Abstract
With the aim of assessing whether hydrological alterations differently affect non-native and native fish species in coastal systems, we investigated fish assemblages along the fluvio-estuarine ecotone of the Neotropical São João River, Brazil. During a year, samplings with standardized efforts and limnological parameter records were carried out bimonthly at 15 sampling points in the last 50 km of the São João River, representing natural (meandering riverine and estuarine) and modified (reservoir, channelized and drained floodplain) stretches. We recorded 48 marine and 51 freshwater fish species, with 12 non-native species among the latter. Several native species were exclusively found within natural stretches, whereas non-native species presented significantly higher biomass catches in modified stretches. Only three species occurred in the entire study area, all of which were non-native species. Non-native species were associated with low dissolved oxygen and pH levels below the dam, whereas native freshwater species responded negatively to water transparency, and marine species responded positively to water conductivity at the hydrologically unaltered points. The hydrological alterations disrupted the fluvio-estuarine ecotone of the lower São João River, which plays the role of a dispersion source for non-native species and where invasive species are favored.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The Neotropics encompass extensive fluvial networks and a large fraction of the freshwater fishes of the world, with 6025 valid species (Albert et al., 2020). However, rivers in this region are increasingly subject to hydraulic alterations that affect especially rheophilic species (Nilsson et al., 2005; Agostinho et al., 2016; Winemiller et al., 2016). By disrupting longitudinal displacements along the river channel, dams isolate formerly continuous fish populations from the up- and downstream stretches, affecting species that depend on lotic conditions and free access to habitats for feeding, reproduction, and nursery grounds (Araújo et al., 2009; Orsi & Briton, 2014; Winemiller et al., 2016). In some cases, the meandering courses of floodplains might experience additional interventions with channelization and marginal embankments. The conversion of extensive sinuous stretches into straighter and shorter channels with elevated banks causes large impacts on fluvial ecosystems since the structural complexity and lateral connectivity between the river and the floodplain are reduced or even eliminated (Brookes, 1985; Amoros & Bornette, 2002). The straightening of the river channel also eliminates beaches and backwaters, which are typical marginal habitats formed by depositional and erosional processes on meandering stretches (Brookes, 1985). In addition to eliminating microhabitats preferentially occupied by small-sized specialist fishes and blocking lateral displacements to nursery areas (Jurajda, 1995; Belliard et al., 1999), the impacts of channelizing and damming floodplain rivers may worsen when followed by species introductions (Dudgeon et al., 2006; Agostinho et al., 2008; Garcia et al., 2018).
Hydrological interventions of rivers favor the introduction of non-native species that may first become established in the altered stretches of a river and then start to threaten the native biota of the natural stretches via competition or predation (Agostinho et al., 2016; Liew et al., 2016; Garcia et al., 2018). In general, highly invasive fish species share some life-history and tolerance characteristics that together tend to maximize their reproductive efforts (e.g., high fecundity, developed parental care, long reproductive season, tolerance to hypoxia), which results in established populations of these invasive species within a few years after their introduction (Pool & Olden, 2012; Orsi & Briton, 2014; Garcia et al., 2018). However, the native fish fauna, in addition to being able to surpass the new environmental filters imposed by hydrological alterations, must be able to successfully coexist with invaders, which are generally better adapted to the current conditions of high water transparency, stable water level, and water residence time on the reservoir as well as flow reduction and turbulent waters just below the dam (Espínola et al., 2010; Orsi & Briton, 2014; Franco et al., 2018). The replacement of native biota, which may include rare and endemic species, by widely spread non-native species results in biotic homogenization, an anthropogenic phenomenon that has been seen in the Neotropics and elsewhere (Pool & Olden, 2012; Vitule et al., 2012; Liew et al., 2016). Considering that biotic homogenization represents one of the main challenges in the conservation of freshwater fish, it is urgent to understand invasion dynamics in ecoregions with high species richness (including areas with high rates of endemism) and whose fluvial courses are subjected to hydrological alterations (Leprieur et al., 2008; Olden et al., 2010; Daga et al., 2015).
The effects of hydrological alterations in the enhancement of species invasions have been mostly explored in Neotropical watersheds regulated by hydroelectric dams (Gomes & Miranda, 2001; Agostinho et al., 2008; Franco et al., 2018). However, even rivers with low capacities for hydropower production, such as those across coastal lowlands, are affected by hydrological interventions and species introductions (Catelani et al., 2014). In these rivers, freshwater species and several marine species that generally enter the rivers to reach feeding or nursery grounds face invaders (Ferreira & Petrere, 2009; Catelani et al., 2021a). The Fluminense ecoregion (FEOW352, sensu Abell et al., 2008) encompasses an important fraction of southeast Brazil and includes the metropolitan area of the capital of the State of Rio de Janeiro. Despite its relatively small area (6,763 km2), the Fluminense ecoregion contains a high number of fish species per area, including endemic species and species that require different habitats to complete their life cycles (Mazzoni et al., 2006; Abell et al., 2008).
Several rivers in the Fluminense ecoregion are subjected to drastic hydrological alterations, which are mainly related to channelization and drainage of the floodplains across the coastal plain (Moulton et al., 2007; Catelani et al., 2014; Marçal et al., 2017). The São João River is an emblematic example of a coastal river in the Fluminense ecoregion in which hydrological alterations preceded the deliberate introduction of non-native species. Such interventions included channelization, the placement of earthen embankments, and drainage of the floodplain since 1960; it also included the construction of a dam ca. twenty years later, before the first species introductions (CILSJ, 2002; Catelani et al., 2021b). With the aims of improving agriculture and controlling water-borne diseases and the water supply, these interventions caused deep alterations in the hydrological cycle of the lower course, where the structural complexity of the river was reduced and the lateral connectivity between the main channel and the floodplain was extirpated (Moulton et al., 2007). Before the interventions, the meandering lower course of the São João River was 130 km long and surrounded by an extensive forested floodplain, where hundreds of swamps and oxbow lagoons covered more than 1400 km2 (CILSJ, 2002). Most of this area is now drained and has been replaced by pasture for livestock activities. After the filling of the reservoir (ca. 30 years ago), sport fishers deliberately introduced the peacock bass (genus Cichla), an Amazonian piscivorous fish greatly appreciated in recreational fisheries (Catelani et al., 2021b). Currently, peacock bass and several other non-native species from other Neotropical watersheds or zoogeographic regions are caught in the lower São João River, such as the Silver Dollar Metynnis lippincottianus (Cope 1870) and the African catfish Clarias gariepinus (Burchell 1822) (Catelani et al., 2021a).
In this study, we aimed to assess the representativeness of non-native species along the transition from riverine to estuarine sections (herein treated as the riverine-estuarine ecotone, sensu Ward et al., 1999) of the São João River, since in addition to being the most hydrologically altered reach, it is the core area of species introductions. By investigating several natural and hydrologically altered stretches along the last 50 km of the river, we provide a limnological characterization, evaluate the representativeness of the non-native and native species in the structure of the fish assemblages, and explore the degree to which the species respond to limnological parameters. We expect to provide evidence that non-native species succeed in altered habitats if we find higher catches of invasive species on the altered compared to the natural stretches. Since species introductions in the São João River took place decades ago, the knowledge gathered on this system might generate important subsidies to foresee the impacts that other coastal rivers may experience due to similar hydrological interventions and species introductions.
Materials and methods
Study area
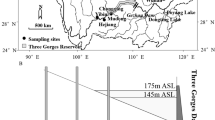
The São João River basin covers 2190 km2 (22° 30′–22° 41′ S and 41° 59′–42° 20′ W) of the Fluminense ecoregion in southeastern Brazil (CILSJ, 2002). The river originates at 800 m above sea level and runs most of the ca. 120 km west across an extensive coastal plain into the Atlantic Ocean (Fig. 1). The mean annual temperature varies between 22 and 24°C, and the annual accumulated rainfall reaches 1000 to 1300 mm, with a rainy and warm season between October and March and a dry season with mild temperatures between April and September (Alvares et al., 2013). Despite the low demographic density of the watershed, remnants of the Atlantic Forest are mostly found on the tops of hills and in two conservation units (the integral protection unit called the Poço das Antas Biological Reserve and the Environmental Protection Area of the São João River Basin/Mico-Leão-Dourado). The extensive floodplain over the last 40 km of the river was mostly drained and converted into pasture for livestock ranching.
Location of the 15 sampling points along the lower course of the São João River, State of Rio de Janeiro, Brazil. The order of the sampling points follows the upstream–downstream gradient and represents the natural free flowing sinuous riverine (1–3) and estuarine (13–15) stretches, the hydrologically altered (in bold) Juturnaíba Reservoir (4–6), and the channelized river downstream of the dam (7–12), which runs in parallel to the sinuous former main channel
The Juturnaíba Reservoir is located 40 km upstream from the river mouth, presents a surface area of ca. 45 km2, and provides drinking water to nearby municipalities (ca. 600,000 inhabitants) located both inside and outside of the São João River basin. The reservoir is surrounded mostly by crops and pastures, and its depth varies between 1 and 5 m, with extensive banks of submerged and floating aquatic plants (CILSJ, 2002). Upstream from the reservoir, the river flows unmodified within the Poço das Antas Biological Reserve and is surrounded by a forested floodplain. Downstream of the dam, the channelized river runs 30 km in parallel to the sinuous former main channel and across a drained and deforested coastal plain, where it receives several low-order channelized tributaries. Due to the regulated flow (4 m3s−1 on average) of the river and the influence of tides, this long-altered stretch presents semilentic characteristics. The last 10 km of the river runs along a natural sinuous stretch, where the river channel is wide and deep (approximately 150 m width and 8 m depth), and the river banks are covered by extensive mangroves due to the influence of marine water intrusion (Fig. 1).
Fish sampling and limnological characterizations
Standardized samplings were carried out bimonthly at 15 fixed points that cover natural and altered stretches across the last 50 km of the São João River (Fig. 1). According to similar environmental characteristics (e.g., riverbank cover, the degree of channel modification), the sampling points were split into two groups. The sampling points located in the vegetated, free flowing, and nonchannelized fluvial stretches upstream of the reservoir (sampling points 1–3) and along the estuary (sampling points 13–15) were designated as natural. Conversely, the altered region encompasses the sampling points located at the hydrologically modified stretches, including the reservoir (sampling points 4–6) and the channelized river that is surrounded by earthen embankments downstream of the dam (sampling points 7–9) that runs straight across floodplain, where several inflows of channelized creeks are located (sampling points 10–12); these sampling points were designated as altered (Fig. 1). Samplings below the dam (sampling points 7–15) were carried out between January 2016 and January 2017, whereas samplings above the dam (sampling points 1–6) occurred between October 2016 and October 2017. At each sampling point, a set of nine gillnets (15, 20-, 25-, 30-, 35-, 40-, 45-, 60-, and 80-mm between-knots) and trammel nets (with mesh sizes of 35 and 110 mm) were set by three individuals between 16:00 and 8:00. During these 16 h in the water, the gill and trammel nets were checked once, between 20:00 and 22:00. Cast nets (10-, 20-, and 25-mm), seines (0.8-cm mesh size), and hand and dipnets were employed to include the small fish as well. Soon after fish sampling, the following limnological parameters were recorded: electrical conductivity (μS/cm) as a proxy of water salinity, dissolved oxygen (mg/l), temperature (°C) (YSI Pro 2030), pH (Merck™ strips), and water transparency (m) and depth (m) (with the use of a Secchi disk).
Captured fish were euthanized using benzocaine and stored on ice until immersion in a 4% formaline solution, then were subsequently stored in 70°GL ethanol. We recorded the standard length (SL, to the nearest mm) and total weight (TW, g) of each specimen. Since the sampling effort was standardized among the 15 points, we expressed the numerical abundance and biomass as numerical catch and biomass catch, respectively. We identified specimens with the use of taxonomic guides and keys (e.g., Figueiredo & Menezes, 1978, 1980, 2000; Menezes & Figueiredo, 1980, 1985; Kullander & Ferreira, 2006) and split them into native and non-native species after checking the species distributions in Fricke et al., (2020). Voucher specimens were deposited in the Fish Collection (NPM) of the Instituto de Biodiversidade e Sustentabilidade, Universidade Federal do Rio de Janeiro (NUPEM/UFRJ). The fish collection and euthanasia procedures were in accordance with the Brazilian requirements (collection permit SISBIO 45745; Committee of Ethics in Animal Experimentation of the Campus UFRJ-Macaé Professor Aloísio Teixeira MAC029).
Data analyses
Physical and chemical characteristics
We summarized the mean values of the six limnological variables recorded on the six campaigns for each sampling point in a matrix that was submitted to a principal component analysis (PCA). PCA ordinated the 15 sampling points according to their similarity in limnological variables and revealed the variables most important to the ordination. Only eigenvalues higher than those produced by a broken stick model were retained for interpretation, and they were assessed with the “bstick” function. We performed PCA with the function “princomp” in the package “vegan” (Oksanen et al., 2019) after standardizing (z-score) all variables.
Fish assemblages and non-native species
The characterization of the fish assemblages along the riverine-estuarine ecotone included an ordination of the sampling points according to species composition as well as comparisons of the attributes of non-native and native species between the assemblages in the natural and hydrologically altered stretches. We ordinated the 15 assemblages based on species composition (presence/absence data) by applying nonmetric multidimensional scaling (NMDS) method to similarity matrices based on Jaccard distance with the package “MASS” (Venables & Ripley, 2002). The function “metaMDS” calculated the distortion (stress) between the similarity matrix and the ordination produced in the graphical representation of the axes. Stress values < 0.2 were considered acceptable for use in interpreting the configurations (Quinn & Keough, 2002). A permutational analysis of variance available in the “permute” package (Simpson, 2019) was further applied to verify whether the distance-based ordination responded to hydrological alteration [natural (1–3, 13–15) and altered (4–12) sampling points]. The attributes of species richness and the numerical and biomass abundances of non-native and native species were compared between the assemblages of natural and altered stretches through Student’s t test. We employed the corresponding Welch test whenever the data were heteroscedastic, even after the appropriate transformation was applied.
Species responses to the physical and chemical characteristics
Species representativeness in the assemblages was expressed by an index of relative importance (IRI), which was based on the frequency of occurrence of each species in the samplings (FO) and on the numerical (N) and biomass (B) catches (IRI% = FO*N*B). To explore the effects of the limnological parameters on the species representativeness along the riverine-estuarine ecotone, we performed a canonical correspondence analysis (CCA) available in the package “vegan” to the IRI and limnological matrices after excluding the species that together represented < 0.05% of the overall IRI. We also applied the Hellinger transformation on the IRI matrix before it was submitted to the CCA to assign low weights to species with low counts and many zeros (Legendre & Gallagher, 2001). With the CCA, we aimed to evaluate how much of the variation in species representativeness could be explained by linear correlations of the physical and chemical variables, as well as to identify species’ environmental preferences (Palmer, 1993).
All statistical analyses were carried out in R version 3.3.3 (R Core Team, 2017), and significance was implied at P < 0.05.
Results
Physical and chemical characteristics
Along the last 50 km of the river, the São João River becomes deeper, and the water conductivity increases suddenly at the beginning of the meandering estuary (Fig. 2a, c). The low pH and less oxygenated waters at the lowermost channelized sampling points differ from those just below the dam, where lower depths and temperatures are the main characteristics (Fig. 2d–f). The warmer and clearer waters in the reservoir differ mostly from the shallow, less transparent waters at the riverine sampling points (Fig. 2b, d). In this riverine stretch, the pH levels were the highest recorded and were similar to those found in the estuary (Fig. 2f). From the longitudinal gradient perspective, the pH and dissolved oxygen levels diverged the most among the sampling points and marked a disruption where the channelized São João River received the channelized tributaries before the freshwater became diluted by marine waters (Fig. 2e, c, f).
Variations in the limnological characteristics along the fluvio-estuarine ecotone of the lower course of the São João River, Brazil. Sampling points 1–3 and 13–15 represent the natural meandering riverine and estuarine stretches, respectively, whereas the hydrologically altered sampling points (in bold) 4–6 are located in the reservoir, and the points 7–12 are located on the channelized stretch below the dam
The first two axes of the PCA (PC1 eigenvalue = 2.382; PC2 eigenvalue = 1.431) were significant and explained most of the variation in the limnological parameters (69%), which varied particularly among sampling points located immediately above and below the dam (Fig. 3). The sampling points in the channelized stretch presented higher PC1 scores and were associated with low dissolved oxygen and pH levels, whereas the sampling points in the meandering riverine stretch presented lower PC1 scores and were associated with lower values of Secchi depth, total depth and temperature. The sampling points located in the meandering estuarine stretch presented higher PC2 scores and were associated with brackish waters, which is a singular characteristic of the lowermost stretch of the São João River (Fig. 3).
Ordination of the 12 sampling points along the fluvio-estuarine ecotone of the lower course of the São João River, Brazil, according to a principal component analysis of the limnological characteristics. Sampling points 1–3 and 13–15 represent the natural meandering riverine and estuarine stretches, respectively, whereas the hydrologically altered sampling points (in bold) 4–6 are located in the reservoir, and the points 7–12 are located on the channelized stretch below the dam
Fish assemblages and non-native species
A total of 6670 specimens belonging to 99 species were caught, 48 of which were marine species and 51 of which were freshwater species (Online resource 1). The ordination of the assemblages based on the species presence/absence data evidenced a higher similarity in species composition among the altered stretches because the sampling points 4–12 were closer to one another when compared to other groups (Fig. 4; Online resource 1). The PERMANOVA detected significant differences between the assemblages of the natural and altered stretches based on the species presence/absence data (Pseudo F1,13 = 4.230; R2 = 0.246; P < 0.001).
Ordination of the 12 fish assemblages along the fluvio-estuarine ecotone of the lower course of the São João River, Brazil, according to a nonmetric multidimensional scaling method applied to species presence/absence data. Sampling points 1–3 and 13–15 represent the natural meandering riverine and estuarine stretches, respectively, whereas the hydrologically altered sampling points (in bold and enclosed in the polygon) 4–6 are located in the reservoir, and the points 7–12 are located on the channelized stretch below the dam
Twelve of the freshwater species are non-native species, and although they are widely spread along the fluvio-estuarine ecotone, they prevailed in the assemblages of the altered stretches (Online resource 1, Fig. 5a). Despite having the lowest species richness, those assemblages proportionally presented the highest numerical catches (4–6 on the reservoir) and the highest catches of biomass of the non-native species (10–12 on the channelized stretch) (Fig. 5b, c). Among the fish assemblage attributes, the catches of non-native species were higher in the altered compared to the natural stretches. For biomass, these differences were statistically significant (Table 1). Non-native species represent 10% of the species recorded on the lower São João River. However, they account for 37% of the overall numerical (Fig. 5b) and 39% of the biomass catches (Fig. 5c). In contrast, the native freshwater and marine species are negatively correlated in number, numerical, and biomass abundance along the fluvio-estuarine ecotone (Online resource 1, Fig. 5). The assemblages in the natural riverine stretch presented only freshwater species, including several rare species and a few dominant species, 16 of which were exclusive, whereas in the estuary, marine species dominated (Online resource 1, Fig. 5).
Species richness (a), numerical catches (b), and biomass catches (c) of the native and non-native freshwater (FSW) and native marine (MRN) species recorded along the fluvio-estuarine ecotone of the São João River. Sampling points 1–3 and 13–15 represent the natural meandering riverine and estuarine stretches, respectively, whereas the hydrologically altered sampling points (in bold) 4–6 are located in the reservoir, and the points 7–12 are located on the channelized river below the dam
Species responses to the physical and chemical characteristics
Only thirty-four species corresponded to 99.5% of the IRI matrix. A large amount of the variance (68%) in the IRI matrix was explained by the physical and chemical variables, with the first two CCA axes being significant (CCA1 eigenvalue = 0.802, P = 0.001; CCA2 eigenvalue = 0.407, P = 0.004) (Fig. 6). Most non-native species and several native marine and freshwater species displayed strong unimodal responses to environmental gradients determined by pH, electrical conductivity, depth, water transparency, and temperature. Downstream of the dam, where low pH levels prevailed, almost all of the non-native species [M. lippincotianus, C. gariepinus, Oreochromis niloticus (Linnaeus 1758), Hoplerythrinus unitaeniatus (Spix & Agassiz 1829) and Cichla kelberi] and a few representatives of native species [Centropomus parallelus Poey 1860, Eugerres brasilianus (Cuvier 1830) and Geophagus brasiliensis (Quoy & Gaimard 1824)] attained their highest representativeness values. The native freshwater species Brycon insignis Steindachner 1877, Trachelyopterus striatulus (Steindachner 1877), Astyanax gr. bimaculatus (Linnaeus 1758) and Psalidodon parahybae (Eigenmann 1908) were exclusive to the assemblages of the riverine stretch, where the water attains a lower transparency and temperature than in other stretches (Table 1; Fig. 6a). Marine species such as Genidens genidens (Cuvier 1829), Lycengraulis grossidens (Spix & Agassiz 1829), Stellifer brasiliensis (Schultz 1945), and Mugil curema Valenciennes 1836 were much more representative in the assemblages of the deeper and brackish estuarine stretch (Fig. 6a). Only three species [C. kelberi, C. gariepinus and Hoplosternum littorale (Hancock 1828)] were recorded in both the natural and altered conditions (Table 1).
Ordination of the native ( ) and non-native (
) and non-native ( ) freshwater and native marine (
) freshwater and native marine ( ) species (a) and sampling points (b) according to a canonical correspondence analysis along the fluvio-estuarine ecotone of the São João River, Brazil. The six vectors correspond to the limnological parameters: electrical conductivity (Cond), dissolved oxygen (DO), temperature (Temp), pH, transparency of Secchi disk (Secchi) depth (Depth). In (b), sampling points 1–3 and 13–15 represent the natural meandering riverine and estuarine stretches, respectively, whereas the hydrologically altered sampling points (in bold) 4–6 are located in the reservoir, and the points 7–12 are located on the channelized stretch below the dam
) species (a) and sampling points (b) according to a canonical correspondence analysis along the fluvio-estuarine ecotone of the São João River, Brazil. The six vectors correspond to the limnological parameters: electrical conductivity (Cond), dissolved oxygen (DO), temperature (Temp), pH, transparency of Secchi disk (Secchi) depth (Depth). In (b), sampling points 1–3 and 13–15 represent the natural meandering riverine and estuarine stretches, respectively, whereas the hydrologically altered sampling points (in bold) 4–6 are located in the reservoir, and the points 7–12 are located on the channelized stretch below the dam
Discussion
Hydrological alterations disrupted the longitudinal gradient of the lower São João River and affected the limnology of the modified stretches that experienced the predominance of non-native species in the fish assemblages. The more homogeneous assemblages of the modified stretches reflect the smaller set of fish species that the reservoir and the channelized lower course of the São João River harbor. Among them are the African C. gariepinus and the Neotropical but also non-native species C. kelberi and H. littorale, medium- to large-size species, which were the only species occurring along all natural and hydrologically altered stretches. In contrast, the fish assemblages of the riverine and estuarine sample sites are composed of dozens of native freshwater and marine species, respectively. Although most of them were small-sized species [a characteristic that prevails in the freshwater fish fauna of the coastal drainages of the Neotropical Atlantic Forest (Abilhoa et al., 2011)] or juveniles of medium- to large-size species of estuaries (Blaber, 2000), the large-sized B. insignis, an endangered rheophilic seed disperser, was recorded only in the assemblages of the uppermost meandering riverine course. Downstream B. insignis and several other rheophilic native species were no longer found.
The absence of almost half of the native freshwater species in the reservoir and channelized stretches and of the totality of the marine species upstream of the dam [except Platanichthys platana (Regan 1917)] suggests inability of most native species to cope with the artificial lentic and semilentic conditions that prevail upstream and downstream of a dam due to flow regulation. These semilentic environments are prone to harboring non-native, preadapted species and several sedentary native species that do not depend on lateral or longitudinal displacements in the floodplain or upstream areas, respectively (Corbacho & Sanchéz, 2001; Poff et al., 2007; Mims & Olden, 2013). Since most non-native species in the lower São João River originate from high-diversity areas (e.g., the Amazon and eastern African basins), their physiological and behavioral adaptations used to survive in such highly competitive environments probably favor successful establishment outside their original ranges (Fitzgerald et al., 2016; Carvalho et al., 2020). On the lower São João River, in addition to sharing less diverse assemblages dominated by non-native species, hydrologically altered stretches experience physical and chemical characteristics directly related to the effects of river regulation, such as high variations in pH, transparency, depth, and oxygenation. Meanwhile, these conditions negatively affect native species that evolved life cycles requiring turbid waters, pH-neutral, and oxygenated waters (Blaber, 2013) and seem to have fewer effects on non-native species that are adapted to hypoxia (C. gariepinus and H. littorale) and are visually oriented (C. kelberi) (Brauner et al., 1995; Espínola et al., 2009; Weyl et al., 2016).
The absence of studies prior to the damming of the São João River hampers our attempt to identify vulnerable species that might be displaced or locally eliminated after interacting with those invaders from which catastrophic effects on freshwater species have been reported (Pelicice & Agostinho, 2009). The few small characins and the curimatid Cyphocharax gilbert (Quoy & Gaimard 1824) that, even with low representativeness, are still recorded on the altered stretches below the dam are strong candidates, but silent local species extinctions in the past cannot be discarded. Although the São João River shares with other watersheds of the Fluminense ecoregion, a lack of former fish compositional data, the high representativeness of the non-native species reported herein reveals the speed of the invasion process, since the invasion history in the area is relatively recent. None of the non-native species reported herein were recorded in the earliest study on the basin (Bizerril, 1995). The first records of non-native species on the São João River date back to the beginning of this century, when C. kelberi, C. gariepinus, and O. niloticus were reported in the Juturnaíba Reservoir and Nannostomus beckfordi Günther 1872 and Cichlasoma dimerus (Heckel 1840) were reported in the stretch upstream from the reservoir (Jaramillo-Villa, 2010; Mendonça et al., 2018). Tank escapes and illegal releases by sport fishers are the most probable sources of the introductions of non-native cichlids and C. gariepinus (Weyl et al., 2016; Catelani et al., 2021b), whereas H. litoralle, whose natural distribution includes a wide range of Neotropical watersheds except coastal watersheds in southeastern Brazil, has been caught in the region since the 1980s (de Moraes et al., 2017). Reservoirs are widely recognized as the main vectors for species introductions, and due to its proximity to the metropolitan area of Rio de Janeiro city (the second most populous Brazilian city), Juturnaíba Reservoir might be viewed as the first, and possibly still the main core of the invasion process along the fluvio-estuarine ecotone. In Brazil, the encouragement by the government of these activities and the inefficient legislation regarding rearing, managing, and combating non-native species (Pelicice et al., 2017; Azevedo Santos et al., 2017; Brito et al., 2018) reinforce aquaculture and sport fisheries as the main vectors of species introduction (Pelicice et al., 2014; Barrella et al., 2014; Casimiro et al., 2018).
From a worldwide dataset that included a thousand river basins, Leprieur et al. (2008) found evidence that human activities ultimately facilitate the establishment of non-native species by disturbing natural landscapes and increasing propagule pressure. The “human activity” hypothesis (sensu Leprieur et al., 2008) that river regulations positively affect non-native species to the detriment of several native species that may experience local extinction has been widely supported in study cases and meta-analyses (Vitule et al., 2012; Liew et al., 2016; Pelicice et al., 2018) and partially also herein. Our results support the expectations that non-native species are favored in hydrologically altered river stretches, and as non-native species are widely spread along the fluvio-estuarine ecotone, they increase the homogeneity in assemblage compositions. However, our data do not support a deleterious effect of non-native species over native species. We raise two non-exclusive hypotheses to explain why, at least at the level of assemblage attributes, we did not find differences in native species between the natural and altered stretches. The first hypothesis is related to the time elapsed since non-native species threatened the native fauna on the lower São João River. Although hydrological modifications took place almost a century ago (floodplain channelization), non-native species have developed sustainable populations almost only in the last 20 years (Catelani et al., 2021b). It might be for a given length of time that the deleterious effects of invaders still appear imperceptible on the assemblage level. The second hypothesis is related to the high carrying capacity that the continuous input of marine resources (i.e., crustaceans and several small-sized shoaling fish species) provides to consumers on the reach below the dam. When included as feeding resources, marine organisms may both alleviate the predation pressure of non-native species over freshwater fish fauna and enhance the population growth of invaders, since self-regulatory mechanisms such as cannibalism are frequently reported in invaded reservoirs (Pereira et al., 2017; Mendonça et al., 2018). Personal and published information confirms the wide consumption of crustaceans and marine fish species by C. kelberi and C. gariepinus (PA Catelani, unpubl. data; Barrella et al., 2014; Rabelo & Soares, 2014) in estuaries, and specially designed studies would shed light on these questions involving the biotic interactions of invasive species across aquatic ecotones.
We recorded more marine species in freshwater than freshwater species crossing the lowermost stretches of the São João River. Marine intrusions represent an additional threat to freshwater species, since freshwater species must share space and feeding resources with the large predatory invaders C. kelberi and C. gariepinus and with the generalist feeders H. littoralle, C. dimerus, M. lippincotianus along the embanked straight channel below the dam. In contrast to most marine species recorded herein (i.e., mugilids, engraulids, ariids), which are euryhaline and typical estuary inhabitants, most freshwater species are intolerant to brackish waters, and their displacements across coastal basins rely on floods of high magnitude, which are unusual in Southeast Brazil (Alvares et al., 2013). However, the marine ancestrality of the cichlids may confer a higher ability to withstand brackish waters. Among the non-native cichlids, only C. kelberi was found in the last meandering estuary. Further investigations regarding the salinity tolerance of this cichlid are highly desirable. As an emblematic coastal river where 24% of the freshwater fish fauna is composed of non-native species, the São João River might rapidly shift from recipient to a passive donor of propagules and imperil adjacent watersheds when invaders successfully overcome the chemical barrier represented by salinity.
Although non-native species are widely spread along the fluvio-marine ecotone of the São João River, only three out the 12 non-native species (the carnivorous C. kelberi, C. gariepinus and H. littorale) occur in all natural and hydrologically altered stretches. Rheophilic species were absent of the dammed, channelized, and drained floodplain stretches. There, most of the biomass catches correspond to the above-mentioned invaders, that due to their largest size within the fish assemblages and feeding habits represent an even greater risk for the native species. We conclude that hydrological alterations disrupted the lower São João River, which plays the role of a dispersion source for non-native species and where invasive species are favored.
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Abell, R., M. L. Thieme, C. Revenga, M. Bryer, M. Kottelat, N. Bogutskaya, B. Coad, N. Mandrak, S. C. Balderas, W. Bussing, M. L. J. Stiassny, P. Skelton, G. R. Allen, P. Unmack, A. Naseka, R. Ng, N. Sindorf, J. Robertson, E. Armijo, J. V. Higgins, T. J. Heibel, E. Wikramanayake, D. Olson, H. L. López, R. E. Reis, J. G. Lundberg, M. H. S. Pérez & P. Petry, 2008. Freshwater ecoregions of the world: a new map of biogeographic units for freshwater biodiversity conservation. BioScience 58(5): 403–414.
Abilhoa, V., R. R. Braga, H. Bornatowski & J. R. S. Vitule, 2011. Fishes of the Atlantic Rain Forest streams: ecological patterns and conservation. In Grillo, O. (ed.), Changing Diversity in Changing Environment. Intech, Rijeka: 259–282.
Agostinho, A. A., F. M. Pelicice & L. C. Gomes, 2008. Dams and the fish fauna of the Neotropical region: impacts and management related to diversity and fisheries. Brazilian Journal of Biology 68(4): 1119–1132.
Agostinho, A. A., L. C. Gomes, N. C. L. Santos, J. C. G. Ortega & F. M. Pelicice, 2016. Fish assemblages in Neotropical reservoirs: colonization patterns, impacts and management. Fisheries Research 173: 26–36.
Albert, J. S., V. A. Tagliacollo & F. Dagosta, 2020. Diversification of neotropical freshwater fishes. Annual Review of Ecology, Evolution, and Systematics 51: 27–53.
Alvares, C. A., J. L. Stape, P. C. Sentelhas, J. L. M. Gonçalves & G. Sparovek, 2013. Köppen’s climate classification map for Brazil. Meteorologische Zeitschrift 22(6): 711–728.
Amoros, C. & G. Bornette, 2002. Connectivity and biocomplexity in waterbodies of riverine floodplain. Freshwater Biology 47: 761–776.
Araújo, F. G., B. C. T. Pinto & T. P. Teixeira, 2009. Longitudinal patterns of fish assemblages in a large tropical river in southeastern Brazil: evaluating environmental influences and some concepts in river ecology. Hydrobiologia 618: 89–107.
Azevedo-Santos, V. M., P. M. Fearnside, C. S. Oliveira, A. A. Padial, F. M. Pelicice, D. P. Lima, D. Simberloff, T. E. Lovejoy, A. L. B. Magalhães, M. L. Orsi, A. A. Agostinho, F. A. Estaves, P. S. Pompeu, W. F. Laurance, M. Petrere Jr., R. P. Mormul & J. R. S. Vitule, 2017. Removing the abyss between conservation Science and policy decisions in Brazil. Biodiversity and Conservation 26: 1745–1752.
Barrella, W., A. G. Martins, M. Petrere Jr. & M. Ramires, 2014. Fishes of the southeastern Brazil Atlantic Forest. Environmental Biology of Fishes 97(12): 1367–1376.
Belliard, J., R. B. Thomas & D. Monnier, 1999. Fish communities and river alteration in the Seine Basin and nearby coastal streams. Hydrobiologia 400: 155–166.
Bizerril, C. R. F. S., 1995. Análise da distribuição da ictiofauna de uma bacia hidrográfica do leste brasileiro. Arquivos de Biologia e Tecnologia 38(2): 477–499.
Blaber, S. J. M., 2000. Tropical estuarine fishes: ecology, exploitation and conservation. Blackwell Science, Queensland.
Blaber, S. J. M., 2013. Fishes and fisheries in tropical estuaries: the last 10 years. Estuarine, Coastal and Shelf Science 135: 57–65.
Brauner, C. J., C. L. Ballantyne & D. J. Randall, 1995. Air breathing in the armoured catfish (Hoplosternum littorale) as an adaptation to hypoxic, acidic, and hydrogen sulphide rich waters. Canadian Journal of Zoology 73: 739–744.
Brito, M. F. G., A. L. B. Magalhães, D. P. Lima-Junior, F. M. Pelicice, V. M. Azevedo-Santos, D. A. Z. Garcia, A. M. Cunico & J. R. S. Vitule, 2018. Brazil naturalizes non-native species. Science 361(6398): 139.
Brookes, A., 1985. River channelization: traditional engineering methods, physical consequences and alternative practices. Progress in Physical Geography 9(1): 44–73.
Carvalho, T. L., E. A. Ferreira, F. M. Pelicice & R. Fernandes, 2020. Comparative functional responses predict the predatory impact of the highly invasive fish Cichla kelberi. Hydrobiologia. https://doi.org/10.1007/s10750-020-04440-6.
Casimiro, A. C. R., D. A. Z. Garcia, A. P. Vidotto-Magnoni, J. R. Britton, A. A. Agostinho, F. S. de Almeida & L. M. Orsi, 2018. Escapes of non-native fish from flooded aquaculture facilities: the case of Paranapanema River, Southern Brazil. Zoologia 35: e14638.
Catelani, P. A., A. C. Petry, F. Di Dario, V. L. M. Santos & M. M. Mincarone, 2014. Fish composition (Teleostei) of the estuarine region of the Macaé River, southeastern Brazil. Check List 10(4): 927–935.
Catelani, P. A., A. C. Petry, F. M. Pelicice & E. García-Berthou, 2021a. When a freshwater invader meets the estuary: the peacock bass and fish assemblages in the São João River, Brazil. Biological Invasions 23: 167–179.
Catelani, P. A., A. C. Petry, F. M. Pelicice & R. A. M. Silvano, 2021b. Fishers’ knowledge on the ecology, impacts and benefits of the non-native peacock bass Cichla kelberi in a coastal river in southeastern Brazil. Ethnobiology and Conservation 10: 04. https://doi.org/10.15451/ec2020-09-10.04-1-16.
CILSJ, 2002. Bacias Hidrográficas dos Rio São João e Rio das Ostras: águas, terras e conservação Ambiental. Consórcio Intermunicipal Lagos São João. Rio de Janeiro. http://www.oads.org.br/files/109.pdf.
Corbacho, C. & J. M. Sánchez, 2001. Patterns of species richness and introduced species in native freshwater fish faunas of a Mediterranean-type basin: the Guadiana River (southwest Iberian Peninsula). Regulated River: Research & Management 17: 699–707.
Daga, V. S., F. Skóra, A. A. Padial, V. Abilhoa, E. A. Gubiani & J. R. S. Vitule, 2015. Homogenization dynamics of the fish assemblages in Neotropical reservoirs: comparing the roles of introduced species and their vectors. Hydrobiologia 746: 327–347.
De Moraes, M. B., C. N. M. Polaz, E. P. Caramaschi, S. dos Santos Júnior, G. Souza & F. L. Carvalho, 2017. Espécies exóticas e alóctones da Bacia do Rio Paraíba do Sul: implicações para a conservação. Revista Biodiversidade Brasileira 7(1): 34–54.
Dudgeon, D., A. H. Arthington, M. O. Gessner, Z. I. Kawabata, D. J. Knowler, C. Lévêque, R. J. Naiman, A. H. Prieur-Richard, D. Soto, M. L. J. Stiassny & C. A. Sullivan, 2006. Freshwater biodiversity: importance, threats, status and conservation challenges. Biological Reviews 81: 163–182.
Espínola, L. A., C. V. Minte-Vera & H. F. Júlio Jr., 2009. Invasibility of reservoirs in the Paraná Basin, Brazil, to Cichla kelberi Kullander and Ferreira, 2006. Biological Invasions 12: 1873–1888.
Espínola, L. A., C. Minte-Vera & H. F. Júlio Junior, 2010. Invasibility of reservoirs in the Paraná Basin, Brazil, to Cichla kelberi Kullander and Ferreira, 2006. Biological Invasions 12: 1873–1888.
Ferreira, F. C. & M. P. Petrere Jr., 2009. The fish zonation of the Itanhaém river basin in the Atlantic Forest of southern Brazil. Hydrobiologia 636: 11–34.
Figueiredo J. L. & N. A. Menezes, 1978. Manual de Peixes Marinhos do Sudeste do Brasil. II. Teleostei (1). Museu de Zoologia da Universidade de São Paulo, São Paulo.
Figueiredo J. L. & N. A. Menezes, 1980. Manual de Peixes Marinhos do Sudeste do Brasil. III. Teleostei (2). Museu de Zoologia da Universidade de São Paulo, São Paulo.
Figueiredo J. L. & N. A. Menezes, 2000. Manual de Peixes Marinhos do Sudeste do Brasil. VI. Teleostei (5) Museu de Zoologia da Universidade de São Paulo, São Paulo.
Fitzgerald, D. B., M. Tobler & K. O. Winemiller, 2016. From richer to poorer: successful invasions by freshwater fishes depends on species richness of donor and recipient basins. Global Change Biology 22: 2440–2450.
Franco, A. C. S., L. N. dos Santos, A. C. Petry & E. García-Berthou, 2018. Abundance of invasive peacock bass increases with water residence time of reservoirs in southeastern Brazil. Hydrobiologia 817: 155–166.
Fricke, R., W. N. Eschmeyer & R. Van der Laan, 2020. Eschmeyer’s catalog of fishes: genera, species, references. http://researcharchive.calacademy.org/research/ichthyology/catalog/fishcatmain.asp). Electronic version. accessed 15 Jan 2020.
Garcia, D. A. Z., J. R. Britton, A. P. Vidotto-Magnoni & M. L. Orsi, 2018. Introductions of non-native fishes into a heavily modified river: rates, patterns and management issues in the Paranapanema River (Upper Paraná ecoregion, Brazil). Biological Invasions 20: 1229–1241.
Gomes, L. C. & L. E. Miranda, 2001. Riverine characteristics dictate composition of fish assemblages and limit fisheries in reservoirs of the Upper Paraná River Basin. Regulated Rivers: Research & Management 17: 67–76.
Jaramillo-Villa, U., 2010. Efeito da retificação de rios sobre as taxocenoses de peixes: estudo em rios costeiros de Mata Atlântica, no estado do Rio de Janeiro, Brasil. Master Thesis in Ecology. Universidade Federal do Rio de Janeiro.
Jurajda, P., 1995. Effect of channelization and regulation on fish recruitment in a flood plain river. Regulated Rivers: Restoration & Management 10: 207–215.
Kullander, S. O. & E. J. G. Ferreira, 2006. A review of the South American cichlid genus Cichla, with descriptions of nine new species (Teleostei: Cichlidae). Ichthyological Exploration of Freshwaters 17(4): 289–398.
Oksanen J. F., G. Blanchet, M. Friendly, R. Kindt, Legendre, P., D. McGlinn, P. R. Minchin, R. B. O’Hara, G. L. Simpson, P. Solymos, M. H. H. Stevens, E. Szoecs & H. Wagner, 2019. vegan: Community Ecology Package. R package version 2.5-6. https://CRAN.R-project.org/package=vegan.
Legendre, P. & E. D. R. Gallagher, 2001. Ecologically meaningful transformations for ordination of species data. Oecologia 129: 271–280.
Leprieur, F., O. Beauchard, S. Blanchet, T. Oberdorff & S. Brosse, 2008. Fish invasions in the world’s river systems: when natural processes are blurred by human activities. PLoS Biology 6(2): e28.
Liew, J. H., H. H. Tan & D. C. J. Yeo, 2016. Dammed rivers: impoundments facilitate fish invasions. Freshwater Biology 61(9): 1421–1429.
Marçal, M., G. Brierley & R. Lima, 2017. Using geomorphic understanding of catchment-scale process relationships to support the management of river futures: Macaé Basin, Brazil. Applied Geography 84: 23–41.
Mazzoni, R., N. Fenerich-Verani, E. P. Caramaschi & R. Iglesias-Rios, 2006. Stream-dwelling fish communities from an Atlantic Rain Forest drainage. Brazilian Archives of Biology and Technology 49(2): 249–256.
Mendonça, H. S., A. C. A. Santos, M. M. Martins & F. G. Araújo, 2018. Size-related and seasonal changes in the diet of the non-native Cichla kelberi Kullander & Ferreira, 2006 in a lowland reservoir in the southeastern Brazil. Biota Neotropica 18(3): e20170493.
Menezes N. A. & J. L. Figueiredo, 1980. Manual de Peixes Marinhos do Sudeste do Brasil. IV. Teleostei (3). Museu de Zoologia da Universidade de São Paulo, São Paulo.
Menezes N. A. & J. L. Figueiredo, 1985. Manual de Peixes Marinhos do Sudeste do Brasil. V. Teleostei (4). Museu de Zoologia da Universidade de São Paulo, São Paulo.
Mims, M. C. & J. D. Olden, 2013. Fish assemblages respond to altered flow regimes via ecological filtering of life history strategies. Freshwater Biology 58: 50–62.
Moulton, T. P., M. L. de Souza & A. F. de Oliveira, 2007. Conservation of catchments: some theoretical considerations and case histories from Rio de Janeiro. Neotropical Biology and Conservation 2(1): 28–35.
Nilsson, C., C. A. Reidy, M. Dynesius & C. Revenga, 2005. Fragmentation and flow regulation of the world’s large river systems. Science 308: 405–408.
Olden, J. D., M. J. Kennard, F. Leprieur, P. A. Tedesco, K. O. Winemiller & E. García-Berthou, 2010. Conservation biogeography of freshwater fishes: recent progress and future challenges. Diversity and Distributions 16: 496–513.
Orsi, M. L. & J. R. Britton, 2014. Long-term changes in the fish assemblage of a neotropical hydroelectric reservoir. Journal of Fish Biology 84: 1964–1970.
Palmer, M. W., 1993. Putting things in even better order: the advantages of canonical correspondence analysis. Ecology 74(8): 2215–2230.
Pelicice, F. M. & A. A. Agostinho, 2009. Fish fauna destruction after the introduction of non-native predator (Cichla kelberi) in a Neotropical reservoir. Biological Invasions 11: 1789–1801.
Pelicice, F. M., J. D. Latini & A. A. Agostinho, 2014. Fish fauna after the introduction of a vorascious predator: main drivers and the role of the invader’s demography. Hydrobiologia 746: 271–283.
Pelicice, F. M., V. M. Azevedo-Santos, J. R. S. Vitule, M. L. Orsi, D. P. Lima Junior, A. L. B. Magalhães, P. S. Pompeu, M. Petrere Jr. & A. A. Agostinho, 2017. Neotropical freshwater fishes imperilled by unsustainable policies. Fish and Fisheries 18(6): 1119–1133.
Pelicice, F. M., V. M. Azevedo-Santos, A. L. H. Esguícero, A. A. Agostinho & M. S. Arcifa, 2018. Fish diversity in the cascade of reservoirs along the Paranapanema River, southeast Brazil. Neotropical Ichthyology 16(2): e170150.
Pereira, L. S., A. A. Agostinho & K. O. Winemiller, 2017. Revisiting cannibalism in fishes. Reviews in Fish and Biology Fisheries 27: 499–513.
Poff, N. L., J. D. Olden, D. M. Merritt & D. M. Pepin, 2007. Homogenization of regional river dynamics by dams and global biodiversity implications. Proceedings of the National Academy of Sciences 104(14): 5732–5737.
Pool, T. K. & J. D. Olden, 2012. Taxonomic and functional homogenization of an endemic desert fish fauna. Diversity and Distributions 18: 366–376.
Quinn, G. P. & M. J. Keough, 2002. Experimental design and data analysis for biologists. Cambridge University Press, Cambridge.
R Core Team, 2017. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing, Vienna.
Rabelo, L. B. & L. S. H. Soares, 2014. Feeding interaction of the non-native african catfish (Clarias gariepinus Burchell, 1822) in Itanhaém River Estuary, Bahia, Brazil. Brazilian Journal of Oceanography 62(3): 179–186.
Simpson, G. L., 2019. Functions for generating restricted permutations of data. vs.09-5. https://github.com/gavinsimpson/permute.
Venables, W. N. & B. D. Ripley, 2002. Modern Applied Statistics with S, 4th ed. Springer, New York.
Vitule, J. R. S., F. Skóra & V. Abilhoa, 2012. Homogenization of freshwater fish faunas after the elimination of a natural barrier by a dam in Neotropics. Diversity and Distributions 18: 111–120.
Ward, J. V., K. Tockner & S. Schiemer, 1999. Biodiversity of floodplain river ecosystems: ecotones and connectivity. Regulated Rivers: Research & Management 15: 125–139.
Weyl, O. L. F., V. S. Daga, B. R. Ellender & J. R. S. Vitule, 2016. A review of Clarias gariepinus invasions in Brazil and South Africa. Journal of Fish Biology 89(1): 386–402.
Winemiller, K. O., P. B. McIntyre, L. Castello, E. Fluet-Chouinard, T. Giarrizzo, S. Nam, I. G. Baird, W. Darwall, N. K. Lujan, I. Harrison, M. L. J. Stiassny, R. A. M. Silvano, D. B. Fitzgerald, F. M. Pelicice, A. A. Agostinho, L. C. Gomes, J. S. Albert, E. Baran, M. Petrere Jr., C. Zarfl, M. Mulligan, J. P. Sullivan, C. C. Arantes, L. M. Sousa, A. A. Koning, D. J. Hoeinghaus, M. Sabaj, J. G. Lundberg, J. Armbruster, M. L. Thieme, P. Petry, J. Zuanon, G. Torrente Villara, J. Snoeks, C. Ou, W. Rainboth, C. S. Pavanelli, A. Akama, A. van Soesbergen & L. Sáenz, 2016. Balancing hydropower and biodiversity in the Amazon, Congo, and Mekong. Science 351(6269): 128–129.
Acknowledgements
This study was financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior—Brasil (CAPES)—Finance Code 001 and Fundação Carlos Chagas Filho de Amparo à Pesquisa do Estado do Rio de Janeiro (E-26/010.001930/2019). The authors are grateful to the reviewers and editor for their comments and suggestions. We are indebted to F.M. Pelicice for the valuable suggestions on the sampling design, to the several undergraduate and graduate students and technicians from the Universidade Federal do Rio de Janeiro, for their help in the fieldwork and processing of the samples, to M.P. Araújo for drawing the Fig. 1, to R. Marques for assistance in samplings on the reservoir, and to S.C. Pinto for the boat loan.
Funding
This study was financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior – Brasil (CAPES) – Finance Code 001 (VLMS and PAC) and Fundação Carlos Chagas Filho de Amparo à Pesquisa do Estado do Rio de Janeiro (E-26/010.001930/2019).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation and data collection were performed by Vagner Leonardo Macedo dos Santos and Paula Araujo Catelani and analyses by Vagner Leonardo Macedo dos Santos and Ana Cristina Petry. The first draft of the manuscript was written by Vagner Leonardo Macedo dos Santos and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The four authors acknowledge no conflict of interest.
Ethical approval
Collection permit SISBIO 45745. Committee of Ethics in Animal Experimentation of the Campus UFRJ-Macaé Professor Aloísio Teixeira MAC029.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Guest editors: Katya E. Kovalenko, Fernando M. Pelicice, Lee B. Kats, Jonne Kotta & Sidinei M. Thomaz / Aquatic Invasive Species III
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
dos Santos, V.L.M., Catelani, P.A., Petry, A.C. et al. Hydrological alterations enhance fish invasions: lessons from a Neotropical coastal river. Hydrobiologia 848, 2383–2397 (2021). https://doi.org/10.1007/s10750-021-04542-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-021-04542-9