Abstract
The tissue-protective properties of erythropoietin (EPO) have been described in several neurodegenerative diseases models, but erythrocytosis following EPO treatment may lead to deleterious effects. Carbamylated erythropoietin, an EPO derivative lacking hematopoietic side effects, has shown protective properties in some studies. However, it is not known if CEPO protects primary hippocampal cells against Aβ25−35 toxicity. The present study aimed to investigate the effect of CEPO-Fc on biochemical alterations in Akt, GSK-3β, and ERK signaling and cell death induced by Aβ25−35 in isolated hippocampal cell culture. The embryonic hippocampal cells were obtained from 18–19 day rat embryos. The cells were exposed with Aβ25−35 (20 µM) in the absence or presence of CEPO-Fc (1 or 5 IU) and PI3k and ERK inhibitors. CEPO-Fc at the dose of 5 IU significantly prevented the cell loss and caspase-3 cleavage caused by Aβ25−35. Additionally, CEPO-Fc noticeably reversed Aβ mediated decrement of Akt and GSK-3β phosphorylation. With exposure to LY294002, PI3 kinase inhibitor, Akt phosphorylation diminished and CEPO-Fc protective effects disappeared. Furthermore, while CEPO-Fc nullified Aβ-induced increment of phospho-ERK, inhibition of ERK activity by PD98059, had no effect on Aβ25−35-mediated caspase-3 cleavage and cell toxicity. These results imply that protective effects of CEPO-Fc seem to be mainly exerted through the PI3K/Akt pathway rather than ERK signaling. This study suggested that CEPO-Fc prevents Aβ-induced cell toxicity as well as Akt/GSK-3β and ERK alterations in isolated hippocampal cells. These findings might provide a new perspective on CEPO-Fc protective properties as a prospective remedial factor for neurodegenerative diseases like AD.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Alzheimer’s disease (AD) is a complex neurodegenerative disease, which is known as the most common cause of dementia. One of the most important structures connected with AD pathology is hippocampus. Specifically, the characteristics of this disease including extracellular deposits of β-amyloid peptide (Aβ) (known as senile plaques), neurofibrillary tangles, and loss of neurons have been observed in hippocampus [1, 2]. It has been suggested that toxicity of Aβ is the main cause of neuronal damage in AD. Aβ is a sequence of 40–42 amino acids (therefore named as Aβ1−42) in which its toxicity is mainly caused by sequence of 25–35 amino acids of this protein (named as Aβ25−35). This sequence is considered as the active site of Aβ [3, 4]. Numerous in-vitro experiments have suggested the toxicity of Aβ peptide for neurons. Furthermore, in transgenic mice with large amounts of Aβ production, pathological symptoms of AD such as behavioral defects are observed. Thus, it has been proposed that Aβ plays a pivotal role in the etiology of AD [5]. There are various studies suggesting that dysregulation of some intracellular signaling pathways are associated with increased production and accumulation of Aβ that can contribute to development of neurodegenerative diseases. For example, it has been reported that cellular toxicity of Aβ can impair natural signaling of Akt, Glycogen Synthase Kinase-3β (GSK-3β), and Extracellular Signal-Regulated Kinase (ERK) [6,7,8].
Erythropoietin (EPO) is a glycohormone with known hematopoietic activities and neuroprotective effects [9]. In the central nervous system (CNS), EPO exerts its biological effects through binding to EPO receptors (EPORs) expressed in cerebral cortex, midbrain, and hippocampus [10, 11]. Various different in vivo and in vitro studies have indicated the neuroprotective effects of recombinant human EPO (rhEPO) [12,13,14]. Nevertheless, in the CNS, the therapeutic dose of EPO is far greater than its dose for hematological effects. Therefore, the main restriction of using EPO is that its long-term use will result in overproduction of red blood cells and elevated blood viscosity and other side effects associated with the hematopoietic system [15, 16]. On the other hand, EPO has a relatively short serum half-life. Carbamylated erythropoietin (CEPO) (a chemical-modified derivative of EPO) has been reported to exhibit the protective effects of rhEPO in various studies [17, 18], but does not bind to the EPOR and thus does not affect the erythropoiesis process. In this regards, experimental evidence indicated that the protective effects of CEPO is mediated through binding a heteroreceptor named EPOR-βcR (β-common receptor or CD131) [19,20,21]. For instance, Ding et al. (2015) showed that CEPO could not support the proliferation of Neuro-2a cells because the lack of CD131 in this cell line [22]. On the other hand, the CEPO protective effects on cell lines that do not express the EPOR also confirm the fact that CEPO protective signaling occurs via binding to a separate receptor from that of EPOR [23]. It has also been reported that CEPO can improve memory in another manner from the EPO function [24]. In a recent in-vivo study, we found that CEPO-Fc (linked to the Fc portion of human IgG1 molecule leading to a prolonged half-life) was able to prevent memory deficit resulting from Aβ25−35 in rats. These observed effects were parallel with changes and modifications in the activity of some signaling molecules including MAPKs and Akt/GSK-3β [8]. Since most studies have investigated the protective effects of EPO and rhEPO while the molecular mechanisms of CEPO protective effects at cellular levels have remained unknown, thus the purpose of the current work was to study the protective effects of CEPO-Fc against Aβ25−35 toxicity in isolated hippocampal cells. Further, at the molecular level, we measured the effect of CEPO-Fc on Aβ25−35-mediated Caspase-3 activation, and changes in Akt/GSK-3β and ERK activity. Additionally, Akt and ERK inhibitors were applied for investigating the possible interplay between these signaling molecules in Aβ25−35 toxicity and/or CEPO-Fc protection.
Materials & methods
Reagents and antibodies
Aβ25− 35 (A4559), Thiazolyl Blue Tetrazolium Blue (MTT) powder (M5655), Sodium pyruvate (P5280), Hanks’ balanced salt (HBS, H2387) and Poly-l-lysine solution (P4707) were from Sigma Aldrich. Preparation of CEPO-Fc was done in our lab (Vienna, Austria), which is described in more detail in succeeding sections. Cell culture materials including DMEM/F12, (32500-035), FBS, (10270-106) and Penicillin-Streptomycin Antibiotic (15140-122) were purchased from Gibco® life technologies™. Selective PI3 kinase inhibitor (LY294002 (ab120243)) was purchased from Abcam, and selective mitogen-activated protein kinase (MEK) inhibitor (PD98059 (9900L)) was from Cell Signaling Technology Company. The following antibodies were also purchased from Cell Signaling Technology Company: Caspase-3 antibody (9665), Beta-actin antibody (4970), the phosphorylated form of antibodies including Akt(Ser473), GSK-3β(Ser9), ERK (with Cat # 4060, 5558, 4377 respectively), their corresponding total forms including Akt, GSK-3β and ERK (with Cat # 4685, 9315, 4695 respectively), and anti-rabbit horseradish peroxidase (HRP) secondary antibody (7074). Western Blotting Detection Reagent kit (Amersham™ ECL™ Select) (RPN2235) was from GE healthcare. Immobilon-P PVDF Membrane (IPVH00010) and Bovine Serum Albumin (BSA, 1120180100) were purchased from Millipore. Halt Protease/phosphatase inhibitor cocktail (78440) was from Thermofisher Scientific.
Culture of hippocampal cells
We performed the experiments based on the animal ethics committee of Shahid Beheshti University of Medical Sciences and in accordance with NIH guidelines. Primary hippocampal cultures were obtained from embryonic rat brains (18–19 day-old), as we described earlier [7, 25]. The hippocampi were gently separated and collected in sterile HBSS (Ca2+ and Mg2+ free Hanks’ balanced salt solution, with 10 mM HEPES, 1 mM sodium pyruvate). For digestion of the hippocampi, they were exposed to trypsin (0.05%) at 37 °C for 20 min. Afterward, the tissues were washed with HBSS and then using a sterile fire-polished Pasteur pipette the tissue gently triturated (8–10 times trituration). After leaving the homogenate on ice (10–15 min) for precipitation of debris, the supernatant was centrifuged (2000 rpm/2 min) and hippocampal cells were pelleted. The cells were re-suspended in DMEM/F12 (1:1) plating medium (containing FBS (10%) and 100 U/ml penicillin and 100 µg/ml streptomycin). After cells counting, they were added to 96-well and 6-well culture plates (coated with Poly-l-lysine) for MTT assay and Western blotting analysis respectively. The cells were seeded at a density of 40 × 103 cells/well in the 96-well plate and 1.2 × 106 cells/well in the 6 well plate. After incubating the cells for 4 days (at 37 °C, 95% humidified atmosphere and 5% CO2), medium was replaced by fresh DMEM/F12 (1:1) medium with FBS (1%) and cell treatments were done.
Drug preparation
A main stock of Aβ25−35 (2 µg/µl) was prepared through dissolving in sterile distilled water and stored in − 20 °C. According to the protocols of previous studies [25, 26], the in vitro incubation of Aβ25–35 (at 37 °C for 4 days) was done for its aggregation process. Production and biochemical characterization of EPO-Fc was previously described by Schriebl et al, this molecule is a fusion protein consisting of two rhEPO molecules linked to the Fc region of a human IgG1 molecule [27]. The EPO-FC complex is subsequently carbamylated until no erythropoietic potency remains. Carbamylation is performed by incubation with potassium cyanate (KOCN) at pH 8 and 37 °C for 48 h. Lysine residues react with potassium cyanate and are modified to homocitrulline. Thereafter unbound cyanate is removed by gel filtration and thereby buffered into PBS pH 7.3. Due to carbamylation hematopoietic properties are lost, but neuroprotective activities are preserved. The main stock (1.91 mg/ml or 2.3 × 105 IU) of CEPO-Fc was prepared by dissolving in phosphate buffered saline and then diluted in medium for treating the cells.
Cell treatments
Cell treatments by aggregated Aβ25−35 (previously incubated for 4 days at 37 °C), CEPO-Fc, or both, with or without PD98059 and LY294002 were done 4 days after seeding the hippocampal cells. Treatments with different drugs were done for 24 h. MTT assay was used to determine toxic dose of Aβ25−35 and protective dose of CEPO-Fc. Accordingly; we selected 20 µM of Aβ25−35 as a toxic dose and 1 and 5 IU of CEPO-Fc for further studies. The inhibitors were dissolved in dimethyl sulfoxide (DMSO) in a stock concentration, which the final DMSO concentration did not exceed 0.1% when added to the cells medium. According to our previous study, the dose 50 µM of PD98059 or LY294002 were selected for treatments [25].
Cell viability assay
The viability of cultured hippocampal cells was assessed using the MTT assay. Twenty-four hours after cell treatments, the medium was completely removed and replaced by MTT solution (0.5 mg/ml); then the cells were incubated for 4 more h at 37 oC. Afterwards, the medium was replaced by DMSO (100 µl/each well). A microplate reader (Synergy HT, Biotek®) was used to measure absorbance at 570 nm. Furthermore, by an inverted phase contrast microscope, we visualized the morphological changes of cells.
Western blot analysis
Before lysing the plated cells, they were rinsed with cold HBSS and then lysed and scraped in a cold RIPA (Radioimmunoprecipitation assay) lysis buffer. This buffer was comprised of Tris-HCl (50 mM, pH8.0), NaCl (150 mM), Triton X-100 (1%), Na-Deoxycholate (0.5%), SDS (sodium dodecyl sulfate, 0.1%)), as well as, protease/phosphatase inhibitors cocktail. Lysate samples were homogenized by needling. Then, the homogenate was centrifuged at 4 °C (13,000 rpm for 25 min). Lowry assay was used for evaluating protein concentration of samples. Protein extracts of each sample (20 µg) were loaded on SDS-PAGE gel electrophoresis (12%) and transferred onto PVDF membranes. BSA (5%) was used for blocking process of the membranes (1 h, room temperature). Then, overnight incubation of blots was done with primary antibodies (Akt, GSK-3β, ERK, caspase-3 and β-actin) at 4 °C. Thereafter, blots were probed with secondary antibody for 1:30 h at room temperature. Using chemiluminescence ECL reagent, the protein bands were then detected. For detection of the total form of proteins and also β-actin, the blots were placed in stripping solution (containing Tris-base (1 M, PH6.8), SDS (10%) and 2-Mercaptoethanol (0.4%)) for 25 min at 54 °C. Image-J software was used to quantify the density of protein bands.
Statistics
Data were analyzed with GraphPad Prism software, version 7.01. All experiments were repeated 3–4 times. Comparisons were performed by one-way analysis of variance (ANOVA) followed by Tukey's post hoc test with a significance level set at p < 0.05. The results are expressed as means ± SEM.
Results
The effects of Aβ25−35, CEPO-Fc, LY294002 and PD98059 on hippocampal cell viability and morphology
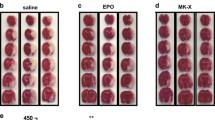
In order to assess the cell viability, MTT assay was performed. The toxic effect of Aβ25−35 was assessed for 4 different doses (5, 10, 20 and 40 µM). (Fig. 1a). According to One-way ANOVA analysis, Aβ25−35 significantly induced hippocampal cell death (F(4, 28) = 16.20, P < 0.0001). Post hoc analysis showed that Aβ25−35 at doses 10 (P < 0.05), 20 and 40 µM (P < 0.001) significantly decreased cell viability in a concentration-dependent manner. In this regards, we considered to select a dose of Aβ25−35 that its toxicity could be reversed by treatment with CEPO-Fc. Since the higher dose (40 µM) caused severe toxicity, treatment with CEPO could not exhibit the protective effects against its toxicity and prevent cell death. Consequently, the dose 20 µM was selected for further experiments. Furthermore, other published studies also have used the dose of 20 µM in their studies. In the same way, the effect of CEPO-Fc at doses 1, 2, 5 and 10 IU against Aβ-induced cell death was assessed by MTT assay (Fig. 1b). One-way ANOVA analysis presented significant differences between groups (F(9, 147) = 10.49, P < 0.0001). Post hoc analysis by Tukey’s test showed that, however, CEPO-Fc treatment at dose 1 IU had no significant effect on Aβ-induced cell death but, doses 2 (P < 0.01), 5 and 10 IU (P < 0.001) markedly reversed cell loss induced by Aβ25−35. Accordingly, two doses of CEPO-Fc (1 and 5 IU) were selected for the rest of experiments. Figure 1c depicts the effects of LY294002 and PD98059 on the protective effect of CEPO-Fc. One-way ANOVA analysis showed a significant difference between groups (F(6,54) = 23.63, P < 0.0001). Tukey’s test revealed that the protective effect of CEPO-Fc is reversed following LY294002 administration (P < 0.001). However, PD98059 did not affect the protective properties of CEPO-Fc. Additionally, the results also showed that LY294002 (at the Aβ20µM + LY group) exacerbated Aβ-induced toxicity (P < 0.05), while PD98059 had not effect on Aβ25−35 toxicity. Figure 2 represents the pics of morphological changes of the cultured cells that captured 24 h after treatments with different drugs. In Aβ25−35-treated group, as shown, Aβ-induced morphological changes including cell body shrinkage and increased dead cells and cell debris are evident. CEPO-Fc treatment at dose 5 IU could prevent these morphological changes. Following LY294002 exposure, not only the protective effect of CEPO-Fc on cells was obviously omitted but also Aβ-induced morphological changes was potentiated. However, PD98059 did not affect the protective properties of CEPO-Fc.
The effect of CEPO-Fc on Aβ25−35 toxicity in the presence or absence of PI3K and ERK inhibitors in cultured hippocampal cells. The cell viability was evaluated by MTT assay 24 hours after treatments. a The cell viability following Aβ25−35 exposure at doses 5, 10, 20 and 40 µM. b The effect of CEPO-Fc at doses 1, 2, 5 and 10 IU on Aβ20µM. c The cell viability after treatments with Aβ20µM and/or CEPO-Fc5IU in the presence or absence of LY294002 and PD98059. Data are reported as mean ± SEM. *P < 0.05 and ***P < 0.001 represent the difference between control versus other treated groups. #P < 0.05, ##P < 0.01 and ###P < 0.001 represent the difference between Aβ20µM group and the others
The effects of Aβ25−35, CEPO-Fc, LY294002 and PD98059 on caspase-3, Akt, GSK-3β and ERK alterations in cultured hippocampal cells
In order to investigate the protective effects of CEPO-Fc against Aβ25−35 toxicity and to see the contribution of caspase-3, Akt, GSK-3β and ERK activity in this interaction, western blotting analysis was done. In the case of proteins with two form of phospho and total (Akt, GSK-3β and ERK), the amount of phospho/total ratio normalized to control was measured.
The results indicating the caspase-3 cleavage (as an indicator of caspase-3 activation) are displayed in Fig. 3. The hippocampal cells treated with Aβ25−35 and/or CEPO-Fc in the presence or absence of inhibitors were analyzed for cleaved caspase-3 protein. The band of cleaved caspase-3 was observed at 17 kDa. As an internal control, β-actin antibody detected a band at 45 kDa. One-way ANOVA showed a significant difference between groups (F(8,26) = 8.88, P < 0.0001). Analysis by Tukey's test showed that Aβ25−35 significantly elevated the cleavage of caspase-3 (P < 0.05) but treatment with CEPO-Fc at dose 5 IU markedly prevented this enhanced activation (P < 0.05). The results also revealed that following LY294002 and PD98059 administration, CEPO-Fc failed to decrease caspase-3 cleavage induced by Aβ25−35. According to the statistical analysis, a significant increment of cleaved caspase-3 was observed between control and Aβ(20 µM) + CEPO-Fc5IU + LY294002 group (P < 0.001) and also between control and Aβ(20 µM) + CEPO-Fc5IU + PD98059 group (P < 0.05).
The results of the western blotting analysis for the relative amount of cleaved caspase-3 in cultured hippocampal cells exposed to Aβ20µM and/or CEPO-Fc in the presence or absence of PI3K and ERK inhibitors. The cell lysate was followed by immunoblotting using anti-caspase-3 and β-Actin. Representative bands for each antibody are shown. Quantitative values represent mean normalized to control ± SEM. *P < 0.05, **P < 0.01 and ***P < 0.001 represent the difference between control and other treated groups. #P < 0.05 represents the difference between Aβ20µM treated group and the others
Figure 4 displays the relative amount of Akt phosphorylation. Specific antibodies for phosphorylated Akt (P.Akt) and total Akt (T.Akt) detected a band at about 60 kDa. Analysis by one-way ANOVA revealed a significant difference between groups (F(8,26) = 10.61, P < 0.0001). Results of Tukey's post hoc test disclosed that while Aβ25−35 significantly decreased Akt phosphorylation in hippocampal cells (P < 0.001), CEPO-Fc at dose 5 IU could significantly reverse Aβ-induced decrement of Akt phosphorylation (P < 0.01). In addition, the results also indicated that CEPO-Fc in the presence of LY294002 was not further able to reverse Aβ-mediated Akt phosphorylation diminution, where the amount of phospho-Akt was significantly decreased in Aβ(20 µM) + CEPO-Fc5IU + LY294002 group compared to control (P < 0.001). However, PD98059 did not represent a significant effect on the Akt phosphorylation.
Anti-phospho and total GSK-3β antibody revealed a 46 kDa band. A significant difference in the amount of phospho-GSK-3β between groups was observed via One-way ANOVA analysis (F(8,18) = 8.519, P < 0.001). Tukey's test indicated that although Aβ25−35 treatment resulted in a significant reduction of the phospho-GSK-3β in hippocampal cells (P < 0.05), CEPO-Fc at dose 5 IU reversed this Aβ-induced phospho-GSK-3β decline (P < 0.01). However, following adding LY294002 to cells, CEPO-Fc could not prevent this Aβ-induced decline effect. As statistical analysis showed, a significant decrease was observed in Aβ(20 µM) + CEPO-Fc5IU + LY294002 group compared to control (P < 0.01). The post hoc test also revealed that in groups receiving PD98059, GSK-3β phosphorylation was markedly raised compared to control (P < 0.05). These findings are illustrated in Fig. 5.
The results of western blotting analysis evaluating the ERK phosphorylation in hippocampal cells are depicted in Fig. 6. Following incubation with anti-phospho and total ERK antibodies, two bands were appeared at 42 and 44 kDa. One-way ANOVA showed a significant difference between groups (F(8,26) = 7.826, P < 0.0001). Post hoc analysis by Tukey’s test demonstrated a significant increment in phospho-ERK after Aβ25−35 exposure (P < 0.05) and subsequently a significant decrement following CEPO-Fc (5 IU) treatment (P < 0.05). Further reduction of phospho-ERK level was shown following the use of ERK inhibitor at groups receiving PD98059 in comparison to Aβ20µM group (P < 0.001). Furthermore, the results also showed that, in the presence of LY294002, phosphorylated ERK level was significantly lower than the Aβ20µM group (P < 0.05).
The results of the western blotting analysis for P.Akt/T.Akt in cultured hippocampal cells exposed to Aβ20µM and/or CEPO-Fc in the presence or absence of PI3K and ERK inhibitors. The cell lysate was followed by immunoblotting using anti-phospho and total Akt and β-Actin. Representative bands for each antibody are shown. Quantitative values represent mean normalized to control ± SEM. ***P < 0.001 displays the difference between control and other treated groups. ##P < 0.01 displays the difference between Aβ20µM treated group and other treated groups
The results of the western blotting analysis for P.GSK-3β/T.GSK-3β in cultured hippocampal cells exposed to Aβ20µM and/or CEPO-Fc in the presence or absence of PI3K and ERK inhibitors. The cell lysate was followed by immunoblotting using anti-phospho and total GSK-3β and β-Actin. Representative bands for each antibody are shown. Quantitative values represent mean normalized to control ± SEM. *P < 0.05 and **P < 0.01 indicate the difference between control and other treated groups. #P < 0.05 and ##P < 0.01 indicate the difference between Aβ20µM treated group and other treated groups
The results of the western blotting analysis for P.ERK/T.ERK in cultured hippocampal cells exposed to Aβ20µM and/or CEPO-Fc in the presence or absence of PI3K and ERK inhibitors. The cell lysate was followed by immunoblotting using anti-phospho and total ERK and β-Actin. Representative bands for each antibody are shown. Quantitative values represent mean normalized to control ± SEM. *P < 0.05 displays the difference between control and Aβ20µM treated groups. #P < 0.05 and ###P < 0.001 display the difference between Aβ20µM treated group and the others
Discussion
Since in vitro studies provide conditions through which the response of isolated cells to different drugs can be assessed, we conducted this research to study the possible protective properties of CEPO-Fc against cell toxicity induced by Aβ25−35. The results presented that CEPO-Fc reduced the cytotoxic effects of Aβ25−35 on hippocampal cultures. In addition, western blot analysis also revealed that the protective effects of CEPO-Fc against Aβ25−35 toxicity were in line with reversing the biochemical changes at the level of caspase-3, Akt, GSK-3β, and ERK proteins.
Treatment of cells with CEPO-Fc alone did not significantly differ from the control group, indicating that CEPO-Fc could be used as a protective factor without affecting cell characteristics. Further, as we explained earlier, CEPO acts through binding to EPOR-βcR. This has been shown in various studies [19,20,21]. Therefore, considering the protective effects of CEPO-Fc in the present study, it can be concluded that these types of receptors are expressed in hippocampal cells.
Extensive studies have concluded that caspase-3 activity could be considered as an indicator of apoptosis initiation [28]. It has also been reported that caspase-3 has a major executive role in the neural cell death in the brain of AD patients. This fact has been shown by elevated levels of caspase-3 activity in the damaged brain regions of these patients [29]. In the present study, diminished cell viability was associated with an obvious increase in caspase-3 cleavage in the cells receiving Aβ25−35. In this regard, numerous studies have found similar results suggesting that Aβ induces cell death through activating caspase-3 and apoptotic pathway [30,31,32]. In our study, CEPO-Fc treatment at 5 IU dose enhanced cell viability and reduced caspase-3 cleavage. The morphological changes of cells in the presence of different treatments also confirmed this result. These results demonstrated that CEPO-Fc is potent enough to protect cells against apoptosis. In accordance with our results, the protective effects of rhEPO or its derivatives in preventing cell death have been reported in other in-vitro studies. For example, distinct studies have indicated that rhEPO protects PC12 cells and cultured neurons against Aβ25−35 [14, 33, 34], glutamate, and ischemia toxicity [11, 35]. It has also been demonstrated that rhEPO protects myocardial cells from apoptosis following intra-amygdala injection of Kainic acid and induction of an epileptic model in rats [36].
Our results also revealed that in addition to increment of cell death and activated caspase-3, Aβ25−35 treatment lowered Akt activity and enhanced GSK-3β activity (via reducing its inhibitory phosphorylation) in the isolated hippocampal cells. The concomitant treatment of Aβ25−35 and LY294002 resulted in a further reduction in cell viability. In agreement with our results, there are ample evidences suggesting that disturbed Akt/GSK-3β pathway have a major role in the Aβ-induced cell toxicity. For instance, reports have revealed that there is an association between impaired Akt activity and AD. They demonstrated that the activated PI3K/Akt signaling protects cells against neurotoxicity mediated by Aβ in an in-vitro model [25, 37, 38]. On the other hand, previous studies have revealed that Akt activation inhibits apoptosis (through modulation of transcription factors involved in apoptosis or inhibition of pro-apoptotic factors) [39]. Once activated, Akt phosphorylates Ser-9 residue in GSK-3β molecule and subsequently inhibits its activity. It has been reported that GSK-3β is contributed in the major phases of apoptotic pathways [40]. According to the "GSK-3β hypothesis in AD", GSK-3β is involved as a strategic mediator in induction of neurodegeneration in AD pathology [41, 42]. Our results also demonstrated that, in addition to decreasing caspase-3 cleavage, administration of CEPO-Fc to cultured cells significantly prevented the reduction of Akt and GSK-3β phosphorylation (thereby increasing Akt activity and decreasing GSK-3β activity) mediated by Aβ25−35. We found that in the presence of the LY294002 inhibitor, CEPO-Fc was no longer able to prevent activation of caspase-3 and cell death caused by Aβ25−35. These findings highlight the importance of Akt/GSK-3β pathway in the protective effects of CEPO-Fc. In line with the current findings, some documents have demonstrated that EPO or CEPO activates the PI3K/Akt signaling pathway. For example, increased activity of Akt/GSK-3β pathway and protective properties have been demonstrated following treatment with EPO [43] and CEPO [44] in a hypoxia/ischemia model. Furthermore, in an induced model of Parkinson's in cultured cells, rhEPO treatment activated PI3K/Akt signaling and exerted neuroprotective effects [45]. These findings, based on previous and current studies, consequently, can conclude that CEPO-Fc exerts protective effects through activating the Akt/GSK-3β pathway. Our recently published in-vivo study also supports these results [8].
Our findings also indicated that whereas Aβ25−35 caused phosphorylation of ERK to be increased, the protective effect of CEPO-Fc treatment was associated with reduction of ERK phosphorylation. Contrary to our findings, some studies have claimed that Aβ inhibits ERK activity and normal activity of ERK protects against Aβ toxicity [46, 47]. In addition, ERK activity enhancement has been reported to prevent scopolamine-induced learning deterioration [48]. However, the increment of activated ERK levels surrounding amyloid deposit in AD patients’ brain [49], as well as in Aβ-dependent disorders in the animal [6, 50, 51] and cellular [7, 25] models confirm our results suggesting that enhanced ERK activity might participate in Aβ-induced cellular loss. In order to further test of this hypothesis, ERK activation was inhibited by an upstream inhibitor (PD98559). Following use of PD98059, we observed that ERK inhibition was not able to reverse cell loss mediated by Aβ25−35; rather, surprisingly a significant growth in caspase-3 cleavage was observed in the presence of PD98059. The time of ERK activation seems to an important factor in determining its cellular role. In this regard, it has been reported that transient activation of ERK has protective effects while persistent activation leads to cell death [52,53,54]. As, in our study, the cells were exposed to Aβ25−35 for 24 h, this persistent activation of ERK is likely to contribute to Aβ25−35-induced apoptosis.
We also observed that following use of PD98059, cleaved caspase-3 remained significantly at a high level and even inhibition of ERK was not able to prevent cell death induced by Aβ25−35. Accordingly, we suggest that the pathways developing toxicity in hippocampal cells do not take one or two specific pathways; rather, a collection of different stimuli are involved in the ultimate response of the cells. In addition, our results also demonstrated that CEPO-Fc not only reversed the Aβ25−35 mediated elevation of ERK phosphorylation but also significantly lowered its level. This finding implies that under the current experimental conditions, CEPO-Fc was able to prevent persistence activation of ERK. This effect can be one of the possible ways through which CEPO-Fc protected hippocampal cells against Aβ25−35 toxicity. So, if ERK plays an adverse role in the current experimental conditions, expectedly the protective effects of CEPO-Fc will be enhanced through inhibiting ERK activity. However, our results in MTT assay test and also western blotting analysis (which clearly showed an increment in caspase-3 cleavage) did not reveal such an outcome in protective properties of CEPO-Fc. On the other hand, there are some documents in contradiction with our findings. For example, Ma et al. revealed that enhanced transient phosphorylation of ERK1/2 is involved in EPO-mediated protection in Aβ-induced cell toxicity. Nevertheless, after using PD98059, the protective effect of EPO was still persist on cell viability. Therefore, they suggested that ERK1/2 signaling does not have a major role in the protection mediated by EPO against Aβ [34]. In contrary to our results, Zhang et al. indicated that EPO protects primary cortical neuron cultures against sevoflurane-induced toxicity through activating ERK1/2, where inhibiting ERK1/2 phosphorylation eliminated the protective effects of EPO [55]. Further, pretreatment with EPO attenuated ischemia/reperfusion-induced apoptosis by transient ERK activation [56]. These results raise a theory that on the one hand CEPO-Fc is positively involved in transient activation of ERK, while it negatively participates in persistence activation of ERK. In both cases, CEPO-Fc contributes to cell protection. Inhibition of ERK activity followed by PD98059, in the current study, may support CEPO-Fc to prevent persistence ERK activation but it may inhibit its transient activation induced by CEPO-Fc. This is why use of PD98059 does not further improve the protective effects of CEPO-Fc.
One of the important points in this research was the possible interaction between Akt and ERK signaling in primary hippocampal cells toxicity or protection mediated by Aβ25−35 or CEPO-Fc respectively. It has previously been reported that there is an inverse relationship between Akt and ERK activation in cell toxicity caused by Aβ, where inhibition of each one leads to enhanced phosphorylation of the other [25]. Our results revealed that following use of LY294002, CEPO-Fc was still able to prevent ERK phosphorylation induced by Aβ25−35. This reduction in phosphorylation of ERK was also observed in cells receiving Aβ25−35 and LY294002 concurrently. In contrary to our findings, some documents suggest that the extent of ERK phosphorylation is increased following inhibition of PI3K/Akt pathway [25]. Our results also demonstrated that inhibition of ERK signaling by PD98059 prevented Aβ-induced decrement in Akt phosphorylation. Additionally, inhibition of ERK partially potentiated the ability of CEPO-Fc to prevent the reduced Akt phosphorylation mediated by Aβ25−35, implying that ERK activation participates in Akt signaling disturbance. Therefore, it can be inferred that ERK inhibition results in a relative rise in Akt phosphorylation. Congruent with the present findings, some studies have indicated that inhibition of ERK prevents reduced Akt activity and cell death [25, 57]. In general, according to the results, it can be concluded that PI3K/Akt signaling has a more pivotal role than ERK in protective effects of CEPO-Fc.
Conclusion
The findings of the current study indicated, for the first time, that CEPO-Fc protects isolated hippocampal cells against cell death induced by Aβ25−35 toxicity. This protective effect of CEPO-Fc is associated with its modulatory effect on the biochemical changes caused by Aβ25−35 in caspase-3, Akt, GSK-3β and ERK. Since a large body of evidence has reported the protective properties of CEPO in various models of neurodegenerative diseases (reviewed in [58]), the current study may be useful in explaining the molecular mechanisms associated with CEPO-Fc protective properties. These findings suggest the use of CEPO-Fc as a potential candidate in the treatment of neurodegenerative diseases.
Change history
20 May 2020
Unfortunately, the original version of this article contained a mistake in the arrangement of representative cell images in Fig.��2. In this figure, the same representative image for A�� group was mistakenly placed for A�����+���LY group. The corrected form of this figure is provided in this correction.
References
Parihar M, Hemnani T (2004) Alzheimer’s disease pathogenesis and therapeutic interventions. J Clin Neurosci 11(5):456–467
Malchiodi-Albedi F, Domenici MR, Paradisi S, Bernardo A, Ajmone‐Cat MA, Minghetti L (2001) Astrocytes contribute to neuronal impairment in βA toxicity increasing apoptosis in rat hippocampal neurons. Glia 34(1):68–72
Holscher C, Gengler S, Gault VA, Harriott P, Mallot HA (2007) Soluble beta-amyloid [25–35] reversibly impairs hippocampal synaptic plasticity and spatial learning. Eur J Pharmacol 561(1–3):85–90
Behl C, Davis JB, Klier FG, Schubert D (1994) Amyloid β peptide induces necrosis rather than apoptosis. Brain Res 645(1–2):253–264
Ivins KJ, Thornton PL, Rohn TT, Cotman CW (1999) Neuronal apoptosis induced by β-amyloid is mediated by caspase-8. Neurobiol Dis 6(5):440–449
Kirouac L, Rajic AJ, Cribbs DH, Padmanabhan J (2017) Activation of Ras-ERK signaling and GSK-3 by amyloid precursor protein and amyloid beta facilitates neurodegeneration in Alzheimer’s disease. Eneuro.https://doi.org/10.1523/ENEURO.0149-16.2017
Hooshmandi E, Ghasemi R, Iloun P, Moosavi M (2019) The neuroprotective effect of agmatine against amyloid β-induced apoptosis in primary cultured hippocampal cells involving ERK, Akt/GSK-3β, and TNF-α. Mol Biol Rep 46(1):489–496
Hooshmandi E, Motamedi F, Moosavi M, Katinger H, Zakeri Z, Zaringhalam J, Maghsoudi A, Ghasemi R, Maghsoudi N (2018) CEPO-Fc (an EPO derivative) protects hippocampus against Aβ-induced memory deterioration: a behavioral and molecular study in a rat model of Aβ toxicity. Neuroscience 388:405–417
Sasaki R (2003) Pleiotropic functions of erythropoietin. Intern Med 42(2):142–149
Juul SE, Anderson DK, Li Y, Christensen RD (1998) Erythropoietin and erythropoietin receptor in the developing human central nervous system. Pediatr Res 43(1):40–49. https://doi.org/10.1203/00006450-199804001-00243
Morishita E, Masuda S, Nagao M, Yasuda Y, Sasaki R (1997) Erythropoietin receptor is expressed in rat hippocampal and cerebral cortical neurons, and erythropoietin prevents in vitro glutamate-induced neuronal death. Neuroscience 76(1):105–116
Brines M, Cerami A (2005) Emerging biological roles for erythropoietin in the nervous system. Nat Rev Neurosci 6(6):484–494. https://doi.org/10.1038/nrn1687
Castaneda-Arellano R, Beas-Zarate C, Feria-Velasco AI, Bitar-Alatorre EW, Rivera-Cervantes MC (2014) From neurogenesis to neuroprotection in the epilepsy: signalling by erythropoietin. Front Biosci 19:1445–1455
Li G, Ma R, Huang C, Tang Q, Fu Q, Liu H, Hu B, Xiang J (2008) Protective effect of erythropoietin on beta-amyloid-induced PC12 cell death through antioxidant mechanisms. Neurosci Lett 442(2):143–147. https://doi.org/10.1016/j.neulet.2008.07.007
Kirkeby A, Torup L, Bochsen L, Kjalke M, Abel K, Theilgaard-Monch K, Johansson PI, Bjorn SE, Gerwien J, Leist M (2008) High-dose erythropoietin alters platelet reactivity and bleeding time in rodents in contrast to the neuroprotective variant carbamyl-erythropoietin (CEPO). Thromb Haemost 99(4):720–728. https://doi.org/10.1160/th07-03-0208
Piloto N, Teixeira HM, Teixeira-Lemos E, Parada B, Garrido P, Sereno J, Pinto R, Carvalho L, Costa E, Belo L, Santos-Silva A, Teixeira F, Reis F (2009) Erythropoietin promotes deleterious cardiovascular effects and mortality risk in a rat model of chronic sports doping. Cardiovasc Toxicol 9(4):201–210. https://doi.org/10.1007/s12012-009-9054-2
Montero M, Poulsen FR, Noraberg J, Kirkeby A, van Beek J, Leist M, Zimmer J (2007) Comparison of neuroprotective effects of erythropoietin (EPO) and carbamylerythropoietin (CEPO) against ischemia-like oxygen-glucose deprivation (OGD) and NMDA excitotoxicity in mouse hippocampal slice cultures. Exp Neurol 204(1):106–117. https://doi.org/10.1016/j.expneurol.2006.09.026
Coleman TR, Westenfelder C, Togel FE, Yang Y, Hu Z, Swenson L, Leuvenink HG, Ploeg RJ, d’Uscio LV, Katusic ZS, Ghezzi P, Zanetti A, Kaushansky K, Fox NE, Cerami A, Brines M (2006) Cytoprotective doses of erythropoietin or carbamylated erythropoietin have markedly different procoagulant and vasoactive activities. Proc Natl Acad Sci USA 103(15):5965–5970. https://doi.org/10.1073/pnas.0601377103
Chamorro ME, Wenker SD, Vota DM, Vittori DC, Nesse AB (2013) Signaling pathways of cell proliferation are involved in the differential effect of erythropoietin and its carbamylated derivative. Biochim Biophys Acta 1833(8):1960–1968. https://doi.org/10.1016/j.bbamcr.2013.04.006
Maltaneri RE, Chamorro ME, Schiappacasse A, Nesse AB, Vittori DC (2017) Differential effect of erythropoietin and carbamylated erythropoietin on endothelial cell migration. Int J Biochem Cell Biol 85:25–34. https://doi.org/10.1016/j.biocel.2017.01.013
Leist M, Ghezzi P, Grasso G, Bianchi R, Villa P, Fratelli M, Savino C, Bianchi M, Nielsen J, Gerwien J (2004) Derivatives of erythropoietin that are tissue protective but not erythropoietic. Science 305(5681):239–242
Ding J, Li QY, Yu JZ, Wang X, Lu CZ, Ma CG, Xiao BG (2015) The lack of CD131 and the inhibition of Neuro-2a growth by carbamylated erythropoietin. Cell Biol Toxicol 31(1):29–38. https://doi.org/10.1007/s10565-015-9292-y
Sturm B, Helminger M, Steinkellner H, Heidari MM, Goldenberg H, Scheiber-Mojdehkar B (2010) Carbamylated erythropoietin increases frataxin independent from the erythropoietin receptor. Eur J Clin Invest 40(6):561–565. https://doi.org/10.1111/j.1365-2362.2010.02292.x
Armand-Ugon M, Aso E, Moreno J, Riera-Codina M, Sanchez A, Vegas E, Ferrer I (2015) Memory improvement in the AβPP/PS1 mouse model of familial Alzheimer’s disease induced by carbamylated-erythropoietin is accompanied by modulation of synaptic genes. J Alzheimer’s Dis 45(2):407–421. https://doi.org/10.3233/jad-150002
Ghasemi R, Moosavi M, Zarifkar A, Rastegar K, Maghsoudi N (2015) The interplay of Akt and ERK in Aβ toxicity and insulin-mediated protection in primary hippocampal cell culture. J Mol Neurosci 57(3):325–334. https://doi.org/10.1007/s12031-015-0622-6
Maurice T, Mustafa MH, Desrumaux C, Keller E, Naert G, de la Rodriguez Cruz CG-BM, Garcia Rodriguez Y (2013) Intranasal formulation of erythropoietin (EPO) showed potent protective activity against amyloid toxicity in the Aβ(2)(5)(-)(3)(5) non-transgenic mouse model of Alzheimer’s disease. J Psychopharmacol 27(11):1044–1057. https://doi.org/10.1177/0269881113494939
Schriebl K, Trummer E, Lattenmayer C, Weik R, Kunert R, Muller D, Katinger H, Vorauer-Uhl K (2006) Biochemical characterization of rhEpo-Fc fusion protein expressed in CHO cells. Protein Exp Purif 49(2):265–275. https://doi.org/10.1016/j.pep.2006.05.018
Gurtu V, Kain SR, Zhang G (1997) Fluorometric and colorimetric detection of caspase activity associated with apoptosis. Anal Biochem 251(1):98–102. https://doi.org/10.1006/abio.1997.2220
Su JH, Zhao M, Anderson AJ, Srinivasan A, Cotman CW (2001) Activated caspase-3 expression in Alzheimer’s and aged control brain: correlation with Alzheimer pathology. Brain Res 898(2):350–357
Marin N, Romero B, Bosch-Morell F, Llansola M, Felipo V, Roma J, Romero FJ (2000) Beta-amyloid-induced activation of caspase-3 in primary cultures of rat neurons. Mech Ageing Dev 119(1–2):63–67
Allen JW, Eldadah BA, Huang X, Knoblach SM, Faden AI (2001) Multiple caspases are involved in beta-amyloid-induced neuronal apoptosis. J Neurosci Res 65(1):45–53. https://doi.org/10.1002/jnr.1126
Xian YF, Mao QQ, Wu JC, Su ZR, Chen JN, Lai XP, Ip SP, Lin ZX (2014) Isorhynchophylline treatment improves the amyloid-beta-induced cognitive impairment in rats via inhibition of neuronal apoptosis and tau protein hyperphosphorylation. J Alzheimer’s Dis 39(2):331–346. https://doi.org/10.3233/jad-131457
Zhi-Kun S, Hong-Qi Y, Zhi-Quan W, Jing P, Zhen H, Sheng-Di C (2012) Erythropoietin prevents PC12 cells from beta-amyloid-induced apoptosis via PI3KAkt pathway. Transl Neurodegener 1(1):7. https://doi.org/10.1186/2047-9158-1-7
Ma R, Hu J, Huang C, Wang M, Xiang J, Li G (2014) JAK2/STAT5/Bcl-xL signalling is essential for erythropoietin-mediated protection against apoptosis induced in PC12 cells by the amyloid beta-peptide Aβ25-35. Br J Pharmacol 171(13):3234–3245. https://doi.org/10.1111/bph.12672
Sakanaka M, Wen TC, Matsuda S, Masuda S, Morishita E, Nagao M, Sasaki R (1998) In vivo evidence that erythropoietin protects neurons from ischemic damage. Proc Natl Acad Sci USA 95(8):4635–4640
Ma BX, Li J, Li H, Wu SS (2015) Recombinant human erythropoietin protects myocardial cells from apoptosis via the janus-activated kinase 2/signal transducer and activator of transcription 5 pathway in rats with epilepsy. Curr Ther Res 77:90–98. https://doi.org/10.1016/j.curtheres.2015.07.001
Martin D, Salinas M, Lopez-Valdaliso R, Serrano E, Recuero M, Cuadrado A (2001) Effect of the Alzheimer amyloid fragment Aβ(25–35) on Akt/PKB kinase and survival of PC12 cells. J Neurochem 78(5):1000–1008
Kong J, Ren G, Jia N, Wang Y, Zhang H, Zhang W, Chen B, Cao Y (2013) Effects of nicorandil in neuroprotective activation of PI3K/AKT pathways in a cellular model of Alzheimer’s disease. Eur Neurol 70(3–4):233–241. https://doi.org/10.1159/000351247
Yamaguchi H, Wang HG (2001) The protein kinase PKB/Akt regulates cell survival and apoptosis by inhibiting bax conformational change. Oncogene 20(53):7779–7786. https://doi.org/10.1038/sj.onc.1204984
Beurel E, Jope RS (2006) The paradoxical pro- and anti-apoptotic actions of GSK3 in the intrinsic and extrinsic apoptosis signaling pathways. Prog Neurobiol 79(4):173–189. https://doi.org/10.1016/j.pneurobio.2006.07.006
Balaraman Y, Limaye AR, Levey AI, Srinivasan S (2006) Glycogen synthase kinase 3β and Alzheimer’s disease: pathophysiological and therapeutic significance. Cell Mol Life Sci 63(11):1226–1235. https://doi.org/10.1007/s00018-005-5597-y
Peineau S, Taghibiglou C, Bradley C, Wong TP, Liu L, Lu J, Lo E, Wu D, Saule E, Bouschet T, Matthews P, Isaac JT, Bortolotto ZA, Wang YT, Collingridge GL (2007) LTP inhibits LTD in the hippocampus via regulation of GSK3β. Neuron 53(5):703–717. https://doi.org/10.1016/j.neuron.2007.01.029
van der Kooij MA, Groenendaal F, Kavelaars A, Heijnen CJ, van Bel F (2008) Neuroprotective properties and mechanisms of erythropoietin in in vitro and in vivo experimental models for hypoxia/ischemia. Brain Res Rev 59(1):22–33. https://doi.org/10.1016/j.brainresrev.2008.04.007
Ding J, Wang J, Li QY, Yu JZ, Ma CG, Wang X, Lu CZ, Xiao BG (2017) Neuroprotection and CD131/GDNF/AKT pathway of carbamylated erythropoietin in hypoxic neurons. Mol Neurobiol 54(7):5051–5060. https://doi.org/10.1007/s12035-016-0022-0
Jia Y, Mo SJ, Feng QQ, Zhan ML, OuYang LS, Chen JC, Ma YX, Wu JJ, Lei WL (2014) EPO-dependent activation of PI3K/Akt/FoxO3a signalling mediates neuroprotection in in vitro and in vivo models of Parkinson’s disease. J Mol Neurosci 53(1):117–124. https://doi.org/10.1007/s12031-013-0208-0
Watson K, Fan GH (2005) Macrophage inflammatory protein 2 inhibits beta-amyloid peptide (1–42)-mediated hippocampal neuronal apoptosis through activation of mitogen-activated protein kinase and phosphatidylinositol 3-kinase signaling pathways. Mol Pharmacol 67(3):757–765. https://doi.org/10.1124/mol.104.004812
Townsend M, Mehta T, Selkoe DJ (2007) Soluble Aβ inhibits specific signal transduction cascades common to the insulin receptor pathway. J Biol Chem 282(46):33305–33312. https://doi.org/10.1074/jbc.M610390200
Moosavi M, Khales GY, Abbasi L, Zarifkar A, Rastegar K (2012) Agmatine protects against scopolamine-induced water maze performance impairment and hippocampal ERK and Akt inactivation. Neuropharmacology 62(5–6):2018–2023. https://doi.org/10.1016/j.neuropharm.2011.12.031
Medina MG, Ledesma MD, Dominguez JE, Medina M, Zafra D, Alameda F, Dotti CG, Navarro P (2005) Tissue plasminogen activator mediates amyloid-induced neurotoxicity via Erk1/2 activation. EMBO J 24(9):1706–1716. https://doi.org/10.1038/sj.emboj.7600650
Ghasemi R, Zarifkar A, Rastegar K, maghsoudi N, Moosavi M (2014) Insulin protects against Aβ-induced spatial memory impairment, hippocampal apoptosis and MAPKs signaling disruption. Neuropharmacology 85:113–120. https://doi.org/10.1016/j.neuropharm.2014.01.036
Ghasemi R, Zarifkar A, Rastegar K, Maghsoudi N, Moosavi M (2014) Repeated intra-hippocampal injection of beta-amyloid 25–35 induces a reproducible impairment of learning and memory: considering caspase-3 and MAPKs activity. Eur J Pharmacol 726:33–40. https://doi.org/10.1016/j.ejphar.2013.11.034
Stanciu M, Wang Y, Kentor R, Burke N, Watkins S, Kress G, Reynolds I, Klann E, Angiolieri MR, Johnson JW, DeFranco DB (2000) Persistent activation of ERK contributes to glutamate-induced oxidative toxicity in a neuronal cell line and primary cortical neuron cultures. J Biol Chem 275(16):12200–12206
Marshall CJ (1995) Specificity of receptor tyrosine kinase signaling: transient versus sustained extracellular signal-regulated kinase activation. Cell 80(2):179–185
Subramaniam S, Unsicker K (2010) ERK and cell death: ERK1/2 in neuronal death. FEBS J 277(1):22–29. https://doi.org/10.1111/j.1742-4658.2009.07367.x
Li R, Zhang LM, Sun WB (2017) Erythropoietin rescues primary rat cortical neurons from pyroptosis and apoptosis via Erk1/2-Nrf2/Bach1 signal pathway. Brain Res Bull 130:236–244
Zou YR, Zhang J, Wang J, Peng L, Li GS, Wang L (2016) Erythropoietin receptor activation protects the kidney from ischemia/reperfusion-induced apoptosis by activating ERK/p53 signal pathway. Transpl Proc 48(1):217–221. https://doi.org/10.1016/j.transproceed.2016.01.009
Sinha D, Bannergee S, Schwartz JH, Lieberthal W, Levine JS (2004) Inhibition of ligand-independent ERK1/2 activity in kidney proximal tubular cells deprived of soluble survival factors up-regulates Akt and prevents apoptosis. J Biol Chem 279(12):10962–10972. https://doi.org/10.1074/jbc.M312048200
Chen J, Yang Z, Zhang X (2015) Carbamylated erythropoietin: a prospective drug candidate for neuroprotection. Biochem Insights 8(Suppl 1):25–29. https://doi.org/10.4137/bci.s30753
Acknowledgements
This article is a part of the thesis written by Mrs "Etrat Hooshmandi" in School of Medicine (Registration No: 558) and supported by a Grant (No. 22534) from Deputy of research and technology and Neuroscience Research Center, Shahid Beheshti University of Medical Sciences, Tehran, Iran.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interests related to this manuscript.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Hooshmandi, E., Moosavi, M., Katinger, H. et al. CEPO (carbamylated erythropoietin)-Fc protects hippocampal cells in culture against beta amyloid-induced apoptosis: considering Akt/GSK-3β and ERK signaling pathways. Mol Biol Rep 47, 2097–2108 (2020). https://doi.org/10.1007/s11033-020-05309-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-020-05309-6