Abstract
Eugenia pohliana DC.(Myrtaceae) is used in folk medicine by communities in Brazil. However, there are no reports on its biological activity. This is the first study to identify the components of E. pohliana essential oil (EpEO) and evaluate their antinociceptive and anti-inflammatory activities in an in vivo model at doses of 25, 50, and 100 mg/kg. The essential oil (EO) was obtained by hydrodistillation, and the analysis was performed by gas chromatography coupled with mass spectrometry. Antinociceptive activity was evaluated by writhing tests, tail movement, and formalin (neurogenic and inflammatory pain); naloxone was used to determine the nociception mechanism. Anti-inflammatory activity was assessed by oedema and peritonitis tests. We found that (E)-β-caryophyllene (BCP) (15.56%), δ-cadinene (11.24%) and α-cadinol (10.89%) were the major components. In the writhing test, there was a decrease in writing by 42.95–70.70%, in the tail movement, an increase in latency time by 69.12–86.63%, and in the formalin test, there was a reduction in pain neurogenic by 29.54–61.74%, and inflammatory pain by 37.42–64.87%. The antinociceptive effect of EpEO occurs through the activation of opioid receptors. In addition, a reduction in inflammation by 74.93‒81.41% was observed in the paw edema test and inhibition of the influx of leukocytes by 51.86‒70.38% and neutrophils by 37.74‒54.72% in the peritonitis test. It was concluded that EpEO has antinociceptive effect by the opioid pathway, as shown by the inhibitory effect of naloxone, and anti-inflammatory actions, and that its use does not cause hemolytic damage or behavioral change.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Inflammation is the response of the immune system to infections or tissue damage (Matsuda et al. 2019). Excessive or persistent activation can compromise organs and systems, leading to decompensation and organ dysfunction (Hirano et al. 2021). Pain is one of the signs of the inflammatory process; when it is constant or intermittent, it is a debilitating factor that impairs an individual's quality of life (McParland et al. 2021).
Analgesic and anti-inflammatory drugs are used to alleviate disturbances caused by the inflammatory process. However, many drugs have several adverse effects ranging from mild to severe. One of the main analgesics, morphine, can promote respiratory depression, constipation, nausea, vomiting, tolerance, and dependence (Azevedo Neto et al. 2020) whereas non-steroidal anti-inflammatory drugs (NSAIDs), such as indomethacin, can cause skin reactions and gastrointestinal, renal, and cardiovascular complaints (Wongrakpanich et al. 2018). Owing to these limitations, there is a need to seek alternative measures to treat pain and inflammation that may also help in the treatment of diseases (Lee et al. 2021; Russo et al. 2021).
Among natural medicinal products, essential oils extracted from plants have great pharmaceutical relevance (Mondal et al. 2021). Several activities have been described for these oils, such as anti-inflammatory (Han and Parker 2017), antinociceptive (Costa et al. 2020), antimicrobial (Souza et al. 2021), leishmanicidal (Nunes et al. 2021), larvicidal (Silva et al. 2021), antifungal (Kujur et al. 2021), antioxidant, and antiprotozoan (Sampaio et al. 2021). The presence of these activities encourages the discovery of secondary metabolites with pharmacological applications (Jacoby et al. 2021).
Many botanical families contain volatile compounds with medicinal properties, and the Myrtaceae family, which is widely distributed in the Caatinga biome, is a source of essential oils responsible for important health benefits associated (Macedo et al. 2021). One of the largest genera of the Myrtaceae family includes Eugenia, which encompasses approximately 1,000 species (Mazine et al. 2014), and contains essential oils with antioxidant properties (Franco et al. 2021) as well as healing (Silva et al. 2018); antimicrobial (Bezerra Filho et al. 2020), anti-inflammatory, and antinociceptive effects (Costa et al. 2020).
Thus, due to the variety of biological activities attributed to the genus, it is expected that the species Eugenia pohliana DC. and its essential oil have pharmacological potential; however, to our knowledge, there are still no reports of chemical studies or pharmacological activities of the preparations from this species. Thus, our objective was to determine, for the first time, the chemical composition of the essential oil of E. pohliana leaves and the antinociceptive and anti-inflammatory properties induced by the action of other species of the genus using in vivo models.
Materials and methods
Herbal material
Leaves of Eugenia pohliana DC. were collected in June 2019 in Serra do Catimbau, in the municipality of Buíque, Pernambuco, Brazil (8° 30′57″S 37° 20′ 59″W) a region of the Caatinga and registered in the National System of Management of Genetic Heritage and Associated Traditional Knowledge (A08E18B). An exsiccata (number 54805) was deposited in the Herbarium of Professor Vasconcelos Sobrinho (PEUFR) of the Department of Biology of the Federal Rural University of Pernambuco (UFRPE), Recife, Brazil. Then, the plant material was subjected to washing with running water, followed by natural drying in a clean, dry, and insect-free place for 6 h.
Extraction
To obtain the essential oil from Eugenia pohliana DC. leaves (EpEO), the plant material after drying was ground and subjected to hydrodistillation (600 g, 4 h). The essential oil (EO) obtained was weighed, and the yield was calculated in % (m/m) and immediately stored in a dark flask under refrigeration (−5 °C) until used for analysis.
Chemical composition of essential oil
Operating conditions, the (CG-MS) Shimadzu GCMS-QP2010 model. Nitrogen was used as gas from the GC transported by an Rtx®-5MS silica capillary column (30 mx 0.25 mm × 0.25 μm), at a flow rate of 1 L/min and an inlet pressure of 30 psi. and MS was optimized as follows: 70 eV, extraction gas (He) 13.6 mL.min-1 and pressure of 53.5 kPa. Using the following temperature program: 100 °C (3 min) to 310 °C (3.5 °C/min). The identification of chemical components was performed by comparing the results obtained through the gas chromatography test coupled to mass spectrometry with results already existing in the literature, specifically in the NIST08 Mass Spectral Library, a mass spectral library of the GC–MS system, Wiley 9th Edition Mass Spectral Ratio RegistryTM and other published data (Adams 2007).
Animals
Male Swiss mice used weighed between 30 and 35 g, aged between 8 and 10 weeks, and were obtained from the bioterium of the Keizo Asami Immunopathology Laboratory (LIKA), Federal University of Pernambuco (UFPE) maintained under standard conditions for 12-h light/dark cycle, at 22 ± 2 °C, with water ad libitum available. The experimental protocols used were approved by the Animal Use Ethics Committee (CEUA) of UFPE, protocol number 0070/2020. On the day of testing, the mice fasted for 6 h before each test.
Haemolytic potential
Briefly, blood was collected by cardiac puncture in mice and centrifuged (3000 rpm, 10 min) where the supernatant was discarded and obtaining the erythrocyte concentrate (pellet) for the assay. A saline solution of the pellet (2%) was incubated in a microtube together with different concentrations of EpEO (156.25–5000 mg/mL) at room temperature (25 °C). After 30 min of incubation, the samples were centrifuged (3000 rpm, 5 min) and the supernatant collected and the hemoglobin released quantified in a spectrophotometer at 540 nm of absorbance (Jimenez et al. 2003).
Antinociceptive activity
Treatments
To evaluate the antinociceptive activity, groups of animals (n = 6) were treated orally with saline solution (negative control), EpEO (25, 50, or 100 mg/kg per os) 1 h before the evaluations, while the reference drug, indomethacin (10 mg/kg/ip) and morphine (10 mg/kg/ip) 30 min before. In the investigation of the antinociceptive mechanism, the animals were pretreated with a non-selective opioid receptor antagonist (naloxone 2 mg/kg) 30 min before treatment with the highest effective concentration of EpEO (100 mg/kg).
Acetic acid-induced abdominal writhing test
The abdominal contortion model was performed according to Oliveira et al. (2018). Thirty-six mice were immobilized and received an intraperitoneal nociception-inducing agent (acetic acid 0.85% v/v; 0.1 mL/10 g b.w) and divided into 6 treatment groups. With the aid of a chronometer, the number of contortions performed by each animal was observed for 5 min after the application of acetic acid.
Tail movement
Initially, the mice were pre-selected where their tails were immersed in a glass container containing distilled water (55 ± 1 °C) of which, which showed withdrawal reflex before 5 s, were selected and divided into five groups. Twenty-four hours later, treatments were administered, and again the same procedure was performed at times 0, 30, 60, 90, and 120 min. With a stopwatch, the time the animal took to perform a reflex action (tail movement) caused by heat, limited to 20 s, was recorded (Khatun et al. 2015).
Formalin test
Following the Hunskaar and Hole (1987) protocol, thirty-six animals were randomly divided into 6 groups. Afterward, the mice were immobilized and received formalin (10%, 20 uL) in the left hind paw and the licking time by the animal at the application site was recorded during two phases: the first phase (0–5 min) representing neurogenic pain; and second (15–30 min) cytokine-mediated inflammatory pain.
Investigation of antinociceptive activity mechanisms
A group of mice received pretreatment with naloxone and 30 min later, 100 mg/kg of EpEO was applied. After 60 min, the formalin test was followed.
Anti-inflammatory activity
Treatments
To evaluate the anti-inflammatory activity, sixty male Swiss mice were used, thirty for each test. Groups of animals (n = 6) were treated orally with saline solution (negative control), EpEO at concentrations (25, 50 or 100 mg/kg b.w) or indomethacin (20 mg/kg) 1 h before the evaluations.
Carrageenan-induced paw edema
Following the protocol by Winter et al. (1962), the paw edema test was performed to determine the mean variation of the volume of the paw (%) measured with a plethysmometer, during a predetermined period (0, 60, 120, 180, and 240 min) after the application of the phlogistic agent (15 μL of 2% carrageenan) in the plantar region of the left paw. For the results, the formula was used: (percentage change (%) = ((final paw volume − initial paw volume))/(initial paw volume) × 100).
Peritonitis
Carrageenan-induced inflammation of the peritoneum test followed the protocol described by Oliveira et al. (2016). Briefly, 1% carrageenan (0.1 ml, 10 g b.w) was administered intraperitoneally and after 4 h the animals were anesthetized and an application of 2 ml of heparinized PBS was injected into the peritoneal region followed by a gentle abdominal massage. Then, an aspiration of the peritoneal exudate for the quantification of leukocytes using an automatic analyzer was performed. Additionally, to investigate the role of regulator proteins in the inflammatory response, the quantification of cytokines levels (IL-1β and TNF-α) in the peritoneal exudate was performed using the Millipore 7-plex kit (Millipore Corp., Billerica, MA, USA) as described by Oliveira et al (2021).
Adverse effects assessment
To assess possible adverse effects caused by treatment with EpEO, groups of animals (n = 6) were submitted to open field and elevated plus-maze tests. Treatments were performed using a negative control group (0.9% saline) and EpEO (1000 mg/kg).
Open field
Following Archer's protocols (1973) Mice from the control and EpEO-treated groups (1000 mg/kg) were positioned in the central part of a circular acrylic arena (75 cm × 75 cm × 50 cm) dividing it into four squares vertically and horizontally. Then, for 5 min, the mice were evaluated for the number of crossings (rearings) in the squares and the act of self-cleaning (crossing).
Elevated plus-maze test
Several solid substances cause anxiety when ingested. Thus, the anxiety level of a mouse submitted to the elevated plus-maze test was evaluated as described by Kraeuter et al. (2019). Both groups of mice are individually allocated in the central part of the platform and recorded the number of entries and the time spent in open and closed arms. The animal's anxiety index was determined using the following equation: AI = 1 − [{(time spent in the open arms / total time in the maze) + (number of open-arm entries/total entries in the maze)}/2], the closer to 1 the value, the more anxious the animal was during the test.
Statistical analysis
Data analysis was performed using GraphPad Prism® version 8.0 and expressed as mean values with standard deviation (± SD). Using one-way analysis of variance (ANOVA) we calculated statistically significant differences (p < 0.001) followed by Bonferroni or Dunnett”s.
Results and discussion
Chemical characterization
This is the first report of the chemical composition of the essential oil extracted from Eugenia pohliana DC. leaves. The chemical components, their respective retention indices (RI), and relative amounts of EO extracted from E. pohliana leaves are shown in Table 1. The EpEO presented a yellowish color, and the chemical characterization showed 38 compounds, comprising 98.73% of the total. The major components were sesquiterpenes: (E)-β-caryophyllene (15.56%), δ-cadinene (11.24%), and α-cadinol (10.89%).
The presence of (E)-β-caryophyllene (BCP), an odorous bicyclic sesquiterpene, common in plants of this family and has been reported as a component of Eugenia brejoensis Mazine species that have antimicrobial activity (Mendes et al. 2018); and Eugenia calycina Cambess., which has larvicidal (Silva et al. 2021), cytotoxic, and antimicrobial activity (Sousa et al. 2015). BCP is also found in Eugenia dysenterica DC., which has healing activity (Silva et al. 2018); Eugenia sulcata Spring ex Mart. which has insecticidal activity (Gonzalez et al. 2014) and Eugenia egensis DC. which has cytotoxic and antioxidant properties (Silva et al. 2017).
BCP, a phytocannabinoid, acts as a ligand for cannabinoid receptor-2 (CB-2) (Gertsch et al. 2008), a part of the endocannabinoid system that is involved in cell signaling (Meccariello et al. 2020). When activated, this receptor inhibits mediators of inflammation, contributing to the relief of pain and inflammation (Maayah et al. 2020). Compounds that interact with the endocannabinoid system have shown promise for the treatment of various diseases (Shah et al. 2021). Other activities can be attributed to BCP, such as antimicrobial activity against Staphylococcus mutans (Yoo and Jwa. 2018), antiherpetic (Astani et al. 2011), wound healing (Koyama et al. 2019), anti-inflammatory (Brito et al. 2019), and protective effects against ischemic brain injury (Chang et al. 2013).
Another compound present in the composition of EpEO was δ-cadinene, found in the oil of Eugenia caryophyllata Thunb. (Wang et al. 2021) which showed antinociceptive and anti-inflammatory activities (Taher et al. 2015) and Eugenia brasiliensis Lam., which showed significant antimicrobial activity (Silva et al. 2019). This compound has demonstrated acaricidal activity against Psoroptes cuniculi (Guo et al. 2017).
The oxygenated sesquiterpene α-cadinol is a termiticide (Morikawa et al. 2014) reported in the composition of E. brejoensis (Mendes et al. 2018) and Eugenia pyriformis Cambess. (Durazzini et al. 2019). To date, there are no reports of antinoceptive and anti-inflammatory activities for these species, but other pharmacological activities have been described, such as antimicrobial activity in E. brejoensis (Bezerra Filho et al. 2020) and E. pyriformis (Souza et al. 2021).
Other species of the Myrtaceae family with biological activity contain α-cadinol, such as Myrcia tomentosa (Aubl.) DC. which has antimicrobial activity (Sa et al. 2017), Eucalyptus occidentalis Endl., which has shown repellent and insecticidal activity (Bande-Borujeni et al. 2018) and Plinia trunciflora (O.Berg) Kausel, which has antimicrobial activity against yeasts and bacteria (Lago et al. 2011).
Hemolytic activity
Haemolysis generally occurs by lysis of erythrocytes, potentially leading to hemolytic anemia. A variety of substances can induce hemolysis, making assessment of this activity extremely important (Barros et al. 2016). Thus, EpEO had a hemolysis rate in the range of 0.74 and 2.2% at the concentrations tested. These data show low toxicity, as they do not present significant hemolytic activity (Jimenez et al. 2003).
Antinociceptive activity
Acetic acid-induced writhing test
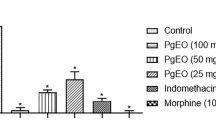
The acetic acid-induced writhing model, although not specific, is a simple and sensitive test that is the standard for evaluating candidate drugs with antinociceptive action (Hunskaar and Hole 1987). EpEO reduced writhing in animals by 42.95%, 52.94%, and 70.70% when treated with doses of 25, 50, and 100 mg/kg, respectively. The standard drugs morphine and indomethacin promoted a reduction of 98.83% and 85.88%, respectively, when compared to the control group (Fig. 1).
The genus Eugenia has shown positive results in tests of writhing induced by acetic acid; for example, the methanolic extract of Eugenia uniflora L. leaves lead to 60% and 74% reduction in writhing at concentrations of 100 and 200 mg/kg, respectively. The EO from E. caryophyllata leaves inhibited 89.6% of abdominal writhing induced by acetic acid in mice in the treatment with a concentration of 100 mg/kg (Taher et al. 2015), which was attributed to the presence of BCP as a major compound in its chemical composition (Wang et al. 2021).
Tail Immersion
The tail-immersion test is a highly sensitive test for opioid drugs and is ideal for the evaluation of drugs that act on the central nervous system (Oliveira et al. 2018; Khatun et al. 2015). EpEO promoted greater analgesic action after 90 min of administration, with an increase in latency time by 86.63–69.12% compared to control (Fig. 2). Treatment with 100 mg/kg of EpEO or morphine showed an antinociceptive action above 50% at assessment time points. These results indicate that EpEO has a central analgesic effect similar to that of morphine (Kotlinska et al. 2013).
Formalin test
In the formalin-induced nociception test, EpEO showed antinociceptive action at all doses and during both phases of the test. A significant reduction (p < 0.001) in time spent licking the paw was achieved; 61.74%, 65.5%, and 29.54% in the first phase (neurogenic pain) and in 64.87%, 64.3%, and 37.42% in the second phase (inflammatory pain) at doses of 100, 50, and 25 mg/kg, respectively, compared to the control (Fig. 3). Morphine, the standard drug, reduced licking time in both phases (86.5–86.3%), while indomethacin was effective only in the second phase of the test (92%) when compared to the control.
The EO of Eugenia candolleana DC. leaves, with composition dominated by BCP, inhibited the licking time after formalin injection by up to 55.65% in the first phase; however, the results were more significant in the second phase, with inhibition of up to 96.7% at a dose of 100 mg/kg (Guimarães et al. 2009). According to the literature, BCP is among the major compounds of this species (Neves et al. 2017). Santos et al. (2020) reported that acetone extract from Myrciaria floribunda (H.West ex Willd.) O.Berg fruits reduced licking time by up to 86.52% in the treatment with 100 mg/kg in the first phase and 82.58% in the second phase. (Silva Barbosa et al. 2020) described the chemical composition of the EO of M. floribunda fruit peels as dominated by BCP and δ-cadinene (Silva Barbosa et al. 2020).
Commercially obtained BCP alone has a significant effect on inflammatory pain in the formalin test; however, it does not have any treatment effect on neurogenic pain (Klauke et al. 2014). Thus, the significant results in the inflammatory pain phase of EpEO treatment may be related to the presence of BCP.
To investigate the mechanism of action and antinociceptive activity using the formalin test, we pretreated the rats with naloxone, a non-selective opioid receptor antagonist, followed by treatment with EpEO 100 mg/kg or morphine. The action of EpEO was almost entirely inhibited, similar to morphine, in the two phases of the formalin test (Fig. 3). These results suggest that the antinociceptive effect of EpEO occurs through the activation of opioid receptors (Lewanowitsch et al. 2006), as suggested by the tail-flick test.
Anti-inflammatory activity
Carrageenan-induced paw oedema
As previously mentioned, there are no studies on the biological activities of E. pohliana; however, based on evidence of anti-inflammatory action of the genus Eugenia in addition to the expressive action of the treatment with EO in the second phase (inflammatory phase) of the formalin test, we investigated the anti-inflammatory potential using the paw edema test to screen for this possible effect. Indeed, treatment with EpEO promoted a reduction of 74.93–81.4% in paw oedema induced by carrageenan, while indomethacin inhibited it by 81.1% at 3 h when compared to control (Fig. 4). The activity of EpEO and indomethacin remained constant until the end of the test.
Similarly, Costa et al. (2020) described that an Eugenia stipitata McVaugh EO had an anti-inflammatory effect on paw oedema, reducing it by up to 96.94% after treatment with 250 mg/kg. According to Sobeh et al. (2019), the paw edema test with the methanolic extract of E. uniflora leaves reduced edema by 32%. In this study, the inhibition potential for Cyclooxygenase-1 (COX-1) and Cyclooxygenas -2 (COX-2) was determined. The results show significant inhibition, with IC50 values of 5.63 μg/mL for COX-1 and 0.18 μg/mL for COX-2. The inhibition of these enzymes prevents the production of inflammatory substances and the action of anti-inflammatory NSAIDs (Trinh et al. 2021). Mesquita et al. (2017) reported that BCP is a major volatile chemical extracted from E. uniflora.
Peritonitis
After confirming the anti-edematogenic effect of the EpEO treatment, the carrageenan-induced peritonitis test was performed as a model of acute inflammation. Oral administration of EpEO promoted a significant reduction (p < 0.001) of 51.86‒70.38% and 37.74‒54.72% in the influx of leukocytes and neutrophils, respectively, at all doses tested. Indomethacin inhibited 79.01% of leukocyte migration by 71.69% of neutrophils (Table 2).
Lazarini et al. (2020) showed that oral treatment with ethanolic extract of Eugenia selloi B.D.Jacks. fruits promoted a decrease in neutrophils by 58% and 70% at concentrations of 3 and 10 mg/kg, respectively. A methanolic extract of E. uniflora leaves (100 mg/kg) reduced the number of leukocytes to 4.84 ± 1.97 × 106 mL compared to untreated animals (10.04 ± 2.64 × 106 mL) (Sobeh et al. 2019). BCP, the main compound of EpEO, reduces the peritoneal migration of neutrophils at a dose of 100 μL (Brito et al. 2019). Such anti-inflammatory effects may be related to its ability to activate CB-2 receptors, as the absence of these receptors suppresses the inflammatory response in animal models (Gertsch et al. 2008).
Costa et al (2022) reported the effects of essential oil from Psidium glaziovianum Kiaersk. P. promoted a reduction of inflammatory cytokines and attributed the presence of monoterpenes in its composition such as 1,8-cineole, α-pinene, and β-pinene. In our study, treatment with EpEO (25, 50 and 100 mg/kg per os) promoted a significant reduction (p < 0.001) in TNF-α levels (51.27, 55.57 and 66.58%) and IL-1β (51.40, 60.16 and 73.64%) (Fig. 5). As expected, the indomethacin group reduced TNF-α and IL-1β by 76.59 and 72.71%, respectively. The hypothesis of anti-inflammatory action is possibly related to the presence of β-caryophyllene (Aguilar-Ávila et al. 2019) in the composition of EpEO. These findings confirm the anti-inflammatory action of EpEO in a dose-dependent.
Adverse effects assessment
Open field and elevated plus-maze test
In general, analgesic agents have direct side effects on the central nervous system (CNS). To test for these side effects, we used the open field and elevated plus maze tests, which have become a standard for measuring anxiety, sedation, and activity not only in rodents but also in several other animals (Prut and Belzung 2003). The absence of the animal's exploratory behavior indicates that the substance tested has sedative and depressant activity (File and Wardill 1975; Mujumdar et al. 2000).
Thus, to investigate the occurrence of this possible effect in the treatment with EpEO, we tested the animals in the open field and elevated plus maze test after a dose of 1000 mg/kg, which is 10 times greater than the therapeutic dose used in our tests. Treatment with EpOE in the open field test showed no significant differences in rearing (20 ± 2) and crossing (58 ± 12). However, diazepam resulted in an absence of the animals' exploratory behavior with values of 8 ± 1 for rearing and 37 ± 6 for crossing. The control group showed rearing of 25 ± 3 and crossing of 75 ± 7.
In the LCE test, treatment with EpEO did not promote anxiety symptoms, presenting an anxiety index of 0.54, which was not significant when compared with the control (0.41). Diazepam, on the other hand, had an anxiety index of 0.09, indicating that its neurological activity caused anxiety in the animals.
Thus, our results revealed that EpEO treatment did not affect exploratory activity and anxiety. In contrast, diazepam at 1 mg/kg showed significant values in relation to the control. Based on these results, EpEO can be considered as a possible herbal medicine candidate for the treatment of pain and inflammation.
Conclusion
The essential oil of Eugenia pohliana leaves showed biological activity and reduced inflammatory and non-inflammatory pain as well as acute inflammatory processes. Antinociceptive activity has a mechanism of action via the opioid pathway, the same pathway as morphine. Regarding the chemical components present in EpEO, the large presence of BCP may be the underlying reason for its activity in relieving pain and inflammation. BCP is a CB-2 ligand that acts in the regulation of inflammatory mediators. Additionally, EpEO showed no effect on behavior and its hemolytic potential was not significant; thus, it is a promising candidate for the development of new analgesic and anti-inflammatory molecules.
Data Availability
We declare for all purposes that the National Council for Scientific Research and Development (CNPq), the Coordination for the Improvement of Higher Education Personnel (Capes), and the Fundação de Amparo a Ciência e Tecnologia do Estado de Pernambuco (FACEPE) have made available scholarships of research and financial support to researchers, for the acquisition of inputs and materials.
References
Adams RP (2007) Identification of essential oil components by gas chromatography/mass spectrometry, 4th edn. Allured Publ. Corp, Illinois
Aguilar-Ávila DS, Flores-Soto ME, Tapia-Vázquez C, Pastor-Zarandona OA, López-Roa RI, Viveros-Paredes JM (2019) β-caryophyllene, a natural sesquiterpene, attenuates neuropathic pain and depressive-like behavior in experimental diabetic mice. J Med Food 22(5):460–468. https://doi.org/10.1089/jmf.2018.0157
Archer J (1973) Tests for emotionality in rats and mice: a review. Anim Behav 21:205–235. https://doi.org/10.1016/S0003-3472(73)80065-X
Astani A, Reichling J, Schnitzler P (2011) Screening for antiviral activities of isolated compounds from essential oils. Evidence-based complement. Altern Med 2011:1–8. https://doi.org/10.1093/ecam/nep187
Azevedo Neto J, Costanzini A, Giorgio R, Lambert DG, Ruzza C, Calò G (2020) Biased versus partial agonism in the search for safer opioid analgesics. Molecules 25:3870. https://doi.org/10.3390/molecules25173870
Bande-Borujeni S, Zandi-Sohani N, Ramezani L (2018) Chemical composition and bioactivity of essential oil from Eucalyptus occidentalis leaves against two stored product pests. Int J Trop Insect Sci 38:216–223. https://doi.org/10.1017/S1742758418000085
Barbosa DCS, Holanda VN, Assis CRD, Aguiar JCROF, Nascimento PH, Silva WV, Navarro DMAF, Silva MV, Lima VLM, Correia MTS (2020) Chemical composition and acetylcholinesterase inhibitory potential, in silico, of Myrciaria floribunda (H. West ex Willd.) O. Berg fruit peel essential oil. Ind Crops Prod 151:112372. https://doi.org/10.1016/j.indcrop.2020.112372
Barros FJ, Costa RJO, Cesário FRAS, Rodrigues LB, Costa JGM, Coutinho HDM, Galvao HBF, Menezes IRA (2016) Activity of essential oils of Piper aduncum anf and Cinnamomum zeylanicum by evaluating osmotic and morphologic fragility of erythrocytes. Eur J Integr Med 8:505–512. https://doi.org/10.1016/j.eujim.2016.02.011
Bezerra Filho CM, Silva LCN, Silva MV, Løbner-Olesen A, Struve C, Krogfelt KA, Correia MTS, Oliva MLV (2020) Antimicrobial and antivirulence action of Eugenia brejoensis essential oil in vitro and in vivo invertebrate models. Front Microbiol 11:424. https://doi.org/10.3389/fmicb.2020.00424
Brito LF, Oliveira HBM, Neves Selis N, Souza CLS, Júnior MNS, Souza EP, da Silva LSC, Souza Nascimento F, Amorim AT, Campos GB, Oliveira MV, Yatsuda R, Timenetsky J, Marques LM (2019) Anti-inflammatory activity of β -caryophyllene combined with docosahexaenoic acid in a model of sepsis induced by Staphylococcus aureus in mice. J Sci Food Agric 99:5870–5880. https://doi.org/10.1002/jsfa.9861
Chang H-J, Kim J-M, Lee J-C, Kim W-K, Chun HS (2013) Protective effect of β-caryophyllene, a natural bicyclic sesquiterpene, against cerebral ischemic injury. J Med Food 16:471–480. https://doi.org/10.1089/jmf.2012.2283
Costa WK, Oliveira JRS, Oliveira AM, Santos IBS, Cunha RX, Freitas AFS, Silva JWLM, Silva VBG, Aguiar JCROF, Silva AG, Navarro DMAF, Lima VLM, da Silva MV (2020) Essential oil from Eugenia stipitata McVaugh leaves has antinociceptive, anti-inflammatory and antipyretic activities without showing toxicity in mice. Ind Crops Prod 144:112059. https://doi.org/10.1016/j.indcrop.2019.112059
Costa WK, Nascimento MF, Santos CRB, Navarro DMAF, Napoleão TH, Correia MTS, Brayner FA, Oliveira AM, Silva MV (2022) Oral administration of essential oil from Psidium glaziovianum Kiaersk leaves alleviates pain and inflammation in mice. Inflammopharmacology 30(2):599–607
Durazzini AMS, Machado CHM, Fernandes CC, Miranda M (2019) Chemical composition and effect of hydrodistillation times on the yield of essential oil from Eugenia pyriformis leaves. Orbital Electron J Chem 11:334–338. https://doi.org/10.17807/orbital.v11i5.1221
File SE, Wardill AG (1975) Validity of head-dipping as a measure of exploration in a modified hole-board. Psychopharmacologia 44:53–59. https://doi.org/10.1007/BF00421184
Franco CJP, Ferreira OO, Moraes ÂABM, Varela ELP, Nascimento LD, Percário S, Oliveira MS, Andrade EHA (2021) Chemical composition and antioxidant activity of essential oils from Eugenia patrisii Vahl, E. punicifolia (Kunth) DC., and Myrcia tomentosa (Aubl.) DC., leaf of family myrtaceae. Molecules 26:3292. https://doi.org/10.3390/molecules26113292
Gertsch J, Leonti M, Raduner S, Racz I, Chen J-Z, Xie X-Q, Altmann K-H, Karsak M, Zimmer A (2008) Beta-caryophyllene is a dietary cannabinoid. Proc Natl Acad Sci 105:9099–9104. https://doi.org/10.1073/pnas.0803601105
Gonzalez MS, Lima BG, Oliveira AFR, Nunes DD, Fernandes CP, Santos MG, Tietbohl LAC, Mello CB, Rocha L, Feder D (2014) Effects of essential oil from leaves of Eugenia sulcata on the development of agricultural pest insects. Rev Bras Farmacogn 24:413–418. https://doi.org/10.1016/j.bjp.2014.05.003
Guimarães AG, Melo MS, Bonfim RR, Passos LO, Machado SMF, Ribeiro AS, Sobral M, Thomazzi SM, Quintans-Júnior LJ (2009) Antinociceptive and anti-inflammatory effects of the essential oil of Eugenia candolleana DC., myrtaceae, on mice. Rev Bras Farmacogn 19:883–887. https://doi.org/10.1590/S0102-695X2009000600016
Guo X, Shang X, Li B, Zhou XZ, Wen H, Zhang J (2017) Acaricidal activities of the essential oil from Rhododendron nivale Hook. f. and its main compund, δ-cadinene against Psoroptes cuniculi. Vet Parasitol 236:51–54. https://doi.org/10.1016/j.vetpar.2017.01.028
Han X, Parker TL (2017) Anti-inflammatory activity of clove (Eugenia caryophyllata) essential oil in human dermal fibroblasts. Pharm Biol 55:1619–1622. https://doi.org/10.1080/13880209.2017.1314513
Hirano SI, Ichikawa Y, Sato B, Yamamoto H, Takefuji Y, Satoh F (2021) Potential therapeutic applications of hydrogen in chronic inflammatory diseases: possible inhibiting role on mitochondrial stress. Int J Mol Sci 22:2549. https://doi.org/10.3390/ijms22052549
Hunskaar S, Hole K (1987) The formalin test in mice: dissociation between inflammatory and non-inflammatory pain. Pain 30:103–114. https://doi.org/10.1016/0304-3959(87)90088-1
Jacoby RP, Koprivova A, Kopriva S (2021) Pinpointing secondary metabolites that shape the composition and function of the plant microbiome. J Exp Bot 72:57–69. https://doi.org/10.1093/jxb/eraa424
Jimenez PC, Fortier SC, Lotufo TMC, Pessoa C, Moraes MEA, Moraes MO, Costa-Lotufo LV (2003) Biological activity in extracts of ascidians (Tunicata, Ascidiacea) from the northeastern Brazilian coast. J Exp Mar Bio Ecol 287:93–101. https://doi.org/10.1016/S0022-0981(02)00499-9
Khatun A, Imam MZ, Rana MS (2015) Antinociceptive effect of methanol extract of leaves of Persicaria hydropiper in mice. BMC Complement Altern Med 15:63. https://doi.org/10.1186/s12906-015-0558-y
Klauke AL, Racz I, Pradier B, Markert A, Zimmer AM, Gertsch J, Zimmer A (2014) The cannabinoid CB2 receptor-selective phytocannabinoid beta-caryophyllene exerts analgesic effects in mouse models of inflammatory and neuropathic pain. Eur Neuropsychopharmacol 24:608–620. https://doi.org/10.1016/j.euroneuro.2013.10.008
Kotlinska JH, Gibula-Bruzda E, Witkowska E, Chung NN, Schiller PW, Izdebski J (2013) Antinociceptive effects of two deltorphins analogs in the tail-immersion test in rats. Peptides 39:103–110. https://doi.org/10.1016/j.peptides.2012.11.008
Koyama S, Purk A, Kaur M, Soini H, Novotny M, Davis K, Kao C, Matsunami H, Mescher A (2019) Beta-caryophyllene enhances wound healing through multiple routes. PLoS ONE 14:1–32. https://doi.org/10.1371/journal.pone.0216104
Kraeuter AK, Guest PC, Sarnyai Z (2019) The elevated plus maze test for measuring anxiety-like behavior in rodents. In: Guest P (eds) Pre-clinical models. Methods in molecular biology. Humana Press, New York, pp 69–74. https://doi.org/10.1007/978-1-4939-8994-2_4
Kujur A, Kumar A, Prakash B (2021) Elucidation of antifungal and aflatoxin B1 inhibitory mode of action of Eugenia caryophyllata L. essential oil loaded chitosan nanomatrix against Aspergillus flavus. Pestic Biochem Physiol 172:104755. https://doi.org/10.1016/j.pestbp.2020.104755
Lago JHG, Souza ED, Mariane B, Pascon R, Vallim MA, Martins RCC, Baroli AA, Carvalho BA, Soares MG, dos Santos RT, Sartorelli P (2011) Chemical and biological evaluation of essential oils from two species of myrtaceae—Eugenia uniflora L. and Plinia trunciflora (O. Berg) Kausel. Molecules 16:9827–9837. https://doi.org/10.3390/molecules16129827
Lazarini JG, Franchin M, Soares JC, Nani BD, Massarioli AP, Alencar SM, Rosalen PL (2020) Anti-inflammatory and antioxidant potential, in vivo toxicity, and polyphenolic composition of Eugenia selloi B.D.Jacks. (pitangatuba), a Brazilian native fruit. PLoS ONE 15:1–16. https://doi.org/10.1371/journal.pone.0234157
Lee W, Lee CH, Lee J, Jeong Y, Park JH, Nam IJ, Lee DS, Lee HM, Lee J, Yun N, Song J, Choi S, Kim S (2021) Botanical formulation, TADIOS, alleviates lipopolysaccharide (LPS)-Induced acute lung injury in mice via modulation of the Nrf2-HO-1 signaling pathway. J Ethnopharmacol 270:113795. https://doi.org/10.1016/j.jep.2021.113795
Lewanowitsch T, Miller JH, Irvine RJ (2006) Reversal of morphine, methadone and heroin induced effects in mice by naloxone methiodide. Life Sci 78:682–688. https://doi.org/10.1016/j.lfs.2005.05.062
Maayah ZH, Takahara S, Ferdaoussi M, Dyck JRB (2020) The molecular mechanisms that underpin the biological benefits of full-spectrum cannabis extract in the treatment of neuropathic pain and inflammation. Biochim Biophys Acta Mol Basis Dis 1866:165771. https://doi.org/10.1016/j.bbadis.2020.165771
Macedo JGF, Rangel JML, Santos MO, Camilo CJ, Costa JGM, Souza MMA (2021) Therapeutic indications, chemical composition and biological activity of native Brazilian species from Psidium genus (myrtaceae): a review. J Ethnopharmacol 278:114248. https://doi.org/10.1016/j.jep.2021.114248
Matsuda M, Huh Y, Ji RR (2019) Roles of inflammation, neurogenic inflammation, and neuroinflammation in pain. J Anesth 33:131–139. https://doi.org/10.1007/s00540-018-2579-4
Mazine FF, Souza VC, Sobral M, Forest F, Lucas E (2014) A preliminary phylogenetic analysis of Eugenia (Myrtaceae: Myrteae), with a focus on neotropical species. Kew Bull 69:9497. https://doi.org/10.1007/s12225-014-9497-x
McParland A, Moulton J, Brann C, Hale C, Otis Y, Ganter G (2021) The brinker repressor system regulates injury-induced nociceptive sensitization in Drosophila melanogaster. Mol Pain 17:17448069211037400. https://doi.org/10.1177/17448069211037401
Meccariello R, Santoro A, D’Angelo S, Morrone R, Fasano S, Viggiano A, Pierantoni R (2020) The epigenetics of the endocannabinoid system. Int J Mol Sci 21:1113. https://doi.org/10.3390/ijms21031113
Mendes JF, Martins HHA, Otoni CG, Santana NA, Silva RCS, Silva AG, Silva MV, Correia MTS, Machado G, Pinheiro ACM, Piccoli RH, Oliveira JE (2018) Chemical composition and antibacterial activity of Eugenia brejoensis essential oil nanoemulsions against Pseudomonas fluorescens. LWT 93:659–664. https://doi.org/10.1016/j.lwt.2018.04.015
Mesquita PRR, Nunes EC, Santos FN, Bastos LP, Costa MAPC, Rodrigues MF, Andrade JB (2017) Discrimination of Eugenia uniflora L. biotypes based on volatile compounds in leaves using HS-SPME/GC–MS and chemometric analysis. Microchem J 130:79–87. https://doi.org/10.1016/j.microc.2016.08.005
Mondal M, Quispe C, Sarkar C, Bepari TC, Alam MJ, Saha S, Ray P, Rahim MA, Islam MT, Setzer WN, Salehi B, Ahmadi M, Abdalla M, Sharifi-Rad J, Kundu SK (2021) Analgesic and anti-inflammatory potential of essential oil of Eucalyptus camaldulensis Leaf. In vivo and in silico studies. Nat Prod Commun 16:1–16. https://doi.org/10.1177/1934578X211007634
Morikawa T, Ashitani T, Kofujita H, Takahashi K (2014) Antitermitic activity of extracts from Chamaecyparis obtusa branch heartwood. Eur J Wood Wood Prod 72:651–657. https://doi.org/10.1007/s00107-014-0830-8
Mujumdar AM, Naik DG, Waghole RJ, Kulkarni DK, Kumbhojkar MS (2000) Pharmacological studies on sterculia foetida leaves. Pharm Biol 38:13–17. https://doi.org/10.1076/1388-0209(200001)3811-BFT013
Neves IA, Rezende SRF, Kirk JM, Pontes EG, Carvalho MG (2017) Composition and larvicidal activity of essential oil of Eugenia candolleana DC. (Myrtaceae) against Aedes aegypti. Rev Virtual Quim 9:2305–2315. https://doi.org/10.21577/1984-6835.20170138.
Nunes TAL, Costa LH, Sousa JMS, Souza VMR, Rodrigues RRL, Val MCA, Pereira ACTC, Ferreira GP, Silva MV, Costa JMAR, Véras LMC, Diniz RC, Rodrigues KAF (2021) Eugenia piauhiensis Vellaff. essential oil and γ-elemene its major constituent exhibit antileishmanial activity, promoting cell membrane damage and in vitro immunomodulation. Chem Biol Interact 339:109429. https://doi.org/10.1016/j.cbi.2021.109429
Oliveira AM, Freire MOL, Silva WAV, Ferreira MRA, Paiva PMG, Soares LAL, Medeiros PL, Carvalho BM, Napoleão TH (2018) Saline extract of Pilosocereus gounellei stem has antinociceptive effect in mice without showing acute toxicity and altering motor coordination. Regul Toxicol Pharmacol 95:289–297. https://doi.org/10.1016/j.yrtph.2018.04.004
Oliveira AM, Nascimento MF, Ferreira MRA, Moura DF, Souza TGS, Silva GC, Ramos EHS, Paiva PMG, Medeiros PL, Silva TG, Soares LAL, Chagas CA, Souza IA, Napoleão TH (2016) Evaluation of acute toxicity, genotoxicity and inhibitory effect on acute inflammation of an ethanol extract of Morus alba L. (Moraceae) in mice. J Ethnopharmacol 194:162–168. https://doi.org/10.1016/j.jep.2016.09.004
Oliveira AM, Freitas AFS, Costa MDS, Torres MKS, Castro YAA, Almeida AMR, Paiva PMG, Carvalho BM, Napoleao TH (2021) Pilosocereus gounellei (Cactaceae) stem extract decreases insulin resistance, inflammation, oxidative stress, and cardio-metabolic risk in diet-induced obese mice. J Ethnopharmacol 265:113327
Prut L, Belzung C (2003) The open field as a paradigm to measure the effects of drugs on anxiety-like behaviors: a review. Eur J Pharmacol 463:3–33. https://doi.org/10.1016/S0014-2999(03)01272-X
Russo C, Edwards KD, Margetts G, Kleidonas S, Zaibi NS, Clapham JC, Zaibi MS (2021) Effects of Salvia officinalis L. and Chamaemelum nobile (L.) extracts on inflammatory responses in two models of human cells: primary subcutaneous adipocytes and neuroblastoma cell line (SK-N-SH). J Ethnopharmacol 268:113614. https://doi.org/10.1016/j.jep.2020.113614
Sa FAS, Paula JAM, Santos PA, Oliveira LAR, Oliveira GAR, Liao LM, Paula JR, Silva MRR (2017) Phytochemical analysis and antimicrobial activity of Myrcia tomentosa (Aubl.) DC. Leaves Mol 22:1–10. https://doi.org/10.3390/molecules22071100
Sampaio MGV, Santos CRB, Vandesmet LCS, Santos BS, Santos IBS, Correia MTS, Martins ALB, Silva LCN, Menezes IRA, Gomez MCV, Silva MV (2021) Chemical composition, antioxidant and antiprotozoal activity of Eugenia gracillima Kiaersk. leaves essential oil. Nat Prod Res 35:1914–1918. https://doi.org/10.1080/14786419.2019.1644506
Santos IBS, Santos B, Oliveira JRS, Costa WK, Zagmignan A, Silva LCN, Ferreira MRA, Lermen VL, Lermen MSBS, Silva AG, Ximenes RM, Soares LAL, Paiva PMG, Lima VLM, Correia MTS, Silva MV (2020) Antioxidant action and in vivo anti-inflammatory and antinociceptive activities of Myrciaria floribunda fruit peels: possible involvement of opioidergic system. Adv Pharmacol Pharm Sci 2020:1–11. https://doi.org/10.1155/2020/1258707
Shah SA, Gupta AS, Kumar P (2021) Emerging role of cannabinoids and synthetic cannabinoid receptor 1/cannabinoid receptor 2 receptor agonists in cancer treatment and chemotherapy-associated cancer management. J Cancer Res Ther 17:1–9. https://doi.org/10.4103/jcrt.JCRT_488_18
Silva JKR, Andrade EH, Barreto LH, Silva NCF, Ribeiro A, Montenegro RC, Maia JG (2017) Chemical composition of four essential oils of Eugenia from the Brazilian Amazon and their cytotoxic and antioxidant activity. Medicines 4:1–10. https://doi.org/10.3390/medicines4030051
Silva MVSG, Silva SA, Teixera TL, Oliveira A, Morais SAL, Silva CV, Espindola LS, Sousa RMF (2021) Essential oil from leaves of Eugenia calycina Cambes: natural larvicidal against Aedes aegypti. J Sci Food Agric 101:1202–1208. https://doi.org/10.1002/jsfa.10732
Silva ROM, Castro JWG, Dantas Junior OM, Araújo ACJ, Leandro MKNS, Costa RJO, Pinto LL, Leandro LMG, Silva LE, Amaral W, Parabocz LD, Ferriani AP, Garcia B, Maia BHLNS, Rocha JE, Bezerra CF, Freitas TS, Costa MS, Campina FF, Matias EFF, Iriti M, Coutinho HDM (2019) Photoinduced antibacterial activity of the essential oils from Eugenia brasiliensis lam and Piper mosenii C. DC. by blue led light. Antibiotics 8:242. https://doi.org/10.3390/antibiotics8040242
Silva SMM, Costa CRR, Gelfuso GM, Guerra ENS, Nóbrega YKM, Gomes SM, Pic-Taylor A, Fonseca-Bazzo YM, Silveira D, Magalhães PO (2018) Wound healing effect of essential oil extracted from Eugenia dysenterica DC (Myrtaceae) leaves. Molecules 24:1–16. https://doi.org/10.3390/molecules24010002
Sobeh M, El-Raey M, Rezq S, Abdelfattah MA, Petruk G, Osman S, El-Shazly AM, El-Beshbishy HA, Mahmoud MF, Wink M (2019) Chemical profiling of secondary metabolites of Eugenia uniflora and their antioxidant, anti-inflammatory, pain killing and anti-diabetic activities: a comprehensive approach. J Ethnopharmacol 240:111939. https://doi.org/10.1016/j.jep.2019.111939
Souza AM, Oliveira VB, Oliveira CF, Betim FCM, Pacheco SDG, Cogo LL, Miguel OG, Miguel MD (2021) Chemical composition and in vitro antimicrobial activity of the essential oil obtained from Eugenia pyriformis Cambess (Myrtaceae). Braz Arch Biol Technol 64:1–10. https://doi.org/10.1590/1678-4324-2021200663
Sousa RMF, Morais SAL, Vieira RBK, Napolitano DR, Guzman VB, Moraes TS, Cunha LCS, Martins CHG, Chang R, Aquino FJT, Nascimento EA, Oliveira A (2015) Chemical composition, cytotoxic, and antibacterial activity of the essential oil from Eugenia calycina Cambess. leaves against oral bacteria. Ind Crops Prod 65:71–78. https://doi.org/10.1016/j.indcrop.2014.11.050
Taher YA, Samud AM, El-Taher FE, ben-Hussin G, Elmezogi JS, Al-Mehdawi BF, Salem HA, (2015) Experimental evaluation of anti-inflammatory, antinociceptive and antipyretic activities of clove oil inmice. Libyan J Med 10:1–8. https://doi.org/10.3402/ljm.v10.28685
Trinh HKT, Pham LD, Le KM, Park HS (2021) Pharmacogenomics of hypersensitivity to non-steroidal anti-inflammatory drugs. Front Genet 12:1–10. https://doi.org/10.3389/fgene.2021.647257
Wang W, Zhang Y, Yang Z, He Q (2021) Effects of incorporation with clove (Eugenia caryophyllata) essential oil (CEO) on overall performance of chitosan as active coating. Int J Biol Macromol 166:578–586. https://doi.org/10.1016/j.ijbiomac.2020.10.215
Winter CA, Risley EA, Nuss GW (1962) Carrageenin-Induced Edema in Hind Paw of the Rat as an Assay for Antiinflammatory Drugs. Exp Biol Med 111:544–547. https://doi.org/10.3181/00379727-111-27849
Wongrakpanich S, Wongrakpanich A, Melhado K, Rangaswami J (2018) A comprehensive review of non-steroidal anti-inflammatory drug use in the elderly. Aging Dis 9:143–150. https://doi.org/10.14336/AD.2017.0306
Yoo H-J, Jwa S-K (2018) Inhibitory effects of β-caryophyllene on Streptococcus mutans biofilm. ArCh Oral Biol 88:42–46. https://doi.org/10.1016/j.archoralbio.2018.01.009
Acknowledgements
The authors express their gratitude to the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) for financial support and investigator research grants (DMAFN, THN, and MTSC). We are also grateful to the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES; Finance Code 001) and the Fundação de Amparo à Ciência e Tecnologia do Estado de Pernambuco (FACEPE) for financial support.
Funding
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Author information
Authors and Affiliations
Contributions
ALD, JBG, WKC, AMO performed the methodologies. WKC, AMO analyzed and interpreted the results. BOV performed the essential oil extraction. ALD, WKC, AMO wrote the article. JCROFA, DMAFN performed the analysis and chemical characterization of essential oil. AMO, MVS performed a critical review of the final version of the article. MTSC, THN, AMO, MVS contributed reagents, materials and analytical tools.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
do Nascimento, A.L., Guedes, J.B., Costa, W.K. et al. Essential oil from the leaves of Eugenia pohliana DC. (Myrtaceae) alleviate nociception and acute inflammation in mice. Inflammopharmacol 30, 2273–2284 (2022). https://doi.org/10.1007/s10787-022-01067-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10787-022-01067-y