Abstract
Two anhydrous salts of piperidine have been prepared with naphthalene-1,5-disulfonic acid, and 4-nitrophthalic acid by slow evaporation technique, and their structures were fully characterized. These two structures adopted homo or hetero supramolecular synthons. Compound 1 crystallizes in the triclinic, space group P-1, with a = 7.728(3) Å, b = 8.042(3) Å, c = 9.150(3) Å, α = 80.631(4)°, β = 79.433(4)°, γ = 86.531(4)°, V = 551.3(3) Å3, Z = 1. Compound 2 crystallizes in the monoclinic, space group P2(1)/c, with a = 6.3466(5) Å, b = 24.008(2) Å, c = 9.0390(7) Å, α = 90°, β = 97.0050(10)°, γ = 90°, V = 1367.01(19) Å3, Z = 4. Analysis of the crystal packing of 1, 2 suggests that there are N–H···O hydrogen bonds between the acids and the piperidine in both compounds. Except the classical hydrogen bonds, the secondary propagating interactions including CH2···O, O···Cπ, and CH2···π interactions also play important roles in structure extension. In conclusion, we have shown that 3D structures can be constructed by collective non-covalent interactions.
Graphical Abstract
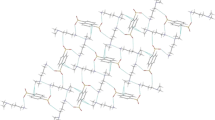
The crystal structures of the salts from piperidine, naphthalene-1,5-disulfonic acid, and 4-nitrophthalic acid are predominantly stabilized by classical hydrogen bonds as well as CH2···O, O···Cπ, and CH2···π interactions.

Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
A productive strategy in the crystal engineering is to build supramolecular structures from molecules that are programmed to engage in multiple interactions such as electrostatic forces, hydrogen bonding, cation···π interaction, C–H···π, and π···π interactions [1–6]. Hydrogen bonding system is the main key for the organization of supramolecules between organic acid and organic base containing the hydrogen bonding sites [7, 8]. Intramolecular, and intermolecular hydrogen bonds are responsible for the construction of several complicated self-assembled structures involving N-containing compounds and acidic components [9, 10].
Through hydrogen bonds cocrystals and organic salts can be generated. In pharmaceuticals, salt formation is often used in order to modify the properties of the compounds [11], such as increasing or decreasing solubility, improving stability and reducing hygroscopicity of a drug product. There are many interesting hydrogen bonded topological structures from infinite 1-D chain to 3-D supramolecular framework [12, 13]. The organic acid bears an important hydrogen bonding functional group for crystal engineering [14–17]. It is interesting to exploit the robust and directional recognition of organic acids with the N-containing basic building blocks [18–25]. In this regard, the most frequently used moieties with hydrogen bonding capability are amine (including aromatic amine and aliphatic amine), and pyridyl derivatives.
Compared with pyridine derivatives, piperidine, besides the NH group, bears the flexible ring with CH2 units, which can provide more complex nonbonding interactions when it interacts with the carboxylic acids. To the best of our knowledge there are very few reports involving acid–base adducts concerning the piperidine [26–29].
As an extension of our study of the weak interactions (hydrogen bonding, π–π interaction, and halogen bonding) concerning the aromatic N-containing derivatives [30, 31], herein we report the preparation and structures of two organic salts assembled from piperidine (L) and the corresponding organic acids (Scheme 1), respectively. The two organic salts are (piperidine)2: (naphthalene-1,5-disulfonic acid) [(HL) 2+2 · (nds2−), nds2− = naphthalene-1,5-disulfonate] (1), and (piperidine): (4-nitrophthalic acid) [(HL) · (Hnpa), Hnpa = hydrogen 4-nitrophthalate] (2).
Experimental Section
Materials and Physical Measurements
The chemicals and solvents used in this work are of analytical grade and available commercially and were used without further purification. The FT-IR spectra were recorded from KBr pellets in range 4000–400 cm−1 on a Mattson Alpha-Centauri spectrometer. Microanalytical (C, H, N, and S) data were obtained with a Perkin-Elmer Model 2400II elemental analyzer. Melting points of the new compounds were recorded on an XT-4 thermal apparatus without correction.
Preparation of the Salts 1–2
(Piperidine)2: (Naphthalene-1,5-Disulfonic Acid) [(HL) 2+2 · (nds2−)] (1)
A solution of naphthalene-1,5-disulfonic acid tetrahydrate (36 mg, 0.1 mmol) in methanol (15 ml) was added dropwise to a vigorously stirred solution of piperidine (8.5 mg, 0.1 mmol) in methanol (3 ml) over a period of 5 min. The solution was further stirred for 10 min, then the solution was filtered into a test tube. The solution was left standing at room temperature for several days. Colorless block crystals were isolated after slow evaporation of the methanol solution in air. The crystals were collected and dried in air to give the compound [(HL) 2+2 · (nds2−)] (1). (yield: 36.0 mg, 78.50 %). mp 184–186 °C. Elemental analysis: Calc. for C20H30N2O6S2 (458.58): C, 52.34; H, 6.54; N, 6.10; S, 13.96. Found: C, 52.28; H, 6.49; N, 6.02; S, 13.88. Infrared spectrum (cm−1): 3389 s(νas(NH)), 3270 s(νs(NH)), 3156, 3090, 2985, 2930, 2852, 1614, 1570, 1523, 1475, 1430, 1386, 1341, 1398, 1255, 1213, 1172, 1128, 1086, 1043, 1001, 958, 914, 870, 827, 784, 743, 697, 655, 630, 612 m.
(Piperidine): (4-Nitrophthalic Acid) [(HL) · (Hnpa)] (2)
A solution of 4-nitrophthalic acid (21.1 mg, 0.1 mmol) in methanol (14 ml) was added dropwise to a vigorously stirred solution of piperidine (8.5 mg, 0.1 mmol) in methanol (3 ml) over a period of 5 min. The solution was further stirred for 10 min, then the solution was filtered into a test tube. The solution was left standing at room temperature for several days. Colorless block crystals were isolated after slow evaporation of the methanol solution in air. The crystals were collected and dried in air to give the compound [(HL) · (Hnpa)] (2). (yield: 26.0 mg, 87.75 %). mp 192–193 °C. Elemental analysis: Calc. for C13H16N2O6 (296.28): C, 52.65; H, 5.40; N, 9.45. Found: C, 52.60; H, 5.32; N, 9.37. Infrared spectrum (cm−1): 3704 s(ν(OH)), 3386 s(νas(NH)), 3268 s(νs(NH)), 3176, 3130, 3084, 2925, 2853 m, 1674 s(ν(C=O)), 1618 s(νas(COO−)), 1570 m, 1529 s(νas(NO2)), 1486 m, 1443 m, 1398 s(νs(COO−)), 1353 m, 1315 s(νs(NO2)), 1270 s(ν(C–O)), 1225, 1178, 1135, 1089, 1044, 972, 933, 887, 844, 799, 757, 712, 668, 625, 601 m.
X-ray Crystallography and Data Collection
The crystals were mounted on a glass fiber on a Bruker SMART 1000 CCD diffractometer operating at 50 kV and 40 mA using Mo Kα radiation (0.71073 Å). Data collection and reduction were performed using the SMART and SAINT software [32]. The structures were solved by direct methods, and the non-hydrogen atoms were subjected to anisotropic refinement by full-matrix least squares on F 2 using SHELXTL package [33].
Hydrogen atom positions for the two structures were located in a difference map and refined independently. Further details of the structural analysis are summarized in Table 1. Selected bond lengths and angles for the compounds 1–2 are listed in Table 2, the relevant hydrogen bond parameters are provided in Table 3.
Results and Discussion
Preparation and General Characterization
The preparation of the compounds 1–2 were carried out with piperidine and the corresponding organic acids in 1:1 ratio in methanolic solution, which was allowed to evaporate at ambient conditions to give the final crystalline products. Both compounds are anhydrous organic salts. The two compounds are not hygroscopic. The molecular structures and their atom labelling schemes for the two structures are illustrated in Figs. 1, 3, respectively. The elemental analysis data for the two crystalline compounds are in good agreement with their compositions. The infrared spectra of the two compounds are consistent with their chemical formulas determined by elemental analysis and further confirmed by X-ray diffraction analysis.
The bands at approximately 3268–3704 cm−1 in the IR spectra of the two compounds arise from the O–H or N–H stretching frequencies. The medium intensity bands in the regions of 1500–1630 and 600–750 cm−1 are attributed to aromatic ring stretching and bending, respectively. The salt 2 exhibits bands for both COO and COOH groups.
Structural Descriptions
X-ray Structure of (Piperidine)2: (Naphthalene-1,5-Disulfonic Acid) [(HL) 2+2 · (nds2−)] (1)
Crystallization of piperidine and naphthalene-1,5-disulfonic acid in a 1:1 ratio from methanol gave single crystals suitable for X-ray diffraction. Structure determination (Table 1) revealed that piperidine and naphthalene-1,5-disulfonic acid are present in a 2:1 ratio in the molecular complex 1. The crystal structure of 1 consists of one protonated L, and half a dianion of naphthalene-1,5-disulfonate in the asymmetric unit (Fig. 1). In the dianion, there exists an inversion centre located at the middle point of the C8–C8A bond. Compound 1 crystallizes in the triclinic space group P-1. The piperidine moiety is protonated at the ring nitrogen. The angle at the protonated ring N atom is 112.06(16)°. The C–N bond lengths in the ring are in the range of 1.489(3)–1.490(2) Å, and the C1–C2, C2–C3, C3–C4, and C4–C5 bond lengths are 1.499(3), 1.515(3), 1.516(3), and 1.507(3) Å, respectively. The piperidine ring adopts the chair conformation. The S–O bond distances in the SO3 − range from 1.4342(16) to 1.4655(15) Å (Δ is 0.0313 Å) which are in the range of the deprotonated SO3 − moiety [34].
The cations and the dianions form 2D sheet extending at the direction that makes an angle of ca. 60° with the ab plane (Fig. 2). Herein one H at the cation (NH2 +) functions as a double hydrogen bond donor with one O atom at the SO3 − with N–O distance of 2.893(2) Å and the S at the same SO3 − with N–S distance of 3.562(2) Å. The other H at the cation (NH2 +) also generates one N–H···O (N(1)–H(1B)···O(2)#3, 2.765(2) Å, 173.4°,#3 −x, −y, −z + 1), and one N–H···S(N(1)–H(1B)···S(1)#3, 3.7999(19) Å, 161.5°) hydrogen bond with the anion. For the presence of the hydrogen bonds there exists the R 21 (3) ring according to Bernstein [35]. There also exists the CH2–O (C(5)–H(5A)···O(1)#2, 143.7°, #2 −x + 1, −y, −z + 1) association at the sheet between the CH2 moiety of the piperidine and the SO3 − with C–O distance of 3.260(3) Å. The 2D sheets were further stacked along the direction that was perpendicular with its extending direction by the CH2–π association between the CH2 of the piperidine and the naphthalene ring of the anion with C–Cg (Cg is the gravity centre of the naphthalene ring) distance of 3.730(2) Å to form 3D layer network structure. Herein the neighboring sheets were slipped some distances from each other along its extending direction.
X-ray Structure of (Piperidine): (4-Nitrophthalic Acid) [(HL) · (Hnpa)] (2)
The compound 2 of the composition [(HL) · (Hnpa)] was also prepared by reacting piperidine and 4-nitro-phthalic acid with 1:1 ratio in a solution of methanol, in which only one proton of the carboxyl units was transferred to the ring N atom of the piperidine. Thus 2 can be classified as a hydrogenphthalate salt. In the asymmetric unit of 2 there existed one cation of piperidine, and one anion of 4-nitro-hydrogenphthalate, as shown in Fig. 3. Salt 2 crystallizes as monoclinic block crystals in the space group P2(1)/c. Structure of 2 is example of a complex with many symmetry-independent components in the asymmetric unit cell (Z > 2). In the compound, there is one pair of ion pair with no solvent molecules. The same as the salt 1, the piperidine ring also adopts the chair conformation.
In the cation, the angle (C(1)–N(1)–C(5)) around the protonated N atom is [112.23(19)°], which is similar to the corresponding angle in 1. The C–N bond lengths in the piperidine ring are in the range of 1.478(3)–1.484(3) Å, and the C1–C2, C2–C3, C3–C4 and C4–C5 bond lengths are 1.498(3), 1.507(3), 1.511(4), and 1.495(3) Å, respectively. The bond lengths at the cation of 2 are similar to the corresponding values at the cation of salt 1 also.
The phenyl ring of the anion with atoms C8–C13 and the piperidine ring with atoms N1–C1–C2–C3–C4–C5 intersects at an angle of 56.4(2)° with each other. The carboxylates O1–C6–O2, and O3–C7–O4 are rotated out of the phenyl plane by 19.1(1)°, and 28.2(1)°, respectively. The two carboxyl moieties intersect at an angle of 34.9(3)° with each other, which is different from the reported adduct of phthalate [36]. The nitro group N6–O5–O6 deviates by 9.9(2)° from the phenyl ring of the anion. The nitro group N6–O5–O6 make angles of 18.9(3)° and 38.1(1)° with the two carboxylates, respectively.
At the cation there is bonded an anion by the bifurcated N–H···O hydrogen bonds between the NH+ and both O of the carboxylate of the anion with N–O distances of 2.932(3)–3.242(3) Å. For the above mentioned bifurcated hydrogen bonds, there generates the R 21 (4) ring motifs between the anion and the cation. 4-nitrohydrogenphthalate is found the intramolecular O–H···O hydrogen bonds between the COOH and the COO− with O–O distance of 2.383(3) Å, giving an S 11 (7) ring motif. The cation bonded with the anion are linked together by the CH2–O (C(1)–H(1D)···O(6)#2, 125.8°; C(2)–H(2A)···O(5)#1, 145.4°) contacts between the CH2 of the cation and the nitro group at the anion with C–O distances of 3.253(3)–3.444(3) Å to form a 1D chain (Fig. 4). The 1D chains are connected together by the O–Cπ contact between the O atom at the carboxylate and the π–C at the carboxyl group with O–C distance of 3.188(3) Å to form 2D sheet extending parallel to the ac plane. Herein, the O–C separation is slightly longer than the archived value of 3.019(4) Å [37]. The 2D sheets are further combined together by the N–H···O hydrogen bond between the NH+ and the C=O at the COOH with N–O distance of 2.854(2) Å, and CH2–O contact (C(5)–H(5B)···O(2), 114.6°) between the CH2 of the cation and the carboxylate group with C–O distance of 3.148(3) Å to form 3D ABAB layer network structure. The geometries of the C(5)–H(5B)···O(2) contact were comparable with those (with D···A distance of 3.154 (3) Å, and <(DHA) of 115.0(3)°) found at the reported compound [38]. In this case the adjacent sheet layers are also slipped some distance from each other along the extending direction.
Supporting Information Available
Crystallographic data for the structural analysis have been deposited with the Cambridge Crystallographic data center, CCDC Nos. 898477 for 1, and 1059210 for 2. Copies of this information may be obtained free of charge from the +44(1223)336-033 or Email: deposit@ccdc.cam.ac.uk or http://www.ccdc.cam.ac.uk.
References
Steiner T (2002) Angew Chem Int Ed 41:48
Ma JC, Dougherty DA (1997) Chem Rev 97:1303
Nishio M (2004) Cryst Eng Commun 6:130
Claessens CG, Stoddart JF (1997) J Phys Org Chem 10:254
Meyer EA, Castellano RK, Diederich F (2003) Angew Chem Int Ed 42:1210
Desiraju GR (1995) Angew Chem Int Ed 34:2311
Whitesides GM, Simanek EE, Mathias JP, Seto CT, Chin D, Mammen M, Gordon DM (1995) Acc Chem Res 28:37 and references therein
Khurram M, Qureshi N, Smith MD (2006) Chem Commun 2006:5006
Shukla R, Lindeman SV, Rathore R (2007) Chem Commun 2007:3717
Wan CQ, Chen XD, Mak TCW (2008) Cryst Eng Commun 10:475
Gould PJ (1986) Int J Pharm 33:201
Shan N, Bond AD, Jones W (2002) Cryst Eng 5:9
Bhogala BR, Basavoju S, Nangia A (2005) CrystEng Commun 7:551
Desiraju GR (1989) Crystal engineering. The design of organic solids. Elsevier, Amsterdam
Leiserowitz L (1976) Acta Crystallogr B 32:775
Kolotuchin SV, Fenlon EE, Wilson SR, Loweth CJ, Zimmerman SC (1995) Angew Chem Int Ed Engl 34:2654
Kuduva SS, Craig DC, Nangia A, Desiraju GR (1999) J Am Chem Soc 121:1936
Pedireddi VR, Prakashareddy J (2002) Tetrahedron Lett 43:4927
Refat MS, Elfalaky A, Elesh E (2011) J Mol Struct 990:217
Metrangolo P, Neukirch H, Pilati T, Resnati G (2005) Acc Chem Res 47:386
Shattock TR, Arora KK, Vishweshwar P, Zaworotko MJ (2008) Cryst Growth Des 8:4533
Biradha K, Mahata G (2005) Cryst Growth Des 5:61
Ma BQ, Coppens P (2003) Chem Commun 2003:504
Beatty AM, Schneider CM, Simpson AE, Zaher JL (2002) Cryst Eng Commun 4:282
Ballabh A, Trivedi DR, Dastidar P, Suresh E (2002) Cryst Eng Commun 4:135
Sowmya NS, Sampathkrishnan S, Sudhahar S, Chakkaravarthi G, Kumar RM (2014) Acta Cryst E70:559
Li ZH, Cheng BW, Mei SK (2008) Acta Cryst E64:o1085
Wang YT, Tang GM, Zhang YC, Wan WZ (2008) Acta Cryst E64:o1753
Smith G, Wermuth DU (2010) Acta Cryst C66:o374
Jin SW, Yan PH, Wang DQ, Xu YJ, Jiang YY, Hu LL (2012) J Mol Struct 1016:55
Jin SW, Zhang WB, Liu L, Gao HF, Wang DQ, Chen RP, Xu XL (2010) J Mol Struct 975:128
Bruker (2004) SMART and SAINT. Madison, Bruker AXS
Sheldrick GM (2000) SHELXTL structure determination software suite, version 6.14. Madison, Bruker AXS
Jin Y (2012) Acta Cryst E68:o90
Bernstein J, Davis RE, Shimoni L, Chang NL (1995) Angew Chem Int Ed 34:1555
Dale SH, Elsegood MRJ, Hemmings M, Wilkinson AL (2004) Cryst Eng Comm 6:207
Eichstaedt K, Olszewska T, Gdaniec M (2013) Acta Cryst E69:o144
Cowan JA, Howard JAK, Mason SA, McIntyre GJ, Lo SMF, Mak T, Chui SSY, Cai JW, Cha JA, Williams ID (2006) Acta Crys C62:o157
Acknowledgments
This research was supported by Zhejiang Provincial Natural Science Foundation of China under Grant No. LY14B010006, the Education Office Foundation of Zhejiang Province under Grant No. Y201017321, the National Training Programs of Innovation and Entrepreneurship of China for Undergraduates under Grant No. 201410341022, the Zhejiang A & F University Science Foundation under Grant No. 2009FK63, and the Open Foundation of Key Laboratory of Chemical Utilization of Forestry Biomass of Zhejiang Province, Zhejiang A & F University under Grant No. 2015CUFB02.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jin, S., Feng, C., Wen, X. et al. Crystal Structures of Two Salts from Piperidine, Naphthalene-1,5-Disulfonic Acid, and 4-Nitrophthalic Acid. J Chem Crystallogr 46, 21–27 (2016). https://doi.org/10.1007/s10870-015-0623-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10870-015-0623-x