Abstract
This study determined the relative influence of nutrients and stream habitat on three biological assemblages in five regions of the USA. Nutrient samples were collected over a four-week period, followed by habitat assessment and the collection of biological samples. Biological sampling included diatoms, invertebrates and fish, with three assemblage metrics used for each taxonomic group. We developed boosted regression tree (BRT) models for each biological assemblage metric within each region. Diatom BRT models indicated that nutrients, primarily orthophosphate or total phosphorus, played a larger role than habitat. Nutrients and habitat were approximately equal for invertebrate models with ammonia nitrogen and total organic nitrogen dominant. For fish, habitat had greater importance than nutrients, with total organic nitrogen and total phosphorus the dominant nutrients. Invertebrates had the highest model performance with average CV (cross-validation) R2 at 0.47, with diatoms at 0.31 and fish at 0.26. Strongest individual metrics included low phosphorus diatom taxa, tolerant invertebrate abundance and fish multimetric (fish MMI). These findings suggest that the influence of nutrient species varies regionally and by metric, with the relative importance of nutrients and habitat changing as one moves up the trophic level.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Elevated nutrient concentrations in streams can lead to a variety of problems for human health, aesthetic impairment, water use, aquatic biota and downstream transport (Dodds & Welch, 2000). Ecological concerns include increased quantities of algae and aquatic plants (Houser & Richardson, 2010), decreased oxygen levels (Correll, 1998; Waite et al., 2019), toxic algal blooms (Lopez et al., 2008; Loftin et al., 2016), loss in important habitat due to excessive plant growth and resulting sedimentation (Sand-Jensen, 1998; Wharton et al., 2006), and a shift in aquatic food webs (Bracewell et al., 2019). Changes in stream primary production, habitat and food webs all contribute to potential increases in exotic species (Carpenter et al., 1998).
The eutrophication of surface waters is partially due to the increased loading of both nitrogen and phosphorus (Carpenter et al., 1998; USEPA, 1998, 2016a; NRC, 2000). A national assessment of 499 streams across the USA found that total nitrogen (TN) and phosphorus (TP) were 2–10 times higher than EPA recommended regional nutrient criteria, with highest TN concentrations associated with agricultural streams and TP elevated in both agricultural and urban streams (Dubrovsky et al., 2010). Point sources play a dominant role in nutrient loads in urban lands, comprising greater than 50% of N and P (Preston et al., 2011). Coastal areas of the USA are particularly impacted due to high transport of nutrients from inland rivers (Alexander et al., 2008) and from disproportionate population growth and urbanization along coastlines (Oelsner & Stets, 2019). Despite limited improvements, many streams and rivers in the USA are near or over nutrient saturation levels (King et al., 2014).
Challenges in managing nutrients involve determining the nutrient concentrations that elicit negative impacts to streams and how nutrient–biota interactions are influenced by other factors. Early work on establishing target nutrient levels was based on the distribution of nutrient concentrations (USEPA, 1998; Dodds & Welch, 2000) and use of reference sites for establishing “acceptable” concentrations (Herlihy & Sifneos, 2008). A major challenge of this effort is the determination of nutrient thresholds, which are defined as transition points of altered ecological conditions due to small changes in nutrient concentration (Huggett, 2005; Groffman et al., 2006; Dodds et al., 2010). Thresholds have been used to assess an ecological change in algal biomass (Dodds et al., 2010), specific taxa (Sunderman et al., 2015) and assemblage metrics (Black et al., 2011; Waite et al., 2019). Because species and ecosystem responses tend to be nonlinear (Clements et al., 2010; Dodds et al., 2010), studies have moved toward the use of nonlinear models, as in CART (Stevenson et al., 2012), gradient forest (GF) (Wagenhoff et al., 2017) and boosted regression tree (Waite et al., 2021). BRT models are among a family of techniques used to advance single-classification or regression trees by combining the results of sequentially fit regression trees to reduce predictive error and improve overall performance (Elith et al., 2008). Boosted regression trees (BRTs) are ideal for addressing the relative importance of variables that may be highly skewed (Elith et al., 2008) and has been shown to be useful in recent stressor studies (Munn et al., 2018; Waite et al., 2019, 2021).
This study builds upon previous work done as part of the U.S. Geological Survey Stream Assessment Program. Waite et al. (2021) examined the response of the multimetric index (MMI) metric to numerous stressors (68) in various stressor categories including habitat, pesticides (herbicides, fungicides, insecticides, and toxicity), nutrients, water quality (conductivity and dissolved oxygen) and stream flow. While Waite et al. (2021) provided important information as to what major groups were having a strong influence on biota, it was a coarse-grain approach in that MMI are nonspecific stressor response indicators and the high number of stressors resulted in not being able to fully parse out nutrient species and habitat. Our study represents a more fine-grain approach in that nutrient species can be separated out as to their relative importance among biota and regions. This study relied on a nutrient gradient design across a large geographic area (434 sites from five regions), repeated nutrient sampling prior to biological sampling and the comparison of three taxonomic assemblages (algae, invertebrates and fish). The objectives of this study were to determine 1) what nutrient species are most associated with each biological assemblage and 2) what is the relative influence of nutrients and instream habitat on biological assemblages. For the first objective, we hypothesize that dissolved forms of dissolved inorganic nitrogen (DIN) and orthophosphate (OP) will be more important to biota due to dissolved nutrients being biologically available. For the second objective, we hypothesize that nutrients will be more important for the diatoms and become less important as one moves up the food chain where the effects from nutrients become indirect (note: an important score is determined for each explanatory variable by the BRT models; this is used as evidence for the stated hypotheses).
Methods
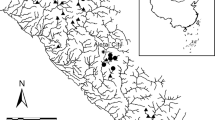
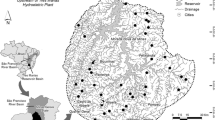
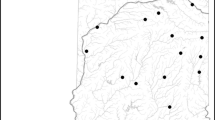
From 2013 to 2017, the USGS evaluated multiple stressors on the ecological condition of streams in five regions of the USA: Northwest (Sheibley et al., 2017), coastal California (May et al., 2020), Upper Midwest (Garrett et al., 2017), Northeast (Coles et al., 2019) and Southeast (Journey et al., 2015) (Fig. 1). A total of 434 wadeable streams were sampled across the five regions. A key design component of assessing nutrients and ecological effects is to select sites that span a gradient of nutrient concentrations, with a portion of sites falling below published threshold levels for TN (0.5 mg l−1) and TP (0.03 mg l−1) (Dodds et al., 2002; Stevenson et al., 2006). Sites were selected along a gradient of urban or agriculture land use to capture a range of nutrient conditions. Site-specific geospatial boundaries and geospatial characteristics are available from Qi & Nakagaki (2020a and 2020b) (Table 1).
Location of sites within the five regions assessed in this study (modified from Waite et al. 2021)
Habitat
Habitat variables followed protocols by USGS (Fitzpatrick et al., 1998) for the Northeast, Southeast and Northwest, whereas USEPA protocols were used for coastal California and Upper Midwest (USEPA, 2010). Habitat variables used in this study were comparable among the two protocols. Stream reaches of approximately 150 m were established at each site, with instream habitat collected at 11 equidistant transects. Quantitative measures used in this study included stream width (m), gradient, percent fines, substrate size (D84) and percent canopy (Table 2). Flow related metrics (peak stage, interval peak, stream depth and depth change) were determined using either established USGS gages or pressure transducers. Median and maximum water temperature prior to sampling were determined using temperature thermistors.
Diatoms, invertebrates and fish
Diatoms, invertebrate and fish were sampled using USGS methods (Moulton et al., 2002) in the Southeast, Northeast and Northwest regions, with USEPA methods (USEPA, 2013) in the Midwest and California. Algae and invertebrates were collected in the richest targeted habitat (e.g., riffles) in the USGS protocols, whereas they were collected along set transects in the USEPA protocol. Concerns about the differences between the two sampling protocols have been addressed by a variety of researchers and have been found to be comparable, especially across large regional studies (Ode et al., 2008; Waite et al., 2014; Cuffney & Kennen, 2018). Fish were collected using standard electroshock methods along the 150 m reach (Moulton et al., 2002; USEPA, 2013). Method details for each region can be seen in Journey et al. (2015, Garrett et al. (2017), Sheibley et al. (2017), Coles et al. (2019) and May et al. (2020).
Nutrients
Discrete water samples were collected weekly over four weeks prior to the collection of biological sampling, with the median of the four samples used for each site. Water samples were collected using either grab samples or the equal-width method based on flow (U.S. Geological Survey, 2006). Colorimetry was used to determine total phosphorus (TP, DL = 0.002 mg/l) concentrations (O’Dell, 1993) with orthophosphate (OP, DL = 0.002 mg/l) by colorimetric analysis (Fishman, 1993). The colorimetric analysis method was also used for dissolved ammonia (NH3 + NH4+, DL = 0.005 mg/l) and nitrite (DL = 0.005 mg/l) (Fishman, 1993), whereas dissolved nitrate plus nitrite was determined by low-level enzyme reduction (DL = 0.02 mg/l) (Patton & Kryskalla, 2011). DIN is the sum of dissolved nitrate, nitrite and ammonia. TON is total organic nitrogen, calculated as the difference between TN and DIN concentrations.
Ecological metrics and stressor variables
For each taxonomic group, we selected up to six metrics for running the initial analyses. The final metrics were selected based upon their known response to nutrient and habitat stressors and their successful use in prior studies (Munn et al., 2018; Waite et al., 2019, 2020; Table 2). There was also an effort to include metrics that represented different assemblage characteristics, like overall assemblage diversity versus trophic status. Diatom metrics included the biological condition index group 4 and 5 (BC 4–5) which include tolerant diatoms; low phosphorus taxa (Potapova & Charles, 2007) which include diatom taxa that respond to low levels of total phosphorus; and relative abundance of moderate and highly motile diatom taxa which indicate increasing nutrient concentrations (Porter et al., 2007).
Invertebrate metrics used in this study were calculated using methods presented in Cuffney et al. (2003, 2007). Metrics selected included scraper richness as a trophic metric, the average tolerance of invertebrate taxa to represent overall invertebrate tolerance to nutrients and a multimetric (invertebrate MMI) which discriminates across land use disturbance gradients (Herlihy et al., 2020).
Fish metrics captured various features of assemblages including richness and tolerances outlined in Meador & Brown (2015) and Meador & Carlisle (2007). Metrics included the fish MMI (USEPA, 2016b), the average of species tolerance to nitrate (Meador, 2012) and the percent benthic invertivore taxa to reflect trophic status of fish assemblages.
Six nutrient species were included as environmental stressors (Table 2), including four measures of nitrogen (TN, DIN, ammonia and TON) and two measures of phosphorus (TP and OP). While many of these measures can be correlated in specific regions, each is used to represent various measures of nutrient condition. Ten physical habitat measures were included in the BRT modeling, including two geomorphic (wetted width and gradient), two substrate (percent fines and substrate size D84), percent canopy cover, median and maximum water temperature, two measures of stream depth (stream depth and change in stream depth) and two measures of stream flow (peak flow and interval peak flow).
Data analysis and model development
For all instream environmental stressors, we focused on summary metrics related to the median of the water quality sampling (Waite et al., 2019). Algal, invertebrate and fish metrics were used to create Boosted regression tree models (Gaussian) using the gbm library in R (Version 4.2.1; R Project for Statistical Computing, Vienna, Austria) (2018). Explanatory variables included 11 habitat and six nutrient variables (Table 2). A minimum of 1000 trees per model was used to optimize the BRT model, with evaluation of interactions and partial dependency responses to reduce potential explanatory variables and overfitting (Elith et al., 2008). Variables with Variable Importance ((VI) scores < 7 were removed to improve interpretation. VI scores are a measure of how often a variable is selected across all the trees and it’s deviance improvement vs. when it is removed compared to all other variables evaluated in the model. Final VI scores are re-scales so all variables add up to 100 (Waite et al., 2021). BRT models were run for each of the regions in this study. Studies have demonstrated the strong influence of regional differences in biological assemblages and therefore the need for separate assessments (Potapova & Charles, 2007; Munn et al., 2009; Herlihy et al., 2020). The VI scores were used to assess the two objectives of the study as to what explanatory variables are most important to the three taxonomic groups.
Partial dependency plots show the stressor plotted against the biotic metric and modeled response form (solid line curve), with Lowess smooth curves (dashed lines) added to aid in the overall interpretation (Waite et al., 2019). CV (cross-validation) R2 values, which are less likely to be inflated by overfitting, were calculated by holding 25% of the sites out of each regression tree split and then using those sites to test the amount of deviance explained by the split (Elith et al., 2008).
Results
Nutrients
All five regions in our study contained a wide range in TP and TN concentrations (Table 3) with sites falling below and above published threshold levels for TP (0.03 mg l−1) and TN (0.5 mg l−1) (Dodds et al., 2002; Stevenson et al., 2006) (Fig. 2). However, the percentage of sites below the threshold levels varied depending on the region, with a low of 9 and 7% for TN and TP, respectively, in the Midwest to a high of 35 and 45% for TN and TP, respectively, in the Northeast. Overall, TN concentrations ranged from < 0.05 to 36.4 mg l−1 and TP ranged from 0.01 to 2.82 mg l−1 for TP (Table 3). Highest average concentrations of nitrogen (TN, DIN, TON and ammonia) occurred in the Midwest, which is the more agriculturally dominated region, with highest averages of phosphorus (TP and OP) from California. Lowest average concentrations of TN and DIN occurred in the Northeast, with the Northwest having the lowest average concentrations of TON, ammonia, TP and OP.
Distribution of TP by region based on the median weekly value for four weeks prior to sampling biological assemblages. The vertical line represents an ecological threshold for TP (0.03 mg l.−1, Dodds et al. 2002)
Pearson correlations of the six nutrient species indicated that all were significantly correlated (P < 0.05, n = 435, r = 0.28–0.95) with all regions combined; however, there were some differences at the regional scale. TN and TP were strongly correlated (P < 0.01) at r = 0.63–0.69 for Northwest, California and Northeast, whereas correlations were weaker in the Midwest and Southeast (r = 0.45 and 0.48, respectively). Studies normally focus on either total nutrients (TN and TP) or on the dissolved fractions (DIN and OP). Our study found that at the regional scale TN and DIN were highly correlated ranging from 0.92 to 0.99, whereas TP and OP ranged from 0.84 to 0.97. Therefore, using the total or dissolved forms of nitrogen or phosphorus provided similar results. In contrast, there were larger differences when comparing TN to either TON or ammonia. TN and TON correlations ranged from 0.43 to 0.87 with lowest correlations in the Midwest and Southeast and highest correlations in California and Northeast. The relationship between TN and ammonia was more varied with correlations ranging from r = − 0.1 (Midwest, P > 0.5) to r = 0.71 (California). However, the average r value was only 0.42 which indicates that ammonia can vary substantially with TN among regions. Correlations of TP with TON can be an indicator of TON source. In our study, TP was correlated with TON in all regions with lowest correlation in the Midwest (r = 0.48) and all other regions between r = 0.67 and 0.77.
BRT models
Forty-two BRT models were run using three taxonomic groups (algae, invertebrates and fish) in five regions; fish were not collected in California (BRT data in Supplemental Table). BRT was unable to construct four diatom models due to insufficient site separation, which reduced the number of BRT models to 38. Overall, invertebrate BRT models were the strongest with CV R2 values ranging from 0.25 to 0.63 (average = 0.47) with fish models the weakest, ranging from 0.04 to 0.55 (average = 0.26). A nutrient species was the dominant variable in most diatom models, with nutrients and habitat equal as the dominant variables for invertebrate models and habitat the dominant variables for fish models.
Diatoms
The low phosphorus diatom metric had the highest CV R values in the Midwest, Northeast and Southeast, whereas in the Northwest the diatom biological condition metric (BC 4–5) CV R2 was highest (Table 4). The motile diatom metric was consistently the poorest performer in all regions. None of the three diatoms metrics consistently outperformed others among the regions; however, diatom metrics were best explained by predictor variables in the Southeast. TP or OP was the dominant variable in 82% of diatom models, resulting in phosphorus having the highest average percent importance across all diatom models (Fig. 3); dissolved OP was found more important than TP in most models. In contrast, while various nitrogen species occurred in 9 of the 11 models (82%), it was total organic nitrogen (TON) that occurred in more BRT models with the highest average across all models (Fig. 3). While habitat variables were important in all diatom models, interval peak was the only habitat variable with the highest VI value for the BC-45 metric in the Northeast. Variables reflecting changes in streamflow were the most common habitat explanatory variable for diatoms, which included interval peak and change in depth; variables of lessor importance included stream width, substrate and peak flow.
Invertebrates
Invertebrate models had higher CV R2 values than diatoms or fish, with only minimal differences among the three metrics (Table 5). The Southeast region generally had the highest BRT CV R2 values and the Midwest with the lowest CV R2 values. Average invertebrate tolerance (RichTol) had the highest CV R2 values in 3 of the 5 regions. Invertebrates were the only taxonomic group that had approximately equal frequency of nutrient species and habitat variables as importance variables. Of the nutrient species, TON had the highest average percent importance value; however, ammonia was second with an average of 11% (Fig. 3). Habitat was equally as important overall in the BRT models, with 53% of models having a habitat variable as the most important explanatory variable. Interval peak and percent fines were the most common habitat variables in the models, with 33% of the models having interval peak as the first explanatory variable.
Fish
Fish metrics differed from diatoms and invertebrates in that habitat variables were generally more important than nutrient species (Table 6). TP occurred more often (50%) than TON (33%); both had similar average percent importance values at 9 and 8%, respectively (Fig. 3). While many habitat variables played a role in the BRT models, percent fines and maximum temperature (Tmax) were the dominant variables. While there were no strong patterns as to the strength of the three fish metrics, fish MMI tended to have a slightly higher score (as CV R2) in the Northwest, Midwest and Northeast, whereas fish invertivores were strongest in the Southeast.
Partial dependency plot comparisons
We used partial dependency plots to assess patterns of nutrient species found to be important in models. For comparison, we examined a single metric within each of the three taxonomic assemblages for three of the regions. The diatom low phosphorus model had the highest VI values among the diatom model with either OP or TP the highest nutrient importance value in the models. For all three regions, the diatom low phosphorus metric decreased with increased concentrations of OP or TP, with the breakpoint occurring around 0.12 mg l−1 (Fig. 4), with little change above 0.2 mg l−1.
Partial dependency plots from selected boosted regression models for the Northwest (left column), Midwest (middle column) and Southeast (right column). Metrics plotted include the diatom low phosphorus taxa metric (upper panel), average invertebrate tolerance metric (middle panel) and fish nitrate tolerance metric (lower panel). Solid line represents the BRT modeled line, and the dashed line represents a Lowess smooth curve
Ammonia was found to be important in the average invertebrate tolerance BRT models. Partial dependency plots indicated that average invertebrate tolerance increased with initial increases in ammonia, but then leveled off rapidly (Fig. 4). Breakpoints for the three regions varied only slightly, with the Northwest at 0.02 mg l−1, Midwest at 0.015 mg l−1 and Southeast a bit higher at 0.1 mg l−1. The ammonia vs invertebrate tolerance metric was unique in that the Lowess curve indicated an initial increase followed by a decrease over increasing ammonia concentrations.
For fish, TP was used to assess nutrient increases versus the nitrate-tolerant metric; TP was found to be positively correlated with TN (r = 0.45, P < 0.001). The BRT modeled line showed no apparent relationship between TP and nitrate-tolerant fish; however, the Lowess curve increased near the higher concentrations of TP (Fig. 4). In contrast, the Midwest showed nitrate-tolerant fish increasing slightly above 0.15 mg l−1. The Southeast region indicated the opposite pattern to the Midwest in that nitrate-tolerant fish decreased at lower levels of TP, near 0.05 mg l−1.
Discussion
The dominant role of nutrients in the diatom models reflects their position at the base of the food web where they are directly influenced by nitrogen and phosphorus (Hering et al., 2006). This supports our second hypothesis that nutrients can be more important than habitat for diatoms. The role of phosphorus in the diatom models indicates its importance as a limiting nutrient (Downing et al., 2001; Smith, 2003; Camargo et al., 2005). However, nitrogen can be the limiting nutrient in streams with low N:P loading ratios (Smith, 2003), which may explain the importance of DIN for diatom biological condition in the Northwest. N:P ratios were highly mixed depending on the site and region with California and the Southeast more nitrogen limited (N:P < 10) and the Midwest more phosphorus limited (N;P > 20). Overall, nutrient limitation varies substantially by taxonomic group and region (Borchardt, 1996; Mebane et al., 2021). Furthermore, anthropogenic factors greatly influence the loadings of nitrogen and phosphorus to streams thereby influencing the relative limitation of either nitrogen or phosphorus (Jarvie et al., 2018). The relative equal mix of TP and OP as dominant factors in models relates to the high correlation between the two measures.
While we hypothesized that DIN would be the dominant form of nitrogen for diatoms, we found TON had the greatest influence on diatom metrics followed by TN and DIN (highly correlated). TON (total organic nitrogen) is not commonly analyzed as part of algal-nutrient studies; however, organic nitrogen can comprise up to 80% of TN in surface waters and is common in both urban (Scott et al., 2007) and agricultural settings (Hussain et al., 2020). In our study, TON composed an average of 36% of TN but varied widely across sites (1–100%). The importance of TON was shown by it occurring in 37% of the diatom BRT models, with it the dominant variable in 50% of the invertebrate models. TON was associated with agricultural land use in California, Northwest and Midwest, whereas TON was associated with urban land use in the Northwest and Northeast. The source of organic nitrogen includes soil runoff, litter decomposition on land and biological production in streams (Hussain et al., 2020). Overall, TON provides a major pool of nitrogen in stream and rivers and is a function of sources, hydrological processes and instream biogeochemical processes (Scott et al., 2007), with the TON pool consisting of a mixture of organic nitrogen compounds. While algae can utilize nitrate and ammonia directly, organic nitrogen needs to be biologically transformed into NH4, urea or nitrate (Berman & Chava, 1999). Taken together, TON can provide an important pool of nitrogen in flowing water systems (Scott et al., 2007), which is reflected in the BRT models.
While nutrients enhance the accrual (colonization plus growth) of benthic algae, light and temperature are also key to algal development, with hydrologic stability and grazing important for biomass loss (Biggs, 1996). Of the habitat variables examined in the BRT models, we found metrics related to stream flow, as in median daily change in depth and the number of days between high flow events (Interval Peak Flow and Depth change), to be the most common, followed by measures of substrate (percent substrate fines and substrate D84). It is well documented that the combination of streamflow and sediment is important controlling factors of benthic algae (Lamberti, 1996; Peterson, 1996; Munn et al., 2018).
As indicated by the Lowess curve, the Subsidy–Stress relationship (Odum et al., 1979) best describes the interaction of nutrients with invertebrates; at low but increasing nutrient concentrations there is often an increase in algae which has a positive influence on invertebrates (Chetelat et al., 1999); however, when nutrients become excessive, streams become more eutrophic which leads to a negative influence on invertebrate assemblages (Wang et al., 2007; Ouyang et al., 2018; Waite et al., 2019). The negative influence of nutrients on invertebrates can be due to a combination of factors as in changes in substrate due to increased primary production (Sand-Jensen, 1998; Wharton et al., 2006), food quantity and quality (King & Richardson, 2007), and dissolved oxygen levels (Correll, 1998). The result is often a change in assemblage diversity and composition of loss of native species (Carpenter et al., 1998; Smith et al., 1999). BRT models indicated that nutrients explained approximately half of the invertebrate metrics, with ammonia the most frequent form of nitrogen. Ammonia includes ionized ammonia (NH4+) and unionized ammonia (NH3), and while NH4+ is normally the dominant form, the relative proportions of each are dependent on pH and temperature (USEPA, 2013b). NH3 is the toxic form of ammonia and has been shown to be particularly detrimental to mussels and some species of fish, but less toxic to aquatic invertebrates (USEPA, 2013b). While the frequency of ammonia in the BRT models (60%) could indicate potential toxicity, concentrations of ammonia only exceeded USEPA chronic criteria (1.3 mg/l at pH 7.7 and water temperature of 20.3 °C) at one site (USEPA, 2013b), and therefore, the importance of ammonia likely indicates overall eutrophication more than toxicity. However, it is important to note that ammonia varies rapidly in streams due to input and uptake, and therefore, concentrations can exceed levels of concern for short periods. Furthermore, the assessment of ammonia toxicity is based upon limited taxa and measures; therefore, low-level effects could be a factor. Ammonia is a key nitrogen species for biological processes given it is assimilated by bacteria and algae, but also can undergo nitrification which converts ammonia to nitrate for additional assimilation (Bernhardt et al., 2002). TN or nitrate, which often accounts for most of the TN, is often reported as an important nutrient associated with a negative (Yuan, 2010) or positive (Piggott et al., 2012) influence on invertebrates.
Stream habitat played an equally important role as nutrients influencing invertebrate metrics in California, Northwest and Northeast, with habitat having a larger influence on invertebrates in the Midwest and Southeast. Of the habitat variables, interval peak stage and substrate fines occurred in more of the invertebrate models than other habitat variables. In an experimental study, Juvigny-Khenafou et al. (2020) found that flow reduction had a larger influence on invertebrate assemblages than fine sediment, which was followed by nutrients. Flow alteration modifies the diffusion of materials and instream habitat (Calapez et al., 2018; Lange et al., 2018) and can dislodge invertebrates from their habitat. Fine sediment, which can change in relation to stream flow, can reduce habitat heterogeneity by filling interstitial spaces (Piggott et al., 2015) and scouring benthic habitat. Fine sediment was equally important for both invertebrates and fish metrics, occurring in 50% of models.
BRT models indicated that fish metrics were influenced by physical habitat more than by nutrients, which supports our second hypothesis. However, nutrients are well documented for influencing fish assemblages, including TP (Meador & Carlisle, 2007; Atique & An, 2018; Waite et al., 2019) and TN and its dissolved forms (Meador & Carlise, 2007; Meador & Frey, 2018; Waite et al., 2019). Except for ammonia as a potential toxicant, nitrogen and phosphorus influence fish assemblages indirectly in the same way they influence invertebrates via increases in primary production (Kratina et al., 2012) which influences the overall food resources (Bracewell et al., 2019; Blocher et al., 2020), while also influencing habitat (e.g., substrate, macrophyte cover, etc.) and dissolved oxygen concentrations (Meador & Carlise, 2007; Munn et al., 2020).
The key habitat variables of importance to fish assemblages included percent fines, water temperature and changes in stream flow. As with invertebrates, percent fines reduce important habitat and limit food resources for insectivorous fish (Sullivan & Watzin, 2010; Meador and Frey, 2018; Blocher et al., 2020). Water temperatures and streamflow are important variables for fish distribution (VanCompernolle et al., 2019); minimum daily temperatures are critical for permitting species to tolerate periods of higher temperature (Farless & Brewer, 2017).
Conclusions
This study demonstrated the varying influences of nutrients and habitat on three biological assemblages in streams. Our hypothesis that the dissolved forms of nutrients (DIN and OP) would dominant the relationships of nutrients to biological metrics was not fully supported. We found both OP and TP to be key variables, but with nitrogen we found TON to dominant as an explanatory variable. However, our second hypothesis was supported in that the BRT models indicated that diatoms were more influenced by nutrients than habitat; invertebrates were equally influenced by both nutrients and habitat, and habitat had a greater control than nutrients on fish. Furthermore, phosphorus (orthophosphate or total phosphorus) was the key nutrient for diatoms, with ammonia and total organic nitrogen of approximate equal importance for invertebrates, and a mix of total organic nitrogen and total phosphorus the dominant nutrients for fish metrics. Diatoms and invertebrates both had a distinct breakpoint in a metric in response to increasing nutrient concentrations; however, fish tended to have more muted changes. Strongest individual biotic metrics included low phosphorus diatom taxa, tolerant invertebrate abundance and fish multimetric index (Fish MMI). This study illustrates that various nutrient species can have a dominant influence on biota depending on the assemblage and conditions, and that nutrients play a more important role at the food base with habitat becoming more important in higher trophic levels.
Data availability
All data supporting the findings of this study are available with the paper and its Supplementary Information.
References
Alexander, R. B., R. A. Smith, G. E. Schwarz, E. W. Boyer, J. V. Noland & J. W. Brakebill, 2008. Differences in phosphorus and nitrogen delivery to the Gulf of Mexico from the Mississippi River Basin. Environmental Science and Technology 42: 822–830.
Atique, U. & K. G. An, 2018. Stream health evaluation using a combined approach of multi-metric chemical pollution and biological integrity models. Water. https://doi.org/10.3390/w10050661.
Berhardt, E. S., R. O. Hall & G. E. Likens, 2002. Whole-system estimates of nitrification and nitrate uptake in streams of the Hubbard Brook experimental forest. Ecosystems 5: 419–430.
Berman, T. & S. Chava, 1999. Algal growth on organic compounds as nitrogen sources. Journal of Plankton Research 21: 1423–1437.
Biggs, B. J. F., 1996. Hydraulic habitat of plants in streams. Regulated Rivers: Research and Management 12: 131–144.
Black, R. W., P. W. Moran & J. D. Frankforter, 2011. Response of algal metrics to nutrient and physical factors and identification of nutrient thresholds in agricultural streams. Environmental Monitoring and Assessment 175: 397–417.
Blocher, J. R., M. R. Ward, C. D. Mattaei & J. J. Piggott, 2020. Multiple stressors and stream macroinvertebrate community dynamics: interactions between fine sediment grain size and flow velocity. Science of the Total Environment 717: 137070.
Borchardt, M. A., 1996. Nutrients. In Stevenson, R. J., M. L. Bothwell & R. L. Lowe (eds), Algal ecology: freshwater benthic ecosystems Academic Press: 183–227.
Bracewell, S., R. C. M. Verdonschot, R. B. Schafer, A. Bush, D. R. Lapen & P. J. Van den Brink, 2019. Qualifying the effects of single and multiple stressors on the food web structure of Dutch drainage ditches using a literature review and conceptual models. Science of the Total Environment 684: 727–740.
Calapez, A. R., S. R. Q. Serra, J. M. Santos, P. Branco, T. Ferreira, T. Hein, A. G. Brito & M. J. Feio, 2018. The effect of hypoxia and flow decrease in macroinvertebrate functional responses: a trait-based approach to multiple-stressors in mesocosms. Science of the Total Environment 637–638: 647–656. https://doi.org/10.1016/j.scitotenv.2018.05.071.
Camargo, J. A., A. Alonso & M. de la Puente, 2005. Eutrophication downstream from small reservoirs in mountain rivers of central Spain. Water Resources 39: 3376–3384.
Carpenter, S. R., N. F. Caraco, D. L. Correll, R. W. Howarth, A. N. Sharpley & V. H. Smith, 1998. Nonpoint pollution of surface waters with phosphorus and nitrogen. Ecological Application 8: 559–568.
Chetelat, J., F. R. Pick, A. Morin & P. B. Hamilton, 1999. Periphyton biomass and community composition in rivers of different nutrient status. Canadian Journal of Fisheries and Aquatic Sciences 56: 560–569.
Clements, W. H., N. K. M. Vieira & D. L. Sonderegger, 2010. Use of ecological thresholds to assess recovery in lotic ecosystems. Journal of the North American Benthological Society 29: 1017–1023.
Coles, J.F., K. Riva-Murray, P.C. van Metre, D.T. Button, A.H. Bell, S.L. Qi, C.A. Journey & R.W. Sheibley, 2019. Design and methods of the U.S. Geological Survey Northeast Stream Quality Assessment (NESQA), 2016. U.S. Geological Survey Open-File Report 2018–1183. 46 p.
Correll, D. L., 1998. The role of phosphorus in the eutrophication of receiving waters: a review. Journal of Environmental Quality 27: 261–266.
Cuffney, T. F. & J. G. Kennen, 2018. Potential pitfalls of aggregating aquatic invertebrate data from multiple agency sources: implications for detecting aquatic assemblage change across alteration gradients. Freshwater Biology 63(8): 1–14. https://doi.org/10.1111/fwb.13031.
Cuffney, T. F., M. D. Bilger & A. M. Haigler, 2007. Ambiguous taxa: effects on the characterization and interpretation of invertebrate assemblages. Journal of the North American Benthological Society 26: 286–307.
Cuffney, T.F., 2003. User’s manual for the National Water-Quality Assessment Program invertebrate data analysis system (IDAS) software: Version 3. Open-File Report 03–172. U.S. Geological Survey, Raleigh, North Carolina.
De’ath, G., 2007. Boosted trees for ecological modeling and prediction. Ecology 88: 243–251.
Dodds, W. K. & E. B. Welch, 2000. Establishing nutrient criteria in streams. Journal of North American Benthological Society 19(1): 186–196.
Dodds, W. K., V. H. Smith & K. Lohman, 2002. Nitrogen and phosphorus relationships to benthic algal biomass in temperature streams. Canadian Journal of Fisheries and Aquatic Science 59: 865–874.
Dodds, W.K., W.H. Clements. K. Gido, R.H. Hilderbrand & R.S. King, 2010. Thresholds, breakpoints, and nonlinearity in freshwaters as related to management Journal of North American Benthological Society 29(3): 988–997.
Downing, J. A., S. B. Watson & E. McCauley, 2001. Predicting cyanobacteria dominance in lakes. Canadian Journal of Fisheries and Aquatic Sciences 58: 1905–1908.
Dubrovsky, N.M., K.R. Burow, G.M. Clark, J.M. Gronberg, P.A. Hamilton, K.J. Hitt, D.K. Mueller, M.D. Munn, B.T. Nolan, L.J. Puckett, M.G. Rupert, T.M. Short, N.E. Spahr, L.A. Sprague & W.G. Wilber, 2010. The quality of our Nation’s waters—nutrients in the Nation’s streams and groundwater, 1992–2004: U.S. Geological Survey Circular 1350, 174 p., https:// pubs.usgs.gov/circ/1350/.
Elith, J., J. R. Leathwick & T. Hastie, 2008. A working guide to boosted regression trees. Journal of Animal Ecology 77: 802–813.
Farless, N. A. & S. K. Brewer, 2017. Thermal tolerances of fishes occupying groundwater and surface-water dominated streams. Freshwater Science 36(4): 866–876.
Fishman, M. J., 1993. Methods of analysis by the US Geological Survey National Water Quality Laboratory—Determination of inorganic and organic constituents in water and fluvial sediments. Open-File Report 93–125. United States Geological Survey, Denver, Colorado. (https://pubs.usgs.gov/of/1993/0125/report.pdf )
Fitzpatrick, F.A., I.R. Waite, P.J. D’Arconte, M.R. Meador, M.A. Maupin & M.E. Gurtz, 1998. Revised methods for characterizing stream habitat in the National Water-Quality Assessment Program: U.S. Geological Survey Water-Resources Investigations Report 98–4052, 67 p.
Garrett, J.D., J.W. Frey, P.C. Van Metre, C.A. Journey, N. Nakagaki, D.T. Button & L.H. Nowell, 2017. Design and methods of the Midwest Stream Quality Assessment (MSQA), 2013. U.S. Geological Survey Open-File Report 2017– 1073, 59 p., Denver, CO.
Groffman, P.M., J.S. Baron, T. Blett, T., A.J. Gold, I. Goodman, L,H, Gunderson, et al., 2006. Ecological thresholds: the key to successful environmental management or an important concept with no practical application? Ecosystems 9: 1-13.
Hering, D., R. K. Johnson, S. Kramm, S. Schmutz, K. Szoszkiewicz & R. F. M. Verdonschot, 2006. Assessment of European streams with diatoms, macrophytes, macroinvertebrates, and fish: a comparative metric-based analysis of organism response to stress. Freshwater Biology 51: 1757–1785.
Herlihy, A. T. & J. C. Sifneos, 2008. Developing nutrient criteria and classification schemes for wadeable streams in the conterminous US. Journal of North American Benthological Society 27(4): 932–948.
Herlihy, A. T., J. C. Sifneos, R. M. Hughes, D. V. Peck & R. M. Mitchell, 2020. The relation of lotic fish and benthic macroinvertebrate condition indices to environmental factors across the conterminous USA. Ecological Indicators 112: 105958.
Houser, J. N. & W. B. Richardson, 2010. Nitrogen and phosphorus in the Upper Mississippi River: transport, processing, and effects on the river ecosystem. Hydrobiologia 640: 71–88.
Huggett, A. J., 2005. The concept and utility of ‘ecological thresholds’ in biodiversity conservation. Biological Conservation 124: 301–310.
Hussain, M. Z., G. P. Robertson, B. Basso & S. K. Hamilton, 2020. Leaching losses of dissolved organic carbon and nitrogen from agricultural soils in the upper US Midwest. Science of the Total Environment 734: 139379.
Jarvie, H. P., D. R. Smith, L. R. Norton, F. K. Edwards, M. J. Bowes, S. M. King, P. Scarlett, S. Davies, R. M. Dils & N. Bachiller-Jareno, 2018. Phosphorus and nitrogen limitation and impairment of headwater streams relative to rivers in Great Britain: a national perspective on eutrophication. Science of the Total Environment 621: 849–862.
Journey, C.A., P.C. Van Metre, A.H. Bell, J.D. Garrett, D.T. Button, N. Nakagaki, S.L. Qi & P.M. Bradley, 2015. Design and methods of the Southeast Stream Quality Assessment (SESQA), 2014. USGS Open File Report 46. U.S. Geological Survey, Reston, VA.
Juvigny-Khenafou, N. P. D., J. J. Piggott, D. Atkinson, Y. Zhang, S. J. Macaulay, N. Wu & C. D. Matthaei, 2020. Impacts of multiple anthropogenic stressors on stream macroinvertebrate community composition and functional diversity. Ecology and Evolution. https://doi.org/10.1002/ece3.6979.
King, R. S. & C. J. Richardson, 2007. Subsidy-stress response of macroinvertebrate community biomass to a phosphorus gradient in an oligotrophic wetland ecosystem. Journal of North American Benthological Society 26(3): 491–508.
King, S. A., J. B. Heffernan & M. J. Cohen, 2014. Nutrient flux, uptake, and autotrophic limitation in streams and rivers. Freshwater Science 33: 85–98.
Kratina, P., H. S. Greig, O. L. Thompson, T. S. A. Carvalho-Pereira & J. B. Shurin, 2012. Warming modifies trophic cascades and eutrophication in experimental freshwater communities. Ecology 93: 1421–1430.
Lamberti, G.A., 1996. The role of periphyton in benthic food webs. In: Stevenson, R.T., Bothwell, M.L., & Lowe, R.L. (eds), Algal Ecology Academic Press, San Diego, California, pp. 533–572.
Lange, K., P. Meier, C. Trautwein, M. Schmid, C. T. Robinson, C. Weber & J. Brodersen, 2018. Basin-scale effects of small hydropower on biodiversity dynamics. Frontiers in Ecology and the Environment 16: 397–404. https://doi.org/10.1002/fee.1823.
Loftin, K.A., J.M. Clark, C.A. Journey, D.W. Kolpin, P.C. Van Metre, D. Carlise & P.M. Bradley, 2016. Spatial and temporal variation in microcystin occurrence in wadeable streams in the southeastern United States. Environmental Toxicology and Chemistry 35(9):2281-2287
Lopez, C.B., E.B. Jewett, Q. Dortch, B.T. Walton & H.K. Hudnell, 2008. Scientific Assessment of Freshwater Harmful Algal Blooms. Interagency Working Group on Harmful Algal Blooms, Hypoxia, and Human Health of the Joint Subcommittee on Ocean Science and Technology. Washington, DC.
May, J.T., L.H. Nowell, J.F. Coles, D.T. Button, A.H. Bell, S.L. Qi & P.C. Van Metre, 2020. Design and Methods of the California Stream Quality Assessment (CSQA), 2017. U.S. Geological Survey Open-File Report 2020–1023.
Meador, M. R., 2012. Nutrient enrichment and fish nutrient tolerance: assessing biological relevant nutrient criteria. Journal of the American Water Resources Association. https://doi.org/10.1111/jawr.12015.
Meador, M. R. & D. M. Carlise, 2007. Quantifying tolerance indicator values for common stream fish species of the United States. Ecological Indicators 7: 329–338.
Meador, M. R. & L. M. Brown, 2015. Life history strategies of fish species and biodiversity in eastern USA streams. Environmental Biology of Fish 98: 663–677. https://doi.org/10.1007/s10641-014-0304-1.
Meador, M. R. & J. W. Frey, 2018. Relative importance of water quality stressors in predicting fish community responses in Midwestern streams. Journal of the American Water Resources Association 54: 708–723.
Mebane, C. A., A. M. Ray & A. M. Marcarelli, 2021. Nutrient limitation of algae and macrophytes in streams: Integrating laboratory bioassays, field experiments, and field data. PLoS ONE 16(6): e0252904. https://doi.org/10.1371/journal.pone.0252904.
Moulton S.R., J.G. Kennen, R.M. Goldstein & J.A. Hambrook, 2002. Revised protocols for sampling algal, invertebrate, and fish communities as part of the National Water-Quality Assessment Program U.S. Geological Survey Open-File Report 02–150.
Munn, M. D., I. R. Waite, D. P. Larsen & A. T. Herlihy, 2009. The relative influence of geographic location and reach-scale habitat on benthic invertebrate assemblages in six ecoregions. Environmental Monitoring and Assessment 154: 1–14. https://doi.org/10.1007/s10661-008-0372-9.
Munn, M. D., I. Waite & C. P. Konrad, 2018. Assessing the influence of multiple stressors on stream diatom metrics in the upper Midwest, USA. Ecological Indicators 85: 1239–1248. https://doi.org/10.1016/j.ecolind.2017.09.005.
Munn, M. D., R. W. Sheibley, I. R. Waite & M. R. Meador, 2020. Understanding the relationship between stream metabolism and biological assemblages. Freshwater Science 39(4): 680–692.
NRC (National Research Council), 2000. Clean Coastal Waters: Understanding and Reducing the Effects of Nutrient Pollution, National Academy Press, Washington, DC:
O’Dell, J.W., 1993. Method 365.1, Revision 2.0: Determination of Phosphorus by Semi-Automated Colorimetry. US Environmental Protection Agency. Cincinnati, OH. 18 pp.
Ode, P. R., C. P. Hawkins & R. D. Mazor, 2008. Comparability of biological assessments derived from predictive models and multimetric indices of increasing geographic scope. Journal of the North American Benthological Society 27: 967–985.
Odum, E. P., J. T. Finn & E. H. Franz, 1979. Perturbation theory and the subsidy-stress gradient. BioScience 29: 349–352.
Oelsner, G. P. & E. G. Stets, 2019. Recent trends in nutrient and sediment loading to coastal areas of the U.S.: Insights and global context. Science of the Total Environment 654: 1225–1240.
Ouyang, Z., S. S. Qian, R. Becker & J. Chen, 2018. The effects of nutrients on stream invertebrates: a regional estimation by generalized propensity score. Ecological Processes 7: 21. https://doi.org/10.1186/s13717-018-0132-x.
Patton, C.J. & J.R. Kryskalla, 2011. Colorimetric determination of nitrate plus nitrite in water by enzymatic reduction, automated discrete analyzer methods. United States Geological Survey Techniques and Methods 5-B8. United States Geological Survey, Reston, Virginia. (https://pubs.er.usgs.gov/publication/tm5B8)
Peterson, C.G., 1996. Response of benthic algal communities to natural physical disturbance. In: Stevenson, R.T., Bothwell, M.L., Lowe, R.L. (Eds.), Algal Ecology Academic Press, San Diego, California, pp. 375–402.
Piggott, J. J., K. Lange, C. R. Townsend & C. D. Matthaei, 2012. Multiple stressors in agricultural streams: a mesocosm study of interactions among raised water temperature, sediment addition and nutrient enrichment. PLoS ONE 7(11): e49873. https://doi.org/10.1371/journal.pone.0049873.
Piggott, J. J., K. Lange, C. R. Townsend & C. D. Matthaei, 2015. Reconceptualizing synergism and antagonism among multiple stressors. Ecological Evolution 7: 1538–1547.
Porter, S. D., D. K. Mueller, N. E. Spahr, M. D. Munn & N. M. Dubrovsky, 2007. Efficacy of algal metrics for assessing nutrient and organic enrichment in flowing waters. Freshwater Biology 53: 1036–1054.
Potapova, M. & D. F. Charles, 2007. Diatom metrics for monitoring eutrophication in rivers of the United States. Ecological Indicators 7: 48–70.
Preston, S. D., R. B. Alexander, G. E. Schwarz & C. G. Crawford, 2011. Factors affecting stream nutrient loads: a synthesis of regional SPARROW model results for the continental United States 1: factors affecting stream nutrient loads: a synthesis of regional SPARROW model results for the continental United States. Journal of American Water Resources Association. 47: 891–915. https://doi.org/10.1111/j.1752-1688.2011.00577.x.
Qi, S.L. & N. Nakagaki, 2020a. Geospatial Database of the Study Boundaries, Sampled Sites, Watersheds, and Riparian Zones for the U.S. Geological Survey Regional Stream Quality Assessment. U.S. Geological Survey Data Release https://doi.org/10.5066/P90YQ9TW.
Qi, S.L. & N. Nakagaki, 2020b. Selected Environmental Characteristics of Sampled Sites, Watersheds, and Riparian Zones for the U.S. Geological Survey Regional Stream Quality Assessment, 2013 to 2017. U.S. Geological Survey Data Release 10. 5066/P962N215.
Sand-Jensen, K., 1998. Influence of submerged macrophytes on sediment composition and near-bed flow in lowland streams. Freshwater Biology 39(4): 663–679.
Scott, D., J. Harvey, R. Alexander & G. Schwarz, 2007. Dominance of organic nitrogen from headwater streams to large rivers across the conterminous United States. Global Biogeochemical Cycles 21:GB1003
Sheibley, R.W., J.L. Morace, C.A. Journey, P.C. Bell, D.T. Button, S.L. Qi & P.C. Van Metre, 2017. Design and methods of the Pacific Northwest Stream Quality Assessment (PNSQA), 2015. U.S. Geological Survey Open-File Report 2017–1103. 46.
Smith, V. H., 2003. Eutrophication of freshwater and coastal marine ecosystems: a global problem. Environmental Science of Polluted Rivers 10: 126–139.
Smith, V. H., G. D. Tilman & J. C. Nekola, 1999. Eutrophication: Impacts of excess nutrient inputs of freshwater, marine, and terrestrial ecosystems. Environmental Pollution 100: 179–196.
Stevenson, R. J., S. R. Rier, C. M. Riseng, R. E. Schulz & M. J. Wiley, 2006. Comparing effects of nutrients on algal biomass in streams in two regions with different disturbance regimes. Hydrobiologia 561: 149–165.
Stevenson, R. J., B. J. Bennett, D. N. Jordan & R. D. French, 2012. Phosphorus regulates stream injury by filamentous green algae, DO, and pH with threshold response. Hydrobiologia 695: 25–42.
Sullivan, S. M. P. & M. C. Watzin, 2010. Towards a functional understanding of the effects of sediment aggradation on stream fish condition. River Research and Applications 26: 1298–1314.
Sunderman, A., M. Leps, S. Leisner & P. Haase, 2015. Taxon-specific physico-chemical change points for stream benthic invertebrates. Ecological Indicators 57: 314–323.
U.S. Geological Survey, 2006. National Field Manual for the Collection of Water-Quality Data. Chapter A4. Collection of Water Samples. U.S. Geological Survey Techniques of Water-Resources Investigations Reston, VA. 231 pp.
USEPA, 1998. National strategy for the development of regional nutrient criteria. U.S. Environmental Protection Agency Office of Water Fact Sheet EPA-822-F-98–002. http://www.epa.gov/ waterscience/criteria/nutrient/nutsi.html. Accessed November 2006
USEPA, 2010. National Rivers and Streams Assessment Quality Assurance Project Plan. United States Environmental Protection Agency Office of Water, Office of Environmental Information, Washington, DC.
USEPA, 2013a. National Rivers and Streams Assessment 2013‐2014: Field Operations Manual – Wadeable. EPA‐841‐B‐12‐009b. U.S. Environmental Protection Agency, Office of Water Washington, DC.
USEPA, 2013b. Aquatic life ambient water quality criteria for ammonia – Freshwater 2013. EPA 822-R-18–002. U.S. Environmental Protection Agency, Office of Water Washington, DC
USEPA, 2016a. Nitrogen and Co-Pollutants: Research Roadmap. EPA 601/R-17/004 Washington, D.C.
USEPA, 2016b. National Rivers and Streams Assessment 2008–2009 Technical Report. EPA 841/R-16/008. Office of Water and Office of Research and Development, US Environmental Protection Agency, Washington, DC.
VanCompernolle, M., J. H. Knouft & D. L. Finklin, 2019. Multispecies conservation of freshwater fish assemblages in response to climate change in the southeastern United States. Biodiversity Research. https://doi.org/10.1111/ddi.12948.
Wagenhoff, A., J. E. Clapcott, K. E. M. Lau, G. D. Lewis & R. G. Young, 2017. Identifying congruence in stream assemblage thresholds in response to nutrient and sediment gradients for limit setting. Ecological Applications 27(2): 469–484.
Waite, I. R., J. G. Kennen, J. T. May, L. R. Brown, T. F. Cuffney, K. A. Jones & J. L. Orlando, 2014. Stream macroinvertebrate response models for bioassessment metrics: addressing the issue of spatial scale. PLoS ONE 9: 1–21.
Waite, I. R., M. D. Munn, P. W. Moran, C. P. Konrad, L. H. Nowell, M. R. Meador, P. C. Van Metre & D. M. Carlisle, 2019. Effects of urban multi-stressors on three stream biotic assemblages. Science of the Total Environment 660: 1472–1485.
Waite, I. R., Y. Pan & P. M. Edwards, 2020. Assessment of multi-stressors on compositional turnover of diatom, invertebrate and fish assemblages along an urban gradient in Pacific Northwest streams (USA). Ecological Indicators 112: 106047.
Waite, I. R., P. C. Van Metre, P. W. Moran, C. P. Konrad, L. H. Nowell, M. R. Meador, M. D. Munn, T. S. Schmidt, A. C. Gellis, D. M. Carlisle, P. M. Bradley & B. J. Mahler, 2021. Multiple in-stream stressors degrade biological assemblages in five U.S. regions. Science of the Total Environment 800: 14950.
Wang, L., D. M. Robertson & P. J. Garrison, 2007. Linkages between nutrients and assemblages of macroinvertebrates and fish in wadeable streams: Implications to nutrient criteria development. Environmental Management 39: 194–212.
Wharton, G., J. A. Cotton, R. S. Wotton, J. A. B. Bass, C. M. Heppell, M. Trimmer, I. A. Sanders & L. L. Warren, 2006. Macrophytes and suspension-feeding invertebrates modify flows and fine sediments in the Frome and Piddle catchments, Dorset (UK). Journal of Hydrology 330: 171–184.
Yuan, L. L., 2010. Estimating the effects of excess nutrients on stream invertebrates from observational data. Ecological Applications 20(1): 110–125.
Funding
This research was funded by the U.S. Geological Survey’s National Water Quality and Assessment Program.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors do not have any relevant financial or non-financial interests to disclose.
Additional information
Handling editor: Verónica Ferreira
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Munn, M.D., Waite, I., Sheibley, R.W. et al. The influence of stream nutrients and habitat on three biological assemblages. Hydrobiologia (2024). https://doi.org/10.1007/s10750-024-05680-6
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10750-024-05680-6