Abstract
Purpose
To evaluate, in a combined treatment strategy for treatment-warranted retinopathy of prematurity (ROP), which of the two is a better treatment sequence, peripheral laser photocoagulation (LPC-IVB) first or intravitreal bevacizumab (IVB-LPC) first.
Methods
Twenty-two babies (44 eyes) with ROP were recruited from 1 July 2014 to 30 March 2016. All the right eyes received LPC on day one followed by IVB on day four (LPC-IVB group). In all left eyes, IVB was injected on day one followed by LPC on day four (IVB-LPC group). The primary outcome measure was the proportion of eyes that had complete ROP regression with no additional treatment within 2 weeks of the onset of therapy. Retinal photography and drawings were used at each visit to document disease course.
Results
In LPC-IVB group, 72.7% (16/22) eyes had complete ROP regression with no additional treatment within 2 weeks of the onset of therapy. In the IVB-LPC group, 95.5% (21/22 eyes) had complete regression within 2 weeks. Additional laser had complete regression in all eyes in each group. One baby (two eyes, one from each group) had late recurrence at 5 months.
Conclusion
The combined therapy strategy was successful for ROP management. Administration of anti-vascular endothelial growth factor injection before the peripheral laser was better than the reverse strategy of laser first. Late recurrences and adverse events were low.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Retinopathy of prematurity (ROP) is an important cause of childhood blindness worldwide. Pan-retinal laser photocoagulation is a safe and effective treatment for ROP [1,2,]–[3]. The early treatment of high-risk pre-threshold ROP with laser significantly reduces unfavourable outcomes to a clinically important degree [4].
Intravitreal bevacizumab (IVB) monotherapy, as compared to conventional laser therapy, in infants with stage 3 + ROP showed a significant benefit for Zone I ROP but not for Zone II ROP [5]. Persistent peripheral avascular retina and late onset recurrent disease, however, have been reported with intravitreal injections [6, 7]. In the setting of media opacity, poor dilatation with neovascularization of iris (NVI), exudative retinal detachment or medical instability, intravitreal injection therapies provide an adjuvant treatment until laser therapy [5]. Combined treatment with both laser and anti-vascular endothelial growth factor (VEGF) therapies has been reported in several studies recently [6,7,8]. These showed promising results in treating stage 3 ROP and minimize the possible disadvantages of laser ablation or intravitreal anti-VEGF monotherapy.
Possible advantages of combined therapy include lower dose of anti-VEGF, less recurrence than monotherapy and preservation of central visual field [2]. In previous studies with combined modality treatment, no study has compared the sequence of peripheral laser photocoagulation (LPC) first followed by IVB treatment with the alternate sequence of IVB first followed by LPC treatment. Most studies have not mentioned the sequence used. The two treatments affect different pathways of the disease process and it is not clear if altering the sequence has any effect on the regression of ROP. Our Null hypothesis was that the outcomes of either of these two approaches would be the same. The purpose of the present study was to evaluate these two different sequences of the combined therapy.
Materials and methods
This prospective case series study was conducted at the Srimati Kanuri Santhamma Centre for Vitreoretinal Diseases and the Jasti V Ramanamma Children’s Eye Care Centre, L V Prasad Eye Institute, Hyderabad, India. Preterm babies presenting to the Out-Patient Department and having vision-threatening ROP that needed non-surgical treatment were included. Babies were also included in our ongoing ITCROPS ethics committee approved database [2].
Inclusion criteria
Type 1 ROP in Zone I, posterior Zone II [4] and aggressive posterior ROP (APROP).
Exclusion criteria
Cases with any ROP beyond stage 3 plus disease.
The study had Institutional Ethics Committee approval (Ethics Ref. No. LEC 06-14-053) including the off-label use of bevacizumab for ROP and followed the tenets of the Declaration of Helsinki. Informed written consent for each case was obtained from the parent or legal guardian of the baby. All prenatal, intra-natal and post-natal events including demography data were collected for each baby in a prospective database form. Pupils were dilated with 1% tropicamide and 2.5% phenylephrine instilled in 3 doses, 5 min apart with wiping of eyelids [2]. Retinal examination findings were documented using fundus photography (Retcam, Clarity Medical Systems, Pleasanton CA, USA) and retinal drawings as seen by indirect ophthalmoscopy. All treatments were done under topical anaesthesia [3] with pulse oximeter monitoring and a stand by neonatal anaesthesiologist. Following the completion of treatment and two hours of observation and feeding, the babies were discharged to home if stable or transported to the neonatologist’s centre if needed. Systemic parameters including anaemia, respiratory problems, sepsis, nutrition, etc. were monitored and managed in coordination with the treating neonatologist [2].
All the right eyes (LPC-IVB) received peripheral laser (LPC) on day 1 and the left eyes (IVB-LPC) received IVB 0.625 mg in 0.025 ml on day 1. On day 4 (± 3 days), before next session of treatment, fundus photographs or drawings were recorded. Then, the right eyes received IVB and left eyes received LPC at this visit. The laser protocol required sparing of retina up to 2 disc diameters from the anterior ridge of vascularization [8]. Confluent laser was done beyond this area up to the ora serrata in a single session. At 1, 2 and 6 weeks follow-up visits, fundus photograph or retinal drawing was done to assess the effect of the two treatment regimens. Additional visits were scheduled based on disease status. In all eyes, the posterior pole was skipped while doing LPC or additional laser except in one eye (in LPC-IVB group) where laser was extended to posterior areas unintentionally while doing LPC. Additional laser was done after 2–6 weeks from onset of therapy in eyes that showed inadequate disease regression. Criteria for doing supplemental laser photocoagulation after 2 weeks or more included poorly dilating pupils with persistent NVI, persistent dilatation and tortuosity in any quadrant, active new vessels or an early traction in any clock hour. Complete regression was defined as clear media, well dilating pupils, attached macula with no active new vessels, no plus disease and no active traction anywhere in the retina both on indirect ophthalmoscopy and on Retcam photographs evaluated by two independent observers.
Primary outcome measure was the proportion of eyes that had complete ROP regression with no additional treatment within 2 weeks of the onset of therapy. Secondary outcome was the proportion of eyes that did not regress even after supplemental laser and needed surgery or those with any complications of therapy. The final effect was measured in all babies at 2 months post-treatment. Babies were also followed up for one year and beyond to evaluate visual and refractive outcomes and any complications.
Statistical analysis was done using STATA version 11 (StataCorp, College Station, TX, USA). Categorical data were described using proportion and analysed using Chi-square test and Fisher's exact test. Continuous data were checked for normality using Shapiro–Wilk test. Normally distributed data were described by using mean and standard deviation and analysed by using Students’ t test, while data which were not normal were described using median and inter-quartile range and analysed by using Mann–Whitney test. P value of < 0.05 was considered statistically significant.
Results
Forty-four eyes of 22 patients with Type 1 ROP in Zone I (4 eyes, 9.1%), posterior Zone II (8 eyes, 18.2%) and APROP (32 eyes, 72.7%) were included in the study. Mean gestational age at birth was 30.7 ± 1.5 weeks. Male–female ratio was 15:7. Mean birth weight was 1246.8 ± 185.8 g. Mean post-menstrual age at treatment was 35.5 ± 1.5 weeks.
Primary Outcome measure
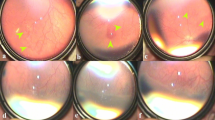
The proportion of eyes that had complete ROP regression within 2 weeks of the onset of therapy with no need for any additional treatment was 72.7% in LPC-IVB group (16/22 eyes) while 6/22 eyes (27.3%) did not have complete regression initially. All these six eyes regressed after one additional session of laser photocoagulation. Hence, all 22 eyes (100%) in LPC-IVB group had complete regression when evaluated at 2 months (Fig. 1) (see figure, Supplemental Digital Content 1, which demonstrates colour fundus images of LPC group). The proportion of eyes that had complete ROP regression within 2 weeks of the onset of therapy with no additional treatment was 95.5% in IVB-LPC group (21/22 eyes). The one eye with residual disease showed complete regression with additional single session of laser when evaluated at 2 months (Fig. 2) (see figure, Supplemental Digital Content 2, which demonstrates colour fundus images of IVB group).
a 1st day before laser showing vascular loops in Zone I APROP; b 4th Day after laser and before IVB, image showing incompletely regressed plus disease and neovascularization inferiorly, laser marks not seen; c 3 weeks post-laser and IVB, before giving additional laser showing large pre-retinal haemorrhage, vitreous haemorrhage covering macula and with few laser spots; d 5 months after additional laser and IVB right eye showing fully regressed ROP
Six of the seven eyes from both groups that needed additional laser had APROP (five in LPC-IVB group and one in IVB-LPC group) and one had threshold ROP in posterior Zone II that belonged to the LPC-IVB group. The need for additional laser was significantly different between LPC-IVB and IVB-LPC groups (Table 1, p = 0.03). Hence in a combined strategy, treatment with IVB followed by laser after 4–7 days was the best strategy to achieve early regression with rare need for additional laser.
Recurrence rate and long-term outcome
Two eyes (one from each group of LPC-IVB and IVB-LPC) of one baby with APROP developed late recurrence and retinal detachment at 5 months post-treatment that was managed by surgery which salvaged the vision of one eye. All the other 42 eyes on follow-up from 12 to 30 months showed attached posterior pole with lasered periphery and well regressed ROP in all areas. None of the eyes had vitreopathy related problems like disc or macular drag or tractional changes in any quadrant.
Adverse events
No systemic adverse events of drug or procedure were noted in any baby. Linear patch of retinal pigmented epithelium (RPE) atrophy was seen during the follow-up in 4 eyes of 2 babies with APROP, 2 eyes in each group (Fig. 3). Transient fresh pre-retinal or vitreous haemorrhage (Fig. 1) was seen within 1–3 weeks of therapy in 4 eyes (3 in LPC group and 1 in IVB group) needing additional laser in 3 eyes.
Discussion
Our study was primarily designed not to test the efficacy or safety of the combined regimen but to specifically address the outcomes of two different protocols of a combined treatment. Most studies on combined treatment do not mention whether laser should be done first or IVB should be administered first. Results of our study showed that intravitreal injection few days before laser seems to have distinct advantage.
The pathogenesis of the neovascular process in ROP involves the secretion of angiogenic factors, mainly VEGF into the vitreous cavity from cells in the avascular retina of a premature baby. Laser photocoagulation destruction of this avascular retina has been the standard of care for management of the neovascular process in ROP following reports of its safe and effective use from multicentric randomized controlled trials and real-world database outcomes [1, 2, 6, 7]. Laser works by destruction of the VEGF producing cells so that the neovascular process can be halted. However, it does not have any effect on the already secreted VEGF, and hence, the laser therapy takes about 2 weeks to have an effect on the ROP neovascular process. Anti-VEGF agents, on the other hand, inhibit and immediately neutralize the already secreted VEGF molecules but do not prevent the secretion of VEGF. Inhibition of VEGF has been shown to be effective in several angiogenic disorders [6]. Bevacizumab is the most commonly reported anti-VEGF agent used in ROP and has been widely used as an off-label treatment for ocular angiogenic conditions [5,6,7, 9,10,11]. The combined regimens, by targeting two different pathways of ROP angiogenic process, are reported to be effective and also prevent adverse effects of each of the modality [7, 8, 10].
The design of our protocol was uniquely placed to address the issue whether altering the sequence of therapy has any effect on outcomes. Firstly, both eyes received treatment from day one and so none of the two eyes of the baby had to wait for the treatment to start and be at a disadvantage. Secondly, risk of infection from bilateral simultaneous injections, though minimal, was avoided. This helped to avoid any risk of a possible bilateral endophthalmitis and also the theoretical possible surge of the drug into the systemic circulation on a single day dosing of the two eyes. The protocol also avoided prolonged stressful bilateral laser session both to the baby and to the treating surgeon because each eye received laser on a different day. This allowed completion of treatment to each eye in half the time on each day of therapy and was friendly to all stakeholders. By including each eye of the same baby into the two arms of the study, individual biologic and systemic factors that could influence outcomes were nullified. During the design of the study, there were concerns on the effect of IVB on the contralateral eye. However, this was taken care because the same situation would prevail for each eye once both sessions were completed as per protocol, and hence, we believe that contralateral effects if any would not be a significant influence on the outcomes. The strategy also allowed us to spare large parts of posterior pole from the destructive effects of laser and overall reduced number of laser burns than what is needed for laser monotherapy [8].
Results of our study showed that the primary outcome measure of completely regressed ROP with no need for additional treatment was seen more in IVB-LPC group eyes than in LPC-IVB group eyes. Rates of repeat laser were significantly higher in LPC-IVB group eyes than IVB-LPC group eyes. This could be possibly explained by the fact that after initial IVB, the pupil dilates well and media haze reduces while NVI and plus disease decrease within 3–4 days. Peripheral laser following an IVB would be easier to perform leading to a more complete and confluent laser delivery. Also due to ease of delivering laser even up to far periphery, there is less pressure on the globe during the procedure. On the other hand, laser at the first session in treatment naive eyes would face poorly dilating pupils and hazy media and hence is more difficult to deliver adequately. Both these situations would possibly be the cause of the new pre-retinal or vitreous haemorrhages that we observed in the LPC-IVB group eyes than the IVB-LPC group eyes (three eyes versus one eye). LPC group eyes hence took longer to regress and also needed additional laser. The final outcome, however, did not seem to be affected by either of the two strategies. These final regression of ROP results are similar to the BEAT ROP study [5]. However, with IVB monotherapy used in that study, early recurrence was seen in 4% eyes [5], while this was only 0.5% (one of 22 eyes) in the eyes with combined therapy where IVB was used first followed by early laser to peripheral retina. While this could partly be due to small sample of our study compared to the BEAT-ROP study, nevertheless the results are worth pursuing further.
Studies have shown that combined treatment with IVB and laser results in rapid ROP regression and development of peripheral retinal vessels within 1–2 weeks with minimal vitreoretinal traction by the fibrovascular membrane, but allowed peripheral, retinal vessel growth over the laser scars subsequently [6]. In our study also, every eye showed normal vascularization up to and beyond through the laser scars with no tractional residual components (Figs. 1, 2). One major problem that has been noted with bevacizumab monotherapy is the higher rate of late recurrences, many months after initial therapy [11]. The need for long-term periodic follow-up possibly till the peripheral retina gets vascularized is hence well recognized. Our combined strategy appeared quite effective in preventing late recurrences though not in all eyes. One patient (2 eyes) had a late recurrence after initial regression and this appeared to be due to new growth of avascular retina at anterior edge of the already treated retina as reported by us earlier [12].
Side effects of anti-VEGF have been reported [13,14,15]. None of our patients had ocular or systemic side effect during the treatment or thereafter that could be attributed to the procedures. However, atrophic changes in RPE or choroid were noted in four eyes and we believe these are related to the anti-VEGF drugs as similar changes have not been reported in laser only eyes. The anatomical outcomes however in all except 2 eyes of one baby were very encouraging. The visual, refractive and long-term cognitive outcomes are being followed and will be reported after completion of 5 years for all babies. The current study was presented at the “Hot Topics in ROP” meet in Chicago in November 2018 [16].
Conclusion
Anti-VEGF injection before laser therapy in a combined treatment regimen for ROP was found to be superior to the alternate strategy of laser first followed by anti-VEGF injection. Good anatomic results were achieved by the combined treatment using either of the two regimens after additional peripheral laser in non-regressed eyes. Recurrence rates with combined therapy were minimal though not eliminated. Pre-retinal or vitreous haemorrhage and RPE atrophic changes were noted in few eyes during the course of treatment.
References
Axer-Siegel R, Maharshak I, Snir M, Friling R, Ehrlich R, Sherf I, Shalev B, Sirota L, Weinberger D (2008) Diode laser treatment of retinopathy of prematurity: anatomical and refractive outcomes. Retina 28:839–846
Jalali S, Kesarwani S, Hussain A (2011) Outcomes of a protocol-based management for zone 1 retinopathy of prematurity: the Indian Twin Cities ROP Screening Program report number 2. Am Jl Ophthalmol 151:719–724
Jalali S, Azad R, Trehan HS, Dogra MR, Gopal L, Narendran V (2010) Technical aspects of laser treatment for acute retinopathy of prematurity under topical anesthesia. Ind Jl Ophthalmol 58:509–515
Early Treatment for Retinopathy of Prematurity Cooperative Group (2003) Revised indications for the treatment of retinopathy of prematurity: results of the early treatment for retinopathy of prematurity randomized trial. Arch Ophthalmol 121:1684–1694
Mintz-Hittner HA, Kennedy KA, Chuang AZ (2011) Efficacy of intravitreal bevacizumab for stage 3+ retinopathy of prematurity. N Eng J Med 364:603–615
Lee JY, Chae JB, Yang SJ, Yoon YH, Kim JG (2010) Effects of intravitreal bevacizumab and laser in retinopathy of prematurity therapy on the development of peripheral retinal vessels. Graefe's Arch Clin Exp Ophthalmol 248:1257–1262
Ahmed AE, Channa R, Durrani J, Ali A, Ahmad K (2010) Early experience with intravitreal bevacizumab combined with laser treatment for retinopathy of prematurity. Mid East Afr Jl Ophthalmol 17:264–267
Kim J, Kim SJ, Chang YS, Park WS (2014) Combined intravitreal bevacizumab injection and zone I sparing laser photocoagulation in patients with zone I retinopathy of prematurity. Retina 34:77–82
Lalwani GA, Berrocal AM, Murray TG, Buch M, Cardone S, Hess D, Johnson RA, Puliafito CA (2008) Off-label use of intravitreal bevacizumab (Avastin) for salvage treatment in progressive threshold retinopathy of prematurity. Retina 28:S13–18
Chung EJ, Kim JH, Ahn HS, Koh HJ (2007) Combination of laser photocoagulation and intravitreal bevacizumab (Avastin) for aggressive zone I retinopathy of prematurity. Graefe's Arch Clin Exp Ophthalmol 245:1727–1730
Hu J, Blair MP, Shapiro MJ, Lichtenstein SJ, Galasso JM, Kapur R (2012) Reactivation of retinopathy of prematurity after intravitreal bevacizumab. Arch Ophthalmol 103:1000–1006
Dave VP, Jalali S, Rani PK, Padhi TR (2014) Characteristics and outcomes of anterior hyaloidal fibrovascular proliferation in lasered retinopathy of prematurity. The Indian Twin Cities Retinopathy of Prematurity Study (ITCROPS) report number 4. Int Ophthalmol 34:511–517
Klufas MA, Chan RV (2015) Intravitreal anti-VEGF therapy as a treatment for retinopathy of prematurity: what we know after 7 years. JAAPOS 52:77–84
Chhablani J, Rani PK, Balakrishnan D, Jalali S (2014) Unusual adverse choroidal reaction to intravitreal bevacizumab in aggressive posterior retinopathy of prematurity: the Indian Twin Cities ROP screening (ITCROPS) data base report number 7. Semin Ophthalmol 29:222–225
Jalali S, Balakrishnan D, Zeynalova Z, Padhi TR, Rani PK (2013) Serious adverse events and visual outcomes of rescue therapy using adjunct bevacizumab to laser and surgery for retinopathy of prematurity. The Indian Twin Cities Retinopathy of Prematurity Screening database Report number 5. Arch Dis Child Fetal Neonatal Ed 98:F327–F333
Jalali S, Laveti V, Balakrishnana D, Rani PK, Mohamed A (2018) Prospective clinical study of two different regimens of combined laser photocoagulation and intravitreal bevacizumab treatment for retinopathy of prematurity. In: Hot topics in ROP meet, Chicago. https://pdfs.semanticscholar.org/2e33/db395c1fe823b73cf54a69cbf9f18757d9ae.pdf Meeting abstract 68
Funding
Hyderabad Eye Research Foundation provided unrestricted support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Human and animal rights
All procedures performed in this study involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. The study had Institutional Ethics Committee approval (Ethics Ref. No. LEC 06-14-053) including the off-label use of bevacizumab for ROP.
Conflict of interest
The authors declare that they have no conflict of interest.
Informed consent
Informed consent for each case was obtained from the parent or legal guardian of the baby.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Off-Label Use: Bevacizumab is an off-label use in ROP mentioned in this paper.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplemental Digital Content 1
(a) 1st day before laser showing vascular loops in Zone I APROP; (b) 4th Day after laser and before IVB, image showing incompletely regressed plus disease and neovascularization inferiorly, laser marks not seen; (c) 3 weeks post-laser and IVB, before giving additional laser showing large pre-retinal haemorrhage, vitreous haemorrhage covering macula and with few laser spots; (d) 5 months after additional laser and IVB right eye showing fully regressed ROP. (TIFF 2339 kb)
Supplemental Digital Content 2
(a): 1st day before IVB showing vascular loops in Zone I APROP; (b): 4th Day after IVB and before laser, loops not regressed but plus reduced; (c): In 3rd month, fully regressed ROP with laser spots. (TIFF 2177 kb)
Rights and permissions
About this article
Cite this article
Laveti, V., Balakrishnan, D., Rani, P.K. et al. Prospective clinical study of two different treatment regimens of combined laser photocoagulation and intravitreal bevacizumab for retinopathy of prematurity: the Indian Twin Cities ROP Study (ITCROPS) database report number 9. Int Ophthalmol 40, 3539–3545 (2020). https://doi.org/10.1007/s10792-020-01543-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10792-020-01543-w