Abstract
Cognitive impairment is a progressive disorder that affects the ageing population. With the increase in the mean age of our population, it is becoming a public health problem. Homocysteinemia has been implicated in cognitive impairment. Whilst it is modulated by vitamins B12 and folate, it acts through MMPs 2 and 9. To assesses the relationship of cognitive impairment with homocysteine, B12, folate and MMPs 2 and 9, so as to detect cases of mild cognitive impairment which are potentially reversible, blood samples were drawn from 73 enrolled subjects, with and without cognitive impairment on basis of Montreal cognitive assessment (MoCA) score < 25 or ≥ 25, respectively. Homocysteine, B12, folate and MMPs2 and 9 were estimated. Correlation between MoCA score and these parameters was elucidated. After adjusting for age and gender, homocysteine was the only significant independent predictor of MoCA score. Cut-off of homocysteine for prediction of MoCA < 25 was derived at 13.5 µmol/L(PPV = 59.6%; NPV = 79.2%). The equation derived for calculation of MoCA score from homocysteine is: MoCA score = 32.893 + [(− 0.223)(homocysteine in μmol/L)]. Homocysteine > 13.5 μmol/L predicts low MoCA (< 25) with 84.8% sensitivity and 50% specificity. Hence, patients with an Hcy > 13.5 μmol/L should be administered B12 and folate to reduce homocysteine, a modifiable risk factor of cognitive decline. Also, a novel equation for calculating MoCA score from homocysteine has been derived. Using this derived equation to calculate MoCA score, it may be possible to identify asymptomatic subjects with early cognitive impairment.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Cognitive impairment is known to progress with age, especially those domains involving measures of speed of information processing, working memory and executive cognitive function [1]. In addition, over the last two centuries, life expectancy has doubled globally [2]. In fact the number of elderly subjects in the world is expected to increase from 93 million observed in 2011 to an expected 323 million in 2050, India being the second highest contributor after China [3]. We are, therefore, faced with a consequent increase in prevalence of cognitive impairment [4]. At the same time, there is a continued burden of nutritional deficiencies in India [5]. Many biomarkers have been implicated in the development of cognitive impairment, notable amongst them being homocysteinemia. It has been demonstrated that subjects in the highest quartile of homocysteine levels were more than two times as likely to be in the lowest quartile of neurobehavioral test scores as compared to those in the lowest quartile of homocysteine levels [6]. Also, serum homocysteine levels have been shown to bear a significant negative correlation with serum levels of vitamin B12 as well as folate [7]. It is pertinent here to note that homocysteinemia has the distinction (as opposed to other markers) of being modifiable through the appropriate administration of vitamins of the B group –B12 and folate [7, 8]. In addition, it has been postulated that the neuromodulatory effects of raised serum homocysteine act through the altered expression of matrix metalloproteinases [MMPs] especially MMPs 2 and 9 [9, 10].
The role of homocysteine in causation of cognitive decline may be segregated into short-term and long-term events as given in the Fig. 1 below. It is postulated that if the early events of vitamin deficiencies and homocysteinemia are corrected, further progression of cognitive decline may be prevented.

Modified from Bhargava, 2018 [11]
The Role of Homocysteine in Cognitive Decline. Short-term and long-term events in the progression of vitamin (B12, B6 and folate) deficiency and homocysteinemia-induced cognitive decline and its prevention: Early preclinical events could include successively – vitamin deficiencies, homocysteinemia, inhibition of post-synaptic GABA receptors, elevated MMP9 and phosphorylation of tau proteins. The long-term events could include accumulation of the phosphorylated tau proteins and amyloid-β-peptides into neurofibrillary tangles and amyloid plaques which would ultimately culminate in cognitive decline.
This study was conducted to evaluate the role of serum levels of homocysteine, vitamin B12, folate and the matrix metalloproteinases (MMP2 and MMP9) in cognitive impairment (CI) as defined by Montreal Cognitive Assessment (MoCA) score in a North Indian urban population [12]. The aim was to identify a nutritional strategy as a simple effective method to ameliorate homocysteinemia and reduce cognitive decline.
Materials and methods
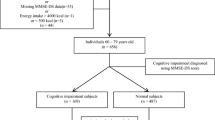
From amongst patients presenting to the neurology OPD with complaint of forgetfulness, a total of 73 subjects were enrolled over a period of 3 months. All subjects were subject to evaluation of higher mental functions, including lobar functions, by a detailed questionnaire. Also, MoCA score was calculated for all subjects. They were then segregated into those with and without CI (MoCA score < 25 and 25–30, respectively). Thus, we included 40 subjects with CI and 33 subjects without CI. Blood samples were drawn and the following analytes were estimated:
-
1.
Vitamin B12 and folate by chemiluminescent immunoassay
-
2.
Homocysteine by microparticle enzyme immunoassay
-
3.
MMP 2 and MMP 9 by ELISA
The data generated was subject to preliminary statistical analysis by SPSS version 20. All numerical variables were expressed as mean ± SEM or median and interquartile range as the case may be. Significance of difference between the two groups was derived. Since MoCA score represents the cognitive ability, the Spearman correlation of all variables with MoCA score was derived along with their significance by univariate analysis. Further, the data was subject to multivariate logistic regression and the emerging significant factor, homocysteine, was evaluated for its prediction of MoCA < 25 through its ROC, AUC NPV, PPV, sensitivity and specificity. Significance was ascertained at a p < 0.05.
Results
The descriptive statistics derived are given in Table 1. It was observed that age and homocysteine were the only parameters found significantly different in the two groups of subjects–those with and those without cognitive impairment; vitamin B12, folate, MMP2 and MMP9 were not significantly different in these two groups [Table 1].
The data was subject to statistical analysis for gender differences in the measured parameters in all subjects as a whole, and then in the two subgroups of those with and without cognitive impairment. It was observed that MoCA score was significantly higher in females as compared to males when all subjects’ data was grouped together. The details are given as mean ± SEM as well as median and interquartile range in Tables 2, 3 and 4 and the p value for each parameter is also tabulated in these.
Vitamin B12 and folate were found to bear a significant negative correlation to homocysteine as shown in Table 5.
Table 6 gives the correlation of MoCA score to all the parameters estimated—homocysteine, folate, vitamin B12, MMP2 and MMP9. Of these, MoCA correlation only to homocysteine and vitamin B12 were significant. Univariate analysis revealed that only homocysteine and vitamin B12 had a significant correlation with MoCA score.
In addition to estimating the circulating levels of the MMPs 2 and 9, the samples were processed for MMP 2 RNA expression as well. MMP2 gene is located on chromosome 16 at position 12.2.
Ct (constant threshold) was measured for expression of the SOI (sequence of interest; in this case, gene of interest) and RS (reference sequence or house-keeping gene) which was GAPDH in this case.
ΔCt was calculated
[Difference between Ct of SOI and Ct of RS, mostly house-keeping gene]
∴ ΔCt denotes the expression of MMP 2 gene.
Expression of MMP2 was calculated after normalization with GAPDH expression.
Pearson’s correlation was performed for the serum homocysteine levels and the MMP 2 gene expression. This is shown in Table 7.
Univariate analysis of the data revealed that MoCA scores significantly correlated with homocysteine and vitamin B12. The correlation graphs are given in Fig. 2.
This data was further subject to multivariate logistic regression for evaluation before and after adjusting for gender and age. It emerged that homocysteine was the only significant independent variable for MOCA scores after adjusting for age and gender, as given in Table 8.
Further, it was elucidated that the Durbin-Watson statistic was 1.75, which indicates a good positive autocorrelation. The statistical output was used to derive an equation for the prediction of MoCA based on serum homocysteine levels.
The ROC curve was then drawn and area under the curve was quantified as shown in Fig. 3.
The characteristics of homocysteine > 13.5 μmol/L (the cut-off derived from the ROC curve) as a predictor for low MOCA score is given in Table 9.
Summary of results
Amongst vitamin B12, folate, homocysteine and the MMPs 2 and 9, cognitive score (MoCA) indirectly correlated with homocysteine and vitamin B12. After adjusting for age and gender, homocysteine was the only significant independent predictor of MoCA score. Regression analysis revealed cut off value of homocysteine for prediction of MoCA < 25 as 13.5 µmol/L (PPV = 59.6%; NPV = 79.2%; Sensitivity = 84.8%; Specificity = 50%). The equation derived for calculation of MoCA score from homocysteine is:
Discussion
The term cognition encompasses all the brain functions related to memory, learning, spatial orientation, reasoning, judgement, planning and problem solving. The Diagnostic and Statistical Manual of Mental Disorders 5 (DSM 5) defines key domains of cognitive function: executive function, learning and memory, perceptual motor function, language complex attention and social cognition. Cognitive impairment is defined as decline in cognitive ability that is acquired (as opposed to developmental), and may have an underlying brain pathology. The causes may vary between different types of disorders but most include damage to the portions of brain involved in cognitive abilities, i.e. frontal and temporal cortex, basal ganglia, limbic system (thalamus, hypothalamus, hippocampus, amygdala, cingulate gyrus) and even the cerebellum. The common causes are degenerative dementias, stroke with vascular/mixed dementia, genetic causes, traumatic brain injuries, metabolic factors of which homocysteinemia is well-known and there is supporting evidence suggesting association of homocysteinemia in cognitive functions from previous studies. Neuropathologically, in such cases, there may be depositions of β amyloid as plaques, tau protein tangles, presence of Lewy bodies, ischemic demyelination, infarcts with angiopathic changes in the brain [12,13,14,15].
In 2002, approximately 5.4 million (22.2%) people of the USA who were elderly (> 70 years old) had cognitive impairment [16]. Of these, 10–15% of those with homocysteinemia progressed to Alzheimer’s Disease, whereas only 1–2.5% of those without homocysteinemia progressed to AD [17]. Qadri et al. measured vitamin B12, folate and homocysteine in 283 subjects. They observed that subjects in the lowest quartile of folate had an increased odds ratio for mild cognitive impairment (3.1) and dementia (3.8). They also observed that those with homocysteinemia had an increased odds ratio (4.3) for dementia [18]. In a population based study by Schafer et al. linear regression models revealed that homocysteine was consistently and strongly associated with worse neurobehavioral test performance. The same study elucidated that subjects in the highest quartile of homocysteine levels were more than two times as likely to be in the lowest quartile of neurobehavioral test scores as compared to those in the lowest quartile of homocysteine levels [6].
Through a case series, McCaddon demonstrated the beneficial effects of vitamin B12 and folate supplements on homocysteine as well as the cognitive score [19]. Malouf and Grimley conducted a randomized clinical trial in elderly subjects with raised circulating homocysteine levels with and without cognitive impairment. In those without cognitive impairment, they administered a daily dose of 800 mcg of folate for 3 years and demonstrated better global functioning (p = 0.033), better memory storage (p = 0.006) and better information processing speed (p = 0.016). Those who had cognitive impairment were daily administered choline esterase inhibitors and 1000 mcg of folate for the same period resulting in better overall response (OR = 4.06; p = 0.02) and a better Nurse’s Observation scale for Geriatric Patients (p = 0.002) [20].
The present study demonstrated that in North Indian urban population, vitamin B12 bears a significant correlation with the cognitive score, whereas folate does not as opposed to the above mentioned studies. Should we then presume that this population is not deficient in folate and therefore it does not impact cognitive scores? Maybe. In favour of this assumption is our retrospective data of approximately 48,000 subjects where we observed that the prevalence of deficiency of vitamin B12 and folate are 44.1% and 2.2%, respectively, which could explain the lack of correlation of the cognitive scores with folate [21]. After adjusting for age and gender, multivariate analysis elucidated that, amongst the analytes studied, homocysteine alone impacted the cognitive score. Regression analysis indicated the cut-off value of homocysteine as 13.5 µmol/L for prediction of MoCA score < 25. On basis of this analysis, an equation has been derived for calculating the MoCA score from homocysteine levels. In one of our previous studies, we demonstrated a reduction in homocysteine levels by supplementation with B12 and folate, irrespective of the baseline levels of these vitamins, which means that with or without prevalent deficiency, homocysteine was lowered [5].
Hence, it may be postulated that if homocysteine is reduced by supplementing with vitamin B12 and folate, the cascade of events leading to cognitive decline may be prevented, as shown in Fig. 1. This could reduce the burden of cognitive impairment and, thence, of dementia/Alzheimer’s disease.
It is, therefore, suggested that all subjects > 50 years of age (the lower age limit of our subjects demonstrating cognitive impairment) should be tested for circulating levels of homocysteine and those with a level > 13.5 µmol/L should be administered supplements of vitamin B12 and folate, and followed up for cognitive impairment. By calculating MoCA score from serum homocysteine using the derived equation, it may be possible to identify asymptomatic subjects with early cognitive impairment.
Conclusion
Homocysteine > 13.5 μmol/L predicts low MoCA (< 25) with 84.8% sensitivity and 50% specificity. So subjects with Hcy > 13.5 μmol/L (which is within its BRI of 5–15 μmol/L) should be carefully evaluated for the presence/progression of dementia and administered B12 and folate as a measure towards amelioration of homocysteinemia, the modifiable risk factor of cognitive decline.
In addition, an equation for calculating MoCA score from homocysteine has been derived in the present study. This, to the best of our knowledge, is a novel equation. By calculating MoCA score from serum homocysteine using this derived equation, it may be possible to identify asymptomatic subjects with early cognitive impairment.
Further studies on a larger population are suggested to corroborate the results using this equation.
Data availability
Enquiries about data availability should be directed to the authors.
References
Murman DL (2015) The impact of age on cognition. Semin Hear 36(3):111–121
https://www.nature.com/scitable/content/life-expectancy-around-the-world-has-increased-19786/
United Nations (2002) World population ageing, 1950–2050. United Nations Department of Economic and Social Affairs, New York
Peters R (2006) Ageing and the brain. Postgrad Med J 82:84–88
Gopalan C (2013) The changing nutrition scenario. Indian J Med Res 138(3):392–397
Schafer JH, Glass TA, Bolla KI, Mintz M, Jedlicka AE, Schwartz BS (2005) Homocysteine and cognitive function in a population-based study of older adults. J Am Geriatr Soc 53:381–388
Bhargava S, Ali A, Bhargava EK, Manocha A, Kankra M, Das S, Srivastava LM (2012) Lowering homocysteine and modifying nutritional status with folate and vitamin B12 in Indian patients of vascular disease. J Clin Biochem Nutr 50(3):222–226
Collaboration HLT (1998) Lowering blood homocysteine with folate based supplements: meta-analysis of randomised trials Homocysteine Lowering Trialists’ Collaboration. BMJ 316(7135):894–898
Lominadze D, Roberts AM, Tyagi N, Moshal KS, Tyagi SC (2006) Homocysteine causes cerebrovascular leakage in mice. Am J Physiol Heart Circ Physiol 290(3):H1206. https://doi.org/10.1152/ajpheart.00376.2005
Hainsworth AH, Yeo NE, Weekman EM (1862) Wilcock DM (2016) Homocysteine, homocysteinemia and vascular contributions to cognitive impairment and dementia (VCID). Biochim Biophys Acta 5:1008–1017
Bhargava S (2018) Homocysteinemia and its neurological effects. In: Bhargava S (ed) The clinical application of homocysteine. Springer Books International, Cham, p 46
Nasreddine ZS, Phillips NA, Bédirian V, Charbonneau S, Whitehead V, Collin I, Cummings JL, Chertkow H (2005) The montreal cognitive assessment, MoCA: a brief screening tool for mild cognitive impairment. J Am Geriatr Soc 53(4):695–699
Regier DA, Kuhl EA, Kupfer DJ (2013) The DSM-5: classification and criteria changes. World Psychiatry 12(2):92–98. https://doi.org/10.1002/wps.20050.PMID:23737408;PMCID:PMC3683251
Smith AD, Refsum H, Bottiglieri T, Fenech M, Hooshmand B, McCaddon A, Miller JW, Rosenberg IH, Obeid R (2018) Homocysteine and dementia: an international consensus statement. J Alzheimers Dis 62(2):561–570. https://doi.org/10.3233/JAD-171042.PMID:29480200;PMCID:PMC5836397
Mufson EJ, Binder L, Counts SE, DeKosky ST, deTolledo-Morell L, Ginsberg SD, Ikonomovic MD, Perez SE, Scheff SW (2012) Mild cognitive impairment: pathology and mechanisms. Acta Neuropathol 123(1):13–30
Bhargava S, Bhandari A, Choudhury S (2018) Role of homocysteine in cognitive impairement and alzheimer’s disease. Indian J Clin Biochem 33(1):16–20. https://doi.org/10.1007/s12291-017-0646-5.PMID:29371765;PMCID:PMC5766465
Plassman BL, Langa KM, Fisher GG, Heeringa SG, Weir DR, Ofstedal MB, Burke JR, Hurd MD, Potter GG, Rodgers WL, Steffens DC, McArdle JJ, Willis RJ, Wallace RB (2008) Prevalence of cognitive impairment without dementia in the United States. Ann Intern Med 148:427–434
Qadri P, Fragiacomo C, Pezzati R, Zanda E, Forloni G, Tettamanti M, Lucca U (2004) Homocysteine, folate and vitamin B12 in mild cognitive impairment, Alzheimers disease and vascular dementia. Am J Clin Nutr 80:114–122
McCaddon A (2006) Homocysteine and cognitive impairment: a case series in general practice setting. Nutr J 5:6
Malouf R, Grimley EJ (2003) Folate with or without B12 for the prevention and treatment of healthy elderly and demented people. Cochrane Database Syst Rev. 4:CD004514
Bhargava S, Srivastava LM, Manocha A, Kankra M, Rawat S (2022) Micronutrient deficiencies and anemia in Urban India—do we need food fortification? Ind J Clin Biochem 37(2):149–158. https://doi.org/10.1007/s12291-021-00966-1
Funding
This study was funded by the Research Development Program (RDP) of Sir Ganga Ram Hospital, New Delhi, India, under project no: 4.9.35-010.
Author information
Authors and Affiliations
Contributions
BS: conceptualized the study, reviewed literature, formulated the design of the study, performed biochemical analyses for estimation of homocysteine, vitamin B12 and folate, compiled the data, participated in analysis of the data, participated in writing and editing the manuscript and formulated the hypothesis. SPK: helped in formulation of the design of the study, identified subjects, assessed their cognitive abilities, reviewed and edited the manuscript. BA: helped in formulation of the design of the study, identified subjects, assessed their cognitive abilities, reviewed and edited the manuscript. CS: helped in formulation of the design of the study, performed analyses of samples for MMPs 2 and 9, helped in statistical analysis, helped in editing the manuscript. TP: helped in compilation of data and performed statistical analyses. MA, KM and SP: helped in review of literature, formulation of the design of the study, performing biochemical analyses for estimation of homocysteine, vitamin B12 and folate, compilation of data, editing of manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Ethical approval
This study was approved by the institutional ethics committee under no: EC/01/16/925.
Informed consent
Informed consent was obtained from all individual subjects included for participating in the study and publishing of the data as per the consent form approved by the ethics committee.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Seema, B., Prahlad, K.S., Anuradha, B. et al. Homocysteine and nutritional biomarkers in cognitive impairment. Mol Cell Biochem 478, 2497–2504 (2023). https://doi.org/10.1007/s11010-023-04679-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-023-04679-2