Abstract
Heterophylly, a type of intraspecific trait variation (ITV), is commonly observed in aquatic and amphibious plants. In this study, we focused on Luronium natans (L.) Raf., as a model species to investigate ITV, specifically looking at variations within different leaf types. The objective was to test general hypotheses regarding ITV across various leaf traits. We collected 210 leaves from the homogeneous environments of softwater lakes in northwestern Poland to measure six leaf traits: leaf area (LA), leaf dry weight (LDW), specific leaf area (SLA), leaf dry mass content (LDMC), circularity, and leaf width/length ratio (shape traits). These leaves were categorized into three types based on the shoot: heterophyllous shoots with floating leaves (HF), heterophyllous shoots with submerged leaves (HS), and submerged rosettes with linear leaves (S). Significant differences were observed in LA and LDMC within these leaf types and in SLA between HF and S types. ITV within each shoot type exhibited a wide range of variation, particularly in LA, LDW, SLA, and LDMC, with shape traits displaying stronger variability than functional traits. Our results indicate lower ITV of functional leaf traits within the same heterophyllous shoots compared to fully submerged leaves in a rosette form. We also noted negative correlations between SLA-LDMC and LA-circularity trait pairs, suggesting a variation–covariation pattern in leaf traits with minimal influence from water environmental factors.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Phenotypic variation has been recognized for centuries, with Darwin’s work in 1859 being a cornerstone in this area (Darwin, 1859). Subsequent studies by Gleason (1926) and Clements (1936) further elucidated how trait variation within plant species contributes to the structure of plant communities. Numerous studies have highlighted the role of phenotypic variation in shaping the diversity of plant forms, species coexistence, community structure (Jung et al., 2010), and ecosystem processes (Lavorel & Garnier, 2002; Funk et al., 2017). In recent decades, functional and trait-based ecology have provided conceptual frameworks and methodologies to study intraspecific trait variation ITV (Albert et al., 2010, 2011). However, trait variation among different species exceeds ITV (Siefert et al., 2015; Westerband et al., 2021). A global meta-analysis found that ITV accounts for approximately 25% of total trait variation within communities (Siefert et al., 2015). ITV can impact competitive interactions, community composition (Violle et al., 2012), and ecosystem functions, such as productivity (Ma et al., 2022). Moreover, ITV manifests across different biologic organizations, from organs and individuals to ecosystems (de Bello et al., 2021). At the individual level, the relative extent of ITV tended to be greater for whole-plant traits (e.g., plant height) vs. organ-level traits (e.g., leaf area, leaf economics spectrum; Siefert et al., 2015).
There are two major complementary mechanisms driving ITV: (1) genetic variability and (2) phenotypic plasticity, which reflects each genotype’s potential to produce different phenotypes in response to environmental conditions (Garnier et al., 2016). Many studies have demonstrated high phenotypic plasticity in macrophytes (Wells & Pigliucci, 2000; Santamaria 2002) and ITV (Fu et al. 2013), differentiation of growth forms with wide variability in leaf size, leaf economics spectrum (LES), and shape (Willby et al. 2000; Pierce et al., 2012; Zhang et al., 2017; Chmara et al., 2021). Consequently, macrophyte species often exhibit higher specific leaf area (SLA) and relative growth rates (Poorter et al., 2009; Fu et al., 2014; Liu et al., 2021). Heterophylly, a form of phenotypic plasticity in plants (Wells & Pigliucci, 2000), is particularly common in aquatic and amphibious plants (Li et al., 2019). Amphibious plants possess the unique ability to produce vastly different leaf types on the same shoot (Koga et al., 2020; van Veen & Sasidharan, 2021). The water surface acts as a distinct boundary between aerial and submerged conditions, necessitating different leaf morphology based on function (Wanke, 2011; Li et al., 2019). Therefore, heterophyllous plants serve as excellent models for studying ITV because they produce two or more leaf types on the same shoot, each adapting to different environments. For example, submerged leaves typically tend to be thinner, narrower, lack a cuticle, and may exhibit dissection, whereas aerial leaves are thicker, cutinized, feature stomata, and have been morphologic and anatomic similarities to terrestrial plants (Sculthorpe, 1967; Wells & Pigliucci, 2000). However, the extent of ITV in heterophyllous plants remains largely unexplored.
In this study, our focus was on Luronium natans (L.) Raf., a known heterophyllous species (Hyldgaard & Brix, 2011; Chmara et al., 2019). The present study includes data measured on six leaf traits from different shoots of L. natans in relatively homogeneous softwater lakes. We hypothesized that heterophyllous amphibious plants represent a unique case of ITV in leaves, with ITV varying across different shoot types. Our specific questions were as follows: (1) Do traits and the extent of ITV vary within different shoot types? (2) What are the trait–trait relationships within leaves in different shoot types? (3) How do environmental conditions influence leaf trait variation at a local scale?
Materials and methods
Study sites and field sampling
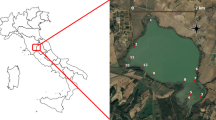
The study took place in the Pomeranian Lakeland (54° 07′ 42″ N, 17° 36′ 08″ E), situated on the outwash plain of the Brda River in northwestern Poland. This region represents a young glacial landscape, with the southern portion falling within the Tuchola Forest UNESCO-MAB Biosphere Reserve (53° 46′ 12″ N, 18° 01′ 05″ E; central point). Currently, the study area is characterized by a high concentration of lakes and peatlands, predominantly covered by Pinus sylvestris forests. The Chojnice weather station reports a mean annual air temperature of 7.4 °C and mean annual precipitation of 580 mm, based on data from 1952 to 2019 (Polish Institute of Meteorology and Water Management—National Research Institute).
The study focused on three relatively homogeneous softwater lakes where L. natans was observed (Fig. 1). These lakes represent a narrow range of softwater habitats, with water pH (5.68–6.19) and calcium concentrations (1.04–4.39 mg L−1). Situated at elevations between 124 and 153 m a.s.l., these shallow lakes were sampled during vegetation seasons in June and August from 2020 to 2021 by SCUBA divers. To investigate trait variation in L. natans, individuals were collected from two depth zones in each lake: 0–1 and 1–2.5 m. Field measurements (from a boat) included (1) visibility (m; Secchi disk) and (2) photosynthetic active radiation (PAR), expressed as a percentage of light reaching the water surface, measured with a Licor LI-250 Light Meter. A total of 25 water samples were collected from different depth zones in the lakes, alongside additional water samples from near-sediment areas, each containing 500 ml, for physicochemical analyses. Physicochemical properties of the water samples were assessed as follows: (1) pH using a pH meter 320/SET1 with a SENTIX 97 T measuring electrode; (2) conductivity; (3) calcium concentration (Ca2+, determined through complexometric titration with calconcarboxylic acid as an indicator); (4) water color assessed comparatively using the Platinum–Cobalt Reference Standards; (5) concentration of humic acids measured spectrophotometrically at 330 nm using a UV–VIS Aquamate spectrophotometer; and (6) total nitrogen (TN) and total phosphorus (TP) analyzed using Merck Spectroquant Cuvette Tests.
Species
L. natans (Alismataceae) is an endemic plant species in Europe, primarily found in the Atlantic and subAtlantic climatic zones in the western and northwestern parts of Europe (Lansdown, 2011). It is included in Annex II of the Europe Habitats Directive 92/43/EEC (European Commission, 1992), which highlights its conservation significance. L. natans is a perennial and evergreen plant that reproduces through both vegetative and generative means. It comprises unevenly aged ramets interconnected by stolons, with new shoots emerging from pseudostolons (Nielsen et al., 2006). The species thrives in aquatic and amphibious environments, exhibiting two distinct growth forms: a fully submerged isoetid form as a bottom-dwelling plant with a rosette and a nymphaeid form with long-petioled oval floating leaves (Fig. 2; Greulich et al., 2001). L. natans can be found in various habitat types, ranging from oligotrophic lakes to eutrophic lakes, canals (Willby & Eaton, 1993; Szańkowski & Kłosowski, 2001; Nielsen et al., 2006), and even dystrophic lakes (Lansdown, 2011).
Leaf traits measurements
Healthy leaves were specifically collected from different shoots of L. natans. The leaves were categorized into three types based on their position on the shoot: heterophyllous shoots with floating leaves (HF, n = 45), heterophyllous shoots with submerged leaves (HS, n = 31), and submerged bottom-dwelling plants with rosette linear leaves (S, n = 134), totaling 210 leaves (Supplementary Information S1). The individuals with floating leaves and submerged leaves with a rosette, belonging to the same shoot (HF and HS types), were collected from the 0 to 1.0 m depth zone, while submerged individuals with a rosette (S type) were collected from the 1.0 to 2.5 m depth zone.
Six leaf traits were measured, including leaf area (LA), leaf dry weight (LDW), specific leaf area (SLA), leaf dry mass content (LDMC), and shape traits: circularity (Circ) [calculated as 4π (area × perimeter−2)] and leaf width/length ratio (w/l; Table 1). The measurement methods followed the standardized protocols of Pérez-Harguindeguy et al. (2013). Each leaf was weighted and scanned while fresh using a balance and ImageJ software, respectively (http://imagej.nih.gov/ij), to determine LA and shape traits (Circ, w/l). Subsequently, the same leaves were dried at 80 °C until a constant weight was achieved and their final dry mass was measured. SLA was calculated as the LA (mm2) per unit of leaf dry mass (mg), which is a component of the leaf economics spectrum representing the trade-off between rapid resource acquisition and conservation (Wright et al., 2004). LDMC is the ratio of dry weight to fresh weight (g).
Data analysis
We conducted basic statistical analyses to assess the differences and extent of ITV within shoot types for various traits, including LA, LDW, SLA, LDMC, Circ, and w/l. To quantify the extent of ITV, we calculated the coefficient of variation (CV) using the formula: CV = traits (SD)/traits (mean) × 100%, where SD represents the standard deviation. The nonparametric Kruskal–Wallis test, followed by Dunn’s multiple comparisons post hoc test, was employed to compare leaf trait values among different leaf types. Spearman rank correlations were utilized to determine trait–trait relationships and trait–environmental variable correlations, and correlation heat maps were generated using OriginPro software (OriginLab Corporation, USA). Kernel density plots were used to visualize the overlap of traits within shoot types, focusing on LDMC, LA, and SLA distributions. To analyze leaf trait variation in the environmental space of sites (lakes), we conducted nonmetric multidimensional scaling (nMDS) separately for four leaf traits and water properties, as well as for shape leaf traits (circularity and w/l ratio) and water properties. Furthermore, an analysis of similarities (ANOSIM) test was performed using Bray–Curtis distance.
For comparing the functional traits of the investigated plant, we utilized R 4.2.1 software (R Core Team, 2022) and employed ggstatsplot to create violin plots with statistical descriptions (Patil, 2021). Additionally, density plots were also generated and visualized using the ggpubr package (Kassambara, 2023). Finally, for the nMDS analysis and ANOSIM test, we utilized the vegan package (Oksanen et al., 2019).
Results
Trait differences and extent of ITV
The ITV values for leaf traits were notably high, ranging from 47.3% for LDMC to 56.6% for LDW (Supplementary Information S2). For LA, LDW, SLA, and LDMC, the ranges were 10.0–333.7 mm2, 0.2–7.0 mg, 7.3–176.8 mm2 mg−1, and 0.8–46.6%, respectively (Supplementary Information S2). Kruskal–Wallis tests revealed significant differences in LA and LDMC within the leaf types (Fig. 3a, d; χ2 = 30.3, P < 0.001 and χ2 = 29.9, P < 0.001). The values of LA and LDMC indicate that floating leaves (HF) are smaller and possess higher LDMC compared to submerged leaves (S). Additionally, there were differences in SLAs between submerged leaves and other shoot types (Fig. 3c; P < 0.001). However, there were no differences in SLA between leaves on the same heterophyllous shoot (HF vs. HS). LDW within the leaf-type groups did not exhibit significant differences (Fig. 3b; P = 0.08).
Functional leaf traits related to shoot types: HF heterophyllous shoot with floating leaves (n = 45), HS heterophyllous shoot with submerged leaves (n = 31), S submerged rosette with linear leaves (n = 134). Differences between leaf traits: a LA leaf area, b LDW leaf dry weight, c SLA specific leaf area, and d LDMC leaf dry mass content. Different letters indicate significant differences (Dunn’s post hoc test of multiple comparisons of independent P < 0.01). The violin plot shows the estimated density of sample distributions with boxes showing interquartile ranges and the red dot indicating the median
The analysis of shape traits revealed high ITV and significant differences in circularity and the ratio of leaf width to length (Fig. 4a, b; χ2 = 91.2, P < 0.001 and χ2 = 93.5, P < 0.001). In general, submerged leaves from individuals with rosettes and heterophyllous shoots were narrower compared to floating leaves. We observed high intraspecific variation in shape traits within shoot types, with CV values of 137.5% for circularity and 144.5% for the leaf w/l ratio (Supplementary Information S2). Specifically, circularity values exhibited a wide range, representing the full spectrum of leaf shapes, ranging from extremely narrow leaves to perfectly circular (0.01–1.0; Supplementary Information S2). Overall, the extent of ITV for shape traits varied more strongly than for functional traits.
Comparisons of shape traits: a circularity and b ratio of leaf to width length between shoot types. For abbreviations, see Fig. 3
Trait–trait relationship and trait overlap
When comparing traits within shoot types, we identified several trait–trait relationships. The SLA values in HF type and S type were negatively related to LDMC (r = − 0.63, r = − 0.41; Fig. S1a, c, Supplementary Materials 1). LDMC was negatively correlated with circularity in the HS shoot type and not correlated with SLA (r = − 0.44, r = − 0.02; Supplementary Information S2). However, SLA for all leaf types was not correlated with leaf shape traits (Circ, w/l). LA showed a negative correlation with circularity in HF type and S type (Fig. S1a, c, Supplementary Information S2). Trait–trait relationships conducted separately for each site confirmed correlation patterns between SLA and LDMC (negatively significantly correlated; Supplementary Information S2) and SLA not significantly correlated with shape traits (Circ and w/l).
We observed a relatively low trait overlap of LDMC and SLA (Fig. 5a, c) and a relatively high overlap of LA within shoot types (Fig. 5b).
Environmental effects on the leaf traits variation
The environmental conditions of the studied sites (lakes) are available in the supplementary materials (S2). These sites exhibited limited diversity in terms of environmental variables: water pH ranged narrowly from 5.68 to 6.19, TN ranged from 0.669 to 2.267 mg N dm−3 water color ranged from 5 to 70, humic acid concentration ranged from 0.50 to 10.25 mg dm−3, and PAR ranged from 23.3% to 59.9%. In general, there was minimal differentiation in environmental variables among the studied lakes. The ANOSIM analysis indicated that environmental conditions in the lakes were not significantly different (ANOSIM statistic: R = 0.12, P = 0.0001). Additionally, environmental conditions in the study lakes explained little of the leaf trait variation within shoot types (ANOSIM statistic: R = 0.14, P = 0.0001).
The nMDS analysis was performed to assess how leaf traits varied within different shoot types, exhibiting distinct separation of the three shoot type groups in ordination space (Fig. 6a; ANOSIM statistic: R = 0.37, P = 0.0001). Another analysis, which was conducted for shape leaf traits showed a clear distinction between shoot types (Fig. 6b; ANOSIM statistic: R = 0.74, P = 0.0001).
Nonmetric multidimensional scaling (nMDS) of leaf trait within different shoot types in ordination space. The color circle represents shoot type: HF heterophyllous shoot with floating leaves, HS heterophyllous shoot with submerged leaves, S submerged rosette with linear leaves. The nMDS analysis was performed for a four leaf traits (LA, LDW, SLA, LDMC) and b shape leaf traits (circularity and w/l ratio)
In general, environmental variables had limited effects on leaf trait variation within shoot types. Some correlations could be partially explained by environmental effects. We observed a significant positive correlation for the LA–pH, LA–Calcium and negative correlation for the LA–TP, LA–TN, LA–Color, and LA–HA trait–environmental variable pairs (Supplementary Information S2). SLA was negatively related to pH, TP, TN, Color, HA, and PAR. However, LDMC and Circularity within leaf types were not correlated with environmental variables.
Discussion
Advancements in trait-based methods in recent decades have contributed significantly to our understanding of ITV (Jung et al., 2014; Ma et al., 2022). Our study, utilizing ITV approaches, revealed clear leaf differences and high ITV (Figs. 3, 4; Supplementary Materials 1) in L. natans, affected by contrasting air–water environments. Overall, we found high ITV across all leaf traits, ranging from 47.3% to 144.4% (Supplementary Information S2). A recent study at the intraspecific level reported significantly lower ITV values (only 10–15%) for leaf functional traits compared to our results (Martin et al., 2017; Bloomfield et al., 2018). For example, traits belonging to the leaf economics spectrum exhibited ITV values similar to mean ITV values within community levels (Siefert et al., 2015). This confirms the well-known high phenotypic plasticity and high ITV of macrophytes and at the same time provides insight into the relatively unexplored extent of ITV at the individual level of macrophytes, especially concerning heterophyllous plants. Several studies have indicated that ITV can be equivalent to or similar to trait variation between species (BTV) for leaf mass per area (LMA; Fajardo and Siefert, 2018) and LDMC (Read et al., 2017). A recent study on grassland species revealed higher ITV than BTV (Tautenhahn et al., 2019). However, it has been shown that ITV measurements may be substantially underestimated (Yang et al., 2020). The authors argue that ITV values can be biased for low sample sizes, emphasizing the need for more extensive sampling. According to these studies, samples greater than 20 are recommended for almost all traits and species. Our research based on the leaf traits of L. natans complies with these requirements.
Even higher ITV were found in leaf shape traits, specifically circularity and the w/l ratio (Fig. 4; Table in Supplementary Information S2). Within the shoot types, we noted a high ITV for circularity and the w/l ratio, expressed as coefficients of variation. Different studies show that leaf shape, being a crucial phenotypic trait, can reflect the adaptation of macrophytes to environmental constraints (Pierce et al., 2012; Maberly & Gontero, 2018; Dalla Vecchia et al., 2020; Dalla Vecchia & Bolpagni, 2022), adaptive strategies related to lake eutrophication and the relationship between water depth and leaf shape (Liu et al., 2021), as well as adaptation to carbon acquisition in air–water environments (Chmara et al., 2021; Pan et al., 2023).
Our investigation revealed differences in SLAs between submerged leaves and other leaf types, but no differences in SLA were observed among leaves on the same heterophyllous shoot (Fig. 3b, c). The highest values of specific leaf area were found in a submerged rosette with linear leave groups. Leaves from these groups tend to be thinner and narrower, characteristics adapted to submerged conditions, categorizing them as true aquatic plants (sensu Veen & Sasidharan 2021). Consequently, high-SLA leaves facilitate better gas exchange and inorganic carbon acquisition (Mommer et al., 2005). In contrast, floating leaves are smaller and have lower SLA and higher LDMC (Fig. 3c, d). Aquatic plants have limited access to carbon and experience reduced light levels (Pedersen et al., 2013). The process of inorganic carbon assimilation varies depending on leaf types. Floating leaves may utilize atmospheric carbon dioxide (CO2), while submerged leaves lack stomata, requiring CO2 to diffuse through their leaf surface (Hyldgaard & Brix, 2011). This suggests that different leaf types within the same heterophyllous shoot serve to optimize carbon concentration mechanisms (CCMs) for photosynthesis and confirm the carbon acquisition plasticity of L. natans (Greulich et al., 2001; Hyldgaard & Brix, 2011).
Overall, we found that environmental variables had minimal impact on trait variation. Why were the leaf trait–environmental variable relationships observed in our study so weak? Firstly, the study was conducted in relatively homogeneous lakes with a narrow range of water environmental conditions (refer to Table in Supplementary Information S2). It is possible that factors at the boundary between water and land, particularly water-level fluctuations, play a significant role. According to Banaś et al. (2023), reduced water levels can lead to a significant reduction in the amphibious form of L. natans, which typically thrives in deep-water populations (submerged rosette with linear leaves). This form is particularly common in lakes in northern Poland.
On the other hand, clear trait–trait correlations were found (Supplementary Information S2). The negative and significant correlations observed for the SLA–LDMC within shoot types may indicate a trade-off relationship in resource allocation within these shoot types. These findings align with previous studies conducted at the intraspecific level in homogeneous environments (Gorné et al., 2021). The authors of those studies suggest that there is a high degree of variation–covariation in leaf traits that is not primarily driven by environmental variables.
Conclusion
Our results highlight high ITV observed in the heterophyllous amphibious plant L. natans. Specifically, ITV of functional leaf traits within the same heterophyllous shoot is lower compared to fully submerged leaves with a rosette. These results offer valuable insights into the extent of ITV in aquatic plants and suggest the inclusion of heterophyllous plants in trait-based studies. Furthermore, it is recommended to consider heterophyllous amphibious plants in global leaf economics spectrum research.
On the other hand, fully submerged leaves with a rosette exhibit significantly different traits from leaves on heterophyllous shoots, indicating their adaptation to submergence. These adaptations are likely driven by ITV within shoot types in L. natans, which plays a crucial role in facilitating adaptation to contrasting air–water environments. Moreover, our findings highlight a variation–covariation pattern in leaf traits and indicate minimal influence from water environmental factors.
Data availability
The data that support the findings of this study are available from the corresponding author (RC) upon request.
References
Albert, C. H., W. Thuiller, N. G. Yoccoz, A. Soudant, F. Boucher, P. Saccone & S. Lavorel, 2010. Intraspecific functional variability: extent, structure and sources of variation. Journal of Ecology 98: 604–613. https://doi.org/10.1111/j.1365-2745.2010.01651.x.
Albert, C. H., F. Grassein, F. M. Schurr, G. Vieilledent & C. Violle, 2011. When and how should intraspecific variability be considered in trait-based plant ecology? Perspectives in Plant Ecology, Evolution and Systematics 13: 217–225. https://doi.org/10.1016/j.ppees.2011.04.003.
Banaś, K., R. Ronowski, R. Chmara & J. Szmeja, 2023. Community structure, environmental conditions and anthropogenic pressure on the habitat of the European endemic aquatic plant Luronium natans (L.) Raf. BMC Plant Biology BioMed Central 23: 596. https://doi.org/10.1186/s12870-023-04518-y.
Bloomfield, K. J., L. A. Cernusak, D. Eamus, D. S. Ellsworth, I. C. Prentice, I. J. Wright, M. M. Boer, M. G. Bradford, P. Cale, J. Cleverly, J. J. G. Egerton, B. J. Evans, L. S. Hayes, M. F. Hutchinson, M. J. Liddell, C. Macfarlane, W. S. Meyer, S. M. Prober, H. F. Togashi & O. K. Atkin, 2018. A continental-scale assessment of variability in leaf traits: within species, across sites and between seasons. Functional Ecology 32: 1492–1506. https://doi.org/10.1111/1365-2435.13097.
Chmara, R., J. Szmeja & A. Robionek, 2019. Leaf traits of macrophytes in lakes: Interspecific, plant group and community patterns. Limnologica 77: 125691. https://doi.org/10.1016/j.limno.2019.125691.
Chmara, R., E. Pronin & J. Szmeja, 2021. Functional macrophyte trait variation as a response to the source of inorganic carbon acquisition. PeerJ 9: e12584. https://doi.org/10.7717/peerj.12584.
Clements, F. E., 1936. Nature and structure of the climax. Journal of Ecology 24: 252–284.
Dalla Vecchia, A. & R. Bolpagni, 2022. The importance of being petioled: leaf traits and resource-use strategies in Nuphar lutea. Hydrobiologia 849: 3801–3812. https://doi.org/10.1007/s10750-022-04803-1.
Dalla Vecchia, A., P. Villa & R. Bolpagni, 2020. Functional traits in macrophyte studies: current trends and future research agenda. Aquatic Botany 167: 103290. https://doi.org/10.1016/j.aquabot.2020.103290.
Darwin, C., 1859. On the origin of species by means of natural selection, John Murray, London:
de Bello, F., C. P. Carmona, A. T. Dias, L. Götzenberger, M. Moretti & M. P. Berg, 2021. Handbook of trait-based ecology: from theory to R tools, Cambridge University Press, Cambridge:
European Commission, 1992. Council Directive 92/43/EEC of 21 May 1992 on the conservation of natural habitats and of wild fauna and flora. Offic. Eur. Communities L206: 7–50.
Fajardo, A. & A. Siefert, 2018. Intraspecific trait variation and the leaf economics spectrum across resource gradients and levels of organization. Ecology 99: 1024–1030. https://doi.org/10.1002/ecy.2194.
Fu, H., G. Yuan, J. Zhong, T. Cao, L. Ni & P. Xie, 2013. Environmental and ontogenetic effects on intraspecific trait variation of a macrophyte species across five ecological scales. PLoS One 8: e62794. https://doi.org/10.1371/journal.pone.0062794.
Fu, H., J. Zhong, G. Yuan, L. Ni, P. Xie & T. Cao, 2014. Functional traits composition predict macrophytes community productivity along a water depth gradient in a freshwater lake. Ecology and Evolution 9: 1516–1523. https://doi.org/10.1002/ece3.1022.
Funk, J. L., J. E. Larson, G. M. Ames, B. J. Butterfield, J. Cavender-Bares, J. Firn, D. C. Laughlin, A. E. Sutton-Grier, L. Williams & J. Wright, 2017. Revisiting the Holy Grail: using plant functional traits to understand ecological processes. Biological Reviews 92: 1156–1173. https://doi.org/10.1111/brv.12275.
Garnier, E., M. L. Navas & K. Grigulis, 2016. Plant functional diversity: organism traits, community structure, and ecosystem properties, Oxford University Press, Oxford:
Gleason, H. A., 1926. The individualistic concept of the plant association. Bulletin of the Torrey Botanical Club 53: 7–26.
Gorné, L. D., S. Díaz, V. Minden, Y. Onoda, K. Kramer, C. Muir, S. T. Michaletz, S. Lavorel, J. Sharpe, S. Jansen, M. Slot, E. Chacon & G. Boenisch, 2021. The acquisitive–conservative axis of leaf trait variation emerges even in homogeneous environments. Annals of Botany 129: 709–722. https://doi.org/10.1093/aob/mcaa198.
Greulich, S., M.-H. Barrat-Segretain & G. Bornette, 2001. Basal rosette or floating leaf canopy—an example of plasticity in a rare aquatic macrophyte. Hydrobiologia 448: 53–59. https://doi.org/10.1023/A:1017530102260.
Hyldgaard, B. & H. Brix, 2011. Plasticity in carbon acquisition of the heterophyllous Luronium natans: an endangered freshwater species in Europe. Aquatic Botany 94: 127–133. https://doi.org/10.1016/j.aquabot.2011.01.002.
Jung, V., C. Violle, C. Mondy, L. Hoffmann & S. Muller, 2010. Intraspecific variability and trait-based community assembly. Journal of Ecology 98: 1134–1140. https://doi.org/10.1111/j.1365-2745.2010.01687.x.
Jung, V., C. H. Albert, C. Violle, G. Kunstler, G. Loucougaray & T. Spiegelberger, 2014. Intraspecific trait variability mediates the response of subalpine grassland communities to extreme drought events. Journal of Ecology 102: 45–53. https://doi.org/10.1111/1365-2745.12177.
Kassambara, A., 2023. Package ‘ ggpubr ’ version 0.6.0. R Found Stat Comput.
Koga, H., Y. Doll, K. Hashimoto, K. Toyooka & H. Tsukaya, 2020. Dimorphic leaf development of the aquatic plant Callitriche palustris L. Through differential cell division and expansion. Frontiers in Plant Science 11: 269. https://doi.org/10.3389/fpls.2020.00269.
Lansdown, R.V. 2011. Luronium natans. The IUCN Red List of Threatened Species 2011: e.T162134A5547543. https://doi.org/10.2305/IUCN.UK.2011-1.RLTS.T162134A5547543.en. Accessed on 20 May 2023
Lavorel, S. & E. Garnier, 2002. Predicting changes in community composition and ecosystem functioning from plant traits: revisiting the Holy Grail. Functional Ecology 16: 545–556. https://doi.org/10.1046/j.1365-2435.2002.00664.x.
Li, G., S. Hu, H. Hou & S. Kimura, 2019. Heterophylly: phenotypic plasticity of leaf shape in aquatic and amphibious plants. Plants 8: 1–13.
Liu, H., G. Liu & W. Xing, 2021. Functional traits of submerged macrophytes in eutrophic shallow lakes affect their ecological functions. Science of the Total Environment 760: 143332. https://doi.org/10.1016/j.scitotenv.2020.143332.
Ma, F., Z. Zuo, L. Yang, D. Li, H. Wang, F. Li, S. Fan, C. Liu & D. Yu, 2022. The effect of trait-based diversity on productivity results mainly from intraspecific trait variability in the macrophyte community. Freshwater Biology 67: 1137–1149. https://doi.org/10.1111/fwb.13906.
Maberly, S. C. & B. Gontero, 2018. Trade-offs and synergies in the structural and functional characteristics of leaves photosynthesizing in aquatic environments. In Adams, W., III. & I. Terashima (eds), The leaf: a platform for performing photosynthesis. Advances in photosynthesis and respiration (including bioenergy and related processes). Springer, Cham.
Martin, A. R., B. Rapidel, O. Roupsard, K. van den Meersche, E. de Melo-Virginio-Filho, M. Barrios & M. E. Isaac, 2017. Intraspecific trait variation across multiple scales: the leaf economics spectrum in coffee. Functional Ecology 31(3): 604–612. https://doi.org/10.1111/1365-2435.12790.
Mommer, L., T. L. Pons, M. Wolters-Arts, J. H. Venema & E. J. W. Visser, 2005. Submergence-induced morphological, anatomical and biochemical responses in a terrestrial species affect gas diffusion resistance and photosynthetic performance. Plant Physiology 139: 497–508. https://doi.org/10.1104/pp.105.064725.
Nielsen, U. N., T. Riss & H. Brix, 2006. The importance of vegetative and sexual dispersal of Luronium natans. Aquatic Botany 84: 165–170. https://doi.org/10.1016/j.aquabot.2005.09.002.
Oksanen, J., F. G. Blanchet, M. Friendly, R. Kindt, P. Legendre, D. Mcglinn, P. R. Minchin, R. B. O’hara, G. L. Simpson, P. Solymos, M. Henry, H. Stevens, E. Szoecs, & H. W. Maintainer, 2019. Package “vegan” Title Community Ecology Package. Community ecology package 2: 1–297, https://cran.r-project.org/web/packages/vegan/vegan.pdf.
Pan, Y., J. García-Girón & L. L. Iversen, 2023. Global change and plant-ecosystem functioning in freshwaters. Trends in Plant Science 28: 646–660. https://doi.org/10.1016/j.tplants.2022.12.013.
Patil, I., 2021. Visualizations with statistical details: The “ggstatsplot” approach. J Open Source Softw 6: 3167. https://doi.org/10.21105/joss.03167.
Pedersen, O., T. D. Colmer & K. Sand-Jensen, 2013. Underwater photosynthesis of submerged plants – recent advances and methods. Frontiers in Plant Science 4: 140. https://doi.org/10.3389/fpls.2013.00140.
Pérez-Harguindeguy, N., S. Díaz, E. Garnier, S. Lavorel, H. Poorter, P. Jaureguiberry, M. S. Bret-Harte, W. K. Cornwell, J. M. Craine, D. E. Gurvich, C. Urcelay, E. J. Veneklaas, P. B. Reich, L. Poorter, I. J. Wright, P. Ray, L. Enrico, J. G. Pausas, A. C. de Vos, N. Buchmann, G. Funes, F. Quétier, J. G. Hodgson, K. Thompson, H. D. Morgan, H. ter Steege, M. G. van der Heijden, L. Sack, B. Blonder, P. Poschlod, M. V. Vaieretti, G. Conti, A. C. Staver, S. Aquino & J. H. Cornelissen, 2013. New handbook for standardised measurement of plant functional traits worldwide. Australian Journal of Botany 61: 167–234. https://doi.org/10.1071/BT12225_CO.
Pierce, S., G. Brusa, M. Sartori & B. E. L. Cerabolini, 2012. Combined use of leaf size and economics traits allows direct comparison of hydrophyte and terrestrial herbaceous adaptive strategies. Annals of Botany 109: 1047–1053. https://doi.org/10.1093/aob/mcs021.
Polish Institute of Meteorology and Water Management - National Research Institute. https://danepubliczne.imgw.pl Accessed 15 May 2023
Poorter, H., Ü. Niinemets, L. Poorter, I. J. Wright & R. Villar, 2009. Causes and consequences of variation in leaf mass per area (LMA): a meta-analysis. New Phytologist 182: 565–588. https://doi.org/10.1111/j.1469-8137.2009.02830.x.
R Core Team 2022. A language and environment for statistical computing. R Found Stat Comput Vienna, Austria URL http//wwwR-project.org/
Read, Q. D., J. A. Henning & N. J. Sanders, 2017. Intraspecific variation in traits reduces ability of trait-based models to predict community structure. Journal of Vegetation Science 28: 1070–1081. https://doi.org/10.1111/jvs.12555.
Santamaría, L., 2002. Why are most aquatic plants widely distributed? Dispersal, clonal growth and small-scale heterogeneity in a stressful environment. Acta Oecologica 23: 137–154. https://doi.org/10.1016/S1146-609X(02)01146-3.
Sculthorpe, C. D., 1967. The biology of aquatic vascular plants, Edward Arnold, London:
Siefert, A., C. Violle, L. Chalmandrier, C. H. Albert, A. Taudiere, A. Fajardo, L. W. Aarssen, C. Baraloto, M. B. Carlucci, M. V. Cianciaruso, L. V. de Dantas, F. de Bello, L. D. S. Duarte, C. R. Fonseca, G. T. Freschet, S. Gaucherand, N. Gross, K. Hikosaka, B. Jackson, V. Jung, C. Kamiyama, M. Katabuchi, S. W. Kembel, E. Kichenin, N. J. B. Kraft, A. Lagerström, Y. L. Bagousse-Pinguet, Y. Li, N. Mason, J. Messier, T. Nakashizuka, J. M. Overton, D. A. Peltzer, I. M. Pérez-Ramos, V. D. Pillar, H. C. Prentice, S. Richardson, T. Sasaki, B. S. Schamp, C. Schöb, B. Shipley, M. Sundqvist, M. T. Sykes, M. Vandewalle & D. A. Wardle, 2015. A global meta-analysis of the relative extent of intraspecific trait variation in plant communities. Ecology Letters 18: 1406–1409. https://doi.org/10.1111/ele.12508.
Szańkowski, M. & S. Kłosowski, 2001. Habitat conditions of the phytocoenoses dominated by Luronium natans (L.) Raf. in Poland. Hydrobiologia 455: 213–222. https://doi.org/10.1023/A:1011914607379.
Tautenhahn, S., C. Grun-Wenzel, M. Jung, S. Higgins & C. Romermann, 2019. On the relevance of intraspecific trait variability–a synthesis of 56 dry grassland sites across Europe. Flora 254: 161–172. https://doi.org/10.1016/j.flora.2019.03.002.
van Veen, H. & R. Sasidharan, 2021. Shape shifting by amphibious plants in dynamic hydrological niches. New Phytologist 229: 79–84. https://doi.org/10.1111/nph.16347.
Violle, C., B. J. Enquist, B. J. McGill, L. Jiang, C. H. Albert, C. Hulshof, V. Jung & J. Messier, 2012. The return of the variance: intraspecific variability in community ecology. Trends in Ecology and Evolution 27: 244–252. https://doi.org/10.1016/j.tree.2011.11.014.
Wanke, D., 2011. The ABA-mediated switch between submersed and emersed life-styles in aquatic macrophytes. Journal of Plant Research 124: 467–475. https://doi.org/10.1007/s10265-011-0434-x.
Wells, C. L. & M. Pigliucci, 2000. Adaptive phenotypic plasticity: the case of heterophylly in aquatic plants. Perspectives in Plant Ecology, Evolution and Systematics 3: 1–18. https://doi.org/10.1078/1433-8319-00001.
Westerband, A. C., J. L. Funk & K. E. Barton, 2021. Intraspecific trait variation in plants: a renewed focus on its role in ecological processes. Annals of Botany 127: 397–410. https://doi.org/10.1093/aob/mcab011.
Willby, N. J. & J. W. Eaton, 1993. The distribution, ecology and conservation of Luronium natans (L.) Rafin. in Britain. Journal of Aquatic Plant Management 31: 70–76.
Willby, N. J., V. J. Abernethy & B. O. L. Demars, 2000. Attribute-based classification of European hydrophytes and its relationship to habitat utilization. Freshwater Biology 43: 43–74. https://doi.org/10.1046/j.1365-2427.2000.00523.x.
Wright, I. J., P. B. Reich, M. Westoby, D. D. Ackerly, Z. Baruch, F. Bongers, J. Cavender-Bares, T. Chapin, J. H. C. Cornelissen, M. Diemer, J. Flexas, E. Garnier, P. K. Groom, J. Gulias, K. Hikosaka, B. B. Lamont, T. Lee, W. Lee, C. Lusk, J. J. Midgley, M. L. Navas, U. Niinemets, J. Oleksyn, N. Osada, H. Poorter, P. Poot, L. Prior, V. I. Pyankov, C. Roumet, S. C. Thomas, M. G. Tjoelker, E. J. Veneklaas & R. Villar, 2004. The worldwide leaf economics spectrum. Nature 428: 821–827. https://doi.org/10.1038/nature02403.
Yang, J., J. H. Lu, Y. Chen, E. Yan, J. Hu, X. Wang & G. Shen, 2020. Large underestimation of intraspecific trait variation and its improvements. Frontiers in Plant Science 11: 9. https://doi.org/10.3389/fpls.2020.00053.
Zhang, L., D. Ma, J. Xu, J. Quan, H. Dang, Y. Chai, X. Liu, Y. Guo & M. Yue, 2017. Economic trade-offs of hydrophytes and neighbouring terrestrial herbaceous plants based on plant functional traits. Basic and Applied Ecology 22: 11–19. https://doi.org/10.1016/j.baae.2017.06.004.
Acknowledgements
This study received financial support from the Polish National Science Center under project number 2019/32/C/NZ8/00147. We extend our gratitude to Małgorzata Fus for her assistance in collecting L. natans specimens from lakes. We would like to thank the Translmed Publishing Group (TPG), a proofreading and copyediting company, for their valuable contributions in proofreading and copyediting this manuscript. Permission to study the protected species L. natans was obtained from the Regional Director for Environmental Protection in Gdańsk, Poland, under decision numbers RDOŚ-Gd WZG.6400.92.2020.AB.2 and RDOŚ-Gd-WOC.6205.24.2020.MaK.2.
Funding
This study was financially supported by the Polish National Science Center under project No. 2019/32/C/NZ8/00147.
Author information
Authors and Affiliations
Contributions
RC contributed to the conception and design of the study. RC and EP co-organized the field study and collected the samples in the field. RC collected the plant samples, analyzed the data, and wrote the manuscript. Finally, RC also prepared all samples for functional trait analyses. Data analysis was conducted by RC and EP. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Guest editors: Rossano Bolpagni, Lars Lønsmann Iversen, Mattia Martin Azzella & Andreas Hussner / A Unified Understanding of Macrophyte Ecology and Adaptations: Plant Functional Traits and Trait-based Approaches
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Chmara, R., Pronin, E. Intraspecific leaf trait variation of the heterophyllous amphibious plant Luronium natans: a trait-based study. Hydrobiologia (2024). https://doi.org/10.1007/s10750-024-05605-3
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10750-024-05605-3