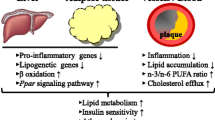
Abstract
Since 40 years, it is known that omega-3 poly-unsaturated fatty acids (ω3 PUFAs) have cardioprotective effects. These include antiarrhythmic effects, improvements of autonomic function, endothelial function, platelet anti-aggregation and inflammatory properties, lowering blood pressure, plaque stabilization and reduced atherosclerosis. However, recently, conflicting results regarding the health benefits of ω3 PUFAs from seafood or ω3 PUFAs supplements have emerged. The aim of this review is to examine recent literature regarding health aspects of ω3 PUFAs intake from fish or supplements, and to discuss different arguments/reasons supporting these conflicting findings.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Fish is a good source of protein and, unlike fatty meat products, it is poor in saturated fat. Fish is also a good source of omega-3 poly-unsaturated fatty acids (ω3 PUFAs). Forty years ago, the cardiovascular beneficial effects of ω3 PUFAs were first noticed when researchers identified lower rates of cardiovascular disease among Greenland Inuit, whose diet consisted of foods rich in ω3 PUFAs, such as whale, fish and seal, compared to the Danish population (Bang et al. 1976). By comparing plasma and food lipid profiles of both populations, the authors concluded that there was an association between dietary pattern and the incidence of cardiovascular disease. Also, a diet rich in ω3 PUFAs is associated with lower serum cholesterol and triglycerides, slower atherosclerotic plaque growing rate, and slightly lower arterial blood pressure (De Caterina 2011). Since then, epidemiological, clinical, animal and cellular studies confirmed these beneficial effects (Billman 2013). Consequently, the American Heart Association (AHA) recommends eating at least two serving (particularly oily) fish per week. ω3 PUFAs have thus generated considerable interest as well as controversies regarding their impact on cardiovascular physiology. A number of observational studies have shown that consumption of fish leads to a reduction of sudden cardiac death incidence (Burr et al. 1989; Siscovick et al. 1995). However, from the end of the 20st century, researchers failed to demonstrate a convincing cardioprotective effect and ignited the debate on whether PUFAs have healthy effects (Salonen et al. 1995; Ascherio et al. 1995; Kromhout et al. 1996; Pietinen et al. 1997; Guallar et al. 1999; Gillum et al. 2000; Oomen et al. 2000; Rissanen et al. 2000; Iso et al. 2006; Wilhelm et al. 2008; Yamagishi et al. 2008). As reviewed by Moreno et al. (2012), ω3 PUFAs have various effects on cardiac ion currents that could explain the contradictory effects of ω3 PUFAs. These differences appeared to depend on whether the ω3 PUFAs were applied to cells or formerly incorporated in membranes modifying its composition and, thus, the effects on cell signalling. In this paper, we review the experimental evidence supporting that the actions of ω3 PUFAs are due to pleiotropic mechanisms.
Environmental parameters
ω3 PUFAs have pleiotropic actions including cardioprotective effects, especially against post-infarction arrhythmias (Saravanan et al. 2010; Schuchardt et al. 2011). Recently, new drugs have been added to the standard treatment of cardiac pathologies, such as statins. The introduction of statins, a drug class used to lower LDL-cholesterol level by inhibiting HMG-COA reductase, can modulate ω3 PUFAs effects by interfering with their metabolism (de Lorgeril et al. 2013). The mechanism of action is still not well understood, but it seems that mitochondria could be involved, especially coenzyme Q10. In agreement with this hypothesis, a European secondary prevention clinical trial reports that ω3 supplementation (alpha-linolenic acid, Eicosapentaenoic acid; EPA and docoxahexaenoic acid; DHA) reduces the incidence of cardiovascular events by half only when statins were not prescribed (Eussen et al. 2012). Surprisingly, ω3 PUFAs cardioprotective effects are still observed in diabetic patients on statins (Kromhout et al. 2011), the same applies for the effects of ω3 PUFAs heart rate (Kim et al. 2011), on plasmatic triglyceride level and on the threshold of occurrence of ventricular palpitations induced by electrical stimulation (Durrington et al. 2001; Schrepf et al. 2004; ORIGIN Trial Investigators et al. 2012). An inhibition of ω3 PUFAs effects by statins cannot be simply explained by loss of efficiency of these lipids, which was demonstrated by the most recent clinical trials and meta-analysis. For example, in a diabetic population, it is possible to have a metabolic pathway altered by this pathology, which is sensitive to ω3 PUFAs and not to statins and vice versa.
Also, patients included in randomized clinical trials (RCT) using ω3 PUFAs can differ from the general population in an important way, which could explain the various results between RCTs (where there is intervention) and epidemiological studies. It was reported that patients who volunteered to join RCTs are frequently healthier and more active than the average population, which could affect the results in an unknown way. Furthermore, patients dropping out of the trials are often more ill and could be the ones benefiting most from ω3 PUFAs.
Nutritionally, cooking method are of importance. Consuming boiled fish is associated with a lower rate of fatal arrhythmias when compared to fried fish consumption (Mozaffarian et al. 2003). Also, mortality linked to coronary heart disease was lower in individuals consuming oily fish as opposed to white meat fish or no fish at all (Oomen et al. 2000).
The AHA nutrition recommendations advise to limit oily fish consumption of children. Indeed, fat fishes, such as farmed salmon, swordfish, king mackerel, or tilefish, are known to accumulate high levels of mercury, polychlorinated biphenyls, dioxins, organochlorines and other environmental contaminants (Hites et al. 2004). The levels of these substances are generally higher in older, larger, predatory fishes and marine mammals. Also, although a high rate of plasmatic DHA was associated with a reduction of the acute coronary artery events, this effect is significantly attenuated when increased levels of mercury are found in patient’s hair (Rissanen et al. 2000), suggesting an interaction between ω3 PUFAs and mercury. Also, it was suggested that a high intake of mercury from non-oily freshwater fish and the subsequent mercury accumulation in the body were associated with a higher an risk of myocardial infarction in Eastern Finnish men and this increased risk may be due to the promotion of lipid peroxidation by mercury (Salonen et al. 1995). More recently, Hu et al. (2016) studied the relationship between Canadian Inuit traditional diet and myocardial infarction risk in 2072 participants aged 18–79. They found that Inuit traditional diet contains high levels of EPA, DHA and mercury. A careful analysis of their results indicates that beneficial effects of EPA and DHA on myocardial infarction are diminished by the adverse effect of mercury (by 5% for men and 4% for women) (Hu et al. 2016). Thus, dietary mercury levels could partially explain certain contradictory results.
Finally, an important factor that is rarely taken into account in these studies is the inter-individual variability of incorporation of ω3 PUFAs in cell membranes (Arterburn et al. 2006; Bougnoux et al. 2009). In 2010, it has been shown that cardioprotective properties of ω3 PUFAs depend on their incorporation into cell membranes (von Schacky 2010), which suggests that positive effects needing incorporation can be masked in a sub-population which do not efficiently incorporate ω3 PUFAs in their cell membrane.
Cellular parameters
The controversy about ω3 PUFAs effects also extends to the cellular level (Billman 2013). The majority of electrophysiological single cell studies found an inhibition of ion currents such as fast sodium current (INa) (Xiao et al. 1995; Kang and Leaf 1996; Leifert et al. 2000), ultrafast activating delayed outward potassium current (IKur) (Honoré et al. 1994; Li et al. 2009), rapidly activating delayed rectifying outward potassium current (IKR) (Guizy et al. 2005), L-type calcium current (ICaL) (Xiao et al. 1997; Hazama et al. 1998; Rodrigo et al. 1999; Ferrier et al. 2002; Verkerk et al. 2006), and Na+–Ca2+ exchanger current (INCX) (Xiao et al. 2004; Ander et al. 2007). On the other hand, ω3 PUFAs were found to enhance slowly activating delayed rectifying outward potassium current (IKS) (Doolan et al. 2002) and outwardly rectifying potassium current (ITO) (Macleod et al. 1998; Judé et al. 2003; Li et al. 2009). Also, incorporation of ω3 PUFAs has been positively correlated with mitochondrial proton leak (Hulbert 2003), and increased mitochondrial DHA content through lipid infusion or dietary intervention augments proton movement and state 4 respiration (Stillwell et al. 1997). In addition, DHA enhances Na+ membrane permeability (Stillwell and Wassall 2003) and Na+/K+-ATPase activity (Else and Wu 1999; Turner et al. 2003; Wu et al. 2004).
Collectively, these studies have led to the idea that ω3 PUFAs influence mitochondrial activity, and excitation–contraction coupling through modulation of ion channels. However, it is surprising that in a lot of epidemiological studies, if not all, no effects on electrocardiogram parameters were observed in agreement with such molecular effects as it is observed with drugs having comparable multi-ion channels effects.
Such contradictory and unspecific effects of ω3 PUFAs on ion membrane channels activity challenge the hypothesis that cardioprotective effects of ω3 PUFA could be due to the modulation of these channels or that in vivo, ω3 PUFAs have any effect on ion channels.
In parallel, some scientists were more interested in the regulation of intracellular Ca2+, which is known to play a role in physio-pathological situations and to be a key actor in cardiac muscle and skeletal excitation–contraction coupling.
Acute application of ω3 PUFAs leads to numerous effects on intracellular Ca2+ and on cellular mechanisms that ensue from it (Judé et al. 2006; Billman 2012). Besides the fact that ω3 PUFAs inhibit many voltage-dependent calcium channels activity, it has also been shown that they inhibit pro-arrhythmic diastolic depolarization due to intracellular calcium elevation and this can be explained by a reduction of NCX exchanger activity (Szentandrássy et al. 2007; Berecki et al. 2007; Den Ruijter et al. 2008; Sankaranarayanan and Venetucci 2012).
ω3 PUFAs have direct effects on mechanisms that regulate sarcoplasmic Ca2+ level by decreasing Ca2+ released by the sarcoplasmic reticulum. Mechanistically, a decreased frequency of the occurrence of spontaneous calcium events (sparks) in isolated ventricular cardiomyocytes has been observed (Negretti et al. 2000; O’Neill et al. 2002; Honen et al. 2003; Sankaranarayanan and Venetucci 2012). In agreement with this observation, ω3 PUFAs reduce the opening probability of ryanodine receptor type 2 (RyR2), responsible for these calcium events (Swan et al. 2003). In 1993, Taffet observed that a ω3 PUFAs-enriched diet is associated with increased DHA incorporation in the cardiac sarcoplasmic reticulum membrane (Taffet et al. 1993). This effect is associated with a reduction of sarcoplasmic reticulum content and a decreased SERCA2a pump activity by dietary interventions that change the composition, and possibly the structure of phospholipid membranes thereby affecting enzyme turnover. However, these observations are in contradiction with other studies (Leifert et al. 2000; Billman et al. 2012). It must be noted that the acute application of ω3 PUFAs induces an augmentation of the sarcoplasmic reticulum calcium content (Negretti et al. 2000; O’Neill et al. 2002; Swan et al. 2003). Also, delayed after-depolarization and early after-depolarization are both prevented by an acute or chronic exposition to ω3 PUFAs (Den Ruijter et al. 2008; Milberg et al. 2011).
In conclusion, ω3 PUFAs can regulate intracellular Ca2+ homeostasis, decreasing the risk of arrhythmias, in particular when these originate from a change of intracellular Ca2+ homeostasis such as during an ischemia/reperfusion episode (Billman 1991).
Dietary supplementation with ω3 PUFAs leads to their incorporation in cardiac membrane phospholipids. This incorporation in mitochondria, sarcoplasmic reticulum and cell membranes plays an essential role in the regulation of excitation–contraction coupling. Thus, several studies have investigated the effects of this incorporation. One important point is that after their incorporation, these lipids can undergo enzymatic and non-enzymatic oxidation, leading to new products derived from ω3 PUFAs. The effects of enzymatic metabolites such as prostaglandins or leukotrienes have been well studied (Calder 2010). However, it is important to note that in presence of a lipid antioxidant, alpha-tocopherol, the beneficial effects of ω3 PUFAs are totally abolished (such as anti-arrhythmic properties) whereas in presence of a pro-oxidant (low concentrations of hydrogen peroxide) enhancing non-enzymatic oxidation, the beneficial effects of these lipids are potentiated (Roy et al. 2015). This might be linked with a similar observation about the effects of DHA on rat ion currents (Judé et al. 2003). Recently, we focused our attention on certain non-enzymatic metabolites of ω3 PUFAs (NEO-PUFAs). We found that some NEO-PUFAs, namely the 4(RS)-4-F4t-neuroprostanes and the 10(S)-10-F4t-neuroprostane, can normalize RyR2 function and thus reduce the occurrence of abnormal extrasystoles (Roy et al. 2015). These results, and others, suggest that the beneficial effects of ω3 PUFAs can be obtained in conditions of oxidative stress (characteristic of many chronic pathologies).
Also, these discoveries open new perspectives for non-enzymatic oxygenated metabolites of ω3 PUFAs as potent healthy mediators in diseases associated with an oxidative stress (Burton et al. 1990; Janero et al. 1991; Sano 2010; Anderson and Taylor 2012; Anderson et al. 2012).
This element of oxidation must be taken into account, because it differs from one individual to another. The intensity of oxidative stress and antioxidant capacity are highly inter-individually different, thus even for a comparable level of PUFAs, the production of NEO-PUFAs will be variable.
Conclusion
To conclude, all these contradictory data leads the scientific community to consider effects and cellular targets of ω3 PUFAs (acutely or chronically applied).
Recently, it has been found that low ω3 PUFAs blood level and dietary intake can potentially increase the risk of non beneficial outcomes and could explain the difference and the contradiction in the effects observed (Stark et al. 2016).
Many factors can explain the contradictory observations made in cells, animal and human studies as described in this review.
Therefore, it is important and essential to take into consideration these parameters in future studies that, otherwise, could generate other contradictory results. Notably, given the challenges of fatty acid analysis and reporting, an international initiative should be considered to lead to standardized approaches and methods before all studies. To limit the conflicting findings, a standardization of study method (taking into account dose and type of fatty acids, rate of oxidation) should be proposed such as the ω3 PUFAs index proposed by Von Schacky (von Schacky 2010).
References
Ander BP, Hurtado C, Raposo CS et al (2007) Differential sensitivities of the NCX1.1 and NCX1.3 isoforms of the Na+ –Ca2+ exchanger to alpha-linolenic acid. Cardiovasc Res 73:395–403. doi:10.1016/j.cardiores.2006.09.013
Anderson EJ, Taylor DA (2012) Stressing the heart of the matter: re-thinking the mechanisms underlying therapeutic effects of n-3 polyunsaturated fatty acids. F1000 Med Rep 4:13. doi:10.3410/M4-13
Anderson EJ, Katunga LA, Willis MS (2012) Mitochondria as a source and target of lipid peroxidation products in healthy and diseased heart. Clin Exp Pharmacol Physiol 39:179–193. doi:10.1111/j.1440-1681.2011.05641.x
Arterburn LM, Hall EB, Oken H (2006) Distribution, interconversion, and dose response of n-3 fatty acids in humans. Am J Clin Nutr 83:1467S–1476S
Ascherio A, Rimm EB, Stampfer MJ et al (1995) Dietary intake of marine n-3 fatty acids, fish intake, and the risk of coronary disease among men. N Engl J Med 332:977–982. doi:10.1056/NEJM199504133321501
Bang HO, Dyerberg J, Hjøorne N (1976) The composition of food consumed by Greenland Eskimos. Acta Med Scand 200:69–73
Berecki G, Den Ruijter HM, Verkerk AO et al (2007) Dietary fish oil reduces the incidence of triggered arrhythmias in pig ventricular myocytes. Heart Rhythm 4:1452–1460. doi:10.1016/j.hrthm.2007.07.015
Billman GE (1991) The antiarrhythmic and antifibrillatory effects of calcium antagonists. J Cardiovasc Pharmacol 18(Suppl 10):S107–S117
Billman GE (2012) Omega-3 polyunsaturated fatty acids and cardiac rhythm: an introduction. Front Physiol 3:457. doi:10.3389/fphys.2012.00457
Billman GE (2013) The effects of omega-3 polyunsaturated fatty acids on cardiac rhythm: a critical reassessment. Pharmacol Ther 140:53–80. doi:10.1016/j.pharmthera.2013.05.011
Billman GE, Carnes CA, Adamson PB et al (2012) Dietary omega-3 fatty acids and susceptibility to ventricular fibrillation: lack of protection and a proarrhythmic effect. Circ Arrhythm Electrophysiol 5:553–560. doi:10.1161/CIRCEP.111.966739
Bougnoux P, Hajjaji N, Ferrasson MN et al (2009) Improving outcome of chemotherapy of metastatic breast cancer by docosahexaenoic acid: a phase II trial. Br J Cancer 101:1978–1985. doi:10.1038/sj.bjc.6605441
Burr ML, Fehily AM, Gilbert JF et al (1989) Effects of changes in fat, fish, and fibre intakes on death and myocardial reinfarction: diet and reinfarction trial (DART). Lancet 2:757–761
Burton KP, Morris AC, Massey KD et al (1990) Free radicals alter ionic calcium levels and membrane phospholipids in cultured rat ventricular myocytes. J Mol Cell Cardiol 22:1035–1047
Calder PC (2010) Omega-3 fatty acids and inflammatory processes. Nutrients 2:355–374. doi:10.3390/nu2030355
De Caterina R (2011) n-3 fatty acids in cardiovascular disease. N Engl J Med 364:2439–2450. doi:10.1056/NEJMra1008153
de Lorgeril M, Salen P, Defaye P, Rabaeus M (2013) Recent findings on the health effects of omega-3 fatty acids and statins, and their interactions: do statins inhibit omega-3? BMC Med 11:5. doi:10.1186/1741-7015-11-5
Den Ruijter HM, Berecki G, Verkerk AO et al (2008) Acute administration of fish oil inhibits triggered activity in isolated myocytes from rabbits and patients with heart failure. Circulation 117:536–544. doi:10.1161/CIRCULATIONAHA.107.733329
Doolan GK, Panchal RG, Fonnes EL et al (2002) Fatty acid augmentation of the cardiac slowly activating delayed rectifier current (IKs) is conferred by hminK. FASEB J 16:1662–1664. doi:10.1096/fj.02-0084fje
Durrington PN, Bhatnagar D, Mackness MI et al (2001) An omega-3 polyunsaturated fatty acid concentrate administered for one year decreased triglycerides in simvastatin treated patients with coronary heart disease and persisting hypertriglyceridaemia. Heart Br Card Soc 85:544–548
Else PL, Wu BJ (1999) What role for membranes in determining the higher sodium pump molecular activity of mammals compared to ectotherms? J Comp Physiol B 169:296–302
Eussen SRBM, Geleijnse JM, Giltay EJ et al (2012) Effects of n-3 fatty acids on major cardiovascular events in statin users and non-users with a history of myocardial infarction. Eur Heart J 33:1582–1588. doi:10.1093/eurheartj/ehr499
Ferrier GR, Redondo I, Zhu J, Murphy MG (2002) Differential effects of docosahexaenoic acid on contractions and L-type Ca2+ current in adult cardiac myocytes. Cardiovasc Res 54:601–610
Gillum RF, Mussolino M, Madans JH (2000) The relation between fish consumption, death from all causes, and incidence of coronary heart disease. The NHANES I epidemiologic follow-up study. J Clin Epidemiol 53:237–244
Guallar E, Aro A, Jiménez FJ et al (1999) Omega-3 fatty acids in adipose tissue and risk of myocardial infarction: the EURAMIC study. Arterioscler Thromb Vasc Biol 19:1111–1118
Guizy M, Arias C, David M et al (2005) {Omega}-3 and {omega}-6 polyunsaturated fatty acids block HERG channels. Am J Physiol Cell Physiol 289:C1251–C1260. doi:10.1152/ajpcell.00036.2005
Hazama H, Nakajima T, Asano M et al (1998) Omega-3 polyunsaturated fatty acids–modulation of voltage-dependent L-type Ca2+ current in guinea-pig tracheal smooth muscle cells. Eur J Pharmacol 355:257–266
Hites RA, Foran JA, Carpenter DO et al (2004) Global assessment of organic contaminants in farmed salmon. Science 303:226–229. doi:10.1126/science.1091447
Honen BN, Saint DA, Laver DR (2003) Suppression of calcium sparks in rat ventricular myocytes and direct inhibition of sheep cardiac RyR channels by EPA, DHA and oleic acid. J Membr Biol 196:95–103. doi:10.1007/s00232-003-0628-9
Honoré E, Barhanin J, Attali B et al (1994) External blockade of the major cardiac delayed-rectifier K+ channel (Kv1.5) by polyunsaturated fatty acids. Proc Natl Acad Sci USA 91:1937–1941
Hu XF, Laird BD, Chan HM (2016) Mercury diminishes the cardiovascular protective effect of omega-3 polyunsaturated fatty acids in the modern diet of Inuit in Canada. Environ Res. doi:10.1016/j.envres.2016.06.001
Hulbert AJ (2003) Life, death and membrane bilayers. J Exp Biol 206:2303–2311
Iso H, Kobayashi M, Ishihara J et al (2006) Intake of fish and n3 fatty acids and risk of coronary heart disease among Japanese: the Japan public health center-based (JPHC) study cohort I. Circulation 113:195–202. doi:10.1161/CIRCULATIONAHA.105.581355
Janero DR, Hreniuk D, Sharif HM (1991) Hydrogen peroxide-induced oxidative stress to the mammalian heart-muscle cell (cardiomyocyte): lethal peroxidative membrane injury. J Cell Physiol 149:347–364. doi:10.1002/jcp.1041490302
Judé S, Bedut S, Roger S et al (2003) Peroxidation of docosahexaenoic acid is responsible for its effects on ITO and ISS in rat ventricular myocytes. Br J Pharmacol 139:816–822. doi:10.1038/sj.bjp.0705308
Judé S, Roger S, Martel E et al (2006) Dietary long-chain omega-3 fatty acids of marine origin: a comparison of their protective effects on coronary heart disease and breast cancers. Prog Biophys Mol Biol 90:299–325. doi:10.1016/j.pbiomolbio.2005.05.006
Kang JX, Leaf A (1996) Evidence that free polyunsaturated fatty acids modify Na+ channels by directly binding to the channel proteins. Proc Natl Acad Sci USA 93:3542–3546
Kim S-H, Kim M-K, Lee H-Y et al (2011) Prospective randomized comparison between omega-3 fatty acid supplements plus simvastatin versus simvastatin alone in Korean patients with mixed dyslipidemia: lipoprotein profiles and heart rate variability. Eur J Clin Nutr 65:110–116. doi:10.1038/ejcn.2010.195
Kromhout D, Bloemberg BP, Feskens EJ et al (1996) Alcohol, fish, fibre and antioxidant vitamins intake do not explain population differences in coronary heart disease mortality. Int J Epidemiol 25:753–759
Kromhout D, Geleijnse JM, de Goede J et al (2011) n-3 fatty acids, ventricular arrhythmia-related events, and fatal myocardial infarction in postmyocardial infarction patients with diabetes. Diabetes Care 34:2515–2520. doi:10.2337/dc11-0896
Leifert WR, Jahangiri A, Saint DA, McMurchie EJ (2000) Effects of dietary n-3 fatty acids on contractility, Na+ and K+ currents in a rat cardiomyocyte model of arrhythmia. J Nutr Biochem 11:382–392
Li G-R, Sun H-Y, Zhang X-H et al (2009) Omega-3 polyunsaturated fatty acids inhibit transient outward and ultra-rapid delayed rectifier K+ currents and Na+ current in human atrial myocytes. Cardiovasc Res 81:286–293. doi:10.1093/cvr/cvn322
Macleod JC, Macknight AD, Rodrigo GC (1998) The electrical and mechanical response of adult guinea pig and rat ventricular myocytes to omega3 polyunsaturated fatty acids. Eur J Pharmacol 356:261–270
Milberg P, Frommeyer G, Kleideiter A et al (2011) Antiarrhythmic effects of free polyunsaturated fatty acids in an experimental model of LQT2 and LQT3 due to suppression of early afterdepolarizations and reduction of spatial and temporal dispersion of repolarization. Heart Rhythm 8:1492–1500. doi:10.1016/j.hrthm.2011.03.058
Mozaffarian D, Lemaitre RN, Kuller LH et al (2003) Cardiac benefits of fish consumption may depend on the type of fish meal consumed: the cardiovascular health study. Circulation 107:1372–1377
Negretti N, Perez MR, Walker D, O’Neill SC (2000) Inhibition of sarcoplasmic reticulum function by polyunsaturated fatty acids in intact, isolated myocytes from rat ventricular muscle. J Physiol 523(Pt 2):367–375
O’Neill SC, Perez MR, Hammond KE et al (2002) Direct and indirect modulation of rat cardiac sarcoplasmic reticulum function by n-3 polyunsaturated fatty acids. J Physiol 538:179–184
Oomen CM, Feskens EJ, Räsänen L et al (2000) Fish consumption and coronary heart disease mortality in Finland, Italy, and The Netherlands. Am J Epidemiol 151:999–1006
Pietinen P, Ascherio A, Korhonen P et al (1997) Intake of fatty acids and risk of coronary heart disease in a cohort of Finnish men. The alpha-tocopherol, beta-carotene cancer prevention study. Am J Epidemiol 145:876–887
Rissanen T, Voutilainen S, Nyyssönen K et al (2000) Fish oil-derived fatty acids, docosahexaenoic acid and docosapentaenoic acid, and the risk of acute coronary events: the Kuopio ischaemic heart disease risk factor study. Circulation 102:2677–2679
Rodrigo GC, Dhanapala S, Macknight AD (1999) Effects of eicosapentaenoic acid on the contraction of intact, and spontaneous contraction of chemically permeabilized mammalian ventricular myocytes. J Mol Cell Cardiol 31:733–743. doi:10.1006/jmcc.1998.0914
Roy J, Oger C, Thireau J et al (2015) Non-enzymatic lipid mediators, neuroprostanes, exert the anti-arrhythmic properties of docosahexaenoic acid. Free Radic Biol Med. doi:10.1016/j.freeradbiomed.2015.04.014
Salonen JT, Seppänen K, Nyyssönen K et al (1995) Intake of mercury from fish, lipid peroxidation, and the risk of myocardial infarction and coronary, cardiovascular, and any death in eastern Finnish men. Circulation 91:645–655
Sankaranarayanan R, Venetucci L (2012) Are the anti-arrhythmic effects of omega-3 fatty acids due to modulation of myocardial calcium handling? Front Physiol 3:373. doi:10.3389/fphys.2012.00373
Sano M (2010) Cardioprotection by hormetic responses to aldehyde. Circ J 74:1787–1793
Saravanan P, Bridgewater B, West AL et al (2010) Omega-3 fatty acid supplementation does not reduce risk of atrial fibrillation after coronary artery bypass surgery: a randomized, double-blind, placebo-controlled clinical trial. Circ Arrhythm Electrophysiol 3:46–53. doi:10.1161/CIRCEP.109.899633
Schrepf R, Limmert T, Claus Weber P et al (2004) Immediate effects of n-3 fatty acid infusion on the induction of sustained ventricular tachycardia. Lancet 363:1441–1442. doi:10.1016/S0140-6736(04)16105-9
Schuchardt JP, Schneider I, Meyer H et al (2011) Incorporation of EPA and DHA into plasma phospholipids in response to different omega-3 fatty acid formulations–a comparative bioavailability study of fish oil versus krill oil. Lipids Health Dis 10:145. doi:10.1186/1476-511X-10-145
Siscovick DS, Raghunathan TE, King I et al (1995) Dietary intake and cell membrane levels of long-chain n-3 polyunsaturated fatty acids and the risk of primary cardiac arrest. JAMA 274:1363–1367
Stark KD, Van Elswyk ME, Higgins MR et al (2016) Global survey of the omega-3 fatty acids, docosahexaenoic acid and eicosapentaenoic acid in the blood stream of healthy adults. Prog Lipid Res 63:132–152. doi:10.1016/j.plipres.2016.05.001
Stillwell W, Wassall SR (2003) Docosahexaenoic acid: membrane properties of a unique fatty acid. Chem Phys Lipids 126:1–27
Stillwell W, Jenski LJ, Crump FT, Ehringer W (1997) Effect of docosahexaenoic acid on mouse mitochondrial membrane properties. Lipids 32:497–506
Swan JS, Dibb K, Negretti N et al (2003) Effects of eicosapentaenoic acid on cardiac SR Ca(2+)-release and ryanodine receptor function. Cardiovasc Res 60:337–346. doi:10.1016/S0008-6363(03)00545-5
Szentandrássy N, Pérez-Bido MR, Alonzo E et al (2007) Protein kinase A is activated by the n-3 polyunsaturated fatty acid eicosapentaenoic acid in rat ventricular muscle. J Physiol 582:349–358. doi:10.1113/jphysiol.2007.132753
Taffet GE, Pham TT, Bick DL et al (1993) The calcium uptake of the rat heart sarcoplasmic reticulum is altered by dietary lipid. J Membr Biol 131:35–42
Trial Investigators ORIGIN, Bosch J, Gerstein HC et al (2012) n-3 fatty acids and cardiovascular outcomes in patients with dysglycemia. N Engl J Med 367:309–318. doi:10.1056/NEJMoa1203859
Turner N, Else PL, Hulbert AJ (2003) Docosahexaenoic acid (DHA) content of membranes determines molecular activity of the sodium pump: implications for disease states and metabolism. Naturwissenschaften 90:521–523. doi:10.1007/s00114-003-0470-z
Verkerk AO, van Ginneken ACG, Berecki G et al (2006) Incorporated sarcolemmal fish oil fatty acids shorten pig ventricular action potentials. Cardiovasc Res 70:509–520. doi:10.1016/j.cardiores.2006.02.022
von Schacky C (2010) Omega-3 index and sudden cardiac death. Nutrients 2:375–388. doi:10.3390/nu2030375
Wilhelm M, Tobias R, Asskali F et al (2008) Red blood cell omega-3 fatty acids and the risk of ventricular arrhythmias in patients with heart failure. Am Heart J 155:971–977. doi:10.1016/j.ahj.2007.11.045
Wu BJ, Hulbert AJ, Storlien LH, Else PL (2004) Membrane lipids and sodium pumps of cattle and crocodiles: an experimental test of the membrane pacemaker theory of metabolism. Am J Physiol Regul Integr Comp Physiol 287:R633–R641. doi:10.1152/ajpregu.00549.2003
Xiao YF, Kang JX, Morgan JP, Leaf A (1995) Blocking effects of polyunsaturated fatty acids on Na+ channels of neonatal rat ventricular myocytes. Proc Natl Acad Sci USA 92:11000–11004
Xiao YF, Gomez AM, Morgan JP et al (1997) Suppression of voltage-gated L-type Ca2+ currents by polyunsaturated fatty acids in adult and neonatal rat ventricular myocytes. Proc Natl Acad Sci USA 94:4182–4187
Xiao Y-F, Ke Q, Chen Y et al (2004) Inhibitory effect of n-3 fish oil fatty acids on cardiac Na+/Ca2+ exchange currents in HEK293t cells. Biochem Biophys Res Commun 321:116–123. doi:10.1016/j.bbrc.2004.06.114
Yamagishi K, Iso H, Date C et al (2008) Fish, omega-3 polyunsaturated fatty acids, and mortality from cardiovascular diseases in a nationwide community-based cohort of Japanese men and women the JACC (Japan collaborative cohort study for evaluation of cancer risk) study. J Am Coll Cardiol 52:988–996. doi:10.1016/j.jacc.2008.06.018
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Roy, J., Le Guennec, JY. Cardioprotective effects of omega 3 fatty acids: origin of the variability. J Muscle Res Cell Motil 38, 25–30 (2017). https://doi.org/10.1007/s10974-016-9459-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10974-016-9459-z