Abstract
Autoimmune diseases (ADs) are featured by the body’s immune responses being directed against its own tissues, resulting in prolonged inflammation and subsequent tissue damage. Currently, the exact pathogenesis of ADs remains not fully elucidated. Semaphorin-3A (Sema3A), a secreted member of semaphorin family, is a potent immunoregulator during all immune response stages. Sema3A has wide expression, such as in bone, connective tissue, kidney, neurons, and cartilage. Sema3A can downregulate ADs by suppressing the over-activity of both T-cell and B-cell autoimmunity. Moreover, Sema3A shows the ability to enhance T-cell and B-cell regulatory properties that control ADs, including systemic lupus erythematosus, rheumatoid arthritis, multiple sclerosis, and systemic sclerosis. However, it can also induce ADs when overexpressed. Together, these data strongly suggest that Sema3A plays a pivotal role in ADs, and it may be a promising treatment target for these diseases. In the present review, we focus on the immunological functions of Sema3A and summarize recent studies on the involvement of Sema3A in the pathogenesis of ADs; the discoveries obtained from recent findings may translate into novel therapeutic agent for ADs.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Autoimmune diseases (ADs) are a pathophysiological state wherein immune responses are directed against, and damage, the body’s own tissues (Guan et al. 2017). ADs contain a broad spectrum of disorders, including systemic lupus erythematosus (SLE), rheumatoid arthritis (RA), multiple sclerosis (MS), and systemic sclerosis (SSc). ADs, being able to activate complement and deposit in many sites, induce multi-organ inflammation and subsequent irreversible organ injuries such as in skin, joint, and kidney (Vadasz and Toubi 2012). Although significant progresses have been achieved in the treatment strategies of ADs, the exact cause and pathogenesis of ADs are still unclear. Unraveling the complex pro-inflammatory mechanisms in the pathogenesis of ADs was the focus in a large number of studies for several decades. These mechanisms include cellular and humoral immune responses followed by the production of pro-inflammatory cytokines such as tumor necrosis factor (TNF), interferons (IFNs), and interleukin 17 (IL-17), as well as the overexpression of co-stimulatory molecules such as CD86 and CD40 on effector T cells, and the elevated production of B-cell activating factor (BAFF) (Kim 2015; Vadasz et al. 2015b). In the last decade, the involvement of regulatory immune responses in ADs has drawn much attention, the normal function of which was proven to maintain immune-mediated homeostasis. The early identified immunoregulators include regulatory T cells (Tregs) and the inhibitory cytokines, such as interleukin 10 (IL-10) and transforming growth factor beta (TGF-β) (Yuan et al. 2014). Nevertheless, more recently, other regulatory responses were emerging, such as the involvement of B regulatory cells (Bregs) and the regulatory semaphorins in suppressing autoimmunity.
In recent years, emerging data indicate that semaphorins, which were identified originally as guidance cues for developing axons, play regulatory roles in the immune system (Kumanogoh et al. 2005; Vadasz and Toubi 2013). The semaphorins are implicated in most stages of both normal and pathological immune responses and have been revealed to participate in autoimmune pathologies (Mizui et al. 2009; Vadasz et al. 2010; Vadasz and Toubi 2013). Semaphorins comprise a large family of proteins that are either secreted or membrane bound. Semaphorin-3A (Sema3A), a secreted member of semaphorin family, is identified as a potent immunoregulator in all stages of immune response, including both the early initiation and the late phase of inflammatory processes (Suzuki et al. 2008; Vadasz and Toubi 2013). Sema3A was also demonstrated to enhance T-cell and B-cell regulatory properties. It could inhibit the responsiveness of human monocytes and T cells to chemokine gradients (Ji et al. 2009; Delaire et al. 2001; Takamatsu et al. 2010a). Therefore, Sema3A has been recently considered as a new paradigm in the pathogenesis of ADs, such as SLE (Vadasz et al. 2011, 2012, 2015a; Vadasz and Toubi 2012), RA (Perez et al. 2016; Yoshida et al. 2014), MS (Costa et al. 2015; Kremer et al. 2015; Eixarch et al. 2013a; Gutiérrez-Franco et al. 2016; Okuno et al. 2011; Rezaeepoor et al. 2017; Williams et al. 2007), and SSc (Rimar et al. 2014; Olewicz-Gawlik and Samborski 2016; Vadasz et al. 2015d). Sema3A exerts an effective immunosuppressive role in these diseases through inhibiting the over-activity of both T-cell and B-cell autoimmunity. In addition, Sema3A could upregulate the expression of forkhead box P3 (FoxP3) in Tregs (Cozacov et al. 2017), further suggesting the potential of Sema3A as an immune regulator of inflammation in ADs.
In the present review, we focus on the immunological functions of Sema3A and summarize recent evidences on the involvement of Sema3A in the pathogenesis of ADs; the discoveries obtained from recent findings may translate into novel therapeutic agent for ADs.
Literature search method
“All relevant studies regarding the association of Sema3A with autoimmune diseases (SLE, RA, MS and SSc) were searched in Pubmed, Embase and the Cochrane Library databases. The following keywords and subject terms were used in the literature search: ‘Systemic lupus erythematosus’ OR ‘SLE’ AND ‘Semaphorin-3A’ OR ‘Sema3A’; ‘Rheumatoid arthritis’ OR ‘RA’ AND ‘Semaphorin-3A’ OR ‘Sema3A’; ‘Multiple sclerosis’ OR ‘MS’ AND ‘Semaphorin-3A’ OR ‘Sema3A’; ‘Systemic sclerosis’ OR ‘SSc’ AND ‘Semaphorin-3A’ OR ‘Sema3A’. Variants for all keywords were used to increase the number of studies returned by the search. All resulting articles were then screened and carefully reviewed.”
Sema3A and its receptors
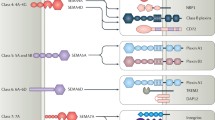
Currently, the semaphorin family contains 20 members in mice and humans, and 5 members in Drosophila; they are classified into 8 classes, namely 1–7 and V (Committee 1999; Yazdani and Terman 2006). The 8 classes of semaphorins have different overall structural and sequence characteristics, but all members of this family possess a conserved extracellular domain that consists of approximately 500 amino acids, which is termed semaphorin domain (Yazdani and Terman 2006). Sema3A, originally named collapsin, is a member of class 3 semaphorins (Piaton et al. 2011); semaphorin domain, plexin–semaphorin–integrin (PSI) domain, immunoglobulin-like domain, and basic domain were constitute of Sema3A (Püschel et al. 1995; Elizabeth et al. 1995; Kolodkin et al. 1993; Fiore and Püschel 2003) (Fig. 1). It was found in the parallel experiments that a neuronal growth cone collapsing factor associated with chicken brain membranes was biochemically purified (Committee 1999; Luo et al. 1993). Sema3A is secreted by a variety of cell types such as neurocyte and several immune cells (Schlahsa et al. 2009).
The structure of semaphorin-3A (Sema3A). Sema3A consists of semaphorin domain, plexin–semaphorin–integrin (PSI) domain, immunoglobulin-like domain, and basic domain. The sema domain is expressed as a seven-bladed ß propeller structure. The PSI domain included cysteine-rich plexin, sema, and integrin. Plexins and transcription factors share the immunoglobulin-like domain. The basic domain contains a conserved extracellular domain of about 500 amino acids
Comparing with other semaphorins, Sema3A has wide expression, such as in bone (Wright et al. 1995), cancer cells (Rieger et al. 2003), connective tissue (Püschel et al. 1995), heart (Wright et al. 1995), kidney (Villegas and Tufro 2002), neurons (Wright et al. 1995), cartilage (Wright et al. 1995), and teeth (Loes et al. 2001). The expression patterns of the Sema3A are the best characterized in the nervous system, particularly during development (Fiore and Püschel 2003). Sema3A also has many functions, for instance, Sema3A participates in the process of cell death (Gagliardini and Fankhauser 1999), cell adhesion and aggregation (Kashiwagi 2005), and cell migration and patterning (Miao et al. 1999). Sema3A repulses peripheral axons and contributes to proper wiring of peripheral nervous system (Kawasaki et al. 2002; Taniguchi et al. 1997). Sema3A also can positively contribute to the observed partial regeneration of peripheral nerves (Scarlato 2003).
Plexin and neuropilin families are two major receptors for semaphorins. Plexin 1 and neuropilin-1 (NRP-1) form a stable complex. Plexin 1 alone does not bind Sema3A, but the NRP-1/plexin 1 complex has a higher affinity for Sema3A than does NRP-1 alone. While Sema3A binding to NRP-1 does not alter non-neuronal cell morphology, Sema3A interaction with NRP-1/plexin 1 complexes induces adherent cells to round up. Expression of a dominant-negative plexin 1 in sensory neurons blocks Sema3A-induced growth cone collapse. Sema3A treatment leads to the redistribution of growth cone NRP-1 and plexin 1 into clusters. Therefore, physiologic Sema3A receptors consist of NRP-1/plexin 1 complexes (Takahashi et al. 1999). Sema3A binds to NRP-1 with a high affinity and signals through plexin 1; however, Sema3A can also signal through plexin-A4 in different pathways (Eixarch et al. 2013b).
Immunological functions of Sema3A
In recent years, there has been increasing evidence regarding the involvement of the semaphorins in the regulation of the immune system. Roth et al. proposed a viewpoint that one of the major discoveries in the field of immunity is probably the identification of a potentially crucial role of semaphorin functions in the immune system (Roth et al. 2008). Suzuki et al. reported that Sema3A is a potent immunoregulator during all immune response stages. Sema3A is involved in lymphocyte migration to the thymus and from the bloodstream to inflamed tissues. It is expressed on many types of immune cells such as B cells, T cells, and dendritic cells (DCs), but mainly on Tregs, leading to the inhibition of T effector cell proliferation and production of pro-inflammatory cytokines (Roth et al. 2008; Vadasz et al. 2010, 2012).
Sema3A normally binds to B cells (including memory/autoreactive B cells); it downregulates the expression of CD100 (a stimulatory molecule) and upregulates the expression of CD72 (inhibitory molecule) on B cells. When cocultured with Sema3A, IL-10 and TGF-β expressions were enhanced in CD19+CD25high Breg cells; Breg cell properties were enhanced by Sema3A through upregulating CD72 expression on B cells (Vadasz et al. 2014). Bregs are a subgroup of activated B cells, which play a crucial role in maintaining self-tolerance and preventing autoimmunity, demonstrating the immune-regulatory role of Sema3A.
When Sema3A binds Hepler T-cell type 1 (Th1) and Hepler T-cell type 17 (Th17) cells, it decreases their proliferation and the production of tumor necrosis factor (TNF), interferons (IFNs), and IL-17 (Vadasz and Toubi 2012). Rimar et al. and Suzuki et al. reported that Sema3A plays a regulatory role in immune responses, mainly serves as a negative regulatory for T cells in physiological and pathological immune responses (Rimar et al. 2015a). The inhibitory effect of Sema3A on T cells is mediated by the blockade of Ras/mitogen-activated protein kinase (MAPK) signaling pathway (Catalano 2006; Vadasz and Toubi 2013). The addition of Sema3A to DC/T-cell co-cultures significantly inhibited allogeneic T-cell proliferation. Sema3A acted directly on T cells as it could block the anti-CD3/CD28-stimulated T-cell proliferation. Sema3A exerts its immunomodulatory functions through the blockage of actin cytoskeleton reorganization, affecting TCR polarization and interfering with early TCR signal transduction events such as ZAP-70 or focal adhesion kinase phosphorylation (Lepelletier et al. 2006). The other immunoregulatory mechanisms of Sema3A may be maintaining self-tolerance; it was shown that Sema3A triggers a proapoptotic program that sensitizes activated T cells to Fas (CD95)-mediated apoptosis (Moretti et al. 2008). The NRP-1-plexin-A4 receptor complex transduces Sema3A-mediated immune suppressive signaling. Plexin-A4 is expressed in various cells, including T cells, DCs, and macrophages. Both NRP-1-mutant T cells, in which the Sema3A binding site is specifically disrupted, and plexin-A4−/− T cells exhibit enhanced in vitro proliferation after TCR stimulation. Moreover, plexin-A4−/− mice have enhanced T-cell priming and exacerbated T-cell-mediated immune responses (Yamamoto et al. 2008), suggesting that plexin-A4 negatively regulates T-cell-mediated immune responses. In viral infection, Sema3A is also involved in virus-induced immune suppression. Measles virus-infected DCs, which fail to promote T-cell expansion, have greater early Sema3A secretion, resulting in a loss of actin-based protrusion on T cells (Tran-Van et al. 2011). A very recent study by Cozacov et al. showed that the co-culture of condition medium with 2 mcg/ml of recombinant human sema3A with CD4 + T cells increased the expression of FoxP3 in Tregs, which play a critical role in preventing autoimmunity (Cvetanovich and Hafler 2010), strongly suggesting its potential as an immune regulator of inflammation (Cozacov et al. 2017) (Figure 2).
Immunological function of Sema3A. Sema3A suppressed B-cell proliferations through increasing the expression of CD72 (inhibitory molecule), and the signaling of BCR. Sema3A decreases the production of IFN and TNF by macrophages and reduces the production of pro-inflammatory cytokines by T cell. Due to the blockage of actin cytoskeleton reorganization, immunomodulatory functions of Sema3A pushing TCR polarization and interfering with early TCR signal transduction events. When increasing it to DC cell and T-cell co-cultures, Sema3A significantly inhibited allogeneic T-cell proliferation. Sema3A also guides the entry of DC cell into the afferent lymphatics by promoting actomyosin contraction
In addition, it has been shown that plexin-A1, a critical receptor component for Sema3A, was crucially involved in the entry of DCs into the lymphatics. In addition, Sema3A was essential for DC transmigration and that Sema3A produced by the lymphatics promoted actomyosin contraction at the trailing edge of migrating DCs. These findings indicate that Sema3A plays an essential role in the migration of DCs, and identify a previously unknown mechanism that induces actomyosin contraction as these cells pass through narrow gaps (Takamatsu et al. 2010b).
Becoming a frontier player in the regulation of immune responses and the maintenance of self-tolerance, Sema3A is thus implicated in the pathogenesis of ADs, including SLE (Vadasz et al. 2011, 2012, 2015a; Vadasz and Toubi 2012), RA (Perez et al. 2016; Takagawa et al. 2013; Yoshida et al. 2014), MS (Costa et al. 2015; Kremer et al. 2015; Eixarch et al. 2013a; Gutiérrez-Franco et al. 2016; Okuno et al. 2011; Rezaeepoor et al. 2017; Williams et al. 2007), and SSc (Rimar et al. 2014; Olewicz-Gawlik and Samborski 2016; Vadasz et al. 2015d).
Semaphorin-3A in autoimmune diseases
ADs arise an inappropriate immune response against self-components, including macromolecules, cells, tissues, and organs.(Pan et al. 2013a). These groups of diseases involve a variety of molecules, cells, and tissues, which are targeted by the autoimmune responses (Pan et al. 2013b). Although the exact etiologies of ADs remain to be further elucidated and the clinical characteristics of these diseases are diverse, current findings have revealed that Sema3A is involved in the pathogenesis of ADs, including SLE (Gao 2017; Vadasz 2012), RA (Takagawa et al. 2013; Yoshida et al. 2014), MS (Rezaeepoor et al. 2017), and SSc (Olewicz-Gawlik and Samborski 2016; Rimar et al. 2015a; Vadasz et al. 2015c) (Table 1).
Semaphorin-3A in systemic lupus erythematosus
SLE is a prototypical ADs, characterized by the production of autoantibodies to components of the cell nucleus in association with a variety of clinical manifestations. The exact etiology of SLE is unknown; it has been confirmed that genetic factors play an important role in the predisposition of SLE, but multiple environmental or yet unclear factors may also be responsible (Mok and Lau 2003; Li et al. 2016). In the interaction of various factors such as genetic factors, environmental factors, and estrogen levels, it leads to a decrease in T lymphocytes, a reduction in T-suppression cell function, an excessive hyperplasia of B cells, a large amount of autoantibodies, and a combination with corresponding autoantigen in vivo (Mok and Lau 2003; Yu et al. 2014). The corresponding immune complexes are deposited on the skin, joints, small blood vessels, and glomeruli (Rua-Figueroa Fernandez de Larrinoa 2015). Antimalarials, steroids, and immunosuppressive drugs have been used in the treatment of SLE for many years. Although these drugs are efficient in improving quality of life, survival rate and well being, and maintaining long remissions, they are associated with several severe side effects. Thus, novel and better focused treatments are urgently needed (Lerang et al. 2012; Ruiz-Irastorza et al. 2010; Vadasz and Toubi 2012). For example, the monoclonal antibody against BAFF and the discovery of many regulatory molecules, some of these are protective and of therapeutic potential (Navarra et al. 2011).
Recently, Vadasz et al. reported that Sema3A was expressed strongly in the tubuli in lupus glomerulonephritis (LGN), suggesting that it can serve as a histological marker for tubular damage (Vadasz et al. 2011). Later, they observed significantly lower serum Sema3A levels in SLE patients compared with RA patients and normal controls. In addition, serum Sema3A levels were inversely correlated with SLE disease activity and renal damage. The expression of both Sema3A and NRP-1 was found to be significantly lower on Bregs of SLE patients in comparison with healthy controls. Furthermore, upon the co-culture of Sema3A with cytosine-phosphodiester-guanine oligodeoxynucleotides (CpG-ODN)-stimulated B cells of SLE patients, their TLR-9 expression was markedly decreased (Vadasz et al. 2012). Bregs have an important position in the immunomodulatory, specifically in preventing SLE with a role in the maintenance of self-tolerance (Lund and Randall 2010). Sema3A enhances Breg properties by upregulating CD72 expression on B cells. The immunomodulatory effect of Sema3A was demonstrated (Vadasz et al. 2014). T-cell dysfunction is evident in SLE patients. The total number of T cells in the peripheral blood usually decreases (Bakke et al. 1983). The expression of Sema3A and its receptor NRP-1 as binding receptors and plexin A protein as signal transducers have been found to increase and activate T cells on differentiated macrophages, showing that they can regulate inflammation situation (Lepelletier et al. 2006). Therefore, Sema3A may be a promising therapeutic agent for SLE, such focused therapies will contribute to the maintenance of self-tolerance and attenuate pro-inflammatory status in this disease (Vadasz et al. 2015a).
A very recent study by Gao et al. also demonstrated similar results; Sema3A and NRP-1 were both significantly decreased in serum and PBMC of SLE patients, suggesting that the circulating expression of Sema3A and NRP-1 was seriously defected in SLE. In addition, circulating Sema3A was significantly correlated with disease activity and blood damage in patients with SLE. Furthermore, the result of receiver operating characteristic (ROC) curve showed that Sema3A had the potential to be a new diagnostic biomarker in SLE (Gao et al. 2017). All these findings above strongly indicate the therapeutic and diagnostic potential of Sema3A in SLE.
Semaphorin-3A in rheumatoid arthritis
RA is a chronic inflammatory disease characterized by the destruction of peripheral joint in which the articular cartilage and bones are destroyed by the proliferative synovitis. The synovial lesion is formed by inflammatory cell infiltration, marked proliferation of the synovial lining cells, and enhanced angiogenesis. Angiogenesis occurs in the synovia from the early stage of RA, which is considered to be one of the key processes for progression of the arthritic lesions in this disease (AE 1998; Vadasz et al. 2010). Although both basic scientific researches and clinical researches have attempted to determine the factors involved in the pathogenesis of RA, the exact cause of this disease is still unclear.
In RA patients, serum vascular endothelial growth factor (VEGF) levels are significantly related to joint destruction and disease activity score (Clavel et al. 2007; Kurosaka et al. 2010). Sema3A is also a receptor for VEGF165 (Miao et al. 1999). Studies have shown that the expression levels of Sema3A and VEGF are correlated with disease activity in several tumors (Barresi and Tuccari 2010; Osada et al. 2006). Takagawa et al. showed that Sema3A expression in synovial lining cells was reduced in RA tissues compared with osteoarthritis (OA) samples. There was also a significant reduction of Sema3A mRNA levels in RA synovial tissue samples than in OA and a significantly negative correlation of the ratio of Sema3A/VEGF-A mRNA expression levels with Disease Activity Score 28-joint count C reactive protein (DAS28-CRP), but there was no significant correlation between Sema3A and VEGF165 (Takagawa et al. 2013).
The RA synovium contains activated T and B cells, all of which are recruited through a vigorous neovascularization process with lymphangiogenesis (Brennan and McInnes 2008). The immunogenetics of RA suggests that the abnormal pathway of T-cell activation plays a key role in the occurrence and/or continuation of the disease (André et al. 2009). Catalano et al. reported on the defective expression of Sema3A on CD4 T cells derived from patients with RA. Sema3A increases the CD4+NP-1+T cell ability to suppress alloresponses, that its transient expression is altered in rheumatoid inflammation and that the reintroduction of Sema3A is sufficient to attenuate collagen-induced arthritis (CIA), an experimental model of human RA, supporting its therapeutic potential in the treatment of this disease (Catalano 2010). Sema3A inhibits osteoclast function and increases osteoblast function (Negishi-Koga and Takayanagi 2012). It may be a key mediator of bone immunity in RA and has immune activation-related bone destruction. However, Yoshida et al. showed that serum concentration of Sema3A was elevated in RA patients, but serum Sema3A level did not associate with disease activity and marker of bone metabolism (Yoshida et al. 2014). These findings suggest that Sema3A might play an important role in RA pathogenesis.
Semaphorin-3A in multiple sclerosis
MS, one of the most prevalent autoimmune diseases, is a chronic and disabling neurological disease characterized by a disturbance in myelination, which mainly occurs in people aged 15–40 years (Arizmendi-Vargas et al. 2011). Its clinical features are focal disturbances of the paroxysmal optic nerve, spinal cord, and brain. These neurological disorders can have varying degrees of remission and recurrence. The most common form of this disease is relapsing–remitting MS (Dendrou et al. 2015). MS tissue damage and neurological symptoms are thought to be caused by an immune response directed against myelin antigens (Huseby et al. 2001). Viral infections or other stimulatory factors may cause T cells and antibodies to enter the central nervous system (CNS) by destroying the blood–brain barrier, resulting in increased expression of cell adhesion molecules, matrix metalloproteinases, and pro-inflammatory cytokines that together act to attract other immune cells and breakdown cells (Ransohoff and Brown 2012; Tubridy et al. 1999). In particular, Th1 cytokines such as IL-2 and IFN-γ may be associated with the incidence of MS (Arizmendi-Vargas et al. 2011). Due to immune attack, the myelin sheath can be exfoliated, which slows nerve conduction and leads to neurological symptoms (Chang et al. 2002; Huseby et al. 2001). At present, the etiology of MS remains incompletely elucidated.
Sema3A is a secreted molecule that inhibits regenerating axons sprouting into glial scars of astrocytes and an axon guidance molecule in the CNS (De Winter et al. 2002). Sema3A is involved not only in MS pathogenesis but also in the nervous systems. Sema3A may have a positive effect in the immune system since it aborts the immune response. In the context experimental autoimmune encephalomyelitis (EAE) model of MS, this may be due to the over-activation of MOG-specific T cells, and plexin-A4-deficient mice exhibit a worse clinical course of disease, suggesting that the Sema3A/NRP-1/plexin-A4 pathway is involved in the pathogenesis of EAE (Yamamoto et al. 2008). Rezaeepoor et al. revealed that the serum level of Sema3A and its expression in peripheral blood mononuclear cells (PBMCs) of relapsing–remitting multiple sclerosis (RRMS) patients were significantly lower than in normal subjects. It suggested that Sema3A could be involved in pathogenesis of MS and might be a potential diagnostic biomarker for the disease (Rezaeepoor et al. 2017). Since a higher expression of Sema3A and its receptors, especially in astrocytes but also in microglia/macrophages, was found in MS lesions compared to progressive multifocal leucoencephalopathy and acute cerebral infarct lesions (Costa et al. 2015). In addition, Sema3A has been shown to be aberrantly expressed in brains with MS (Williams et al. 2007), suggesting that Sema3A is involved in the regeneration of oligodendrocytes or axons (Okuno et al. 2011). Inhibition of Sema3A during remyelination showed reversible blockade of oligodendrocyte precursor cell (OPC) differentiation in the presence of Sema3A. Although some remyelination has occurred in MS, this reinforces the deregulation of sema3A expression in the CNS that may be an important factor in impaired OPC recruitment and/or differentiation during MS injury, which ultimately limits myelin repair (Majed et al. 2006; Piaton et al. 2011; Syed et al. 2011). Nevertheless, further studies are still required to precisely evaluate its potential utility as a circulating biomarker and a novel therapeutic target for MS.
Semaphorin-3A in systemic sclerosis
SSc is a multisystem disease with varied course from being a relatively benign condition involving the skin and peripheral vasculature to a rapidly progressive disease affecting internal organs with a consequent high mortality risk, and represents one of the systemic autoimmune diseases with worse prognosis (Bryan et al. 1996; Rubio-Rivas et al. 2014). SSc can be identified as two subsets: a stable, limited cutaneous group (limited systemic sclerosis, ISSc), representing the majority of patients, and a more rapidly advancing smaller group with diffuse cutaneous (diffuse systemic sclerosis, dSSc) involvement (LeRoy et al. 1998). The etiology of SSc remains not fully understood. It may be due to genetic, environmental factors (viral infections, chemical substances such as silicon, etc.), cellular and humoral immune abnormalities, and other factors; fibroblasts synthesis and secretion of collagen increased, leading to skin and visceral fibrosis (LeRoy 1988; Steen and Medsger 2007).
Sema3A as an immunomodulatory molecule mainly affects the activation of Tregs. Most recent studies demonstrated lower serum Sema3A expression levels in SSc patients compared to normal controls (Rimar et al. 2014; Vadasz et al. 2015d). Rimar et al. reported that the expression of Sema3A on Tregs was also lower in SSc patients compared to normal control. In addition, serum Sema3A level was negatively correlated with the disease duration and low C4 level. A lower serum Sema3A level was associated with SCL-70 antibody positivity (Rimar et al. 2015b). The decreased expression of sema3A on Tregs in SSc patients may be associated with impaired regulation. The lower expression level of Sema3A both in serum and Tregs in SSc patients may help explain the attenuated activation of Tregs in SSc. However, there are also studies showing no significant change in serum level of Sema3A (Romano et al. 2016) or even higher level of Sema3A (Olewicz-Gawlik and Samborski 2016) in SSc patients when compared with normal controls. The expression level of VEGF in the dermis of SSc patients is significantly increased (Distler et al. 2004; Mackiewicz et al. 2002). VEGF exerts its biological function by binding to NRP-1 of Sema3A receptor and VEGFR-1, VEGFR-2 of tyrosine kinase receptor, both of which are upregulated in dermal endothelial cells in SSc-infected skin (Distler et al. 2002; Mackiewicz et al. 2002). Therefore, the role of Sema3A in SSc still needs further research.
Semaphorin-3A as a promising therapeutic agent for autoimmune diseases
Because of its immunosuppressive role in systemic autoimmunity, targeting Sema3A or its receptors may have promise as a potential therapeutic strategy for ADs. With known of the specific mechanisms of Sema3A in ADs, together with the knowledge on the capacity of current treatment strategy to target, this process may open a door to novel therapeutic options for ADs. In fact, studies in animal models of several ADs have demonstrated encouraging results. Overexpression of Sema3A partially prevents and attenuates the progression of CIA, the animal model of RA (Catalano 2010). In EAE, the animal model of MS, mice deficient in plexin-A4 demonstrate a worse clinical course Dendrou et al. 2015. Furthermore, NRP-1-deficient CD4 + T cells result in increased EAE severity; the conditional knockout mice exhibit preferential Th17 lineage commitment and decreased Tregs functionality. Conversely, CD4 + T cells expressing NRP-1 inhibit effector T-cell proliferation and cytokine production independent of Tregs (Solomon et al. 2011). Serum Sema3A levels were inversely correlated with SLE disease activity and renal damage (Vadasz et al. 2011), and the expansion of Bregs in SLE patients was enhanced when stimulated with Sema3A, suggesting that this regulatory molecule may be considered as a potential therapy for SLE (Vadasz et al. 2012). Collectively, these evidence illustrated that Sema3A may serve as a promising therapeutic agent for ADs.
The prospective and the limitation of Semaphorin-3A in clinical application
Since its suppressive activity in ADs has been appeared in many reports, both in vitro and in vivo, Sema3A may act as a therapeutic agent for these diseases in clinical application. Recently, there were several studies about Sema3A and ADs. Gao et al. suggested that Sema3A was significantly correlated with disease activity and blood damage in SLE patients; it had the potential to be a new diagnostic biomarker in SLE (Gao et al. 2017). Perez et al. demonstrated that class 3 semaphorins are differentially expressed in the synovium of early patients depending on the severity and the progression of the disease, and that Sema3A plays an important role in the invasive capacity of RA (Perez et al. 2016). Rezaeepoor et al. found that the serum level of Sema3A was significantly lower in MS patients; it could be involved in pathogenesis of MS (Rezaeepoor et al. 2017). Vadasz et al. demonstrated that Sema3A levels were significantly decreased in both serum and Tregs in SSc patients (Rimar et al. 2015b). These results suggest that overexpressing Sema3A provides a useful approach for basic research on treatment of ADs.
Despite emerging evidence that Sema3A has therapeutic potential in several ADs studies, it is still too early to ensure the efficacy of Sema3A in clinical application. Researches into the involvement of Sema3A in pathogenesis and treatment of ADs are relatively limited and with contradictory results. In addition, the clinical features of ADs are diverse, and the causes of these diseases are complicated. Furthermore, the pathogenesis of MS includes both the immune and the nervous systems. Sema3A appears to play a protective role in the immune system, but it also has a damaging effect in the CNS due to its inhibitory effect on OPC migration toward the site of lesion, or the repulsive activity that impedes the outgrowth of injured neurons. Therefore, the effect of manipulating expression of sema3A on MS outcome remains inconclusive.
Conclusions
Although Sema3A has confirmed roles in immune responses or immune cell differentiation. The Sema3A expressed in the vast majority of immune cells remains ill defined. The challenge going forward will be to identify the Sema3A-based mechanisms that regulate immune responses, and to dissect the specific functional sequences and domains that execute the biological functions of Sema3A. New technologies are being developed and applied to study the biochemistry of Sema3A, its structures, and receptor, which will advance research in this field.
The available evidence clearly supports the involvement of Sema3A in the pathogenesis in human diseases; however, Sema3A research in ADs is a nascent field. Future studies will undoubtedly uncover additional and novel insights into the functions of Sema3A in immunomodulatory and ADs. Studying Sema3A that is differentially expressed in immune cell subtypes or damaged tissues between patients and healthy individuals should provide further insights into the pathogenesis of ADs and further elucidate the molecular mechanisms of Sema3A in the regulation of immune responses and inflammation. SNPs within Sema3A-encoding genes may affect the expression or function of Sema3A in ADs.
Further investigation is also needed to unveil how Sema3A-related loci contribute to pathogenesis, and will enhance our understanding of ADs. Advances in genome-editing technologies such as CRISPR/Cas9 should enable researchers to determine the involvement of a given Sema3A-related SNP in pathogenesis. We believe that Sema3A has potential as disease biomarkers and therapeutic targets, and that researches in this area should help the development of novel diagnostic and therapeutic approaches to treat ADs.
Abbreviations
- ADs:
-
Autoimmune diseases
- αB-crystallin:
-
Alpha B-crystallin
- BAFF:
-
B-cell activating factor
- Bregs:
-
B regulatory cells
- CIA:
-
Collagen-induced arthritis
- CNS:
-
Central nervous system
- CpG-ODN:
-
Cytosine-phosphodiester-guanine oligodeoxynucleotides
- DAS28-CRP:
-
Disease Activity Score 28-joint count C reactive protein
- DCs:
-
Dendritic cells
- dSSc:
-
Diffuse systemic sclerosis
- EAE:
-
Experimental autoimmune encephalomyelitis
- FoxP3:
-
Forkhead box P3
- IL-2:
-
Interleukin 2
- IL-10:
-
Interleukin 10
- IL-17:
-
Interleukin 17
- IFNs:
-
Interferons
- ISSc:
-
Limited systemic sclerosis
- LGN:
-
Lupus glomerulonephritis
- MAG:
-
Myelin binding glycoprotein
- MAPK:
-
Ras/mitogen-activated protein kinase
- MOG:
-
Oligodendrocyte glycoprotein
- MS:
-
Multiple sclerosis
- NRP-1:
-
Neuropilin-1
- OA:
-
Osteoarthritis
- OPC:
-
Oligodendrocyte precursor cell
- PBMCs:
-
Peripheral blood mononuclear cells
- PSI:
-
Plexin–semaphorin–integrin
- RA:
-
Rheumatoid arthritis
- ROC:
-
Receiver operating characteristic
- RRMS:
-
Relapsing–remitting multiple sclerosis
- Sema3A:
-
Semaphorin-3A
- SLE:
-
Systemic lupus erythematosus
- SSc:
-
Systemic sclerosis
- TGF-β:
-
Transforming growth factor beta
- Th1:
-
Helper T-cell type 1
- Th17:
-
Helper T-cell type 17
- TNF:
-
Tumor necrosis factor
- Tregs:
-
Regulatory T cells
- VEGF:
-
Vascular endothelial growth factor
- VEGFR:
-
Vascular endothelial growth factor receptor
References
Ae K (1998) Angiogenesis: implication for rheumatoid arthritis. Arthritis Rheum 41:951–962
André S, Tough DF, Lacroix-Desmazes S, Kaveri SV, Bayry J (2009) Surveillance of antigen-presenting cells by CD4 + CD25 + regulatory T cells in autoimmunity. Am J Pathol 174:1575–1587. https://doi.org/10.2353/ajpath.2009.080987
Arizmendi-Vargas J, Carrillo-Ruiz JD, Lopez-Lizarraga ME, Martinez-Menchaca H, Serrato-Ávila JL, Rendón-Molina A, Rivera-Silva G (2011) Multiple sclerosis: an overview of the disease and current concepts of its pathophysiology. J Neurosci Behav Health 3:44–50
Bakke AC, Kirkland PA, Kitridou RC (1983) T lymphocyte subsets in systemic lupus erythematosus. Arthritis Rheum 26:745–750
Barresi V, Tuccari G (2010) Increased ratio of vascular endothelial growth factor to semaphorin3A is a negative prognostic factor in human meningiomas. Neuropathology 30:537–546. https://doi.org/10.1111/j.1440-1789.2010.01105.x
Brennan FM, McInnes IB (2008) Evidence that cytokines play a role in rheumatoid arthritis. J Clin Investig 118:3537–3545. https://doi.org/10.1172/jci36389
Bryan C, Howard Y, Brennan P, Black C, Silman A (1996) Survival following the onset of scleroderma: results from a retrospective inception cohort study of the UK patient population. J Rheumatol 35:1122–1126
Catalano A (2006) Semaphorin-3A is expressed by tumor cells and alters T-cell signal transduction and function. Blood 107:3321–3329. https://doi.org/10.1182/blood-2005-06-2445
Catalano A (2010) The neuroimmune semaphorin-3A reduces inflammation and progression of experimental autoimmune arthritis. J Immunol 185:6373–6383. https://doi.org/10.4049/jimmunol.0903527
Chang A, Tourtellotte WW, Rudick R, Trapp BD (2002) Premyelinating oligodendrocytes in chronic lesions of multiple sclerosis. N Engl J Med 346:165–173. https://doi.org/10.1056/nejmoa010994
Clavel G et al (2007) Angiogenesis markers (VEGF, soluble receptor of VEGF and angiopoietin-1) in very early arthritis and their association with inflammation and joint destruction. Clin Immunol 124:158–164
Committee SN (1999) Unified nomenclature for the semaphorin/collapsins. Cell 97:551–552
Costa C, Martínez-Sáez E, Gutiérrez-Franco A, Eixarch H, Castro Z, Ortega-Aznar A, Ramón y Cajal S, Montalban X, Espejo C (2015) Expression of semaphorin 3A, semaphorin 7A and their receptors in multiple sclerosis lesions. Mult Scler J 21:1632–1643. https://doi.org/10.1177/1352458515599848
Cozacov R, Halasz K, Haj T, Vadasz Z (2017) Semaphorin 3A: is a key player in the pathogenesis of asthma. Clin Immunol 184:70–72. https://doi.org/10.1016/j.clim.2017.05.011
Cvetanovich GL, Hafler DA (2010) Human regulatory T cells in autoimmune diseases. Curr Opin Immunol 22:753–760. https://doi.org/10.1016/j.coi.2010.08.012
de Larrinoa IRFF (2015) What is new in systemic lupus erythematosus. Reumatol Clin 11:27–32
De Winter F et al (2002) Injury-induced class 3 semaphorin expression in the rat spinal cord. Exp Neurol 175:61–75. https://doi.org/10.1006/exnr.2002.7884
Delaire S, Billard C, Tordjman R, Chédotal A, Elhabazi A, Bensussan A, Boumsell L (2001) Biological activity of soluble CD100. II. Soluble CD100, similarly to H-SemaIII, inhibits immune cell migration. J Immunol 166:4348–4354
Dendrou CA, Fugger L, Friese MA (2015) Immunopathology of multiple sclerosis. Nat Rev Immunol 15:545–558. https://doi.org/10.1038/nri3871
Distler O et al (2002) Angiogenic and angiostatic factors in systemic sclerosis: increased levels of vascular endothelial growth factor are a feature of the earliest disease stages and are associated with the absence of fingertip ulcers. Arthritis Res 4:30
Distler O et al (2004) Uncontrolled expression of vascular endothelial growth factor and its receptors leads to insufficient skin angiogenesis in patients with systemic sclerosis. Circ Res 95:109–116
Eixarch H, Gutiérrez-Franco A, Montalban X, Espejo C (2013) Semaphorins 3A and 7A: potential immune and neuroregenerative targets in multiple sclerosis. Trends Mol Med 19:157–164. https://doi.org/10.1016/j.molmed.2013.01.003
Fiore R, Püschel AW (2003) The function of semaphorins during nervous system development. Front Biosci 8:s484–s499
Gagliardini V, Fankhauser C (1999) Semaphorin III can induce death in sensory neurons. Mol Cell Neurosci 14:301–316
Gao H et al (2017a) Expression and clinical significance of semaphorin 3A in serum and mononuclear cells in patients with systemic lupus erythematosus. Zhonghua Yi Xue Za Zhi 97:370–374. https://doi.org/10.3760/cma.j.issn.0376-2491.2017.05.010
Gao H, Ma XX, Guo Q, Zou YD, Zhong YC, Xie LF, Shao M, Zhang XW (2017b) Expression and clinical significance of semaphorin 3A in serum and monouclear cells in patients with systemic lupus erythematous. Natl Med J China 97:370–374
Guan SY, Leng RX, Khan MI, Qureshi H, Li XP, Ye DQ, Pan HF (2017) Interleukin-35: a potential therapeutic agent for autoimmune diseases. Inflammation 40:303–310. https://doi.org/10.1007/s10753-016-0453-9
Gutiérrez-Franco A et al (2016) Differential expression of sema3A and sema7A in a murine model of multiple sclerosis: implications for a therapeutic design. Clin Immunol 163:22–33. https://doi.org/10.1016/j.clim.2015.12.005
Huseby ES, Liggitt D, Brabb T, Schnabel B, Öhlén C, Goverman J (2001) A pathogenic role for myelin-specific Cd8 + T cells in a model for multiple sclerosis. J Exp Med 194:669–676. https://doi.org/10.1084/jem.194.5.669
Ji JD, Park-Min KH, Ivashkiv LB (2009) Expression and function of semaphorin 3A and its receptors in human monocyte-derived macrophages. Hum Immunol 70:211–217. https://doi.org/10.1016/j.humimm.2009.01.026
Kashiwagi H (2005) Negative regulation of platelet function by a secreted cell repulsive protein, semaphorin 3A. Blood 106:913–921. https://doi.org/10.1182/blood-2004-10-4092
Kawasaki T et al (2002) Requirement of neuropilin 1-mediated Sema3A signals in patterning of the sympathetic nervous system. Development 129:671–680
Kim SJ (2015) Immunological function of Blimp-1 in dendritic cells and relevance to autoimmune diseases. Immunol Res 63:113–120. https://doi.org/10.1007/s12026-015-8694-5
Kolodkin AL, Matthes DJ, Goodman CS (1993) The semaphorin genes encode a family of transmembrane and secreted growth cone guidance molecules. Cell 75:1389–1399. https://doi.org/10.1016/0092-8674(93)90625-Z
Kremer D, Hartung HP, Küry P (2015) Targeting semaphorins in MS as a treatment strategy to promote remyelination: a tale of mice, rats and men. Mult Scler J 21:1616–1617. https://doi.org/10.1177/1352458515608693
Kumanogoh A et al (2005) Semaphorins in the immune system. Int Congr Ser 1285:202–206. https://doi.org/10.1016/j.ics.2005.08.008
Kurosaka D et al (2010) Clinical significance of serum levels of vascular endothelial growth factor, angiopoietin-1, and angiopoietin-2 in patients with rheumatoid arthritis. J Rheumatol 37:1121–1128
Lepelletier Y et al (2006) Immunosuppressive role of semaphorin-3A on T cell proliferation is mediated by inhibition of actin cytoskeleton reorganization. Eur J Immunol 36:1782–1793. https://doi.org/10.1002/eji.200535601
Lerang K, Gilboe IM, Gran JT (2012) Differences between rheumatologists and other internists regarding diagnosis and treatment of systemic lupus erythematosus. Rheumatology (Oxford) 51:663–669. https://doi.org/10.1093/rheumatology/ker318
LeRoy E (1988) Scleroderma (systemic sclerosis): classification, subsets and pathogenesis. J Rheumatol 15:202–205
LeRoy EC, Black C, Fleischmajer R, Jablonska S, Krieg T, Medsger TA Jr, Rowell N, Wollheim F (1998) Scleroderma (systemic sclerosis): classification, subsets and pathogenesis. J Rheumatol 15:202–205
Li HM et al (2016) Emerging role of adipokines in systemic lupus erythematosus. Immunol Res 64:820–830. https://doi.org/10.1007/s12026-016-8808-8
Loes S, Kettunen P, Kvinnsland IH, Taniguchi M, Fujisawa H, Luukko K (2001) Expression of class 3 semaphorins and neuropilin receptors in the developing mouse tooth. Mech Dev 101:191–194. https://doi.org/10.1016/S0925-4773(00)00545-1
Lund FE, Randall TD (2010) Effector and regulatory B cells: modulators of CD4 + T cell immunity. Nat Rev Immunol 10:236. https://doi.org/10.1038/nri2729
Luo Y, Raible D, Raper JA (1993) Collapsin: a protein in brain that induces the collapse and paralysis of neuronal growth cones. Cell 75:217–227. https://doi.org/10.1016/0092-8674(93)80064-L
Mackiewicz Z, Sukura A, Povilenaite D, Ceponis A, Virtanen I, Hukkanen M, Konttinen YT (2002) Increased but imbalanced expression of VEGF and its receptors has no positive effect on angiogenesis in systemic sclerosis skin. Clin Exp Rheumatol 20:641–646
Majed HH et al (2006) A novel role for Sema3A in neuroprotection from injury mediated by activated microglia. J Neurosci 26:1730–1738. https://doi.org/10.1523/jneurosci.0702-05.2006
Messersmith EK, Leonardo ED, Shatz CJ, Tessier-Lavigne M, Goodman M, Kolodkin AL (1995) Semaphorin III can function as a selective chemorepellent to pattern sensory projections in the spinal cord. Neuron 14:949–959
Miao HQ, Soker S, Feiner L, Alonso JL, Raper JA, Klagsbrun M (1999) Neuropilin-1 mediates collapsin-1/semaphorin III inhibition of endothelial cell motility: functional competition of collapsin-1 and vascular endothelial growth factor-165. J Cell Biol 146:233–242
Mizui M, Kumanogoh A, Kikutani H (2009) Immune semaphorins: novel features of neural guidance molecules. J Clin Immunol 29:1–11. https://doi.org/10.1007/s10875-008-9263-7
Mok CC, Lau CS (2003) Pathogenesis of systemic lupus erythematosus. J Clin Pathol 56:481–490
Moretti S, Procopio A, Lazzarini R, Rippo MR, Testa R, Marra M, Tamagnone L, Catalano A (2008) Semaphorin3A signaling controls Fas (CD95)-mediated apoptosis by promoting Fas translocation into lipid rafts. Blood 111:2290–2299. https://doi.org/10.1182/blood-2007-06096529
Navarra SV et al (2011) Efficacy and safety of belimumab in patients with active systemic lupus erythematosus: a randomised, placebo-controlled, phase 3 trial. Lancet 377:721–731. https://doi.org/10.1016/s0140-6736(10)61354-2
Negishi-Koga T, Takayanagi H (2012) Bone cell communication factors and semaphorins. Bonekey Rep 1:183
Okuno T, Nakatsuji Y, Kumanogoh A (2011) The role of immune semaphorins in multiple sclerosis. FEBS Lett 585:3829–3835. https://doi.org/10.1016/j.febslet.2011.03.033
Olewicz-Gawlik ASD, Samborski W (2016) Soluble semaphorin 3a and neuropilin-1: new markers for dysregulation of angiogenesis in systemic sclerosis. Ann Rheum Dis 75:749. https://doi.org/10.1136/annrheumdis-2016-eular.5233
Osada R, Horiuchi A, Kikuchi N, Ohira S, Ota M, Katsuyama Y, Konishi I (2006) Expression of semaphorins, vascular endothelial growth factor, and their common receptor neuropilins and alleic loss of semaphorin locus in epithelial ovarian neoplasms: increased ratio of vascular endothelial growth factor to semaphorin is a poor prognostic factor in ovarian carcinomas. Hum Pathol 37:1414–1425
Pan HF, Leng RX, Li XP, Zheng SG, Ye DQ (2013a) Targeting T-helper 9 cells and interleukin-9 in autoimmune diseases. Cytokine Growth Factor Rev 24:515–522
Pan HF, Li XP, Zheng SG, Ye DQ (2013b) Emerging role of interleukin-22 in autoimmune diseases. Cytokine Growth Factor Rev 24:51–57. https://doi.org/10.1016/j.cytogfr.2012.07.002
Perez SG et al (2016) Class 3 semaphorins modulate the invasive capacity of rheumatoid arthritis fibroblast-like synoviocytes. Anna Rheum Dis. https://doi.org/10.1136/annrheumdis-2016-209124.140
Piaton G et al (2011) Class 3 semaphorins influence oligodendrocyte precursor recruitment and remyelination in adult central nervous system. Brain 134:1156–1167. https://doi.org/10.1093/brain/awr022
Püschel AW, Adams RH, Betz H (1995) Murine semaphorin D/collapsin is a member of a diverse gene family and creates domains inhibitory for axonal extension. Neuron 14:941–948
Ransohoff RM, Brown MA (2012) Innate immunity in the central nervous system. J Clin Investig 122:1164–1171. https://doi.org/10.1172/jci58644
Rezaeepoor M, Shapoori S, Ganjalikhani-hakemi M, Etemadifar M, Alsahebfosoul F, Eskandari N, Mansourian M (2017) Decreased expression of Sema3A, an immune modulator, in blood sample of multiple sclerosis patients. Gene 610:59–63. https://doi.org/10.1016/j.gene.2017.02.013
Rieger J, Wick W, Weller M (2003) Human malignant glioma cells express semaphorins and their receptors, neuropilins and plexins. Glia 42:379–389. https://doi.org/10.1002/glia.10210
Rimar D, Rosner I, Slobodin G, Boulman N, Rozenbaum M, Halasz K, Haj T, Jiries N, Kaly L, Vadasz Z (2014) Semaphorin 3a as a possible immunoregulator in systemic sclerosis. Ann Rheum Dis 73:868. https://doi.org/10.1136/annrheumdis-2014-eular.2948
Rimar D, Rosner I, Slobodin G, Rozenbaum M, Halasz K, Jiries N, Kaly L, Boulman N, Vadasz Z (2015a) Semaphorin 3A, a potential immune regulator in Familial Mediterranean fever. Pediatr Rheumatol 13:O46. https://doi.org/10.1186/1546-0096-13-s1-o46
Rimar D et al (2015b) Semaphorin 3A: an immunoregulator in systemic sclerosis. Rheumatol Int 35:1625–1630. https://doi.org/10.1007/s00296-015-3269-2
Romano E et al (2016) Decreased expression of neuropilin-1 as a novel key factor contributing to peripheral microvasculopathy and defective angiogenesis in systemic sclerosis. Ann Rheum Dis 75:1541–1549. https://doi.org/10.1136/annrheumdis-2015-207483
Roth L, Koncina E, Satkauskas S, Crémel G, Aunis D, Bagnard D (2008) The many faces of semaphorins: from development to pathology. Cell Mol Life Sci 66:649–666. https://doi.org/10.1007/s00018-008-8518-z
Rubio-Rivas M, Royo C, Simeón CP, Corbella X, Fonollosa V (2014) Mortality and survival in systemic sclerosis: systematic review and meta-analysis. Semin Arthritis Rheum 44:208–219. https://doi.org/10.1016/j.semarthrit.2014.05.010
Ruiz-Irastorza G, Ramos-Casals M, Brito-Zeron P, Khamashta MA (2010) Clinical efficacy and side effects of antimalarials in systemic lupus erythematosus: a systematic review. Ann Rheum Dis 69:20–28. https://doi.org/10.1136/ard.2008.101766
Scarlato M (2003) Induction of neuropilins-1 and -2 and their ligands, Sema3A, Sema3F, and VEGF, during Wallerian degeneration in the peripheral nervous system. Exp Neurol 183:489–498. https://doi.org/10.1016/s0014-4886(03)00046-3
Schlahsa L, Zenk J, Aufderbeck S, Figueiredo C, Blasczyk R (2009) Soluble semaphorin 3A regulates the immune response. Hum Immunol 70:s157
Solomon BD, Mueller C, Chae WJ, Alabanza LM, Bynoe MS (2011) Neuropilin-1 attenuates autoreactivity in experimental autoimmune encephalomyelitis. Proc Natl Acad Sci USA 108:2040–2045. https://doi.org/10.1073/pnas.1008721108
Steen VD, Medsger TA (2007) Changes in causes of death in systemic sclerosis, 1972–2002. Ann Rheum Dis 66:940–944. https://doi.org/10.1136/ard.2006.066068
Suzuki K, Kumanogoh A, Kikutani H (2008) Semaphorins and their receptors in immune cell interactions. Nat Immunol 9:17–23. https://doi.org/10.1038/ni1553
Syed YA, Hand E, Mobius W, Zhao C, Hofer M, Nave KA, Kotter MR (2011) Inhibition of CNS remyelination by the presence of semaphorin 3A. J Neurosci 31:3719–3728. https://doi.org/10.1523/jneurosci.4930-10.2011
Takagawa S, Nakamura F, Kumagai K, Nagashima Y, Goshima Y, Saito T (2013a) Decreased Semaphorin3A expression correlates with disease activity and histological features of rheumatoid arthritis. BMC Musculoskelet Disord 14:1–11
Takagawa S, Nakamura F, Kumagai K, Nagashima Y, Goshima Y, Saito T (2013b) Decreased semaphorin3A expression correlates with disease activity and histological features of rheumatoid arthritis. BMC Musculoskelet Disord 14:40. https://doi.org/10.1186/1471-2474-14-40
Takahashi T, Fournier A, Nakamura F, Wang LH, Murakami Y, Kalb RG, Fujisawa H, Strittmatter SM (1999) Plexin-neuropilin-1 complexes form functional semaphorin-3a receptors. Cell 99:59–69
Takamatsu H, Okuno T, Kumanogoh A (2010a) Regulation of immune cell responses by semaphorins and their receptors. Cell Mol Immunol 7:83–88. https://doi.org/10.1038/cmi.2009.111
Takamatsu H et al (2010b) Semaphorins guide the entry of dendritic cells into the lymphatics by activating myosin II. Nat Immunol 11:594–600. https://doi.org/10.1038/ni.1885
Taniguchi M, Yuasa S, Fujisawa H, Naruse I, Saga S, Mishina M, Yagi T (1997) Disruption of semaphorin III/D gene causes severe abnormality in peripheral nerve projection. Neuron 19:519–530. https://doi.org/10.1016/S0896-6273(00)80368-2
Tran-Van H, Avota E, Bortlein C, Mueller N, Schneider-Schaulies S (2011) Measles virus modulates dendritic cell/T-cell communication at the level of plexinA1/neuropilin-1 recruitment and activity. Eur J Immunol 41:151–163. https://doi.org/10.1002/eji.201040847
Tubridy N et al (1999) The effect of anti-α4 integrin antibody on brain lesion activity in MS. Neurology 53:466. https://doi.org/10.1212/wnl.53.3.466
Vadasz Z, Toubi E (2012) Semaphorin 3A: a marker for disease activity and a potential putative disease-modifying treatment in systemic lupus erythematosus. Lupus 21:1266–1270. https://doi.org/10.1177/0961203312456753
Vadasz Z, Toubi E (2013) Semaphorins: their dual role in regulating immune-mediated diseases. Clinic Rev Allerg Immunol 47:17–25. https://doi.org/10.1007/s12016-013-8360-4
Vadasz Z, Attias D, Kessel A, Toubi E (2010) Neuropilins and semaphorins: from angiogenesis to autoimmunity. Autoimmun Rev 9:825–829. https://doi.org/10.1016/j.autrev.2010.07.014
Vadasz Z, Ben-Izhak O, Bejar J, Sabo E, Kessel A, Storch S, Toubi E (2011) The involvement of immune semaphorins and neuropilin-1 in lupus nephritis. Lupus 20:1466–1473. https://doi.org/10.1177/0961203311417034
Vadasz Z et al (2012a) Semaphorin 3A is a marker for disease activity and a potential immunoregulator in systemic lupus erythematosus. Arthritis Res Ther 14:R146. https://doi.org/10.1186/ar3881
Vadasz Z, Haj T, Halasz K, Rosner I, Slobodin G, Attias D, Kessel A, Kessler O, Neufeld G, Toubi E (2012b) Semaphorin 3A is a marker for disease activity and a potential immunoregulator in systemic lupus erythematosus. Arthritis Res Ther 14:1–8
Vadasz Z et al (2014) A regulatory role for CD72 expression on B cells in systemic lupus erythematosus. Semin Arthritis Rheum 43:767–771. https://doi.org/10.1016/j.semarthrit.2013.11.010
Vadasz Z, Peri R, Eiza N, Slobodin G, Balbir-Gurman A, Toubi E (2015a) The expansion of CD25 high IL-10 high FoxP3 high B regulatory cells is in association with SLE disease activity. J Immunol Res 2015:254245. https://doi.org/10.1155/2015/254245
Vadasz Z, Rainis T, Nakhleh A, Haj T, Bejar J, Halasz K, Toubi E (2015b) The involvement of immune semaphorins in the pathogenesis of inflammatory bowel diseases (IBDs). PLoS ONE 10:e0125860. https://doi.org/10.1371/journal.pone.0125860
Vadasz Z, Rimar D, Toubi E (2015c) A6.14 Semaphorin 3A, an immunoregulator and potential biomarker for disease severity in systemic sclerosis. Ann Rheum Dis 74:A61. https://doi.org/10.1136/annrheumdis-2015-207259.140
Villegas G, Tufro A (2002) Ontogeny of semaphorins 3A and 3F and their receptors neuropilins 1 and 2 in the kidney. Mech Dev 199:S149–S153
Williams A et al (2007) Semaphorin 3A and 3F: key players in myelin repair in multiple sclerosis? Brain 130:2554–2565. https://doi.org/10.1093/brain/awm202
Wright DE, White FR, Gerfen RW, Silos-Santiago I, Snider W (1995) The guidance molecule semaphorin III is expressed in regions of spinal cord and periphery avoided by growing sensory axons. J Comp Neurol 361:321–333
Yamamoto M et al (2008) Plexin-A4 negatively regulates T lymphocyte responses. Int Immunol 20:413–420. https://doi.org/10.1093/intimm/dxn006
Yazdani U, Terman JR (2006) The semaphorins. Genome Biol 7:211. https://doi.org/10.1186/gb-2006-7-3-211
Yoshida Y et al (2014) AB0114 Semaphorin3A and semaphorin4d in rheumatoid arthritis. Ann Rheum Dis 72:A820. https://doi.org/10.1136/annrheumdis-2013-eular.2437
Yu C, Gershwin ME, Chang C (2014) Diagnostic criteria for systemic lupus erythematosus: a critical review. J Autoimmun 49:10–13
Yuan X, Cheng G, Malek TR (2014) The importance of regulatory T-cell heterogeneity in maintaining self-tolerance. Immunol Rev 259:103–114. https://doi.org/10.1111/imr.12163
Acknowledgements
This work was supported by grants from the National Natural Science Foundation of China (81573222). The authors have no relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending, or royalties.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Liu, LN., Li, XM., Ye, DQ. et al. Emerging role of semaphorin-3A in autoimmune diseases. Inflammopharmacol 26, 655–665 (2018). https://doi.org/10.1007/s10787-018-0484-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10787-018-0484-y