Abstract
Hypotension induced by general anesthesia is associated with postoperative complications, increased mortality, and morbidity, particularly elderly patients. The aim of this study was to investigate the effectiveness of corrected carotid artery flow time (FTc) for predicting hypotension following anesthesia induction in patients over 65 years old. After faculty ethical committee approval and written informed consent, 138 patients (65 years and older, ASA physical status I–III) who scheduled for elective surgery were included in this study. In the pre-operative anesthesia unit, the carotid artery FTc value was measured by ultrasound and hemodynamic values were recorded. Following anesthesia induction with propofol, hemodynamic data were recorded at 1-minute intervals for 3 min. Measurements were terminated prior to endotracheal intubation, as direct laryngoscopy and endotracheal intubation could cause sympathetic stimulation and hemodynamic changes. Hypotension occurred in 52 patients (37.7%). The preoperative FTc value of the patients who developed hypotension was statistically lower (312.5 ms) than the patients who did not (345.0 ms) (p < 0.001). The area under the ROC curve for carotid artery FTc was 0.93 (95% CI for AUC:0.89–0.97; p < 0.001) with an optimal cut-off of value for predicting post-anesthesia hypotension 333 ms, a sensitivity of 90.4% and a specificity of 84.9%. As a result of the multiple logistic regression model, carotid artery FTc emerged as the sole independent risk factor for hypotension following anesthesia induction. Preoperative carotid artery FTc measurement is a simple, bedside, noninvasive, and reliable method for predicting anesthesia-induced hypotension in elderly patients.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
Hypotension following general anesthesia induction may be related to drugs, comorbidities, inadequate fluid intake, and a prolonged fasting period. It can lead to tissue hypoperfusion, organ failure, postoperative complications, and increased morbidity and mortality, especially in elderly patients [1, 2]. Therefore, early diagnosis and treatment are crucial in this population. Vasoactive drugs and fluid therapy are the most preferred agents to maintain blood pressure [3, 4].
Corrected carotid artery flow time (FTc) is primarily influenced by left ventricular preload and is associated with myocardial contractility and afterload [5]. Therefore, it may serve not only as an indicator of fluid responsiveness but also as an index reflecting the complexity of blood pressure [5, 6]. Recent studies have demonstrated that carotid artery FTc is an indicator that can be used to predict anesthesia-related hypotension [7, 8]. However, there is currently a gap in the existing literature, specifically in the context of studies related to elderly patients who have comorbidities and a high ASA score.
The aim of our study is to investigate the predictive value of preoperative carotid artery FTc for post-induction hypotension in patients with comorbidities over 65 years of age (ASA I–III) and to determine an appropriate cut-off value.
2 Methods
2.1 Study population
After obtaining written informed consent from the patients and receiving approval from the faculty ethical committee (date: Jan/06/2023, IRB number: 129/10), we enrolled 138 patients who met the inclusion criteria in this prospective and cross-sectional study. The study was registered on ClinicalTrials.gov. (NCT05896514).
Patients over 65 years of age who scheduled for elective surgery under general anesthesia were included in this study. Exclusion criteria were preoperative hypotension [mean arterial blood pressure (MAP) < 70 mmHg] and preoperative hypertension [systolic blood pressure (SBP) ≥ 180 mmHg or diastolic blood pressure (DBP) ≥ 110 mmHg], age < 65 years old, American Society of Anesthesiologists (ASA) physical status ≥ 4, carotid artery stenosis > 50%, unstable angina, left ventricular ejection fraction < 40%, cardiac rhythm other than sinus, serious vascular disease, implanted pacemaker, autonomic nervous system disorders, mild to severe valvular heart disease, emergency or urgent surgery.
2.2 Carotid artery FTc measurement
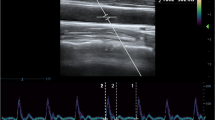
In the pre-operative anesthesia unit, all patients were monitored with 3-lead electrocardiography (ECG), noninvasive blood pressure, and pulse oximetry. The carotid artery FTc value of each patient was measured in B-mode using ultrasonography (Esaote, MyLab™ X6, Italy) 10 min before anesthesia induction.
To acquire measurements, patients were placed in a supine position with their heads turned 30 degrees to the left. Using a 4–12 MHz linear ultrasound probe directed towards the head, a long-axis B-mode image of the right common carotid artery was captured from the lower border of the thyroid cartilage. Subsequently, the probe was positioned approximately 2 cm proximal to the carotid bifurcation and at the center of the lumen. The angle correction cursor was placed parallel to the direction of blood flow. The insonation angle was maintained between 60 and 72 degrees for accurate blood flow velocity measurement. A pulsed wave Doppler image of the blood flow along the artery was then obtained, and recordings of carotid artery blood flow wave images were made. The cycle time was determined by measuring the interval between heartbeats from the onset of the systolic upstroke, utilizing the length measurement function on the ultrasound device. Flow time was calculated by measuring the duration between the systolic upstroke and the dicrotic notch in increments of tenths of a millisecond (ms). The FTc measurement was taken consecutively 3 times, and the mean of these was used for analysis. FTc values were calculated using Wodey’s formula, FTc = flow time + [1.29 × (heart rate − 60), to decrease the effect of heart rate, instead of Bazett’s formula ( \({\text{FTC = }}\frac{{{\text{Flow time}}}}{{\sqrt {{\text{RR interval}}} }}\) ) [9]. All carotid artery FTc measurements were analyzed by the same anesthesiologist who did not participate in the follow-up evaluations.
2.3 Anesthesia induction
Patients did not receive any premedication. Following the carotid artery FTc measurement, all patients were taken to the operating room, where standard monitoring, including 3-lead ECG, non-invasive blood pressure, and pulse oximetry, was conducted using an Infinity Kappa monitor (GE Medical Systems, Inc, Telford, PA).
Anesthesia induction was initiated with 2 mg/kg of propofol (20 mg in 10 s), and muscle relaxation was induced with 0.6 mg of rocuronium. Mask ventilation during induction utilized sevoflurane (2%) and a 50% oxygen-50% nitrous oxide mixture. Opioid agents were not administered during the anesthesia induction process.
2.4 Data collection
Ten minutes before induction of anesthesia (T0), SBP, DBP, MAP, and HR were recorded. Following anesthesia induction, SBP, DBP, MAP, and HR were measured at 1-minute intervals for 3 min (T1, one minute after anesthesia induction; T2, two minutes after anesthesia induction; T3, three minutes after anesthesia induction).
As direct laryngoscopy and endotracheal intubation could induce sympathetic stimulation, measurements were concluded 3 min after anesthesia induction. Hemodynamic values during and after endotracheal intubation were not included in the study data.
Post-induction hypotension was defined as a 30% decrease in SBP, a 20% decrease in MAP, or an absolute SBP less than 90 mmHg and MAP less than 65 mmHg within 3 min after anesthesia induction. Hypotension was addressed with intravenous norepinephrine administered in bolus doses of 4 µg, with repetition as necessary.
2.5 Sample size
The sample size was calculated assuming that carotid artery FTc has a diagnostic predictive validity of at least 0.70 in developing post-induction hypotension in elderly patients. In this calculation, the prevalence of hypotension was assumed to be 50%. We concluded that at least 112 patients were required to detect a difference of 0.20 units between the area under the ROC curve of carotid artery FTc (AUC = 0.70) and the null hypothesis (AUC = 0.50) with 95% power and 5% two-tailed type I error. Considering a possible 20% data loss (dropout rate or missing data), the study was planned to be conducted with 135 patients.
2.6 Statistical analysis
When summarizing the data, numbers and percentages were used for categorical measurements and mean ± standard deviation or median (Q1–Q3) was used for numerical measurements. Chi-square test was used to analyze categorical measurements. The Kolmogrov Smirnov test was used to test whether the numerical measurements met the assumption of normal distribution. In the comparison of numerical measurements between the groups, independent samples t test was used if the assumptions were met and Mann Whitney U test was used if the assumptions were not met. Repeated measures ANOVA analysis was used to compare the change of repeated numerical measurements over time between groups. Relationship between continuous variables was determined by Spearman correlation coefficient (r). Coefficient was taken as very weak between 0.0 and 0.02, weak 0.2–0.4, moderate 0.4–0.6, strong 0.6–0.8 and very strong 0.8–1.0. Multiple Logistic Regression analysis was used to determine the risk factors for the development of hypotension after induction of anesthesia. Variables that were significant in the univariate analysis were added to the model. Although SBP, DBP and MAP variables are significant, MAP variable is used in the model since these three variables cannot be included in the model at the same time due to the correlation between them. ROC curve analysis was performed to determine the diagnostic value of the FTc variable in determining the development of post-induction hypotension. Youden index was used to determine the optimum cut-off point after ROC curve analyis. Sensitivity, specificity and their confidence intervals were presented. IBM SPSS 20.0 package program was used for statistical analysis of the data. A value of p < 0.05 was considered significant in all tests.
3 Results
A total of 184 patients were evaluated, however 138 patients included in this study (Fig. 1).
The mean age of the patients who participated in the study was 72.5 ± 5.2 years and 50 of the patients were female, 88 of them were male. Seventy patients had hypertension, 42 had diabetes mellitus, 25 had coronary artery disease (CAD), and 23 had asthma-chronic obstructive pulmonary disease (COPD). Hypotension occurred in 52 cases (37.7%) following anesthesia induction. Sex, age, ASA, comorbidities, fasting time, propofol, and FTc levels were compared between the groups with and without hypotension. In the group that developed hypotension, the age, the proportion of ASA physical status III patients, the proportion of patients with a history of hypertension, and fasting time were higher than those who did not develop hypotension, while the FTc level was lower (Table 1).
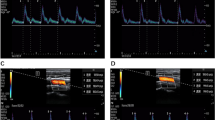
The follow-up results of vital parameters in the study groups with and without post-induction hypotension are presented in Fig. 2. According to the results of repeated measures analysis, the time-dependent trends (time*group interaction) of HR levels were similar in the groups (Fig. 2d), whereas the time-dependent changes of SBP, DBP, and MAP levels were significantly different in the groups (Fig. 2a, b and c).
The relationship between carotid artery FTc and drop in MAP and SBP in patients with hypotension after induction of anesthesia was presented in Table 2. There was a strong negative correlation between carotid artery FTc and percentage change (Δ%) from baseline SBP at T1 and T2 (r = − 0.63, p < 0.001; r = − 0.61, p < 0.001, respectively). Similarly, the percentage changes at T3 had moderate negative correlation between carotid artery FTc (r = − 0.59, p < 0.001). There was also strong negative correlation between FTc and percentage change (Δ%) from baseline MAP at T1, T2, and T3 (r = − 0.64, p < 0.001; r = − 0.60, p < 0.001; r = − 0.61, p < 0.001, respectively). At lower carotid artery FTc levels, percentage change in MAP and SBP was greater.
According to SBP criteria (SBP less than 90 mmHg or 30% decrease in SBP), median FTc value in hypotension group (317.5 ms) was significantly lower than in non-hypotension group (345.5 ms) (p < 0.001) (Fig. 3a). According to MAP criteria (MAP less than 65 mmHg), median FTc value in hypotension group (317.5 ms) was significantly lower than in non-hypotension group (345.5 ms) (p < 0.001) (Fig. 3b).
ROC curve analysis was performed to test the predictive value of carotid artery FTc measurement for post-induction hypotension and diagnostic accuracy measures were obtained (Fig. 4). The area under the ROC curve for carotid artery FTc was 0.93 (95% CI for AUC:0.89–0.97; p < 0.001) with an optimal cut-off of 333 ms, a sensitivity of 90.4% (95% CI: 78.9–96.8) and a specificity of 84.9% (95% CI: 75.5–91.7).
The outcome of the multiple logistic regression model, designed to identify risk factors influencing the occurrence of hypotension after anesthesia induction in elderly patients, incorporated variables such as age, ASA III score, history of hypertension, fasting time, mean arterial pressure (MAP), and carotid artery flow time (FTc), all of which were found to be significant in individual analyses. The analysis revealed that the sole independent determinant was carotid artery FTc (Odds Ratio [OR] = 0.88; 95% Confidence Interval [CI]: 0.84–0.92; p < 0.001) (refer to Table 3). In elderly patients, each ms decrease in carotid artery FTc was associated with a 13% increase in the risk of post-induction hypotension (OR: 1/0.88 = 1.13).
4 Discussion
The data we gathered highlights the significance of preoperative carotid artery flow time (FTc) values as a crucial determinant in the occurrence of hypotension following anesthesia induction in patients aged 65 and above. A reduction in carotid artery FTc is associated with a rapid and substantial decline in blood pressure post-general anesthesia induction. According to our findings, early identification of elderly patients prone to developing hypotension can be facilitated through preoperative carotid artery FTc measurements.
In recent times, carotid artery FTc measurement has been utilized either independently or in conjunction with other methods to assess current volume status in both critical and non-critical patients [6, 10,11,12,13]. Barjaktarevic et al. demonstrated that carotid artery FTc values increased in correlation with elevated cardiac output resulting from volume infusion [14]. Additionally, carotid artery FTc proves to be a reliable diagnostic tool for estimating fluid status in dehydrated individuals during fasting [10]. This suggests that assessing cardiac functions through carotid artery FTc becomes more feasible when patients undergo prolonged fasting before anesthesia. Hypotension after general anesthesia induction can be attributed to various factors, and prolonged fasting time is particularly crucial for elderly patients who are more susceptible to dehydration and the adverse effects of inadequate fluid intake. In our study, patients who developed hypotension after anesthesia induction exhibited longer fasting times and lower preoperative carotid artery FTc values compared to those without hypotension. Our findings support the hypothesis that extended fasting time and low carotid artery FTc values contribute to anesthesia-induced hypotension.
There is limited literature on the use of carotid artery FTc in elderly patients. Chen et al. employed carotid artery FTc as an alternative method to stroke volume variation (SVV) for assessing volume responsiveness in elderly patients during general anesthesia [15]. Maitra et al. suggested that a carotid artery FTc value below 330.2 ms, with a sensitivity of 85.7% and specificity of 96.8%, can predict hypotension after propofol-induced anesthesia in ASA I–II patients aged between 18 and 65 years [8]. In a separate investigation, the association between FTc values and fluid responsiveness was evaluated in spontaneously breathing awake patients spanning an age range of 19 to 80 years. The determined cut-off value for fluid responsiveness in this study was 349.4 ms, exhibiting a sensitivity of 72.7% and specificity of 87.1% [16]. Notably, these investigations did not exclusively focus on elderly patients. Wang et al. proposed that a carotid artery FTc value below 379.1 ms reliably indicated post-induction hypotension in elderly patients [17]. However, it is crucial to acknowledge that their study specifically enrolled elderly patients aged 65–75 years with ASA physical status I–II, excluding those with hypertension, diabetes, cardiovascular disease, and obesity. They uniformly administered crystalloid at a rate of 10 mL/kg/h to all patients before measuring carotid artery FTc and calculated FTc values using Bazett’s formula. In our research, all patients aged over 65 with ASA scores of I–III were included, with the oldest participant being 87 years old, and no fluid infusion was administered before carotid artery FTc measurement. In addition; given prior suggestions that Wodey’s formula could be as accurate as Bazett’s formula or even superior, we opted for Wodey’s formula for FTc calculation [18]. Our findings aligned with those reported in existing literature.
The ROC curve analysis in our study revealed an area under the curve for carotid artery FTc of 0.93, with an optimal cut-off of 333 ms, a sensitivity of 90.4%, and a specificity of 84.9%. Consequently, a preoperative carotid artery FTc value below 333 ms was identified as a significant indicator for clinical hypotension. In Wang et al.’s study, investigating the efficacy of FTc in elderly patients, the identification of a higher cut-off value (379.1 ms) may be attributed to the exclusion of patients over 75 years of age and those with comorbidities [17]. Additionally, all patients received fluid infusion before carotid artery FTc measurement in their study.
The duration of FTc is influenced by factors such as left ventricular preload, myocardial contractility, and afterload. Consequently, the presence of hypertension, which elevates left ventricular afterload, may result in a shortened carotid artery FTc [19]. Moreover, individuals of advanced age and with a high ASA physical status might be more susceptible to hemodynamic fluctuations due to factors such as left ventricular diastolic dysfunction, reduced vascular reactivity, and heightened sensitivity to anesthetics [17]. Indeed, our study observed a higher risk of hypotension post-anesthesia induction in elderly patients with preoperative hypertension, elevated ASA physical status, and a diminished carotid artery FTc value. However, based on logistic regression analysis, only a low carotid artery FTc value emerged as an independent risk factor for anesthesia-induced hypotension among these variables. This finding underscores the effectiveness of carotid artery FTc value in predicting hypotension following the induction of general anesthesia.
The strength of our study lies in its uniqueness as, to the best of our knowledge, it is the first to investigate the effectiveness of FTc in predicting hypotension after anesthesia induction in all patients aged over 65, including those with comorbidities and an ASA physical status of less than 4. However, the study had some limitations. First, it was conducted as a single-center study. Second, the absence of BIS monitoring during anesthesia induction is noteworthy. Although the propofol dose remained consistent across patients with and without hypotension, the inclusion of BIS monitoring could have provided valuable insights into the depth of anesthesia. Third, since several studies have shown that FTc is an indicator of afterload, preload, cardiac contractility, and fluid response, additional ECHO-related parameters were not included in the study data. Fourth, continuous blood pressure monitoring provides more accurate information. However, given that we included patients with very low surgical risk (ophthalmic surgery, examination under general anesthesia, etc.) and the incorporation of hemodynamic data between induction of anesthesia and endotracheal intubation in the study, we used non-invasive arterial monitoring.
5 Conclusion
Based on our study results, a preoperative carotid artery flow time (FTc) of less than 333 ms is identified as an independent risk factor for hypotension following the induction of anesthesia with propofol in elderly patients with comorbidities and ASA physical statuses I–III. The measurement of preoperative carotid artery FTc emerges as a straightforward, bedside, noninvasive, and dependable method for predicting anesthesia-induced hypotension within this specific patient demographic.
Data availability
No datasets were generated or analysed during the current study.
References
Futier E, Lefrant JY, Guinot PG, Godet T, Lorne E, Cuvillon P, Bertran S, Leone M, Pastene B, Piriou V, Molliex S, Albanese J, Julia JM, Tavernier B, Imhoff E, Bazin JE, Constantin JM, Bruno P, Jaber S, INPRESS Study Group. Effect of individualized vs standard blood pressure management strategies on postoperative organ dysfunction among high-risk patients undergoing major surgery: a randomized clinical trial. JAMA. 2017;318(14):1346–57. https://doi.org/10.1001/jama.2017.14172.
Yokose M, Takaki R, Mihara T, Saigusa Y, Yamamoto N, Masui K, Goto T. Hypotension after general anesthesia induction using remimazolam in geriatric patients: protocol for a double-blind randomized controlled trial. PLoS ONE. 2022;17(9):e0275451. https://doi.org/10.1371/journal.pone.0275451.
Myrberg T, Lindelöf L, Hultin M. Effect of preoperative fluid therapy on hemodynamic stability during anesthesia induction, a randomized study. Acta Anaesthesiol Scand. 2019;63(9):1129–36. https://doi.org/10.1111/aas.13419.
Legrand M, Kothari R, Fong N, Palaniappa N, Boldt D, Chen LL, Kurien P, Gabel E, Sturgess-DaPrato J, Harhay MO, Pirracchio R, Bokoch MP. VEGA-1 trial investigators 2023 Norepinephrine versus phenylephrine for treating hypotension during general anaesthesia in adult patients undergoing major noncardiac surgery: a multicentre, open-label, cluster-randomised, crossover, feasibility, and pilot trial. Br J Anaesth. 2023;130(5):519–27. https://doi.org/10.1016/j.bja.2023.02.004.
Ma IWY, Caplin JD, Azad A, Wilson C, Fifer MA, Bagchi A, Liteplo AS, Noble VE. Correlation of carotid blood flow and corrected carotid flow time with invasive cardiac output measurements. Crit Ultrasound J. 2017;9(1):10. https://doi.org/10.1186/s13089-017-0065-0.
Kimura A, Suehiro K, Juri T, Tanaka K, Mori T. Changes in corrected carotid flow time induced by recruitment maneuver predict fluid responsiveness in patients undergoing general anesthesia. J Clin Monit Comput. 2022;36(4):1069–77. https://doi.org/10.1007/s10877-021-00736-7.
Kim HJ, Choi YS, Kim SH, Lee W, Kwon JY, Kim DH. Predictability of preoperative carotid artery-corrected flow time for hypotension after spinal anaesthesia in patients undergoing caesarean section: a prospective observational study. Eur J Anaesthesiol. 2021;38(4):394–401. https://doi.org/10.1097/EJA.0000000000001376.
Maitra S, Baidya DK, Anand RK, Subramanium R, Bhattacharjee S. Carotid artery corrected flow time and respiratory variations of peak blood flow velocity for prediction of hypotension after induction of general anesthesia in adult patients undergoing elective surgery: a prospective observational study. J Ultrasound Med. 2020;39(4):721–30. https://doi.org/10.1002/jum.15151.
Wodey E, Carre F, Beneux X, Schaffuser A, Ecoffey C. Limits of corrected flow time to monitor hemodynamic status in children. J Clin Monit Comput. 2000;16(3):223–8. https://doi.org/10.1023/a:1009981024804.
Shokoohi H, Berry GW, Shahkolahi M, King J, King J, Salimian M, Poshtmashad A, Pourmand A. The diagnostic utility of sonographic carotid flow time in determining volume responsiveness. J Crit Care. 2017;38:231–5. https://doi.org/10.1016/j.jcrc.2016.10.025.
Hossein-Nejad H, Mohammadinejad P, Lessan-Pezeshki M, Davarani SS, Banaie M. Carotid artery corrected flow time measurement via bedside ultrasonography in monitoring volume status. J Crit Care. 2015;30(6):1199–203. https://doi.org/10.1016/j.jcrc.2015.08.014.
Beier L, Davis J, Esener D, Grant C, Fields JM. Carotid ultrasound to predict fluid responsiveness: a systematic review. J Ultrasound Med. 2020;39(10):1965–76. https://doi.org/10.1002/jum.15301.
Antiperovitch P, Iliescu E, Chan B. Carotid systolic flow time with passive leg raise correlates with fluid status changes in patients undergoing dialysis. J Crit Care. 2017;39:83–6. https://doi.org/10.1016/j.jcrc.2017.02.017.
Barjaktarevic I, Chiem A, Cannesson M. Time to correct the flow of corrected flow time. Crit Ultrasound J. 2017;9(1):18. https://doi.org/10.1186/s13089-017-0076-x.
Chen Y, Liu Z, Fang J, Xie Y, Zhang M, Yang J. Correlation of carotid corrected flow time and respirophasic variation in blood flow peak velocity with stroke volume variation in elderly patients under general anaesthesia. BMC Anesthesiol. 2022;22(1):246. https://doi.org/10.1186/s12871-022-01792-5.
Kim DH, Shin S, Kim N, Choi T, Choi SH, Choi YS. Carotid ultrasound measurements for assessing fluid responsiveness in spontaneously breathing patients: corrected flow time and respirophasic variation in blood flow peak velocity. Br J Anaesth. 2018;121(3):541–9. https://doi.org/10.1016/j.bja.2017.12.047.
Wang J, Li Y, Su H, Zhao J, Tu F. Carotid artery corrected flow time and respiratory variations of peak blood flow velocity for prediction of hypotension after induction of general anesthesia in elderly patients. BMC Geriatr. 2022;22(1):882. https://doi.org/10.1186/s12877-022-03619-x.
Mohammadinejad P, Hossein-Nejad H. Calculation of corrected flow time: Wodey’s formula vs. Bazett’s formula. J Crit Care. 2017;44:154–5. https://doi.org/10.1016/j.jcrc.2017.10.046.
Singer M, Allen MJ, Webb AR, Bennett ED. Effects of alterations in left ventricular filling, contractility, and systemic vascular resistance on the ascending aortic blood velocity waveform of normal subjects. Crit Care Med. 1991;19(9):1138–45. https://doi.org/10.1097/00003246-199109000-00008.
Acknowledgements
Assistance with the special article: the authors would like to thank to True Editors Customer Service (order 4458) for English editing of this study.
Funding
The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by ID, MT, EG, YKA, and MO. The first draft of the manuscript was written by ID and MT, all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors have no relevant financial or non-financial interests to disclose.
Ethical approval
This study was performed in line with the principles of the Declaration of Helsinki. The approval for this study was obtained from the institutional review board of Cukurova University Ethics Committee (Date: Jan/06/2023, IRB number: 129/10).
Consent to participate
Our study did NOT use any diagnostic images (x-rays, laparoscopic images, ultrasound images, brain scans, pathology slides, etc.) or any part of the participants' bodies.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Demir, I., Turktan, M., Gulec, E. et al. The predictive role of carotid artery flow time for anesthesia-induced hypotension in high-risk elderly patients. J Clin Monit Comput 38, 859–867 (2024). https://doi.org/10.1007/s10877-024-01145-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10877-024-01145-2