Abstract
We used HPLC to identify and quantify pigments in a Holocene sediment record from large, shallow Lake Peipsi, Estonia. The aim of our study was to track the influence of long-term climate change (i.e. temperature fluctuations) on past dynamics of aquatic primary producers. Sedimentary pigments were separated and quantified in 182 samples that span the last ca. 10,000 years. There was an increasing trend in sedimentary pigment concentrations from basal to upper sediment layers, suggesting a gradual increase in lake trophic status through time. Using additive models, our results suggested that primary producer dynamics in Lake Peipsi were closely related to temperature fluctuations. We, however, identified two periods (early Holocene and after ca. 2.5 cal ka BP) when the relationship between primary producer composition and temperature was weak, suggesting the influence of additional drivers on the primary producer community. We postulate that: (a) the increase of primary producer biomass in the early Holocene could have been caused by input of allochthonous organic matter and nutrients from the flooded areas when water level in Lake Peipsi was increasing, and (b) changes in the abundance and structure of primary producer assemblages since ca. 2.5 cal ka BP was related to widespread agricultural activities in the Lake Peipsi catchment. These results suggest that human activities can disrupt the relationship between the primary producer community and temperature in large, shallow lakes.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Global warming and intensification of human activities are expected to have major impacts on inland waters (Adrian et al. 2009). Among other consequences, rising water temperature and anthropogenic nutrient enrichment will enhance primary production and lead to such problems as harmful algal blooms (Havens and Paerl 2015). Because of the limited timescale of limnological monitoring data, it is difficult to assess the extent of global change impacts on lakes. Fortunately, lacustrine sediment archives can provide insights into past ecological conditions in lakes and enable evaluation of the responses of water bodies to global change (Smol 1992).
Pelagic and benthic algae are often the main food resource at the base of aquatic food webs, making them a dominant control on the whole-lake ecosystem (Vadeboncoeur et al. 2002; Jeppesen et al. 2014). Short-lived primary producers respond to a wide range of environmental variables (Anderson et al. 1996), including nutrient concentrations and water temperature (Fietz et al. 2005; Tõnno et al. 2013). Moreover, primary producers contain photosynthetic pigments that are archived in lake sediments even after cells senesce and lose their morphological structure (Cohen 2003). Therefore, sedimentary pigments are a useful proxy for exploring the past dynamics of both planktonic and benthic primary producers (Leavitt and Hodgson 2001). Temporal dynamics of sedimentary pigment composition have been associated with an immediate response to nutrient supply and/or to past climate variability (Bennett et al. 2001; Fietz et al. 2005; Tõnno et al. 2013; Deshpande et al. 2014).
Large, shallow lakes are important at the regional scale, as they offer multiple ecosystem services to human society (e.g. potable water, fisheries, recreation, etc.). Because of intensive anthropogenic activities, however, large, shallow lakes are subjected to multiple pressures, such as inputs of excessive nutrients and toxic pollutants (Nõges et al. 2008a). In addition, shallow lakes are generally more vulnerable to climate change than deeper, thermally stratified lakes, because temperatures in shallow waterbodies change rapidly in response to shifts in air temperature because of their large area/volume ratio (Arvola et al. 2010). A better understanding of the driving factors of long-term primary producer dynamics in large, shallow lakes is needed to improve predictions concerning their responses to global changes.
Lake Peipsi, the fourth largest lake in Europe, is located on the border of Estonia and Russia, in the north temperate zone of Europe (Fig. 1). Monitoring of Lake Peipsi phytoplankton composition began in the early twentieth century and revealed the dominance of diatoms and cyanobacteria. Other phytoplankton groups, including Chlorophyta, Cryptophyta and Dinophyta, were also represented, but their abundances were less important (Laugaste et al. 1996). Paleolimnological investigations, however, revealed that past algal composition of Lake Peipsi was probably dominated by benthic algae, i.e. diatoms (Hang et al. 2008; Punning et al. 2008; Heinsalu et al. 2007). Nowadays, primary production in Lake Peipsi is dominated by phytoplankton, while contributions from vascular plants, which occupy 1.7% of the area in the northern basin of the lake, and microphytobenthos, are thought to be negligible (Haberman et al. 2008). Although Lake Peipsi is currently well-studied, historical conditions in the lake are poorly known. A few paleolimnological studies have documented the lake’s eutrophication during the twentieth century, a likely consequence of agricultural expansion, massive use of mineral fertilizers, and ineffective wastewater treatment (Leeben et al. 2008). Other studies revealed that water-level fluctuations at the end of the early Holocene caused dramatic changes in the diatom community composition (Davydova 1985; Davydova and Kimmel 1991; Punning et al. 2008), and analyses of sediment geochemical composition indicated the dominance of autochthonous organic matter in the lake sediment in the late Holocene (Leeben et al. 2010; Makarõtševa et al. 2010; Belle et al. 2018). The influence of Holocene climate change on the development of Lake Peipsi, however, has received little attention.
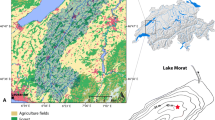
Location map of Lake Peipsi, modified from Hang et al. (2001). Distribution of bottom deposits on the Estonian side is shown. The white circle marks the sediment coring station
This study was undertaken to investigate the influence of Holocene temperature fluctuations on the primary producer community composition in Lake Peipsi. We inferred changes in the primary producer community at high-temporal resolution using sedimentary pigment analysis. We used additive models to quantify the influence of climate change, i.e. temperature fluctuations, on primary producer dynamics. We hypothesized that temperature was an important driver of the primary producer community composition in Lake Peipsi. We also hypothesized that increasing agricultural activities during the recent period were an important driver of primary producer assemblages.
Study site
Lake Peipsi (57°51′–59°01′N, 26°57′–28°10′E, 30 m a.s.l.) is a shallow, non-stratified water body with mean and maximum depths of 7 m and 15 m, respectively. The lake surface area is 3555 km2, with a maximum length of 150 km and width that varies between 23 and 42 km. Lake Peipsi has three basins: (1) Peipsi proper (surface area 2611 km2, mean depth 8.3 m) in the north, and (2) Lake Pihkva (surface area 708 km2, mean depth 3.8 m) in the south, which are connected by narrow, river-like (3) Lake Lämmijärv (surface area 236 km2, mean depth 2.5 m). The present study utilized a sediment core from the largest basin, Peipsi proper (Fig. 1). The Lake Peipsi catchment is large (~ 47,800 km2) and flat (Miidel et al. 2001), and possesses ~ 240 tributaries. Water level of Lake Peipsi is not regulated and average annual water level fluctuation is about 1.15 m (Haberman et al. 2008).
The Peipsi proper basin is considered to be moderately eutrophic, with average total phosphorus and total nitrogen concentrations of 40 and 658 mg m−3, respectively, during the primary producer growing season. Mean Secchi disk depth during the growth period in the Peipsi proper basin (hereafter Lake Peipsi) is about 1.7 m, pH is 8.3 and mean Chlorophyll a content is 17.5 mg m−3 (Kangur and Möls 2008; Haberman et al. 2008).
Materials and methods
Coring, chronology and sedimentological analysis
A 4.3-m-long sediment core was retrieved from the ice in March 2007 from the widest part (58°47″13′N; 27°19″18′E) of Lake Peipsi, using a 10-cm-diameter, Russian-type peat corer. Topmost unconsolidated sediments were sampled using a Willner-type gravity corer. Sediment cores were transported to the laboratory in the dark, sliced into contiguous 1-cm-thick sub-samples and stored at − 24 °C until further analysis.
Radiocarbon dates, including 5 conventional and 4 accelerator mass spectrometry (AMS) analyses, were used to establish a chronology for the deeper sediment section (Leeben et al. 2010). The upper section of the core was dated by correlation (loss-on-ignition, diatom composition and spheroidal fly-ash particles) with a core taken in 2002, which was dated by measurements of 210Pb, 226Ra and 137Cs, and by spheroidal fly-ash particle distribution in the sediments (Heinsalu et al. 2007). Details of the chronology and sediment lithostratigraphy for the 2002 core are described in Heinsalu et al. (2007), and details about the deeper, 2007 core are described in Leeben et al. (2010). All radiocarbon dates were calibrated and ages are expressed in thousands of years before present (cal ka BP), where AD 1950 = 0 cal ka BP.
Sediments were analysed for water content, organic matter (OM) and carbonate matter (CM) content. Water content was assessed by drying the samples to constant weight at 105 °C. The OM content was estimated by weight loss-on-ignition (LOI) at 550 °C for 4 h and CM was determined from weight LOI between 550 and 950 °C for 2 h (Heiri et al. 2001). Results of LOI are expressed in terms of percentage of dry weight (hereafter % of DW).
We used the annual mean temperature reconstruction provided by Ilvonen et al. (2016) as an estimate of past climate variability in the area (Fig. 2). This temperature reconstruction is a pollen-based inference and consisted of a modern pollen sample “training set” (e.g. modern climate variables and associated pollen compositional data) and corresponding fossil pollen records from lake sediments of the Baltic region (Estonia and South Sweden; Ilvonen et al. 2016).
Sedimentary pigment extraction and chromatographic analysis
Sedimentary pigment subsamples were analysed at 1-cm resolution. Altogether, 182 samples were analysed for sedimentary pigments, following the method of Leavitt and Hodgson (2001). Sediment samples were freeze-dried, and sedimentary pigments were extracted in the dark over 24 h using a solution of acetone and methanol (80:20 v:v) at − 20 °C. Extracts were clarified before chromatographic analysis by filtration through 0.45-µm Millex-LCR hydrophilic PTFE membrane filters. Reversed-phase, high-performance liquid chromatography (HPLC) was applied, using a Shimadzu Prominence (Japan) series system with a photodiode-array (PDA) detector to separate the pigments.
The pigment identification method was slightly modified from Airs et al. (2001). Prior to injection, the ion-pairing reagent 0.5 M ammonium acetate was added in a volume ratio of 2:3 to each sample. To avoid chemical decomposition of pigments, the auto-sampler was cooled to + 5 °C (Reuss et al. 2005). The sample injection volume was 100 µL. Separations were performed in a reversed-phase mode using two Waters Spherisorb ODS2 3-µm columns (150 mm × 4.6 mm ID) in-line with a pre-column (10 mm × 5 mm ID) containing the same phase. A binary gradient elution method (Table 1) was used with isocratic holds between 0–2 and 30–43 min. The flow rate remained constant during the elution (0.8 mL min−1). Absorbance was detected at wavelengths from 350 to 700 nm. The software “LC solution 1.22” (Shimadzu) was applied to collect and analyse the pigment data. Peak areas were integrated at each pigment absorbance maximum (Roy et al. 2011). Commercially available external standards from DHI Water and Environment (Denmark) were used for peak identification and quantification. The concentrations of studied sedimentary pigments were expressed as nanomoles per gram sediment organic matter (nmol g OM−1).
Chlorophyll a (Chl a), it’s derivative pheophytin a (Phe a) and β, β-carotene (β-car) were selected as indicators of total algal abundance and primary production (Leavitt and Hodgson 2001; Waters et al. 2013). Diatoms were represented by fucoxanthin (Fuco), diadinoxanthin (Diadino) and diatoxanthin (Diato). Fuco, however, can also be the dominant pigment in chrysophytes and Diadino can also represent the dinophyte group (Roy et al. 2011). In the xanthophyll cycle, Diadino can be transformed to Diato under excessive light conditions (Roy et al. 2011), therefore these two marker pigments are presented together (Diadino + Diato). Zeaxanthin (Zea) was chosen to represent colonial cyanobacteria (Bianchi et al. 2002), whereas canthaxanthin (Cantha) and echinenone (Echin) were selected to indicate abundance of colonial and filamentous cyanobacteria and N2-fixing filamentous cyanobacteria, respectively (Waters et al. 2013; Deshpande et al. 2014). Zea can also be a dominant pigment in chrysophytes and some dinophytes, but is a minor pigment in green algae (Roy et al. 2011). Finally, lutein (Lut) and chlorophyll b (Chl b) were selected to track green algae dynamics, whereas alloxanthin (Allo) was chosen to track Cryptophyta, keeping in mind that Lut and Chl b are also pigments in higher plants (Waters et al. 2013). According to Tamm et al. (2015), in nearby large, shallow Lake Võrtsjärv, marker pigments Zea and Fuco display significant correlations with cyanobacteria and diatoms, respectively. The Chl a/Phe a ratio was used to assess pigment preservation in the lake sediments, and should remain stable under steady-state preservation conditions (Ady and Patoine 2016).
Data analysis
Zones, defined by major patterns in sedimentary pigment composition, were determined by constrained hierarchical cluster analysis, using a Bray–Curtis distance and the CONISS linkage method, and significance of the zones was assessed using the broken-stick model (Bennett 1996). Detrended correspondence analysis (DCA) was then performed on the individual pigment data to assess the compositional turn-over and to select the appropriate ordination method (Legendre and Birks 2012). The first axis length of the DCA was short (< 2.5 SD), suggesting a low compositional turn-over, and a linear approach (Euclidean distance) was then used in the following part of the study. First, a principal component analysis (PCA) was performed on the sedimentary pigment data (excluding Fuco), and the PCA axis significance was assessed using the broken-stick model. PCA scores were considered as indicators of sedimentary pigment temporal variability. Statistical relationships between the explanatory variable (annual mean air temperature) and response variables (PCA1 and PCA2 scores) were examined using generalized additive models (GAM; fitted using the mgcv package for R), with a continuous-time, first-order autoregressive process to account for potential temporal autocorrelation (Simpson and Anderson 2009). Temporal trends in GAMs residuals were also checked to reveal possible contributions of untargeted drivers to primary producer dynamics. We expected to observe an increase in GAM residuals during periods when temperature fluctuation explained less pigment variability. For graphical displays, temporal trends were smoothed using additive models. All statistical tests and graphical displays were performed using R 3.4.0 statistical software (R Core Team 2017).
Results
Sediment geochemistry
Gyttja accumulation atop glacio-lacustrine clays at our coring site began ca. 10.4 cal ka BP (Leeben et al. 2010). The OM content in sediments followed a gradual increase, from 10% to 27%, from the basal part of the core until ca. 7.5 cal ka BP (Fig. 2), then remained stable until ca. 2.5 cal ka BP (~ 23%), followed by a small increase to 26%. The CM values remained nearly stable until ca. 4.0 cal ka BP (~ 4%), followed by a small decrease to the top of the sediment core, where it reached its minimum of ~ 2% (Fig. 2).
Sedimentary pigment dynamics
No temporal trend in Chl a/Phe a ratio was observed in the early Holocene sediments, whereas from 7.5 to 0.06 cal ka BP the Chl a/Phe a ratio decreased steadily (Fig. 3). In the topmost sediments, since ca. 0.06 cal ka BP, the ratio increased ~ 2.5-fold (Fig. 3). Sedimentary pigments fluctuated in sediment samples older than ca. 7.4 cal ka BP (Fig. 3). Concentrations of marker pigments Fuco, Diadino + Diato and Allo increased gradually in the period 7.5–2.5 cal ka BP, while Zea remained low and concentration of Lut and Chl b remained relatively stable over time (Fig. 3). Contents of cyanobacterial marker pigments Cantha and Echin were high from 7.5 to 4.5 cal ka BP, with a decreasing trend thereafter. All measured sedimentary pigments except Echin and Diadino + Diato started to increase after ca. 2.5 cal ka BP. Finally, steep increases (1–8×) in all marker pigments except Echin were detectable in the upper sediment core since ca. 0.06 cal ka BP, i.e. during the twentieth century (Fig. 3).
a Temporal dynamics of sedimentary pigments in Lake Peipsi. Individual pigments are expressed in terms of nanomoles per gram sediment organic matter (nmol g OM−1). Dendrogram is based on the pigment data, and constructed by hierarchical clustering analysis (Bray–Curtis distance, CONISS linkage method). Horizontal black lines indicate zones (from A to F) defined by major patterns in sedimentary pigment composition. b Without zone F. Chlorophyll a—Chl a; Pheophytin a—Phe a; β,β-Carotene—β-car; Fucoxanthin—Fuco; Diadinoxanthin + Diatoxanthin—D + D; Zeaxanthin—Zea; Canthaxanthin—Cantha; Echinenone—Echin; Lutein—Lut; Chlorophyll b—Chl b; Alloxanthin—Allo
Cluster analysis (Bray–Curtis distance, CONISS linkage method) on individual pigment data revealed a large number of significant zones (> 10). We decided to consider only the 6 major zones, according to their overall patterns of sedimentary pigment composition (Fig. 3a). To avoid spurious correlations, very high sedimentary pigment values in the topmost sediments (cluster F, 8 samples; Fig. 3a), from the twentieth century, the period of intensive anthropogenic activity, were omitted from the statistical analysis, as they deviated substantially from the average values and could thus dominate the analysis. Furthermore, microbial activity, and thus degradation of settled material (including pigments) in upper sediment layers is intense compared with such processes in deeper sediment layers (Wetzel 2001). In upper sediment layers, where degradation processes are still in progress, sedimentary pigment abundances are much higher than in the rest of the sediment core, but not because of greater production, and could therefore lead to misinterpretation of the statistical analysis.
Additive models
The first two axes of the PCA applied to the sedimentary pigment data accounted for 73.7% of their variability. The first axis of the PCA (PCA1) represented 59.9% of the overall variability, whereas the second axis (PCA2) accounted for 13.8% (Fig. 4a, b). PCA1 evenly explained Zea, Chl a, and β-car, and negative PCA1 values represented pigment-rich sediment layers. PCA2 was mainly explained by Cantha, with substantial contributions of Echin and Allo (Fig. 4b). PCA1 scores followed a gradual decrease over time, corresponding to a gradual switch from positive to negative values (Fig. 4a, c) whereas PCA2 scores showed a more contrasting temporal trend (Fig. 4c). PCA2 scores followed a steep rise from 10 to 7.5 cal ka BP, with a switch from negative to positive values. Then, PCA2 scores remained nearly stable until ca. 5.0 cal ka BP, and a decrease followed, from 5.0 to 3.0 cal ka BP, after which values remained relatively stable until the uppermost sample (Fig. 4c).
a Factorial map of the principal component analysis (PCA) performed on the individual sedimentary pigment data (PCA2 versus PCA1). A grey-scale was used to identify the sample age: light-grey colors correspond to the oldest samples, whereas black symbols represent the youngest samples. b Correlation circle representing the sedimentary pigment contributions to the first two axes of the PCA. For sedimentary pigment abbreviations, see Fig. 3 legend. c Temporal trends in first and second axis of the PCA scores (PCA1 and PCA2)
A GAM approach, fitted with a continuous-time, first-order autoregressive (CAR[1]) process, was applied to the PCA scores to study the response of the primary producer community to long-term changes in temperature. The final model, built on PCA1 values (GAM1; Table 2), showed a non-linear relationship with annual mean air temperature (F = 16.43; edf = 5.31), and explained 39.9% of the PCA1 variability (Fig. 5). Final PCA2 GAM (GAM2) revealed a non-linear relationship with annual mean air temperature (F = 45.08; edf = 6.47), and explained 67.7% of the PCA2 variability (Table 2).
Fitted smooth function between explanatory variables and PCA1 and PCA2 scores from general additive models (GAM1 and GAM2) fitted with a continuous-time, first-order autoregressive process to account for temporal autocorrelation. Grey surface marks the 95% uncertainty interval of the fitted function. On the x-axis, black ticks show the distribution of observed values for variables. Numbers in brackets on the y-axis are the effective degrees of freedom (edf) of the smooth function. Relationships between predicted and observed PCA scores plotted for Axis1 and Axis2 (Pearson’s correlation test; p value < 0.05)
Fitted relationships between PCA1 scores and annual mean air temperature were non-linear (Fig. 5). PCA2 showed a monotonic and positive relationship with temperature fluctuations, whereas PCA1 scores were not monotonically related to them. A PCA1 fitted function showed a negative relationship with temperature at values colder than 5 °C.
Assessment of GAM residuals revealed two temporal patterns. GAM1 residuals (based on PCA1 scores) increased slightly during the early Holocene, and decreased during the modern period, from ca. 2.3 cal ka BP (Fig. 6), suggesting influences of untargeted forcing factors. In contrast, GAM2 residuals (built on PCA2 scores) did not reveal any specific temporal trend (Fig. 6), and remained relatively stable throughout the record.
Discussion
Organic matter, climate fluctuations and changes in water level
We hypothesize that Lake Peipsi began to develop between ca. 12.4 and 11.7 cal ka BP, with isolation from the Baltic Ice Lake caused by tectonic uplift in northeast Estonia (Rosentau et al. 2009). This was the low-water-level period in the lake`s history, with stage about 10 m lower than today, caused by heterogenous glacio-isostatic uplift in different areas (Rosentau et al. 2009). At the coring site in this study, this low-water period is marked by a sedimentation hiatus. Thereafter, in the early Holocene, the water level increased gradually (Rosentau et al. 2009; Punning et al. 2008) and by ca. 10.4 cal ka BP the lake area had increased, enabling gyttja accumulation at our coring site.
Sediment composition in Lake Peipsi changed remarkably in the early Holocene, with a continuous rise in OM content, from ~ 5 to ~ 25% of DW. The gradual increase in OM content between ca. 10.4 and 7.6 cal ka BP has been attributed to progressive deepening of the water body (Leeben et al. 2010; Makarõtševa et al. 2010), with present water level probably reached ca. 7.6 cal ka BP (Makarõtševa et al. 2010). A possible consequence of the ~ 10 m rise in water level was flooding of vast areas around the lake and erosion of allochthonous organic matter and nutrients from the catchment.
In North Europe, changes in early Holocene climate were rather intense, starting with low temperatures at the beginning of the period, followed by gradual warming, interrupted periodically by short cooling periods (Antonsson and Seppä 2007). During the Holocene Thermal Maximum (HTM), the period from 8.0 to 4.0 cal ka BP, average temperatures in Northern Europe were approximately 2.5–3.5 °C higher than today (Antonsson and Seppä 2007; Heikkilä and Seppä 2010; Ilvonen et al. 2016). In the Baltic region, summer conditions in the HTM were probably warm and dry, linked with strong blocking of anticyclonic atmospheric conditions over North Europe (Heikkilä and Seppä 2010). In the late Holocene, ca. 4.5 cal ka BP, temperature decreased gradually until it reached the present value (Ilvonen et al. 2016). After stable conditions in the middle Holocene, the increase in sedimentary OM in Lake Peipsi ca. 2.5 cal ka BP indicates a rise in the lake’s trophic state, probably initiated by early agricultural activity in Estonia (Poska et al. 2004; Leeben et al. 2010).
Long-term changes in sedimentary pigments
We used sedimentary pigment analysis of the Lake Peipsi sediment core to identify the main temporal patterns of Holocene primary producer dynamics in the lake. Two zones can be identified in early Holocene sediments, using the content of sedimentary pigments. In zone A, the beginning of the early Holocene, contents of sedimentary pigments were low, indicating modest in-lake production. Thereafter, in zone B, together with rising water level, in-lake productivity also began to increase (Fig. 3). Greater erosion of allochthonous OM (Leeben et al. 2010) and nutrients from the catchment, together with higher temperatures, might have led to prolonged growth periods and higher rates of OM mineralization (Nõges et al. 2005; Havens and Paerl 2015), enhancing aquatic primary production in zone B (Fig. 3). Aquatic primary production in zones A and B could have been primarily benthic, as previously suggested by Punning et al. (2008). They demonstrated that benthic diatoms dominated in Lake Peipsi during the early Holocene, probably as a consequence of the low water depth. Given the low Chl a/Phe a ratio, preservation of sedimentary pigments at the beginning of the early Holocene, until ca. 9.5 cal ka BP, was evidently rather poor (Fig. 3). Flooding of the lake’s surroundings and consequent enhanced input of pigments of terrestrial origin likely occurred during this period. Before being buried in lake sediments, these terrestrial pigments were probably subject to substantial degradation. At the same time, lacustrine primary production of mainly benthic origin was quite low, thus there were fewer pigments deposited in the sediment. In the subsequent part of the early Holocene, after ca. 9.5 cal ka BP (zone B), the Chl a/Phe a ratio increased, indicating improved conditions for preservation of sedimentary pigments (Fig. 3). With increasing biomass of benthic primary producers, relatively more pigment could be buried quickly in the sediments, limiting its exposure to light and oxygen, which favour pigment degradation (Leavitt 1993; Cuddington and Leavitt 1999).
After this rapid initial phase, Leeben et al. (2010) suggested that Lake Peipsi development remained rather stable in the middle Holocene (7.5–2.5 cal ka BP), although some changes can be seen in the dynamics of the quantified sedimentary pigments used to identify zones C and D (Fig. 3). Chl a, β-car and Zea had negative contributions to PCA1, revealing the rise in trophic status since the middle Holocene (Figs. 4b, c). The rising concentration of the sedimentary marker pigment Allo, which is found only in planktonic cryptophytes, suggests an increase in aquatic primary production in the middle Holocene, especially in zone D (Lami et al. 2000; Fig. 3). Indeed, according to Punning et al. (2008), the abundance of planktonic relative to benthic diatoms in Lake Peipsi increased continuously, from 20 to 80%, since the middle Holocene, indicating a rise in aquatic ecosystem trophic status. In this study, pigments Fuco and D + D, which represent chrysophytes, dinophytes, and diatoms, also increased within this period, especially in zone D (Fig. 3). Therefore, we postulate that PCA1 could be interpreted as evidence of gradual, natural eutrophication of Lake Peipsi, a process influenced by multiple factors including paleoclimate, catchment processes, in-lake productivity and sediment infilling (Engstrom and Fritz 2006; Fritz and Anderson 2013).
Cyanobacterial marker pigments Echin and Cantha display a positive relation with temperature and a positive contribution to the PCA2 axis (Fig. 4b). Cyanobacteria benefit from warmer water because of their higher optimum growth temperatures (Kosten et al. 2012), and experienced favourable environmental conditions during the HTM (Fig. 3). Besides, higher water temperature during the HTM likely induced in-lake nitrogen limitation and low N/P ratio as a consequence of intense phosphorus recycling from the lake sediments (Nõges et al. 2008b; Elliott 2012), as suggested by diatom analysis (Punning et al. 2008). Cyanobacteria, many of which are N-fixers, have a competitive advantage over other algal groups in lakes with low N/P ratio, as they compete better for nitrogen than other algae groups (Huisman and Hulot 2005). Thus, it is likely that conditions for cyanobacteria proliferation in Lake Peipsi during the HTM were favourable, as the lake’s trophic state had increased naturally (Punning et al. 2008) and climate was warmer than today (Ilvonen et al. 2016).
The Chl a/Phe a ratio decreased steadily from the middle Holocene onward, indicating deteriorating conditions for preservation of sedimentary pigments (Fig. 3). The increased number of planktonic primary producers, resulting from higher water level, probably also led to accelerated pigment degradation. In the water column, carotenoids and chlorophylls break down rapidly under conditions of high light and oxygen, prior to permanent burial in the sediments (Leavitt 1993). We suggest that unfavourable pigment preservation conditions could account for the modest increase in Chl a and β-car in the middle Holocene, including the HTM. All the potential drivers of primary producer community composition may account for the changes seen in the Lake Peipsi pigment record, and the general rise in trophic state that occurred during the period 7.5–4.5 cal ka BP. Previous studies also reported an increase in lacustrine primary production during the HTM (Hammarlund et al. 2003).
Climate change and human impact
Nutrient supply and water temperature are two of the main regulating factors for primary producers in aquatic ecosystems (Scheffer 1998). An increase in OM in lake sediments usually accompanies eutrophication (Dean 2006). Generally, epilimnetic water temperatures are well correlated with regional air temperatures and exhibit a direct response to climate forcing (Adrian et al. 2009).
GAM, applied to the sedimentary pigment data, revealed that primary producer dynamics in Lake Peipsi are strongly related to temperature fluctuations, matching well with the interpretations of the ecological requirements of primary producers that generate the sediment pigments (Figs. 4, 5). Specific temporal trends in GAM1 residuals, however, indicate potential untargeted driving factors of primary producer community development during the early Holocene (Fig. 6). We suggest that, in addition to increasing temperature, input of allochthonous OM (Leeben et al. 2010) to the lake caused by the rise in water level, also increased the biomass of primary producers in the early stage of lake development. The primary producer community was probably nutrient-limited, but added inputs and mineralization of terrestrial OM enhanced in-lake nutrient concentrations and fueled the rise in primary production during this period. After the water level stabilized ca. 7.6 cal ka BP, rising temperature affected the primary producer community in Lake Peipsi. Therefore, the rise in trophic status and change in the primary producer community in the middle Holocene can be explained by a combination of climate change and ontogeny of Lake Peipsi, i.e. natural nutrient enrichment (Anderson et al. 2008).
Our analysis also revealed another, recent driver of primary producer community change during the late Holocene, corresponding to zone E. The temporal trend in GAM1 residuals decreased from ca. 2.3 cal ka BP, revealing that the pigment variability detected by our model, built with temperature as a unique variable, is low. This recent change in lake functioning coincided well with the intensification of agricultural activities in Estonia (Poska et al. 2004) (Fig. 6). Extensive agricultural activity required widespread forest clearance (Poska et al. 2004; Niinemets and Saarse 2009), which led to nutrient leaching into the lake, a process that probably favoured in-lake productivity (Tõnno et al. 2013). This is confirmed by previous studies that showed that autochthonous OM predominated in Lake Peipsi sediments from ca. 2.5 cal ka BP onwards (Leeben et al. 2010; Makarõtševa et al. 2010). Kisand et al. (2017) showed that the content of sedimentary organic P increased remarkably since ca. 2.3 cal ka BP in Lake Peipsi, linked to the rise in lake productivity (Reitzel et al. 2012).
Finally, the considerable increase in all paleopigments except Echin, in the uppermost part of the sediment core (zone F), could be related to the anthropogenic eutrophication of Lake Peipsi during the twentieth century, with clear acceleration in the 1950s, and could have been caused by massive use of mineral fertilizers and rapid expansion of urban areas (Heinsalu et al. 2007; Kangur and Möls 2008; Leeben et al. 2008). Also Taranu et al. (2015) reported recent widespread anthropogenic eutrophication of Northern Hemisphere lakes, leading to the dominance of potentially toxic cyanobacteria.
Conclusions
This study provided insights into the influence of long-term, Holocene climate fluctuations on changes in the trophic state and development of the primary producer community in large, shallow European Lake Peipsi. An increase in sedimentary pigment concentrations from basal to upper sediment layers suggests natural nutrient enrichment and a gradual increase in lake trophic status. GAM analysis of the pigment data indicates that temperature was an important driver of primary producer dynamics in the lake. Two periods, however, were identified, during which the relationship between primary producer composition and temperature was weaker, suggesting the influence of other potential drivers. In the early Holocene, from 10.4 to 7.5 cal ka BP, that additional driver was probably water level. At that time, water level in so-called “Small Peipsi” was increasing, and additional allochthonous OM and nutrients, transported from the flooded areas, contributed to greater primary producer biomass in the lake. The other anomalous period was after ca. 2.5 cal ka BP, when early human activity (agricultural practices) in the lake watershed probably changed the abundance and structure of primary producer assemblages. These results demonstrate the vulnerability of the lake ecosystem to human activities.
This study demonstrated that water temperature and nutrient supply were the main factors that regulated primary producer community abundance and composition in Lake Peipsi during the early and middle Holocene. That link was disrupted in the late Holocene when human activities became an additional important driver of primary producer dynamics in the lake.
References
Adrian R, O’Reilly CM, Zagarese H, Baines SB, Hessen DO, Keller W, Livingstone DM, Sommaruga R, Straile D, Van Donk E, Weyhenmeyer GA, Winder M (2009) Lakes as sentinels of climate change. Limnol Oceanogr 54:2283–2297
Ady FD, Patoine A (2016) Impacts of land use and climate variability on algal communities since ~ 1850 CE in an oligotrophic estuary in northeastern New Brunswick, Canada. J Paleolimnol 55:151–165
Airs RL, Atkinson JE, Keely BJ (2001) Development and application of a high resolution liquid chromatographic method for the analysis of complex pigment distributions. J Chromatogr A 917:167–177
Anderson NJ, Odgaard BV, Segerström U, Renberg I (1996) Climate-lake interactions recorded in varved sediments from a Swedish boreal forest lake. Glob Change Biol 2:399–405
Anderson NJ, Brodersen KP, Ryves DB, McGowan S, Johansson LS, Jeppesen E, Leng MJ (2008) Climate versus in-lake processes as controls on the development of community structure in a low-arctic lake (South-West Greenland). Ecosystems 11:307–324
Antonsson K, Seppä H (2007) Holocene temperatures in Bohuslän, southwest Sweden: a quantitative reconstruction from fossil pollen data. Boreas 36:400–410
Arvola L, George G, Livingstone DM, Järvinen M, Blenckner T, Dokulil MT, Jennings E, Nic Aonghusa C, Nõges P, Nõges T, Weyhenmeyer GA (2010) The impact of the changing climate on the thermal characteristics of lakes. In: George G (ed) The impact of climate change on European Lakes, 1st edn. Springer, Berlin, pp 85–102
Belle S, Freiberg R, Poska A, Agasild H, Alliksaar T, Tõnno I (2018) Contrasting responses to long-term climate change of carbon flows to benthic consumers in two different sized lakes in the Baltic area. Quat Sci Rev 187:168–176
Bennett KD (1996) Determination of the number of zones in a biostratigraphical sequence. New Phytol 132:155–170
Bennett JR, Cumming BF, Leavitt PR, Chiu M, Smol JP, Szeicz J (2001) Diatom, pollen, and chemical evidence of postglacial climatic change at Big Lake, South-Central British Columbia, Canada. Quat Res 55:332–343
Bianchi TS, Rolff C, Widbom B, Elmgren R (2002) Phytoplankton pigments in Baltic Sea seston and sediments: seasonal variability, fluxes, and transformations. Estuar Coast Shelf Sci 55:369–383
Cohen AS (2003) Paleolimnology: the history and evolution of lake systems. Oxford University Press, New York
Cuddington K, Leavitt PR (1999) An individual-based model of pigment flux in lakes: implications for organic biogeochemistry and paleoecology. Can J Fish Aquat Sci 56:1964–1977
Davydova N (1985) Diatoms—indicators of natural conditions of water reservoir in Holocene. Nauka, Leningrad (in Russian)
Davydova N, Kimmel K (1991) Palaeogeography of Lake Peipsi on the basis of biostratigraphical studies of bottom sediments. Proc Est Acad Sci Geol 40:16–23 (in Russian)
Dean WE (2006) Characterization of organic matter in lake sediments from Minnesota and Yellowstone National Park. U.S. Geological Survey Open-File Report 2006-1053. U.S Geological Survey, Reston, Virginia
Deshpande BN, Tremblay R, Pienitz R, Vincent WF (2014) Sedimentary pigments as indicators of cyanobacterial dynamics in a hypereutrophic lake. J Paleolimnol 52:171–184. https://doi.org/10.1007/s10933-014-9785-3
Elliott JA (2012) Is the future blue–green? A review of the current model predictions of how climate change could affect pelagic freshwater cyanobacteria. Water Res 46:1346–1371
Engstrom DR, Fritz SC (2006) Coupling between primary terrestrial succession and the trophic development of lakes at Glacier Bay, Alaska. J Paleolimnol 35:873–880
Fietz S, Nicklisch A, Oberhänsli H (2005) Phytoplankton response to climate changes in Lake Baikal during the Holocene and Kazantsevo Interglacials assessed from sedimentary pigments. J Paleolimnol 37:177–203. https://doi.org/10.1007/s10933-006-9012-y
Fritz SC, Anderson NJ (2013) The relative influences of climate and catchment processes on Holocene lake development in glaciated regions. J Paleolimnol 49:349–362
Haberman J, Timm T, Raukas A (2008) Peipsi. Eesti Loodusfoto, Tartu (in Estonian)
Hammarlund D, Björck S, Buchardt B, Israelson C, Thomsen CT (2003) Rapid hydrological changes during the Holocene revealed by stable isotope records of lacustrine carbonates from Lake Igelsjön, southern Sweden. Quat Sci Rev 22:353–370
Hang T, Miidel A, Kalm V, Kimmel K (2001) New data on the distribution and stratigraphy of the bottom deposits of Lake Peipsi, Eastern Estonia. Proc Est Acad Sci Geol 50:233–253
Hang T, Kalm V, Kihno K, Milkevičius M (2008) Pollen, diatom and plant macrofossil assemblages indicate a low water level phase of Lake Peipsi at the beginning of the Holocene. Hydrobiologia 599:13–21
Havens KE, Paerl HW (2015) Climate change at a crossroad for control of harmful algal blooms. Environ Sci Technol 49:12605–12606. https://doi.org/10.1021/acs.est.5b03990
Heikkilä M, Seppä H (2010) Holocene climate dynamics in Latvia, eastern Baltic region: a pollen-based summer temperature reconstruction and regional comparison. Boreas 39:705–719
Heinsalu A, Alliksaar T, Leeben A, Nõges T (2007) Sediment diatom assemblages and composition of pore-water dissolved organic matter reflect recent eutrophication history of Lake Peipsi (Estonia/Russia). Hydrobiologia 584:133–143
Heiri O, Lotter AF, Lemcke G (2001) Loss on ignition as a method for estimating organic and carbonate content in sediments: reproducibility and comparability of results. J Paleolimnol 25:101–110
Huisman J, Hulot F (2005) Population dynamics of harmful cyanobacteria. Factors affecting species composition. In: Huisman J, Matthijs HCP, Visser PM (eds) Harmful cyanobacteria. Springer, Dordrecht, pp 143–176
Ilvonen L, Holmström L, Seppä H, Veski S (2016) A Bayesian multinomial regression model for palaeoclimate reconstruction with time uncertainty. Environmetrics 27:409–422. https://doi.org/10.1002/env.2393
Jeppesen E, Meerhoff M, Davidson TA, Trolle D, Sondergaard M, Lauridsen TL, Beklioglu M, Brucet S, Volta P, Gonzalez-Bergonzoni I, Nielsen A (2014) Climate change impacts on lakes: an integrated ecological perspective based on a multi-faceted approach, with special focus on shallow lakes. J Limnol 73:84–107
Kangur K, Möls T (2008) Changes in spatial distribution of phosphorus and nitrogen in the large north-temperate lowland Lake Peipsi (Estonia/Russia). Hydrobiologia 599:31–39
Kisand A, Kirsi A-L, Ehapalu K, Alliksaar T, Heinsalu A, Tõnno I, Leeben A, Nõges P (2017) Development of large shallow Lake Peipsi (North-Eastern Europe) over the Holocene based on the stratigraphy of phosphorus fractions. J Paleolimnol 58:43–56. https://doi.org/10.1007/s10933-017-9954-2
Kosten S, Huszar VLM, Bécares E, Costa LS, van Donk E, Hansson LA, Jeppesen E, Kruk C, Lacerot G, Mazzeo N, Meester LD, Moss B, Lürling M, Nõges T, Romo S, Scheffer M (2012) Warmer climates boost cyanobacterial dominance in shallow lakes. Glob Change Biol 18:118–126
Lami A, Guilizzoni P, Marchetto A (2000) High resolution analysis of fossil pigments, carbon, nitrogen and sulphur in the sediment of eight European Alpine lakes: the MOLAR project. In: Lami A, Cameron N, Korhola A (eds) Paleolimnology and ecosystem dynamics at remote European Alpine lakes. J Limnol 59:15–28
Laugaste R, Jastremskij VV, Ott I (1996) Phytoplankton of Lake Peipsi-Pihkva: species composition, biomass and seasonal dynamics. Hydrobiologia 338:49–62
Leavitt PR (1993) A review of factors that regulate carotenoid and chlorophyll deposition and fossil pigment abundance. J Paleolimnol 9:109–127
Leavitt PR, Hodgson DA (2001) Sedimentary pigments. In: Smol JP, Birks HJB, Last WM (eds) Tracking environmental change using lake sediments. Terrestrial, algal, and siliceous indicators, vol 3. Kluwer, Dordrecht, pp 295–325
Leeben A, Tõnno I, Freiberg R, Lepane V, Bonningues N, Makarõtševa N, Heinsalu A, Alliksaar T (2008) History of anthropogenically mediated eutrophication of Lake Peipsi as revealed by the stratigraphy of fossil pigments and molecular size fractions of pore-water dissolved organic matter. Hydrobiologia 599:49–58
Leeben A, Heinsalu A, Alliksaar T, Vassiljev J (2010) High-resolution spectroscopic study of pore-water dissolved organic matter in Holocene sediments of Lake Peipsi (Estonia/Russia). Hydrobiologia 646:21–31
Legendre P, Birks HJB (2012) From classical to canonical ordination. In: Birks HJB, Lotter AF, Juggins S, Smol JP (eds) Tracking environmental change using lake sediments. Developments in paleoenvironmental research. Springer Netherlands, Amsterdam, pp 201–248
Makarõtševa N, Lepane V, Alliksaar T, Heinsalu A (2010) A 10,000 year record of sediment pore-water dissolved organic matter characteristics from Lake Peipsi as revealed by HPSEC. Chem Ecol 26:13–24
Miidel A, Noormets R, Hang T, Flodén T, Bjerkéus M (2001) Bedrock geology and topography of the Lake Peipsi depression, eastern Estonia. GFF 123:15–22
Niinemets E, Saarse L (2009) Holocene vegetation and land-use dynamics of south-eastern Estonia. Quat Int 207:104–116
Nõges T, Järvet A, Laugaste R, Loigu E, Leisk Ü, Tõnno I, Nõges P (2005) Consequences of catchment processes and climate changes on the ecological status of large shallow temperate lakes. In: Ahalya N, Murty CR (eds) Aquatic ecosystems, conservation, restoration and management. Capital Publishing Company, New Delhi, pp 88–99
Nõges P, Kangur K, Nõges T, Reinart A, Simola H, Viljanen M (2008a) Highlights of large lake research and management in Europe. Hydrobiologia 599:259–276
Nõges T, Laugaste R, Nõges P, Tõnno I (2008b) Critical N:P ratio for cyanobacteria and N2-fixing species in large shallow temperate lakes Peipsi and Võrtsjärv, North-East Europe. Hydrobiologia 599:77–86
Poska A, Saarse L, Veski S (2004) Reflections of pre- and early-agrarian human impact in the pollen diagrams of Estonia. Palaeogeogr Palaeocl 209:37–50
Punning JM, Kapanen G, Hang T, Davydova N, Kangur M (2008) Changes in the water level of Lake Peipsi and their reflection in a sediment core. Hydrobiologia 599:97–104
R Core Team (2017) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Reitzel K, Ahlgren J, Rydin E, Egemose S, Turner BL, Hupfer M (2012) Diagenesis of settling seston: identity and transformations of organic phosphorus. J Environ Monit 14:1098–1106
Reuss N, Conley DJ, Bianchi TS (2005) Preservation conditions and the use of sediment pigments as a tool for recent ecological reconstruction in four Northern European estuaries. Mar Chem 95:283–302
Rosentau A, Vassiljev J, Hang T, Saarse L, Kalm V (2009) Development of the Baltic Ice Lake in the eastern Baltic. Quat Int 206:16–23
Roy S, Llewellyn CA, Egeland ES, Johnsen G (2011) Phytoplankton pigments. Characterization, chemotaxonomy and applications in oceanography. Cambridge University Press, Cambridge
Scheffer M (1998) Ecology of shallow lakes. Chapman & Hall, London
Simpson GL, Anderson NJ (2009) Deciphering the effect of climate change and separating the influence of confounding factors in sediment core records using additive models. Limnol Oceanogr 54:2529–2541
Smol JP (1992) Paleolimnology: an important tool for effective ecosystem management. J Aquat Ecosyst Health 1:49–58
Tamm M, Freiberg R, Tõnno I, Nõges P, Nõges T (2015) Pigment-based chemotaxonomy—a quick alternative to determine algal assemblages in large shallow eutrophic lake? PLoS ONE 10:e0122526. https://doi.org/10.1371/journal.pone.0122526
Taranu ZE, Gregory-Eaves I, Leavitt PR, Bunting L, Buchaca T, Catalan J, Domaizon I, Guilizzoni P, Lami A, McGowan S, Moorhouse H (2015) Acceleration of cyanobacterial dominance in north temperate-subarctic lakes during the Anthropocene. Ecol Lett 18:375–384
Tõnno I, Kirsi A-L, Freiberg R, Alliksaar T, Lepane V, Kõiv T, Kisand A, Heinsalu A (2013) Ecosystem changes in a large shallow Lake Võrtsjärv, Estonia—evidence from sediment organic matter and phosphorus fractions. Boreal Environ Res 18:195–208
Vadeboncoeur Y, Vander Zanden MJ, Lodge DM (2002) Putting the lake back together: reintegrating benthic pathways into lake food web models. Bioscience 52:44–54
Waters MN, Smoak JM, Saunders CJ (2013) Historic primary producer communities linked to water quality and hydrologic changes in the northern Everglades. J Paleolimnol 49:67–81. https://doi.org/10.1007/s10933-011-9569-y
Wetzel RG (2001) Limnology. Lake and river ecosystems. Academic Press, London
Acknowledgements
This research was supported by institutional research grants IUT21-2, IUT1-8 and PRG323 and Estonian Science Foundation Grants Nos. 6741, 7888 and 9102. We acknowledge colleagues S. Veski, A. Heinsalu and J. Vassiljev for sediment coring and establishing the chronology, and A. Leeben for participating in the early stage of manuscript development. We are grateful for the comments and corrections made by two anonymous reviewers of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Tõnno, I., Nauts, K., Belle, S. et al. Holocene shifts in the primary producer community of large, shallow European Lake Peipsi, inferred from sediment pigment analysis. J Paleolimnol 61, 403–417 (2019). https://doi.org/10.1007/s10933-019-00067-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10933-019-00067-3