Abstract
Recent studies have shown that, coupled with other environmental factors, aluminium exposure may lead to neurodegeneration resulting in cognitive impairment resembling Alzheimer's disease. Menaquinone, a form of vitamin K2, aids in maintaining healthy bones and avoids coronary calcification. It also has anti-inflammatory and antioxidant properties. Here, we study the neuroprotective effects of vitamin K2 (MK-7) using the animal model of Alzheimer's disease (AD). Aluminium chloride (AlCl3; 100 mg/kg for 3 weeks orally) was administered to Swiss albino mice to induce neurodegeneration and Vitamin K2 (100 g/kg for 3 weeks orally) was applied as treatment. This was followed by behavioural studies to determine memory changes. The behavioural observations correlated with proinflammatory, oxidative, and brain histopathological changes in AlCl3-treated animals with or without vitamin K2 treatment. AlCl3 administration led to memory decline which was partially restored in Vitamin K2 treated animals. Myeloperoxidase levels in the brain increased due to AlCl3-mediated inflammation, which Vitamin K2 prevented. The acetylcholine esterase and oxidative stress markers induced by AlCl3 were reversed by Vitamin K2. Also, Vitamin K2 helps to restore hippocampal BDNF levels and reduced the amyloid β accumulation in AlCl3-administered animals. Additionally, Vitamin K2 protected the hippocampal neurons against AlCl3-mediated damage as observed in histopathological studies. We conclude that Vitamin K2 could partially reverse AlCl3-mediated cognitive decline. It increases acetylcholine and BDNF levels while reducing oxidative stress, neuroinflammation, and β-amyloid deposition, thus protecting the hippocampal neurons from AlCl3-mediated damage.
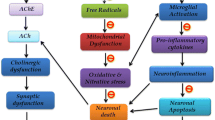
Graphical abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
A variety of endogenous and exogenous agents, such as metals and environmental toxins (aluminum, mercury, etc.), can contribute to neurodegeneration, which is of multifactorial clinical occurrence (del Blanco and Barco 2018; McLachlan et al. 2019). Neurodegeneration can result in different disease conditions, including Alzheimer's Disease (AD) (Zameer et al. 2018). AD is characterized by cognition decline and visuospatial impairment, eventually leading to progressive dementia. It is pathologically characterized by neurofibrillary tangles (NFT), amyloid beta plaque formation, and neuroinflammation primarily in the brain's hippocampal region. Changes in cholinergic neurotransmission, neuroinflammation, and oxidative stress have all been identified as critical contributors to AD.
According to epidemiological evidence, aluminium (Al) in drinking water is linked to neurological conditions including AD (Exley and Esiri 2006; Saba et al. 2017). Prolonged Al exposure can result in pathological alterations similar to Alzheimer's dementia (Exley 2017; Yang et al. 2014). Numerous preclinical studies suggest that long-term exposure to aluminium chloride (AlCl3) leads to a considerable decline in cognitive abilities and increases the activity of the AChE enzyme and oxidative stress (Chiroma et al. 2018; Wei et al. 2017). Al has been associated with the formation of NFTs in AD patients (Saba et al. 2017).
Menaquinone-7 (MK-7) is a variant of Vitamin K2 or Menaquinone is clinically used to improve bone health. A study by Hadipour and co-workers found using PC12 cells that Vitamin K2 can protect against amyloid β (Aβ) and H2O2-mediated cytotoxicity by decreasing the reactive oxygen species (ROS) in the cells. It was also found Aβ and H2O2 can induce apoptosis, while Vitamin K2 pre-treatment showed a decrease in apoptosis (Hadipour et al. 2020).
The aim of the study is to determine the neuroprotective effect of MK-7 against AlCl3-induced neurodegeneration. The current study show that AD-like condition was induced in mice by AlCl3 treatment affecting spatial and recognition memory. Neuropathological alterations included neuroinflammation, oxidative stress, an increase in brain amyloid β levels, and loss of hippocampal neurons. MK-7 partially reversed AlCl3-associated cognitive decline by protecting hippocampal neurons from AlCl3-mediated damage.
Materials and methods
Animal
Male swiss albino mice weighing 22–28 gm were used in the study in accordance with approved protocol of the CPCSEA (Committee for the Purpose of Control and Supervision of Experiments on Animals) Govt. of India under Approved Protocol No. 1972/PH/BIT/ 16/17. Animals were grouped (6 animals/group) and housed in different cages and were exposed to 12 h cycle of light and dark cycle and controlled temperature (21 ± 1 °C) along with the same diet and ad libitum to water.
Experimental design
The mice were acclimatized for 1 week and then were divided randomly to the below-mentioned groups with six animals in each group. All animals were provided with identical diet. Detailed experimental design is shown in Fig. 1.
After completion of dosing, behavioural studies were initiated. After the end of the behavioural studies, the animals from the relevant group were sacrificed under anesthesia followed by the collection of blood and brain for further biochemical and histopathological studies.
Behavioral studies (Sarkar et al. 2021)
Elevated plus maze (EPM) The EPM equipment has two open arms (25 cm × 5 cm) in length with a low (0.5 cm) walls and two closed arm high-walled (16 cm) arms with a open space on center (5 cm × 5 cm). The equipment is elevated at 50 cm (Komada et al. 2008). The time the animal takes to reach to the closed arm from the edge of open arm facing the outside is noted. Four paws inside the closed arms are called Transfer Latency (TL). TL is noted on day 1 also known as familiarising phase and after 24 h on day 2 also known as the trial phase to observe the retention memory (Gupta et al. 2012). The time for each animal is 120 s. The animals which could not go inside the closed arm within 120 s are excluded from the study. To avoid the trail after each animal trial, the maze was wiped with 70% ethanol.
Passive avoidance test (PAT) The step-down avoidance test was performed in a two-chamber attached equipment containing one light and one dark chamber connected with an open gate. The dark compartment is connected with a negative stimulus as with the help of step through the fear and emotional memory is evaluated. To evaluate the step through avoidance on the initial day, the animals were allowed to explore the area for 5 min. Next day, the same animals were placed, and when it enters the dark chamber, they experienced mild shock (0.2–0.4 mA) for 1 s. The animals failed to enter to the dark chamber for up to 60 s is not used in the evaluation. The retention memory is tested after 24 h without the shock application in the dark compartment, with 5 min durations (Gupta et al. 2012). To avoid the trial after each animal trial, the maze was wiped with 70% ethanol.
Morris water maze (MWM) The Morris water maze consists of a circular cistern with a 60 cm radius and 50 cm depth which perimeter was spotted as north (N), south (S), east (E), and west (W). The cistern was filled with water of room temperature (26 ± 2 °C) up to 30 cm. A black-coloured round (4 cm radius) platform was placed in the center of any quadrant submerged 1 cm underneath the water. To conceal the position of the platform, a black synthetic opacifier was used. The trials lasted for 120 s initiating at the most distal position which is the opposite quadrant. For 4 consecutive days, the animals were given trial to memorise the location of the platform, and the animal could not reach the platform for 120 s is excluded from the examination. The 5th day was the probe trial day, where the platform was removed and animals were placed and observed for 120 s. (Wang et al. 2015). As MWM is used to check the special memory, the escape latency and the time duration during the probe trial were recorded through a computer-imaging system (Ethovision ver. 3.0) by Noldus.
Novel object recognition (NOR) A rectangular plastic open box (65 cm × 45 cm × 65 cm) was used. On the initiation day, the animals were placed to explore the whole area. After 24 h, similar object (A + A) was placed and the animals were allowed to explore the objects for familiarization. The animals which did not explore the objects are discarded from the experiment. Next day was the test day where the animals were allowed to explore the arena with two different objects (A + B) where one similar object (A) is replaced with a novel/ new object (B) (Antunes and Biala 2012; Ennaceur 2010). To avoid the trail after each animal trial, the maze was wiped with 70% ethanol. Cognitive function was evaluated by Preference Index (PI) which is the ratio between the exploration time on a familiar and novel object on the test day. More than 50% PI suggested a preference of a new object and less than 50% shows a preference for a familiar object, whereas 50% PI shows that none of the objects were preferred.
Biochemical studies
Estimation of acetylcholinesterase activity (AchE) The neurotransmitter acetylcholine, which is responsible for memory and learning, is broken down by the enzyme acetylcholinesterase through catalysis and promotion. The Ellman's approach was used to evaluate AchE. Using acetylthiocholine iodide (0.075 M) as a substrate and dithiobisnitrobenzoate (0.01 M DTNB) in the hippocampus tissue homogenate, AchE was measured using Ellman's technique. In 30 s intervals, the change in absorption at 412 nm was measured for 3 min.
Estimation of myeloperoxidase activity (MPO) Azurophilic granules of neutrophilic granulocytes store the pro-inflammatory enzyme MPO. Neutrophil activation that lasts for a long time might cause tissue damage and inflammation in the brain. Pulli's approach can be used to measure MPO activity in the brain. Corticohippocampal area was removed from the brain and colon of slaughtered animals. The corticohippocampal tissue was homogenised (10% w/v) with 0.5% hexadecyltrimethylammonium bromide in 0.05 M potassium PBS with a pH of 6.0. The supernatant from the sonicated tissue homogenates was removed after one minute and centrifuged at 4 °C, 10,000 g, for 15 min. H2O2 (0.7 mM) and 3,3′,5,5′-tetramethylbenzidine (2.9 mM constituted with 14.5% DMSO together with 150 mM of sodium PBS of pH 5.4) were added to the supernatant. After adding 2 M of sulfuric acid to the reaction mixture, it was incubated for another 5 min at 37 °C. The absorbance at 450 nm was then measured.
Estimation of reduced glutathione (GSH) In cellular systems, GSH functions as an antioxidant to combat free radicals. Oxidative damage in neurons is caused by GSH deficiency. The Moron's approach was used to estimate GSH. The corticohippocampal area was isolated after the animal was slaughtered. 10% Trichloroacetic acid and pH 8.0 PBS were used to homogenise the tissue (TCA). After centrifuging the supernatant at 15,000 rpm for 15 min at 4 °C and adding 0.6 mM DTNB, absorbance at 412 nm was measured.
Estimation of malondialdehyde (MDA) The supernatant of corticohippocampal brain homogenate was reacted in a test tube with 30% trichloroacetic acid (TCA) and 0.8% thiobarbituric acid (TBA) to quantify the amount of MDA present. Aluminum foil was placed over the test tube, which was then heated in a water bath at 80 C for 30 min. The test tube was then submerged in ice water for 30 min, followed by 15 min of centrifugation at 3000 rpm. At a wavelength of 540 nm, the supernatant was collected, and absorbance was measured.
Estimation of superoxide dismutase (SOD) activity All aerobic cells have a large supply of SODs. The superoxide anion is broken down by SODs into oxygen (O2) and hydrogen peroxide (H2O2). Supernatant of corticohippocampal homogenate was used as the sample, along with reagents of 50 mM sodium carbonate, 25 M nitroblue tetrazolium (NBT), and 0.1 mM ethylenediaminetetraacetic acid (EDTA). Additionally, the reaction was started with 1 mM hydroxylamine hydrochloride. At 560 nm, the absorbance was measured. SOD activity is indicated as a 50% reduction in the total amount of enzyme required to stop the NBT.
Cytokine and chemokine estimation Proinflammatory cytokines called tumour necrosis factor (TNF)-α induce neuroinflammation in the CNS, which impairs learning and memory in animals. The hippocampus region of the brain was removed from slaughtered animals. TNF-α was detected in hippocampus homogenates' supernatant using an ELISA kit (Sigma-Aldrich in St. Louis, Missouri, USA) in accordance with the manufacturer's protocol.
BDNF estimation Brain-derived neurotrophic factor (BDNF) is a neuroprotective and crucial neuronal transmitter. The hippocampus region of the brain was removed from sacrificed animals. Hippocampal homogenates' supernatant was utilized to measure BDNF using an ELISA kit from Sigma-Aldrich in St. Louis, Missouri, USA, in accordance with the manufacturer's recommended technique.
Amyloid β estimation The hippocampus region of the brain was removed from sacrificed animals. Supernatants from hippocampal homogenates were used for Western blot analysis of β-Amyloid. Amyloid beta 1–42 (Mouse; Bioss Inc.) as primary Ab and HRP conjugate anti-mouse secondary Ab (Cell Signalling Technology Inc.) was used for the study. Densitometric estimation was conducted using "Image Studio Digits Ver 5.2".
Histopathology The cervical dislocation procedure was followed by anesthesia and sacrifice of the animals. The brain was removed and preserved for 2 days in 10% formalin (prepared in buffer solution with pH 7.6). The tissues were then immersed in liquefied wax that had been heated to a temperature of between 60 and 70 degree Celsius over the course of the next 2 days, going from 70% alcohol to 100% alcohol in stages. The tissues were divided into sections of 5 m thickness after solidification and put on a microscope slide. To get rid of wax, sectioned tissues were successively cleaned with 100% ethanol once, 95% ethanol twice, and 70% ethanol once. Haematoxylin and eosin (H and E) dye was used to stain the tissues, and then, 95% and 100% ethanol were used to remove any remaining moisture. Using a microscope, the slide was examined after being histocleared. Each mouse's five consecutive coronal slices were produced as slides. Each slide's hippocampus portion was photographed with a Leica camera under 10× and 40× magnification. Twenty consecutive portions of each slide's hippocampus's CA (Cornu Ammonis) 1 and CA3 area were measured for their number of neurons, and the average was computed to yield neurons per millimeter2. Using Image J software, neurons were counted at a magnification of 40 times. Two researchers counted the neurons in the study regions.
Results
Behavioural studies
Compared to control animals, the transfer latency (A) for the AlCl3-treated group was significantly higher (p < 0.001). Also, the preferential index for novel object recognition (E) and step-down latency (B) were significantly lower (p < 0.001) in the AlCl3-treated rats than the control group. The number of entries (C) and the amount of time (D) spent in the target quadrant were significantly lower (p < 0.01) and (p < 0.001), respectively, in the AlCl3-treated animals than in the control animals indicating cognitive impairment. However, supplementing the AlCl3-treated rats with vitamin K2 raised (p < 0.05) the step-down latency, decreased (p < 0.001) the transfer latency, and increased (p < 0.001) the preference for novel objects. In MWM, Vitamin K2 treatment to the AlCl3-treated animals increased the no. of entries (p < 0.01) to the target quadrant and the time spent in the target quadrant (p < 0.001) (Fig. 2).
Effect of AlCl3 and Vitamin K2 on fear-conditioned, recognition, and spatial memory. The transfer latency in (A) EPM amplified (p < 0.001) in the AlCl3-administered animal and reduced (p < 0.001) after Vitamin K2 was administered with AlCl3. The step-down latency (B) in PAT and the (E) preference index (PI) in NOR decreased (p < 0.001) in the AlCl3 group. The step-down latency (p < 0.05) and the preference index (p < 0.001) in NOR were enhanced after Vitamin K2 administration. The number of entries (C) and time spent by animals in a particular quadrant (D) during the probe test reduced (p < 0.01, p < 0.001) significantly in the AlCl3 group compared to the untreated control. The number of entries and time were enhanced after Vitamin K2 administration (p < 0.01, p < 0.001) in the AlCl3 group of animals. *p < 0.05, **p < 0.01, and ***p < 0.001 are statistically significant. All values are expressed as Mean + SEM, n = 10. p < 0.05 is considered to be statistically significant
Biochemical studies
AchE activity When compared to untreated controls, corticohippocampal homogenates from mice that had received AlCl3 showed enhanced AChE activity (p < 0.01). Following the treatment of vitamin K2, AChE levels were dramatically (p < 0.001) lowered (Fig. 3A).
Effect of AlCl3 and Vitamin K2 on AchE and oxidative stress markers. A AchE activity of the brain was enhanced after AlCl3 treatment. Vitamin K2 administration reduced AchE activity in AlCl3-treated animals. B GSH levels decreased in the AlCl3 administered group, whereas Vitamin K2 could resort to the AlCl3-mediated reduced GSH levels. C MDA in the brain was enhanced after AlCl3 treatment. Co-administration of Vitamin K2 reduced the MDA in the brain in AlCl3-treated animals. In the case of SOD, the % inhibition D diminished in the AlCl3 group, whereas Vitamin K2 co-administrated with AlCl3 increased the SOD level. *p < 0.05, **p < 0.01, and ***p < 0.001 are statistically significant. All values are expressed as Mean + SEM, n = 10. p < 0.05 is considered to be statistically significant
Oxidative stress markers Corticohippocampal homogenates of AlCl3-treated animals demonstrated reduced (p < 0.01) GSH (B) and SOD (D) along with increased (p < 0.01) MDA levels compared to untreated controls. GSH and SOD levels increased significantly (p < 0.05, p < 0.001), while MDA (C) levels decreased (p < 0.001) significantly after Vitamin K2 administration to the AlCl3 group of animals (Fig. 3).
BDNF levels AlCl3-treated animal corticohippocampal homogenates showed decreased (p < 0.05) BDNF levels in comparison to untreated controls. Following the administration of vitamin K2, BDNF levels increased considerably in the AlCl3-treated animals (p < 0.05) (Fig. 4A).
Effect of AlCl3 and Vitamin K2 on BDNF and pro-inflammatory markers. A BDNF concentration in the brain was decreased (p < 0.05) after AlCl3 treatment. Vitamin K2 administration increases (p < 0.05) the concentration of BDNF in AlCl3-treated animals. B TNF-α levels enhanced (p < 0.01) in the AlCl3 administered group, whereas Vitamin K2 could resort (p < 0.05) to the AlCl3-mediated enhanced TNF-α levels. C MPO in the brain was enhanced (p < 0.01) after AlCl3 treatment. Co-administration of Vitamin K2 reduced (p < 0.05) the MPO in the brain in AlCl3-treated animals. *p < 0.05, **p < 0.01, and ***p < 0.001 are statistically significant. All values are expressed as Mean + SEM, n = 10. p < 0.05 is considered to be statistically significant
Inflammatory markers
TNF-α When compared to untreated controls, corticohippocampal homogenates from rats that received AlCl3 showed higher (p < 0.01) TNF-α levels, which were considerably reduced (p < 0.05) in vitamin K2-AlCl3 co-administered animals (Fig. 4B).
MPO Corticohippocampal homogenates of AlCl3-treated mice showed higher (p < 0.01) MPO levels than control, which were considerably reduced (p < 0.05) in vitamin K2-AlCl3 co-administered animals (Fig. 4C).
Amyloid β AlCl3-treated animal's corticohippocampal homogenates showed increased (p < 0.05) Aβ protein levels in comparison to untreated controls. AlCl3-treated animals upon administration of vitamin K2 showed a significant decrease in Aβ levels (p < 0.05) (Fig. 5).
Effect of AlCl3 and Vitamin K2 on β-Amyloid levels. Amyloid β activity in the brain was enhanced (p < 0.05) after AlCl3 treatment. Vitamin K2 administration reduced (p < 0.05) Amyloid β activity in AlCl3-treated animals. *p < 0.05 is statistically significant. All values are expressed as Mean + SEM, n = 6. p < 0.05 is considered to be statistically significant
Hippocampal neuronal density Learning and memory have been linked to hippocampal neuronal health (Gareau et al. 2011; Heijtz et al. 2011). Compared to the control, the number of neurons in the hippocampal CA1 and CA3 regions of AlCl3 animals was considerably low (p < 0.001). However, AlCl3 animals on vitamin K2 showed a significant increase in CA1 and CA3 neuronal density (p < 0.001) (Fig. 6).
Effect of AlCl3 and Vitamin K2 on hippocampal neurons. Histopathology of hippocampal sections of (A, B) Control, (C, D) AlCl3, (E, F) AlCl3 + Vitamin K2, (G, H) Vitamin K2, and (I) Neuronal count. AlCl3-treated animals had significantly lower neuronal density in the CA1 (p < 0.001) and CA3 (p < 0.001) regions of the hippocampus when compared to control animals. Treatment with Vitamin K2 and AlCl3 protected neurons in the CA1 (p < 0.001) and CA3 (p < 0.001) regions. Under 10X magnification, representative images from hematoxylin and eosin-stained hippocampus slices. ***p < 0.001. All values are expressed as Mean + SEM n = 6. p < 0.05 is considered statistically significant. CA stands for Cornu Ammonis
Discussion
AlCl3 is a chlorotoxin (neurotoxin) that provokes functional alterations in the cholinergic, dopaminergic, and noradrenergic neurotransmission. Acetylcholine is one of the major neurotransmitters required for memory, while a reduction in Ach leads to impaired recognition memory. The impairment of cholinergic transmission is due to a decrease in acetyltransferase activity or an increase of Acetylcholinesterase. AlCl3 has the propensity to cause impaired cholinergic transmission by affecting the synthesis and release of the neurotransmitter as well as increasing its breakdown (Borai et al. 2017). The administration of AlCl3 can mediate AD (Shunan et al. 2021). In the current study, administration of AlCl3 affected cognitive parameters as revealed using different behavioral studies. It was found to increase transfer latency using plus maze, reduce step-down latency in the passive avoidance test, and preference index in the Novel object recognition test suggesting impaired memory. Morris water maze test which is used to determine spatial memory, AlCl3-administered mice showed decrease in the number of entries and time spent in the target quadrant compared to the control animals indicating reduced special memory. In the biochemical study, we observed AlCl3-mediated increase in AchE level indicating a decrease in corticohippocampal Ach as reported previously (Borai et al. 2017). Here, we show that the co-administration of Vitamin K2 with AlCl3 could partially reverse the cognitive decline produced by AlCl3 by the previously mentioned behavioral evaluation. Antioxidants like GSH and SOD were low, while oxidative stress marker MDA (Ameen and Shafi 2016) was elevated compared to the control group of animals suggesting hippocampal oxidative stress (Firdaus et al. 2022). Lipid peroxidation is caused by free radicals interacting with the lipid parts of the cortex and hippocampus. Additionally, these free radicals cause protein carbonylation by forming oxidised adduct products. Both the phenomena may prove to be toxic to the cells. It is established that brain oxidative damage in the synapses of the cortex and hippocampus causes cognitive impairment (Akter et al. 2020). Neuroinflammation characterized by microglial activation and astrogliosis is one of the primary mediators of neurodegeneration. MPO an inflammatory marker and TNF-α a pro-inflammatory cytokine were also elevated in the AlCl3-administered animals. The anti-inflammatory action of Vitamin K2 was also evident in our results where Vitamin K2 reduced MPO an inflammatory marker and TNF-α a pro-inflammatory cytokine in the corticohippocampal homogenates of AlCl3-treated animals. The amount of BDNF, a marker of neurodegeneration, was reduced in the AlCl3 mice. Amyloid β, a hallmark of AD, got elevated in the AlCl3 mice confirming the development of AD-like conditions in these animals (Abbas et al. 2022). Histopathological studies also showed a decrease in neuronal density in both hippocampal CA1 and CA3 regions compared to control indicating neuronal death in these animals.
In the previous studies, it has been reported that Vitamin K2 can prevent neuronal apoptosis by reducing the ROS levels (Hadipour et al. 2020). In the current study, different oxidative stress markers like GSH and SOD levels increased and MDA levels decreased compared to the AlCl3 group suggesting a decrease in AlCl3-mediated oxidative stress in-vivo. The anti-inflammatory action of Vitamin K2 was also evident in our results where Vitamin K2 reduced MPO an inflammatory marker and TNF-α a pro-inflammatory cytokine in the corticohippocampal homogenates of AlCl3-treated animals. Reduced BDNF expression has been commonly associated with aging and psychiatric disorders (Wang et al. 2021), while this neurotropic factor can also support memory formation (Miranda et al. 2019). BDNF levels increased in the Vitamin K2 treated animals suggesting its neuroprotective functions. Amyloid β aggregation a pathological hallmark of AD was reduced in the vitamin K2-treated mice hippocampus suggesting an improvement in the given disease conditions by Vitamin K2. Histopathological studies substantiated the biochemical observations showing Vitamin K2-associated increase in neuronal density in both CA1 and CA3 regions of the hippocampus compared to AlCl3 animals suggesting neuroprotective actions of Vitamin K2 and improvement of disease conditions.
Conclusion
We conclude that Vitamin K2 could partially reverse AlCl3-mediated cognitive decline. It increases hippocampal acetylcholine and BDNF levels while reducing oxidative stress, neuroinflammation, and β-Amyloid deposition, thus protecting the hippocampal neurons from AlCl3-mediated damage.
Data availability
The available data has been shared. Further quaries ma be directed to authors.
References
Abbas F, Eladl MA, El-Sherbiny M, Abozied N, Nabil A, Mahmoud SM, Mokhtar HI, Zaitone SA, Ibrahim D (2022) Celastrol and thymoquinone alleviate aluminum chloride-induced neurotoxicity: Behavioral psychomotor performance, neurotransmitter level, oxidative-inflammatory markers, and BDNF expression in rat brain. Biomed Pharmacother 151:113072. https://doi.org/10.1016/j.biopha.2022.113072
Akhtar A, Dhaliwal J, Saroj P, Uniyal A, Bishnoi M, Sah SP (2020) Chromium picolinate attenuates cognitive deficit in ICV-STZ rat paradigm of sporadic Alzheimer’s-like dementia via targeting neuroinflammatory and IRS-1/PI3K/AKT/GSK-3β pathway. Inflammopharmacol 28:385–400. https://doi.org/10.1007/s10787-019-00681-7
Ameen N, Shafi S (2016) Biochemical and in-vivo antioxidant parameters for evaluation of memory enhancing activity. Int J Pharm Chem Biol Sci 6(3):265–270
Antunes M, Biala, (2012) The novel object recognition memory: neurobiology, test procedure, and its modifications Cogn. Process 13(2):93–110. https://doi.org/10.1007/s10339-011-0430-z
Borai IH, Ezz MK, Rizk MZ, Aly HF, El-Sherbiny M, Matloub AA, Fouad GI (2017) Therapeutic impact of grape leaves polyphenols on certain biochemical and neurological markers in AlCl3-induced Alzheimer’s disease. Biomed Pharmacother 93:837–851. https://doi.org/10.1016/j.biopha.2017.07.038
Chiroma SM, MohdMoklas MA, Mat Taib CN, Baharuldin MTH, Amon Z (2018) D-galactose and aluminium chloride induced rat model with cognitive impairments. Biomed Pharmacother 103:1602–1608
del Blanco B, Barco A (2018) Impact of environmental conditions and chemicals on the neuronal epigenome. Curr Opin Chem Biol 45:157–165. https://doi.org/10.1016/j.cbpa.2018.06.003
Ellman EL, Courtney KD, Andres V Jr, Featherstone RM (1961) A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem Pharmacol 7(2):88–95. https://doi.org/10.1016/0006-2952(61)90145-9
Ennaceur A (2010) One-trial object recognition in rats and mice: methodological and theoretical issues. Behav Brain Res 215(2):244–254. https://doi.org/10.1016/j.bbr.2009.12.036
Exley C (2017) Aluminum should now be considered a primary etiological factor in Alzheimer’s disease. J Alzheimers Dis Rep 1(1):23–25. https://doi.org/10.3233/ADR-170010
Exley C, Esiri MM (2006) Severe cerebral congophilic angiopathy coincident with increased brain aluminium in a resident of Camelford, Cornwall, UK. J Neurol Neurosurg Psychiatry 77(7):877–879. https://doi.org/10.1136/jnnp.2005.086553
Firdaus Z, Kumar D, Singh SK, Singh TD (2022) Centella asiatica alleviates AlCl3-induced cognitive impairment, oxidative stress, and neurodegeneration by modulating cholinergic activity and oxidative burden in rat brain. Biol Trace Elem Res 200:5115–5126. https://doi.org/10.1007/s12011-021-03083-5
Gareau MG, Wine E, Rodrigues DM, Cho JH, Whary MT, Philpott DJ, MacQueen G, Sherman PM (2011) Bacterial infection causes stress-induced memory dysfunction in mice. Gut 60(3):307–317 (gut.2009.202515)
Gupta A, Jalhan SH, Jindal A, Upmanyu N (2012) Various animal models to check learning and memory—a review. Int J Pharm 4:91–95
Hadipour E, Tayarani-Najaran Z, Fereidoni M (2020) Vitamin K2 protects PC12 cells against Aβ (1–42) and H2O2-induced apoptosis via p38 MAP kinase pathway. Nutr Neurosci 23(5):343–352. https://doi.org/10.1080/1028415X.2018.1504428
Heijtz RD, Wang S, Anuar F, Qian Y, Bjorkholm B, Samuelsson A, Hibberd ML, Forssberg H, Pettersson S (2011) Normal gut microbiota modulates brain development and behaviour. Proc Natl Acad Sci 108(7):3047–3052. https://doi.org/10.1073/pnas.1010529108
Komada M, Takao K, Miyakawa T (2008) Elevated plus maze for mice. JoVE-J vis Exp 22:e1088. https://doi.org/10.3791/1088
McLachlan DRC, Bergeron C, Alexandrov PN, Walsh WJ, Pogue AI, Percy ME, Kruck TPA, Fang Z, Sharfman NM, Jaber V, Zhao Y, Li W, Lukiw WJ (2019) Aluminum in neurological and neurodegenerative disease. Mol Neurobiol 56:1531–1538. https://doi.org/10.1007/s12035-018-1441-x
Miranda M, Morici JF, Zanoni MB, Bekinschtein P (2019) Brain-derived neurotrophic factor: a key molecule for memory in the healthy and the pathological brain. Front Cell Neurosci. https://doi.org/10.3389/fncel.2019.00363
Moron MS, Depierre JW, Mannervik B (1979) Levels of glutathione, glutathione reductase and glutathione S-transferase activities in rat lung and liver. BBA-Gen Subjects 582(1):67–78. https://doi.org/10.1016/0304-4165(79)90289-7
Pulli B, Ali M, Forghani R, Schob S, Hsieh KL, Wojtkiewicz G, Linnoila JJ, Chen JW (2013) Measuring myeloperoxidase activity in biological samples. PLoS ONE. https://doi.org/10.1371/journal.pone.0067976
Saba K, Rajnala N, Veeraiah P, Tiwari V, Rana RK, Lakhotia SC, Patel AB (2017) Energetics of excitatory and inhibitory neurotransmission in aluminum chloride model of Alzheimer’s disease: reversal of behavioral and metabolic deficits by Rasa Sindoor. Front Mol Neurosci 10:323. https://doi.org/10.3389/fnmol.2017.00323
Saputra WD, Aoyama N, Komai M, Shirakawa H (2019) Menaquinone-4 suppresses lipopolysaccharide-induced inflammation in MG6 mouse microglia-derived cells by inhibiting the NF-κB signaling pathway. Int J Mol Sci 20(9):2317. https://doi.org/10.3390/ijms20092317
Sarkar SR, Mazumder PM, Chatterjee K, Sarkar A, Adhikary M, Mukhopadhyay K, Banerjee S (2021) Saccharomyces boulardii ameliorates gut dysbiosis associated cognitive decline. Physiol Behav 236:113411. https://doi.org/10.1016/j.physbeh.2021.113411
Shunan D, Yu M, Guan H, Zhou Y (2021) Neuroprotective effect of Betalain against AlCl3-induced Alzheimer’s disease in Sprague Dawley Rats via putative modulation of oxidative stress and nuclear factor kappa B (NF-κB) signaling pathway. Biomed Pharmacothe 137:111369. https://doi.org/10.1016/j.biopha.2021.111369
Tanaka S, Kondo H, Kanda K, Ashino T, Nakamachi T, Sekikawa K, Iwakura Y, Shioda S, Numazawa S, Yoshida T (2011) Involvement of interleukin-1 in lipopolysaccaride-induced microglial activation and learning and memory deficits. J Neurosci Res 89(4):506–514. https://doi.org/10.1002/jnr.22582
Wang T, Hu X, Liang S, Li W, Wu X, Wang L, Jin (2015) Lactobacillus fermentum NS9 restores the antibiotic induced physiological and psychological abnormalities in rats. Benef Microbes 6(5):707–717. https://doi.org/10.3920/BM2014.0177
Wang CS, Kavalali ET, Monteggia LM (2021) BDNF signaling in context: from synaptic regulation to psychiatric disorders. Cell. https://doi.org/10.1016/j.cell.2021.12.003
Wei Y, Liu D, Zheng Y, Li H, Hao C, Ouyang W (2017) Protective effects of kinetin against aluminum chloride and D-galactose induced cognitive impairment and oxidative damage in mouse. Brain Res Bull 134:262–272. https://doi.org/10.1016/j.brainresbull.2017.08.014
Yang WN, Hu XD, Han H, Shi LL, Feng GF, Liu Y, Qian YH (2014) The effects of valsartan on cognitive deficits induced by aluminum trichloride and d-galactose in mice. Neurol Res 36(7):651–658. https://doi.org/10.1179/1743132813Y.0000000295
Zameer S, Najmi AK, Vohora D, Akhtar M (2018) Bisphosphonates: future perspective for neurological disorders. Curr Pharmacol Rep PR 70(5):900–907. https://doi.org/10.1016/j.pharep.2018.03.011
Funding
The work is not funded by any agency.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chatterjee, K., Mazumder, P.M. & Banerjee, S. Vitamin K2 protects against aluminium chloride-mediated neurodegeneration. Inflammopharmacol 31, 2675–2684 (2023). https://doi.org/10.1007/s10787-023-01290-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10787-023-01290-1