Abstract
Acceleromyography is characterised by an increase of the twitch response T1 (first twitch of the train-of-four [TOF]) during first 30 min of monitoring known as the staircase phenomenon. In adults the staircase phenomenon can be avoided by tetanic prestimulation. This study examined, if tetanic prestimulation eliminates the staircase phenomenon in children. After written informed consent, the neuromuscular function of 80 children, 10 in each age group (< 6 months, 6–12 months, 12–24 months, 2–3 years, 3–6 years, 6–12 years, 12–18 years, and ≥ 18 years) was measured on both arms simultaneously over 30 min under general anaesthesia. The ulnaris nerve was stimulated using the TOF technique every 15 s. The twitch response (T1, TOF ratio [TOFR]) was measured by acceleromyography. Before calibration, tetanic prestimmulation (50 Hz for 5 s) was administered to one randomly selected arm. The effect of tetanic prestimulation and age was analysed using general linear models based on the normalized T1 and TOFRs of both arms. Tetanic prestimulation significantly affected T1 values avoiding the staircase phenomenon (p < 0.0001). After 5.8 min [1.0–17.2 min] the normalized T1 values increased to 117% [102–147%] without prestimulation (p < 0.0001) independent of the age group (p = 0.539). The normalized TOFR was stable throughout the observation period of 30 min 100% [95–107%]. Infants (> 12 weeks), children, and young adults (< 18 years) develop similar characteristics of the staircase phenomenon than adults. Tetanic prestimulation prevents the staircase phenomenon in these age groups. The stability of the TOFR reading confirms its value to monitor neuromuscular function over time.
Registration: The study was registered as NCT02552875 on Clinical Trials.gov on July 29, 2014.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Acceleromyography (AMG) is the most frequently used technique to monitor neuromuscular function when neuromuscular blocking drugs are given during anaesthesia [1]. Furthermore, AMG is a recommended quantitative technique to investigate the pharmacodynamic characteristics of neuromuscular blocking agents (NMBA) or their reversal agents [2]. Following supramaximal stimulation of the ulnaris nerve the acceleration of the thumb is recorded as a measure of contraction force of the adductor pollicis muscle, the twitch response. The depth of the neuromuscular block is quantified by the twitch response related to a baseline value, which has been obtained before injection of the NMBA. A major limitation of the AMG technique is the increase of the twitch’s baseline value referred to as the staircase phenomenon, which does bias pharmacokinetic studies [3].
The staircase phenomenon develops during low-frequency nerve stimulation [4, 5]. Studies in animals and humans demonstrated heterogeneity of the staircase phenomenon intraindividually at different muscles but also interindividually at the distinctive measuring site, the adductor pollicis muscle [6]. Although, it is not possible to predict whether a staircase phenomenon will appear, 30 min are commonly sufficient to get a stable baseline using single twitch (1 Hz) or train-of-four (2 Hz every 12–15 s) stimulation [7]. In adult patients, time for signal stabilization can be shortened to 2–5 min when a 50 Hz-tetanus over 5 s is applied before calibration [7]. However, there is scarce knowledge about AMG signal stabilisation in pediatric patients [8, 9]. In particular, experience about the effects of tetanic prestimulation is not available in children.
Therefore, we examined if a 50 Hz tetanus applied for 5 s to the ulnaris nerve before initial calibration of the acceleromyograph effectively prevents the staircase phenomenon in infants, children and young adults in a similar fashion than in adults. Monitoring the neuromuscular function simultanously at both forearms, we additionally examined if there is an age related difference of the extent of the staircase phenomenon.
2 Methods
The study was performed between September 20, 2014 and September 15, 2015. The protocol was approved by the Institutional Ethics Committee (University of Regensburg, Regensburg, Germany; protocol No. 14-101-0114). After written informed consent, 142 infants, children and young adults were recruited. Classified as American Society of Anesthesiologists physical status I or II, they were scheduled to undergo general anesthesia for elective surgical procedures. Exclusion criteria included patients with diseases or on medications known to interfere with neuromuscular transmission, hepatic or renal dysfunction, and allergies to medications used in this trial.
2.1 Study design and data collection
This randomized, controlled, single-blinded trial was designed to characterize the staircase phenomenon in eight age related groups: < 6 months, 6–12 months, 12–24 months, 2–3 years, 3–6 years, 6–12 years, 12–18 years, ≥ 18 years (age control). The effect of tetanic stimulation was evaluated using an intraindividual comparison. The ulnar nerve of the control arm was stimulated by train-of-four (TOF) pattern only. Simultaneously the ulnar nerve of the contralateral arm was prestimulated with a 50 Hz tetanus for 5 s (tetanus arm) and continued also by TOF stimulation. Control and tetanus arm as well as the monitoring devices were allocated using a randomization module based in the hospital’s internal network. Furthermore, randomization was organized in two blocks of four patients in each age related group. A final block was sized to end up with ten patients in each age group to allow substitution of excluded patients.
2.2 Anaesthesia management
Children < 8 months received no premedication while older children and young adults received weight-adapted oral midazolam. They were monitored by non-invasive arterial blood pressure (placed on the leg), electrocardiogram, and peripheral oxygen saturation (SpO2). Anesthesia was induced either with inhaled sevoflurane (4% to 6%) or propofol (2–3 mg kg−1) and remifentanil (5 µg kg−1 min−1) according to patient´s ability to cooperate while obtaining venous access. Tracheal intubation or insertion of a laryngeal mask was performed without neuromuscular blocking drugs. Anaesthesia was maintained with propofol (5–8 mg kg−1 h−1) and remifentanil (0.2–0.5 µg kg−1 min−1) to achieve a bispectral index of 40–60 (Draeger Medical Systems, Telford, Pennsylvania, USA). The lungs were ventilated with a 50% oxygen in air to achieve an end-tidal CO2 partial pressure of 35–40 mmHg. Before starting the neuromuscular monitoring the absence of endtidal sevoflurane was proved to avoid influence of sevoflurane on neuromuscular function. Core body and palmar skin temperature were kept constant above 36 °C and 32 °C, respectively using a warming blanket (WARMTOUCH™ WT 600, Covidien Deutschland GmbH, Neustadt, Germany) and isolating body covers for the arms.
2.3 Neuromuscular monitoring
Neuromuscular monitoring followed the good clinical research practice guidelines in pharmacodynamic studies of NMBA [2]. After cleaning of the skin two paediatric surface electrodes (Neotrode®, ConMed Corporation, NY, USA) were placed over the ulnar nerves on the volar side of both wrists. A piezocristal transducer (TOF-Watch SX® acceleromyograph, Organon Ltd., Dublin, Ireland) was attached to the volar side of the thumbs at the interphalangeal joint. Both arms were immobilized. In particular, movement of the fingers was restricted in extended position with surgical tape on the armboard. The thumbs were extended with a preload (50–150 g) using a double strand of an elastic rubber band (2 mm thick) to decrease signal variability in all subjects [2, 10, 11]. Data from the two TOF Watch® SX devices were registered and stored on two computers (software: TOF Watch® SX Monitor Version 2.5.Int, 2007).
Before application of preload and calibration of AMG device, a 50-Hz-tetanus was administered for 5 s with 50 mA current at the ulnar nerve of the tetanus arm. Within the next 60 s, both AMG devices were calibrated (CAL 2 mode) using the built-in calibration mode to determine the supramaximal current [10]. Then, both AMG devices were started in TOF mode delivering four square wave pulses of 0.2 ms at a frequency of 2 Hz every 15 s for 30 min. To control for baseline variations, the amplitude of the first twitch of TOF (T1) as well as TOF ratio (TOFR) were normalized, i.e. the first values obtained after calibration were set to 100% [2, 9, 10]. After 30 min of TOF stimulation, the study protocol was finished and the scheduled surgical procedure was started.
Repeatability of AMG values is impaired [12]. Therefore, an anesthesiologist (C.U.), blinded to the intervention, reviewed the AMG traces to guarantee data quality. If either T1-to-T1 or TOFR-to-TOFR variations were higher than 7% it was classified as an artefact and the respective data were excluded from analysis. These patients were substituted adding additional cases to the last block of the randomization list. Furthermore, we assumed that maximum values of T1 are given if the T1 value has reached the individual 97% percentile of a respective patient.
2.4 Statistical analysis
Primary outcome variable was the T1 values measured at both aductor pollicis muscles over 30 min. Secondary outcome variable was the analogously measured TOFR and the time to the maximum increase of the T1 values of the control arm.
Sample size was calculated according to the primary outcome variable. We assumed a mean difference of 25% between the maximum T1 values with and without tetanic prestimulation, a standard deviation of 20% of the difference, and a correlation coefficient of 0.3 between both arms. To detect this effect by using a paired t test at a significance level α = 0.05 with a power of 0.8 a total of n = 10 patients per age group were needed.
Statistical analyses were performed with general linear models (GLM) for each tested dependent variable (T1 and TOFR) in a hierarchical fashion. We tested two within-group factors, the development of the staircase phenomenon by the factor time and the effect of the tetanic prestimulation by the factor intervention (control vs. tetanus). The age groups served as between-group factor. Post-hoc tests in sub-cohorts were performed if the respective main factor was significant by Dunnett-T tests. The time to the maximal increase of T1 values was compared between the age groups using the Kruskal–Wallis-H test. Values are given as medians and 95% confidence intervals. A p-value < 0.05 was considered statistical significant.
Statistics was performed using SAS 9.4 (SAS Institute Inc., Cary, NC).
3 Results
In 104 of 142 recruited patients neuromuscular measurement was performed. Finally 80 participants were enrolled and evaluated (Fig. 1), whose baseline characteristics are presented in Table 1. Technical difficulties such as inability to find a supramaximal stimulation current or difficulties to fix the devices within the time frame of 60 s between tetanus and TOF stimulation more frequently appeared in the youngest age groups. Minor local skin irritation and redness at the stimulation site developed in 18 patients independently of tetanic stimulation and age. Induction of anesthesia with sevoflurane by facemask was performed in 17 patients preferrently in the younger children (Table 1). No patient needed a manual adjustment of the gain factor of the TOF Watch SX®.
CONSORT Flow Diagram. Patients were excluded if the time delay between tetanus and start of TOF stimulation was longer than 60 s (application) or if either supramaximal stimulation current was not found (device failure). Posthoc exclusions were necessary if the difference between two consecutive T1 values was > 7%
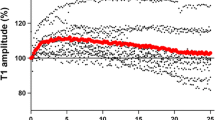
Tetanic prestimulation significantly affected the T1 values avoiding the staircase phenomenon (upper panel of Fig. 2, p < 0.0001). At every control arm (without tetanic prestimulation) T1 values increased during the observation period of 30 min (staircase phenomenon). The maximum increase of the normalized T1 values was 117% [102–147%] and did not differ between groups (central panel of Fig. 2, p = 0.539). It was reached after 5.8 min [1.0–17.2 min] independent of the patients age (age groups: p = 0.785). At the tetanus arm in most patients the T1 values decreased during the observation period of 30 min (lower panel of Fig. 2). T1 values of all patients reached a median of 85% [68–103%] 30 min after tetanic prestimulation and did not differ between groups (p = 0.102).
Twitch response (T1% of normalized control measurement). The upper panel shows the means in the eight different age groups of T1 over 30 min measured at the control and the tetanus arm. General linear models revealed a significant effect of time (square fashion, p < 0.0001) as well as intervention (control vs. tetanus arm, p < 0.0001) but no effect of the age group (p = 0.539). T1 means of the control arm were > 100% at any time illustrating the staircase phenomenon, while T1 means of the tetanus arm did not increase over time illustrating that tetanic prestimulation prevented the staircase phenomenon. Even more, the T1 means of the prestimulated tetanus arm decreased within the observation period. The central panel shows the extent of the staircase phenomenon as indicated by the maximum T1 increase of the control arm, which did not differ between the eight age groups (p = 0.539). The lower panel shows that the maximum decrease of T1 values of the tetanus arm did also not differ between ages (p = 0.102)
The normalized TOFR was 100% [95–107%] at the end of the observation period 30 min after start of the monitoring. It did not change over time, between intervention, and between age groups (Fig. 3).
Train-of-four ratio (TOFR of normalized control measurement). The upper panel shows the means in the eight different age groups of the TOFR [%] over 30 min measured at the control and the tetanus arm. General linear models revealed not any significant effect of time (p = 0.879),intervention (control vs. tetanus arm, p = 0.267), and age group (p = 0.102). The lower panel shows distribution of the TOFR of the control arm in th different age groups 30 min after start of the measurements
4 Discussion
We confirmed that the staircase phenomenon develops in any age when neuromuscular function is monitored with AMG [9]. For the first time, however, we could demonstrate that a 50 Hz tetanus applied for 5 s to the ulnaris nerve prevents the staircase phenomenon in particular in infants, children and young adults. Importantly, we also confirmed that the TOFR is not affected by either the staircase phenomenon or the tetanic prestimulation [9].
In contrast to electromyography, where repetitive stimulation does not increase the size of the compound action potential [4], mechanomyography [13] and AMG [10] develop the staircase phenomenon indicating a muscular origin for this stepwise initial increase of the twitch response. It is most probably a biological attribute to repetitive muscle contraction. The twitch potentiation is most likely caused by a Ca2+-calmodulin-dependent phosphorylation of the regulatory light chains of myosin with an increased calcium sensitivity and an increased rate of cross-bridges [14, 15].
In the first weeks of life muscle fibres undergo functional and structural changes. They are transformed from slow-twitch (type I) to fast-twitch fibres (type II) [16]. However, the adductor pollicis muscle does not follow this transformation; type I muscle fibres still predominante in adults [17]. Therefore, the staircase phenomenon is supposed to be comparably developed in children of any age [9]. In accordance to these physiological considerations, our data confirm age independency of the staircase phenomenon. This is in contrast to Zhou and colleagues, who demonstrated that in smaller infants (1–5 months) the maximum staircase effect appears earlier (7.25 min vs. 9.5 min) and to lower degrees (121 ± 15% vs. 143 ± 14%) compared to older children (6–24 months) [9]. The poor repeatability of any AMG device [12] determination of the maximum T1 but even more the time to maximum is challenging. Assuming an imprecision of 3% we measured a maximum increase of median T1 values of 117% [102–147] at 5.8 min [1.0–17.2 min].
Accordingly, it was expected that tetanic prestimulation may have the same effect on the staircase phenomenon in children compared to adults. Kopman and collegues demostrated that 50 Hz tetanic prestimulation for 5 s prevents the staircase phenomenon in adults resulting in stable T1 values between 97 ± 5% and 105 ± 15% of baseline [10]. Using the same stimulation patterns we demonstrated that tetanic prestimulation eradicates the staircase phenomenon in each age group.
In pharmacodynamic and pharmacokinetic studies of NMBA a stable baseline of control T1 value is essential for valid results [2]. The staircase phenomenon artificially prolongates the onset times of neuromuscular blocking drugs, because they are determined during T1 increase and before the plateau is reached [3]. Shortening the stabilization period by up to 20 min, the tetanic prestimulation added to validity of pharmocodynamic studies within the induction period of children. Therefore, the recommendation to use tetanic prestimulation with stable T1 values (< 5% variation) for 2–5 min before application of the neuromuscular blocking drug has to be transferred from adults to children [2].
Following tetanic prestimulation T1 values significantly decreased in an age-independent manner to 85% [68–103%] after 30 min. In this respect, data from adults are inconsistent. While Kopman and collegues pointed out that tetanic prestimulation reduces variability of T1 values without any decrease ranging between 87 and 136% after 22.5 min [10], Mazzinari and collegues reported a decrease to 79% [70–92%] after 20 min consistant with our results [3]. Following tetanic stimulation, however, twitch responses are typically increased by approximately 27% [18]. This effect, also referred as posttetanic potentiation, continues for 6 to 10 min [18]. Therefore, the observed decrease of T1 following tetanic prestimulation is reasonable.
The TOFR was stable at each arm during the observation period in any age group. In accordance with previous studies [3, 19], we can confirm that the staircase phenomenon but also the tetanic prestimulation influences the first and the fourth twitch response uniformly in the same extent. This highlights the advantage of TOFR as the clinical parameter to monitor neuromuscular function over time, in particular to decide sufficient neuromuscular recovery before extubation [20]. Important to note, AMG has to be normalized when sufficient neuromuscular recovery is determined, i.e. the TOFR has to recover to at least 90% of the TOFR measured before giving a neuromuscular blocking drug [21].
4.1 Limitations
The study has limitations. In some children < 6 years we induced anaesthesia by inhalation of sevoflurane, which is known to potentiate the action of neuromuscular blocking drugs [22]. However, we applied sevoflurane not longer than 10 min and equilibration of volatile agents between alveolar and muscle compartment requires almost 45 min [23]. Furthermore, we started our measurements not before the endtidal sevoflurane concentration was 0.0 vol%. Finally, there is no evidence that volatile anaesthetics depress the twitch response per se [24]. Nevertheless, we cannot doubtlessly exclude that the staircase phenomenon was influenced using this technique.
The slope of staircase phenomenon is maximal during the first stimulations. Accordingly, the single twitch stimulation during calibration (CAL 2 mode) most likely induced a relevant increases of the twitch response, which is not recorded. Accordingly, Zhou and collegues, who calibrated their devices just with 60 mA refraining from determining the supramaximal current, had higher maxima of the T1 values [9]. We decided to follow the consensus recommendations and calibrated as well as normalized the AMG devices and therefore might underestimate the extent of the T1 increase [2].
Depending on maturation of the neuromuscular junction through the first 8–12 weeks of life, infants younger than 4 weeks show a TOF fading [25]. We did not include infants of this age class. Therefore, we have to restrict our conclusions respectively.
The study was done on an immobilized forearm with a preload applied to the thumb. Even in such optimized conditions there was a 9% failure rate of the device predominantly in younger patients and a 14% rate of unacceptable variability. Thus, even under optimal conditions nearly a quarter of patients can not be adequately monitored with acceleromyography.
5 Conclusions
In conclusion, infants > 12 weeks, children, and young adults (< 18 years) develop similar characteristics of the staircase phenomenon than adults. Tetanic prestimulation prevents children of any age from the staircase phenomenon and shortens the period of siganal stabilization. Neither staircase phenomenon nor tetanic prestimulation influence the TOFR.
References
Kirmeier E, Eriksson LI, Lewald H, Jonsson Fagerlund M, Hoeft A, Hollmann M, Meistelman C, Hunter JM, Ulm K, Blobner M, Contributors P. Post-anaesthesia pulmonary complications after use of muscle relaxants (POPULAR): a multicentre, prospective observational study. Lancet Respir Med. 2018. https://doi.org/10.1016/S2213-2600(18)30294-7.
Fuchs-Buder T, Claudius C, Skovgaard LT, Eriksson LI, Mirakhur RK, Viby-Mogensen J, 8th International Neuromuscular M. Good clinical research practice in pharmacodynamic studies of neuromuscular blocking agents II: the Stockholm revision. Acta Anaesthesiol Scand. 2007;51(7):789–808. https://doi.org/10.1111/j.1399-6576.2007.01352.x.
Mazzinari G, Errando CL, Diaz-Cambronero O, Martin-Flores M. Influence of tetanic stimulation on the staircase phenomenon and the acceleromyographic time-course of neuromuscular block: a randomized controlled trial. J Clin Monit Comput. 2019;33(2):325–32. https://doi.org/10.1007/s10877-018-0157-9.
Krarup C. Enhancement and diminution of mechanical tension evoked by staircase and by tetanus in rat muscle. J Physiol. 1981;311:355–72.
Ritchie JM, Wilkie DR. The effect of previous stimulation on the active state of muscle. J Physiol. 1955;130(2):488–96.
Deschamps S, Trager G, Mathieu PA, Hemmerling TM. The staircase phenomenon at the corrugator supercilii muscle in comparison with the hand muscles. Br J Anaesth. 2005;95(3):372–6. https://doi.org/10.1093/bja/aei183.
Lee GC, Iyengar S, Szenohradszky J, Caldwell JE, Wright PM, Brown R, Lau M, Luks A, Fisher DM. Improving the design of muscle relaxant studies. Stabilization period and tetanic recruitment. Anesthesiology. 1997;86(1):48–54.
Driessen JJ, Robertson EN, Booij LH. Acceleromyography in neonates and small infants: baseline calibration and recovery of the responses after neuromuscular blockade with rocuronium. Eur J Anaesthesiol. 2005;22(1):11–5.
Zhou ZJ, Wang X, Zheng S, Zhang XF. The characteristics of the staircase phenomenon during the period of twitch stabilization in infants in TOF mode. Paediatr Anaesth. 2013;23(4):322–7. https://doi.org/10.1111/pan.12041.
Kopman AF, Kumar S, Klewicka MM, Neuman GG. The staircase phenomenon: implications for monitoring of neuromuscular transmission. Anesthesiology. 2001;95(2):403–7.
Claudius C, Skovgaard LT, Viby-Mogensen J. Is the performance of acceleromyography improved with preload and normalization? A comparison with mechanomyography. Anesthesiology. 2009;110(6):1261–70. https://doi.org/10.1097/ALN.0b013e3181a4f239.
Liang SS, Stewart PA, Phillips S. An ipsilateral comparison of acceleromyography and electromyography during recovery from nondepolarizing neuromuscular block under general anesthesia in humans. Anesth Analg. 2013;117(2):373–9. https://doi.org/10.1213/ANE.0b013e3182937fc4.
McCoy EP, Mirakhur RK, Connolly FM, Loan PB. The influence of the duration of control stimulation on the onset and recovery of neuromuscular block. Anesth Analg. 1995;80(2):364–7.
Rassier DE, Macintosh BR. Coexistence of potentiation and fatigue in skeletal muscle. Braz J Med Biol Res. 2000;33(5):499–508.
Rassier DE, Tubman LA, MacIntosh BR. Staircase in mammalian muscle without light chain phosphorylation. Braz J Med Biol Res. 1999;32(1):121–9.
Curless RG. Developmental patterns of peripheral nerve, myoneural junction and muscle: a review. Prog Neurobiol. 1977;9(4):197–209.
Round JM, Jones DA, Chapman SJ, Edwards RH, Ward PS, Fodden DL. The anatomy and fibre type composition of the human adductor pollicis in relation to its contractile properties. J Neurol Sci. 1984;66(2–3):263–72.
Krarup C. Electrical and mechanical responses in the platysma and in the adductor pollicis muscle: in normal subjects. J Neurol Neurosurg Psychiatry. 1977;40(3):234–40.
Diaz-Cambronero O, Flor Lorente B, Mazzinari G, Vila Montanes M, Garcia Gregorio N, Robles Hernandez D, Olmedilla Arnal LE, Argente Navarro MP, Schultz MJ, Errando CL, group IPs. A multifaceted individualized pneumoperitoneum strategy for laparoscopic colorectal surgery: a multicenter observational feasibility study. Surg Endosc. 2019;33(1):252–60. https://doi.org/10.1007/s00464-018-6305-y.
Ali HH, Utting JE, Gray C. Stimulus frequency in the detection of neuromuscular block in humans. Br J Anaesth. 1970;42(11):967–78.
Suzuki T, Fukano N, Kitajima O, Saeki S, Ogawa S. Normalization of acceleromyographic train-of-four ratio by baseline value for detecting residual neuromuscular block. Br J Anaesth. 2006;96(1):44–7. https://doi.org/10.1093/bja/aei273.
Motamed C, Donati F. Sevoflurane and isoflurane, but not propofol, decrease mivacurium requirements over time. Can J Anaesth. 2002;49(9):907–12. https://doi.org/10.1007/BF03016872.
Hemmerling TM, Schuettler J, Schwilden H. Desflurane reduces the effective therapeutic infusion rate (ETI) of cisatracurium more than isoflurane, sevoflurane, or propofol. Can J Anaesth. 2001;48(6):532–7. https://doi.org/10.1007/BF03016828.
Miller RD, Eger EI 2nd, Way WL, Stevens WC, Dolan WM. Comparative neuromuscular effects of Forane and halothane alone and in combination with d-tubocurarine in man. Anesthesiology. 1971;35(1):38–42.
Goudsouzian NG. Maturation of neuromuscular transmission in the infant. Br J Anaesth. 1980;52(2):205–14.
Funding
There was no funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
CU, MW, TM, RZ, SK, TS, BG, FZ, BS declare that they have no conflict of interest. MB received honoraria for board services and lectures from GE Health Care (Helsinki, Finland), MSD (Haar, Germany), and Grünenthal (Aachen, Germany).
Ethical approval
All procedures performed in this study involving human participants were in accordance with the ethical standards of the institutional and/or national research committee (Institutional Ethics Committee,University of Regensburg, Regensburg, Germany; protocol No. 14-101-0114) and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Unterbuchner, C., Werkmann, M., Ziegleder, R. et al. Shortening of the twitch stabilization period by tetanic stimulation in acceleromyography in infants, children and young adults (STSTS-Study): a prospective randomised, controlled trial. J Clin Monit Comput 34, 1343–1349 (2020). https://doi.org/10.1007/s10877-019-00435-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10877-019-00435-4