Abstract
Diffusion properties are technologically important in the understanding of semiconductors for the efficent formation of defined nanoelectronic devices. In the present study we employ experimental data to show that bulk materials properties (elastic and expansivity data) can be used to describe gold and silver diffusion in germanium for a wide temperature range (702–1177 K). Here we show that the so-called cBΩ model thermodynamic model, which assumes that the defect Gibbs energy is proportional to the isothermal bulk modulus and the mean volume per atom, adequately metallic diffusion in germanium.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Germanium (Ge) was a material of traditional importance to the semiconductor industry, however, in the past decade it rivals silicon (Si) as it has superior materials properties (better carrier mobilities, low dopant activation temperatures and smaller band-gap) [1–5]. This is driven by the advent of high-k gate dielectric materials as these have eliminated the requirement of a good quality native oxide in advanced nanoelectronic devices [6, 7]. Additionally, the recent investigation of diffusion phenomena such as the discovery that boron diffuses slower in Ge than originally thought has also assisted the exploitation of Ge [8].
In Ge metals such as gold (Au) and silver (Ag) diffuse via the dissociative mechanism [9–11]. In the dissociative mechanism, proposed by Frank and Turnbull [11] there is a requirement for vacancies (V) that excange position with the dopant intersititials (D i ) to form a dopant substitutional to a Ge site (D Ge ) via the relation \( D_{i} + V \leftrightarrow D_{Ge} \). The dissociative mechanism requires vacancies and this is compatible to Ge as this is the dominant intrinsic defect [5, 11].
The association of the defect Gibbs energy gi (where i = defect formation f, diffusion activation act, or migration m) with bulk properties in solids is an issue that has led to different models for numerous decades [12–20]. A notable example is the model of Zener [12], which proposed that gi is proportional to the shear modulus of the solid. The more recent model by Varotsos and Alexopoulos [13–19] (the so-called cBΩ model) postulated that gi is proportional to the isothermal bulk modulus B and the mean volume per atom Ω.
In previous studies [21–31] the cBΩ model was used to investigate the point defect processes in a wide range of crystalline materials, but was not employed to describe Au and Ag diffusion processes in Ge. In the present study we use the cBΩ to model Au and Ag diffusion in Ge by using isothermal bulk modulus and the mean volume per atom in a wide temperature range (827–1176 K).
2 Methodology
2.1 Point defect parameters
Formally, the defect formation parameters in a system can be defined by comparing a real (i.e. defective) crystal to an isobaric ideal (non-defective) crystal [21, 24]. In this framework, the isobaric parameters are defined in terms of the corresponding Gibbs energy (gf) as [21, 24]:
where P is the pressure, T is the temperature; sf, hf and υf represent the defect formation entropy, enthalpy and volume respectively.
Typically, in a monoatomic crystal with a single diffusion mechanism, the diffusion process can be described by the activation Gibbs energy (gact). This is the sum of the Gibbs formation (gf) and the Gibbs migration (gm) processes. The activation entropy sact and the activation enthalpy \( {\text{h}}^{\text{act}} \) can be defined by [21, 24]:
The diffusivity D can be defined by [21, 24]:
where f depends upon the diffusion mechanism and structure, a 0 is the lattice parameter, \( \nu \) is the attempt frequency and k B is Boltzmann’s constant.
2.2 The cBΩ model
The key of the cBΩ model is the description of the defect Gibbs energy gi in terms of the bulk properties of the solid [21, 24]:
where ci is dimensionless.
where β is the thermal (volume) expansion coefficient.
In principle, employing Eqs. (6) and (7) the diffusivity can be calculated in the framework of the cBΩ model at any temperature and pressure from a single experimental measurement using:
Considering a single experimental measurement for a diffusivity D 1 value at a temperature T1 the \( c^{act} \) can be calculated if we assume that the pre-exponential factor \( fa_{0}^{2} \nu \) can be estimated. Then by using this \( c^{act} \) and Eq. 11 the diffusivity D 2 at any temperature T2 can be calculated if we have the elastic data and expansivity data for T2. In previous studies it was proposed that \( c^{act} \) is a constant that is independent of temperature and pressure to least to a first approximation [21, 24]. Finally, for constant temperature the D can be studied at any pressure (refer for example to [32–34]).
3 Results and discussion
3.1 Background
The intrinsic point defects are the main vehicles that facilitate self- and dopant diffusion in materials. The control of diffusion processes is necessary to achieve well defined regions in devices. Regarding self-diffusion in Ge it has been previously established that it is mediated by vacancies [35–38]. Consequent studies revealed that the diffusion of most dopants in Ge is mediated by vacancies [39–42]. Exceptions include copper (Cu), palladium (Pd), Au and Ag (refer to [43] and references therein). Au [10, 44–46] and Ag [47, 48] diffusion in Ge has been investigated for about six decades. Figure 1 represents the dissociative mechanism (Frank and Turnbull mechanism [11]) that is the mechanism for Au and Ag transport in Ge [10]. In this the dopant interstitial migrates until it recombines with a vacancy to form a dopant substitutional (via \( D_{i} + V \leftrightarrow D_{Ge} \)).
The Au and Ag diffusion in the temperature range 873 K to 1193 K can be described through the Arrhenius following relations [10]:
3.2 Au and Ag diffusion in Ge
In the present study we used the expansivity data of Kagaya et al. [49] and the isothermal bulk modulus data of Krishnan et al. [50]. As the single experimental measurement method may lead to errors other ways have been previously used to calculate \( c^{act} \) including the compensation law and the “mean value” method [21, 24, 26, 51, 52].
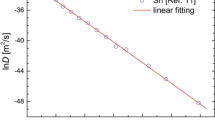
In the present study we used the mean value method because we wanted to limit the dependence of \( c^{act} \) on experimental uncertainties in the determination of a diffusivities [10], the expansivity [49] and isothermal bulk modulus [50]. In the mean value method a linear behavior of \( lnD^{{}} \) with respect to \( \frac{{B\Omega }}{{k_{B} T}} \) testifies the applicability of the cBΩ model, whereas from the slope \( c^{act} \) can be derived (refer to Eq. 11). Figure 2 reports the experimental [10] Au and Ag diffusion coefficients in Ge with respect to \( \frac{{B\Omega }}{{k_{B} T}} \) verifying in essence that the relations are linear and can be represented by the following relations
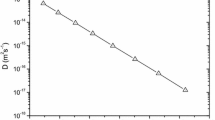
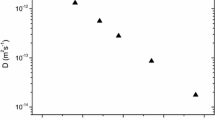
Relations 14 and 15 can be used to calculate the other Au and Ag diffusivity values with respect to temperature. The only requirement is to have the corresponding elastic and expansivity data. As it can be observed from Table 1 the cBΩ model is in excellent agreement with experiment [10], as the experimental and calculated Au and Ag diffusion coefficients in Ge vary by less than 2 %. Figure 3 is the Arrhenius plot for Au and Ag diffusion coefficients obtained by calculated by the cBΩ model. Interestingly, the cBΩ model is in excellent agreement with experiment in a diffusion mechanism that is non-trivial. As it has been previously discussed [21, 24] the cBΩ model is appropriate when a single-diffusion mechanism is operating. Although Au and Ag diffusion in characterized by a single diffusion mechanism dissociative diffusion can be complicated as it requires both vacancies and interstitials, whereas the steps are not as well defined as in other mechanisms (for example the ring-mechanism for vacancy diffusion in Ge [43]).
The experimental [10] Au and Ag diffusion coefficients in Ge with respect to \( \frac{{B\Omega }}{{k_{B} T}} \)
4 Conclusions
Point defect processes in materials can be linked to the bulk properties through the cBΩ model. This in turn can be beneficial to numerous issues including novel nanoelectronic devices. In the present study, the efficacy of the cBΩ model has been tested to model diffusivity via the Frank and Turnbull mechanism. There is an excellent agreement between the calculated and experimental Au and Ag diffusivities in the temperature range considered. It is therefore evident that the Au and Ag dissociative mechanism in Ge can described with the cBΩ model. This in turn implies that the cBΩ model should be tested in systems with complicated diffusion mechanisms and defect processes [53–59].
References
C. Claeys, E. Simoen, Germanium-Based Technologies: From Materials to Devices (Elsevier, Amsterdam, 2007)
A. Chroneos, J. Appl. Phys. 105, 056101 (2009)
G. Impellizzeri, S. Boninelli, F. Priolo, E. Napolitani, C. Spinella, A. Chroneos, H. Bracht, J. Appl. Phys. 109, 113527 (2011)
A. Chroneos, J. Mater. Sci.: Mater. Electron. 24, 1741 (2013)
A. Chroneos, H. Bracht, Appl. Phys. Rev. 1, 011301 (2014)
A. Ritenour, A. Khakifirooz, D.A. Antoniadis, R.Z. Lei, W. Tsai, A. Dimoulas, G. Mavrou, Y. Panayiotatos, Appl. Phys. Lett. 88, 132107 (2006)
A. Chroneos, U. Schwingenschlögl, A. Dimoulas, Ann. Phys. (Berlin) 524, 123 (2012)
S. Uppal, A.F.W. Willoughby, J.M. Bonar, N.E.B. Cowern, T. Grasby, R.J.H. Morris, M.G. Dowsett, J. Appl. Phys. 96, 1376 (2004)
N.A. Stolwijk, W. Frank, J. Hölzl, S.J. Pearton, E.E. Haller, J. Appl. Phys. 57, 5211 (1985)
H. Bracht, N.A. Stolwijk, E. Mehrer, Phys. Rev. B 43, 14465 (1991)
H. Mehrer, Diffusion in Solids (Springer, New York, 2007)
C. Zener, J. Appl. Phys. 22, 372 (1951)
P. Varotsos, K. Alexopoulos, Phys. Rev. B 15, 4111 (1977)
P. Varotsos, K. Alexopoulos, Phys. Rev. B 15, 2348 (1977)
P. Varotsos, K. Alexopoulos, J. Phys. (Paris) Lett. 38, L455 (1977)
P. Varotsos, W. Ludwig, K. Alexopoulos, Phys. Rev. B 18, 2683 (1978)
P. Varotsos, K. Alexopoulos, Phys. Rev. B 22, 3130 (1980)
P. Varotsos, K. Alexopoulos, Phys. Rev. B 24, 904 (1981)
P. Varotsos, K. Alexopoulos, Phys. Rev. B 30, 7305 (1984)
J. Philibert, Defect Diffus. Forum 249, 61 (2006)
P. Varotsos, K. Alexopoulos, Thermodynamics of Point Defects and their Relation with the Bulk Properties (North-Holland, Amsterdam, 1986)
P. Varotsos, N. Sarlis, M. Lazaridou, Phys. Rev. B 59, 24 (1999)
P. Varotsos, Phys. Rev. B 75, 172107 (2007)
P. Varotsos, J. Appl. Phys. 101, 123503 (2007)
H.B. Su, D.O. Welch, W. Wong-Ng, L.P. Cook, Z. Yang, Appl. Phys. Lett. 91, 172510 (2007)
B.H. Zhang, X.P. Wu, Appl. Phys. Lett. 100, 051901 (2012)
I. Sakelis, J. Appl. Phys. 112, 013504 (2012)
B.H. Zhang, AIP Adv. 4, 017128 (2014)
F. Vallianatos, V. Saltas, Phys. Chem. Miner. 41, 181 (2014)
E.S. Skordas, Solid State Ion. 261, 26 (2014)
P. Varotsos, K. Eftaxias, V. Hadjicontis, Phys. Rev. B 38, 6328 (1988)
V. Saltas, F. Vallianatos, Mater. Chem. Phys. 163, 507 (2015)
M.W.D. Cooper, R.W. Grimes, M.E. Fitzpatrick, A. Chroneos, Solid State Ion. 282, 26 (2015)
V. Saltas, A. Chroneos, F. Vallianatos, RSC Adv. 6, 53324 (2016)
M. Werner, H. Mehrer, H.D. Hochheimer, Phys. Rev. B 32, 3930 (1985)
A. Chroneos, D. Skarlatos, C. Tsamis, A. Christofi, D.S. McPhail, R. Hung, Mater. Sci. Semicond. Process. 9, 640 (2006)
E. Hüger, U. Tietze, D. Lott, H. Bracht, D. Bougeard, E.E. Haller, H. Schmidt, Appl. Phys. Lett. 93, 162104 (2008)
A. Chroneos, H. Bracht, R.W. Grimes, B.P. Uberuaga, Appl. Phys. Lett. 92, 172103 (2008)
S. Brotzmann, H. Bracht, J. Appl. Phys. 103, 033508 (2008)
A. Chroneos, R.W. Grimes, B.P. Uberuaga, H. Bracht, Phys. Rev. B 77, 235208 (2008)
R. Kube, H. Bracht, A. Chroneos, M. Posselt, B. Schmidt, J. Appl. Phys. 106, 063534 (2009)
H. Tahini, A. Chroneos, R.W. Grimes, U. Schwingenschlögl, H. Bracht, Appl. Phys. Lett. 99, 072112 (2011)
H.A. Tahini, A. Chroneos, S.C. Middleburgh, U. Schwingenschlögl, R.W. Grimes, J. Mater. Chem. A 3, 3832 (2015)
W.C. Dunlop Jr., Phys. Rev. 97, 614 (1955)
M.F. Millea, J. Phys. Chem. Solids 27, 309 (1966)
A. Almazouzi, J. Bernardini, E.G. Moya, H. Bracht, N.A. Stolwijk, H. Mehrer, J. Appl. Phys. 70, 1345 (1991)
L.Y. Wei, J. Phys. Chem. Solids 18, 162 (1961)
V.E. Kosenko, Sov. Phys. Solid State (Engl. Transl.) 4, 42 (1962)
H.M. Kagaya, N. Shoji, T. Soma, Phys. Status Solidi B 139, 417 (1987)
R. Krishman, R. Srinivasan, S. Deverayanan, In Thermal Expansion of Crystals (Pergamon Press, Oxford, 1979)
B.H. Zhang, X.P. Wu, J.S. Xu, R.L. Zhou, J. Appl. Phys. 108, 053505 (2010)
A. Chroneos, R.V. Vovk, Solid State Ion. 274, 1 (2015)
L.I. Murin, J.L. Lindstrom, B.G. Svensson, V.P. Markevich, A.R. Peaker, C.A. Londos, Solid State Phenom. 108–109, 267 (2005)
S.T. Murphy, A. Chroneos, C. Jiang, U. Schwingenschlögl, R.W. Grimes, Phys. Rev. B 82, 073201 (2010)
A. Chroneos, C.A. Londos, E.N. Sgourou, J. Appl. Phys. 110, 093507 (2011)
I.D. Seymour, A. Chroneos, J.A. Kilner, R.W. Grimes, Phys. Chem. Chem. Phys. 13, 15305 (2011)
D. Rupasov, A. Chroneos, D. Parfitt, J.A. Kilner, R.W. Grimes, S.Ya. Istomin, E.V. Antipov, Phys. Rev. B 79, 172102 (2009)
I.D. Seymour, A. Tarancon, A. Chroneos, D. Parfitt, J.A. Kilner, R.W. Grimes, Solid State Ion. 216, 41 (2012)
M.J.D. Rushton, A. Chroneos, Sci. Rep. 4, 6068 (2014)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Panayiotatos, Y., Vovk, R.V. & Chroneos, A. Gold and silver diffusion in germanium: a thermodynamic approach. J Mater Sci: Mater Electron 28, 1966–1970 (2017). https://doi.org/10.1007/s10854-016-5750-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10854-016-5750-4