Abstract
Purpose
To compare the clinical features, visual outcomes and causative organisms between endophthalmitis secondary to cataract surgery or to intravitreal injections (IVI).
Setting
Meir Medical Center, Kfar Saba, Israel.
Design
Retrospective, non-randomized comparative chart review.
Methods
Medical records of patients with proven or suspected endophthalmitis admitted to the Ophthalmology Department at Meir Medical Center 2/2002–2/2017 were reviewed. Clinical characteristics including presenting and final visual acuity (VA) outcomes, causative organisms and time to admission were assessed.
Results
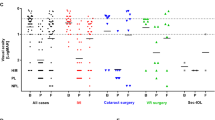
Among 84 patients in our study, 35 had preceding cataract surgery and 12 had preceding IVI. The post-cataract group showed a significant improvement in VA following treatment with a presenting and final VA (logMar ± SD) of (1.80 ± 0.54 and 1.39 ± 0.65, P < 0.01) as opposed to the post-IVI group (1.72 ± 0.26 and 1.81 ± 0.32, P = 0.692), while most patients in the cataract group exhibited some degree of VA recovery (70.96%). Patients undergoing cataract surgery were divided into two separate groups; those who underwent cataract surgery in a private center and those operated at a public center. Patients undergoing surgery at a private medical center showed improvement in VA outcomes following treatment (1.80 ± 0.57 and 1.13 ± 0.66, P < 0.01) as opposed to those operated on at our public medical center.
Conclusions
Overall, patients with endophthalmitis following cataract surgery had better visual outcomes and were more likely to show a VA improvement following treatment when compared with endophthalmitis following IVI. Final VA outcomes of patients with endophthalmitis after cataract surgery performed in a private center were better than those operated on and treated in a public medical center.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Endophthalmitis is a rare, yet disastrous complication of cataract surgery and intravitreal injections. The reported incidence after cataract procedures is 0.023–0.41% [1,2,3] and 0.038–0.056% [4,5,6] after intravitreal injections (IVI). The most common causative organisms of endophthalmitis following penetrating ocular procedures are coagulase-negative Staphylococcus (CoNs) species [1, 2, 7].
Substantial improvements in cataract surgery techniques, such as the use of injectable lenses, micro-incisions, and sutureless surgical wounds have dramatically changed the nature of this surgery in the past few decades, causing it to become faster, more efficient and with a shorter recovery time. The introduction of iodine for preoperative sterilization, intraoperative injection of cefuroxime and postoperative topical antibiotics help minimize the risk of this severe complication. However, endophthalmitis remains a visually devastating complication of this very common procedure [8], essentially because of its poor visual prognosis, as one-third of individuals will not regain visual acuity beyond counting fingers [9].
Concurrently, progress in ocular pharmacotherapeutics and the addition of new medical indications, has led to an exponential increase in IVI. Various complications may occur after IVI, including elevated intraocular pressure and retinal detachment [10]; yet, infectious endophthalmitis remains one of the most dreaded complications due to its poor visual prognosis.
Two recent meta-analyses demonstrated that Staphylococcus spp. are still the most commonly cultured causative organisms in endophthalmitis occurring after cataract surgery and IVI [1, 11]. McCannel [11] reported a significantly greater rate of endophthalmitis due to Streptococcus species following IVI as compared with postoperative endophthalmitis.
This study objective was to assess the difference in VA outcomes between endophthalmitis secondary to cataract surgery or intravitreal injection and to further characterize the main causative organisms and other clinical characteristics between these two groups of patients.
Methods
A retrospective, non-randomized comparative chart review was conducted in accordance with the tenets of the Declaration of Helsinki. The Institutional Review Board of Meir Medical Center, Kfar Saba, Israel, approved the study protocol. Medical records of all cases with proven or suspected endophthalmitis admitted to the Ophthalmology Department at Meir Medical Center from 02/2002 to 02/2017 were reviewed. All cases were included, regardless of the type of ophthalmic surgery, intraocular procedure or injections preceding the endophthalmitis or the site of operation or injection.
Patient records were first classified by etiology of endophthalmitis. They were then retrospectively evaluated for the following case-related data: demographic factors; systemic diseases; previous ocular diseases; lens status; presenting and final Snellen VA; number of days from injection or surgery to presentation in the clinic; clinical signs and symptoms at presentation; preoperative, intraoperative and postoperative prophylactic antibiotic regimen; species of bacterial infection and management of the infection.
Endophthalmitis was diagnosed clinically by identifying symptoms consistent with the condition, predominantly increasing ocular pain, loss of vision, anterior chamber inflammation, hypopyon and vitreous opacities. At least one vitreoretinal specialist confirmed the presumed diagnosis of endophthalmitis in all cases.
All study patients diagnosed with endophthalmitis underwent a standard trans-pars plana vitreous ‘tap and inject’ protocol or primary vitrectomy, according to the decision of the managing ophthalmologists. Vitreous sample taken for microbiological assessment was followed by intravitreal injection of vancomycin (1 mg/0.1 mL) associated with ceftazidime (2.25 mg/0.1 mL) along with injection of intravitreal dexamethasone in several cases.
Visual acuities were converted to logMAR values for statistical analysis. As described by Schulze-Bonsel et al. [12] the following conversion to logMAR was used for vision worse than 6/120: counting fingers = 1.7, hand motion = 2.0, light perception = 2.3 and no light perception = 3.0. Data were analyzed with SPSS for Windows (SPSS Inc, Chicago, IL). Statistical analysis was carried out using the two-tailed t-test. Statistical significance was considered at P < 0.05.
Results
Eighty-four patients were treated for endophthalmitis at Meir Medical Center from February 2002 to February 2017. Among them, 42 (50%) had undergone ocular or periocular surgery up to 1 week prior to presentation, which was regarded as the precipitating factor for the infection. Of these, cataract surgery was the most common, accounting for 35 cases (83%). This was followed by oculoplastic procedures in 3 (7%), penetrating keratoplasty in 2 (4%), pars plana vitrectomy in 2 (4%) and implantation of Ahmed glaucoma valve in 1 patient (2%). Among the 42 patients who presented with non-postoperative endophthalmitis, intravitreal injections were the most common proceeding factor, accountable for 12 cases (28%), followed by ocular trauma in 5 cases and bleb-associated infections in another 5 cases (12% each).
The cataract surgery patients consisted of two sub-groups: 21 patients who underwent cataract surgery in our public medical center and 14 patients operated in private centers.
The average age of the post-cataract surgery and post-intravitreal injection groups was similar, 72 years (range, 49–88) and 74 years (range, 62–97), respectively. Overall, average age of patients was 68 years. Median time for the initial admission and treatment of endophthalmitis varied, ranging from 3 days for the post-IVI group to 4.5 days for pseudophakic patients operated at our center and 7 days for those operated on a private medical center. Median time to admission for all endophthalmitis cases following cataract surgery was 5 days.
Among the 84 cases included in the study, 47 were culture positive. The most prevalent pathogen was CoNs followed by Staphylococcus aureus and Streptococcus pneumoniae. Detailed culture results for the 35 post-cataract and 12 post-IVI patients are presented in Table 1.
A statistically significant improvement in VA was attained following treatment in patients with endophthalmitis post-cataract surgery, with most patients showing at least some degree of VA recovery (Table 2). Conversely, the difference between the presenting and final VA in the post-IVI group did not reach statistical significance, with vision in most patients deteriorating despite treatment. In the post-cataract group, patients who underwent surgery at a private medical center showed statistically significant improvement in VA following treatment as opposed to those operated on at our public medical center, whose VA improvement was not statistically significant (Table 3).
Details concerning the 12 cases of endophthalmitis following IVI are presented in Table 4. Most cases of endophthalmitis in this group occurred after Avastin injection, as it is the most prevalent anti-vitreal injection in Israel. The median time to presentation was 3 days following injection (range 1–25 days). Ten patients underwent pars plana vitrectomy with IVI of vancomycin and ceftazidime and some had dexamethasone added as the primary treatment modality. Two patients underwent vitreous tap and injection of antibiotics into the vitreous. Vitreous cultures were positive in eight cases. Of these, the most commonly isolated organism was CoNs.
Discussion
This study reviewed consecutive cases of endophthalmitis following various precipitating events treated at a single center over 15 years. The current study is one of few retrospective studies to directly compare the visual outcomes and the spectrum of causative organisms of acute endophthalmitis following IVI or cataract surgery. The direct comparison of a large group of patients treated and followed at a single center provides a methodical advantage.
The rate of endophthalmitis following cataract surgery in this study was 0.001% or 1/819 surgeries compared with 0.00017% or 1/5867 injections after IVI. These rates are lower than the aforementioned incidences (0.023–0.41% [1,2,3] and 0.038–0.056% [4,5,6], respectively). To note, the reported incidence of endophthalmitis following other common ocular surgeries is notably higher; 0.02–0.84% for 20G/25G pars plana vitrectomy [13], 0.2–0.382% for penetrating keratoplasty [14] and 0.00197% and 6.3% for Ahmed glaucoma valve implantation [15].
Similar presenting features and causative organisms of endophthalmitis following clear cornea incision cataract surgery have been consistently reported over the past 2 decades. Most cases of postsurgical endophthalmitis are attributed to seeding from the patient’s conjunctiva and adnexa [16, 17]. The presenting characteristics of our cohort are similar to those reported in other large-scale multicenter studies [18,19,20]. In this study, Gram-positive infections continued to be the most prevalent causative factor for endophthalmitis following cataract surgery, with CoNS comprising slightly over 30% of culture-positive cases and Staphylococcus aureus accounted for 17%. CoNS also accounted for more than 40% of all positive culture results in the post-IVI group, similar to the percentage described in a meta-analysis by McCannel [11]. In the current study, streptococcus species accounted for less than 10% of positive cultures Among the post-IVI patients, in contrast to a higher incidence reported by several previous studies [6, 21,22,23,24]. Simunovic et al. [24] reported an increased incidence of Streptococcus spp. endophthalmitis among patients presenting post-IVI compared with patients presenting with endophthalmitis post-cataract surgery. This trend was not demonstrated in our study, possibly due to the small number of patients in the post-IVI group.
Median time to presentation in the current study was 5 days after cataract surgery and 3 days in the post-IVI group, slightly shorter than the 6 days reported from the Endophthalmitis Vitrectomy Study [25] and other studies investigating endophthalmitis after IVI (4 days) [17, 26]. Our observation regarding shorter time to presentation in cases of endophthalmitis post-IVI compared to post-cataract cases agrees with results of studies directly comparing these two etiologies [24, 27]. This finding could be explained by the differences among patients with known retinal disease receiving monthly intravitreal injections, who are more likely to be aware of any changes in their ocular and visual status as compared to healthy patients admitted for routine cataract surgery. In the post-cataract groups, an interesting finding was the longer time to presentation in patients who had initial surgery at a private center (7 days post-op) compared with those operated on at our public center (4.5 days post-op). This difference might be attributable to the lack of ocular emergency services at private centers, which could delay patients from seeking help, in contrast to public center’s patients who may simply return to the same establishment as soon as any complication arise. If this is the case, it may be disconcerting, given the critical importance of immediate medical attention when a complication such as endophthalmitis arises.
The post-IVI group was less likely to show VA improvement following treatment compared with the post-cataract group, a finding which correlates with the study by Simunovic et al. [24] The poorer final VA in this group might not be solely due to the infection-induced pathology, as inferior visual potential might be present due to retinal structural damage from the underlining pathology.
A unique analysis in the current study was the comparison between post-cataract cases who underwent surgery at our public medical center as opposed to private medical institutions. Interestingly, although presenting VA was very similar between those groups and the median time to presentation was shorter among patients who were operated at our medical center (4.5 vs. 7 days), the final VA was better in patients undergoing surgery at private medical centers, as was the percentage of patients showing any improvement in final VA. These unexpected observations might be the result of lower initial visual potential, higher rates of ocular comorbidities and poorer compliance with treatment that are more characteristic of patients undergoing cataract surgery in a public medical center, as described by Sommer [28]. Nevertheless, this observation may simply imply that VA outcomes in patients with endophthalmitis post-cataract surgery has no correlation with the type of the institution in which the surgery was conducted.
Three cases of endophthalmitis following IVI had received intravitreal triamcinolone, comprising 25% of all post-IVI cases in our study. Intravitreal triamcinolone acetonide is an increasingly popular treatment for inflammatory eye disease, neovascular age-related macular degeneration and macular edema [29,30,31,32]. Although infections and sterile endophthalmitis following intravitreal triamcinolone injections have been previously reported, the true incidence remains unknown [33, 34]. Our data are comparable with previous studies reporting an increased incidence (0.74–0.8%) of sterile endophthalmitis following intravitreal triamcinolone injection [35,36,37].
Limitations of the current study include its retrospective design, lack of complete information on the use of perioperative or postoperative prophylactic intracameral or topical antibiotics in the referred cases, the absence of data regarding baseline visual acuity and that clinical assessment and surgeon preference determined the initial treatment)initial tap and inject versus vitrectomy and antibiotic injection). In addition, the relatively small number of culture-positive cases might be the reason we could not demonstrate a correlation between the causative organism and the final VA.
In conclusion, this study showed that patients with endophthalmitis following IVI had significantly worse visual outcomes, were less likely to show any VA improvement following treatment, and presented earlier when compared with patients who developed endophthalmitis following cataract surgery. In addition, the data analysis suggests that although presented with delay, VA outcomes of endophthalmitis cases following cataract surgery performed in private centers might be better than those of patients who were operated on and treated in the same public institution.
References
Jabbarvand M, Hashemian H, Khodaparast M, Jouhari M, Tabatabaei A, Rezaei S (2016) Endophthalmitis occurring after cataract surgery: outcomes of more than 480 000 cataract surgeries, epidemiologic features, and risk factors. Ophthalmology 123:295–301
Miller JJ, Scott IU, Flynn HW Jr et al (2005) Acute-onset endophthalmitis after cataract surgery (2000–2004): incidence, clinical settings, and visual acuity outcomes after treatment. Am J Ophthalmol 139:983–987
Prashant G, Aravind R, Savitri S (2017) Endophthalmitis after cataract surgery: epidemiology, risk factors, and evidence on protection. Curr Opin Ophthalmol 28:67–72
Reibaldi M (2018) Pooled estimates of incidence of endophthalmitis after intravitreal injection of anti-vascular endothelial growth factor agents with and without topical antibiotic prophylaxis. Retina 38:1–11
Sigford DK, Reddy S, Mollineaux C, Schaal S (2015) Global reported endophthalmitis risk following intravitreal injections of anti-VEGF: a literature review and analysis. Clin Ophthalmol 9:773–781
Fileta JB, Scott IU, Flynn HW (2014) Meta-analysis of infectious endophthalmitis after intravitreal injection of anti-vascular endothelial growth factor agents. Ophthalmic Surg Lasers Imaging Retina 45:143–149
Yannuzzi NA, Si N, Relhan N, Kuriyan AE, Albini TA, Berrocal AM, Davis JL, Smiddy WE, Townsend J, Miller D et al (2017) Endophthalmitis after clear corneal cataract surgery: outcomes over two decades. Am J Ophthalmol 174:155–159
Peyman GA, Lad EM, Moshfegh DM (2009) Intravitreal injection of therapeutic agents. Retina 29:875–912
Lalwani GA, Flynn HW Jr, Scott IU, Quinn CM, Berrocal AM, Davis JL, Murray TG, Smiddy WE, Miller D (2008) Acute-onset endophthalmitis after clear corneal cataract surgery (1996–2005). Clinical features, causative organisms, and visual acuity outcomes. Ophthalmology 115:473–476
Falavarjani KG, Nguyen QD (2013) Adverse events and complications associated with intravitreal injection of anti-VEGF agents: a review of literature. Eye 27:787–794
McCannel CA (2011) Meta-analysis of endophthalmitis after intravitreal injection of anti-vascular endothelial growth factor agents: causative organisms and possible prevention strategies. Retina 31:654–661
Schulze-Bonsel K, Feltgen N, Burau H et al (2006) Visual acuities ‘‘hand motion’’ and ‘‘counting fingers’’ can be quantified with the Freiburg visual acuity test. Investig Ophthalmol Vis Sci 47:1236–1240
Scott IU, Flynn HW Jr, Acar N et al (2011) Incidence of endophthalmitis after 20-gauge vs 23-gauge vs 25-gauge pars plana vitrectomy. Graefes Arch Clin Exp Ophthalmol 249:377–380
Taban M, Behrens A, Newcomb RL, Nobe MY, McDonnell PJ (2005) Incidence of acute endophthalmitis following penetrating keratoplasty: a systematic review. Arch Ophthalmol 123(5):605–609
Al-Torbak AA, Al-Shahwan S, Al-Jadaan I et al (2005) Endophthalmitis associated with the Ahmed glaucoma valve implant. Br J Ophthalmol 89:454–458
Mino de Kaspar H, Shriver EM, Nguyen EV et al (2003) Risk factors for antibiotic-resistant conjunctival bacterial flora in patients undergoing intraocular surgery. Graefes Arch Clin Exp Ophthalmol 241:730–733
Kernt M, Kampik A (2010) Endophthalmitis: pathogenesis, clinical presentation, management, and perspectives. Clin Ophthalmol 4:121–135
Gower EW, Keay LJ, Stare DE, Arora P, Cassard SD, Behrens A, Tielsch JM, Schein OD (2015) Characteristics of endophthalmitis after cataract surgery in the United States medicare population. Ophthalmology 122:1625–1632
Morlet N, Li J, Semmens J, Ng J (2003) The endophthalmitis population study of Western Australia (EPSWA). Br J Ophthalmol 87:574–576
Barry P, Seal DV, Gettinby G et al (2006) ESCRS study of prophylaxis of postoperative endophthalmitis after cataract surgery: preliminary report of principal results from a European multicenter study. J Cataract Refract Surg 32:407–410
Shah CP, Garg SJ, Vander JF et al (2011) Outcomes and risk factors associated with endophthalmitis after intravitreal injection of anti-vascular endothelial growth factor agents. Ophthalmology 118:2028–2034
Lemley CA, Han DP (2007) Endophthalmitis: a review of current evaluation and management. Retina 27:662–680
Pathengay A, Moreker MR, Puthussery R et al (2011) Clinical and microbiologic review of culture-proven endophthalmitis caused by multidrug-resistant bacteria in patients seen at a tertiary eye care center in southern India. Retina 31:1806–1811
Simunovic MP, Rush RB, Hunyor AP, Chang AA (2012) Endophthalmitis following intravitreal injection versus endophthalmitis following cataract surgery: clinical features, causative organisms and post-treatment outcomes. Br J Ophthalmol 96:862–866
Endophthalmitis Vitrectomy Study Group (1995) A randomized trial of immediate vitrectomy and of intravenous antibiotics for the treatment of postoperative bacterial endophthalmitis. Arch Ophthalmol 113:1479–1496
Storey P, Dollin M, Pitcher J et al (2014) The role of topical antibiotic prophylaxis to prevent endophthalmitis after intravitreal injection. Ophthalmology 121:283–289
Rizzo JL, Chin EK, Rashid S, Park SS (2012) Exogenous endophthalmitis: post-operative versus post-intravitreal injection. Vis Pan Am 11:38–43
Sommer A, Belkin A, Ofir S, Assia E (2017) Profile of patients presenting for cataract surgery in a public hospital: a 15 year perspective. Isr Med Assoc J 19:109–113
Antcliff RJ, Spalton DJ, Stanford MR et al (2001) Intravitreal triamcinolone for uveitic cystoid macular edema: an optical coherence tomography study. Ophthalmology 108:765–772
Greenberg PB, Martidis A, Rogers AH et al (2002) Intravitreal triamcinolone acetonide for macular oedema due to central retinal vein occlusion. Br J Ophthalmol 86:247–248
Jonas JB, Sofker A (2001) Intraocular injection of crystalline cortisone as adjunctive treatment of diabetic macular edema. Am J Ophthalmol 132:425–427
Sutter FKP, Gillies MC (2003) Pseudo-endophthalmitis after intravitreal injection of triamcinolone. Br J Ophthalmol 87:972–974
Nelson ML, Tennant MTS, Sivalingam A, Regillo CD, Belmont JB, Martidis A (2003) Infectious and presumed noninfectious endophthalmitis after intravitreal triamcinolone acetonide injection. Retina 23:686–691
Taban M, Singh RP, Chung J, Lowder CY, Perez VL, Kaiser PK (2007) Sterile endophthalmitis after intravitreal triamcinolone: a possible association with uveitis. Am J Ophthalmol 144:50–54
Sakamoto T, Enaida H, Kubota T, Nakahara M, Yamakiri K, Yamashita T, Yokoyama M, Hata Y, Murata T, Miyata K et al (2004) Incidence of acute endophthalmitis after triamcinolone-assisted pars plana vitrectomy. Am J Ophthalmol 138:137–138
Fong A, Chan C (2017) Presumed sterile endophthalmitis after intravitreal triamcinolone (Kenalog)-more common and less benign than we thought? Asia Pac J Ophthalmol 6:45–49
Marticorena J, Romano V, Gómez-Ulla F (2012) Sterile endophthalmitis after intravitreal injections. Mediat Inflamm 2012:92812
Funding
None.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
No conflicting relationship exists for any author.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Dar, N., Pillar, S., Friehmann, A. et al. Endophthalmitis after intravitreal injections versus cataract surgery: a 15-year cohort. Int Ophthalmol 40, 73–79 (2020). https://doi.org/10.1007/s10792-019-01153-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10792-019-01153-1