Abstract
Alzheimer’s disease (AD), a chronic, progressive, neurodegenerative disorder, is the most common type of dementia. Beta amyloid (Aβ) peptide aggregation and phosphorylated tau protein accumulation are considered as one of the causes for AD. Our previous studies have demonstrated the neuroprotective effect of the Rho kinase inhibitor fasudil, but the mechanism remains elucidated. In the present study, we examined the effects of fasudil on Aβ1–42 aggregation and apoptosis and identified the intracellular signaling pathways involved in these actions in primary cultures of mouse hippocampal neurons. The results showed that fasudil increased neurite outgrowth (52.84%), decreased Aβ burden (46.65%), Tau phosphorylation (96.84%), and ROCK-II expression. In addition, fasudil reversed Aβ1–42-induced decreased expression of Bcl-2 and increases in caspase-3, cleaved-PARP, phospho-JNK(Thr183/Tyr185), and phospho-ASK1(Ser966). Further, fasudil decreased mitochondrial membrane potential and intracellular calcium overload in the neurons treated with Aβ1–42. These results suggest that inhibition of Rho kinase by fasudil reverses Aβ1–42-induced neuronal apoptosis via the ASK1/JNK signal pathway, calcium ions, and mitochondrial membrane potential. Fasudil could be a drug of choice for treatment of Alzheimer’s disease.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Alzheimer’s disease (AD), a progressive neurodegenerative disorder, has become a serious health and social problem, in particular with the increasing aged population. More than 47 million people worldwide suffer from AD and this number will increase to 131 million by 2050 (Whitaker et al. 2014). AD is pathologically characterized by β-amyloid (Aβ) peptide plaques and phosphorylated tau protein neurofibrillary tangles (Choi et al. 2018; J. Yu et al. 2018). The formation of senile plaques is the core pathogenic substance in the pathogenesis (Korkmaz et al. 2018). The nucleus of senile plaques is mainly composed of Aβ peptide deposition (a short peptide containing 39–43 amino residues), which is formed from the amyloid precursor protein (APP) (Skaper 2012; Zameer et al. 2018). Abnormal folding of Aβ peptide generates the most neurotoxic Aβ oligomers (Gotz et al. 2004; Sharma et al. 2016; Wang et al. 2016). A large number of in vitro studies have demonstrated that Aβ oligomers activate neuronal apoptosis (Dean et al. 2016; Lin et al. 2017; Morishima et al. 2001; J. Wang et al. 2018). Clinical symptoms of AD are progressive memory loss and cognitive dysfunction due to extensive damage of hippocampal and cortical neurons (Amoroso et al. 2018). The apoptosis of neurons caused by Aβ oligomers is characterized by typical apoptosis, including mitochondrial dysfunction (Van Giau et al. 2018), endoplasmic reticulum dysfunction (Dhivya Bharathi et al. 2018), synaptic damage (Jadhav et al. 2015), swelling of the membrane system, chromatin shrinkage (Zhao et al. 1997), and nuclear disintegration (Han et al. 2017).
There are several apoptosis signaling pathways, among which the caspases-3, 8, 9, 12 families are important regulatory molecules for apoptosis of major types of neurons (Jiang et al. 2018; Wei et al. 2018). The apoptosis signal-regulating kinase 1 (ASK1)/c-Jun N-terminal kinase (JNK) signal pathways, which are the upstream of caspases, are vital signaling of central nervous system apoptosis. They are involved in the whole process of neuron apoptosis caused by Aβ oligomers, leading to the occurrence of Aβ neurotoxicity in the brain of AD (Hashimoto et al. 2003; Kanamoto et al. 2000; Sheng et al. 2001; Song et al. 2014). JNK, also known as stress activated protein kinase (SAPK), exerts apoptotic regulation via the Bcl-2 protein family, including anti-apoptotic proteins (Bcl-2, Bcl-xl, and Bcl-w) and pro-apoptotic proteins (Bad, Bax, and Bak) (Zhou et al. 2015). ASK1 is located in the upstream of the JNK signal pathway, initiating the apoptosis process of endothelial cells (Amin et al. 2017; X. Guo et al. 2017; Kim et al. 2018; Mu et al. 2015;). In the pathogenesis of AD, neuronal apoptosis involves the imbalance of reactive oxygen species (ROS) and intracellular calcium overload, which directly causes the activation of ASK1 and initiates the cell apoptosis pathway (Qi and Shuai 2016). Anti-apoptotic members Bcl-2 and Bcl-xl are mainly located in the mitochondrial outer membrane (Ding et al. 2014), endoplasmic reticulum (Urresti et al. 2016) and nuclear membrane (Losonczi et al. 2000), which play a protective role in the cell substructure. Pro-apoptotic members such as Bax and Bad, which are opposite to anti-apoptotic members, have the effect on cell substructure damage (Obulesu and Lakshmi 2014; Salminen et al. 2013; Sharma et al. 2016; Szymanski et al. 2017).
Rho kinase (ROCK) is one of the main kinases involved in cellular activities. The complete ROCK signal pathway includes the upstream active receptor Rho, the downstream ROCK and its substrate. The conversion of the active Rho GTP binding form to the inactive Rho GDP binding form serves as a molecular switch (Tan et al. 2018). It plays an important role in regulating cell growth, division, contraction, adhesion, migration, and secretion. ROCK plays an important role in repair of central nerve cell apoptosis (Chen et al. 2017a, b, c; Chong et al. 2017; Henderson et al. 2016; Lai and McLaurin 2018). Abnormal synaptic function and synaptic loss are also widely recognized as neuronal apoptosis mechanism in AD (Zolezzi et al. 2018).
Fasudil, a selective RhoA/ROCK inhibitor (Wang et al. 2017), has been demonstrated to enhance memory and improve pathogenesis of Alzheimer’s patients (Koch et al. 2018). Several studies have shown that fasudil has multiple functions in the CNS, including activation of endogenous neural stem cells, promotion of neurotrophic factor release, inhibition of intracellular calcium release, dilation of cerebral vessels, protection of nerve cells, improvement of the nerve function, and promotion of axonal regeneration (Ding et al. 2009; Liu et al. 2015; Zhao et al. 2015). In the present study, we examined the effects of fasudil on Aβ1–42-induced changes in expression of the proteins related to apoptosis and identified the intracellular signal pathway involved in apoptosis in primary cultures of mouse hippocampal neurons. It was demonstrated that fasudil promoted neuroprotection against apoptosis via ASK1/JNK signaling.
Materials and methods
All the experiments were performed in compliance with the guidelines and regulations of Administration Office of the International Council for Laboratory Animal Science. The experimental protocols were approved by the Animal Ethics Committee of Shanxi Datong University, Datong, China. Animals had ad libitum access to food and water.
Animals and primary neuronal cell cultures
C57BL/6 mice were purchased from Beijing HFK Bioscience CO., LTD, China. All animals were housed in pathogen-free animal housing facilities at the Institute of Brain Science, Shanxi Datong University and maintained at a 12/12-h light/dark cycle (25 ± 2 °C, humidity 50 ± 5%). Adult male and female mice were housed for mating and vaginal plug was checked for female mice every day. Observation of plug was considered day 1 of the pregnancy, 18 days after which, the female mice were sacrificed by cervical dislocation. Hippocampal tissues of 18-day embryonic mice (E18) were isolated and digested for 10 min with 0.125% trypsin. A 200-mesh sieve was used to filter out connective tissues, centrifuged at 1500 rpm for 5 min. The primary neurons were cultured in neurobasal-A-medium supplemented with 2% B27 (Gibco), 100 U/ml penicillin, 100 μg/ml streptomycin, and 1% L-glutamine (Gibco) at 37 °C, 5% CO2. All the process of cell separation was performed quickly on ice. Cells were identified according to cell morphology and immunofluorescence staining.
Amyloid β protein fragment 1–42 (Aβ1–42) and fasudil treatment
Aβ1–42 (Sigma USA; A9810) was dissolved in PBS to prepare 100 μM as stock solution, stored in −20 °C. The stock was diluted to 2 μM as the working solution. Fasudil hydrochloride was purchased from Tianjin Chase Sun Pharmaceutical Co., Ltd. (China) and prepared as 15 μg/ml solution (Chen et al. 2014; Guo et al. 2014). After 7-day cultures, mouse primary neurons were treated with Aβ and fasudil for 48 h before harvesting cells for further processing.
Immunofluorescence staining with confocal microscopy
Cells were grown on coverslips (10 mm) in 24-well plates. After Aβ-induced apoptosis, the cells were washed 3 times with PBS and fixed with 4% cold paraformaldehyde for 30 min at room temperature. The washed cells were permeabilized with 0.1% Triton X-100 for 15 min, rinsed again with PBS and blocked with 1% bovine serum albumin (BSA) for 30 min at room temperature. Cells were then incubated with anti-MAP2 (HM-2) (diluted at 1:500, Abcam, USA), anti-phospho-Tau (Ser396) (diluted at 1:200, Cell Signaling, USA), phospho-JNK (Thr183/Tyr185) (diluted at 1:250, Cell Signaling, USA) with mouse monoclonal primary antibodies, anti-Aβ1–42 (diluted at 1:500, Millipore, USA), anti-ROCK II (diluted at 1:500, Abcam, USA), anti-Bcl-2 (diluted at 1:200, Abcam, USA), anti-caspase-3 (diluted at 1:200, Abcam, USA), anti-phospho-ASK-1 (Ser966) (diluted at 1:300, Immuno Way, USA) rabbit monoclonal primary antibodies at 4 °C overnight. Cells were washed 3 times with PBS, incubated with FITC or Cy3-conjugated secondary antibodies (Invitrogen, USA) for 1 h at RT, and then thoroughly washed with PBS for 3 times. The modified coverslips were mounted onto glass slides and observed with confocal microscopy (Olympus FV1000, Japan).
Western blotting
The primary cultures of neuronal cells were rinsed twice with PBS, lysed in reaction buffer (120 μl RIPA with 1 mM PMSF, KeyGEN, Nanjing, China, and 1 μM PhosSTOP, Roche, USA), and centrifuged at 12,000 rpm for 15 min. Protein concentration was determined using BCA protein assay (KeyGEN). The supernatants were mixed with 5 × SDS (sodium dodecyl sulfate) sample buffer with 50 mM dithiothreitol. Aliquots of 5 μg of protein extracts were separated on 15% SDS-polyacrylamide gels and transferred to nitrocellulose membranes. Membranes were block with 5% milk in Tris-buffered saline (TBS), then probed with rabbit anti-phospho-JNK (Thr183/Tyr185) (diluted at 1:1000, Cell Signaling, USA), anti-caspase-3 (diluted at 1:1000, Abcam, USA), anti-PARP (diluted at 1:1000, Abcam, USA), anti-β-actin (diluted at 1:7500, Abcam, USA). The secondary antibodies against primary antibodies were anti-rabbit primary antibodies, i.e. the goat anti-rabbit IgG-HRP (1:5000 dilution, Cell Signaling, USA) secondary antibodies, were used before detection of chemiluminescence. Western blotting results were analyzed using Image-Lab analysis software. All results were normalized by β-actin (Yu et al. 2017).
TUNEL analysis of cell apoptosis
TUNEL (terminal deoxynucleotidyl transferase dUTP nick end labeling) assay was performed using One Step TUNEL Apoptosis Kit (Beyotime, China). Neuronal cells were put onto poly-L-lysine-coated glass slides, fixed, permeabilized, and incubated with TUNEL reaction mixture at 37 °C for 1 h as described in the manufacturer’s protocol. The cells were analyzed by fluorescence microscopy (Shen et al. 2011).
JC-1 analysis of mitochondrial membrane potential
Mitochondrial membrane potential detection kits (JC-1, Beyotime, China) were used to analyze mitochondrial membrane potential of neurons. Cells were rinsed with PBS before staining at 37 °C for 20 min using 1 ml JC-1 working solution. During the incubation period, 4 ml distilled water was added to each 1 ml JC-1 dyeing buffer (5x) to prepare an appropriate amount of JC-1 dyeing buffer (1x) before placing in an ice bath. After the incubation, supernatant was removed and washed twice with JC-1 dye buffer (1x). Laser confocal microscope was used for observation (Gao et al. 2011).
Fluo-3 analysis calcium imaging
Fluo-3 AM is one of the most commonly used fluorescent probes to detect the concentration of calcium ions in cells. After entering the cell, Fluo-3 AM is cleaved by the esterase to form Fluo-3, which binds to calcium ions and produces strong fluorescence. Cells were incubated with 5 μM Fluo-3 AM at 37 °C in the dark for 1 h. After washing, cells were re-incubated for 20–30 min to ensure a complete conversion of Fluo-3 AM to Fluo-3. Laser confocal microscopy was used for observation.
Statistical analysis
The SPSS software (International Business Machines Corporation, IBM, USA) was used for statistical analysis. All data were expressed as means ± SEM. Differences among multiple groups were analyzed by one-way analysis of variance (ANOVA) while differences between two groups were analyzed using Dunnett tests. A value of p < 0.05 was considered statistically significant.
Results
Promoting effect of fasudil on neurite outgrowth in Aβ1–42-treated primary neurons
Primary cultures of hippocampal neurons from E18 mice were established for morphometric analysis. The MAP2 marker was used to characterize neuronal cell types. Immunofluorescence staining with confocal microscopy revealed that MAP2 was expressed exclusively in hippocampal neurons. There were no significant changes between groups (Fig. 1A). Further, we explored the morphological analysis of neurons by measuring the neurites length (μm). Vehicle-treated cells (control) showed normal neurite outgrowth. However, there was tendency of decreased neurite growth by Aβ1–42 alone (13.39%), which was not statistically significant. While combination with fasudil robustly increased neurite outgrowth compared to Aβ1–42 alone (52.84%) (Fig. 1B), suggesting the neuro-promoting effect.
MAP2 expression (A) and neurite length (B) in primary cultures of hippocampal neurons. In panel A, (a) DAPI-labelled nuclei of neurons treated with vehicle, fasudil, Aβ1–42 (Aβ), or fasudil + Aβ, (b) neuronal marker MAP2 expression in the different treatment conditions, (c) merging of rows a and b. In panel B, (a) MAP2 in the different treatment conditions, (b) Neurite length measured with Imaris surface tool, (c) merging of both a and b. Neurons cultured for 7 days were treated with vehicle or fasudil (15 μg/ml) 2 h before incubation with Aβ (2 μM) for 48 h, followed by immunofluorescent staining with MAP2 and DAPI antibodies. Data shown are means ± S.E.M; n = 3; ** p < 0.01 versus control; ###p < 0.001 versus Aβ alone. One-way ANOVA followed by post hoc Dunnett tests was used for statistical analysis
Fasudil attenuated Aβ burden and tau phosphorylation
Our results showed that control and fasudil alone did not cause any Aβ1–42 plaques. In contrast, Aβ1–42-treated neurons displayed significant increases in Aβ plaques (145.19%), which was reversed by fasudil (15 μg/ml) (46.65%), suggesting that fasudil reduced Aβ burden (Fig. 2A).
Expression of Aβ (A) and p-Tau (B) in primary cultures of hippocampal neurons. In panel A, (a) DAPI-labelled nuclei of neurons treated with vehicle, fasudil, Aβ, or fasudil + Aβ, (b) Aβ expression in the different treatment conditions, (c) merging. In panel B, (a) DAPI-labelled nuclei of neurons in the different treatment conditions, (b) p-Tau (Ser396), (c) merging. Experimental procedures were the same as Fig. 1, followed by immunofluorescent staining with DAPI, Aβ and p-Tau (Ser396) antibodies. Data shown are means ± S.E.M; n = 3; *** p < 0.001 versus control; ###p < 0.001 versus Aβ alone. One-way ANOVA followed by post hoc Dunnett tests was used for statistical analysis
Tau phosphorylation is the key factor that plays an important role in pathogenicity of AD (Ando et al. 2016). In the present study, expression of phospho-Tau (Ser396) in Aβ1–42-treated neurons was significantly increased as compared with control (3274.08%); this was reversed by fasudil (96.84%), as demonstrated by immunofluorescent intensity quantification, suggesting a protective effect of fasudil on NFT formation in AD (Fig. 2B).
Fasudil protected the neurite by inhibiting Aβ-induced apoptosis and increasing expression of Bcl-2
The mechanism by which neurite growth was increased by fasudil in Aβ1–42-treated cells was dissected with the apoptotic pathways investigated. Neurite length loss due to apoptosis induced by Aβ1–42 was confirmed by TUNEL assay. The number of TUNEL-positive cells was significantly increased in Aβ1–42-treated neurons as compared to control (57.59%), which was reversed by fasudil (49.15%), suggesting the anti-apoptotic effect (Fig. 3A). Anti-apoptotic factors such as Bcl-2 are mainly located in mitochondrial outer membranes, endoplasmic reticulum and nuclear membranes. Immunofluorescence staining was used to determine the role of Bcl-2 in the anti-apoptotic effect of fasudil. Bcl-2 expression was significantly decreased (34.38%) in the Aβ1–42-treated neurons and restored to the normal levels by fasudil (Fig. 3B), suggesting anti-apoptotic and neuroprotective properties of fasudil.
TUNEL-positive cells (A) and Bcl-2 (B) expression in primary cultures of hippocampal neurons. In panel A, (a) DAPI-labelled nuclei of neurons treated with vehicle, fasudil, Aβ, or fasudil + Aβ, (b) TUNEL-positive cells (Cyanine 3, Cy3) in the different treatment conditions. Red fluorescent probe (Cy3) labelled dUTP for breakage DNA fragments after apoptosis; (c) merging. In panel B, (a) DAPI-labelled nuclei of neurons in the different treatment conditions, (b) Bcl-2 on the different conditions, (c) merging. Experimental procedures were the same as Fig. 1, followed by immunofluorescent staining with DAPI and Bcl-2 antibodies, and One Step TUNEL Apoptosis Assay. Data shown are means ± S.E.M, * p < 0.05, ** p < 0.01 versus control; ## p < 0.01, ### p < 0.001 versus Aβ alone. One-way ANOVA followed by post hoc Dunnett tests was used for statistical analysis
Fasudil protected the neurite by inhibiting Aβ-induced apoptosis and decreasing the expression of caspase-3 and cleaved-PARP
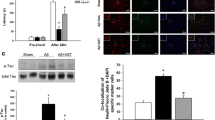
Aβ1–42-induced apoptosis through the caspase pathway was evaluated by determining the expression of caspase-3. Consistent with the TUNEL-positive cell data, fasudil alone did not alter caspase-3 positive cells, which, in contrast, was significantly increased by Aβ1–42 (322.13%). This was reversed by fasudil (70.14%), suggesting the anti-apoptotic effect of fasudil via decreasing caspase-3 expression (Fig. 4A). Similarly, cleaved-PARP is another important indicator of apoptosis. Neuronal DNA damage is repaired by enzymatic activity of the poly(ADP-ribose) polymerase-1 (PARP-1) enzyme (Martire et al. 2016). Hence, in the current study, PARP-1 (Fig. 4B) expression levels were examined with western blotting. Aβ1–42 significantly increased the expression of cleaved PARP (257.66%) (Fig. 4B). This was reversed by fasudil (57.36%).
Expression of caspase-3 (A) and cleaved PARP (B) in primary cultures of hippocampal neurons. In panel A, (a) DAPI-labelled nuclei of neurons treated with vehicle, fasudil, Aβ, or fasudil + Aβ, (b) caspase-3 expression in the different treatment conditions, (c) merging. In panel B, the top panel is the represent immunoblots of cleaved PARP detected by Western blotting; the bottom panel is the corresponding quantification. Experimental procedures were the same as fig. 1, followed by immunofluorescent staining with DAPI and caspase-3 antibodies, and immunoblot analysis with cleaved PARP antibodies. Data shown are means ± S.E.M, * p < 0.05, ** p < 0.01, *** p < 0.001 versus control; ###p < 0.001 versus Aβ alone. One-way ANOVA followed by post hoc Dunnett tests was used for statistical analysis
Fasudil ameliorated mitochondrial stress and decreased intracellular calcium overload in Aβ1–42 exposed neurons
The above data showed Aβ1–42-induced programmed cell death (TUNEL, caspase 3, PARP and Bcl-2), which may be due to mitochondrial stress. Thus, changes in mitochondrial function of primary neurons due to apoptosis induced by Aβ1–42 was investigated. Mitochondrial membrane potential was measured with JC-1 staining. When mitochondrial membrane potential is high, JC-1 accumulates in the matrix of mitochondria by forming J-aggregates with red fluorescence for healthy cells. However, when mitochondrial membrane potential is low, JC-1 becomes monomer with green fluorescence for apoptotic cells. Our data showed decreased ratio of red to green signal in Aβ1–42-treated neurons relative to the control (57.18%); this was attenuated by fasudil to a level that was not significant relative to control (Fig. 5A). Therefore, Aβ1–42 caused apoptosis possibly through decreased mitochondrial membrane potential and fasudil may protect neuron against apoptosis through stabilization of mitochondrial membrane potential.
Changes in JC-1 (A) and Fluo-3 (B) in primary cultures of hippocampal neurons. In panel A, (a) red fluorescent (JC-1 red) intensity in the different conditions; JC-1 formed J-aggregates with red fluorescence. (b) green fluorescent (JC-1 green) intensity in the different conditions; JC-1 becomes monomer with green fluorescence. In panel B, (a) DAPI-labelled nuclei of neurons in the different treatment conditions, (b) Fluo-3 expression in the different conditions; the fluorescence of Fluo-3 increases with higher calcium ion concentrations. (c) merging. Experimental procedures were the same as Fig. 1, followed by immunofluorescent JC-1 and Fluo-3 staining. Data shown are means ± S.E.M, ** p < 0.01 versus control; ###p < 0.001 versus Aβ alone. One-way ANOVA followed by post hoc Dunnett tests was used for statistical analysis
Our results showed elevation of cytosolic Ca2+, an indicator of mitochondrial stress-induced apoptosis, in Aβ1–42-treated neurons (58.10%). This was reversed by fasudil (50.25%) (Fig. 5B), suggesting that fasudil may produce an anti-apoptotic effect through reduction of cytosolic Ca2+ overload and mitochondrial stress and protect organelle functions in neurons.
Fasudil attenuated ROCK-II expression in neurons
ROCK II is involved in the mediation of CNS diseases (Wen et al. 2014). Using immunofluorescence, we showed that expression of ROCK-II in primary neurons was significantly increased by Aβ1–42 (26.14%) and decreased by fasudil (44.59%), both used alone (Fig. 6), as compared with the control; Aβ1–42-induced increase in ROCK II was reversed by fasudil.
Expression of ROCK-II in primary cultures of hippocampal neurons. Left panel: (a) DAPI-labelled nuclei of neurons treated with vehicle, fasudil, Aβ, or fasudil + Aβ, (b) ROCK-II expression in the different conditions, (c) merging. Right panel: Quantification. Experimental procedures were the same as Fig. 1, followed by immunofluorescent staining with ROCK-II antibodies. Data shown are means ± S.E.M, * p < 0.05, ** p < 0.01, *** p < 0.001 versus control; ###p < 0.001 versus Aβ alone. One-way ANOVA followed by post hoc Dunnett tests was used for statistical analysis
Fasudil produced anti-apoptotic and neuroprotective effects through phospho-JNK(Thr183/Tyr185) and phospho-ASK-1(Ser966) pathways
Aβ1–42-induced mitochondrial dysfunction (i.e. decreased JC-1 red/green ratio and increased fluo-3) are also responsible for JNK activation (Hu et al. 2016). ASK1, a member of the mitogen-activated protein kinase kinase (MAPK) family, is located in the upstream of JNK. We determined the role of phospho-JNK(Thr183/Tyr185) and phospho-ASK-1(Ser966) activation in apoptosis induced by Aβ1–42 and measured the intensity of immunofluorescence staining in primary neurons in the four groups. Our results showed that expression of both phospho-JNK(Thr183/Tyr185) (234.39%) and phospho-ASK-1(Ser966) (36.77%) was significantly increased by Aβ1−relative to the control (Fig. 7A, B), which were reversed by fasudil, suggesting that fasudil produced anti-apoptotic and neuroprotective effects by decreasing phospho-JNK(Thr183/Tyr185) and phospho-ASK-1(Ser966).
Expression of p-JNK (A) and p-ASK1 (B) in primary cultures of hippocampal neurons. In panel A, (a) DAPI-labelled nuclei of neurons treated with vehicle, fasudil, Aβ, or fasudil + Aβ, (b) p-JNK expression in the different conditions, (c) merging. In panel B, (a) DAPI-labelled nuclei of neurons, (b) p-ASK1 expression in the different conditions, (c) merging. Experimental procedures were the same as Fig. 1, followed by immunofluorescent staining with p-JNK (Thr183/Tyr185) and p-ASK1(Ser966) antibodies. Data shown are means ± S.E.M, ** p < 0.01, *** p < 0.001 versus control; ##p < 0.01, ###p < 0.001 versus Aβ alone. One-way ANOVA followed by post hoc Dunnett tests was used for statistical analysis
Discussion
One of the key pathological hallmarks of AD is the deposition of extracellular Aβ peptides in the hippocampus by triggering a series of processes called the amyloid cascade that eventually leads to dementia (Gotz et al. 2004; Kirkitadze and Kowalska 2005). The inhibition of Aβ production is therefore considered as a vital strategy for improvement of early AD (Zhong et al. 2018). Tau phosphorylation at proline-rich region (upstream of the microtubule-binding domains) and C-terminal tail region inhibit the microtubule assembly activity, promote its self-aggregation, and disrupt microtubules, leading to formation of neurofibrillary tangles (NFT) and eventually to neurodegeneration (Eidenmuller et al. 2001). Here, we set up an in vitro model of AD using primary cultures of neuronal cells to examine the neuroprotective effects of fasudil, a selective RhoA/ROCK inhibitor, and investigate the intracellular mechanisms involved.
Recent studies have demonstrated that inhibition of ROCK increases axonal regeneration and decreases Aβ formation, suggesting that ROCK can be a potential target for treatment of AD (Liu et al. 2013). Consistent with this, our previous studies have demonstrated fasudil reduces Aβ deposition and ROCK-II expression in the brain of APP/PS1 Tg mice (Yu et al. 2017). In addition, we also have shown that fasudil in combination with BMSCs decreases the severity of AD-related symptoms through peripheral immune system responses (Yu et al. 2018). In the present study, fasudil reversed the tendency of Aβ1–42 to reduce neurite outgrowth primary neurons, suggesting the promoting effect of fasudil. Fasudil treatment clears plaques by attenuating Tau phosphorylation and ROCK II expression in Aβ1–42-induced in vitro model of AD.
Fasudil also prevented Aβ1–42-induced increases in mitochondrial apoptosis-related proteins, including JNK and caspase-3, suggesting that apoptosis of neuronal cells induced by Aβ1–42 is dependent on activation of these proteins as well as ROCK II. Therefore, inhibition of ROCK II and JNK-dependent apoptosis gene expression may be an important approach to the blockade of neuron apoptosis in AD.
Caspases are aspartate-specific cysteine proteases, which play a critical role in apoptosis. Among the members of caspases, caspase-3 is the major contributor to apoptosis and has been identified in several models (Choudhary et al. 2015; Olivera Santa-Catalina et al. 2017). In apoptotic processes, activated caspase-3 increases cleaved PARP, which is considered as a marker of apoptosis (Lazebnik et al. 1994). During apoptotic responses, caspases-3 and 7 cleave PARP, leading to inhibition of its activity; up-regulation of PARP occurs in response to apoptotic processes following caspase activation (West et al. 2005).
Calcium is associated with the function of mitochondria (Szymanski et al. 2017), in which apoptosis resulted from intracellular Ca 2+ overload (Chen et al. 2017a, b, c; Tian et al. 2018). Calcium is the second messenger of cells; however, excessive intracellular calcium ion concentrations cause cytotoxicity (Guo et al. 2013). Aβ1–42-induced increases in calcium ion concentrations in neuronal cells was reversed by fasudil, which is also an aggregator of calcium ions. This result suggests that fasudil may protect mitochondria from apoptosis by inhibiting calcium ions.
The Bcl-2 protein family is the main regulator of mitochondrial apoptosis factor release. Fasudil not only reversed Aβ1–42-induced increases in TUNEL-positive cells, but also blocked Aβ1–42-induced decreases in expression of Bcl-2. These results suggest that fasudil produces anti-apoptotic effects via upregulation of Bcl-2.
ASK1 can be activated by a series of inflammatory factors, such as TNF, IL-1, reactive oxygen radicals, and the death receptor Fas, causing damage to the intracellular microtubule structure and gene transcription in the nucleus (Imarisio et al. 2017). In the pathogenesis of AD, neuronal apoptosis involves the imbalance of reactive oxygen species (ROS) and intracellular calcium overload, which directly leads to the activation of ASK1 and the initiation of apoptosis pathways (Guo et al. 2017).
Conclusions
Fasudil, a Rho kinase inhibitor, protects neurons through anti-apoptosis by reducing ROCK II, JNK, ASK1, and calcium and protecting mitochondrial membrane potential in the cellular model of AD. The results suggest that fasudil can be a novel drug for treatment of AD, although the in vivo mechanisms still need to be clarified.
References
Amin FU, Shah SA, Badshah H, Khan M, Kim MO (2017) Anthocyanins encapsulated by PLGA@PEG nanoparticles potentially improved its free radical scavenging capabilities via p38/JNK pathway against Abeta1-42-induced oxidative stress. J Nanobiotechnology 15(1):12. https://doi.org/10.1186/s12951-016-0227-4
Amoroso N, La Rocca M, Bruno S, Maggipinto T, Monaco A, Bellotti R, Tangaro S (2018) Multiplex networks for early diagnosis of Alzheimer's disease. Front Aging Neurosci 10:365. https://doi.org/10.3389/fnagi.2018.00365
Ando K, Oka M, Ohtake Y, Hayashishita M, Shimizu S, Hisanaga S, Iijima KM (2016) Tau phosphorylation at Alzheimer's disease-related Ser356 contributes to tau stabilization when PAR-1/MARK activity is elevated. Biochem Biophys Res Commun 478(2):929–934. https://doi.org/10.1016/j.bbrc.2016.08.053
Chen C, Li YH, Zhang Q, Yu JZ, Zhao YF, Ma CG, Xiao BG (2014) Fasudil regulates T cell responses through polarization of BV-2 cells in mice experimental autoimmune encephalomyelitis. Acta Pharmacol Sin 35(11):1428–1438. https://doi.org/10.1038/aps.2014.68
Chen C, Yu JZ, Zhang Q, Zhao YF, Liu CY, Li YH, Yang WF, Ma CG, Xiao BG (2015) Role of rho kinase and Fasudil on synaptic plasticity in multiple sclerosis. NeuroMolecular Med 17(4):454–465. https://doi.org/10.1007/s12017-015-8374-6
Chen C, Gu S, Jiang X, Zhang Z (2017a) Arsenite-induced endoplasmic reticulum-dependent apoptosis through disturbance of calcium homeostasis in HBE cell line. Environ Toxicol 32(1):197–216. https://doi.org/10.1002/tox.22226
Chen H, Shao X, Li L, Zheng C, Xu X, Hong X, Li X, Wu M (2017b) Electroacupuncture serum inhibits TNFalphamediated chondrocyte inflammation via the RasRafMEK1/2ERK1/2 signaling pathway. Mol Med Rep 16(5):5807–5814. https://doi.org/10.3892/mmr.2017.7366
Chen J, Sun Z, Jin M, Tu Y, Wang S, Yang X, Chen Q, Zhang X, Han Y, Pi R (2017c) Inhibition of AGEs/RAGE/rho/ROCK pathway suppresses non-specific neuroinflammation by regulating BV2 microglial M1/M2 polarization through the NF-kappaB pathway. J Neuroimmunol 305:108–114. https://doi.org/10.1016/j.jneuroim.2017.02.010
Choi, H., Kim, C., Song, H., Cha, M. Y., Cho, H. J., Son, S. M., Kim H. J. Mook-Jung, I. (2018). Amyloid beta-induced elevation of O-GlcNAcylated c-Fos promotes neuronal cell death. Aging Cell, e12872. https://doi.org/10.1111/acel.12872
Chong CM, Ai N, Lee SM (2017) ROCK in CNS: different roles of isoforms and therapeutic target for neurodegenerative disorders. Curr Drug Targets 18(4):455–462. https://doi.org/10.2174/1389450117666160401123825
Choudhary GS, Al-Harbi S, Almasan A (2015) Caspase-3 activation is a critical determinant of genotoxic stress-induced apoptosis. Methods Mol Biol 1219:1–9. https://doi.org/10.1007/978-1-4939-1661-0_1
Compagnucci C, Barresi S, Petrini S, Bertini E, Zanni G (2015) Rho-kinase signaling controls nucleocytoplasmic shuttling of class IIa histone deacetylase (HDAC7) and transcriptional activation of orphan nuclear receptor NR4A1. Biochem Biophys Res Commun 459(2):179–183. https://doi.org/10.1016/j.bbrc.12.033
Dean DN, Pate KM, Moss MA, Rangachari V (2016) Conformational dynamics of specific Abeta oligomers govern their ability to replicate and induce neuronal apoptosis. Biochemistry 55(15):2238–2250. https://doi.org/10.1021/acs.biochem.6b00161
Dhivya Bharathi M, Justin-Thenmozhi A, Manivasagam T, Ahmad Rather M, Saravana Babu C, Mohamed Essa M, Guillemin GJ (2018) Amelioration of aluminum Maltolate-induced inflammation and endoplasmic reticulum stress-mediated apoptosis by Tannoid principles of Emblica officinalis in neuronal cellular model. Neurotox Res 35:318–330. https://doi.org/10.1007/s12640-018-9956-5
Ding J, Yu JZ, Li QY, Wang X, Lu CZ, Xiao BG (2009) Rho kinase inhibitor Fasudil induces neuroprotection and neurogenesis partially through astrocyte-derived G-CSF. Brain Behav Immun 23(8):1083–1088. https://doi.org/10.1016/j.bbi.2009.05.002
Ding J, Mooers BH, Zhang Z, Kale J, Falcone D, McNichol J, Huang B, Zhang XC, Xing C, Andrews DW, Lin J (2014) After embedding in membranes antiapoptotic Bcl-XL protein binds both Bcl-2 homology region 3 and helix 1 of proapoptotic Bax protein to inhibit apoptotic mitochondrial permeabilization. J Biol Chem. 25;289(17):11873-96. https://doi.org/10.1074/jbc.M114.552562
Eidenmuller J, Fath T, Maas T, Pool M, Sontag E, Brandt R (2001) Phosphorylation-mimicking glutamate clusters in the proline-rich region are sufficient to simulate the functional deficiencies of hyperphosphorylated tau protein. Biochem J 357(Pt 3):759–767
Gao Y, Su Y, Qu L, Xu S, Meng L, Cai SQ, Shou C (2011) Mitochondrial apoptosis contributes to the anti-cancer effect of Smilax glabra Roxb. Toxicol Lett 207(2):112–120. https://doi.org/10.1016/j.toxlet.2011.08.024
Gotz J, Schild A, Hoerndli F, Pennanen L (2004) Amyloid-induced neurofibrillary tangle formation in Alzheimer's disease: insight from transgenic mouse and tissue-culture models. Int J Dev Neurosci 22(7):453–465. https://doi.org/10.1016/j.ijdevneu.2004.07.013
Guo D, Bi H, Wang D, Wu Q (2013) Zinc oxide nanoparticles decrease the expression and activity of plasma membrane calcium ATPase, disrupt the intracellular calcium homeostasis in rat retinal ganglion cells. Int J Biochem Cell Biol 45(8):1849–1859. https://doi.org/10.1016/j.biocel.2013.06.002
Guo MF, Meng J, Li YH, Yu JZ, Liu CY, Feng L, Yang WF, Li JL, Feng QJ, Xiao BG, Ma CG (2014) The inhibition of rho kinase blocks cell migration and accumulation possibly by challenging inflammatory cytokines and chemokines on astrocytes. J Neurol Sci 343(1–2):69–75. https://doi.org/10.1016/j.jns.2014.05.034
Guo X, Namekata K, Kimura A, Harada C, Harada T (2017) ASK1 in neurodegeneration. Adv Biol Regul 66:63–71. https://doi.org/10.1016/j.jbior.2017.08.003
Han XJ, Hu YY, Yang ZJ, Jiang LP, Shi SL, Li YR, Guo MY, Wu HL, Wan YY (2017) Amyloid β-42 induces neuronal apoptosis by targeting mitochondria. Mol Med Rep 16:4521–4528. https://doi.org/10.3892/mmr.2017.7203
Hashimoto Y, Niikura T, Chiba T, Tsukamoto E, Kadowaki H, Nishitoh H, Yamagishi Y, Ishizaka M, Yamada M, Nawa M, Terashita K, Aiso S, Ichijo H, Nishimoto I (2003) The cytoplasmic domain of Alzheimer's amyloid-beta protein precursor causes sustained apoptosis signal-regulating kinase 1/c-Jun NH2-terminal kinase-mediated neurotoxic signal via dimerization. J Pharmacol Exp Ther 306(3):889–902. https://doi.org/10.1124/jpet.103.051383
Henderson BW, Gentry EG, Rush T, Troncoso JC, Thambisetty M, Montine TJ, Herskowitz JH (2016) Rho-associated protein kinase 1 (ROCK1) is increased in Alzheimer's disease and ROCK1 depletion reduces amyloid-beta levels in brain. J Neurochem 138(4):525–531. https://doi.org/10.1111/jnc.13688
Hu J, Ramshesh VK, McGill MR, Jaeschke H, Lemasters JJ (2016) Low dose acetaminophen induces reversible mitochondrial dysfunction associated with transient c-Jun N-terminal kinase activation in mouse liver. Toxicol Sci 150(1):204–215. https://doi.org/10.1093/toxsci/kfv319
Imarisio C, Alchera E, Bangalore Revanna C, Valente G, Follenzi A, Trisolini E, Boldorini R, Carini R (2017) Oxidative and ER stress-dependent ASK1 activation in steatotic hepatocytes and Kupffer cells sensitizes mice fatty liver to ischemia/reperfusion injury. Free Radic Biol Med 112:141–148. https://doi.org/10.1016/j.freeradbiomed.2017.07.020
Jadhav S, Cubinkova V, Zimova I, Brezovakova V, Madari A, Cigankova V, Zilka N (2015) Tau-mediated synaptic damage in Alzheimer's disease. Transl Neurosci 6(1):214–226. https://doi.org/10.1515/tnsci-2015-0023
Jiang Y, Liu H, Ji B, Wang Z, Wang C, Yang C, Pan Y, Chen J, Cheng B, Bai B (2018) Apelin13 attenuates ER stressassociated apoptosis induced by MPP+ in SHSY5Y cells. Int J Mol Med 42(3):1732–1740. https://doi.org/10.3892/ijmm.2018.3719
Kanamoto T, Mota M, Takeda K, Rubin LL, Miyazono K, Ichijo H, Bazenet CE (2000) Role of apoptosis signal-regulating kinase in regulation of the c-Jun N-terminal kinase pathway and apoptosis in sympathetic neurons. Mol Cell Biol 20(1):196–204
Kim IH, Kwon MJ, Jung JH, Nam TJ (2018) Protein extracted from Porphyra yezoensis prevents cisplatin-induced nephrotoxicity by downregulating the MAPK and NF-kappaB pathways. Int J Mol Med 41(1):511–520. https://doi.org/10.3892/ijmm.2017.3214
Kirkitadze MD, Kowalska A (2005) Molecular mechanisms initiating amyloid beta-fibril formation in Alzheimer's disease. Acta Biochim Pol 52(2):417–423
Koch JC, Tatenhorst L, Roser AE, Saal KA, Tonges L, Lingor P (2018) ROCK inhibition in models of neurodegeneration and its potential for clinical translation. Pharmacol Ther 189:1–21. https://doi.org/10.1016/j.pharmthera.2018.03.008
Korkmaz OT, Ay H, Aytan N, Carreras I, Kowall NW, Dedeoglu A, Tuncel N (2018) Vasoactive intestinal peptide decreases beta-amyloid accumulation and prevents brain atrophy in the 5xFAD mouse model of Alzheimer's disease. J Mol Neurosci 68:389–396. https://doi.org/10.1007/s12031-018-1226-8
Lai AY, McLaurin J (2018) Rho-associated protein kinases as therapeutic targets for both vascular and parenchymal pathologies in Alzheimer's disease. J Neurochem 144(5):659–668. https://doi.org/10.1111/jnc.14130
Lazebnik YA, Kaufmann SH, Desnoyers S, Poirier GG, Earnshaw WC (1994) Cleavage of poly(ADP-ribose) polymerase by a proteinase with properties like ICE. Nature 371(6495):346–347. https://doi.org/10.1038/371346a0
Lin J, Yu J, Zhao J, Zhang K, Zheng J, Wang J, Huang C, Zhang J, Yan X, Gerwick WH, Wang Q, Cui W, He S (2017) Fucoxanthin, a marine carotenoid, attenuates beta-amyloid oligomer-induced neurotoxicity possibly via regulating the PI3K/Akt and the ERK pathways in SH-SY5Y cells. Oxidative Med Cell Longev 2017:6792543–6792510. https://doi.org/10.1155/2017/6792543
Liu Y, Liu Z, Wang Y, Liang YR, Wen X, Hu J, Yang X, Liu J, Xiao S, Cheng D (2013) Investigation of the performance of PEG-PEI/ROCK-II-siRNA complexes for Alzheimer's disease in vitro. Brain Res 1490:43–51. https://doi.org/10.1016/j.brainres.2012.10.039
Liu CY, Guo SD, Yu JZ, Li YH, Zhang H, Feng L, Chai Z, Yuan HJ, Yang WF, Feng QJ, Xiao BG, Ma CG (2015) Fasudil mediates cell therapy of EAE by immunomodulating encephalomyelitic T cells and macrophages. Eur J Immunol 45(1):142–152. https://doi.org/10.1002/eji.201344429
Losonczi JA, Olejniczak ET, Betz SF, Harlan JE, Mack J, Fesik SW (2000). NMR studies of the anti-apoptotic protein Bcl-xL in micelles. Biochemistry. 12;39(36):11024-33
Martire S, Fuso A, Mosca L, Forte E, Correani V, Fontana M et al (2016) Bioenergetic impairment in animal and cellular models of Alzheimer's disease: PARP-1 inhibition rescues metabolic dysfunctions. J Alzheimers Dis 54(1):307–324. https://doi.org/10.3233/JAD-151040
Morishima Y, Gotoh Y, Zieg J, Barrett T, Takano H, Flavell R, Davis RJ, Shirasaki Y, Greenberg ME (2001) Beta-amyloid induces neuronal apoptosis via a mechanism that involves the c-Jun N-terminal kinase pathway and the induction of Fas ligand. J Neurosci 21(19):7551–7560
Mu JS, Lin H, Ye JX, Lin M, Cui XP (2015) Rg1 exhibits neuroprotective effects by inhibiting the endoplasmic reticulum stress-mediated c-Jun N-terminal protein kinase apoptotic pathway in a rat model of Alzheimer's disease. Mol Med Rep 12(3):3862–3868. https://doi.org/10.3892/mmr.2015.3853
Obulesu M, Lakshmi MJ (2014) Apoptosis in Alzheimer's disease: an understanding of the physiology, pathology and therapeutic avenues. Neurochem Res 39(12):2301–2312. https://doi.org/10.1007/s11064-014-1454-4
Olivera Santa-Catalina M, Caballero Bermejo M, Argent R, Alonso JC, Centeno F, Lorenzo MJ (2017) JNK signaling pathway regulates sorbitol-induced tau proteolysis and apoptosis in SH-SY5Y cells by targeting caspase-3. Arch Biochem Biophys 636:42–49. https://doi.org/10.1016/j.abb.2017.11.004
Qi H, Shuai J (2016) Alzheimer's disease via enhanced calcium signaling caused by the decrease of endoplasmic reticulum-mitochondrial distance. Med Hypotheses 89:28–31. https://doi.org/10.1016/j.mehy.2016.01.022
Salminen A, Kaarniranta K, Kauppinen A, Ojala J, Haapasalo A, Soininen H, Hiltunen M (2013) Impaired autophagy and APP processing in Alzheimer's disease: the potential role of Beclin 1 interactome. Prog Neurobiol 106-107:33–54. https://doi.org/10.1016/j.pneurobio.2013.06.002
Sharma S, Verma S, Kapoor M, Saini A, Nehru B (2016) Alzheimer's disease like pathology induced six weeks after aggregated amyloid-beta injection in rats: increased oxidative stress and impaired long-term memory with anxiety-like behavior. Neurol Res 38(9):838–850. https://doi.org/10.1080/01616412.2016.1209337
Shen X, Zhu W, Zhang X, Xu G, Ju S (2011) A role of both NF-kappaB pathways in expression and transcription regulation of BAFF-R gene in multiple myeloma cells. Mol Cell Biochem 357(1–2):21–30. https://doi.org/10.1007/s11010-011-0871-9
Sheng JG, Jones RA, Zhou XQ, McGinness JM, Van Eldik LJ, Mrak RE, Griffin WS (2001) Interleukin-1 promotion of MAPK-p38 overexpression in experimental animals and in Alzheimer's disease: potential significance for tau protein phosphorylation. Neurochem Int 39(5–6):341–348
Skaper SD (2012) Alzheimer's disease and amyloid: culprit or coincidence? Int Rev Neurobiol 102:277–316. https://doi.org/10.1016/B978-0-12-386986-9.00011-9
Song J, Park KA, Lee WT, Lee JE (2014) Apoptosis signal regulating kinase 1 (ASK1): potential as a therapeutic target for Alzheimer's disease. Int J Mol Sci 15(2):2119–2129. https://doi.org/10.3390/ijms15022119
Szymanski J, Janikiewicz J, Michalska B, Patalas-Krawczyk P, Perrone M, Ziolkowski W et al (2017) Interaction of mitochondria with the endoplasmic reticulum and plasma membrane in calcium homeostasis, lipid trafficking and mitochondrial structure. Int J Mol Sci 18(7). https://doi.org/10.3390/ijms18071576
Tan D, Wen J, Li L, Wang X, Qian C, Pan M, Lai M, Deng J, Hu X, Zhang H, Guo J (2018) Inhibition of RhoA-subfamily GTPases suppresses Schwann cell proliferation through regulating AKT pathway Rather than ROCK pathway. Front Cell Neurosci 12:437. https://doi.org/10.3389/fncel.2018.00437
Tian J, Gan Y, Pan C, Zhang M, Wang X, Tang X, Peng X (2018) Nerol-induced apoptosis associated with the generation of ROS and ca(2+) overload in saprotrophic fungus Aspergillus flavus. Appl Microbiol Biotechnol 102(15):6659–6672. https://doi.org/10.1007/s00253-018-9125-z
Urresti J, Ruiz-Meana M, Coccia E, Arévalo JC, Castellano J, Fernández-Sanz C, Galenkamp KM, Planells-Ferrer L, Moubarak RS, Llecha-Cano N, Reix S, García-Dorado D, Barneda-Zahonero B, Comella JX (2016) Lifeguard inhibits Fas ligand-mediated endoplasmic reticulum-calcium release mandatory for apoptosis in type II apoptotic cells. J Biol Chem. 15;291(3):1221-34. https://doi.org/10.1074/jbc.M115.677682
Van Giau V, An SSA, Hulme JP (2018) Mitochondrial therapeutic interventions in Alzheimer's disease. J Neurol Sci 395:62–70. https://doi.org/10.1016/j.jns.2018.09.033
Wang X, Hu X, Yang Y, Takata T, Sakurai T (2016) Nicotinamide mononucleotide protects against beta-amyloid oligomer-induced cognitive impairment and neuronal death. Brain Res 1643:1–9. https://doi.org/10.1016/j.brainres.2016.04.060
Wang ZH, Zhu D, Xie S, Deng Y, Pan Y, Ren J, Liu HG (2017) Inhibition of rho-kinase attenuates left ventricular remodeling caused by chronic intermittent hypoxia in rats via suppressing myocardial inflammation and apoptosis. J Cardiovasc Pharmacol 70(2):102–109. https://doi.org/10.1097/FJC.0000000000000496
Wang J, Zheng J, Huang C, Zhao J, Lin J, Zhou X, Naman CB, Wang N, Gerwick WH, Wang Q, Yan X, Cui W, He S (2018) Eckmaxol, a Phlorotannin extracted from Ecklonia maxima, produces anti-beta-amyloid oligomer neuroprotective effects possibly via directly acting on glycogen synthase kinase 3beta. ACS Chem Neurosci 9(6):1349–1356. https://doi.org/10.1021/acschemneuro.7b00527
Wei X, Xu X, Chen Z, Liang T, Wen Q, Qin N, Huang W, Huang X, Li Y, Li J, He J, Wei J, Huang R (2018) Protective effects of 2-Dodecyl-6-Methoxycyclohexa-2,5 -Diene-1,4-Dione isolated from Averrhoa Carambola L. (Oxalidaceae) roots on neuron apoptosis and memory deficits in Alzheimer's disease. Cell Physiol Biochem 49(3):1064–1073. https://doi.org/10.1159/000493289
Wen X, Wang L, Liu Z, Liu Y, Hu J (2014) Intracranial injection of PEG-PEI/ROCK II-siRNA improves cognitive impairment in a mouse model of Alzheimer's disease. Int J Neurosci 124(9):697–703. https://doi.org/10.3109/00207454.2013.877014
West JD, Ji C, Marnett LJ (2005) Modulation of DNA fragmentation factor 40 nuclease activity by poly(ADP-ribose) polymerase-1. J Biol Chem 280(15):15141–15147. https://doi.org/10.1074/jbc.M413147200
Whitaker R, Fossey J, Ballard C, Orrell M, Moniz-Cook E, Woods RT, Murray J, Stafford J, Knapp M, Romeo R, Carlton B, Testad I, Khan Z (2014) Improving well-being and health for people with dementia (WHELD): study protocol for a randomised controlled trial. Trials 15:284. https://doi.org/10.1186/1745-6215-15-284
Xiao WD, Yu AX, Liu DL (2014) Fasudil hydrochloride could promote axonal growth through inhibiting the activity of ROCK. Int J Clin Exp Pathol 7(9):5564–5568
Xu T, Niu C, Zhang X, Dong M (2018) Beta-Ecdysterone protects SH-SY5Y cells against beta-amyloid-induced apoptosis via c-Jun N-terminal kinase- and Akt-associated complementary pathways. Lab Investig 98(4):489–499. https://doi.org/10.1038/s41374-017-0009-0
Yu JZ, Li YH, Liu CY, Wang Q, Gu QF, Wang HQ, Zhang GX, Xiao BG, Ma CG (2017) Multitarget therapeutic effect of Fasudil in APP/PS1transgenic mice. CNS Neurol Disord Drug Targets 16(2):199–209. https://doi.org/10.2174/1871527315666160711104719
Yu J, Yan Y, Gu Q, Kumar G, Yu H, Zhao Y, Liu C, Gao Y, Chai Z, Chumber J, Xiao BG, Zhang GX, Zhang HT, Jiang Y, Ma CG (2018) Fasudil in combination with bone marrow stromal cells (BMSCs) attenuates Alzheimer's disease-related changes through the regulation of the peripheral immune system. Front Aging Neurosci 10:216. https://doi.org/10.3389/fnagi.2018.00216
Zameer S, Kaundal M, Vohora D, Ali J, Kalam Najmi A, Akhtar M (2018) Ameliorative effect of alendronate against intracerebroventricular streptozotocin induced alteration in neurobehavioral, neuroinflammation and biochemical parameters with emphasis on Abeta and BACE-1. Neurotoxicology 70:122–134. https://doi.org/10.1016/j.neuro.2018.11.012
Zhao B, Chrest FJ, Horton WE Jr, Sisodia SS, Kusiak JW (1997) Expression of mutant amyloid precursor proteins induces apoptosis in PC12 cells. J Neurosci Res 47(3):253–263
Zhao YF, Zhang X, Ding ZB, Yang XW, Zhang H, Yu JZ, Li YH, Liu CY, Zhang Q, Zhang HZ, Ma CG, Xiao BG (2015) The therapeutic potential of rho kinase inhibitor fasudil derivative FaD-1 in experimental autoimmune encephalomyelitis. J Mol Neurosci 55(3):725–732. https://doi.org/10.1007/s12031-014-0411-7
Zhong L, Liu H, Zhang W, Liu X, Jiang B, Fei H, Sun Z (2018) Ellagic acid ameliorates learning and memory impairment in APP/PS1 transgenic mice via inhibition of beta-amyloid production and tau hyperphosphorylation. Exp Ther Med 16(6):4951–4958. https://doi.org/10.3892/etm.2018.6860
Zhou YY, Li Y, Jiang WQ, Zhou LF (2015) MAPK/JNK signalling: a potential autophagy regulation pathway. Biosci Rep 35(3). https://doi.org/10.1042/BSR20140141
Zolezzi JM, Lindsay CB, Serrano FG, Ureta RC, Theoduloz C, Schmeda-Hirschmann G, Inestrosa NC (2018) Neuroprotective effects of Ferruginol, Jatrophone, and Junicedric acid against amyloid-beta injury in hippocampal neurons. J Alzheimers Dis 63(2):705–723. https://doi.org/10.3233/JAD-170701
Funding
This work was supported by research grants from the National Natural Science Foundation of China (No. 81272163 and 81,371,414 to C-GM), the Department of Science and Technology, Shanxi Province of China (201803D421073 to YY, 201803D31079 to NPZ, 201805D111009 to CGM, 201805D131005 to JZY), Datong Municipal Science and Technology Bureau (2,017,134 to YY, 2019152 to NPZ, 2019072 to YG, 2019198 to C-GM), PhD Initiation Grant of Datong University (2016-B-01 to YY, 2018-B-01 to YG), Key Discipline of Internal Medicine Grant of Datong University (100,201 to NPZ)and the Open Project of State Key Laboratory of Molecular Developmental Biology, Institute of Genetics and Developmental Biology, Chinese Academy of Sciences, Beijing, China (2018-MDB-KF-07 to YY).
Author information
Authors and Affiliations
Contributions
YG, YY, QLF designed the study, carried out the primary neuron cell tests, immunoassays and western blotting. CGM, HTZ and YY conceived the study, participated in its design and coordination and helped draft the manuscript. NZ, GK participated in its design and revised the manuscript. HTZ revised and finalized the manuscript. JHZ and LHZ participated in the statistical analysis. LJS and JZY participated in figures revision, statistics and text revision. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Gao, Y., Yan, Y., Fang, Q. et al. The Rho kinase inhibitor fasudil attenuates Aβ1–42-induced apoptosis via the ASK1/JNK signal pathway in primary cultures of hippocampal neurons. Metab Brain Dis 34, 1787–1801 (2019). https://doi.org/10.1007/s11011-019-00487-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-019-00487-0