Abstract
In this study, we investigated the preparation of chitin nanofiber (CNF)-reinforced cellulose films through stepwise regeneration procedures from the respective ion gels with ionic liquids. Self-assembled CNF dispersions were prepared by regeneration from the chitin ion gel with the ionic liquid, 1-allyl-3-methylimidazolium bromide, using methanol, followed by dilution with adjusted amounts of methanol. Cellulose ion gels with the ionic liquid, 1-butyl-3-methylimidazolium chloride, were then prepared, soaked in the CNF dispersions, and centrifuged to simultaneously occur regeneration of cellulose and compatibilization with the CNFs. Soxhlet extraction with methanol and subsequent drying of the resulting materials gave the CNF/cellulose composite films. The IR and SEM results of the films indicated the presence of CNFs not only on the surfaces of the films but also inside the films. Powder X-ray diffraction patterns showed the amorphous structure of the cellulose in the film. Tensile testing of the films suggested the reinforcing effect of the CNFs on the mechanical properties of the films.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Natural polysaccharides are widely distributed in nature, and they are vital materials for important in vivo functions in biological systems [1]. For example, cellulose and chitin act as structural materials in the cell walls of plants and in the exoskeletons of crustaceans, shellfish, and insects, respectively [2–5]. Cellulose is composed of β-(1 → 4)-linked glucose residues; chitin has a similar structure, but it is an aminopolysaccharide composed of N-acetylglucomsamine residues, where hydroxy groups at position 2 in cellulose are replaced by acetamido groups. Cellulose and chitin have become increasingly important because they are the most abundant organic substances on earth and they possess unique structures and properties that are much different from those of typical synthetic polymers [6, 7]. Although considerable effort is being made to extend the novel applications of cellulose and chitin in order to make efficient use of biomass resources, the lack of solubility of these structural polysaccharides in water and common organic solvents makes it difficult to improve their processability, fusibility, and functionality. The solubility problem is due to their stiff molecules and close chain packing via the numerous intra- and inter-molecular hydrogen bonds among the hydroxy groups. It is also due to acetamido groups in the repeating sugar residues.
Over the past decade, ionic liquids, which are low melting-point salts that form liquids at temperatures below the boiling point of water, have been identified as good solvents for polysaccharides such as cellulose and chitin [8–15] since Rogers et al. [16] reported that an ionic liquid, 1-butyl-3-methylimidazolium chloride (BMIMCl), dissolved cellulose in relatively high concentrations. In a previous study, we carried out the dissolution of chitin with an ionic liquid, 1-allyl-3-methylimidazolium bromide (AMIMBr), in concentrations up to ~5 wt% at elevated temperature [17]. Through the dissolution with ionic liquids, moreover, the material processing of polysaccharides such as gelation and film formation has also been performed [17–29], such as our studies on the production of a cellulose/BMIMCl ion gel [20] and a chitin/AMIMBr ion gel [17]. We further reported the fabrication of a binary gelling system from a mixture of an individually prepared cellulose solution with BMIMCl and a chitin solution with AMIMBr [30, 31]. The cellulose/chitin composite films were then obtained by the regeneration procedure from the binary gels [31–33].
In previous studies, a dispersion of self-assembled chitin nanofibers (CNFs) with ca. 20–60 nm in width and several hundred nanometers in length was facilely obtained by regeneration from a chitin ion gel with AMIMBr using methanol, followed by sonication [34, 35]. The filtration of the resulting CNF/methanol dispersion gave a film with a highly entangled nanofiber morphology. Because chitin has free amino groups due to partial N-deacetylation of the acetamido groups (generally several percent of the total number of repeating units), the CNFs fabricated from the chitin ion gel were employed as a reinforcing agent for an acidic cellulose derivative, that is, carboxymethyl cellulose (CMC), by electrostatic interaction to construct a robust composite film [36]. The reinforcing effect of the CNFs present on the surface of the composite film was confirmed by the enhanced mechanical properties under tensile mode.
In this paper, we report the fabrication of CNF-reinforced cellulose films by means of the regeneration technique from ion gels. First, self-assembled CNF dispersions in methanol were prepared by regeneration from chitin ion gels with AMIMBr according to our previously reported procedure [34]. Individually prepared cellulose ion gels with BMIMCl [20] were soaked in CNF dispersions of varying concentrations, followed by centrifugation, to simultaneously occur regeneration of cellulose and compatibilization with the CNFs, leading to the composite films. The morphology and mechanical properties of the resulting films were evaluated by scanning electron microscopic (SEM) analysis and tensile testing, respectively. In relation to the present study, composite materials of cellulose with chitin nanocrystals/nanowhiskers, which were obtained by the acid-hydrolysis method from a native chitin, were also fabricated by different approaches, e.g., mainly blending [37, 38]. The present approach provides a facile procedure for producing new bio-based chitin/cellulose composite materials. It therefore has potential for practical applications in biomedical and environmentally benign research fields in the future.
Experimental
Materials
Microcrystalline cellulose in a commercial reagent from Merck, USA (Avicel, No. 2331), was used. Chitin powder from crab shells was purchased from Wako Pure Chemical Industries, Ltd., Japan. The ionic liquids BMIMCl and AMIMBr were purchased from Sigma-Aldrich Co., USA, and prepared by reaction of 1-methylimidazole and 3-bromo-1-propene according to a method modified from that reported in the literature, respectively [39]. Other reagents were used as received.
Preparation of Self-Assembled CNF Dispersion
The preparation of a self-assembled CNF dispersion was carried out according to our previous study [34]. A mixture of chitin (0.120 g, 0.59 mmol) with AMIMBr (1.00 g, 4.92 mmol) was left standing at room temperature for 24 h and then subsequently heated with stirring at 100 °C for 24 h to obtain a chitin ion gel (10 wt%). The gel was then soaked in methanol (40 mL) at room temperature for 48 h, followed by sonication for 10 min to give a self-assembled CNF dispersion (3.0 mg/mL). The dispersion was diluted with methanol to prepare 2.34, 0.46, and 0.21 mg/mL CNF dispersions. For the analytical measurements, the 3.0 mg/mL dispersion was subjected to filtration to isolate the CNFs as a film.
Preparation of Cellulose Ion Gel with BMIMCl and Film
The preparation of a cellulose ion gel with BMIMCI was performed according to our previous study [20]. Cellulose (0.151 g, 0.93 mmol) was dissolved in BMIMCl (1.50 g, 8.59 mmol) by heating it at 100 °C for 6 h. After the solution was cooled to room temperature, it was cast onto a Petri dish and left standing at room temperature for 7 days to form an ion gel. The BMIMCl left over from the gelation was removed by washing the gel with methanol. To calculate the cellulose content in the gel, 0.285 g of the gel was soaked in methanol (10 mL) and further subjected to Soxhlet extraction with methanol (100 mL) for 16 h. The regenerated cellulose was sandwiched between two glass plates and dried under ambient conditions for 3 days and under reduced pressure at room temperature for 3 h to give a cellulose film (0.0325 g). On the basis of the weights of the ion gel and film, the cellulose content in the gel was calculated to be 11.4 wt%.
Preparation of Chitin Nanofiber-Reinforced Cellulose Film
A typical experimental procedure was carried out as follows. The cellulose ion gel (0.287 g, 3.3 cm × 1.3 cm, amount of cellulose: 32.7 mg) was soaked in 0.21 mg/mL of the CNF dispersion (10 mL, amount of chitin: 2.1 mg) and the mixture was subjected to centrifugation at 2000 rpm for 30 min twice. Then, the resulting composite material was subjected to Soxhlet extraction with methanol (100 mL) for 16 h. The regenerated film was sandwiched between two glass plates and dried under ambient conditions for 3 days and under reduced pressure at room temperature for 3 h to give a CNF-reinforced cellulose film.
Measurements
IR spectra were recorded on a PerkinElmer Spectrum Two spectrometer. SEM images were obtained using a Hitachi SU-70 electron microscope. X-ray diffraction (XRD) measurements were conducted at a scanning speed of 2θ = 0.2°/min using a Rigaku Geigerflex RADIIB diffractometer with Ni-filtered CuKα radiation (λ = 0.15418 nm). The stress–strain curves were measured using a tensile tester (Little Senster LSC-1/30, Tokyo Testing Machine Co.).
Results and Discussion
To prepare CNF-reinforced cellulose films by the stepwise regeneration procedures, first the chitin/AMIMBr and cellulose/BMIMCl ion gels were individually prepared according to the methods reported in our previous publications (Fig. 1) [20, 34]. As we reported, small amounts of BMIMCl were leached out during the gelation in the cellulose experiment, whereas the chitin/AMIMBr mixture was totally turned into a gel by heating. To calculate the cellulose content in the ion gel, therefore, BMIMCl was removed from the ion gel via regeneration and Soxhlet extraction using methanol, giving rise to a regenerated cellulose film. On the basis of the weights of the ion gel and film, the cellulose content in the ion gel was calculated to be 11.4 wt%. On the other hand, the chitin/AMIMBr ion gel was soaked in methanol, followed by sonication, to produce the self-assembled CNF dispersion (3.0 mg/mL) by the regeneration of chitin, which was further diluted with different amounts of methanol to provide dispersions with different CNF contents (2.34, 0.46, and 0.21 mg/mL). For compatibilization of the cellulose with the CNFs by means of the regeneration of cellulose, the cellulose/BMIMCl ion gels (ca. 0.3 g) were soaked in 10 mL of the resulting CNF dispersions with methanol (Fig. 1). The samples were then subjected to centrifugation to facilitate regeneration, and the regenerated materials were further subjected to Soxhlet extraction with methanol for the complete removal of BMIMCl, followed by drying to give the CNF/cellulose composite films (Fig. 1). The unit ratios of CNF to cellulose in the resulting films were estimated by their weight differences from the cellulose weight in the cellulose/BMIMCl ion gel. Consequently, it was found that the CNF/cellulose unit ratios increased with increasing CNF content in the dispersions as follows; 0.032, 0.067, and 0.086 from 0.21, 0.46, and 2.34 mg/mL CNF dispersions, respectively.
The presence of CNFs in the films was confirmed by the IR analysis. The IR pattern of the composite film shown in Fig. 2b is similar as that of the cellulose film (Fig. 2a), but it also shows characteristic carbonyl absorption at 1639 cm−1 due to the acetamido groups in the chitin structure, as detected in the CNF (Fig. 2c). The IR results clearly indicate that the cellulose films were compatibilized with the CNFs during the regeneration procedure from the cellulose ion gels using the CNF dispersions.
The surface and cross-sectional morphologies of the composite films on a nano-scale were evaluated by SEM measurements. The SEM images of the surfaces of all the films show the nanofiber morphologies. Part of the surface of the film obtained from the 0.21 mg/mL CNF dispersion is not covered by the CNFs (Fig. 3b), and thus its morphology is similar to that of the cellulose film (Fig. 3a), However, CNFs largely cover the surfaces of the films obtained from the 0.46 and 2.34 mg/mL CNF dispersions, as shown in Fig. 3c and d, respectively. Furthermore, Fig. 3d also shows that some of the CNFs merged, as indicated by the presence of larger amounts of fibers on the film surface. The SEM results are in good agreement with the CNF amounts determined by the weight calculations. The SEM images of the cross sections of the CNF-reinforced cellulose films show the tips of the nanofibers extending from the solid films (Fig. 4b–d), which is not observed for the cellulose film (Fig. 4a). These SEM images suggest that the CNFs were present not only on the surfaces but also inside the films. It is speculated from the SEM results that during the regeneration procedure by soaking the cellulose/BMIMCl ion gels in the CNF dispersions with methanol followed by centrifugation, the CNFs with methanol penetrated into the ion gels to produce the present composite films with good compatibility. In contrast, when the CNF/CMC composite films were prepared by electrostatic interaction between the two polysaccharides, the CNFs were present only on the surfaces of the films, as previously reported [36]. During the regeneration process in the present study, methanol was probably progressed to be miscible with BMIMCl through the dispersion/ion gel interface, gradually migrating inside the gel matrix. Accordingly, the CNFs could penetrate into the ion gel as miscibility progressed. Because the aggregates of the stiff CNFs created spaces in the cellulose matrix during regeneration, the SEM images of the cross sections (Fig. 4b–d) show the presence of vacancies around the CNFs in the composite films.
XRD measurements of the films were conducted to evaluate their crystalline structure. The XRD pattern of the CNF-reinforced cellulose film in Fig. 5c does not show obvious diffraction peaks for cellulose crystalline structure I, as detected in the XRD pattern of the cellulose powder (Fig. 5a). However, it does show a broad peak at approximately 21°, which is assignable to the amorphous structure of cellulose, as seen for the cellulose film (Fig. 5b). These data indicate that the cellulose crystalline structure was not reconstructed during the regeneration procedure from the cellulose/BMIMCl ion gel, as previously reported [28]. Furthermore, diffraction peaks resulting from an antiparallel α-chitin crystalline structure, as detected in the XRD pattern of the CNF film (Fig. 5d), are not detected in the XRD pattern of the composite film (Fig. 5c). This is because the amounts of CNFs in the films were too low to detect their diffraction peaks. Even the largest diffraction peak at 19.5° was certainly overlapped with the broad peak of the amorphous cellulose.
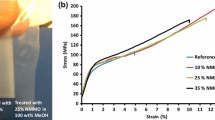
The mechanical properties of the composite films were then evaluated by tensile testing. The stress–strain curves of the cellulose film (Fig. 6a) and the composite films (Fig. 6b–d) indicate that the Young’s moduli of the composite films are comparable to that of the cellulose film. However, the tensile strength and elongation at break of the composite films increased as compared with those of the cellulose film, as listed in the table inserted in Fig. 6. Furthermore, the mechanical properties improve significantly with increasing amounts of CNFs in the composite films, as shown in Fig. 6b–d. These results strongly suggest the reinforcing effect of the CNFs in the composite films.
Conclusion
In this study, we investigated to prepare the CNF-reinforced cellulose films by means of the regeneration process from the cellulose/BMIMCl ion gels by soaking in the CNF dispersions with methanol, which were obtained by regeneration from the chitin/AMIMBr ion gels using methanol. The regenerated materials were further subjected to Soxhlet extraction with methanol, followed by drying to produce the composite films. The IR analysis supported the presence of chitin in the films and the SEM images suggested that the CNFs were present from the surface to the inside of the films. The amorphous structure of cellulose in the composite films was suggested by the XRD analysis. The reinforcing effect of CNFs present in the films was confirmed by tensile testing and the amounts of CNFs strongly affected enhancement of the mechanical property. The present study reveals that regeneration from the cellulose ion gels using the self-assembled CNF dispersions is the efficient approach to fabricate the CNF-reinforced materials with good compatibility. Because the ion gels with ionic liquids have been obtained from various natural polysaccharides [21–29], the present approach will be applicable to the other polysaccharides to produce new bio-based materials in the future.
References
Schuerch C (1986) Polysaccharides. In: Mark HF, Bilkales N, Overberger CG (eds) Encyclopedia of polymer science and engineering, vol 13, 2nd edn. Wiley, New York, pp 87–162
Klemm D, Heublein B, Fink H-P, Bohn A (2005) Angew Chem Int Ed 44:3358
Kurita K (2006) Mar Biotechnol 8:203
Rinaudo M (2006) Prog Polym Sci 31:603
Pillai CKS, Paul W, Sharma CP (2009) Prog Polym Sci 34:641
Klemm D, Kramer F, Moritz S, Lindström T, Ankerfors M, Gray D, Dorris A (2011) Angew Chem Int Ed 50:5438
Muzzarelli RAA (2012) Polym Sci Compr Ref 10:153
Liebert T, Heinze T (2008) BioResources 3:576
Feng L, Chen ZI (2008) J Mol Liq 142:1
Pinkert A, Marsh KN, Pang S, Staiger MP (2009) Chem Rev 109:6712
Zakrzewska ME, Lukasik EB, Lukasik RB (2010) Energy Fuels 24:737
Wang WT, Zhu J, Wang XL, Huang Y, Wang YZ (2010) J Macromol Sci Part B Phys 49:528
Muzzarelli RAA (2011) Marine Drugs 9:1510
Jaworska MM, Kozlecki T, Gorak A (2012) J Polym Eng 32:67
Bochek AM, Murav’ev AA, Novoselov NP, Zaborski M, Zabivalova NM, Petrova VA, Vlasova EN, Volchek BZ, Lavrent’ev VK (2012) Russ J Appl Chem 285:1718
Swatloski RP, Spear SK, Holbrey JD, Rogers RD (2002) J Am Chem Soc 124:4974
Prasad K, Murakami M, Kaneko Y, Takada A, Nakamura Y, Kadokawa J (2009) Int J Biol Macromol 45:221
Turner MB, Spear SK, Holbrey JD, Rogers RD (2004) Biomacromolecules 5:1379
Turner MB, Spear SK, Holbrey JD, Daly DT, Rogers RD (2005) Biomacromolecules 6:2497
Kadokawa J, Murakami M, Kaneko Y (2008) Carbohydr Res 348:769
Kadokawa J, Murakami M, Takegawa A, Kaneko Y (2009) Carbohydr Polym 75:180
Prasad K, Kaneko Y, Kadokawa J (2009) Macromol Biosci 9:376
Prasad K, Izawa H, Kaneko Y, Kadokawa J (2009) J Mater Chem 19:4088
Izawa H, Kaneko Y, Kadokawa J (2009) J Mater Chem 19:6969
Izawa H, Kadokawa J (2010) J Mater Chem 20:5235
Mine S, Prasad K, Izawa H, Sonoda K, Kadokawa J (2010) J Mater Chem 20:9220
Kadokawa J, Kato T, Setoyama M, Yamamoto K (2013) J Polym Environ 21:512
Setoyama M, Yamamoto K, Kadokawa J (2014) J Polym Environ 22:298
Kadokawa J (2015) RSC Adv 5:12736
Yamazaki S, Takegawa A, Kaneko Y, Kadokawa J, Yamagata M, Ishikawa M (2009) Electrochem Commun 11:68
Takegawa A, Murakami M, Kaneko Y, Kadokawa J (2010) Carbohydr Polym 79:85
Kadokawa J, Hirohama K, Mine S, Kato T, Yamamoto K (2012) J Polym Environ 20:37
Setoyama M, Kato T, Yamamoto K, Kadokawa J (2013) J Polym Environ 21:795
Kadokawa J, Takegawa A, Mine S, Prasad K (2011) Carbohydr Polym 84:1408
Tajiri R, Setoguchi T, Wakizono S, Yamamoto K, Kadokawa J (2013) J Biobased Mater Bioenergy 7:655
Hatanaka D, Yamamoto K, Kadokawa J (2014) Int J Biol Macromol 69:35
Huang Y, Zhang L, Yang J, Zhang X, Xu M (2013) Macromol Mater Eng 298:3030
Butchosa N, Brown C, Larsson PT, Berglund LA, Bulone V, Zhou Q (2013) Green Chem 15:3404
Zhao D, Fei Z, Geldbach TJ, Scopelliti R, Laurenczy G, Dyson PJ (2005) Helv Chim Acta 88:665
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kadokawa, Ji., Endo, R., Hatanaka, D. et al. Preparation of Chitin Nanofiber-Reinforced Cellulose Films Through Stepwise Regenerations from Individually Prepared Ion Gels. J Polym Environ 23, 348–355 (2015). https://doi.org/10.1007/s10924-015-0723-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10924-015-0723-x