Abstract
Accumulating evidence supports that Sirtuin 6 (SIRT6) may play a vital role in the pathogenesis of spinal cord injury. The current study was designed to investigate the specific effects of SIRT6 on spinal cord injury (SCI). HE and Nissl staining were performed for pathological analysis in SCI rats. SIRT6 expression was detected by RT-qPCR. CCK8 assay was applied for the detection of cell viability of LPS-injured PC12 cells. TNF-a, IL-1β, IL-6, MCP-1 levels and ROS, MPO, SOD levels were assessed to evaluate inflammation and oxidative stress in spinal cord injury. Cell apoptosis were evaluated by morphological examination using AO/EB fluorescent staining methods and key proteins related to apoptosis were explored via western blot. HE staining revealed increased cavity involving the dorsal white matter and central gray matter, and Nissl staining discovered the loss of motor neurons in the ventral horn in SCI rats. SIRT6 had lower expression in SCI rats. Lipopolysaccharide (LPS) exposure induced cell apoptosis and reduced the expression of SIRT6. Mechanistically, we revealed that up-regulation of SIRT6 alleviated inflammation and oxidative stress and inhibited cell apoptosis in spinal cord injury. Together, our findings indicated that SIRT6 attenuated spinal cord injury by suppressing inflammation, oxidative stress, and cell apoptosis. This study demonstrates that SIRT6 may represent a protective effect against spinal cord injury.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
INTRODUCTION
Spinal cord injury (SCI) is a serious disabling disease caused by a series of internal and external factors in orthopedics and neuroscience [1]. It usually results in loss of movement, sensation, and excretory function below the injured segment of patients [2]. SCI not only brings unimaginable psychological distress and life obstacles to the patients but also causes high burden to the families and the community [3]. Thus, the treatment of SCI has been the research priorities in basic medicine and clinical medicine in recent years.
The pathological process of SCI starts with the primary injury process directly caused by a mechanical force and follows the secondary injury process caused by microcirculation disorders and inflammatory responses [4, 5]. Primary injuries of the spinal cord are irreversible in the pathophysiological process [6]. Hence, most studies focus on prevention against secondary injuries, by reducing neuronal apoptosis and necrosis as well as decreasing inflammation reaction [7, 8].
Sirtuins are a class of proteins that possess either mono-ADP-ribosyltransferase, or deacylase activity [9]. They have been implicated in influencing a wide range of cellular processes, such as apoptosis, inflammation, and stress resistance [10]. SIRT6, a member of the sirtuins family, is known to have NAD+-dependent histone deacetylase, mono-ADP-ribotransferase, and deadipoacylase catalysis [11]. Additionally, SIRT6 plays an important role in regulating DNA repair, telomere maintenance, and inflammatory response [12]. Previously, He et al. indicated that overexpression of SIRT6 could inhibit TNF-α-induced expression of MCP-1 and IL-6 in vascular adventitial fibroblasts and knockdown of SIRT6 could augment generation of reactive oxygen species (ROS) [13]. Besides, a research revealed that SIRT6 was able to protect human dental pulp cells from LPS-induced apoptosis [14]. The above researches suggest that SIRT6 reduces inflammatory response and apoptosis. However, the expression and significance of SIRT6 in SCI and LPS-induced nerve cells are still unknown.
In this paper, SCI model in vivo and vitro were established to study the regulatory role of SIRT6 in SCI so as to provide experimental evidence for the possibility of SIRT6 as a target for SCI therapy.
MATERIALS AND METHODS
Animals and Ethics Statement
A total of 20 adult Sprague–Dawley rats (220–250 g) were obtained from the Animal Center of the Chinese Academy of Sciences in Shanghai, China. All animals were raised in a temperature-controlled environment (23–25 °C) with 12-h light/dark cycles and free access to food and water. Experimental protocols were approved by the Medical Ethics Committee of Hunan Children’s Hospital. The experimental procedure followed the Guidelines for the Care and Use of Laboratory Animals.
Spinal Cord Injury
An animal model of SCI was established as described. After being anesthetized by intraperitoneal injection of chloral hydrate (0.3 mg/g body weight), the experimental rats were shaved, with backs cleaned, and then fixed in the prone position. The laminectomy was carried out at the T9 vertebrae, and the vertebrae were then subjected to a crushing injury via compression with a vascular clip to expose the spinal cord. Rats in the sham control group were subjected to the same surgical without compression injury.
Hematoxylin and Eosin (H&E) Staining
For histopathological examination, T7–T9 spinal cord segments near the lesion epicenter were collected and fixed in 4% formalin for 24 h. Following that, the tissues were embedded in paraffin and sliced into 5 μm-thick slices using a microtome. The sections were deparaffinized with xylene and hydrated gradually with ethanol. Subsequently, the sections were stained with hematoxylin and eosin for H&E staining in accordance with the manufacturer’s protocol. The histopathological changes in the placenta were observed under an optical microscope (200×).
Nissl Staining
For histopathological examination, T7–T9spinal cord segments near the lesion epicenter were collected and fixed in 4% formalin for 24 h. Following that, the tissues were embedded in paraffin and sliced into 5 μm-thick slices using a microtome. The sections were deparaffinized with xylene and hydrated gradually with ethanol. Subsequently, the sections were stained with crystal violet for Nissl staining in accordance with the manufacturer’s protocol. Nissl-positive cells were automatically counted in five randomly selected fields and quantified using IPP software (400×).
Cell Culture and LPS Treatment
Rat pheochromocytoma PC12 cells were obtained from the American Type Culture Collection (ATCC, USA) and maintained in Dulbecco’s Modified Eagle Medium (Sigma-Aldrich, USA) supplemented with 10% fetal bovine serum (FBS; Gibco, USA), 100 U/ml penicillin and 100 μg/ml streptomycin (Gibco, USA) in the incubator under suitable humidity condition at 37 °C.
LPS was obtained from Sigma-Aldrich (St Louis, MO, USA). PC-12 cells were exposure to LPS (1, 2.5, 5, and 10 μg/mL) for 24 h in this research.
Cell Transfection
For transfection, pcDNA-SIRT6 and its negative control (NC) were purchased from GenePharma Corporation (Shanghai, China). Cells reaching 80–85% confluence were then transfected with plasmids using Lipofectamine 2000 (Invitrogen, USA) according to the manufacturer’s instructions. Transfection efficiency was evaluated using qRT-PCR.
Cell Counting Kit-8 (CCK-8) Assay
Cell viability was detected using CCK-8 assay (Beyotime, China) according to the protocol in this research. After relevant treatment, cells with a concentration of 1 × 104 cells/well were seeded into the 96-well plate and incubated for 24 h. After treatments, 10 μL of CCK-8 solution was added to the culture medium and received further incubation for 2 h in the dark at 37 °C. Following that, the absorbance of each well was measured and recorded at 450 nm on Micro-plate reader (Bio-Rad, USA). Each experiments were performed in triplicate.
Quantitative Reverse Transcription PCR (qRT-PCR)
qRT-PCR was conducted to measure the expression level of SIRT6. Total RNA in treated or transfected cells was extracted using TRIzol TM Plus RNA Purification kit (Invitrogen, USA). RNAs were reverse transcribed to complementary DNA (cDNA) with PrimeScript Reverse Transcriptase (Takara, Dalian, China). The expression level of SIRT6was measured using mirVana TM qRT-PCR miRNA Detection Kit (Invitrogen, USA). The thermocycling conditions were as follows: pre-denaturation at 95 °C for 10 min, 35 cycles of denaturation at 95 °C for 10 s, annealing at 60 °C for 30 s, and final extension at 72 °C for 30 s. GAPDH acted as the endogenous control and classic 2-ΔΔCt method was adopted to analyze the relative quantities. Each experiment was performed in triplicate. The primer sequences were as follows: SIRT6, forward 5’-CCAAGTTCGACACCACCTTT-3′, reverse 5’-CGGACGTACTGCGTCTTACA-3′; GAPDH, forward 5’-AATGGGCAGCCGTTAGGAAA-3′, reverse 5’-GCGCCCAATACGACCAAATC-3′.
Enzyme-Linked Immunosorbent Assay (ELISA)
The concentrations of tumor necrosis factor-α (TNF-α), interleukin-1 beta (IL-1β), interleukin-6 (IL-6), and monocyte chemotactic protein 1 (MCP-1) were examined by the corresponding enzyme-linked immunosorbent assay (ELISA) kits (R&D Systems, Abingdon, UK) based on the manufacturer’s instruction. Each experiment was performed in triplicate.
Measurement of Intracellular ROS, SOD, and MPO
In brief, the reactive oxygen species (ROS) levels in PC12 cells were detected by fluorescent probe dichloro-dihydro-fluorescein diacetate (DCFH-DA) assay using a ROS reagent kit (Beyotime, Shanghai, China). Besides, the superoxide dismutase (SOD) and myeloperoxidase (MPO) activities in PC12 cells were respectively evaluated by a SOD detection kit and a MPO detection kit (Nanjing Jiancheng Bioengineering Institute, Nanjing, China), following manufacturer’s protocol. Each experiment was performed in triplicate.
Acridine Orange and Ethidium Bromide (AO/EB) Staining
After relevant treatment or transfection, PC12 cells were fixed with 4% paraformaldehyde for 20 min in room temperature and 1 × PBS was used to wash cells for three times. Then, PC12 cells were incubated with 10 μg/ml AO/EB (Aladdin, China) for 15 min at 37 °C. The fluorescence of PC12 cells was observed under a fluorescent microscope (Leica, Germany). Each experiment was performed in triplicate.
Western Blot Analysis
After relevant treatment or transfection, protein from PC12 cells was extracted using RIPA lysis buffer (Beyotime, China) containing protease inhibitors. BCA Protein Assay Kit (Pierce, USA) was used to quantify the concentration of proteins according to the manufacturer’s instructions. Equal amount of protein was fractionated by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) and transferred onto polyvinylidene difluoride (PVDF) membranes. Subsequently, membranes were blocked in 5% freshly prepared milk-TBST for 1 h at room temperature. Membranes were incubated with appropriate primary antibodies displayed as follows: SIRT6 (Abcam, ab88494, 1:5000), Bcl2 (Abcam, ab196495, 1:2000), Bax (Abcam, ab32503, 1:5000), cleaved caspase3 (Abcam, ab2302, 1:500), and procaspase3 (Abcam, ab13847, 1:500). Thereafter, membranes were washed with TBST and further incubated with secondary antibody marked by horseradish peroxidase (Abcam, ab205718, 1:20000) for 2 h at room temperature. GAPDH served as the internal control. Proteins were developed using an enhanced chemiluminescence (ECL; Beyotime, China) kit and visualized by densitometry using Image Lab 3.0 software (Bio-Rad, USA). Each experiment was performed in triplicate.
Statistical Analysis
All experiments in this research were performed in triplicate. Data are presented as the mean ± standard deviation (SD) from three independent experiments. Statistical evaluation of the data was analyzed by one-way analysis of variance followed by the Scheffe post-hoc test. Analyses were conducted using SPSS 12.0 software (SPSS, Inc., Chicago, IL, USA). Differences were considered to be statistically significant at p < 0.05.
RESULTS
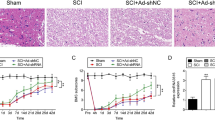
SIRT6 Had Lower Expression in SCI Rats
To confirm the vital role of SIRT6 in spinal cord injury, a well-accepted rat model of SCI was introduced for investigating the specific effects of SIRT6. HE staining and Nissl staining were conducted to analyze the histological level at the seventh day postsurgery. As shown in Fig. 1a, HE staining revealed that cavity involving the dorsal white matter and central gray matter increased in SCI rats compared with that in the sham control group. Nissl staining was utilized to examine the loss of motor neurons in the ventral horn in SCI rats. As exhibited in Fig. 1b, Nissl staining presented a massive loss of ventral motor neurons at and around the epicenter of lesion area. To explore whether SIRT6 was associated with spinal cord injury, western blot assay was applied to determine SIRT6 levels in SCI rats and the sham control rats. As demonstrated in Fig. 1c, SIRT6 was lowly expressed in the lesion epicenter of SCI rats. All these results suggested that SIRT6 participated in the pathological process of spinal cord injury.
LPS Exposure Induced Cell Apoptosis and Reduced the Expression of SIRT6 in PC12 Cells
As shown in Fig. 2a, LPS treatment (1, 2.5, 5, and 10 μg/mL) significantly decreased cell viability in PC12 cells in a dose-dependent manner. In addition, the expression of SIRT6 in PC12 cells exposed to LPS was evaluated using western blot and RT-qPCR analysis. As displayed in Fig. 2b and Fig. 2c, LPS treatment (1, 2.5, 5, and 10 μg/mL) down-regulated SIRT6 level in PC12 cells with increasing dosage. Considering that 5 μg/ml LPS produced significant decreases of both cell viability and SIRT6 expression, 5 μg/ml was selected as a LPS stimulating condition for use in the following experiments.
Up-regulation of SIRT6 Alleviated Inflammation and Oxidative Stress in Spinal Cord Injury
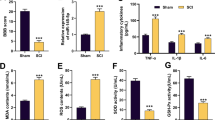
To further analyze the specific effects of SIRT6 in spinal cord injury, SIRT6 mimic and its negative control were transfected into PC12 cells exposed to LPS for the following experiments. As shown in Fig. 3a and Fig. 3b, the protein and mRNA levels of SIRT6 were determined by western blot and RT-qPCR analysis for transfection efficiency. Considering the important role of inflammation and oxidative stress in SCI, we analyzed the levels of pro-inflammatory and oxidative stress markers in PC12 cells under exposure to LPS. Results in Fig. 3c and d unveiled higher levels of inflammatory and oxidative stress in PC12 cells induced by LPS. As expected, up-regulation of SIRT6 caused a significant decrease in the production of TNF-a, IL-1β, IL-6, and MCP-1, with concomitant decreases in ROS and MPO levels and increase in SOD. Overall, up-regulation of SIRT6 alleviated inflammation and oxidative stress in spinal cord injury.
Up-regulation of SIRT6 alleviated inflammation and oxidative stress in spinal cord injury. a Western blot analysis of SIRT6 level. b RT-qPCR analysis of SIRT6 level. c TNF-a, IL-1β, IL-6, and MCP-1 levels assessed by ELISA assay. d Measurement of intracellular ROS, MPO, and SOD. **p < 0.01, ***p < 0.001 vs. control group; # p < 0.05, ## p < 0.01, ### p < 0.001 vs. LPS 5 μg/ml group.
Up-regulation of SIRT6 Inhibited Cell Apoptosis in Spinal Cord Injury
In this study, we evaluated cell apoptosis in two different ways: morphological examination using AO/EB fluorescent staining via the fluorescent microscope and detection of key proteins related to apoptosis via western blot. As shown in Fig. 4a, LPS-treated PC12 cells showed a significant reduction in cell viability and more apoptotic views (chromatin condensation and nuclear fragmentation), while up-regulation of SIRT6 exerted the opposite effects. In addition, it was found in Fig. 4b that Bcl-2 protein expression decreased, and Bax and cleaved caspase-3 protein expression increased in PC12 cells under exposure to LPS. Furthermore, up-regulation of SIRT6 enhanced the expression of anti-apoptotic protein and reduced the expression of pro-apoptotic protein. These results above indicated that up-regulation of SIRT6 could hinder cell apoptosis in spinal cord injury.
Up-regulation of SIRT6 inhibited cell apoptosis in spinal cord injury. a The morphology of pyroptotic cells visualized by AO/EB staining. b Western blot analysis of anti-apoptotic protein and pro-apoptotic protein. **p < 0.01, ***p < 0.001 vs. control group; # p < 0.05, ## p < 0.01 vs. LPS 5 μg/ml group.
DISSCUSSION
SCI is a fearful injury to the central nervous system, leading to serious dysfunction of the lower limbs, cumulatively affecting the respiratory and urinary system, and even endangering life [15]. However, its repair remains challenging owing to the inhibitory environment of the injury site and the irreversibility of primary injuries [16]. At present, major therapies for SCI are secondary injury, including inhibiting the release of inflammatory factors, oxidative stress, promoting neuronal regeneration and repair, and alleviating neuropathic pain [17]. In this study, we aimed to search for early, effective treatment targets to exert a significant impact on reducing the extent of injury and improving the quality of life for SCI patients.
SIRT6 is a NAD+-dependent histone deacetylase which participates in numerous biological activities such as genomic stability, inflammation, and so on. In vitro studies have demonstrated that SIRT6 exerts protective effects against LPS-induced cell damage [13, 14]. Besides, SIRT6 protects mice from LPS-induced acute kidney injury via inhibiting inflammation and apoptosis and inducing autophagy [18]. However, as a key regulatory protein of inflammatory response, the specific mechanism of SIRT6 on regulating the pathologic progress of SCI remains unclear. Data in the present study demonstrated that SIRT6 was down-regulated in SCI rats and LPS-induced PC12 cells, which indicated a potential role of SIRT6 in spinal cord injury. Therefore, modulating the expression of SIRT6 may shed light on SCI therapy.
Recent research reported that improving antioxidative capacity and inhibiting inflammation could ameliorate neurological impairment in SCI [19, 20]. The moderating effect of SIRT6 on inflammation and oxidative stress in LPS-injured neural cells were evaluated in this study. Significant increases in the inflammatory factors including TNF-a, IL-1β, IL-6, and MCP-1 and oxidative stress accompanied with decreased antioxidant capacity were observed in LPS-induced PC12 cells. By contrast, overexpression of SIRT6 could markedly inhibit inflammation and oxidative stress in PC12 cell induced by LPS.
Bcl-2 family is one of the regulatory factors for cell apoptosis involved in the process. The proportion of Bcl-2/Bax determines whether cells are dying [21]. Besides, caspase associated cascade reaction is the important pathway for apoptosis, and caspase3 is the key protease in the process of apoptosis [22]. Here, overexpression of SIRT6 significantly increased the expression of Bcl-2 and inhibited the expression of Bax and cleaved caspase3. As expected, SIRT6 could inhibit apoptosis of PC12 induced by LPS.
In summary, our study demonstrated the neuroprotective effect of SIRT6 against spinal cord injury. This study provides evidence for further understanding the anti-inflammatory, anti-oxidative stress, and anti-apoptosis effects of SIRT6 in SCI. Our findings may provide a new insight into the possibility of SIRT6 being a therapeutic target in SCI and offer theoretical basis for deeply exploring the prevention and treatment of SCI.
References
Duo, Z., and X.J. He. 2015. Advances in mechanisms of treatment for spinal cord injury with lithium. Zhongguo Gu Shang 28: 679–682.
White, A.R., and G.M. Holmes. 2019. Investigating neurogenic bowel in experimental spinal cord injury: Where to begin. Neural Regeneration Research 14: 222–226.
LaVela, S.L., K. Landers, B. Etingen, V.P. Karalius, and S. Miskevics. 2015. Factors related to caregiving for individuals with spinal cord injury compared to caregiving for individuals with other neurologic conditions. The Journal of Spinal Cord Medicine 38: 505–514.
Sobrido-Cameán, D., and A. Barreiro-Iglesias. 2018. Role of Caspase-8 and Fas in cell death after spinal cord injury. Frontiers in Molecular Neuroscience 11: 101.
Fournely, M., Y. Petit, É. Wagnac, J. Laurin, V. Callot, and P.J. Arnoux. 2018. High-speed video analysis improves the accuracy of spinal cord compression measurement in a mouse contusion model. Journal of Neuroscience Methods 293: 1–5.
Pinchi, E., A. Frati, S. Cantatore, S. D'Errico, R. Russa, A. Maiese, M. Palmieri, A. Pesce, R.V. Viola, P. Frati, and V. Fineschi. 2019. Acute spinal cord injury: A systematic review investigating miRNA families involved. International Journal of Molecular Sciences 20.
Zhong, Z.X., S.S. Feng, S.Z. Chen, Z.M. Chen, and X.W. Chen. 2019. Inhibition of MSK1 promotes inflammation and apoptosis and inhibits functional recovery after spinal cord injury. Journal of Molecular Neuroscience 68: 191–203.
Park, C.S., J.Y. Lee, H.Y. Choi, B.G. Ju, I. Youn, and T.Y. Yune. 2019. Protocatechuic acid improves functional recovery after spinal cord injury by attenuating blood-spinal cord barrier disruption and hemorrhage in rats. Neurochemistry International 124: 181–192.
Dai, H., D.A. Sinclair, J.L. Ellis, and C. Steegborn. 2018. Sirtuin activators and inhibitors: Promises, achievements, and challenges. Pharmacology & Therapeutics 188: 140–154.
Strycharz, J., Z. Rygielska, E. Swiderska, J. Drzewoski, J. Szemraj, L. Szmigiero, and A. Sliwinska. 2018. SIRT1 as a therapeutic target in diabetic complications. Current Medicinal Chemistry 25: 1002–1035.
Gertler, A.A., and H.Y. Cohen. 2013. SIRT6, a protein with many faces. Biogerontology 14: 629–639.
Kugel, S., and R. Mostoslavsky. 2014. Chromatin and beyond: The multitasking roles for SIRT6. Trends in Biochemical Sciences 39: 72–81.
He, Y., Y. Xiao, X. Yang, Y. Li, B. Wang, F. Yao, C. Shang, Z. Jin, W. Wang, and R. Lin. 2017. SIRT6 inhibits TNF-α-induced inflammation of vascular adventitial fibroblasts through ROS and Akt signaling pathway. Experimental Cell Research 357: 88–97.
Zhang, L., L. Bai, Q. Ren, G. Sun, and Y. Si. 2018. Protective effects of SIRT6 against lipopolysaccharide (LPS) are mediated by deacetylation of Ku70. Molecular Immunology 101: 312–318.
Powell, A., and L. Davidson. 2015. Pediatric spinal cord injury: A review by organ system. Physical Medicine and Rehabilitation Clinics of North America 26: 109–132.
Nas, K., L. Yazmalar, V. Şah, A. Aydın, and K. Öneş. 2015. Rehabilitation of spinal cord injuries. World Journal of Orthopedics 6: 8–16.
Sánchez-Ventura, J., J. Amo-Aparicio, X. Navarro, and C. Penas. 2019. BET protein inhibition regulates cytokine production and promotes neuroprotection after spinal cord injury. Journal of Neuroinflammation 16: 124.
Zhang, Y., L. Wang, L. Meng, G. Cao, and Y. Wu. 2019. Sirtuin 6 overexpression relieves sepsis-induced acute kidney injury by promoting autophagy. Cell Cycle 18: 425–436.
Hu, W., H. Wang, Z. Liu, Y. Liu, R. Wang, X. Luo, and Y. Huang. 2017. Neuroprotective effects of lycopene in spinal cord injury in rats via antioxidative and anti-apoptotic pathway. Neuroscience Letters 642: 107–112.
Liu, G., G. Fan, G. Guo, W. Kang, D. Wang, B. Xu, and J. Zhao. 2017. FK506 attenuates the inflammation in rat spinal cord injury by inhibiting the activation of NF-κB in microglia cells. Cellular and Molecular Neurobiology 37: 843–855.
Reed, J.C. 2006. Proapoptotic multidomain Bcl-2/Bax-family proteins: Mechanisms, physiological roles, and therapeutic opportunities. Cell Death and Differentiation 13: 1378–1386.
Crowley, L.C., and N.J. Waterhouse. 2016. Detecting cleaved Caspase-3 in apoptotic cells by flow cytometry. Cold Spring Harbor Protocols.
Availability of Data and Materials
The analyzed data sets generated during the present study are available from the corresponding author on reasonable request.
Author information
Authors and Affiliations
Contributions
All authors made substantial contributions to the design of the study, performed the experiments, and analyzed the data.
Corresponding author
Ethics declarations
Competing Interests
All authors read and approved the final version of the manuscript. All authors declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhaohui, C., Shuihua, W. Protective Effects of SIRT6 Against Inflammation, Oxidative Stress, and Cell Apoptosis in Spinal Cord Injury. Inflammation 43, 1751–1758 (2020). https://doi.org/10.1007/s10753-020-01249-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10753-020-01249-2