Abstract
Rivers of tropical islands formed by volcanic eruptions experience unpredictable and heavy rainfall and are characterised by irregular topography. During the 2019 and 2020 low-water season, we investigated the responses of benthic macroinvertebrate assemblages to variations in local environmental conditions in four rivers located in areas of different rainfall intensity. We found that changes in local environmental conditions associated with lower rainfall intensity reduced abundance and taxonomic richness and modified taxonomic composition but did not impact the balance of functional feeding groups of macroinvertebrate communities amongst streams, except for piercer herbivores. Heterotrophy associated with substrate instability and autotrophy with a more stable substrate co-occurred in semi-arid climate rivers. In contrast, a change from heterotrophy upstream to autotrophy and substrate stability downstream was apparent in the two humid rivers studied. Surrogates of stream ecosystem function revealed a very low ratio of coarse to fine particulate organic matter related to the quasi-absence of shredders in these streams and suggested a low allochthonous input contribution, which could be the result of the 5% decline in forest cover over the two past decades.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Studies that explore the taxonomic and functional responses of aquatic macroinvertebrate communities to environmental variations are numerous in continental temperate (Heino et al., 2004; 2007 in Finland; Bonada et al., 2007 in the Mediterranean Basin) and tropical regions (Tomanova et al., 2007; 2008 in Bolivia; Kohlmann et al., 2021 in Costa-Rica) but are scarce in tropical islands. With regards to the Caribbean archipelago, most studies on aquatic biota consist of species checklists and reveal high regional biological richness and high variability; however, very little is known about the ecology and functioning of Caribbean Island rivers (Cinéas & Dolédec, 2022).
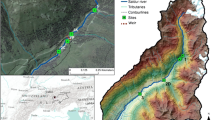
The Caribbean Island of Hispaniola, which originated from volcanic activity, is characterised by rapid changes in elevation over short distances and watersheds that generally consist of 1–4 order streams with short, straight and steep channels and swift currents. The high annual variations in tropical rainfall combined with the steep topography of volcanic islands may greatly influence the in-stream hydraulic conditions (Li et al., 2022 in Puerto Rico), which in turn, may determine the availability of food resources and habitat for benthic macroinvertebrates (Mackey & Currie, 2001; Lepori & Malmqvist, 2007; Nava et al., 2015; Min et al., 2019). Haiti occupies one-third of Hispaniola Island. Seventy-five percent of its territory consists of mountains, of which 52% have slopes steeper than 40%. Air temperature varies between 15 and 35°C year-round, and rainfall ranges between 400 and 4000 mm depending on the relief and exposure (CANARI, 2019). Covered by an important tropical forest and considered a water reservoir for the southern peninsula of Haiti, the Massif de la Hotte was declared a biosphere reserve in March 2016 by UNESCO [Réserve Biosphère de la Hotte—(RBH) green area in Fig. 1].
Map showing the location of the study area in the western part of Haiti within the Caribbean archipelago and selected rivers and sampling sites (RBH frontiers are coloured in dark green). Land cover for the study area was extracted from Pauleus & Aide (2020)
Indeed, RBH has the world's largest concentration of endemic amphibians (CANARI, 2019), whereas the benthic macroinvertebrates of the area remain almost unknown. According to the climate maps (https://agriculture.gouv.ht/statistiques_agricoles/Atlas/thematique_generale.html), the southwestern peninsula of Haiti usually experiences a humid climate along the northern border and a semi-arid climate along the southern border. Therefore, invertebrate sampling was carried out in four of the eight main rivers flowing within the Massif de la Hotte. Two rivers were selected from the northern humid border, the Roseau River (ROS) and the Voldrogue River (VOL) with more intense monthly rainfall and lower interannual variation in rainfall. In addition, two rivers were chosen from the southern semi-arid border experiencing lower monthly rainfall intensity and higher interannual variation (including drying out in some sections), the Acul River (ACU) and the Port-à-Piment River (PAP) (Fig. 1; Supplementary information Fig. S1). Linked to climate differences, the northern humid border was generally more forested than the southern semi-arid border.
According to the Global Forest Watch platform (https://www.globalforestwatch.org), in 2022, forest cover (defined by tree height > 5 m) in the district involving ROS (Corail) and VOL (Jérémie) represented 82% and 74% of the land cover, respectively. In contrast, forest cover in the district involving the ACU (Chardonnières) and the PAP (Jérémie) represented 29% and 41% of the land cover, respectively. Finally, deforestation was included in the study area as elsewhere in Haiti with plantations dominating the northern humid border and pastures being prominent on the southern semi-arid border (Fig. 1).
Benthic macroinvertebrates are known to reflect the ecological status of a stream (Rosenberg & Resh, 1993). Their rather sedentary lifestyles and relatively short life cycle durations (~ 1 year) allow them to respond to short- and long-term disturbances (Park et al., 2008). In addition, macroinvertebrates occupy an intermediate position in aquatic food webs because they mostly feed on basal food resources (litter, aquatic plants, detritus and biofilms) and may serve as prey for fish and amphibians (Wallace & Webster, 1996; Villanueva et al., 2012) or birds (Recalde et al., 2021). Since the 1970s, macroinvertebrates have been grouped into functional feeding groups (FFGs) according to their behavioural food acquisition and food types (Cummins, 1973). FFG distribution and relative abundance, which reflect the food resources available in streams (Allan & Castillo, 2007), allow the indirect assessment of the functional integrity of streams (Cortés-Guzmán et al., 2021). In addition, the proportion of the different FFGs in a community may change in response to hydrological disturbances (Hershey et al., 1988; Hart & Robinson, 1990). For example, in frequently flooded rivers, shredders, which feed on coarse particulate organic matter (CPOM), and scrapers, which feed on periphyton, have been shown to be less abundant than filter feeders, which feed on fine particulate organic matter (FPOM) in the water column (Bhawsar et al., 2015; Min et al., 2019). Finally, FFG ratios have been proposed as surrogates for stream ecosystem attributes mainly focussing on the balance between gross primary production and community respiration and the transfer of coarse and fine particulate organic matter in the water column (Merritt et al., 2002).
The present study aims to compare the abundance and diversity of benthic macroinvertebrate communities inhabiting Haitian rivers, which experience different levels of rainfall intensity, and to provide a functional assessment of the functional integrity of these streams. We hypothesise that in semi-arid climate rivers experiencing lower rainfall intensity, including long periods of drought and unpredictable rainfall, resulting changes in local environmental conditions should (i) induce a decrease in abundance and taxonomic richness and diversity and (ii) modify the taxonomic composition and the resulting balance amongst FFGs, which in turn (iii) should influence ecosystem processes. Finally, we expect (iv) annual differences in biological metrics to be more important in the semi-arid climate rivers associated with higher interannual rainfall variations and longer periods of low flow.
Methods
Climate time series
Rainfall and air temperature time series were extracted from Climate Hazards Group InfraRed Precipitation with Station (CHIRPS) and Climate Hazards Group InfraRed Temperature with Station (CHIRTS), (available at https://iridl.ldeo.columbia.edu) which are global satellite-derived rainfall and air temperature data with a 0.05-degree grid resolution (~ 3 km). Monthly rainfall and air temperature at each sampling site were approximated considering the best available rainfall and air temperature measures collected at the closest meteorological stations, i.e. located ≤ 3 km from each sampling site over the period 2006–2016. These time series did not cover the sampling period (2019–2020; see below). However, we can consider that the range of rainfall intensities in the area was covered by these time series, including torrential rains in 2007–2008 and catastrophic flooding in 2016 (Hurricane Matthew) as well as low-flow periods. No major storm events were recorded in 2017–2019.
Benthic macroinvertebrate assemblages
Benthic macroinvertebrates were sampled in two seasons during the low-flow period. The first sampling campaign (end of February to the beginning of March 2019) occurred later in the low-flow period than the second sampling campaign (beginning January 2020), which was performed after recent rainfall (personal field observations). Sites were selected at different elevations along each river [between 16 and 706 m above sea level (a.s.l.); Table 1] and according to the relative ease of access. Four sites (S1, S2, S3 and S4) were sampled upstream to downstream in the Port-à-Piment River (PAP). The Acul (ACU) and Roseau (ROS) rivers were sampled at three sites (S1, S3, S4), and the Voldrogue River (VOL) was sampled at two sites (S3, S4) (Fig. 1, Table 1). The sampling design was not totally adequate due to the Haitian political context, which included roadblocks and riots occurring during this period, which prevented us from reaching the expected river sites. However, a similar altitudinal gradient was covered in the north and south sides.
A total of 12 samples were collected at each site over a section with a length ten times the width using a Surber net (0.05 m2, 500 µm mesh). Samples were collected by visually considering substrate and current velocity distributions to cover most of the habitat diversity across a given site. A total of 286 samples were collected over the two sampling campaigns (n = 144 and 142 for the first and second campaigns, respectively). Samples were preserved in 95% ethanol until macroinvertebrates were sorted, counted and identified at the family level in the laboratory (in France) using a stereomicroscope and available taxonomic keys (Bernadet et al., 2014; Hamada et al., 2018). Two samples dried out during travel from Haiti to France, and only 11 samples were available for sites S3 and S4 in the ROS for the second sampling campaign. For each sample, we calculated the total number of individuals, the family richness and the Shannon diversity index.
Functional feeding groups
We assigned the macroinvertebrate families to FFGs using the information provided by Ramirez & Guttiérez-Fonseca (2014), and we computed the proportion of individuals belonging to collector gatherers (CG), filtering collectors (Ft), predators (Pr), scrapers (Sc), piercer herbivores (Pc-Hb), shredder detritivores (Sh-Dt) and shredder herbivores (Sh-Hb) in each site and each sampling campaign (see Appendix 1).
Ecosystem functions
We further used FFG ratios as surrogates for three stream ecosystem attributes as proposed by Merritt et al. (2002). The ratio between primary production and respiration (abbreviated PRR) was computed as the ratio between the sum of individuals that were scrapers (Sc), piercers (Pc-Hb) and shredder herbivores (Sh-Hb) and the sum of individuals that were shredder detritivores (Sh-Dt) and collectors (CG, Ft). In-stream substrate stability (SUS) was computed as the ratio of the sum of scrapers (Sc) and filtering collectors (Ft) and the sum of shredder detritivores (Sh-Dt) and collector-gatherers (CG). Finally, the ratio of coarse to fine particulate organic matter (abbreviated CPO) was estimated as the ratio between the number of shredder detritivores (Sh-Dt) and the number of collectors (CG, Ft). We considered threshold values for each ecosystem attribute as follows: PRR˃0.75 demonstrated the prominence of autotrophic processes; SUS > 0.50 corresponded to a more stable substrate; and CPO > 0.50 (dry season) was associated with normal riparian functioning (see Merritt et al., 2002).
Physical and chemical variables
For each invertebrate sample, water temperature, dissolved oxygen, oxygen saturation and conductivity were recorded using a multiparameter probe (HQ40D), and water depth was measured with a ruler. Current velocity was assessed visually using the following categories: very slow (v < 5 cm s−1), slow (5 < v < 25 cm s−1), medium (25 < v < 50 cm s−1), rapid (50 < v < 75 cm s−1) and very rapid (> 75 cm s−1) (Supplementary information Fig. S2). The substrate was also recorded visually, and we used the following categories and their phi values (i.e. log2 of the particle diameter): boulder (Phi = − 8), cobble-pebble (− 6), gravel (− 2), sand (2), silt (1), root and organic debris (3). We further calculated the frequency distribution of the current velocity and substrate categories at each site and for the two sampling campaigns, which allowed us to compute weighted average current velocity (VEL) and phi values (GME) at each site and for both campaigns. Finally, we computed the Simpson index on the proportions of substrate categories to assess the diversity of sediment size (GDI) within a site.
Data analysis
To assess differences in climate across rivers, we compared rainfall and temperature time series at similar altitudes (~ 100 m a.s.l.) using Kruskal–Wallis’s test for global difference followed by Dunn’s test for paired differences between rivers. In addition, since our climate time series covered the period 2006–2016 and our invertebrate sampling was done over 2019–2020, we decomposed the series to address long-term and seasonal trends using classical seasonal decomposition by moving averages and the R stats package.
We performed a normalised principal component analysis (PCA) to derive scores for sites for each sampling campaign based on the physical and chemical parameters measured in the field, averaged by site and sampling campaign (n = 24). We assessed temporal (year) and spatial (site, river) effects on the physical and chemical parameters, abundance, family richness and Shannon diversity using mixed ANOVA with rivers and sampling campaigns as fixed effects and sampling sites as random effects (n = 286). This was followed by Games-Howell post hoc tests to handle unequal variances and sample sizes between groups. When the normality assumption was not met, we used the Box‒Cox transformation (Box & Cox, 1964). We investigated the relationships between each biological metric and physical and chemical parameters using multiple regression models after correcting for multicollinearity amongst the parameters with a variance inflation factor (VIF < 4) (Zuur et al., 2010). Finally, we tested each FFG and ecosystem metric against rivers, sites and sampling campaigns using one-way ANOVA type II (n = 24).
To investigate the biological similarity amongst rivers, sites and sampling campaigns, we performed non-metric multidimensional scaling (NMDS) analysis using log(x + 1) transformation and the Bray‒Curtis distance between macroinvertebrate communities (Minchin, 1987). We tested the effect of rivers, sampling campaigns and the upstream–downstream gradient using permutational multivariate analysis of variance (PERMANOVA) (McArdle & Anderson, 2001). Physical and chemical variables were fitted to the NMDS ordination after correcting for multicollinearity (see above). To that end, we used the envfit() command of the vegan package of R that allowed the estimate of the strengths of the correlation between the NMDS dimensions and each environmental variable. This command provides direction of maximal correlation between the NMDS dimensions and each environmental variable (see Jongman et al., 1987, pp.132–136). For each environmental variable, the goodness-of-fit was assessed by a squared correlation coefficient, and the statistical significance of the explained variance was obtained from a permutation test. These operations allowed us to indicate if one or more environmental variables were associated with differences between sites and rivers as shown in the NMDS ordination.
All statistical analyses were performed with R software version 4.3.2 (R Core Team, 2022) and the vegan (Oksanen et al., 2022), ade4 and adegraphics (Thioulouse et al., 2018) packages. Specific graphics were generated using the R package ggplot2 (Wickham, 2016).
Results
Environmental variables
Rainfall and air temperature variations
Looking at long-term and seasonal trends, monthly rainfall available on comparable sites (~ 100 m a.s.l.) showed that the differences between the four rivers remained rather constant over the period 2006–2016, with the PAP river experiencing higher seasonal variations and longer periods of low flow (Supplementary information Figs. S3A, S4). The distribution of monthly rainfall values differed significantly across rivers (Kruskal–Wallis = 50.0, P < 0.000, n = 528; Fig. 2). Dunn’s test further showed evidence for a lower rainfall in PAP (median = 63.7 mm) compared to the three other rivers [ACU (89.7 mm), P < 0.003; ROS (126 mm), P < 0.000; VOL (125 mm), P < 0.000]. In addition, ACU rainfall significantly differed from ROS (P < 0.007) and only slightly differed from VOL (P < 0.068), whereas no difference was apparent between the ROS and VOL rivers (P > 0.345).
As expected, monthly air temperature values were high and ranged between [26.7–30.7°C] for ACU, [26.1–31.3°C] for PAP, [25.7–30.5°C] for ROS, and [25.6–30.5°C] for VOL (Fig. 2; Supplementary information Figs. S3B, S5). There was evidence for a global statistical difference (Kruskal–Wallis = 8.7, P < 0.033, n = 528). However, neither differences in seasonal variations amongst rivers nor long-term trends of air temperature were apparent (Dunn P > 0.09; Supplementary information Fig. S3B).
Physical and chemical variables
The first axis of a PCA performed on the physical and chemical parameters represented 33.7% of the total variance and separated PAP from ROS (Fig. 3B). This difference was specifically linked to water oxygenation (DOX in Fig. 3A), temperature (MWT), current velocity (VEL) and depth (DEP). The axis also described a gradient of increase in water temperature, conductivity (CON) and oxygenation from upstream to downstream. The second PCA axis (20.6%) described a gradient associated with higher grain size diversity (GDI) upstream than downstream, which was especially apparent in the PAP River.
Results of the normalized PCAs performed on the climate variables and physical and chemical variables shown along the first two axes of each PCA. A Position of physical and chemical variables (CON, conductivity; DEP, water depth; GME, sediment size; GDI, sediment diversity; OSA, oxygen saturation; DOX, dissolved oxygen, MWT, water temperature; VEL, current velocity); and B position of sites along each river associated with changes in physical and chemical variables (VOL, Voldrogue; ROS, Roseau; ACU, Acul; PAP, Port-à-Piment)
The results of the ANOVA mixed models suggested that rivers differed in all parameters (Table 2). Waters were well oxygenated, with few differences between rivers and across years (Table 2). PAP and ACU demonstrated higher water temperature variations than ROS and VOL (Table 2). PAP waters were generally shallower (~ 15 cm; Table 2) than in the three other rivers (> 25 cm). In parallel, ROS demonstrated lower current velocities (~ 0.2 m s−1; Table 2) than the three other rivers (> 0.45 m s−1). Finally, PAP had a smaller sediment size (phi > − 5) than the three other rivers (phi < − 5).
Responses of macroinvertebrate assemblages
Abundance, richness and diversity
A total of 51,126 macroinvertebrates belonging to Bivalvia, Clitellata, Gastropoda, Insecta, Malacostraca and Trepaxonemata and 50 families were collected (Appendix 1). In the first survey, 33,813 individuals and 47 families were collected, whereas 17,313 individuals and 38 families were collected during the second survey. Insects represented 72% of the taxonomic richness (36 families) and had the highest abundance (42,690 individuals, i.e. 83.3% of the total). Ephemeroptera was the most abundant order (35.6%), followed by Trichoptera (32.7%). Finally, Diptera was the richest insect order (11 families), amongst which 10 families were present in the PAP, with 4 families (Culicidae, Dixidae, Psychodidae and Stratiomyidae) found only in the PAP but at very low abundance (between 1 and 4 individuals).
ANOVA mixed models demonstrated stronger evidence for higher abundance and taxonomic richness during the first sampling campaign than during the second sampling campaign in all rivers, whereas Shannon diversity did not show such strong evidence (Fig. 4; Table 3). In addition, there was positive evidence for differences in abundance and taxonomic richness across rivers (Fig. 4; Table 3). Post hoc Games-Howell tests indicated (P < 0.05 for any pair) that the macroinvertebrate abundance was lower in the PAP than in the ACU, ROS and VOL, and the taxonomic richness was higher in the ROS and ACU than in the VOL and PAP (Table 3). In contrast, Shannon diversity did not demonstrate any differences amongst rivers (Table 3). Finally, ANOVA models between each biological metric and site position (from S1 to S4) demonstrated a significant difference between the upper (S1) and lower site (S4) for richness (Games–Howell P < 0.000; median = 9 vs. 7 for S1 and S4, respectively) and Shannon diversity (Games–Howell P < 0.000; median = 1.4 vs. 1.2 for S1 and S4, respectively; and Games–Howell P < 0.015; median = 1.4 vs. 1.2 for S2 and S4, respectively).
Probability density curves of changes in A abundance (log transformed), B family richness and C Shannon diversity in each study site along each river course. The first sampling campaign is represented in light grey, and the second sampling campaign is represented in dark grey (n = 12 per site per sampling campaign except S3 and S4 in the ROS during the represented sampling campaign)
Finally, multiple regression models revealed that Shannon diversity was positively related to sediment size and water temperature in the samples (Table 4). Family richness was positively related to water temperature and oxygen saturation (Table 4). Dissolved oxygen, water depth and sediment size showed a positive correlation with macroinvertebrate abundance (Table 4).
Taxonomic composition
NMDS provided a good representation of sites in two dimensions (Stress = 0.092; Fig. 5A). PERMANOVA revealed that 32.3% of the total variance of the distance matrix was associated with differences in taxonomic composition amongst rivers (P < 0.001), 21.4% amongst sites (P < 0.001), and 6.7% between sampling campaigns (P < 0.007). More specifically, the first NMDS dimension (NMDS1 in Fig. 5A) demonstrated differences between rivers (ANOVA, F3,24 = 17.4, P < 0.000), sampling campaigns (F1,24 = 10.2, P < 0.008) and sites (LRT = 7.1, P < 0.008). Along NMDS1, PAP communities were well separated from those in the ROS, as well as between sampling campaigns (Fig. 5A). The second NMDS dimension (NMDS2) demonstrated no significant difference in composition amongst rivers, whereas there was evidence for differences between sampling campaigns (F1,24 = 15.5, P < 0.002) and along a gradient from upstream (S1) to downstream (S4; LRT = 14.0, P < 0.000). Rheophilic taxa, i.e. adapted to resist flow constraints (Psychodidae, Hydrobiosidae, Simuliidae) were separated from taxa generally found in more lentic habitats (Lymnaeidae, Glossiphoniidae, Sphaeriidae) along NMDS1 (Fig. 5B), a feature that was also expressed along NMDS2 (Fig. 5B). NMDS2 separated the molluscs (Lymnaeidae, Sphaeriidae, Physidae, Planorbidae, Cochliopidae, Neritidae, Thiaridae; Fig. 5B) that were more abundant and diversified downstream than upstream.
First two dimensions of a NMDS analysis performed on the benthos data collected in the two sampling campaigns. A Position of sites separated by river (VOL, Voldrogue; ROS, Roseau; ACU, Acul; PAP, Port-à-Piment). B Position of families (grey dots represent families that show little evidence for discriminating sites at P > 0.1, whereas black dots represent families that discriminate sites (P < 0.1)) (Baet, Baetidae; Caen, Caenidae; Cera, Ceratopogonidae; Chir, Chironomidae; Coch, Cochliopidae; Cord, Corduliidae; Corixid, Corixidae; Doli, Dolichopodidae; Duge, Dugesiidae; Empi, Empididae; Gloss, Glossiphoniidae; Heli, Helicopsychidae; Hbio, Hydrobiosidae; Hphi, Hydrophilidae; Hpti, Hydroptilidae; Lcer, Leptoceridae; Lhyp, Leptohyphidae; Libe, Libellulidae; Lymn, Lymnaeidae; Neri, Neritidae; Phys, Physidae; Plan, Planorbidae; Psep, Psephenidae; Psyc, Psychodidae; Simu, Simuliidae; Spha, Sphaeriidae; Thia, Thiaridae). C Position of environmental variables [CON, conductivity; DEP, water depth; GME, sediment size; GDI, sediment diversity; DOX, oxygen saturation; MWT, mean water temperature; VEL, current velocity. Dashed arrows stand for parameters that show no evidence for site discrimination (P > 0.05)]
Projecting environmental variables along the first two NMDS dimensions showed that differences in taxonomic composition between PAP and ROS along NMDS1 were related to a higher water depth (DEP) and water oxygenation (DOX) as well as lower current velocity (VEL) of the latter (Fig. 5; Table 5). Differences in taxonomic composition between sites depicted along NMDS2 were mainly related to water temperature (MWT), which was expectedly higher downstream, and sediment diversity (GDI), which was higher upstream (Fig. 5; Table 5).
Functional feeding groups
Overall, the bulk of the community included collector gatherers [mean proportion = 0.56 (0.21–0.92) of the total communities in all rivers for the two sampling campaigns], scrapers [0.33 (0.00–0.78)], piercers [0.06 (0.00–0.43)] filter feeders [0.03 (0.00–0.09)], predators [0.02 (0.00–0.09)], shredder herbivores [0.00 (0.00–0.03)] and shredder detritivores [0.00 (0.00–0.02)], which were much less represented.
Collector gatherers, scrapers and filtering collectors showed no differences across rivers, sites, or sampling campaigns (Fig. 6; Table 6). Piercers were especially prominent in the PAP River (Fig. 6; Table 6). They dominated upstream (S1, in Fig. 6), which was confirmed by the positive relationship between their proportions and the altitude of the sites (r = 0.65, P < 0.001). In addition, they were caught in higher proportions during the first sampling campaign (Fig. 6; Table 6). Finally, predators were generally more prominent upstream (S1, S2 in Fig. 6; Table 6), which was confirmed by the positive relationship between their proportions in sites and altitude (r = 0.69, P < 0.001).
Proportions of individuals belonging to a given FFG (see acronyms in Appendix 1) in each river (ACU, Acul; PAP, Port-à-Piment; ROS, Roseau; VOL, Voldrogue), each site and each sampling campaign (for each river, the first sampling campaign corresponds to the first column of pies and the second sampling campaign to the second column of pies)
Ecosystem functions
Overall, values of PRR or SUS showed no clear differences between the northern and the southern rivers (Fig. 7). In addition, there was a high variability between sampling campaigns for some sites (S1 in the ACU, S3 in the PAP, S3 and S4 in the ROS, S3 in the VOL; Fig. 7A and Fig. 7B) and along the course of the rivers. On average, the values of PRR in the ACU (1.22 ± 0.60), ROS (1.06 ± 1.49) and VOL (1.50 ± 1.21) were generally > 0.75, whereas on average, the values in the PAP were < 0.75 (0.52 ± 0.51). In addition, the average values of substrate stability in the ACU (1.46 ± 0.80), ROS (1.04 ± 1.46) and VOL (1.52 ± 1.22) were > 0.5, suggesting greater stability of the substrate, whereas the values in the PAP were < 0.5 (0.33 ± 0.50). Autotrophy and substrate stability tended to increase downstream in the ROS and VOL (Fig. 7A, B). However, ANOVA showed no strong differences in the above processes between rivers, sites, or sampling campaigns. Finally, CPO had very low values (Fig. 7C).
Discussion
Our hypotheses were partly supported since the environmental conditions prevailing in the mid-arid climate river with less stable and low rainfall intensity (i) induced the expected decrease in abundance and taxonomic richness but not in diversity and (ii) expectedly modified the taxonomic composition but not the resulting balance amongst FFGs except for piercers. In contrast, the ecosystem processes as measured in our study did not depend on the exposure to the rainfall intensity as expected (iii). Finally, (iv) annual differences in composition, abundance and richness between sampling campaigns were expectedly associated with the less stable mid-arid climate river.
Abundance and taxonomic richness
Comparative analyses of biological metrics showed that the more stable river experiencing the highest rainfall intensity (ROS) had a higher abundance and taxonomic richness than the less stable river with the lowest rainfall intensity (PAP). Several factors explain these differences. First, such variations can be related to the precipitation regime and resulting flow, as observed in previous studies (Jones et al., 2012; Ferreira et al., 2014; Kohlmann et al., 2021). The lower macroinvertebrate richness and abundance observed in the PAP river with high current velocity contrasts with the result of a few tropical river studies showing a higher macroinvertebrate richness and abundance in higher current velocity conditions (Nelson & Lieberman, 2002; Boyero & Bosch, 2004; Szczerkowska-Majchrzak & Grzybkowska, 2015). The range of velocity in the PAP probably produces more severe hydraulic conditions, which result in the selection of specific taxa. Nevertheless, shortcomings arise from our current velocity measurements that were only rough estimates of the true current velocities in the water column, thus limiting the strength of our results.
Second, in comparison to pebbles and cobbles, gravel sediment observed in the PAP tends to limit periphyton growth, a main food resource for many macroinvertebrates, and provides fewer refugia to effectively protect organisms against hydrological disturbances (Rezende et al., 2014; Wolmarans et al., 2017).
Third, according to the geology of Haiti, the bedrock of the PAP is composed of crystalline basalts, mainly downstream, whilst the ROS flows throughout its course on limestone (Robart, 1984). The dissolution of crystalline basalts, which mainly releases Fe++, is rather unfavourable to macroinvertebrates (Rasmussen & Lindegaard, 1988; Gudbrandsson et al., 2011). In contrast, the dissolution of limestone, which releases mainly Ca++, may explain the higher abundance and richness of molluscs observed in the ROS compared to the PAP, where only a few individual Thiaridae were found (Skeldon et al., 2007).
The other river under higher rainfall intensity (VOL) had a lower abundance and richness than the ROS, which can be attributed to the low altitude of the sampled sites (< 110 m, Table 1). Indeed, we found that on average sites upstream (S1) had slightly higher richness and diversity than sites downstream suggesting an effect of altitude, which is consistent with observations made elsewhere in insular streams (e.g. Leyte Island: Philippine archipelago; see also Forio et al., 2017). This positive relationship in tropical insular rivers contrasts with the negative relationships between altitude and taxonomic richness generally observed in tropical mainland rivers in Ecuador (Monaghan et al., 2000; Jacobsen, 2003) and Brazil (Feio et al., 2015; Rezende et al., 2014). In fact, the sites considered downstream (400 to 780 m a.s.l) in the Ecuador and Brazil studies approximately correspond to our upstream sites. In addition, the colonisation of insular taxa at high altitude may compensate for the small size of insular rivers, since they are generally of 1st to 4th order, as opposed to continental rivers, which generally cover higher orders ≥ 5th (Smith et al., 2003). Unfortunately, the difficult field conditions in Haiti did not allow us to sample sites at higher altitudes in every river to strongly confirm or refute the observed positive relationship between family richness and altitude in Haitian mountain rivers.
Taxonomic composition
Differences in taxonomic composition were apparent amongst rivers and sites in relation to local environmental conditions (water depth, oxygenation, current velocity, water temperature and substrate diversity). The PAP communities tend to be dominated by rheophilic families (e.g. Psychodidae, Hydrobiosidae), which reflect the higher lotic constraints in this river, as observed elsewhere (Tomanova & Usseglio-Polatera, 2007). For instance, Psychodidae are known to adapt to fast-flowing and shallow rivers (Hamada et al., 2018). Atopsyche, the only genus of Hydrobiosidae identified to date in Hispaniola (Cinéas & Dolédec, 2022), was found to inhabit small and fast-flowing streams in Ecuador (Vasquez et al., 2020). In contrast, some families found only in the PAP had morphological traits characterising organisms living in poorly oxygenated lentic areas. For instance, posterior spiracles (e.g. Dixidae, Psychodidae and Stratiomyidae) or siphons (e.g. Culicidae) allow them to directly breathe atmospheric air (Tomanova & Usseglio-Polatera, 2007; Hamada et al., 2018). Although only a few specimens were found, the presence of these families only in the PAP suggests highly disturbed flow conditions, including drought.
In contrast, Simuliidae were present in the rivers under low (PAP) and high (ROS) rainfall intensities. Overall, Simuliidae prefer fast-flowing streams but can be opportunistic in small channels (Vasquez et al., 2020). For instance, Figueiró et al. (2008) demonstrated that four species of Simuliidae in Brazil had distinct current velocity preferences (from slow to fast), suggesting that the diversity of Simuliidae depends on the current velocity range. Finally, our results agree with those of Grillet & Barrera (1997), who concluded that the distribution of neotropical Simuliidae larvae depended on the stream size, hydraulics and precipitation regime.
The ROS River, characterised by a higher water depth and a lower current velocity, hosted a high diversity and abundance of molluscs. On Leyte Island (Philippines), Forio et al. (2017) found that mollusc families (including Neritidae and Physidae found in the ROS) preferred low current velocity and high-water depth. Apart from molluscs, other families known for their low current velocity preferences, such as Corixidae, Hydrophilidae, Ceratopogonidae (Borkent & Spinelli, 2007), Leptoceridae (Gutiérrez et al., 2002), Caenidae (Tripole et al., 2008 in Argentina; Firmiano et al., 2017 in Brazil), Chironomidae (Serra et al., 2016) and Glossiphoniidae (Kazanci et al., 2015) dominated the ROS communities.
Functional feeding groups
The dominance of collector gatherers in Haitian rivers is consistent with previous studies in subtropical rivers (Sun et al., 2023 in China), neotropical rivers (Ramirez & Pringle, 1998 in Costa-Rica; Tomanova et al., 2006 in Bolivia) and tropical rivers (Addo-Bediako, 2021 in Kenya). Their prominence in all rivers, sites, and across the two sampling campaigns indicates the abundance of organic detritus, resulting from a rapid and continuous litter breakdown due to a constant and elevated water temperature (> 25°C) (Dobson et al., 2003; Tomanova et al., 2006). In addition, collector gatherers are known to be generalist feeders that are tolerant to hydraulic disturbances (Park et al., 2008; Bhawsar et al., 2015) and can cope with the frequent tropical storms occurring in the area.
Contrastingly, filtering collectors were found in low proportions in all rivers, which reflects a low amount of FPOM in the water column (Merritt et al., 2002), despite the thermal conditions of the Haitian rivers that are favourable to rapid litter degradation and production of FPOM in the water column. Our results suggest that the high hydraulic constraints associated with steep slopes and high hydraulic disturbances caused by frequent storms may yield high sediment suspension, likely to damage the pre-mandibular fans of Simuliidae or silk nets of Hydropsychidae, thus limiting the prevalence of filtering collectors. Studies that have observed a high proportion of filtering collectors in tropical environments (Oliveira & Nessimian, 2010 in Brazil; Addo-Bediako, 2021 in Kenya) were carried out on continental rivers with much less steep slopes than island rivers. One exception concerns the work of Tomanova et al. (2006) in Bolivia, with which our results on filter feeders probably agree because all their sites were characterised by steep slopes.
The low proportion of shredders has three possible causes. First, the frequent torrential rains occurring in the study area may affect shredders known to be sensitive to hydraulic disturbances (Bhawsar et al., 2015). Second, most studies carried out in tropical rivers have shown that shredders were scarce or even absent due to the non-palatability of tropical tree leaves richer in secondary compounds used as a defence against herbivores (Li & Dudgeon, 2009). Tree leaves on islands can exhibit even higher tannin concentrations than their mainland counterparts, which can exacerbate the scarcity of shredders (Moreira et al., 2019). Third, the low forest cover, even in reserves (Dolisca et al., 2007), showing a possible decline by 5% over the period 2000–2015 (Pauleus & Aide, 2020) may also limit the development of shredders (Winterbourn et al., 1981). Unfortunately, the patchy nature of Haitian deforestation makes precise forest cover estimates difficult, which vary between 7% and 32.3% of the Haitian land cover according to Churches et al. (2014).
Scrapers, which mainly feed on periphyton and diatoms attached to stable substrates (Bhawsar et al., 2015; Cummins, 2019), demonstrated high proportions in all rivers, suggesting significant levels of biofilm and primary production in Haitian rivers. However, their high proportions do not conform to the potential effects of hydraulic disturbances that generally scour substrates and depress biofilm and primary production (Barbour et al., 1996; Uehlinger & Naegeli, 1998). Our sampling campaigns took place during the low-flow season, and the significant proportions of scrapers in our samples suggest rapid periphyton recovery after flow disturbances, as observed in temperate streams (Steinman & McIntire, 1990; Calapez et al., 2014).
Piercers (mainly Hydroptilidae) that feed on algal cell fluids were especially prominent in the PAP upstream site, suggesting a higher development of algae during low-flow periods in this frequently disturbed river. These local conditions probably facilitate the development of Hydroptilidae, characterised by a short emergence period and preferring smaller water bodies (Hering et al., 2009; Houghton, 2012). However, this prominence can also be related to forest canopy openness resulting from deforestation, which in turn favour the development of periphyton biomass (Majdji et al., 2015).
Finally, the upstream increase in the proportion of predators opposes the River Continuum Concept (RCC) expectations of uniform distribution (Vannote et al., 1980). This is probably a response to the higher diversity and abundance of their prey upstream, a phenomenon also observed in temperate studies (e.g. Wallace et al., 2015).
In summary, on the one hand, the significant proportion of collector gatherers and scrapers in the four rivers may be associated with the constant elevated water temperature and solar incidences, which accelerate litter breakdown and primary development, promoting rapid renewal of basal resources during low-flow periods. On the other hand, high and unpredictable rainfall, a characteristic of tropical areas exacerbated by steep slopes, modifies the food quality and quantity of organic matter (Power et al., 1995), affecting the dietary flexibility of organisms (Tamaris-Turizo et al., 2020). Finally, whereas Tomanova et al. (2007) have shown that neotropical streams conformed to the RCC, Haitian rivers generally deviate from RCC expectations, especially by their lower-than-expected proportions of shredders, as indicated by Statzner & Higler (1985).
Ecosystem functions
Ecosystem function surrogates demonstrated very low CPOM/FPOM ratios, suggesting low allochthonous contributions with scarce vegetation and poor riparian areas (Wantzen & Wagner, 2006) potentially associated with deforestation (Pellek, 1990). In addition, the ratio between primary production and respiration showed that three of the four rivers (ACU, ROS, VOL) were dominated by autotrophic processes, suggesting primary production as the dominant base of the food web for Haitian invertebrate communities. In addition, the apparent increase of autotrophy downstream humid rivers (ROS, VOL) corresponds to the mechanisms advocated by the RCC for temperate rivers (Vannote et al., 1980). In contrast, the prominence of heterotrophic processes and substrate instability in the PAP river, which is under lower rainfall intensity, and with fine sediment fractions on the river bottom, agree with patterns observed in rivers elsewhere on continents (Uehlinger et al., 2002; Bernhardt et al., 2018; Cargill et al., 2021).
Temporal effect of sampling campaigns
For some metrics, such as abundance, taxonomic richness and FFG ratio, we observed significant differences between sampling campaigns, which can be attributed to the rainfall that preceded the second campaign (personal observation). The main families whose abundance decreased dramatically during the second campaign have traits conferring them to have a low ability to resist high flows (e.g. Caenidae, Coenagrionidae, Helicopsychidae, Hydroptilidae, Physidae, Planorbidae, Thiaridae). This result suggests that by increasing the flow and shear stress, the rains immediately preceding our second sampling campaign induced a probable drifting of these lentic families, as found by others in temperate rivers (Chanut et al., 2019).
Outcome
This study is the first to analyse the responses of aquatic macroinvertebrates in tropical island rivers to climate, hydraulic and habitat variability, which contributes to understanding how climate change can impact biological diversity and functioning (FFG balance) in these ecosystems. Our results on ecosystem function partly demonstrate the deleterious effect of deforestation even inside a nature reserve. However, they must be taken with some caution since the families were assigned to FFGs using neotropical literature, which does not specifically consider that potential shift in the diet of these taxa on the island. In addition, Ramirez & Guttiérez-Fonseca’s (2014) data, which we used to document FFGs for our island taxa, assigned the functional role of some neotropical taxa using their temperate counterparts (e.g. aquatic insects from North America), whereas there is evidence that related species occurring in different regions do not share the same diets (Cheshire et al., 2005; Chará-Serna et al., 2012). Therefore, future in-depth studies on FFGs should consider mouthpart morphology and gut contents to assign Haitian invertebrate families to proper FFGs and assess their level of dietary flexibility.
Data availability
The data that support the findings of this study are available from the authors upon request.
References
Addo-Bediako, A., 2021. Spatial distribution patterns of benthic macroinvertebrate functional feeding groups in two rivers of the olifants river system, South Africa. Journal of Freshwater Ecology 36: 97–109. https://doi.org/10.1080/02705060.2021.1901789.
Allan, J. D., & M. M. Castillo, 2007. Stream Ecology. 2nd ed. Springer Netherlands. https://doi.org/10.1007/978-1-4020-5583-6.
Barbour, M. T., J. Gerritsen, G. E. Griffith, R. Frydenborg, E. McCarron, J. S. White & M. L. Bastian, 1996. A framework for biological criteria for Florida streams using benthic macroinvertebrates. Journal of the North American Benthological Society 15: 185–211. https://doi.org/10.2307/1467948.
Bernadet, C., H. Touron-Poncet, N. Bargier & R. Céréghino, 2014. Guide méthodologique pour la mise en œuvre de l’Indice Biologique Macroinvertébré des Antilles (IBMA). Rapport Offices de l’Eau de Martinique et de Guadeloupe, Directions de l’Environnement, de l’Aménagement et du Logement de Martinique et Guadeloupe, Version 1.0, 40 pp.
Bernhardt, E. S., J. B. Heffernan, N. B. Grimm, E. H. Stanley, J. W. Harvey, M. Arroita, A. P. Appling, M. J. Cohen, W. H. McDowell, R. O. Hall Jr., J. S. Read, B. J. Roberts, E. G. Stets & C. B. Yackulic, 2018. The metabolic regimes of flowing waters. Limnology and Oceanography 63: 99–118. https://doi.org/10.1002/lno.10726.
Bhawsar, A., M. A. Bhat & V. Vyas, 2015. Distribution and composition of macroinvertebrates functional feeding groups with reference to catchment area in Barna Sub-Basin of Narmada River Basin. International Journal of Scientific Research in Environmental Sciences 3: 385–393. https://doi.org/10.12983/ijsres-2015-p0385-0393.
Bonada, N., M. Rieradevall & N. Prat, 2007. Macroinvertebrate community structure and biological traits related to flow permanence in a Mediterranean river network. Hydrobiologia 589: 91–106. https://doi.org/10.1007/s10750-007-0723-5.
Borkent, A. & G. R. Spinelli, 2007. Neotropical ceratopogonidae (Diptera: Insecta). In Adis, J., J. R. Arias, G. Rueda-Delgado & K. M. Wattzen (eds), Aquatic biodiversity in Latin America (ABLA), Vol. 4. Pensoft Publishers, Sofia-Moscow: 198.
Box, G. E. & D. R. Cox, 1964. An analysis of transformations. Journal of the Royal Statistical Society: Series B (methodological) 26: 211–243. https://doi.org/10.1111/j.2517-6161.1964.tb00553.x.
Boyero, L. & J. Bosch, 2004. The effect of riffle-scale environmental variability on macroinvertebrate assemblages in a tropical stream. Hydrobiologia 524: 125–132. https://doi.org/10.1023/B:HYDR.0000036127.94781.3c.
Calapez, A. R., C. L. Elias, S. F. P. Almeida & M. J. Feio, 2014. Extreme drought effects and recovery patterns in the benthic communities of temperate streams. Limnetica 33: 281–296. https://doi.org/10.23818/limn.33.22.
Cargill, S. K., C. Segura, S. R. Villamizar & D. R. Warren, 2021. The influence of lithology on stream metabolism in headwater systems. Ecohydrology 14: e2284. https://doi.org/10.1002/eco.2284.
Caribbean Natural Resources Institute (CANARI), 2019. The Caribbean islands biodiversity hotspot. 50 pp.
Chanut, P. C. M., T. Datry, C. Gabbud & C. T. Robinson, 2019. Direct and indirect effects of flood regime on macroinvertebrate assemblages in a floodplain riverscape. Ecohydrology 12: e2095. https://doi.org/10.1002/eco.2095.
Chará-Serna, A. M., J. D. Chara, M. del Carmen Zúñiga, R. G. Pearson & L. Boyero, 2012. Diets of leaf litter-associated invertebrates in three tropical streams. Annales De Limnologie—International Journal of Limnology 48: 139–144. https://doi.org/10.1051/limn/2012013.
Cheshire, K., L. Boyero & R. G. Pearson, 2005. Food webs in tropical Australian streams: the prevalence of shredders and predators. Freshwater Biology 50: 748–769. https://doi.org/10.1111/j.1365-2427.2005.01355.x.
Churches, C. E., P. J. Wampler, W. Sun & A. J. Smith, 2014. Evaluation of forest cover estimates for Haiti using supervised classification of Landsat data. International Journal of Applied Earth Observation and Geoinformation 30: 203–216. https://doi.org/10.1016/j.jag.2014.01.020.
Cineas, C. & S. Dolédec, 2022. Species richness and composition of Caribbean aquatic entomofauna: role of climate, island area, and distance to mainland. Frontiers of Biogeography 14: e54479. https://doi.org/10.21425/f5fbg54479.
Cortés-Guzmán, D., J. Alcocer & K. W. Cummins, 2021. Benthic macroinvertebrates of tropical streams: functional and trophic diversity of the Lacantún River, Mexico. Limnology 22: 313–328. https://doi.org/10.1007/s10201-021-00658-y.
Cummins, K. W., 1973. Trophic relations of aquatic insects. Annual Review of Entomology 18: 183–206. https://doi.org/10.1146/annurev.en.18.010173.001151.
Cummins, K. W., 2019. Functional analysis of stream macroinvertebrates. In Gokce, D. (ed), Limnology-some new aspects of inland water ecology. IntechOpen, London.
Dobson, M., J. M. Mathooko, F. K. Ndegwa & C. M’Erimba, 2003. Leaf litter processing rates in a Kenyan highland stream, the Njoro River. Hydrobiologia 519: 207–210. https://doi.org/10.1023/b:hydr.0000026592.50734.ea.
Dolisca, F., J. M. McDaniel, L. D. Teeter & C. M. Jolly, 2007. Land tenure, population pressure, and deforestation in Haiti: the case of Forêt des Pins Reserve. Journal of Forest Economics 13: 277–289. https://doi.org/10.1016/j.jfe.2007.02.006.
Feio, M. J., W. R. Ferreira, D. R. Macedo, A. P. Eller, C. B. M. Alves, J. S. França & M. Callisto, 2015. Defining and testing targets for the recovery of tropical streams based on macroinvertebrate communities and abiotic conditions. River Research and Applications 31: 70–84. https://doi.org/10.1002/rra.2716.
Ferreira, W. R., R. Ligeiro, D. R. Macedo, R. M. Hughes, P. R. Kaufmann, L. G. Oliveira & M. Callisto, 2014. Importance of environmental factors for the richness and distribution of benthic macroinvertebrates in tropical headwater streams. Freshwater Science 33: 860–871. https://doi.org/10.1086/676951.
Figueiró, R., E. S. D. Nascimento, L. H. Gil-Azevedo, M. Maia-Herzog & R. F. Monteiro, 2008. Local distribution of blackfly (Diptera, Simuliidae) larvae in two adjacent streams: the role of water current velocity in the diversity of blackfly larvae. Revista Brasileira De Entomologia 52: 452–454. https://doi.org/10.1590/s0085-56262008000300020.
Firmiano, K. R., R. Ligeiro, D. R. Macedo, L. Juen, R. M. Hughes & M. Callisto, 2017. Mayfly bioindicator thresholds for several anthropogenic disturbances in neotropical savanna streams. Ecological Indicators 74: 276–284. https://doi.org/10.1016/j.ecolind.2016.11.033.
Forio, M. A. E., K. Lock, E. D. Radam, M. Bande, V. Asio & P. L. Goethals, 2017. Assessment and analysis of ecological quality, macroinvertebrate communities and diversity in rivers of a multifunctional tropical island. Ecological Indicators 77: 228–238. https://doi.org/10.1016/j.ecolind.2017.02.013.
Grillet, M. E. & R. Barrera, 1997. Spatial and temporal abundance, substrate partitioning and species co-occurrence in a guild of Neotropical blackflies (Diptera: Simuliidae). Hydrobiologia 345: 197–208. https://doi.org/10.1023/a:1002901612957.
Gudbrandsson, S., D. Wolff-Boenisch, S. R. Gislason & E. H. Oelkers, 2011. An experimental study of crystalline basalt dissolution from 2≤pH≤11 and temperatures from 5 to 75°C. Geochimica Et Cosmochimica Acta 75: 5496–5509. https://doi.org/10.1016/j.gca.2011.06.035.
Gutiérrez, J. D., W. Riss & R. Ospina, 2002. Bioindicación de la calidad del agua con lógica difusa Sugeno y macroinvertebrados acuáticos de la Sabana de Bogotá. Acta Biológica Colombiana 7: 37–51.
Hamada, N., J. H. Thorp & D. C. Rogers, 2018. Keys to neotropical Hexapoda. In Thorp, J. H. & A. Covich (eds), Freshwater invertebrates. Academic press, New York.
Hart, D. D. & C. T. Robinson, 1990. Resource limitation in a stream community: phosphorus enrichment effects on periphyton and grazers. Ecology 71: 1494–1502. https://doi.org/10.2307/1938286.
Heino, J., P. Louhi & T. Muotka, 2004. Identifying the scales of variability in stream macroinvertebrate abundance, functional composition and assemblage structure. Freshwater Biology 49: 1230–1239. https://doi.org/10.1111/j.1365-2427.2004.01259.x.
Heino, J., H. Mykrä, H. Hämäläinen, J. Aroviita & T. Muotka, 2007. Responses of taxonomic distinctness and species diversity indices to anthropogenic impacts and natural environmental gradients in stream macroinvertebrates. Freshwater Biology 52: 1846–1861. https://doi.org/10.1111/j.1365-2427.2007.01801.x.
Hering, D., A. Schmidt-Kloiber, J. Murphy, S. Lücke, C. Zamora-Muñoz, M. J. López-Rodríguez, T. Huber & W. Graf, 2009. Potential impact of climate change on aquatic insects: a sensitivity analysis for European caddisflies (Trichoptera) based on distribution patterns and ecological preferences. Aquatic Sciences 71: 3–14. https://doi.org/10.1007/s00027-009-9159-5.
Hershey, A. E., A. L. Hiltner, M. A. J. Hullar, M. C. Miller, J. R. Vestal, M. A. Lock, S. Rundle & B. J. Peterson, 1988. Nutrient Influence on a stream grazer: Orthocladius microcommunities respond to nutrient input. Ecology 69: 1383–1392. https://doi.org/10.2307/1941635.
Houghton, D., 2012. Biological diversity of the Minnesota caddisflies (Insecta, Trichoptera). ZooKeys 189: 1–389. https://doi.org/10.3897/zookeys.189.2043.
Jacobsen, D., 2003. Altitudinal changes in diversity of macroinvertebrates from small streams in the Ecuadorian Andes. Archiv Fur Hydrobiologie 158: 145–168. https://doi.org/10.1127/0003-9136/2003/0158-0145.
Jones, J. I., J. F. Murphy, A. L. Collins, D. A. Sear, P. S. Naden & P. D. Armitage, 2012. The impact of fine sediment on macro-invertebrates. River Research and Applications 28: 1055–1071. https://doi.org/10.1002/rra.1516.
Jongman, R. H., C. J. F. Braak & O. F. R. Van Tongeren, 1987. Data analysis in community and landscape ecology. Pudoc Wageningen, 299 pp.
Kazanci, N., P. Ekingen, M. Dügel & G. Türkmen, 2015. Hirudinea (Annelida) species and their ecological preferences in some running waters and lakes. International Journal of Environmental Science & Technology 12: 1087–1096. https://doi.org/10.1007/s13762-014-0574-3.
Kohlmann, B., D. Vásquez, A. Arroyo & M. Springer, 2021. Taxonomic and functional diversity of aquatic macroinvertebrate assemblages and water quality in rivers of the dry tropics of Costa Rica. Frontiers in Environmental Science. https://doi.org/10.3389/fenvs.2021.660260.
Lepori, F. & B. Malmqvist, 2007. Predictable changes in trophic community structure along a spatial disturbance gradient in streams. Freshwater Biology 52: 2184–2195. https://doi.org/10.1111/j.1365-2427.2007.01846.x.
Li, A. O. & D. Dudgeon, 2009. Shredders: species richness, abundance, and role in litter breakdown in tropical Hong Kong streams. Journal of the North American Benthological Society 28: 167–180. https://doi.org/10.1899/08-043.1.
Li, Y., D. B. Wright & B. P. Bledsoe, 2022. Watershed controls and tropical cyclone-induced changes in river hydraulic geometry in Puerto Rico. Journal of Hydrology: Regional Studies 44: 101268. https://doi.org/10.1016/j.ejrh.2022.101268.
Mackey, R. L. & D. J. Currie, 2001. The diversity-disturbance relationship: is it generally strong and peaked? Ecology 82: 3479–3492. https://doi.org/10.1890/0012-9658(2001)082[3479:tddrii]2.0.co;2.
Majdi, N., A. Boiché, W. Traunspurger & A. Lecerf, 2015. Community patterns and ecosystem processes in forested headwater streams along a gradient of riparian canopy openness. Fundamental and Applied Limnology 187: 63–78. https://doi.org/10.1127/fal/2015/0740.
McArdle, B. H. & M. J. Anderson, 2001. Fitting multivariate models to community data: a comment on distance-based redundancy analysis. Ecology 82: 290–297. https://doi.org/10.1890/0012-9658(2001)082[0290:fmmtcd]2.0.co;2.
Merritt, R. W., K. W. Cummins, M. B. Berg, J. A. Novak, M. J. Higgins, K. J. Wessell & J. L. Lessard, 2002. Development and application of a macroinvertebrate functional-group approach in the bioassessment of remnant river oxbows in southwest Florida. Journal of the North American Benthological Society 21: 290–310. https://doi.org/10.2307/1468416.
Min, J. K., Y. J. Kim & D. S. Kong, 2019. Spatial distribution patterns of benthic macroinvertebrate functional feeding groups by stream size and gradient in Republic of Korea. Journal of Freshwater Ecology 34: 715–738. https://doi.org/10.1080/02705060.2019.1677793.
Minchin, P. R., 1987. An evaluation of relative robustness of techniques for ecological ordinations. Vegetatio 69: 89–107. https://doi.org/10.1007/bf00038690.
Monaghan, K. A., M. R. Peck, P. A. Brewin, M. Masiero, E. Zarate, P. Turcotte & S. J. Ormerod, 2000. Macroinvertebrate distribution in Ecuadorian hill streams: the effect of altitude and land use. Fundamental and Applied Limnology 149: 421–440. https://doi.org/10.1127/archiv-hydrobiol/149/2000/421.
Moreira, X., B. Castagneyrol, R. de la Mata, N. M. Fyllas, A. Galmán, C. García-Verdugo, A. R. Larrinaga & L. Abdala-Roberts, 2019. Effects of insularity on insect leaf herbivory and chemical defences in a Mediterranean oak species. Journal of Biogeography 46: 1226–1233. https://doi.org/10.1111/jbi.13589.
Nava, D., R. M. Restello & L. U. Hepp, 2015. Intra- and inter-annual variations in Chironomidae (Insecta: Diptera) communities in subtropical streams. Zoologia (curitiba) 32: 207–214. https://doi.org/10.1590/s1984-46702015000300005.
Nelson, S. M. & D. M. Lieberman, 2002. The influence of flow and other environmental factors on benthic invertebrates in the Sacramento River, USA. Hydrobiologia 489: 117–129. https://doi.org/10.1023/a:1023268417851.
Oksanen J., G. Simpson, F. Blanchet, R. Kindt, P. Legendre, P. Minchin, R. O'Hara, P. Solymos, M. Stevens, E. Szoecs, H. Wagner, M. Barbour, M. Bedward, B. Bolker, D. Borcard, G. Carvalho, M. Chirico, M. De Caceres, S. Durand, H. Evangelista, R. FitzJohn, M. Friendly, B. Furneaux, G. Hannigan, M. Hill, L. Lahti, D. McGlinn, M. Ouellette, E. Ribeiro Cunha, T. Smith, A. Stier, C. Ter Braak & J. Weedon, 2022. vegan: Community Ecology Package.R package version 2.6–4, <https://CRAN.R-project.org/package=vegan>
Oliveira, A. L. H. D. & J. L. Nessimian, 2010. Spatial distribution and functional feeding groups of aquatic insect communities in Serra da Bocaina streams, south-eastern Brazil. Acta Limnologica Brasiliensia 22: 424–441. https://doi.org/10.4322/actalb.2011.007.
Park, Y. S., S. Lek, T. S. Chon & P. F. M. Verdonschot, 2008. Evaluation of environmental factors to determine the distribution of functional feeding groups of benthic macroinvertebrates using an artificial neural network. Journal of Ecology and Field Biology 31: 233–241. https://doi.org/10.5141/jefb.2008.31.3.233.
Pauleus, O. & T. M. Aide, 2020. Haiti has more forest than previously reported: land change 2000–2015. PeerJ 8: e9919. https://doi.org/10.7717/peerj.9919.
Pellek, R., 1990. Combating tropical deforestation in Haiti: implementing an action plan requires a more stable political environment. Journal of Forestry 88: 15–19.
Power, M. E., A. Sun, M. Parker, W. E. Dietrich & J. T. Wootton, 1995. Hydraulic food chain models, an approach to the study of food-web dynamics in large rivers. BioScience 45: 159–167. https://doi.org/10.2307/1312555.
R Core Team, 2022. R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/.
Ramírez, A. & P. E. Gutiérrez-Fonseca, 2014. Functional feeding groups of aquatic insect families in Latin America: a critical analysis and review of existing literature. Revista De Biología Tropical 62: 155–167. https://doi.org/10.15517/rbt.v62i0.15785.
Ramirez, A. & C. M. Pringle, 1998. Structure and production of a benthic insect assemblage in a neotropical stream. Journal of the North American Benthological Society 17: 443–463. https://doi.org/10.2307/1468365.
Rasmussen, K. & C. Lindegaard, 1988. Effects of iron compounds on macroinvertebrate communities in a Danish lowland river system. Water Research 22: 1101–1108. https://doi.org/10.1016/0043-1354(88)90004-8.
Recalde, F. C., C. P. B. Breviglieri, M. F. Kersch-Becker & G. Q. Romero, 2021. Contribution of emergent aquatic insects to the trophic variation of tropical birds and bats. Food Webs 29: e00209. https://doi.org/10.1016/j.fooweb.2021.e00209.
Rezende, R. S., A. M. Santos, C. Henke-Oliveira & J. F. Jr Gonçalves, 2014. Effects of spatial and environmental factors on benthic a macroinvertebrate community. Zoologia (curitiba) 31: 426–434. https://doi.org/10.1590/s1984-46702014005000001.
Robart, G., 1984. Végétation de la République d'Haïti. PhD thesis, University of Grenoble.
Rosenberg, D. M. & V. H. Resh (eds), 1993. Freshwater biomonitoring and benthic macroinvertebrates. Chapman & Hall, New York.
Serra, S. R. Q., F. Cobo, M. A. S. Graça, S. Dolédec & M. J. Feio, 2016. Synthesising the trait information of European Chironomidae (Insecta: Diptera): towards a new database. Ecological Indicators 61: 282–292. https://doi.org/10.1016/j.ecolind.2015.09.028.
Skeldon, M. A., M. A. Vadeboncoeur, S. P. Hamburg & J. D. Blum, 2007. Terrestrial gastropod responses to an ecosystem-level calcium manipulation in a northern hardwood forest. Canadian Journal of Zoology 85: 994–1007. https://doi.org/10.1139/z07-084.
Smith, G. C., A. P. Covich & A. M. Brasher, 2003. An ecological perspective on the biodiversity of tropical island streams. BioScience 53: 1048–1051. https://doi.org/10.1641/0006-3568(2003)053[1048:aepotb]2.0.co;2.
Statzner, B. & B. Higler, 1985. Questions and comments on the river continuum concept. Canadian Journal of Fisheries and Aquatic Sciences 42: 1038–1044. https://doi.org/10.1139/f85-129.
Steinman, A. D. & C. D. McIntire, 1990. Recovery of lotic periphyton communities after disturbance. Environmental Management 14: 589–604. https://doi.org/10.1007/bf02394711.
Sun, C., L. Xia, M. Zhang, Q. He, N. Yu & H. Xiang, 2023. Macroinvertebrate functional feeding groups and its relationship with environmental factors in the Jingui River of Shenzhen. Authorea. https://doi.org/10.22541/au.167894686.67052193/v1.
Szczerkowska-Majchrzak, E. & M. Grzybkowska, 2015. Effects of hydrological disturbance of different magnitude on riverine habitats and benthic invertebrates. Polish Journal of Ecology 63: 135–141. https://doi.org/10.3161/15052249pje2015.63.1.012.
Tamaris-Turizo, C. E., G. A. Pinilla-A, C. J. Guzmán-Soto & C. E. Granados-Martínez, 2020. Assigning functional feeding groups to aquatic arthropods in a Neotropical Mountain river. Aquatic Biology 29: 45–57. https://doi.org/10.3354/ab00724.
Thioulouse, J., S. Dray, A.-B. Dufour, A. Siberchicot, T. Jombart & S. Pavoine, 2018. Multivariate analysis of ecological data with ade4, Springer, New York: https://doi.org/10.1007/978-1-4939-8850-1.
Tomanova, S. & P. Usseglio-Polatera, 2007. Patterns of benthic community traits in neotropical streams: relationship to mesoscale spatial variability. Fundamental and Applied Limnology 170: 243–256. https://doi.org/10.1127/1863-9135/2007/0170-0243.
Tomanova, S., E. Goitia & J. Helešic, 2006. Trophic levels and functional feeding groups of macroinvertebrates in neotropical streams. Hydrobiologia 556: 251–264. https://doi.org/10.1007/s10750-005-1255-5.
Tomanova, S., P. A. Tedesco, M. Campero, P. A. Van Damme, N. Moya & T. Oberdorff, 2007. Longitudinal and altitudinal changes of macroinvertebrate functional feeding groups in neotropical streams: a test of the River Continuum Concept. Fundamental and Applied Limnology 170: 233–241. https://doi.org/10.1127/1863-9135/2007/0170-0233.
Tomanova, S., N. Moya & T. Oberdorff, 2008. Using macroinvertebrate biological traits for assessing biotic integrity of neotropical streams. River Research & Applications 24: 1230–1239. https://doi.org/10.1002/rra.1148.
Tripole, S., E. A. Vallania & M. D. C. Corigliano, 2008. Benthic macroinvertebrate tolerance to water acidity in the Grande River sub-basin (San Luis, Argentina). Limnetica 27: 29–38. https://doi.org/10.23818/limn.27.03.
Uehlinger, U. & M. W. Naegeli, 1998. Ecosystem metabolism, disturbance, and stability in a prealpine gravel bed river. Journal of the North American Benthological Society 17: 165–178. https://doi.org/10.2307/1467960.
Uehlinger, U., M. Naegeli & S. G. Fisher, 2002. A hetero-trophic desert stream? The role of sediment stability. Western North American Naturalist 62: 466–473.
Vannote, R. L., G. W. Minshall, K. W. Cummins, J. R. Sedell & C. E. Cushing, 1980. The river continuum concept. Canadian Journal of Fisheries and Aquatic Sciences 37: 130–137. https://doi.org/10.1139/f80-017.
Vázquez, R. F., D. Vimos-Lojano & H. Hampel, 2020. Habitat suitability curves for freshwater macroinvertebrates of Tropical Andean Rivers. Water 12: 2703. https://doi.org/10.3390/w12102703.
Villanueva, V. D., R. Albarino & C. Canhoto, 2012. Positive effect of shredders on microbial biomass and decomposition in stream microcosms. Freshwater Biology 57: 2504–2513. https://doi.org/10.1111/fwb.12023.
Wallace, J. B. & J. R. Webster, 1996. The role of macroinvertebrates in stream ecosystem function. Annual Review of Entomology 41: 115–139. https://doi.org/10.1146/annurev.en.41.010196.000555.
Wallace, J. B., S. L. Eggert, J. L. Meyer & J. R. Webster, 2015. Stream invertebrate productivity linked to forest subsidies: 37 stream-years of reference and experimental data. Ecology 96: 1213–1228. https://doi.org/10.1890/14-1589.1.
Wantzen, K. M. & R. Wagner, 2006. Detritus processing by invertebrate shredders: a neotropical–temperate comparison. Journal of the North American Benthological Society 25: 216–232. https://doi.org/10.1899/0887-3593(2006)25[216:dpbisa]2.0.co;2.
Wickham, H., 2016. ggplot2: Elegant Graphics for Data Analysis, Springer, New York:
Winterbourn, M. J., J. S. Rounick & B. Cowie, 1981. Are New Zealand stream ecosystems really different? New Zealand Journal of Marine and Freshwater Research 15: 321–328. https://doi.org/10.1080/00288330.1981.9515927.
Wolmarans, C. T., M. Kemp, K. N. De Kock & V. Wepener, 2017. The possible association between selected sediment characteristics and the occurrence of benthic macroinvertebrates in a minimally affected river in South Africa. Chemistry and Ecology 33: 18–33. https://doi.org/10.1080/02757540.2016.1261121.
Zuur, A. F., E. N. Ieno & C. S. Elphick, 2010. A protocol for data exploration to avoid common statistical problems. Methods in Ecology and Evolution 1: 3–14. https://doi.org/10.1111/j.2041-210x.2009.00001.x.
Funding
Chevelie Cineas received funds from the French Embassy in Haiti in the context of a co-tutelle PhD thesis between the University of Lyon 1 (France) and the State University of Haiti (Haiti). This work was also performed under the auspice of the EUR H2O’Lyon (ANR-17-EURE-0018) of Université de Lyon (UdL) within the programme ‘Investissements d’Avenir’ operated by the French National Research Agency (ANR). This article was previously edited for proper English language, grammar, punctuation, spelling, and overall style by two highly qualified native English-speaking editors at American Journal Experts (certification verification key 46E4-0CD9-C540-699E-534P issued on October 28, 2023, and certification key 200-249-228-302-385 issued on March 11, 2024).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Chevelie Cinéas and Sylvain Dolédec. The first draft of the manuscript was written by Chevelie Cinéas and Sylvain Dolédec commented on previous versions of the manuscript. Both authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors have no competing interests to declare that are relevant to the content of this article.
Additional information
Handling editor: Verónica Ferreira
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Cinéas, C., Dolédec, S. Influence of climate, physical and chemical variables on the taxonomic and functional responses of macroinvertebrate communities in tropical island rivers. Hydrobiologia 851, 3735–3754 (2024). https://doi.org/10.1007/s10750-024-05532-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-024-05532-3