Abstract
The monarch butterfly (Danaus plexippus) and golden-winged warbler (Vermivora chrysoptera) are two disturbance-dependent, migratory species that have both suffered from breeding habitat loss and degradation. Given the overlap in distribution and ecological needs, efforts to manage golden-winged warbler habitat may also benefit monarchs. In 2021, we surveyed monarchs, milkweed (Asclepias spp.), nectar resources, and structural vegetation characteristics at 49 sites managed for golden-winged warblers (“GWWA sites”) in the western Great Lakes region. We compared our observations to data from nearby sites managed specifically for monarchs (“reference sites”) and assessed the influence of site and landscape characteristics on monarch (adult/immature) and milkweed presence within GWWA sites. Immature monarchs, adult monarchs, and milkweed were 22x, 5x, and 110x more abundant at reference sites compared to GWWA sites, respectively. At GWWA sites, the presence of immature monarchs was positively associated with floral abundance and the presence of immature monarchs and milkweed presence were both positively associated with proportion of emergent herbaceous wetland cover within 500 m and 1 km. Additionally at GWWA sites, immature monarch density increased with milkweed density. Lastly, the presence of adult monarchs was positively associated with size of treatment area.
Implications for insect conservation: Our findings suggest that sites managed as golden-winged warbler habitat can contribute to monarch habitat goals. This will best be done by conducting conservation practices that increase milkweed and floral abundance (e.g., via supplemental plantings) and targeting sites with more semi-natural herbaceous cover (e.g., pastures, herbaceous wetlands), while still meeting golden-winged warbler land cover requirements.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The abundance and quality of early-successional communities in the eastern United States have declined substantially during the past several decades due to land conversion, habitat degradation, and the suppression of natural disturbance regimes (King and Schlossberg 2014; Fogarty et al. 2020). For example, woody stem encroachment has increased by 2.3% per year between 1984 and 2018 in central Nebraska (Filippelli et al. 2020), reducing high-quality grasslands, while the area of young forest and shrubland has decreased by 2.4% and 1.7% per year (1900–2000) in the Northeast and Midwest, respectively (King and Schlossberg 2014). Consequently, many native wildlife species dependent upon these communities have declined as well (Askins 2001; Rosenberg et al. 2019). Early-successional communities were once maintained by large ungulates browsing woody vegetation (e.g., Bison [Bison bison]), flooding caused by beaver dams (Castor canadensis), woody vegetation die back post wildfire and anthropogenic fire (e.g., Native Americans), and high wind events creating canopy gaps (McClain et al. 2021). Since European settlement, natural disturbances have largely been suppressed by prevention of wildfire, flooding control, and extirpation of large ungulates (Mueller et al. 2021) and beavers (Wohl 2021). As a result, active management that creates and maintains early-successional communities has become a conservation priority for many wildlife species (Litvaitis et al. 2021).
In light of widespread suppression of natural disturbances, management of early-successional communities has become necessary for reliant wildlife species, though efforts are often limited due to various challenges (Lutter et al. 2019). These challenges include, but are not limited to, lack of funding and resources (e.g., competitive grant funds; Litvaitis et al. 2021), logistics (e.g., weather conditions), and negative public perception (Lutter et al. 2019). For example, lack of funding and logistical challenges are often reported as barriers for not using prescribed fire to the extent it is needed (Hunter et al. 2020). Additionally, early-successional communities are ephemeral (i.e., the structural openness of these communities diminishes overtime due to natural succession; King and Schlossberg 2014), requiring frequently implemented disturbances to maintain a balance of this young age class on the landscape (Litvaitis et al. 2021). For instance, regenerating timber harvests maintain early-successional conditions for approximately 10–15 years (DeGraaf and Yamasaki 2003) and recommended prescribed fire frequency to preserve early-successional conditions in some communities is about four fires per decade (e.g., oak savannas in the upper Midwest; Peterson and Reich 2001). Due to these challenges associated with managing early-successional communities, aligning management for multiple objectives (e.g., focal species) could make more efficient use of conservation resources.
The golden-winged warbler (Vermivora chrysoptera) requires early-successional habitat within heavily forested landscapes across eastern North America (Confer et al. 2020). Basic breeding habitat needs for the golden-winged warbler include young forests or young shrublands within a deciduous forest-dominated landscape (McNeil et al. 2020) and include habitat features like widely spaced overstory trees (Roth et al. 2014), high stem density (Martin et al. 2007; Buckhardt Thomas et al. 2023), and interspersed patches of herbaceous vegetation (Klaus and Buehler 2001; Bulluck and Buehller 2008; Bakermans et al. 2015a). This at-risk migratory songbird (Rosenberg et al. 2016; Confer et al. 2020) is currently being considered for listing under the U.S. Endangered Species Act (Sewell 2010). Because of this, several habitat-based conservation programs for the golden-winged warbler have been employed through the United States Department of Agriculture (USDA) Natural Resources Conservation Service (NRCS). These programs provide financial and technical assistance to private landowners in portions of the Appalachian Mountain and upper Great Lakes regions (Litvaitis et al. 2021). Primary management activities employed by these private landowners include timber harvesting and shrub-shearing (Buckhardt Thomas et al. 2023), activities designed to leave widely-spaced overstory trees intermixed with patches of shrubs/saplings and herbaceous forbs/grasses (Roth et al. 2019). In addition to golden-winged warblers, these management actions have been shown to benefit a variety of other species associated with early-successional communities (Bakermans et al. 2015b; Mathis et al. 2021).
One of these species includes the monarch butterfly (Danaus plexippus), which also relies on early-successional communities that host nectar-rich plants and their obligate larval host plant, milkweed (Asclepias spp.; Oberhauser and Solensky 2004). Like the golden-winged warbler, the monarch has declined drastically due to habitat loss and degradation (Pleasants and Oberhauser 2013; Thogmartin et al. 2017b). The eastern migratory population of monarchs (Danaus plexippus plexippus) has declined by approximately 80% during the last two decades (Semmens et al. 2016) and listing under the U.S. Endangered Species Act in 2020 was found to be “warranted but precluded” (Federal Register 2020). Because abundant floral and ovipositing resources are pillars of monarch habitat (Dinsmore et al. 2019; Lukens et al. 2020) and golden-winged warblers selectively place nests within structurally open areas with high levels of sunlight and shade-intolerant herbaceous vegetation cover (common species: goldenrod [Solidago spp.], fleabane [Erigeron spp.], blackberry [Rubus spp.]; Confer 1992), best management practices (BMPs) for these two species may align. Indeed, monarchs have benefited from habitat management for other species with similar habitat associations, such as, herbaceous plantings for the greater sage-grouse (Centrocercus urophasianus) in Wyoming (Dumroese et al. 2016) and prescribed fire and thinning treatments for the red-cockaded woodpecker (Leuconotopicus borealis) in Arkansas (Rudolph et al. 2006). Associations between monarchs (abundance or presence) and habitat characteristics within a forest-dominated landscape are nonexistent within peer-reviewed literature, despite growing recognition of forests playing an important role for pollinator diversity (Ulyshen et al. 2023). Additionally, forested communities have not been included within analyses addressing where milkweed restoration should be prioritized (Thogmartin et al. 2017a) nor region-wide monitoring efforts (Kasten et al. 2016; Lukens et al. 2020). State-led planning efforts, like the Mid-America Monarch Conservation Strategy, suggest that “open forest and brushy areas” provide habitat for monarchs, but offer very little additional information (Midwest Association of Fish and Wildlife Agencies 2018).
Efforts to better understand monarch response to management of woody-dominated early-successional communities are warranted given that monarchs have been observed within this community type (Dinsmore et al. 2019; Lee et al. 2021) and management of this community type supports dense pollinator populations (Mathis et al. 2021). In the western Great Lake states, NRCS conservation programs of two at-risk wildlife species overlap. The golden-winged warbler is a focal species for NRCS’ Regional Conservation Partnership Program, and the monarch is a focal species for NRCS’ Working Lands for Wildlife partnership. The purpose of this study was to assess how habitat management through the NRCS-Regional Conservation Partnership Program effort for golden-winged warblers can augment habitat goals for NRCS-Working Lands for Wildlife partnership for monarchs. By 2050, conservation planners recommend that 1,190,000 ha of golden-winged warbler breeding habitat should be created on private and public lands in the Great Lakes Conservation Region (Roth et al. 2019). The ambitious acreage goal for golden-winged warblers has the potential to contribute to monarch conservation as well. The objectives of this project were to: (1) compare adult monarch abundance, immature monarch densities, and milkweed densities between woody-dominated early-successional communities managed for golden-winged warblers (hereafter “GWWA sites”) and herbaceous-dominated sites managed specifically for monarchs (hereafter “reference sites”), and (2) identify within-site and landscape habitat characteristics of only GWWA sites that are associated with monarch (adult and immature) and milkweed presence. We discuss our results with respect to their implications for how existing golden-winged warbler BMPs can simultaneously benefit the monarch.
Methods
Study area
We studied monarch and milkweed responses to two habitat treatment types for the golden-winged warbler in the western Great Lakes region of northern Minnesota and Wisconsin, including shrub-shearing and regenerating timber harvests (Roth et al. 2019). We focused on these two treatment types because they are most commonly implemented for golden-winged warbler habitat creation within the Great Lakes (Buckhardt Thomas et al. 2023). This region is especially notable for golden-winged warbler conservation because it hosts 95% of the species’ breeding population (Roth et al. 2019), likely due to abundant shrub-wetlands and active management of aspen (Populus spp.) timber stands (Peterson et al. 2016). The desired goal of shrub-shearing is to create structural complexity (i.e., patchiness of regenerating shrubs and herbaceous vegetation) within homogenous stands of mature and dense alder-dominated wetlands (Buckhardt Thomas et al. 2023). This is done by brush hogging/mowing strips or blocks (approximately 50%) within this vegetation community during the winter (Golden-winged Warbler Working Group 2019). Regenerative timber harvesting is a silvicultural tool (e.g., clearcut with residuals) similarly used to create structural complexity within upland deciduous forests. Desired conditions post-harvest are the retention of few large overstory trees, dense sapling and shrub layer, and patches of herbaceous vegetation (Bakermans et al. 2015a; Golden-winged Warbler Working Group 2019). For both treatment types it is recommended that 12–37 canopy trees/ha are retained and that sites within heavily-forested landscapes (> 50% forest cover containing at least 70% deciduous forest within 2.4 km; Golden-winged Warbler Working Group 2019) are prioritized for management. Dominant woody species at our shrub-sheared sites included alder (Alnus spp.), willow (Salix spp.), and tamarack (Larix laricina), whereas regenerating timber harvested sites were dominated by aspen, oak (Quercus spp.), and red maple (Acer rubrum).
Selection of GWWA sites
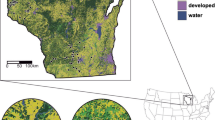
GWWA sites (n = 49) were selected using the following criteria: (1) majority of sites enrolled within NRCS-Regional Conservation Partnership Program for golden-winged warbler management (enrolled = 32, not enrolled = 17), (2) half treated with shrub-shearing (n = 24) and half treated with a regenerating timber harvest (n = 25), (3) treatment conducted 1–5 years prior to monitoring, (4) treated area ≥ 2.6 ha (this is the minimum area needed to fit a 1 ha monitoring plot with a 30 m buffer, see below), and (5) sites of the same treatment type were at least 700 m apart (this distance was chosen because it is greater than the average daily travel distance by breeding-season monarch adults [500 m]; Fisher and Bradbury 2021). In only one instance, two sites of different treatment types were < 700 m apart (413 m). GWWA sites selected for this study ranged in treated area from 2.6 to 52.2 ha (n = 49, mean = 10.1 ha, median = 7.6 ha, SD = 8.6 ha). Site locations were within 45–47 ̊ N and 89–94 ̊ W, at elevations 209–483 m (mean = 378 m), and the majority of sites (94%) were within two ecoregions (Northern Minnesota Wetlands and Northern Lakes and Forests; Fig. 1). Most GWWA sites were privately owned (n = 39, public: n = 10). Other funding programs for private sites included U.S. Fish and Wildlife Service (USFWS) Partners for Fish and Wildlife Program (n = 2), NRCS’s Environmental Quality Incentives Program (n = 3), and USFWS Great Lakes Restoration Initiative (n = 2). Public sites were located on lands managed by the state of Minnesota (Cloquet Area Forest), Minnesota county offices (Carlton, Aikin, Saint Louis), or Wisconsin county offices (Douglas).
A) Distribution of sites throughout northern Minnesota and Wisconsin where monarchs and milkweed were surveyed. Two habitat treatment types for the golden-winged warbler, shrub-shearing (“Alder sites”, n = 24, white circles) and a regenerating timber harvest (“Upland sites”, n = 25, gray circles), were investigated from June-August 2021. Reference sites (black circles) were surveyed in 2020 and 2021 by Monarch Joint Venture Integrated Monarch Monitoring Program (IMMP) participants. B) Representative photo of a site treated with shrub-shearing and C) site treated with a regenerating timber harvest. Note: locations of private sites are randomly shifted by ~ 20 km to protect landowner privacy. The golden-winged warbler breeding range map was acquired from USGS (2018) and the eastern monarch butterfly breeding range map was acquired from USFWS (2022)
Selection of reference sites
We acquired reference site data from the Monarch Joint Venture’s Integrated Monarch Monitoring Program database (hereafter “IMMP”; MJV 2020) (see Supplementary Information for selection criteria). The database includes counts of adult and immature monarchs, habitat variables (e.g., blooming plant and milkweed species richness, milkweed density), and sampling variables (e.g., temperature, wind) for each site visit. We used survey data from 2020 to 2021 to achieve a sufficient sample size while minimizing temporal differences with sampling timing of GWWA sites. We used a Mann-Whitney U test, due to non-normality, to assess whether immature monarch density, adult monarch abundance, and milkweed density were different between 2020 and 2021 reference sites (see Results section).
We created criteria by which to filter reference sites to ensure that they were comparable with GWWA sites (see Supplementary Information). As a result, 22 and 15 unique reference sites were identified from 2020 to 2021, respectively. We removed eight sites from the 2020 dataset since they were sampled in both years to avoid over-representing them. In total, we used data from 29 unique reference sites (2020 = 14 and 2021 = 15) to compare with GWWA sites. Almost all reference sites (n = 28, 97%) were located in the North Central Hardwood Forests ecoregion (not the same as GWWA sites), within latitudes 44–46 ̊N and longitudes 89–95 ̊W, and at elevations of 212–422 m (mean = 334 m; Fig. 1).
Study design
At each GWWA site, we delineated a 1 ha monitoring plot (hereafter “plot”) via ArcGIS 10.7 (ESRI 2018), within which all surveys were conducted. In accordance with the IMMP, each site’s plot shape was rank selected from three options: rectangle (50 m x 200 m, 1st preferred shape), square (100 m x 100 m, 2nd preferred shape) and irregular (variable dimensions, 3rd preferred shape). To minimize edge effects, we created a 30 m buffer extending inside the boundary of the golden-winged warbler managed area. Next, we systematically decided which plot shape to use and where it should be placed based on the size and shape of buffered managed area. A rectangle plot (n = 28) was selected if the plot fit within the buffered treatment area and was randomly placed within the managed area when possible. A square plot (n = 5) was selected if a rectangle plot would not fit within the buffered managed area, and an irregular plot (n = 17) was used if neither a square nor rectangle were viable options. Each site was visited three times in 2021, once during the months of June (2–30 June), July (1–29 July), and August (30 July – 26 Aug).
Data Collection
Monarch and milkweed survey
Upon arrival to a site, we completed a 25 min (rate of 1 m/3 sec) timed visual survey of adult monarchs along the 500 m perimeter of each plot (IMMP Activity 3: Adult Monarch Survey; MJV 2020). We marked the perimeter approximately every 5–10 m with brightly colored flagging tape to make the transect easily visible during each visit. We recorded the behavior(s) (nectaring, oviposting, flying, resting) and substrates (e.g., plant species) for all adult monarch observations. Visual surveys were conducted during appropriate time (10:00–17:00) and weather conditions (≥ 15.6 ℃, no rain, low winds [Beaufort wind code ≤ 5]; MJV 2020). While conducting the adult monarch survey, we also visually searched for milkweed (part of IMMP Activity 1: Milkweed and Blooming Plant Survey, MJV 2020). If milkweed was present within five meters of the transect, we recorded the species, number of plants, number of stems, and stopped the adult monarch survey timer to search the entire plant for immature monarchs (eggs and larvae; IMMP Activity 2: Monarch Egg and Larva Survey; MJV 2020). If milkweeds were detected beyond five meters of the transect, we recorded the same data but did not search them for immature monarchs.
Blooming plant survey
A blooming plant survey was conducted to quantify floral resource availability during each visit. Multiple transects were established within the plot following part of the IMMP’s Activity 1: Milkweed and Blooming Plant Survey protocol (MJV 2020). For rectangle-shaped plots, we created three transects (200 m, 200 m, 100 m, spaced 50 m apart). For square-shaped plots, we created five transects (all 100 m, spaced 25 m apart). For irregular-shaped plots, we established multiple transects of different lengths, spaced 25 m apart (MJV 2020). Regardless of plot shape, the total transect length at all sites was 500 m (MJV 2020). To easily follow transects during each visit, the blooming plant transects were marked every 5–10 m with different colored flagging tape. Due to the number of technicians available to help during each site visit, a 1 m2 subplot was placed every five or ten meters along each transect, for a total of 50 or 100 subplots. To reduce vegetation disturbance resulting from visiting each site three times, we offset the subplot by one meter from the transect during each subsequent visit. Within each subplot, we counted the number of stems with flowers and the number of individual flowers on each stem. We counted individual stems and flowers if < 20. If there were > 20 stems, we estimated the number of stems to the nearest 10 and if there were > 20 flowers/stem, we estimated the number of flowers to the nearest 10 (Mathis et al. 2022). A similar protocol was employed for inflorescences that contained multiple florets (e.g., Solidago spp.).
Structural vegetation survey
We conducted a structural vegetation survey during the second visit to each GWWA site. Structural vegetation surveys were designed to characterize aspects of vegetation structure at each site as well as basal area. We used an ocular tube to quantify percent occurrence of 12 vegetation strata at 25 stops/site, each stop being spaced 20 m apart along the blooming plant transects (James and Shugart 1970). The 12 vegetation strata are defined as: canopy (living tree with DBH ≥ 10 cm), sapling (young [< 10 cm DBH] tree and generally has one main stem), shrub (woody and generally with multiple stems that branch at/below the soil), Rubus spp. (any member of genus Rubus), forb (broad-leaved, herbaceous flowering plants), fern (any seedless vascular plant), coarse woody debris (any woody debris ≥ 10 cm diameter), grass (narrow-leaved sedge, grass, or rush), leaf litter (dead vegetation < 10 cm diameter), and bare ground. Saplings and shrubs were further characterized according to their height and classified as either short (< 1 m) or tall (> 1 m). At each stop, we recorded those vegetation strata that intersected with the ocular tube crosshairs (James and Shugart 1970). Percent occurrence was calculated for each of the 12 vegetation strata categories by dividing the number of intersections by the number of stops (25) and multiplying by 100. We also used a 10 basal area factor prism at the corners and center of the plot (n = 5) to estimate stand level basal area per hectare. Lastly, we also quantified the percent cover of trees, shrubs, forbs, and grass within each plot using the IMMP’s Site Description Survey (MJV 2020) using the following 5 bins (0%, 1–10%, 11–25%, 26–50%, > 50%).
Landscape variables
We calculated the percent area of six land cover categories adjacent to GWWA sites, extracted from the 2019 National Land Cover Database (30 m resolution; Homer et al. 2012), to assess if the presence of monarchs and milkweed was associated with aspects of the surrounding landscape. Land cover categories included three woody cover types (deciduous forest, mixed forest, and woody wetlands) and three semi-natural herbaceous cover types (grassland/herbaceous, pasture/hay, and emergent herbaceous wetlands), all of which are dominant cover types within our Great Lakes study area. We used the extract function within the raster package in R (Hijmans 2021) to extract land cover pixels, which were then converted to percent cover for each of six spatial extents (100 m, 200 m, 500 m, 1 km, 5 km, 10 km) from the centroid of the plot. These extents were chosen based on previous studies that have examined the influence of landscape context on monarchs and other butterflies (Davis et al. 2007; Dinsmore et al. 2019; Bruce et al. 2021).
Statistical analyses
Immature monarch density was calculated by multiplying the number of eggs and larvae by the area searched. Similarly, milkweed density (milkweed stems/ha) was calculated by multiplying the number of milkweed stems divided by the area searched and log-transformed (ln[milkweed stems/ha + 1]) due to non-normality. All response variables (immature monarch density, adult monarch counts, and milkweed density) were averaged for each site. Floral frequency refers to the proportion of subplots in which a blooming plant was present. Floral richness refers to the number of unique blooming plant species present. Floral abundance refers to the number of flowers observed per area sampled during a single visit (# flowers/m2). For only GWWA sites, floral abundance was log-transformed (ln[flowers/m2 + 1]) due to non-normality (floral abundance was not measured at reference sites). For comparisons between reference and GWWA sites, we converted the vegetation 5-bin categorical variable into a continuous variable by selecting the lowest bin value (0% = 0, 1–10% = 1, 11–25% = 11, 26–50% = 26, > 50% = 50). Floral frequency, floral richness, and floral abundance were averaged for each site.
Comparison of reference and GWWA sites
We used a Mann-Whitney U test with the wilcox.test function in the R stats package (R Core Team 2021; due to non-normality) to compare averages of immature monarch densities, adult monarch counts, and milkweed densities between reference and GWWA sites (all run separately). Additionally, to determine which within-site variables were different between GWWA and reference sites, we used a Mann-Whitney U test to compare averages of floral frequency, floral richness, and percent cover of trees, shrubs, forbs, and grass (all run separately).
GWWA site habitat associations
At GWWA sites, we used Spearman’s rank correlation (cor.test function, stats package) to assess the strength of the relationship with (1) per-plant density of immature monarchs and site-specific milkweed density, (2) absolute density of immature monarchs (immatures/ha) and site-specific milkweed density, and (3) adult monarch abundance and site-specific milkweed density. We used logistic regression models to identify within-site and landscape variables that best predicted the presence of immature monarchs, adult monarchs, and milkweed (response variables). Single variable, logistic regression models were created using the glm function in the R package stats. All continuous variables were scaled to have a mean of 0 and standard deviation of 1 using the R scale function.
We created five model sets (see Table 1): (1) treatment type (only one covariate), (2) patch characteristics (years post treatment, treatment area [ha]), (3) floral characteristics (average floral frequency, average floral abundance, average floral richness), (4) structural vegetation characteristics (stand basal area [m2/ha], canopy, tall saplings, short saplings, tall shrubs, short shrubs, Rubus spp., forb, fern, grass, coarse woody debris, leaf litter, bare ground), and (5) landscape characteristics (elevation [m], latitude, longitude, percent cover for six land cover types [grassland/herbaceous, pasture/hay, emergent herbaceous wetland, woody wetland, deciduous forest, mixed forest] at multiple spatial scales [100 m, 200 m, 500 m, 1 km, 5 km, 10 km]).
Within each model set we created univariate models for each explanatory variable (see Table 1) and a null (intercept-only) model. We used Akaike’s Information Criterion adjusted for small sample size (AICc; Burnham and Anderson 2015) to rank and assess models. We considered models to be predictive of presence (monarchs [adult or immature] or milkweed) if AICc values were at least 2.00 AICc values lower than that of the null model and ꞵ 85% confidence intervals did not include zero (Arnold 2010; Burnham and Anderson 2015). This predictive ability was further assessed by calculating the Brier’s score (BrierScore function, DescTools package; Signorell 2021) and area under the curve (AUC) for a receiver operating characteristic curve (roc function, pROC package; Robin et al. 2011). Brier’s score, which is a value from 0 to 1, is interpreted as 0 representing perfect accuracy and 1 representing complete inaccuracy. Area under the curve is also a value between 0 and 1, is interpreted with values above 0.5 indicating increasing ability of the model to predict a good fit. For models within the landscape characteristics set, we tested for spatial autocorrelation (Zuckerberg et al. 2020) using a Moran’s I test (simulateResiduals and testSpatialAutocorrelation functions, DHARMa package; Hartig 2022). We found no evidence of spatial autocorrelation (P > 0.05).
Results
Immature monarch counts and presence
We conducted immature monarch surveys at 49 GWWA sites (147 visits) and acquired similar data for 25 reference sites (35 visits) from the MJV’s IMMP database. The average density of immature monarchs/ha at reference sites significantly differed between 2020 and 2021 (W = 84, P < 0.001). At the GWWA sites, we searched for immature monarchs on 13.75 ± 21.73 (mean ± SD) milkweed stems per visit. A total of 252 immature monarchs (eggs = 186, larvae = 66) were recorded at 26% (13/49) of GWWA sites (117 and 135 immatures at shrub-sheared and timber harvested sites, respectively). MJV surveyors searched for immature monarchs on 123.21 ± 91.62 (mean ± SD) milkweed stems per visit at reference sites, observing a total of 396 immature monarchs (eggs = 301, larvae = 95) at 92% (23/25) reference sites. Reference sites had significantly higher densities (283.52 immatures/ha) of immature monarchs compared to GWWA sites (12.92 immatures/ha, W = 84, P < 0.001; Table 2). At GWWA sites, the per-plant density of immature monarchs tended to decrease as site-specific milkweed density increased, but the trend was not statistically significant (P = 0.816, ρ = -0.066; Fig. 2A). The absolute density of immature monarchs (individual eggs and larvae per ha) increased with site-specific milkweed density (P < 0.001; ρ = 0.836; Fig. 2B).
Assessing immature monarch relationships with milkweed at GWWA sites (forest-dominated and managed for golden-winged warblers). Data were collected in northern Minnesota and Wisconsin during 2021. (A) A comparison of immature monarchs per milkweed plants searched by density of milkweed/ha (log-transformed). Immature monarchs per plant decreased as milkweed density increased, although not significantly (P = 0.816, ρ = -0.066). (B) Density of immature monarchs/ha by milkweed/ha (log-transformed). Density of immature monarchs increased significantly as milkweed density increased (P < 0.001, ρ = 0.836). Lines plotted are regression lines. Milkweed density was log-transformed to show linear relationship but was not needed for Spearman’s rank correlation test
The null model was the top-ranked model in both the immature monarch treatment type and vegetation characteristics model sets (Table 3). Our top model within the patch characteristics model set was treatment area, but the null model was within the competing set (Table 3). Our top model for the floral characteristics model indicated a positive relationship between floral abundance and the presence of immature monarchs (Table 3). The top model within the landscape characteristics model set was proportion of emergent herbaceous wetlands at 500 m (positive) and proportion of emergent herbaceous wetlands at 1 km (positive) was a competing model (Table 3).
Adult monarch butterfly counts and presence
Adult monarch surveys were conducted at 49 GWWA sites (147 visits) and 28 reference sites (53 visits). At GWWA sites, a total of 30 adult monarchs were recorded (11 and 19 at shrub-sheared and timber harvested sites, respectively) at 40% (20/49) of sites. At reference sites, a total of 96 adult monarchs were recorded and an adult was detected at 75% (21/28) of sites. When comparing the two survey years for reference sites, the average number of adult monarchs per site was not significantly different between 2020 and 2021 (W = 105, P = 0.47). The average number of adult monarchs per site was significantly higher at reference sites (2.65 adults) than GWWA sites (0.50 adults, W = 255, P < 0.001; Table 2). At GWWA sites, the abundance of adult monarchs did not increase with milkweed density (P = 0.581, ρ = -0.255). Adult monarchs were recorded nectaring on five flowering plant species at GWWA sites (geranium [Geranium spp.], swamp thistle [Cirsium muticum], joe-pye weed [Eutrochium spp.], bull thistle [Cirsium vulgare], and flat-topped white aster [Doellingeria umbellata]).
The null model was the top-ranked model in both the adult monarch treatment type and floral characteristics model sets (Table 4). Our top model within the patch characteristics model set suggested a positive relationship between hectares of treatment area and adult monarch presence (Table 4). Our top model for the vegetation characteristics model set was short shrub occurrence (positive) and bare ground occurrence was a competing model (negative; Table 4). Our top model within the landscape characteristics model set was proportion of mixed forest within 200 m, but the null model was within the competing set (Table 4).
Milkweed counts and presence
Milkweed surveys were conducted at the same number of sites and visits as the immature monarch surveys for both GWWA and reference sites. We recorded a total of 820 milkweed stems at 30% (15/49) of GWWA sites. A total of 7,369 milkweed stems were recorded at 100% (25/25) of reference sites. The average milkweed stem density (stems/ha) at reference sites was significantly different between 2020 and 2021 (W = 8, P < 0.001). Two GWWA sites had abnormally high average milkweed stem densities (113 and 75 stems/ha) compared to thirteen other sites in which milkweed was present (range = 1–29 mean milkweed stems per site). Milkweed density was higher at reference sites (3996.42 stems/ha) compared to GWWA sites (36.44 stems/ha, W = 8, P < 0.001; Table 2). We recorded three species of milkweed at the GWWA sites: common (Asclepias syriaca, 653 stems), swamp (A. incarnata, 143 stems), and poke (A. exaltata, 24 stems). Six milkweed species were recorded at reference sites: common, whorled (A. verticillata), butterfly weed (A. tuberosa), swamp, oval-leaf (A. ovalifolia), and poke.
The null model was our top-ranked model in both our treatment and structural vegetation sets for predicting milkweed presence (Table 5). Our top model within the patch characteristics model set was treatment area, but the null model was also within the competing set. The top model within the landscape characteristics model set was proportion of emergent herbaceous wetlands at 500 m (positive) and proportion of emergent herbaceous wetlands at 1 km was a competing model (positive; Table 5).
Within-site and landscape characteristics
At GWWA sites, we counted a total of 324,562 flowers (per site metrics: mean = 28.14 flowers/m2, SD = 33.69 flowers/m2, min = 2.65 flowers/m2, max = 198.12 flowers/m2) from 163 species (Supporting Table 1). Of these 163 species, 69% (n = 113) were native, 18% (n = 30) were non-native, and 12% were unknown (unidentified to species, n = 20; Supporting Table 1). Floral abundance was not recorded at reference sites. Structural vegetation characteristics were different between GWWA and reference sites. At GWWA sites, percent cover of trees and shrubs were greater than at reference sites (P < 0.001; Table 6). At reference sites, average floral frequency and percent cover of forbs and grass were greater than at GWWA sites (P < 0.05; Table 6). Floral richness was similar between GWWA and reference sites (P = 0.32; Table 6). At reference sites, 101 total blooming species were recorded, 62% (n = 63) of which were native, 30% (n = 30) non-native, and 9% (n = 8) unknown (unidentified to species).
At GWWA sites, percent occurrence of structural vegetation strata varied between the two treatment types (Supporting Table 2). Stand basal area was 4.33 m2/ha and 3.68 m2/ha at shrub-sheared and regenerating timber harvest sites, respectively (Supporting Table 2). At 1 km, land cover surrounding GWWA sites were dominated by woody wetlands (mean = 38%) or deciduous forest (mean = 33%) and sites were less dominated by grassland/herbaceous (mean = 1%), pasture/hay (mean = 4%), emergent herbaceous wetlands (mean = 6%), and mixed forests (mean = 7%; Supporting Table 3).
Discussion
Our results provide empirical evidence that early-successional communities managed for an at-risk songbird in the western Great Lakes region provide habitat for monarchs and modestly contribute to conservation goals for this imperiled butterfly. Additionally, we found that monarch (adult and immature) and milkweed presence was not influenced by treatment type, suggesting that shrub-shearing and regenerating timber harvest can equally contribute to monarch conservation goals. From 2015 to 2022 in Minnesota, Wisconsin, and Michigan, 10,964 ha of golden-winged warbler habitat was created on private (7,665 ha; L. Rowse, personal communication) and public (3,299 ha; P. Dieser, personal communication) lands. According to our recorded averages of adult (mean = 0.5 adults/ha) and immature (mean = 12.92 immatures/ha) monarch densities at GWWA sites, this area of golden-winged warbler management has created habitat for an estimated 5,502 adults (20 adults extrapolated using the mean egg to pupation survival rate [0.014%; Grant et al. 2020] on a total of 141,655 immature monarchs). Furthermore, this golden-winged warbler managed area has provided growing space for an estimated 399,528 milkweed stems, contributing approximately 0.02% of the 1.6 billion milkweed stems needed to reach eastern monarch population goals (Pleasants 2017). The quasi-extinction risk (the probability of inevitable extinction) for the eastern monarch butterfly population is estimated to be 56–74% by 2080 (U.S. Fish and Wildlife Service 2020), suggesting that current conservation efforts are not sufficient to support the population. Although habitat requirements between these two species are not perfectly aligned, any breeding habitat gains for monarchs via golden-winged warbler conservation will aid in reaching monarch habitat targets. Thus, we provide recommendations for enhancing golden-winged warbler habitat projects to maximize benefits to monarchs without compromising warbler BMPs.
The interspersion of herbaceous vegetation (> 40% cover) at a site is a priority for nesting golden-winged warbler BMPs (Roth et al. 2019) and our results (as well as other studies) indicate that such herbaceous vegetation – especially blooming plants and milkweed – benefit monarchs (Davis et al. 2007; Moranz et al. 2012; Dumroese et al. 2016). Average milkweed density for our GWWA sites (36 stems/ha) was considerably lower than those of studies conducted in herbaceous-dominated communities (1,390 stems/ha; Lukens et al. 2020 and 4,340 stems/ha; Bruce et al. 2021). However, mean floral frequency was similar (40% vs. 45% of subplots; Lukens et al. 2020). To enhance floral and ovipositing resources within GWWA sites, we encourage land managers to consider sowing native seed mixes (for nectar resources) and establishing host plants (i.e., milkweed), within retired log landings and skid trails since log landings have been found to support dense pollinator communities in Pennsylvania (Lee et al. 2021). Nectar plant species likely suitable for this purpose include blazing star (Liatris spp.), flat-topped white aster, geraniums, goldenrod, joe-pye weed, swamp thistle, and wild bergamont (Monarda fistulosa), to name a few. These species are known to be used by monarchs in this study (flat-topped white aster, geraniums, joe-pye weed, swamp thistle) and by other studies (blazing star, goldenrod, and wild bergamont; Lukens et al. 2020; Antonsen et al. 2021). Within-site characteristics of golden-winged warbler habitat and monarch BMPs do not align regarding density of woody regeneration. As young forests and shrublands age, shade intolerant plants (e.g., milkweed) are outcompeted by woody regeneration (Mathis et al. 2022). Golden-winged warbler abundance is positively associated with woody stem density (Martin et al. 2007) and cover (Buckhardt Thomas et al. 2023), but pollinator density has been shown to decline with woody stem density (Mathis et al. 2021). As a result, managers and private landowners should expect monarchs to benefit most from GWWA sites within six years post-management (Mathis et al. 2021) while golden-winged warblers may benefit immediately (Buckhardt Thomas et al. 2023) as well as during several years post-management (McNeil et al. 2020). Furthermore, if managers or private landowners are interested in maintaining monarch habitat, alternative silviculture practices (e.g., group selection harvest; Roberts et al. 2017) could be implemented to retain open structural conditions and provide pockets of sun exposure for herbaceous plants (e.g., milkweed).
According to golden-winged warbler BMPs within the Great Lakes, the size of the treated patch area should be 2.02–10.12 ha, depending on the distance from existing nesting habitat (Golden-winged Warbler Working Group 2019). Based on our finding that adult monarchs were more likely to occur at larger sites, managers intending to benefit monarchs as well as warblers should focus on creating larger (> 10 ha) treatment areas. Our findings relating to patch size (treatment area) are not consistent with other studies. For example, two studies that detected a negative relationship between patch size and monarch presence recorded average patches that were relatively large (93 ha, Bruce et al. 2021; and 34 ha, Davis et al. 2007), whereas our study and another that detected a positive relationship between patch size and monarch presence recorded average patch sizes that were relatively small (10 ha, this study; and 0.11 ha; Dinsmore et al. 2019). This may suggest that when available habitat is limited, adult monarchs will benefit from sites with greater available habitat (e.g., contiguous grassland or harvested area in forested landscapes). Additionally, sites with larger treatment areas would likely provide more floral and ovipositing resources that would attract adult monarchs. This is because a greater treatment area will provide a larger area of early-successional characteristics (e.g., grass, bramble, and forb cover) which is known to increase blooming plant density and diversity (Mathis et al. 2022).
In contrast to golden-winged warbler forest cover requirements (> 50%; Golden-winged Warbler Working Group 2019), our models detected a positive relationship between immature monarch and milkweed presence and percent area of emergent herbaceous wetlands (maximum = 31–38% within 500 m or 1 km). Similarly, other studies support that monarchs (Davis et al. 2007; Dinsmore et al. 2019) and milkweed (Zaya et al. 2017) are positively associated with semi-natural herbaceous land cover types. Therefore, if managers want to prioritize support of monarch populations, alongside those of golden-winged warblers, they should target sites with more semi-natural herbaceous land cover, while still meeting golden-winged warbler land cover requirements. Additionally, these sites may more likely be colonized by milkweed because it is wind dispersed and colonizes disturbed areas (Kaul and Wilsey 2019). Emergent herbaceous wetlands may be especially beneficial to monarchs in our study region because this land cover type is frequently disturbed (e.g., flooding) which maintains structurally open early-successional characteristics, resulting in abundant floral densities and diversity (Mathis et al. 2022). Although there is a conflict in landscape requirements, golden-winged warbler sites may still be enhanced for monarchs by increasing quality of the herbaceous community via native seed sowing as discussed above. Enhancing the herbaceous community component may mitigate the amount of semi-natural herbaceous land cover in the surrounding area required by monarchs.
Although monarchs and golden-winged warblers contrast in their ideal percent forest cover, they have similar preferences for forest type (i.e., deciduous) based on known forest type preference for golden-winged warblers (McNeil et al. 2020) and trends from this study for monarchs. Our models provide some evidence that the presence of adult monarchs was negatively predicted by mixed forest cover, and this may be caused by higher conifer tree cover. Golden-winged warblers, likewise, avoid conifer-dominated forests (McNeil et al. 2020). Coniferous forests are generally characterized as having low light transmission, acidic, nutrient-poor soils, and understories comprised of shade-tolerant herbaceous plants, whereas deciduous forests have high light transmission, richer soils, and a mixture of full sun and shade-tolerant herbaceous plants (Hart and Chen 2006). It is likely that our models detected no association with deciduous forests because our study area is overwhelmingly dominated by deciduous forest (especially aspen) which supports denser herbaceous plant communities compared to conifer stands (Hart and Chen 2006; Tavernia et al. 2016). This is promising as managing for deciduous-dominated forests for golden-winged warbler BMPs (Roth et al. 2019) will not negatively impact monarchs and milkweed.
This study provides promising evidence of co-benefits from golden-winged warbler management in the western Great Lakes for monarchs, but there are some study limitations worth keeping in mind. Although our results indicated that reference sites were strongly favorable for monarchs, the magnitude of difference between GWWA and reference remains unclear due to geographic limitations with our comparison. GWWA and reference sites were within different USDA level III ecoregions, and therefore differed in their landscape contexts (reference sites were all within a grassland-dominated landscape context). Therefore, the stark differences in monarch and milkweed metrics between GWWA and reference sites may be driven less by within-site conditions and more by landscape context. A second limitation is that we used two consecutive years (2020-21) for reference site data but only sampled GWWA sites during 2021. Monarch survival (Nail et al. 2015) and milkweed density (Cope et al. 2023) are known to vary greatly between years due to weather differences, which may have contributed to differences in densities detected between years for reference sites.
Our findings suggest that sites managed with golden-winged warbler BMPs provide marginal but potentially valuable habitat for monarchs in forest-dominated landscapes. Beyond monarchs, our findings add to the discussion on how pollinators in general respond to forest and shrubland management (Roberts et al. 2017; Lee et al. 2021; Mathis et al. 2021; Urban-Mead et al. 2021; Zitomer et al. 2023). Our findings may also have future implications for monarch habitat availability, as the eastern migratory monarch population is expected to experience northward shifts due to climate change (Batalden et al. 2007; Zylstra et al. 2022) and therefore may increasingly encounter forest-dominated landscapes. Extensive woody-dominated early-successional habitat management for golden-winged warblers is necessary to restore bird populations (McNeil et al. 2020) and this study, as well as others (Lee et al. 2021; Mathis et al. 2021), provides clear evidence that these communities are beneficial for monarchs and other pollinators given that certain conditions are met. Furthermore, implications of climate change (Batalden et al. 2007; Zylstra et al. 2022) alongside growing appreciation for pollinator biodiversity trends in forested landscapes (Ulyshen et al. 2023) suggest this is a strategic multi-benefit opportunity space. As a result, forest managers can play an important role in supporting monarch recovery while simultaneously achieving habitat goals for golden-winged warbler conservation. NRCS programs such as Workings Lands for Wildlife and Regional Conservation Partnership Program, offer an effective mechanism to achieve this shared vision.
References
Antonsen AK, Kral-O’Brien KC, Hovick TJ, Limb RF, Geaumont BA, Harmon JP (2021) Intra-annual spatiotemporal dynamics of the monarch butterfly (Lepidoptera: Danaidae), regal fritillary (Lepidoptera: Heliconiinae), and their floral resources in North Dakota, United States. Ann Entomol Soc Am 114(6):727–737. https://doi.org/10.1093/aesa/saab013
Arnold TW (2010) Uninformative parameters and model selection using Akaike’s Information Criterion. J Wildl Manage 74(6):1175–1178. https://doi.org/10.2193/2009-367
Askins RA (2001) Sustaining biological diversity in early successional communities: the challenge of managing unpopular habitats. Wildl Soc Bull 29(2):407–412
Bakermans MH, Smith BW, Jones BC, Larkin JL (2015a) Stand and within-stand factors influencing Golden-winged Warbler use of regenerating stands in the central Appalachian Mountains. Avian Conserv Ecol 10(1). https://doi.org/http://dx.doi.org/10.5751/ACE-00747-100110
Bakermans MH, Ziegler CL, Larkin JL (2015b) American woodcock and golden-winged warbler abundance and associated vegetation in managed habitats. Northeastern Naturalist 22(4):690–703. https://doi.org/10.1656/045.022.0405
Batalden RV, Oberhauser K, Peterson AT (2007) Ecological niches in sequential generations of eastern north american monarch butterflies (Lepidoptera: Danaidae): the ecology of migration and likely climate change implications. Environ Entomol 36(6):1365–1373
Bruce AS, Thogmartin WE, Trosen C, Oberhauser K, Gratton C (2021) Landscape- and local-level variables affect monarchs in Midwest grasslands. Res Square 1–15. https://doi.org/10.21203/rs.3.rs-346846/v1
Buckhardt Thomas A, McNeil DJ, Larkin JL, Johnson KE, Roth AM (2023) Evaluating Golden-winged Warbler use of alder and aspen communities managed with shearing in the western great lakes. Ecosphere 14(3):1–15. https://doi.org/10.1002/ecs2.4443
Bulluck LP, Buehller DA (2008) Factors influencing Golden-winged Warbler (Vermivora chrysoptera) nest-site selection and nest survival in the Cumberland mountains of Tennessee. Auk 125(3):551–559. https://doi.org/10.1525/auk.2008.07075
Burnham KP, Anderson DR (2015) A practical information-theoretic approach. Model selection and multimodel interence, 2nd edn. Springer, New York. https://doi.org/10.1016/b978-0-12-801370-0.00011-3
Confer JL (1992) Golden-winged warbler. Migratory nongame birds of management concern in the Northeast. US Fish and Wildlife Service
Confer JL, Porter C, Aldinger KR, Canterbury RA, Larkin JL, Mcneil DJ (2020) Implications for evolutionary trends from the pairing frequencies among golden-winged and blue-winged warblers and their hybrids. Ecol Evol 10(19):10633–10644. https://doi.org/10.1002/ece3.6717
Cope OL, Zehr LN, Agrawal AA, Wetzel WC (2023) The timing of heat waves has multiyear effects on milkweed and its insect community. Ecology 104(4). https://doi.org/10.1002/ecy.3988
Davis JD, Debinski DM, Danielson BJ (2007) Local and landscape effects on the butterfly community in fragmented Midwest USA prairie habitats. Landscape Ecol 22:1341–1354. https://doi.org/10.1007/s10980-007-9111-9
DeGraaf RM, Yamasaki M (2003) Options for managing early-successional forest and shrubland bird habitats in the northeastern United States. For Ecol Manag 185:179–191. https://doi.org/10.1016/S0378-1127(03)00254-8
Dinsmore SJ, Vanausdall RA, Murphy KT, Kinkead KE, Frese PW (2019) Patterns of Monarch Site occupancy and Dynamics in Iowa. Front Ecol Evol 7(169):1–14. https://doi.org/10.3389/fevo.2019.00169
Dumroese RK, Luna T, Pinto JR, Landis TD (2016) Forbs: Foundation for Restoration of Monarch butterflies, other pollinators, and Greater Sage-Grouse in the Western United States. Nat Areas J 36(4):499–511. https://doi.org/10.3375/043.036.0415
ESRI (2018) Release 10.7. Documentation Manual. Environmental Systems Resarch Institute
Federal Register (2020) Endangered and threatened wildlife and plants; 12-month finding for the monarch butterfly (Vol. 85, Issue 243). https://www.govinfo.gov/content/pkg/FR-2020-12-17/pdf/2020-27523.pdf
Filippelli SK, Vogeler JC, Falkowski MJ, Meneguzzo DM (2020) Monitoring conifer cover: Leaf-off lidar and image-based tracking of eastern redcedar encroachment in central Nebraska. Remote Sensing of Environment, 248. https://doi.org/10.1016/j.rse.2020.111961
U.S. Fish and Wildlife Service (2020) Monarch (Danaus plexippus) species status assessment report, version 2.1. https://www.fws.gov/savethemonarch/pdfs/Monarch-SSA-report.pdf
Fisher KE, Bradbury SP (2021) Influence of habitat quality and resource density on breeding-season female monarch butterfly Danaus plexippus movement and space use in north-central USA agroecosystem landscapes. J Appl Ecol. https://doi.org/10.1111/1365-2664.14061
Fogarty DT, Roberts CP, Uden DR, Donovan VM, Allen CR, Naugle DE, Jones MO, Allred BW, Twidwell D (2020) Woody plant encroachment and the sustainability of priority conservation areas. Sustainability 12(8321):1–15. https://doi.org/10.3390/su12208321
Golden-winged Warbler Working Group (2019) Best Management Practices for Golden-winged Warbler Habitats in the Great Lakes Region. 2nd Edition
Grant TJ, Flockhart DTT, Blader TR, Hellmich RL, Pitman GM, Tyner S, Norris DR, Bradbury SP (2020) Estimating arthropod survival probability from field counts: a case study with monarch butterflies. Ecosphere 11(4). https://doi.org/10.1002/ecs2.3082
Hart SA, Chen HYH (2006) Understory vegetation dynamics of north american boreal forests. CRC Crit Rev Plant Sci 25:381–397. https://doi.org/10.1080/07352680600819286
Hartig F (2022) DHARMa: Residual Diagnostics for Hierarchical (Multi-Level / Mixed) Regression Models. R package version 0.4.5. https://cran.r-project.org/package=DHARMa
Hijmans RJ (2021) raster: Geographic Data Analysis and Modeling. R package version 3.5-2. https://cran.r-project.org/package=raster
Homer CH, Fry JA, Barnes CA (2012) The National Land Cover Database. US Geol Surv Fact Sheet 3020(4):1–4. http://pubs.usgs.gov/fs/2012/3020/
Hunter ME, Colavito MM, Wright V (2020) The Use of Science in Wildland Fire Management: a review of barriers and facilitators. Curr Forestry Rep 6:354–367. https://doi.org/10.1007/s40725-020-00127-2
James FC, Shugart HH (1970) A quantitative method of habitat description. Audubon Field Notes 24:727–736
Kasten K, Stenoien C, Caldwell W, Oberhauser KS (2016) Can roadside habitat lead monarchs on a route to recovery? J Insect Conserv 20:1047–1057. https://doi.org/10.1007/s10841-016-9938-y
Kaul AD, Wilsey BJ (2019) Monarch butterfly host plant (milkweed Asclepias spp.) abundance varies by habitat type across 98 prairies. Restor Ecol 27(6):1274–1281. https://doi.org/10.1111/rec.12993
King DI, Schlossberg S (2014) Synthesis of the conservation value of the early-successional stage in forests of eastern North America. For Ecol Manag 324:186–195. https://doi.org/10.1016/j.foreco.2013.12.001
Klaus NA, Buehler DA (2001) Golden-winged Warbler breeding habitat characteristics and nest success in clearcuts in the southern Appalachian mountains. Wilson Bull 113(3):297–301. https://doi.org/10.1676/0043-5643(2001)113[0297:GWWBHC]2.0.CO;2
Lee MR, McNeil DJ, Mathis CL, Grozinger CM, Larkin JL (2021) Microhabitats created by log landings support abundant flowers and insect pollinators within regenerating mixed-oak stands in the Central Appalachian Mountains. For Ecol Manag 497. https://doi.org/10.1016/j.foreco.2021.119472
Litvaitis JA, Larkin JL, McNeil DJ, Keirstead D, Costanzo B (2021) Addressing the early-successional habitat needs of at-risk species on privately owned lands in the eastern united states. Land 10(1116). https://doi.org/10.3390/land10111116
Lukens L, Kasten K, Stenoien C, Cariveau A, Caldwell W, Oberhauser K (2020) Monarch Habitat in Conservation Grasslands. Front Ecol Evol 8(13):1–13. https://doi.org/10.3389/fevo.2020.00013
Lutter SH, Dayer AA, Rodewald AD, McNeil DJ, Larkin JL (2019) Early successional forest management on private lands as a coupled human and natural system. Forests 10(6):1–14. https://doi.org/10.3390/f10060499
Martin KJ, Lutz RS, Worland M (2007) Golden-winged Warbler habitat use and abundance in northern Wisconsin. Wilson J Ornithol 119(4):523–532. https://doi.org/10.1676/06-103.1
Mathis CL, McNeil DJ, Lee MR, Grozinger CM, King DI, Otto CRV, Larkin JL (2021) Pollinator communities vary with vegetation structure and time since management within regenerating timber harvests of the Central Appalachian Mountains. For Ecol Manag 496. https://doi.org/10.1016/j.foreco.2021.119373
Mathis CL, McNeil DJ, Lee MR, Grozinger CM, Otto CRV, Larkin JL (2022) Can’t see the flowers for the trees: factors driving floral abundance within early-successional forests in the central Appalachian Mountains. Can J For Res 52:1–12. https://doi.org/10.1139/cjfr-2022-0014
McClain WE, Ruffner CM, Ebinger JE, Spyreas G (2021) Patterns of Anthropogenic Fire within the midwestern Tallgrass Prairie 1673–1905: evidence from Written Accounts. Nat Areas J 41(4):283–300. https://doi.org/10.3375/20-5
McNeil DJ, Rodewald AD, Ruiz-Gutierrez V, Johnson KE, Strimas-Mackey M, Petzinger S, Robinson OJ, Soto GE, Dhondt AA, Larkin JL (2020) Multiscale drivers of restoration outcomes for an imperiled songbird. Restor Ecol 28(4):880–891. https://doi.org/10.1111/rec.13147
Midwest Association of Fish and Wildlife Agencies (2018) Mid-America Monarch Conservation Strategy, 2018–2038, Version 1.0http://www.mafwa.org/wp-content/uploads/2018/07/MAMCS_June2018_Final.pdf
MJV (2020) Integrated Monarch Monitoring Program (Issue Version 3.0). https://monarchjointventure.org/images/uploads/documents/IMMP_Guidebook_2021.pdf
Moranz RA, Debinski DM, McGranahan DA, Engle DM, Miller JR (2012) Untangling the effects of fire, grazing, and land-use legacies on grassland butterfly communities. Biodivers Conserv 21(11):2719–2746. https://doi.org/10.1007/s10531-012-0330-2
Mueller NG, Spengler RN, Glenn A, Lama K (2021) Bison, anthropogenic fire, and the origins of agriculture in eastern North America. Anthropocene Rev 8(2):141–158. https://doi.org/10.1177/2053019620961119
Nail KR, Stenoien C, Oberhauser KS (2015) Immature monarch survival: Effects of site characteristics, density, and time. Ann Entomol Soc Am 108(5):680–690. https://doi.org/10.1093/aesa/sav047
Oberhauser KS, Solensky MJ (2004) Monarch butterfly biology and conservation. Cornell University Press
Peterson DW, Reich PB (2001) Prescribed fire in oak savanna: fire frequency effects on stand structure and dynamics. Ecol Appl 11(3):914–927. https://doi.org/10.1890/1051-0761(2001)011[0914:PFIOSF]2.0.CO;2
Peterson SM, Streby HM, Andersen DE (2016) Spatially explicit models of full-season productivity and implications for landscape management of Golden-winged warblers in the western great lakes region. Stud Avian Biology, 141–160
Pleasants J (2017) Milkweed restoration in the Midwest for monarch butterfly recovery: estimates of milkweeds lost, milkweeds remaining and milkweeds that must be added to increase the monarch population. Insect Conserv Divers 10:42–53. https://doi.org/10.1111/icad.12198
Pleasants JM, Oberhauser KS (2013) Milkweed loss in agricultural fields because of herbicide use: effect on the monarch butterfly population. Insect Conserv Divers 6:135–144. https://doi.org/10.1111/j.1752-4598.2012.00196.x
R Core Team (2021) R: a language and environment for statistical computing. R Foundation for Statistical Computing. R Foundation for Statistical Computing. https://www.r-project.org/
Roberts HP, King DI, Milam J (2017) Factors affecting bee communities in forest openings and adjacent mature forest. For Ecol Manag 394:111–122. https://doi.org/10.1016/j.foreco.2017.03.027
Robin X, Turck N, Hainard A, Tiberti N, Lisacek F, Sanchez J-C, Müller M (2011) pROC: an open-source package for R and S + to analyze and compare ROC curves. BMC Bioinformatics. 12th edn.https://doi.org/10.1186/1471-2105-12-77
Rosenberg KV, Will T, Buehler DA, Swarthout SB, Thogmartin WE, Bennett RE, Chandler R (2016) Dynamic distributions and population declines of Golden-winged warblers: chap. 1. Stud Avian Biology 49:3–28
Rosenberg KV, Dokter AM, Blancher PJ, Sauer JR, Smith AC, Smith PA, Stanton JC, Panjabi A, Helft L, Parr M, Marra PP (2019) Decline of the north american avifauna. Science 366(6461):120–124. https://doi.org/10.1126/science.aaw1313
Roth AM, Flaspohler DJ, Webster CR (2014) Legacy tree retention in young aspen forest improves nesting habitat quality for golden-winged warbler (Vermivora chrysoptera). For Ecol Manag 321:61–70. https://doi.org/10.1016/j.foreco.2013.07.047
Roth AM, Rohrbaugh RW, Will T, Swarthout SB, Buehler DA (2019) Golden-winged Warbler Status Review and Conservation Plan. 2nd Edition. https://gwwa.org/wp-content/uploads/2020/06/GWWA_Conservation-Plan_191007_low-res.pdf
Rudolph DC, Ely CA, Schaefer RR, Williamson JH, Thill RE (2006) Monarch (Danaus plexippus L. Nymphalidae) migration, nectar resources and fire regimes in the Ouachita Mountains of Arkansas. J Lepidopterists’ Soc 60(3):165–170
Semmens BX, Semmens DJ, Thogmartin WE, Wiederholt R, López-Hoffman L, Diffendorfer JE, Pleasants JM, Oberhauser KS, Taylor OR (2016) Quasi-extinction risk and population targets for the Eastern, migratory population of monarch butterflies (Danaus plexippus) OPEN. https://doi.org/10.1038/srep23265
Sewell A (2010) Petition to list the Golden-winged Warbler (Vermivora chrysoptera) as a threatened or endangered species under the US Endangered Species Act. The Golden-Winged Warbler Working Group. www.fws.gov/midwest/es/soc/birds/GoldenWingedWarbler/%0AGoldenWingedWarblerPetition.pdf
Signorell A (2021) DescTools: Tools for descriptive statistics. R package version 0.99.44
Tavernia BG, Nelson MD, Garner JD, Perry CH (2016) Spatial characteristics of early successional habitat across the Upper Great Lakes states. For Ecol Manag 372:164–174. https://doi.org/10.1016/j.foreco.2016.04.003
Thogmartin WE, López-Hoffman L, Rohweder J, Diffendorfer J, Drum R, Semmens D, Black S, Caldwell I, Cotter D, Drobney P, Jackson LL, Gale M, Helmers D, Hilburger S, Howard E, Oberhauser K, Pleasants J, Semmens B, Taylor O, …, Wiederholt R (2017a) Restoring monarch butterfly habitat in the Midwestern US: “All hands on deck.” Environmental Research Letters, 12. https://doi.org/10.1088/1748-9326/aa7637
Thogmartin WE, Wiederholt R, Oberhauser K, Drum RG, Diffendorfer JE, Altizer S, Taylor OR, Pleasants J, Semmens D, Semmens B, Erickson R, Libby K, Lopez-Hoffman L (2017b) Monarch butterfly population decline in north america: identifying the threatening processes. Royal Soc Open Sci 4(9). https://doi.org/10.1098/rsos.170760
Ulyshen MD, Urban-Mead KR, Dorey JB, Rivers JW (2023) Forests are critically important to global pollinator diversity and enhance pollination in adjacent crops. Biol Rev 98(4):1118–1141. https://doi.org/10.1111/brv.12947
Urban-Mead KR, Muñiz P, Gillung J, Espinoza A, Fordyce R, van Dyke M, McArt SH, Danforth BN (2021) Bees in the trees: Diverse spring fauna in temperate forest edge canopies. Forest Ecology and Management, 482. https://doi.org/10.1016/j.foreco.2020.118903
Eastern monarch butterfly breeding range. USFWS, Fish US (2022) & Wildlife Service Environmental Conservation Online System. https://ecos.fws.gov/ecp/species/9743
USGS (2018) Golden-winged Warbler (Vermivora chrysoptera) bGWWAx_CONUS_2001v1 Range Map. U.S. Geological Survey Data Release - Gap Analysis Project (GAP). https://doi.org/10.5066/F747492D
Wohl E (2021) Legacy effects of loss of beavers in the continental United States. Environmental Research Letters, 16. https://doi.org/10.1088/1748-9326/abd34e
Zaya DN, Pearse IS, Spyreas G (2017) Long-term trends in midwestern milkweed abundances and their relevance to monarch butterfly declines. BioScience, vol 67. Oxford University Press, pp 343–356. 4 https://doi.org/10.1093/biosci/biw186
Zitomer RA, Galbraith SM, Betts MG, Moldenke AR, Progar RA, Rivers JW (2023) Bee diversity decreases rapidly with time since harvest in intensively managed conifer forests. Ecol Appl. https://doi.org/10.1002/eap.2855
Zuckerberg B, Cohen JM, Nunes LA, Bernath-Plaisted J, Clare JDJ, Gilbert NA, Kozidis SS, Maresh Nelson SB, Shipley AA, Thompson KL, Desrochers A (2020) A review of overlapping landscapes: pseudoreplication or a Red Herring in Landscape Ecology? Curr Landsc Ecol Rep 5:140–148. https://doi.org/10.1007/s40823-020-00059-4
Zylstra ER, Neupane N, Zipkin EF (2022) Multi-season climate projections forecast declines in migratory monarch butterflies. Glob Change Biol 28(21):6135–6151. https://doi.org/10.1111/gcb.16349
Acknowledgements
This project was primarily funded by the United States Department of Agriculture Natural Resources Conservation Service “Conservation Effects Assessment Project” (CEAP) award # A-3A75-16-522. Additional funding was provided by Pennsylvania Wildlife for Everyone Foundation and Commonwealth of Pennsylvania University Biologists. The Minnesota Outdoor Heritage Fund supported habitat management efforts on public sites in Minnesota. We are grateful to Claire Ratcliff and Spencer Henkel for collecting field data. We thank Cloquet Area Forest, Carlton, Aikin, Douglas, and Saint Louis counties for providing us access to these public places. We are especially thankful to NRCS and ABC staff for helping us coordinate access to study sites, especially Kevin Sheppard, Callie Bertsch, and Patrick Weber. Lastly, we are greatly appreciative of the private landowners who provided us with access to their property. Wildlife monitoring efforts on private lands would not be possible without these passionate and curious individuals, we greatly appreciate all that they have done for wildlife conservation and science.
Author information
Authors and Affiliations
Contributions
ECK, DJM, JLL conceived the ideas and designed the study; ECK, EB collected data; ECK and DJM analyzed data; JLL secured funding for the study; all authors wrote and edited manuscript and gave final approval for publication.
Corresponding author
Ethics declarations
Competing interests
None of the authors of this manuscript have competing interests or conflicts of interests to report.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Keele, E.C., McNeil, D.J., Duchamp, J.E. et al. Assessing the benefits of managed golden-winged warbler (Vermivora chrysoptera) nesting habitat for breeding monarch butterflies (Danaus plexippus) in the western Great Lakes. J Insect Conserv 27, 879–894 (2023). https://doi.org/10.1007/s10841-023-00505-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10841-023-00505-8