes driving population structure [47]. In this study, the correlation coefficient was 0.25 (r). In the molecular characterization study conducted by Singh et al. [50] on jujube cultivars using 14 ISSR and 17 SCoT primers, the correlation coefficient was 0.24 (r). Compared to the mentioned study, our correlation coefficient was similar. This low correlation between SCoT and ISSR markers may be due to the specificity of each primer to divergent target regions in the genomes.
Conclusions
Determination of the degree and distribution of genetic diversity is crucial for future breeding studies to be improved. A remarkable genetic diversity was achieved among 94 laurel populations. ISSR and SCoT markers were utilized in this study, and they were evaluated as very effective to detect the genetic diversity of laurel germplasm. Zonguldak5 and Antalya1 and Kastamonu2 and Antalya5 genotypes had the highest genetic distances in the ISSR and SCoT, respectively. These genotypes are considered the most recommended genotypes as parents for laurel breeding. In general, there was almost no difference between the two marker systems in evaluating genetic distances of the laurel populations. However, when 94 laurel genotypes are assessed in terms of all genetic diversity parameters, the ISSR marker system resulted in more informatively. On the other hand, SCoT markers are known to be produced from the functional region of the genome. Therefore, we suggest that these markers should be used in QTL mapping and the construction of linkage maps. Since high genetic diversity was obtained within vast geographical boundaries, the main purpose of the next studies should be to develop new varieties of laurel.
References
Yilmaz A, Ciftci V (2021) Status of laurel plant (Laurus nobilis L.) in Turkey. Eur J Sci Technol 22:325–330
Yilmaz A, Guler E, Soydemir HE, Demirel S, Mollahaliloglu S, Karadeniz T, Ciftci V (2021) Miracle plant: Aronia (Aronia melanocarpa). MAS J Appl Sci 6:83–94. https://doi.org/10.52520/masjaps.20
Srivastava A, Srivastava P, Pandey A, Khanna VK, Pant AB (2019) Phytomedicine: a potential alternative medicine in controlling neurological disorders. In new look to phytomedicine. Academic Press, Cambridge, pp 625–655
Davis PH (1982) Flora of Turkey, vol 7. Edinburg University Press, Edinburg, p 947
Nadeem MA, Aasim M, Kırıcı S, Karık Ü, Nawaz MA, Yılmaz A, Maral H, Khawar KM, Baloch FS (2018) Laurel (Laurus nobilis L.): a less-known medicinal plant to the world with diffusion, genomics, phenomics, and metabolomics for genetic improvement. In: Kumar N (ed) Biotechnological approaches for medicinal and aromatic plants. Springer, Singapore, pp 631–653
Onay Ö (2014) Effects of catalyst on pyrolysis of laurel (Laurus nobilis L.) seed in a fixed bed tubular reactor. Chem Eng Trans 37:127–132
Sellami IH, Wannes WA, Bettaieb I, Berrima S, Chahed T, Marzouk B, Limam F (2011) Qualitative and quantitative changes in the essential oil of Laurus nobilis L. leaves as affected by different drying methods. Food Chem 126:691–697
Gulsen O, Mutlu N (2005) Genetic markers used in plant sciences and their utilization. Alatarim 4:27–37
Nadeem MA, Nawaz MA, Shahid MQ, Doğan Y, Comertpay G, Yıldız M, Hatipoglu R, Ahmad F, Alsaleh A, Labhane N, Özkan H, Chung G, Baloch FS (2018) DNA molecular markers in plant breeding: current status and recent advancements in genomic selection and genome editing. Biotechnol Biotechnol Equip 32:261–285. https://doi.org/10.1080/13102818.2017.1400401
Zietkiewicz E, Rafalski A, Labuda D (1994) Genome fingerprinting by simple sequence repeat (SSR)-anchored polymerase chain reaction amplification. Genomics 20:176–183
Jafari SH, Sepehry A, Soltanloo H, Karimian AA (2019) Genetic differentiation between bitter and sweet asafetida plants using ISSR markers. Mol Biol Rep 46:1069–1078. https://doi.org/10.1007/s11033-018-4565-1
Ghariani S, Amari M, Charfeddine A, Chakroun M, Trifi-Farah N (2019) Molecular and morpho-agronomic characterization among introduced bristle oat collection using ISSR fingerprinting. Agron J 111:156–164. https://doi.org/10.2134/agronj2017.11.0672
De Keyser E, De Riek J, Van Bockstaele E (2009) Discovery of species-wide EST-derived markers in Rhododendron by intron-flanking primer design. Mol Breed 23:171–178
Collard BC, Mackill DJ (2009) Start codon targeted (SCoT) polymorphism: a simple, novel DNA marker technique for generating gene-targeted markers in plants. Plant Mol Biol Rep 27:86
Sadhu S, Jogam P, Thampu RK, Abbagani S, Penna S, Peddaboina V (2020) High efficiency plant regeneration and genetic fidelity of regenerants by SCoT and ISSR markers in chickpea (Cicer arietinum L.). Plant Cell Tissue Organ Cult (PCTOC) 141:465–477
Sesli M, Yegenoglu ED (2017) Genetic relationships in wild olives (Olea europaea ssp. oleaster) by ISSR and RAPD markers. Biotechnol Biotechnol Equip 31:897–904
Ali F, Nadeem MA, Habyarimana E, Yılmaz A, Nawaz MA, Khalil IH, Ercisli S, Chung G, Chaudhary HJ, Baloch FS (2020) Molecular characterization of genetic diversity and similarity centers of safflower accessions with ISSR markers. Braz J Bot 43:109–121. https://doi.org/10.1007/s40415-019-00574-7
Agarwal A, Gupta V, Haq SU, Jatav PK, Kothari SL, Kachhwaha S (2019) Assessment of genetic diversity in 29 rose germplasms using SCoT marker. J King Saud Univ-Sci 31:780–788. https://doi.org/10.1016/j.jksus.2018.04.022
Igwe DO, Afiukwa CA, Ubi BE, Ogbu KI, Ojuederie OB, Ude GN (2017) Assessment of genetic diversity in Vigna unguiculata L. (Walp) accessions using inter-simple sequence repeat (ISSR) and start codon targeted (SCoT) polymorphic markers. BMC Genet 18:98. https://doi.org/10.1186/s12863-017-0567-6
Karık Ü, Nadeem MA, Habyarimana E, Ercişli S, Yildiz M, Yılmaz A, Baloch FS (2019) Exploring the genetic diversity and population structure of Turkish laurel germplasm by the iPBS-retrotransposon marker system. Agronomy 9:647
Mohamed ASH, Ahmed W, Rabia SS, Mourad MM (2016) Implications of morphology and molecular criteria in taxonomy of Lauraceae Juss. Egypt J Exp Biol 12:45–52
Sevindik E (2019) Molecular genetic diversity of some Laurus nobilis L. (Lauraceae) populations grown in The Aegean Region/Turkey. Afs-Adv Food Sci 41:28–31
Doyle JJ, Doyle JL (1990) Isolation of plant DNA from fresh tissue. Focus 12:13–15
Baloch FS, Alsaleh A, Andeden EE, Hatipoglu R, Nachit M, Ozkan H (2016) High levels of segregation distortion in the molecular linkage map of bread wheat representing West Asia and NorthAfrica region. Turk J Agric For 40:352–364. https://doi.org/10.3906/tar1508-27
Baloch FS, Alsaleh A, de Miera LES, Hatipoğlu R, Çiftçi V et al (2015) DNA based iPBS-retrotransposon markers for investigating the population structure of pea (Pisum sativum) germplasm from Turkey. Biochem Syst Ecol 61:244–252. https://doi.org/10.1016/j.bse.2015.06.017
Yeh TBJ, Yang FC, Boyle RC (2000) Popgen ver 1.32: The user-friendly shareware for population genetic analysis. Molecular biology and biotechnology center. University of Alberta, Alberta
Schliep KP (2011) Phangorn: phylogenetic analysis in R. Bioinformatics 27:592–593
Evanno G, Regnaut S, Goudet J (2005) Detecting the number of clusters of individuals using the software STRUCTURE: a simulation study. Mol Ecol 14:2611–2620
Peakall ROD, Smouse PE (2006) GENALEX 6: genetic analysis in Excel. Population genetic software for teaching and research. Mol Ecol Notes 6:288–295
Arroyo-García R, Martínez-Zapater JM, Prieto JF, Álvarez-Arbesú R (2001) AFLP evaluation of genetic similarity among laurel populations (Laurus L.). Euphytica 122:155–164
Nemli S, Kianoosh T, Tanyolac MB (2015) Genetic diversity and population structure of common bean (Phaseolus vulgaris L.) accessions through retrotransposon-based inter primer binding sites (iPBSs) markers. Turk J Agric For 39:940–948. https://doi.org/10.3906/tar-1505-59
Gedik A, Ates D, Erdogmus S, Comertpay G, Tanyolac MB, Ozkan H (2017) Genetic diversity of Crocus sativus and its close relative species analyzed by iPBS-retrotransposons. Turk J Agric For 22:243–252. https://doi.org/10.17557/tjfc.357426
Törün B, Sözen E (2018) Determination of the genetic variation among some Turkish rice (Oryza sativa L.) cultivars using ISSR technique. J Anim Plant Sci 28:1766–1773
Golkar P, Mokhtari N (2018) Molecular diversity assessment of a world collection of safflower genotypes by SRAP and SCoT molecular markers. Physiol Mol Biol Plants 24:1261–1271. https://doi.org/10.1007/s12298-018-0545-0
Kurt C, Arioglu H (2018) Assessment of genetic diversity among Turkish sesame (Sesamum indicum L.) genotypes using ISSR markers. IJAER 4(1):188–199
Etminan A, Pour-Aboughadareh A, Noori A, Ahmadi-Rad A, Shooshtari L, Mahdavian Z, Yousefiazar-Khanian M (2018) Genetic relationships and diversity among wild salvia accessions revealed by ISSR and SCoT markers. Biotechnol Biotechnol Equip 32:610–617. https://doi.org/10.1080/13102818.2018.1447397
Guliyev N, Sharifova S, Ojaghi J, Abbasov M, Akparov Z (2018) Genetic diversity among melon (Cucumis melo L.) accessions revealed by morphological traits and ISSR markers. Turk J Agric For 42:393–401. https://doi.org/10.3906/tar-1707-18
Camacho Villa TC, Maxted N, Scholten M, Ford-Lloyd B (2006) Defining and identifying crop landraces. Plant Genet Resour 3:373–384
Agarwal T, Gupta AK, Patel AK, Shekhawat NS (2015) Micropropagation and validation of genetic homogeneity of Alhagi maurorum using SCoT, ISSR, and RAPD markers. Plant cell Tissue Organ Cult (PCTOC) 120:313–323. https://doi.org/10.1007/s11240-014-0608-z
Gogoi B, Wann SB, Saikia SP (2020) Comparative assessment of ISSR, RAPD, and SCoT markers for genetic diversity in Clerodendrum species of North East India. Mol Biol Rep 47:7365–7377. https://doi.org/10.1007/s11033-020-05792-x
Dilipan E, Ramachandran M, Arulbalachandran D (2020) Population genetics and gene flow of the seagrass, Syringodium isoetifolium based on start codon targeted (SCoT) marker from Palk Bay and Chilika Lake India. Meta Gene 26:100774. https://doi.org/10.1016/j.mgene.2020.100774
Tanya P, Taeprayoon P, Hadkam Y, Srinives P (2011) Genetic diversity among Jatropha and Jatropha-related species based on ISSR markers. Plant Mol Biol Rep 29:252–264
Elframawy A, Deif H, El-Bakatoushi R (2016) Genetic variation among fragmented populations of Atriplex halimus L. using start codon targeted (SCoT) and ITS1-5.8 S-ITS2 region markers. Am J Mol Biol 6:101–115. https://doi.org/10.4236/ajmb.2016.62011
Zabet M, Rahimi A, Izanlo A, Alizadeh Z (2019) Investigation of genetic variation in cumin (Cuminum cyminum) ecotypes of Khorasan Provinces using RAPD and ISSR markers. Agric Biotechnol J 11:75–98. https://doi.org/10.22103/jab.2019.12102.1050
Hadipour M, Kazemitabar SK, Yaghini H, Dayani S (2020) Genetic diversity and species differentiation of medicinal plant Persian Poppy (Papaver bracteatum L.) using AFLP and ISSR markers. Ecol Genet Genom 16:100058. https://doi.org/10.1016/j.egg.2020.100058
Ali F, Yılmaz A, Nadeem MA, Habyarimana E, Subaşı I, Nawaz MA, Chaudhary HJ, Shahid MQ, Ercisli S, Zia MAB, Chung G (2019) Mobile genomic element diversity in world collection of safflower (Carthamus tinctorius L.) panel using iPBS-retrotransposon markers. PLoS ONE 14:e0211985. https://doi.org/10.1371/journal.pone.0211985
Diniz-Filho JAF, Soares TN, Lima JS, Dobrovolski R, Landeiro VL, Telles MPDC, Bini LM (2013) Mantel test in population genetics. Genet Mol Biol 36:475–485
Pour AH, Karahan F, Ilhan E, Ilçim A, Haliloglu K (2019) Genetic structure and diversity of Adonis L. (Ranunculaceae) populations collected from Turkey by inter-primer binding site (iPBS) retrotransposon markers. Turk J Bot 43:585–596. https://doi.org/10.3906/bot-1810-1
Solouki M, Mehdikhani H, Zeinali H, Emamjomeh AA (2008) Study of genetic diversity in Chamomile (Matricaria chamomilla) based on morphological traits and molecular markers. Sci Hortic 117:281–287
Singh SK, Chhajer S, Pathak R, Bhatt RK, Kalia RK (2017) Genetic diversity of Indian jujube cultivars using SCoT, ISSR, and rDNA markers. Tree Genet Genomes 13:12
Acknowledgements
This study was produced from the doctoral dissertation number 620825 conducted at the Bolu Abant İzzet Baysal University Institute of Science. The authors express their gratitude to the BAP (Institution of Scientific Research Projects—Bolu Abant Izzet Baysal University) for their support. The authors are thankful to Associate Prof. Dr. Faheem Shahzad BALOCH, Assistant Prof. Dr. Muhammad Azhar NADEEM, and Dr. Emrah GULER for assistance in the experiments.
Author information
Authors and Affiliations
Contributions
Conceptualization: AY. Data curation: AY. Methodology: AY. Resource: AY. Software: AY. Supervision: VC. Validation: VC. Writing—original draft: AY. Writing—review & editing: VC.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
The whole or part of this article has not been sent/published in any other journal.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yilmaz, A., Ciftci, V. Genetic relationships and diversity analysis in Turkish laurel (Laurus nobilis L.) germplasm using ISSR and SCoT markers. Mol Biol Rep 48, 4537–4547 (2021). https://doi.org/10.1007/s11033-021-06474-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-021-06474-y