Abstract
Context
The problem of how ecological mechanisms create and interact with patterns across different scales is fundamental not only for understanding ecological processes, but also for interpretations of ecological dynamics and the strategies that organisms adopt to cope with variability and cross-scale influences.
Objectives
Our objective was to determine the consistency of the role of individual habitat patches in pattern-process relationships (focusing on the potential for dispersal within a network of patches in a fragmented landscape) across a range of scales.
Methods
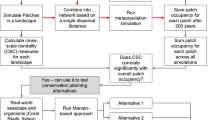
Network analysis was used to assess and compare the potential connectivity and spatial distribution of highland fynbos habitat in and between protected areas of the Western Cape of South Africa. Connectivity of fynbos patches was measured using ten maximum threshold distances, ranging from five to 50 km, based on the known average dispersal distances of fynbos endemic bird species.
Results
Network connectivity increased predictably with scale. More interestingly, however, the relative contributions of individual protected areas to network connectivity showed strong scale dependence.
Conclusions
Conservation approaches that rely on single-scale analyses of connectivity and context (e.g., based on data for a single species with a given dispersal distance) are inadequate to identify key land parcels. Landscape planning, and specifically the assessment of the value of individual areas for dispersal, must therefore be undertaken with a multi-scale approach. Developing a better understanding of scaling dependencies in fragmenting landscapes is of high importance for both ecological theory and conservation planning.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The problem of pattern and scale (as defined by Levin 1992) is concerned with understanding how the properties of units (e.g., habitat patches, neurons, or atoms) measured at one ecological scale aggregate at coarser or broader scales to create new units (e.g., ecosystems, brains, or materials). Scale in landscape ecology is generally defined as consisting of two components: extent (the area that is being considered), and resolution, or grain, which refers more generally to the precision of the measurement or to the pixel size (Gibson et al. 2000; Turner et al. 2001). Ecosystems are multi-scale entities, in which pattern-process interactions occur both within and across many different scales (Wu and Loucks 1995; Poiani et al. 2000; Mateo Sánchez et al. 2013). For example, the scales at which species-habitat relationships exist may differ from the scales at which biological interactions occur between organisms (Mateo Sánchez et al. 2013). In ecosystems the problem of pattern and scale is fundamental not only for understanding ecological processes, but also for interpretation of ecological dynamics and the strategies that organisms adopt in order to cope with variability and cross-scale influences (Levin 2000; Bakun and Broad 2003; Walters 2007). As Levin (1992) observed, these interpretations are in turn of fundamental importance for applications of ecological theory in the conservation and management of ecosystems (e.g., Boyd et al. 2008). Studies are therefore needed that consider pattern-process relationships interacting across a range of scales (Peters et al. 2007).
One of the major foci for pattern-process research in landscape ecology has been around the question of connectivity in fragmenting landscapes (Brooks 2003; Lindenmayer and Fischer 2006). As landscapes become fragmented, both theory and simulation models suggest that if fragmentation is either randomly distributed in space or structured by fine-scale processes such as localized land tenure, a rapid breakdown in apparent landscape connectivity within a given habitat type occurs when around 45 % of that habitat remains (Stauffer 1985; With and Crist 1995; Turner et al. 2001). This breakdown is predicted to have implications for the movements of animals across the landscape, affecting resource availability as well as a range of ecological processes (Driscoll et al. 2013). Most empirical and mechanistic studies of habitat connectivity have, however, focused on movements of individuals between populations of the same species and hence at a single scale of analysis (Swindle et al. 1999; Dunk et al. 2004). With a few exceptions (e.g., Matisziw et al. 2015), they have not gone beyond merely reporting the existence of scale effects and to the point of exploring their generalities across different landscapes (Wu 1999, 2004a). Similarly, fragmentation experiments at the community level have generally been undertaken at a single scale of analysis (Debinski and Holt 2000). Identifying and representing the relationships among many habitats simultaneously, for example as a network, is important when considering the broader ecological environment that a species utilizes (Matisziw et al. 2015). Many studies make the assumption that if organisms can move far enough to go between habitat patches at a single scale of analysis, they will be able to disperse effectively over the entire landscape. It is therefore unclear whether, or how much, the scale dependency of dispersal processes matters for analyses of fragmentation and connectivity.
Concerns over connectivity and corridors have had a major influence on conservation planning approaches, which often seek to identify and conserve areas that facilitate the movements of organisms across the landscape (Simberloff et al. 1992; Tigas et al. 2002; Pressey et al. 2007; Uezu et al. 2008). Most formal conservation planning exercises begin with the creation in a GIS environment of a ‘fishnet’ or vector grid of planning unit cells. These cells are then overlaid on GIS coverages of conservation-relevant features, such a species occurrences or river networks, and spatially explicit information about conservation features (e.g., number of lion sightings or length of stream per planning unit) is extracted into the planning unit coverage. The attribute table from the coverage is then run through decision support software, such as MARXAN (Ball and Possingham 2000), to generate conservation plans that prioritise particular parts of the landscape as conservation targets. Clearly it is important for conservation planning that suitable pieces of land are selected. Most conservation plans are, however, based on information that is collected or interpreted at only one or two scales of analysis (Ball and Possingham 2000; Geselbracht et al. 2009). For example, The Nature Conservancy’s ‘Conservation by Design’ approach adopted a two-scale approach, with areas first being stratified by ecoregion and then using a single grain and extent of planning unit to run existing data through MARXAN (TNC 2003). MARXAN includes a function that attempts to minimize patch edge lengths, but it does not directly consider connectivity; and estimation of the lengths of patch edges is strongly and non-linearly dependent on the grain of analysis. Empirical and theoretical questions have also emerged around scale in the role of the matrix in dispersal (Prugh et al. 2008; Franklin and Lindenmayer 2009), the importance of edge effects (Cadenasso and Pickett 2001; Cadenasso et al. 2003), and the role of relaxation and starting community effects in small and/or isolated patches (Gonzalez 2000; Terborgh et al. 2001; Kuussaari et al. 2009). Comprehensive empirical studies using real landscape data are needed to tell us what kinds of scaling relations may exist, and how variable or consistent they are.
Implicit in the use of a single analytical scale for conservation planning (such as the choice of a single grain and extent at which to develop a conservation plan, or the use of a single organism’s movement data as the basis for describing connectivity) is the assumption that the ecological importance of a given area (whether a single habitat patch or a larger area) is consistent across the full range of scales at which organisms of conservation concern use the landscape. In other words, it assumes that a high-priority patch for conservation action (for instance, as identified by consistent inclusion of that planning unit in a conservation plan generated by MARXAN at one grain of analysis) will remain a high-priority patch if the grain of analysis is changed. This assumption, which is an important but largely untested hypothesis, is of critical importance for our understanding of the ways in which ecological processes scale up and down. If the same area is important in the same way across multiple scales then there will be a scale-invariant relationship between spatial heterogeneity and ecological processes, and hence between landscape structure and ecological function. This would imply linear predictability in pattern-process relationships, meaning that an assessment at one scale of the role of a patch in a landscape could legitimately be extrapolated to its role in the broader extent/ecosystem. Alternatively, if our understanding of the ecological importance of individual areas is scale-dependent, single-species or single-grain studies cannot be generalized to the broader landscape. Pattern-process research must then focus more explicitly on understanding scaling functions and the relationships between landscape context, connectivity, and ecological processes. If supported, the second hypothesis also implies that conservation approaches that rely on analyses of connectivity and context using methods that consider only one grain size are inadequate to identify important land parcels for conservation.
We used a network analysis of the connectivity of protected areas in the Western Cape province of South Africa as a case study to contrast these two hypotheses. We considered that either (1) the importance of individual protected areas for habitat connectivity would remain constant over a range of different scales (i.e., each stepping stone or hub location would be important across a broad range of potential dispersal distances), supporting the first hypothesis, that of scale consistency; or (2) the importance of individual protected areas for habitat connectivity would change with scale, supporting the scale inconsistency hypothesis. Either outcome has significant implications for both theory and practice in ecology and conservation.
Methods
Study area
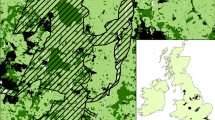
The Western Cape Province of South Africa contains the fynbos biome, which is renowned for its high levels of plant diversity and endemism and includes the Cape fynbos endemic bird area (EBA) (Cowling et al. 1994; Birdlife 2010). The EBA composed of at least 20 range-restricted and biome-restricted bird species, including six biome endemics that play an important pollinating role in fynbos and have varying dispersal distances (Huntley and Barnard 2012), (Table 1).
The fynbos in the Western Cape is highly fragmented. One-third of the area has been transformed into agricultural lands, plantations and urbanisation (Huntley and Barnard 2012). The Cape Town Metropolis contains the world’s highest concentration of threatened endemic fynbos species (Rebelo 1992). The Western Cape contains several hundred protected areas that are managed by various institutions and organizations. The statutory protected areas (Fig. 1) include national parks, which are regulated by South African National Parks (SANParks), and provincial parks that are managed by Cape Nature and governed by the Western Cape Conservation Board Act 15 of 1998. The study area also consists of numerous private protected areas, usually managed and owned by individuals or private organizations, either with or without formal government recognition (Mitchell 2005).
Data collection
We used the South African National Land Cover Dataset (NLCD), which is a national product generated from 2000 to 2001 Landsat Thematic Mapper (TM) satellite imagery, to identify the land cover classes in the Western Cape. This raster map consists of a panchromatic band with 15 m spatial resolution (band 8). Using GIS software (ArcGIS10, ESRI 2012), we extracted the highland fynbos, which includes thicket, bushland, bush clumps and high fynbos. This latter is defined as ‘fynbos communities between 2 and 5 m in height, >70 % cover, composed of multi-stemmed evergreen bushes typically growing on infertile soil and dominated by the Proteaceae family’ (Thompson 1996).
We used Conefor Inputs, a custom-made GIS extension for ArcGIS, to calculate the distances between all habitat patches using the nearest edge distance (Saura and Pascual-Hortal 2007). Intra-patch connectivity may present analytical problems as these measurements are based on the assumption that all habitat patches are equal in their individual contributions to connectivity (Matisziw and Murray 2008), when in fact, they may differ in size and quality. We avoided these problems by first converting fynbos habitat patches into a point dataset and then overlaying these data with the protected areas coverage to extract protection status and classify each habitat patch according to the protected area it fell within.
Spatial scales
Connectivity of the fynbos patches within the given protected areas were measured using threshold distances between patch polygons. Threshold distances are usually derived from data, such as the maximum dispersal distance for the species under study (Urban et al. 2009). We used the average dispersal distances of fynbos endemic bird species (Table 1) to generate ten maximum threshold distances, ranging from five to 50 km. These dispersal distances corresponded to the ten distance values (which we treat as measures of the spatial scale of landscape use by individual organisms) that we used in the analyses.
Protected area network
We used network analysis (Wasserman and Faust 1994; Borgatti et al. 2009; Cumming et al. 2010) to assess the functional connectivity of fynbos patches in protected areas. High fynbos patches within protected areas were represented as nodes and the links or edges between them represented the geographic distance between fynbos patches. Maximum threshold distances were used to define habitat networks; i.e., a pair of nodes in the network became connected if the Euclidean distance between the nodes was shorter than or equal to a certain threshold distance. This process was repeated ten times, creating ten different networks (i.e., one for each of our ten different spatial scales of analysis). A Fruchterman–Reingold Algorithm was used to visualize the resulting networks; this is a force-directed layout algorithm where the sum of the force vectors between the nodes uses a spring action to determine which direction a node should move (Fruchterman and Reingold 1991).
Network-level connectivity measures
Network-level connectivity, which has been identified as an influence on system resilience (Cumming 2011; Moore et al. 2014), was evaluated using network density, transitivity, number of clusters, network diameter, average path length and average degree for every network (see Table 2 for definitions). A Kolmogorov–Smirnov test was used to test for normality and then we used Pearson’s correlation coefficient to measure the correlation of these indices with increasing network size.
To determine habitat connectivity for each spatial scale we calculated average path length between the edges of each protected area habitat patch, which represents the average distance a bird that is randomly placed in the network is capable of dispersing between patches before reaching a barrier. We used bidirected edges because the direction of dispersal (A to B vs. B to A) was irrelevant for our analysis of network connectivity. The statistical significance of each network metric was determined by comparison to 1000 random networks at each scale, using the Erdös–Renyi algorithm, which preserves the number of nodes and edges in the real network while randomly modifying edge locations (Erdös and Renyi 1959).
Organizational level contribution to connectivity
To determine the role that protected areas that are managed by different organizations play in the network connectivity at each spatial scale we calculated the Eigenvector centrality (which is proportional to the sum of the neighbours’ centrality) for fynbos habitat patches under the jurisdiction of each organization at each maximum threshold distance (Table 2).
Protected area contribution to connectivity
The importance of individual protected area habitat patches to overall network connectivity at each different scale was determined by sequentially removing each habitat patch from the network, calculating network density using the ‘igraph’ package version 1.0.1 (Csardi and Nepusz 2006) in R, and then replacing it before repeating the process. An increase in network density increases network connectivity and should make the network more resilient to node removal (Kim and Anderson 2013). The removal of a node that causes a decrease in network density therefore represents a node with higher than average number of connections and hence, identifies a node that plays an important connecting role in the network. We used the node removal technique to identify individual protected area habitat patches that contribute the most to the overall connectivity of the network at each scale. We then ranked habitat patches according to their relative importance.
To explore the relationship between protected area habitat patch size and relative importance, we ran regression analyses and plotted network importance of each habitat patch against its extent and perimeter-to-area ratio at each scale of analysis. Pearson’s correlation coefficient was used to test for statistical significance.
Results
Network connectivity
When we increased the spatial scale of analysis from small (localized analysis) to large (regional analysis) between highland fynbos patches within protected areas, the number of connected nodes increased from 114 to 139 from a scale of 5–30 km and then remained constant until 50 km. The number of connected habitat patches therefore did not change when increasing the spatial scale from 30 to 50 km. As expected, the number of edges significantly increased (t = 25.69, p < 0.05, r = 0.99, df = 8, n = 10) with the shift from a small (243 edges at 5 km) to a larger spatial scale (1659 edges at 50 km; Fig. 2). The non-linear increase in the number of nodes and edges resulted in a significant decrease in network density (t = −3.296, p < 0.05, r = 0.576, df = 8, n = 10, Fig. 3a) as the density of a network is defined as a ration of the number of edges to the number of possible edges (Wasserman and Faust 1994). Conversely, transitivity and average degree of the protected area increased with scale (Fig. 3b, c). From a scale of 5 to 10 km the average path length significantly increased (t = 21.54, p < 0.05, r = 0.99, df = 8, n = 10) from 2.13 to 11.94 (Fig. 3d), largely due to the significant increase in the number of edges.
These trends in general are exactly what might be expected, but the non-linear nature of the trend is interesting. The non-linear transition from a disconnected distribution of habitat to a well-connected network appeared as an inflection in the plot of average path length against spatial scale (Fig. 3d). Increasing spatial scale from 5 to 10 km almost doubled the average path length, which increased from 2.36 to 3.77 km. At the 15 km scale the average path length (commuting distance across the network) dropped, and it continued to decrease until a spatial scale of 45 km (Fig. 3d).
The same pattern emerged when we compared the network metrics at each spatial scale to the randomly simulated networks (Fig. 4a–d). The networks at all spatial scales displayed transitivity values that were larger and number of clusters that were smaller than expected (Fig. 4a, b). Networks at all spatial scales except 20 km displayed average path lengths that were smaller than expected (Fig. 4c). The average path length in the 20 km network was longer than expected (20 km spatial scale = 4.241 km). The 5 and 20 km networks were the only two networks that had longer network diameters (5 km spatial scale = 7 km, 20 km spatial scale = 13 km) than expected (Fig. 4d).
The difference in network metrices: a transitivity, b number of clusters, c average path length and d diameter of networks when compared to the average of 1000 random networks using the Erdös–Renyi algorithm (Erdös and Renyi 1959) with spatial scale between high fynbos patches in protected areas in the Western Cape province of South Africa
Organization level contribution to connectivity
Increasing the spatial scale at which connection was possible from 5 to 10 km resulted in a significant increase in average Eigenvector centrality in national and provincial parks (Table 3; Fig. 5). As spatial scale increased from 10 to 15 km, degree centrality in provincial, national parks and private protected areas significantly decreased (Table 3; Fig. 5). The network contribution of provincial, national and private parks respectively continued to increase significantly between the 15 and 20 km networks, the 20 and 25 km networks, and the 25 and 30 km networks (Table 3; Fig. 5). Increasing the spatial scale between 30 and 35 km only resulted in a significant increase in centrality in provincial and private protected areas (Table 3; Fig. 5). Degree centrality increased in national, provincial and private protected areas when spatial scale increased from 35 to 40 km, 40 and 45 km, and 45–50 km (Table 3; Fig. 5).
Protected area contribution to connectivity
The relative importance in terms of connectivity of individual protected areas changed with spatial scale. Protected areas that ranked as important nodes at the finest scale (5 km) did not play an important connecting role at the other scales with the one exception of Hawequas Mountain Catchment Area. This protected area was ranked within the top three in the 10–35 km networks (Fig. 6). Hottentots-Holland Mountain Catchment Area was ranked within the top three in all protected area networks except the 5 km network (Fig. 6). Kogelberg Nature Reserve played an important role in the larger networks, 35–50 km (Fig. 6).
Protected area size was not statistically significant (p = 0.149) at any scale, suggesting that area and perimeter-to-area ratio were not good predictors of the relative importance of protected areas for connectivity.
Discussion
Our results show clearly that different protected areas in the Western Cape make different contributions to landscape connectivity at different scales. We thus found unequivocal evidence in support of the scale inconsistency hypothesis. While it is not surprising to find scale dependency in landscape pattern (Wu 2004b), the degree to which shifts in the relative importance of different individual protected areas (and correspondingly, of the mandates of different protected area organizations) occurred with scale was unexpectedly large.
Inconsistencies in the connectivity contribution of individual areas across the full range of scales, especially at the finer scales where different protected areas emerged as being more important in the protected area network, have important implications for both the ecology and the conservation of organisms. If organisms within the same community use the landscape at different scales, and the importance of individual patches of habitat is scale-dependent, then habitat structure will influence ecological interactions that have a strong movement-related component, such as predation, pathogen transmission, or pollination (Cumming et al. 2010). If mobile organisms are channeled through bottlenecks at certain points within a landscape then the probability of chance ecological interactions between or with them should be highest in patches that are hubs at several different scales, because dispersing organisms will have to move through these patches in order to access a broader landscape. For example, transmission of avian influenza viruses at waterbodies may be commonest at wetlands that function as hub sites for both far-ranging and more localized waterbirds (Brown et al. 2012; Gaidet et al. 2012; van Dijk et al. 2014).
In the conservation context, our analysis shows that conservation approaches that rely on an analysis of connectivity and context for a single species with a given dispersal distance are inadequate to identify important land parcels. Although it is already recognized that seemingly less significant patches may play an important role in the dispersal of individual species (Bodin et al. 2006), the relevance of scale for such analyses has been unclear and may lead to inappropriate and misleading planning conclusions (Pascual-Hotal and Saura 2007). As Mateo Sánchez et al. (2013) found in their study on brown bears, obtaining reliable predictions of species distribution and habitat strongly relies on independently optimizing the scale of analysis of each predictor variable. Unfortunately, it appears that the results of single-species or single-scale studies that identify key habitat patches within a landscape cannot be easily generalized. Developing a better understanding of scaling functions and the relationships between landscape context, connectivity, and ecological processes thus seems extremely important for conservation planning and related land use decisions. Doing so requires that multi-scale analysis is undertaken, in which the scale dependency of underlying assumptions about the appropriate grain at which to undertake a given exercise is explicitly confronted with comparable data from different grain sizes. This is most easily done by sequentially resampling the finest-grained land cover data set at coarser grains, and/or sequentially changing the sizes of the planning units adopted for conservation planning exercises, and then rerunning relevant analyses to explore whether and how these changes influence resulting conclusions and recommendations. For example, developing MARXAN-based conservation plans for the same area at five different grain sizes (planning unit dimensions), keeping MARXAN parameters the same each time, will distinguish between areas that are consistently selected by MARXAN as conservation priorities and those that are selected at one grain size only.
Moving from a fine to a coarse scale of analysis in the ecological protected area network of the Western Cape not only increased overall network connectivity, but indicated that scale breaks (inflection points where network connectivity changes with scale) occurred at distances of 10, 15 and 35 km. These scale breaks may explain the decrease in degree centrality in the protected area network when threshold distance was increased from 10 to 15 km. Particularly in a fragmented landscape, critical scales associated with changes in landscape connectivity should be considered when quantifying habitat pattern and the influence of habitat pattern on movement (Keitt et al. 1997). The average path length was shortest at the finest (5 km) and 35 km scale. At the 5 and 20 km threshold distances the large network diameter suggested fewer shortcuts than expected (Minor and Urban 2007). Organisms that cannot disperse more than 5 km therefore appear to be particularly vulnerable in Western Cape highland fynbos, as they have access to fewer habitat patches. In local terms, our results have important conservation implications for the protection of endemic, highland fynbos-dependent bird species (particularly Victorin’s Warbler and Cape Rock-Jumper) that disperse at average distances smaller than 5 km (Hockey et al. 2005). In 2012, for instance, the Cape Rock-Jumper was considered a species in decline that required urgent conservation attention (Lee and Barnard 2012).
The network at the 20 km scale also exhibited a larger average path length and higher number of clusters than was expected, compared to the other networks. A high clustering coefficient and large average path length may result in slow movement in a network (Reppas et al. 2011). This result may therefore have conservation implications for the Cape Sugarbird, Cape Rock-Jumper, the Southern Double-Collared Sunbird, Cape Siskin and Malachite Sunbird, which all disperse at this scale.
The socioeconomic implications of our analysis are also interesting. Areas falling under the jurisdiction of different organizations played different scale-dependent roles in the ecological connectivity of the protected area network. Provincial parks played the most important role across all scales when compared to the other organizations, as illustrated by their high centrality values. Within each organization, provincial parks and private protected areas displayed high centrality values at the finer and broadest scales. Centrality values of national parks increased with scale. This may relate to the spatial arrangement of these different organizations; provincial parks serve as key corridors promoting overall connectivity, private protected areas are small and scattered throughout the province, and national parks occur mostly in remote areas. These results suggest that the overall connectivity (and potentially the ecological resilience) of the protected area network is fostered by having a diversity of conservation organizations and different kinds and sizes of protected areas. The relevance of tenure diversity for social-ecological resilience has been well documented (Poteete and Ostrom 2004; Norberg et al. 2008), but it has not been previously shown to influence ecological connectivity.
In summary, multi-scale analysis shows that scientific perceptions of the relative contributions of individual areas to ecological connectivity are strongly scale-dependent. In the Western Cape, where protected areas play an important role in maintaining the geographic connectivity of the fynbos biome, the connectivity of the protected area network varies significantly with scale. Scale considerations are therefore of central importance for understanding ecological pattern-process relationships in this landscape and for effective conservation planning.
References
Baggio R, Scott N, Cooper C (2010) Network acience: a review focused on tourism
Bakun A, Broad K (2003) Environmental ‘loopholes’ and fish population dynamics: comparative pattern recognition with focus on El Nino effects in the Pacific. Fish Oceanogr 12(4/5):458–473
Ball I, Possingham H (2000) MARXAN (V1.8.2): marine reserve design using spatially explicit annealing, a manual. The Ecology Centre, University of Queensland, Brisbane
Bettsetter C (2002) On the minimum node degree and connectivity of a wireless multihop network. In: Proceedings of ACM international symposium. Mobile Ad Hoc Netw. and Como (MobiHoc)
BirdLife International (2010) Endemic Bird Area factsheet: Cape fynbos. Downloaded from 450 http://www.birdlife.org. Accessed 20 Aug 2014
Bodin Ö, Tengö M, Norman A, Lundberg J, Elmqvist T (2006) The value of small size: loss of forest patches and ecological thresholds in southern Madagascar. Ecol Appl 16(2):440–451
Borgatti SP, Mehra A, Brass DJ, Labianca G (2009) Network analysis in the social sciences. Science 323:892–895
Boyd C, Brooks TM, Butchart SHM, Edgar GJ, da Fonseca GAB, Hawkins F, Hoffmann M, Sechrest W, Stuart SN, van Dijk PP (2008) Spatial scale and the conservation of threatened species. Conserv Lett 1(1):37–43
Brooks CP (2003) A scalar analysis of landscape connectivity. Oikos 102(2):433–439
Brown JD, Berghaus RD, Costa TP, Poulson R, Carter DL, Lebarbenchon C, Stallknecht DE (2012) Intestinal excretion of a wild bird-origin H3N8 low pathogenic avian influenza virus in Mallards (Anas Platyrhynchos). J Wildl Dis 48(4): 991-998.
Cadenasso ML, Pickett STA (2001) Effect of edge structure on the flux of species into forest interiors. Conserv Biol 15(1):91–97
Cadenasso ML, Pickett STA, Weathers KC, Bell SS, Benning TL, Cerreiro MM, Dawson TE (2003) An interdisciplinary and synthetic approach to ecological boundaries. BioScience 53:717–722
Cowling RM, Hilton-Taylor C (1994) Patterns of plant diversity and endemism in Southern Africa: an overview. In: Huntley BJ (ed) Botanical Diversity in Southern Africa. National Botanical Institute, Pretoria, pp 31–52
Csardi G, Nepusz T (2006) The igraph software package for complex network research. Int J Complex Syst 1695(5):1–9
Cumming GS (2011) Spatial resilience: integrating landscape ecology, resilience, and sustainability. Landscape Ecol 26(7):899–909
Cumming GS, Bodin O, Ernstson H, Elmqvist T (2010) Network analysis in conservation biogeography: challenges and opportunities. Divers Distrib 16:414–425
Debinski DM, Holt RD (2000) A survey and overview of habitat fragmentation experiments. Conserv Biol 14(2):342–355
Driscoll DD, Banks SC, Barton PS, Lindenmayer DB, Smith AL (2013) Conceptual domain of the matrix in fragmented landscapes. Trends Ecol Evol 28(10):605–613
Dunk JR, Zielinski WJ, Priesler HK (2004) Predicting the occurrence of rare mollusks in northern California forests. Ecol Appl 14:713–729
Erdös P, Rényi R (1959) On random graphs. Publ Math Debr 6:290–297
ESRI (2012) ArcGIS desktop: release 10. Environmental Systems Research Institute, Redlands
Franklin JF, Lindenmayer DB (2009) Importance of matrix habitats in maintaining biological diversity. Proc Natl Acad Sci 106(2):349–350
Fruchterman TM, Reingold EM (1991) Graph drawing by force-directed placement. Softw Pract Exp 21(11):1129–1164
Gaidet N, El Mamy ABO, Cappelle J, Caron A, Cumming GS, Grosbois V, Gil P, Hammouni S, de Almeida RS, Fereidouni SR, Cattoli G, Abolnik C, Mundava J, Fofana B, Ndlovu M, Diawara Y, Hurtado R, Newman SH, Dodman T, Balanca G (2012) Investigating avian influenza infection hotspots in old-world shorebirds. PLoS One 7(9):e46049
Geselbracht L, Torres R, Cumming GS, Dorfman D, Beck M, Shaw D (2009) Identification of a spatially efficient portfolio of priority conservation sites in marine and estuarine areas of Florida. Aquat Conserv-Mar Freshw Ecosyst 19(4):408–420
Gibson CC, Ostrom E, Ahn T-K (2000) The concept of scale and the human dimensions of global change: a survey. Ecol Econ 32(2):217–239
Gonzalez A (2000) Community relaxation in fragmented landscapes: the relation between species richness, area and age. Ecol Lett 3(5):441–448
Hockey PAR, Dean WRJ, Ryan P (2005) Roberts Birds of Southern Africa, VIIth edn. The Trustees of the John Voelcker Bird Book Fund, Cape Town
Huntley B, Barnard P (2012) Potential impacts of climatic change on southern African birds of fynbos and grassland biodiversity hotspots. Divers Distrib 18(8):769–781
Janssen MA, Orjan B, Anderies JM, Elmqvist T, Ernstson H, McAllister RRJ, Per O, Ryan P (2006) Toward a network perspective of the study of resilience in social-ecological systems. Ecol Soc 11(1):15
Keitt TH, Urban DL, Milne BT (1997) Detecting critical scales in fragmented landscapes. Conserv Ecol 1(1):4
Kim H, Anderson R (2013) An experimental evaluation of robustness of networks. Syst J IEEE 7(2):179–188
Kuussaari M, Bommarco R, Heikkinen RK et al (2009) Extinction debt: a challenge for biodiversity conservation. Trends Ecol Evol 24(10):564–571
Lee ATK, Barnard P (2012) Endemic fynbos avifauna: comparative range declines a cause for concern. Ornithol Observ 3:19–28
Levin SA (1992) The problem of pattern and scale in ecology: the Robert H. MacArthur award lecture. Ecology 73(6):1943–1967
Levin SA (2000) Multiple scales and the maintenance of biodiversity. Ecosystems 3(6):498–506
Lindenmayer DB, Fischer J (2006) Habitat fragmentation and landscape change. Island Pres, Washington
Mateo Sánchez MC, Cushman SA, Saura S (2013) Scale dependence in habitat selection: the case of the endangered brown bear (Ursus arctos) in the Cantabrian Range (NW Spain). Int J Geogr Inf Sci 28(8):1531–1546
Matisziw TC, Murray AT (2008) Connectivity change in habitat networks. Landscape Ecol 24(1):89–100
Matisziw TC, Alam M, Trauth KM, Inniss EC, Semlitsch RD, McIntosh S, Horton J (2015) A vector approach for modeling landscape corridors and habitat connectivity. Environ Model Assess 20(1):1–16
Minor ES, Urban DL (2007) Graph theory as a proxy for spatially explicit population models in conservation planning. Ecol Appl 17(6):1771–1782
Mitchell BA (2005) Editorial. -Parks 15:6
Moore C, Cumming GS, Slingsby J, Grewar J (2014) Tracking socioeconomic vulnerability using network analysis: insights from an avian influenza outbreak in an ostrich production network. PLoS One 9(1):e86973
Norberg J, Wilson J, Walker B, Ostrom E (2008) Diversity and Resilience of social-ecological systems. In: Norberg J, Cumming GS (eds) Complexity theory for a sustainable future. Columbia University Press, New York, pp 46–79
Pascual-Hortal L, Saura S (2007) Impact of spatial scale on the identification of critical habitat patches for the maintenance of landscape connectivity. Landscape Urban Plan 83:176–186
Peters DPC, Bestelmeyer BT, Turner MG (2007) Cross-scale interactions and changing pattern-process relationships: consequences for system dynamics. Ecosystems 10(5):790–796
Poiani KA, Richter BD, Anderson MG, Richter HE (2000) Biodiversity conservation at multiple scales: functional sites, landscapes, and networks. Bioscience 50:133–146
Poteete A, Ostrom E (2004) Heterogeneity, group size, and collective action: the role of institutions in forest management. Dev Change 35(3):435–461
Pressey RL, Cabeza M, Watts ME, Cowling RM, Wilson KA (2007) Conservation planning in a changing world. Trends Ecol Evol 22(11):583–592
Prugh LR, Hodges KE, Sinclair ARE, Brashares JS (2008) Effect of habitat area and isolation on fragmented animal populations. Proc Natl Acad Sci 105:20770–20775
Rebelo A (1992) Red Data Book species in the Cape Floristic Region: threats, priorities and target species. Trans R Soc S Afr 48(1):55–86
Reppas AI, Spiliotis KG, Siettos CI (2011) On the effect of path length of small-world networks on epidemic dynamics. Virulence 3:146–153
Saura S, Pascual-Hortal L (2007) A new habitat availability index to integrate connectivity in landscape conservation planning: comparison with existing indices and application to a case study. Landscape and Urban Plan 83(2):91–103
Simberloff D, Farr JA, Cox J, Mehlman DW (1992) Movement corridors—conservation bargains or poor investments. Conserv Biol 6(4):493–504
Stauffer D (1985) Introduction to percolation theory. Taylor and Francis, London
Swindle KA, Ripple WJ, Meslow EC, Schafer D (1999) Old-forest distribution around spotted owl nests in the central Cascade Mountains, Oregon. J Wildl Manag 63(4):1212–1221
Terborgh J, Lopez L, Nunez P et al (2001) Ecological meltdown in predator-free forest fragments. Science 294(5548):1923–1926
Thompson M (1996) A standard land-cover classification scheme for remote-sensing applications in South Africa. S Afr J Sci 92:34–42
Tigas LA, Van Vuren DH, Sauvajot RM (2002) Behavioral responses of bobcats and coyotes to habitat fragmentation and corridors in an urban environment. Biol Conserv 108(3):299–306
TNC (2003) The five-s framework for site conservation: a practitioner’s handbook for site conservation planning and measuring conservation success. The Nature Conservancy, Washington, DC
Turner MG, Gardner RH, O’Neill RV (2001) Landscape ecology in theory and practice: pattern and process. Springer, Berlin
Uezu A, Beyer DD, Metzger JP (2008) Can agroforest woodlots work as stepping stones for birds in the Atlantic forest region? Biodivers Conserv 17(8):1907–1922
Urban DL, Minor ES, Treml EA, Schick RS (2009) Graph models of habitat mosaics. Ecol Lett 12(3):260–273
van Dijk JGB, Hoye BJ, Verhagen JH, Nolet BA, Fouchier RAM, Klaassen M (2014) Juveniles and migrants as drivers for seasonal epizootics of avian influenza virus. J Anim Ecol 83:266–275
Walters S (2007) Modeling scale-dependent landscape pattern, dispersal, and connectivity from the perspective of the organism. Landscape Ecol 22(6):867–881
Wasserman S, Faust K (1994) Social network analysis—methods and applications. Cambridge University Press, Cambridge
Watts DJaSHS (1998) Collective dynamics of ‘small-world’ networks. Nature 393:440–442
Weisstein EW (2015) Fast Fourier Transform, from MathWorld–A Wolfram Web Resource. http://mathworld.wolfram.com/FastFourierTransform.html
With KA, Crist TO (1995) Critical thresholds in species responses to landscape structure. Ecology 76(8):2446–2459
Wu J (1999) Hierarchy and scaling: extrapolating information along a scaling ladder. Can J Remote Sens 25:367–380
Wu J (2004a) Effects of changing scale on landscape pattern analysis: scaling relations. Landscape Ecol 19:125–138
Wu J (2004b) Effects of changing scale on landscape pattern analysis: scaling relations. Landscape Ecol 19(2):125–138
Wu J, Loucks OL (1995) From balance of nature to hierarchical patch dynamics: a paradigm shift in ecology. Q Rev Biol 70:439–466
Acknowledgments
This research was supported by the DST/NRF Centre of Excellence at the Percy FitzPatrick Institute of African Ornithology, a CPRR Grant to GSC from the National Research Foundation of South Africa, and a James S. McDonnell Foundation complexity scholar award to GSC.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Maciejewski, K., Cumming, G.S. Multi-scale network analysis shows scale-dependency of significance of individual protected areas for connectivity. Landscape Ecol 31, 761–774 (2016). https://doi.org/10.1007/s10980-015-0285-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10980-015-0285-2