Abstract
Human activities have disrupted the functioning of river ecosystems around the world. In the Amazon basin, hydropower expansion has affected diversity patterns, but no study has investigated how phytoplankton assemblages respond to impoundments. This study investigated the hypothesis that phytoplankton diversity is highly sensitive to river damming in large tropical rivers, declining in impoundment areas. We examined patterns of taxonomic diversity (i.e., richness, biovolume, and composition), functional diversity (Reynolds Functional Groups, RFGs), and functional redundancy (i.e., number of taxa in each RFG) along the Tocantins River, before and after the construction of Estreito Hydropower Dam. We monitored five sites along the river (ca. 260 km) between December 2009 and May 2013. The dam changed environmental conditions related to flow, nutrient supply, and depth. We recorded 140 taxa and 26 RFGs in the area. Taxa richness, biovolume, and the number of RFGs declined significantly after river regulation. Composition and abundance ranks changed over periods, and the codons C, Y, TC, W1, and LO were eliminated; functional redundancy declined significantly. These results show that phytoplankton diversity is sensitive to river regulation in large tropical rivers, where changes in hydrology and environmental filters lead to significant losses in taxonomic and functional diversity.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Human activities have degraded most freshwater ecosystems in the world, changing biodiversity patterns (Chapin et al., 2000; Dudgeon et al., 2005). The construction of large dams in tropical rivers, in particular, has been a major disturbance to aquatic communities (Agostinho et al., 2016; Grill et al., 2019). Dams have multiple negative effects because they change the hydrological regime, habitat distribution, and dispersion dynamics (Poff et al., 1997; Pelicice et al., 2015; Rahel & Mclaughlin, 2018). The expansion of hydropower plants in the Neotropical region is a matter of great concern, considering that most basins are severely regulated and new dams have been planned for the Amazon system (Winemiller et al., 2016).
Phytoplankton diversity is greatly affected by changes in hydrology, as the distribution of these organisms is regulated by local (e.g., light, nutrients, and herbivory) and regional filters (e.g., hydrological regime, dispersion, climate) (Margalef, 1978; Reynolds, 1999; Beyruth, 2000; Rodrigues et al., 2018; Pineda et al., 2020). Because they have a short generation time and respond quickly to disturbances (Reynolds, 1997), they can be used as a model to understand short-term impacts caused by river regulation. In fact, phytoplankton respond rapidly to environmental variation (Bicudo & Menezes, 2006; Naselli-Flores & Barone, 2007; Kruk et al., 2010), and previous studies have shown that impoundments impact phytoplankton diversity (Braun & Schagerl, 2010; Nogueira et al., 2010; Okuku et al., 2016). Reservoirs with lentic eutrophic conditions favor algae growth, dominance, and the occurrence of blooms (Lürling et al., 2018); in oligotrophic conditions, impoundments may impact negatively algal biomass and productivity (Teixeira de Oliveira et al., 2011; Moreti et al., 2013). However, little is known about how riverine assemblages respond to river damming in the Neotropical region, especially because few studies have compared pre and post periods (Souza et al., 2016). Most studies investigate algae diversity in the impoundment, especially in the Paraná River Basin (Henry, 1992; Tundisi & Matsumura-Tundisi, 2003; Soares et al., 2007; Rodrigues et al., 2015). No study has investigated this question in the megadiverse Amazon system, whose tributaries show different trophic states (Forsberg et al., 2017), including clear water systems.
Functional approaches constitute an important complimentary method to understand the interaction between diversity and disturbance (Mouillot et al., 2013), because niche dimensions determine organism’s responses to environmental filters (Keddy, 1992; Violle et al., 2007). Functional traits describe the bio-ecological functioning of the species (Violle et al., 2007), irrespective to its taxonomic identity and phylogenetic position. A relevant aspect is the fact that many species share traits, as a result of common ancestry or convergent evolution (Winemiller et al., 2015), allowing their classification into functional groups, i.e., a set of species (monophyletic or polyphyletic) that show similar ecological behavior. This aspect also creates functional redundancy in local assemblages, an important mechanism generating community stability and resilience against disturbances and species removal (Walker et al., 1999; Fonseca & Ganade, 2001). The functional approach can be applied to study freshwater phytoplankton, as these organisms show a diversity of traits related to floatability (e.g., cell size, shape and surface–volume ratio), motility (e.g., flagella and aerotopes), nutrient demands, and nitrogen fixation (e.g., heterocytes) (Naselli-Flores & Barone, 2011; Kruk & Segura, 2012; Naselli-Flores et al., 2020). In addition, several species share functional traits, allowing their classification into functional groups (Reynolds et al., 2002; Kruk et al., 2010; Salmaso et al., 2015; Naselli-Flores et al., 2020). Reynolds et al. (2002) proposed a broad classification for phytoplankton groups, which was reviewed by Reynolds (2006) and updated by Padisák et al. (2009). This classification proved to be useful, widely used in ecological studies and applicable to different types of environments worldwide (Kruk et al., 2017)—including impoundments (Dantas et al., 2012; Souza et al., 2016). This approach may reveal how phytoplankton diversity responds to river regulation, in terms of environmental tolerance, habitat characteristics, and trophic status (Reynolds, 2006; Padisák et al., 2009). Its application may improve predictions about population dynamics and species occurrence under regime-shift scenarios (i.e., lotic to lentic conditions; Kimmel et al., 1990).
In this sense, this work investigated the response of phytoplankton diversity to the regulation of a large tropical river. We investigated variations in taxonomic (i.e., taxa richness, biovolume and composition) and functional diversity (Reynolds Functional Groups—sensu Reynolds et al., 2002; Padisák et al., 2009) between periods that preceded and followed the construction of Estreito Hydropower Dam, Tocantins River, a clear water Amazonian system. Previous studies in this basin have reported the loss of fish diversity in impounded areas (Araújo et al., 2013; Lima et al., 2016; Perônico et al., 2019), and this work investigated the hypothesis that phytoplankton diversity is also sensitive to river regulation and declines in the impoundment, because riverine species depend on specific hydrological conditions (Poff et al., 1997), and riverine algae are adapted to live in flowing waters (Reynolds et al., 1994; Soares et al., 2007; Brasil & Huszar, 2011; Souza et al., 2016). Specifically, we investigated the following predictions: (i) changes in the hydrological regime negatively affect species richness, biovolume, and functional diversity, which decline in the impoundment; (ii) composition and biovolume ranks (species and functional groups) change in the impoundment, considering that these algae are adapted to lotic waters; and (iii) due to reductions in species richness, we expect decreased functional redundancy in the impoundment.
Materials and methods
Study area
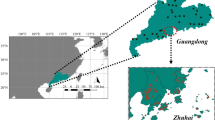
The Tocantins River is the main axis of the Tocantins-Araguaia hydrographic basin, the largest watershed located entirely within Brazilian territory, with approximately 918,273 km2 (10.8% of the country) (MMA—Ministério do Meio Ambiente & Secretaria de Recursos Hídricos, 2006). Savanna vegetation is the main biome (Cerrado), but Amazonian rainforests cover north and northwest regions. The Tocantins River is a clear water river (Sioli, 1984) with average discharge ranging between 13,624 m3/s in the rainy season and 2,412 m3/s in the dry season (ANA, 2009). The river flows through 1,960 km from its sources in central Brazil to the Marajó Island in the Lower Amazon, near the Atlantic Ocean. Irrigation, agriculture, and hydropower production are the main activities in the basin, which developed markedly during the last 20 years. The basin has the second largest hydroelectric potential installed in the country, with seven large dams on the main channel of the Tocantins River, and several small dams on tributaries (Winemiller et al., 2016).
The present study was carried out in the area affected by Estreito Hydropower Plant, Middle Tocantins River, the sixth dam along the cascade. Completed in 2011, the dam converted the main channel of the Tocantins River into a huge, lentic, and long impoundment (400 km2 and ca. 260 km long). It affected hydrological conditions, as minimum flows decreased from 2,412 m3 s−1 (monthly averages between 1931 and 2015; Brasil—Agência Nacional de Água—ANA, 2018) to 744 m3 s−1 (Brasil—Agência Nacional de Água—ANA, 2014). It is a run-of-the-river hydropower dam, with a retention time of 15 days (Pelicice et al., 2015). Large tributaries flow to the studied stretch, such as the Manuel Alves Grande, Manuel Alves Pequeno, and Farinha Rivers, which have economic and touristic relevance.
Sampling and laboratory analysis
Five sampling sites were established along the Tocantins River, between Tupiratins and Babaçulândia municipalities, Tocantins State (Fig. 1).These sites covered the stretch affected by the impoundment, in a linear distance of 138 km: one site in the lotic region upstream (S1—08° 23′ 22.4″ S—48° 06′ 38.6″ W), two in the transition region (S2—07° 51′ 29.6″ S–47° 55′ 17.0″ W and S3—07° 41′ 02.2″ S–47° 40′ 58.1′ W), and two located in the lacustrine region near the dam (S4—07° 20′ 08.5″ S–47° 28′ 32.0″ W and S5—07° 11′ 55.6″ S–47° 45′ 02.7″ W).
Phytoplankton and limnological variables were sampled at the subsurface (30 cm deep) in the central region of each sampling site. We sampled 4 years (December 2009–May 2013), quarterly during the pre-damming (PRE: December 2009–September 2010; N = 20 samples) and post-damming periods (POST: January 2012–May 2013; N = 20 samples), and monthly during the filling period (FILL: February–May 2011; N = 20 samples). In total, 60 samples were collected and analyzed. Phytoplankton samples were fixed in situ with acetic lugol. Taxa were identified under microscope to the lowest taxonomic level, based on morphological characteristics and specialized literature. Individuals (cells, colonies, or filaments) were counted randomly under inverted microscope (Lund et al., 1958; Utermöhl, 1958). Species biovolume was estimated by multiplying the number of individuals by mean unit volume (Sun & Liu, 2003).
Water temperature (°C), pH, dissolved oxygen (mg l−1), electrical conductivity (µS cm−1), and turbidity (NTU) were measured in situ using a multiparameter probe (YSI 6920). Maximum depth (Zmax − m) at each site was estimated using a 10-kg ballast attached to a graduated rope. Water samples were taken concomitantly with phytoplankton sampling to determine dissolved nutrients. In the laboratory, water samples were filtered through a pre-calcined Whatman GF/F filter and divided into two portions, one for immediate analysis (N-NH4+ and N-NH3) and the other was refrigerated for further analysis (P-PO4). Dissolved nitrogen (ammonium N-NH4+ and nitrite N-NH3 − mg l−1) was determined by the method of Koroleff, (1978) and Mackereth et al. (1978), respectively, using Cadmium treated with copper sulfate for the reduction. Total phosphorus (mg l−1) was determined by the colorimetric method after digestion with sulfuric acid (H2SO4) (Chapman, & Pratt, 1961); the determination of inorganic phosphorus (P-PO4) (mg l−1) followed Golterman et al. (1978).
Classification into Reynolds Functional Groups (RFGs)
Species that summed more than 5% of total biovolume were grouped into Reynolds Functional Groups (RFGs). Taxa were classified in codons based on information provided by Reynolds et al. (2002) and Padisák et al. (2009). The number of RFGs was considered as a measure of functional diversity (FD) (Reynolds et al., 2002), and the number of taxa in each RFGs as measure of functional redundancy (FR) (Fonseca & Ganade, 2001).
Data analysis
The euphotic zone (Zeu, m) was calculated by multiplying water transparency (Secchi depth, m) by 2.7 (Cole & Weihe, 2016). The mixing zone (Zmix, m) was calculated based on the thermal profile; the ratio between the euphotic and mixing depths (Zeu:Zmix) was used as a measure of light availability in the mixing zone (Jensen et al., 1994).
All analyses focused on environmental conditions and phytoplankton diversity (taxonomic and functional) considering periods (PRE, FILL, and POST) and sites (S1–S5) as factors.
To characterize the environmental conditions, we calculated mean values and standard deviation for each abiotic variable. Principal Component Analysis (PCA) was also applied to investigate spatial and temporal gradients. For this analysis, we considered the following variables: ammonium, nitrogen ammonia, conductivity, inorganic phosphorus, total phosphorus, dissolved oxygen, pH, water temperature, turbidity, total depth, euphotic zone, and mixing zone. All variables, except pH, were log-transformed.
To investigate the first prediction (i.e., loss of taxa richness, biovolume, and FD after river regulation), we used two-way Analysis of Variance to test the effect of sites, periods, and their interaction on taxa richness, biovolume, and FD. Taxa richness is the number of taxa, and FD is the number of functional groups in each sample. Tukey’s test compared means among groups.
To investigate the second prediction (i.e., changes in composition and biovolume ranks), we examined temporal variations in composition and dominance patterns among taxa and functional groups. A Non-Metric Multidimensional Scaling (NMDS) analysis was applied to analyze variations in taxonomic and functional structure (composition and biovolume), based on Bray–Curtis similarity. The effect of period on assemblage structure was tested with one-way Analysis of Similarity (ANOSIM). We also calculated abundance ranks based on the distribution of biovolume among taxa and RFGs, comparing the rank observed in PRE (reference) with the others periods.
To test the third prediction (i.e., lower functional redundancy (FR) after river regulation), we evaluated temporal variations in the number of taxa within each functional group. A one-way ANOVA was used to test the effect of period on FR.
All analyses were performed using the software Past version 2.16 (Hammer et al., 2001) and Statistica version 7.1 (StatSoft, 2005). Statistical differences implied P < 0.05.
Results
Environmental conditions
Most abiotic variables (temperature, pH, oxygen, turbidity, and conductivity) showed little variation over periods (Fig. 2). Light availability was low across sampling sites (Zeu:Zmix < 1) in the POST period. The ratio between the euphotic zone and maximum depth decreased in the reservoir. Mean depth increased after river regulation, especially at sites near the dam (Online Resource 2). The water column was not stratified throughout the study period.
Environmental conditions along the Tocantins River (sites S1–S5), before (PRE), during (FILL), and after (POST) the construction of Estreito Hydropower Dam. Mean values and standard deviation are shown. Variables: NH4+ (ammonium mg l−1), NO3 (nitrate mg l−1), Cond (conductivity µS cm−1), P-PO4 (inorganic phosphorus mg l−1), PT (total phosphorus mg l−1), OD (dissolved oxygen mg l−1), pH, Temp (water temperature °C), Turb (turbidity NTU), Zmax (maximum depth, m), Zeu (euphotic zone, m), and Zmix (mixing zone, m)
Considering the trophic status, ammonium (N-NH4+) concentration was higher during the PRE period, declining markedly after river regulation. Total and inorganic phosphorus (P-PO4) showed a weak increase in the FILL and POST periods (Fig. 2).
The first two axes of the Principal Component Analysis (PCA) explained 86% of data variability (Table 1). The first axis summarized a strong temporal gradient, separating samples according to each period (Fig. 3). This axis was affected by N-NH4+, oxygen, NO3, and turbidity, which correlated with PRE samples. Other variables correlated with FILL and POST samples, such as PT, PO4, Zmax, Zmix, and pH. The second axis summarized a limnological gradient affected mainly by conductivity, Zeu and NO3, but it was not related to temporal gradients (Fig. 3).
Principal component analysis (PCA) applied to investigate variation in environmental conditions along the Tocantins River (sites S1–S5), before (PRE), during (FILL) and after (POST) the construction of Estreito Hydropower Dam. Variables: electrical conductivity (Cond.); total depth (Zmax.); euphotic zone (Zeu); mixing zone (Zmix); ammonium (N-NH4+.); nitrate (N-NO3); dissolved oxygen (DO); total phosphorus (PT); inorganic phosphorus (P-PO4); water temperature (Temp.); turbidity (Turb.)
Taxonomic and functional diversity
We recorded 140 taxa (Online Resource 1) and 26 RFGs (Table 2). The PRE period was the most diverse with 117 taxa and 23 RFGs; the FILL period had 40 taxa and 17 RFGs, and the POST 37 taxa and 14 RFGs.
We observed higher values of taxa richness, biovolume, and functional diversity (number of RFGs) before river regulation, with little variation among sites (Fig. 4). Biovolume, in particular, showed a sharp decrease in the impoundment. ANOVA results confirmed that these metrics differed significantly over periods, with no significant effect of Site and the interaction Period x Site (Table 3). Tukey’s test indicated that PRE samples differed from other periods, with higher values.
Variations in the structure of phytoplankton along the Tocantins River, before (PRE), during (FILL), and after (POST) the construction of Estreito Hydropower Dam. Taxa richness, functional diversity (number of RFGs), and biovolume (mm3 ml−1). Symbols are mean values and bars represent the standard error
Taxa abundance changed over periods, considering that biovolume ranks showed different patterns (Fig. 5). Diatoms (especially Aulacoseira ambigua (Grunow) Simonsen − 29% of total biovolume) dominated in the PRE, but they declined after river regulation. Actually, all taxa showed low biovolume values during the FILL and POST periods; Raphidiopsis raciborskii (formerly Cylindrospermopsis raciborskii) (Wołoszyńska) Aguilera, Berrendero Gómez, Kastovsky, Echenique & Salerno (36%), and Aulacoseira granulata var. granulata (Ehrenberg) Simonsen (31%) dominated in the FILL, while Aulacoseira sp. (16%), Staurastrum sp. (15%), Trachelomonas armata (Ehrenberg) F. Stein (14%), Raphidiopsis raciborskii (13%), and A. granulata var. granulata (12%) were the main taxa in the POST.
Biovolume ranks among RFGs also changed over periods (Fig. 6). Codons C (30%), P (18%), and W2 (12%) showed greater biovolume in the PRE period, while Sn (39%) and P (34%) dominated during the FILL. In the POST period, the main codons were P (30%), N (18%), W2 (15%), and Sn (13%). Groups C, Y, TC, W1, and LO were exclusive to the PRE period.
Non-Metric Multidimensional Scaling (NMDS) indicated temporal changes in taxonomic (stress = 0.107; Fig. 7a) and functional structure (stress = 0.097; Fig. 7b). In both cases, greater dissimilarity was found between PRE and POST periods. ANOSIM confirmed significant differences among all periods for the taxonomic (R = 0.98; P < 0.0001) and functional structure (R = 1; P < 0.0001).
Non-metric multidimensional scaling (NMDS) applied to investigate variations in taxonomic (a) and functional (b) composition in phytoplankton assemblages along the Tocantins River, before (PRE), during (FILL), and after (POST) the construction of Estreito Hydropower Dam. The line connecting sites represents the river corridor (S1 upstream; S5 downstream)
Functional redundancy declined over periods (Fig. 8) for the most redundant RFGs. ANOVA results confirmed that the mean number of taxa/RFGs was higher in the PRE period (F = 4.75; P < 0.01). Many RFGs recorded in the PRE were not registered in the POST period.
Discussion
The construction of Estreito Hydropower Dam caused the loss of phytoplankton taxonomic and functional diversity along the Tocantins River, a clear water tributary of the Amazon system. In general, total biovolume, taxa richness, and the number of functional groups declined consistently after river regulation, confirming the prediction that changes in hydrology negatively impact algae diversity. We also detected strong changes in composition and abundance ranks, confirming the prediction that communities are reassembled in the impoundment. Finally, functional redundancy declined, confirming the prediction that RFGs become less diverse in the reservoir. Taken together, these results show how algae diversity respond to river damming in the Amazon river system, and support the hypothesis that riverine phytoplankton, a group poorly known in the region (de Cunha et al., 2013), is highly sensitive to changes in hydrology. Previous studies have shown dramatic changes in fish diversity after river regulation (Lima et al., 2016; Perônico et al., 2019), and our study confirmed that phytoplanktonic diversity (taxonomic and functional) is also deeply impacted by the impoundment.
Large dams impact the functioning of river ecosystems, because the hydrological regime, sediment loads, and local environmental conditions are permanently changed (Poff et al., 1997). This impact is pronounced in tropical regions (Turgeon et al., 2019), where a new community state may emerge rapidly (Perônico et al., 2019). Estreito Hydropower Dam created a large lentic impoundment (400 km2 and ca. 260 km long) in the main channel of the Tocantins River, inducing significant reductions to river discharge. Limnological conditions were also affected, as ammonia (NH4+) declined considerably and the water column expanded (depth increased). It probably affected nutrient supply for algae, and may explain the sharp decrease in biovolume, taxa richness, and functional groups in the impoundment. However, we observed weak environmental gradients along the reservoir. The formation of zones or environmental gradients is a common pattern in large Neotropical impoundments (Agostinho et al., 2008). Considering that sites close to the dam (S4 and S5) showed particular conditions after river regulation (Online Resource 1), it is possible that longitudinal differences emerge in next years (i.e., reservoir aging; Agostinho et al., 1999). The expansion of human activities in the region may also change limnological conditions in the impoundment, particularly the input of pollutants and nutrients from urban centers and agriculture (Trinh et al., 2016), with effects on phytoplankton diversity and productivity (Reynolds, 2006).
Impoundments change environmental filters and select for novel aquatic communities (Pringle et al., 2000; Arantes et al., 2019). In Neotropical rivers, the lentic zone of large impoundments is shallow and vast, with no water flow and high retention times (Pelicice et al., 2015; Agostinho et al., 2016). These conditions are unfavorable for the riverine biota, which rely on the natural flow regime, fluvial habitats, and nutrient dynamics to persist (Pringle et al., 2000). The prominent decline in algae diversity indicates that the impoundment was unfavorable for most taxa, pointing to the action of severe filters, probably related to the absence of flow, reduced nitrogen, and light conditions. In flowing rivers, turbulence, light, and nutrients are the main drivers of algae diversity and growth (Soares et al., 2007; Barbosa & Chícharo, 2012; Domingues et al., 2012). The impoundment probably redistributed phytoplankton in the water column, as adjacent areas were permanently flooded and water level increased. In addition, nitrogen deficiency may have imposed severe constraints on algae growth, as many taxa are sensitive to variations in nutrient supply (Padisák et al., 2009). The lower retention time, in particular, may play a crucial role, because some algae rely on the water flow to persist in the column. When flow is reduced, they probably sink to deep layers where solar radiation is limited. It might be the case of the diatom Aulacoseira ambigua, which declined and disappeared after river regulation. These species, among other diatoms, are biological indicators of physical changes in the environment, being associated with turbulent waters (Tilman et al., 1982; Costa-Böddeker et al., 2012; Souza et al., 2016), as they have siliceous cell walls that protect against abrasions (Reynolds et al., 2002; Hamm et al., 2003). This siliceous wall, however, may increase sedimentation losses in lentic waters (Margalef, 1978; Reynolds et al., 2002; Padisák et al., 2003).
Variations in the biomass of RFGs illustrate how phytoplankton algae respond to the disturbance. For example, in a small reservoir in south Brazil (Souza et al., 2016), the RFGs responded either positively or negatively to the impoundment. In our study, however, all RFGs were equally impacted and declined in the impoundment. Codon C (diatoms) was the most affected and disappeared from the impoundment, probably because it needs more eutrophic conditions. A similar pattern was observed by the study mentioned above (Souza et al., 2016), where codon C dominated riverine assemblages, but declined in the reservoir. Codon J (non-mobile Chlorophycea) was another highly impacted group, probably because it is commonly associated with enriched systems and clear waters (Padisák et al., 2009). In the reservoir, the main codons were P (diatoms and Zygnemaphycea), N (Zygnemaphycea), Sn (nitrogen fixers cyanobacteria), and W2 (Euglenophycea), although they showed low biovolume. Some of these groups are sensitive to nutrient limitation, column stratification, and alkaline conditions (i.e., P and N), while others usually inhabit shallow mesotrophic lakes (i.e., W2) (Reynolds et al., 2002). These groups may sustain low biomass in Estreito Reservoir while present conditions persist. If eutrophication occurs, some groups may develop and dominate, particularly Sn (Raphidiopsis raciborskii), the most adapted to tropical eutrophic conditions. This group is associated with warm, well-mixed environments (Padisák et al., 2009), rich in nutrients and tolerant to vertical mixing (Reynolds et al., 2002).
The presence and persistence of the RFGs in the system depend not only on population dynamics. The functional redundancy is another crucial attribute, and may explain the persistence of specific groups. For example, the most redundant RFGs in the river environment were J, F, N, MP, and P, but redundancy reduced significantly in the impoundment, although none of these groups were eliminated. On the other hand, groups with few species in the PRE period (e.g., TB, A, Y, H1, S2) disappeared after river regulation. Redundancy, in this case, might have increased the persistence of certain groups, an important mechanism (insurance effect; Johnson et al., 1996) against the disturbance. It is able to maintain ecosystem functions or functional diversity when species are lost and composition is changed (Walker et al., 1999; Hooper et al., 2005). However, overall functional redundancy decreased after the disturbance, with most groups summing less than two species. Therefore, the community became vulnerable to new disturbances or to stochastic variation (demographic or environmental).
In fluvial systems, phytoplankton evolved in a lotic environment, controlled by hydraulic, limnological, and sedimentological factors (Margalef, 1978; Soares et al., 2007; Naselli-flores et al., 2020). The present study provided strong evidence that algae are very sensitive to changes in riverine conditions, because the construction of Estreito Dam reduced taxonomic and functional diversity in the Tocantins River. The loss of algae diversity is concerning because these organisms generate a number of ecosystem functions and services, particularly primary and oxygen production. Changes in producers induce bottom-up effects across the food web, affecting direct consumers (e.g., zooplankton, insect larvae, fish) and upper trophic levels (Scheffer et al., 1993). We emphasize that phytoplankton algae support food webs in some systems, including fish biomass in tropical river systems (Benedito-Cecilio et al., 2000), in addition to regulating global atmospheric and climate regimes (Falkowski, 2012). Algae diversity is also closely related to human health, since environmental degradation may elicit algal blooms, including toxic cyanobacteria (Carmichael, 1997; Errera & Campbell, 2011; Lürling et al., 2018). We predict that the installation of future hydroelectric plants in clear water rivers in the Amazon system will cause the loss of phytoplankton diversity, with risk of inducing blooms of noxious species and bottom-up effects on the structure and functioning of the system.
References
Agostinho, A. A., L. E. Miranda, L. M. Bini, L. C. Gomes, S. M. Thomaz & H. I. Suzuki, 1999. Patterns of colonization in Neotropical reser; and prognoses on aging. In Tundisi, J. G. & M. Straskraba (eds), Theoretical Reservoir Ecology and its Application. International Institute of Ecology, Oldendorf: 227–265.
Agostinho, A. A., F. M. Pelicice & L. C. Gomes, 2008. Dams and the fish fauna of the Neotropical region: impacts and management related to diversity and fisheries. Brazilian Journal of Biology = Revista brasleira de biologia 68: 1119–1132.
Agostinho, A. A., L. C. Gomes, N. C. L. Santos, J. C. G. Ortega & F. M. Pelicice, 2016. Fish assemblages in Neotropical reservoirs: Colonization patterns, impacts and management. Fisheries Research 173: 26–36.
ANA, 2009. Plano estratégico de recursos hídricos da bacia hidrográfica dos rios Tocantins e Araguaia : relatório síntese. ANA, Brasília.
Arantes, C. C., D. B. Fitzgerald, D. J. Hoeinghaus & K. O. Winemiller, 2019. ScienceDirect Impacts of hydroelectric dams on fishes and fisheries in tropical rivers through the lens of functional traits. Current Opinion in Environmental Sustainability Elsevier BV 37: 28–40.
Araújo, E. S., E. E. Marques, I. S. Freitas, A. L. Neuberger, R. Fernandes & F. M. Pelicice, 2013. Changes in distance decay relationships after river regulation: Similarity among fish assemblages in a large Amazonian river. Ecology of Freshwater Fish 22: 543–552.
Barbosa, A. B. & M. A. Chícharo, 2012. Hydrology and Biota Interactions as Driving Forces for Ecosystem Functioning. Treatise on Estuarine and Coastal Science. Elsevier Inc., Waltham. https://doi.org/10.1016/B978-0-12-374711-2.01002-0.
Benedito-Cecilio, E., B. R. Forsberg & L. C. Martinelli, 2000. Carbon sources of Amazonian fisheries. Fisheries Management and Ecology 7: 305–315.
Beyruth, Z., 2000. Periodic disturbances, trophic gradient and phytoplankton characteristics related to cyanobacterial growth in Guarapiranga Reservoir, São Paulo State, Brazil. Hydrobiologia 424: 51–65.
Brasil, J., & V. L. M. Huszar, 2011. O papel dos traços funcionais na ecologia do fitoplâncton continental. Oecologia Australis 15: 799–834.
Brasil - Agência Nacional de Água - ANA, 2014. Manual de Procedimentos da Operação- Módulo 10 - Submódulo 1021. ANA, Brasilia.
Brasil - Agência Nacional de Água - ANA, 2018. Boletim diário de monitoramento da bacia do rio Paraíba do Sul. Agência Nacional das Águas., https://www.ana.gov.br/sala-de-situacao/tocantins/colecao-para-boletim-diario-do-tocantins?b_start:int=510.
Braun, B. & M. Schagerl, 2010. Algae-environment relationships in an impoundment stretch of the River Grosse Erlauf (Austria). River Systems 19: 3–13.
Carmichael, W. W., 1997. The Cyanotoxins Advances in Botanical Research. Academic Press, London.
Chapin, F. S., E. S. Zavaleta, V. T. Eviner, R. L. Naylor, P. M. Vitousek, H. L. Reynolds, D. U. Hooper, S. Lavorel, O. E. Sala, S. E. Hobbie, M. C. Mack & S. Díaz, 2000. Consequences of changing biodiversity. Nature 405: 234–242.
Chapman, H. D. & P. F. Pratt, 1961. Plant analysis. Methods of analysis for soils, plants and waters. Division of Agricultural Sciences, Davis, CA.
Cole, G. A. & P. E. Weihe, 2016. Textbook of Limnology. Waveland Press, Long Grove, IL.
Costa-Böddeker, S., H. Bennion, T. A. de Jesus, A. L. S. Albuquerque, R. C. L. Figueira & D. C. de Bicudo, 2012. Paleolimnologically inferred eutrophication of a shallow, tropical, urban reservoir in southeast Brazil. Journal of Paleolimnology 48: 751–766.
De Bicudo, C. E. M. & M. Meneze, 2006. Gêneros de algas de águas continentais do Brasil (chave para identificação e descrições). RIMA Editora, São Carlos.
de Cunha, E. D. S., A. C. da Cunha, A. M. da Silveira Junior & S. M. M. Faustino, 2013. Phytoplankton of two rivers in the eastern amazon: characterization of biodiversity and new occurrences. Acta Botanica Brasilica 27: 364–377.
Domingues, R. B., A. B. Barbosa, U. Sommer & H. M. Galvão, 2012. Phytoplankton composition, growth and production in the Guadiana estuary (SW Iberia): Unraveling changes induced after dam construction. Science of the Total Environment Elsevier B.V. 416: 300–313.
Dudgeon, D., A. H. Arthington, M. O. Gessner, Z. I. Kawabata, D. J. Knowler, C. Lévêque, R. J. Naiman, A. H. Prieur-Richard, D. Soto, M. L. J. Stiassny & C. A. Sullivan, 2005. Freshwater biodiversity: importance, threats, status and conservation challenges. Biological Reviews of the Cambridge Philosophical Society 81: 163–182.
ênio Dantas, W., M. C. Bittencourt-Oliveira & A. N. do Moura, 2012. Dynamics of phytoplankton associations in three reservoirs in northeastern Brazil assessed using Reynolds’ theory. Limnologica 42: 72–80.
Errera, R. M. & L. Campbell, 2011. Osmotic stress triggers toxin production by the dinoflagellate Karenia brevis. Proceedings of the National Academy of Sciences 109: 17723–17724.
Falkowski, P., 2012. The power of plankton. Nature 483: 7–10.
Fonseca, C. R. & G. Ganade, 2001. Species functional redundancy, random extinctions and the stability of ecosystems. Journal of Ecology. https://doi.org/10.1046/j.1365-2745.2001.00528.x.
Forsberg, B. R., J. M. Melack, T. Dunne, R. B. Barthem, M. Goulding, R. C. D. Paiva, M. V. Sorribas, U. L. Silva & S. Weisser, 2017. The potential impact of new Andean dams on Amazon fluvial ecosystems. PLoS ONE 12(8): e0182254.
Golterman, H. L., R. S. Clymo & M. A. M. Ohnstad, 1978. Methods for physical and chemical analysis of freshwater. Blackwell Scientific Publications, Oxford.
Grill, G., B. Lehner, M. Thieme, B. Geenen, D. Tickner, F. Antonelli, S. Babu, P. Borrelli, L. Cheng, H. Crochetiere, H. Ehalt Macedo, R. Filgueiras, M. Goichot, J. Higgins, Z. Hogan, B. Lip, M. E. McClain, J. Meng, M. Mulligan, C. Nilsson, J. D. Olden, J. J. Opperman, P. Petry, C. Reidy Liermann, L. Sáenz, S. Salinas-Rodríguez, P. Schelle, R. J. P. Schmitt, J. Snider, F. Tan, K. Tockner, P. H. Valdujo, A. van Soesbergen & C. Zarfl, 2019. Mapping the world’s free-flowing rivers. Nature Springer, US 569: 215–221.
Hamm, C. E., R. Merkel, O. Springer, P. Jurkojc, C. Maier, K. Prechtel & V. Smetacek, 2003. Architecture and material properties of diatom shells provide effective mechanical protection. Nature 421: 841–843.
Hammer, Ø., D. A. T. Harper & P. D. Ryan, 2001. PAST: paleontological statistics software package for education and data analysis. Palaeontologia Electronica 4(1): 9.
Henry, R., 1992. The Oxygen deficit in Jurumirim (Paranapanema Reservoir. Japanese Journal of Limnology 53: 379–384.
Hooper, D. U., I. F. S. Chapin, J. J. Ewel, A. Hector, P. Inchausti, S. Lavorel, J. H. Lawton, D. M. Lodge, M. Loreau, S. B. Naeem, B. Schmid, H. Setala, A. J. Symstad, J. Vandermeer & D. A. Wardle, 2005. Effects of Biodiversity on Ecosystem Functioning: a Consensus of Current. Ecological Monographs 75: 3–35.
Jensen, J. P., E. Jeppesen, K. Blrik, & P. Kristensen, 1994. Impact of Nutrients and Physical Factors on the Shift from Cyanobacterial to Chlorophyte Domiance in Shallow Danish Lakes. Canadian Journal of Fisheries and Aquatic Sciences 51: 1692–1699.
Johnson, K. H., K. A. Vogt, H. J. Clark, O. Schmitz & D. Vogt, 1996. Resilience and stability of ecosystems. Trends in Ecology & Evolution 11: 372–377.
Keddy, P. A., 1992. A pragmatic approach to functional ecology. Functional Ecology 6: 621–626.
Kimmel, B. L., O. T. Lind & L. J. Paulson, 1993. Reservoir primary production. In Thornton, K. W., B. L. Kimmel & F. E. Payne (eds), Reservoir Limnology: Ecological Perspectives. Wiley, New York: 246.
Koroleff, K., 1978. Determination of ammonia. In Grasshoff, K. & E. Kremling (eds), Methods of Seawater Analysis. Verlag Chemie, Winhein.
Kruk, C. & A. M. Segura, 2012. The habitat template of phytoplankton morphology-based functional groups. Hydrobiologia 698: 191–202.
Kruk, C., V. L. M. Huszar, E. T. H. M. Peeters, S. Bonilla, L. Costa, M. LüRling, C. S. Reynolds & M. Scheffer, 2010. A morphological classification capturing functional variation in phytoplankton. Freshwater Biology 55: 614–627.
Kruk, C., M. Devercelli, V. L. M. Huszar, E. Hernández, G. Beamud, M. Diaz, L. H. S. Silva & A. M. Segura, 2017. Classification of Reynolds phytoplankton functional groups using individual traits and machine learning techniques. Freshwater Biology 62: 1681–1692.
Lima, A. C., C. S. Agostinho, D. Sayanda, F. M. Pelicice, A. M. V. M. Soares & K. A. Monaghan, 2016. The rise and fall of fish diversity in a neotropical river after impoundment. Hydrobiologia 763: 207–221.
Lund, J., C. Kipling & E. Le Cren, 1958. The inverted microscope method of estimating algal numbers and the statistical basis of estimations by counting. Hydrobiologia 11: 143–170.
Lürling, M., M. M. Mello, F. van Oosterhout, L. S. de Domis & M. M. Marinho, 2018. Response of natural cyanobacteria and algae assemblages to a nutrient pulse and elevated temperature. Frontiers in Microbiology 9: 1–14.
Mackereth, F. Y. H., J. Heron & J. J. Talling, 1978. Water analysis: some revised methods for limnologist. Freshwater Biology Association, Scientific Publication, Ambleside.
Margalef, R., 1978. Life-forms of phytoplankton as survival alternatives in an unstable environment. Oceanologica Acta 1: 493–509.
MMA - Ministério do Meio Ambiente, & Secretaria de Recursos Hídricos., 2006. Caderno da região hidrográfica Tocantins - Araguaia..
Moreti, L. O. R., L. Martos, V. M. Bovo-Scomparin & L. C. Rodrigues, 2013. Variação espacial e temporal dos grupos funcionais do fitoplâncton em um reservatório tropical. Acta Scientiarum - Biological Sciences 35: 359–366.
Mouillot, D., N. A. J. Graham, S. Villéger, N. W. H. Mason & D. R. Bellwood, 2013. A functional approach reveals community responses to disturbances. Trends in Ecology and Evolution 28: 167–177.
Naselli-Flores, L. & R. Barone, 2007. Pluriannual morphological variability of phytoplankton in a highly productive Mediterranean reservoir (Lake Arancio, Southwestern Sicily). Hydrobiologia 578: 87–95.
Naselli-Flores, L. & R. Barone, 2011. Fight on Plankton! or, Phytoplankton Shape and Size as Adaptive Tools to Get Ahead in the Struggle for Life. Cryptogamie, Algologie 32: 157–204.
Naselli-Flores, L., T. Zohary & J. Padisák, 2020. Life in suspension and its impact on phytoplankton morphology: an homage to Colin S. Reynolds. Hydrobiologia. https://doi.org/10.1007/s10750-020-04217-x.
Nogueira, M. G., M. Ferrareze, M. L. Moreira & R. M. Gouvêa, 2010. Phytoplankton assemblages in a reservoir cascade of a large tropical – subtropical river (SE, Brazil). Brazilian Journal of Biology Revista brasleira de biologia 70: 781–793.
Okuku, E. O., M. Tole, L. I. Kiteresi & S. Bouillon, 2016. The response of phytoplankton and zooplankton to river damming in three cascading reservoirs of the Tana River, Kenya. Lakes and Reservoirs: Research and Management 21: 114–132.
Padisák, J., G. Borics, G. Fehér, I. Grigorszky, I. Oldal, A. Schmidt & Z. Zámbóné-Doma, 2003. Dominant species, functional assemblages and frequency of equilibrium phases in late summer phytoplankton assemblages in Hungarian small shallow lakes. Hydrobiologia 502: 157–168.
Padisák, J., L. O. Crossetti & L. Naselli-Flores, 2009. Use and misuse in the application of the phytoplankton functional classification: a critical review with updates. Hydrobiologia 621: 1–19.
Pelicice, F. M., P. S. Pompeu & A. A. Agostinho, 2015. Large reservoirs as ecological barriers to downstream movements of Neotropical migratory fish. Fish and Fisheries 16: 697–715.
Perônico, P. B., C. S. Agostinho, R. Fernandes & F. M. Pelicice, 2019. Community reassembly after river regulation: rapid loss of fish diversity and the emergence of a new state. Hydrobiologia 847: 519–533.
Pineda, A., P. Iatskiu, S. Jati, A. C. M. Paula, B. F. Zanco, C. C. Bonecker, G. A. Moresco, L. A. Ortega, Y. R. Souza, & L. C. Rodrigues, 2020. Damming reduced the functional richness and caused the shift to a new functional state of the phytoplankton in a subtropical region. Hydrobiologia. https://doi.org/10.1007/s10750-020-04311-0.
Poff, N. L., J. D. Allan, M. B. Bain, J. R. Karr, K. L. Prestegaard, D. Brian, R. E. Sparks, J. C. Stromberg, N. L. Poff, J. D. Allan, M. B. Bain, J. R. Karr, K. L. Prestegaard, B. D. Richter, R. E. Sparks & J. C. Stromberg, 1997. A paradigm for river conservation and restoration. BioScience 47: 769–784.
Pringle, C. M., M. C. Freeman & B. J. Freeman, 2000. Regional effects of hydrologic alterations on riverine Macrobiota in the New World: Tropical – Temperate Comparisons. BioScience 50: 807–823.
Rahel, F. J. & R. L. Mclaughlin, 2018. Selective fragmentation and the management of fish movement across anthropogenic barriers. Ecological Applications 28: 2066–2081.
Reynolds, C. S., 1997. Vegetation processes in the pelagic: a model for ecosystem theory. Journal of the Marine Biological Association of the United Kingdom. 77: 919.
Reynolds, C. S., 1999. Modelling phytoplankton dynamics and its application to lake management. Hydrobiologia 396: 123–131.
Reynolds, C. S., 2006. The Ecology of Phytoplankton. Cambridge University Press, New York.
Reynolds, C. S., J. P. Descy & J. Padisák, 1994. Are phytoplankton dynamics in rivers so different from those in shallow lakes? Hydrobiologia 289: 1–7.
Reynolds, C. S., V. Huszar, C. Kruk, L. Naselli-Flores, & S. Melo, 2002. Towards a functional classification of the freshwater phytoplankton. Journal of Plankton Research. 24: 417–428, http://plankt.oxfordjournals.org/cgi/content/abstract/24/5/417.
Rodrigues, L. C., N. R. Simões, V. M. Bovo-Scomparin, S. Jati, N. F. Santana, M. C. Roberto & S. Train, 2015. Phytoplankton alpha diversity as an indicator of environmental changes in a neotropical floodplain. Ecological Indicators Elsevier Ltd 48: 334–341.
Rodrigues, L. C., B. M. Pivato, L. C. G. Vieira, V. M. Bovo-Scomparin, J. C. Bortolini, A. Pineda & S. Train, 2018. Use of phytoplankton functional groups as a model of spatial and temporal patterns in reservoirs: a case study in a reservoir of central Brazil. Hydrobiologia 805: 147–161.
Salmaso, N., L. Naselli-Flores & J. Padisák, 2015. Functional classifications and their application in phytoplankton ecology. Freshwater Biology 60: 603–619.
Scheffer, M., S. Hosper, M. Meijer, B. Moss & E. Jeppesen, 1993. Alternative equilibria in shalow lakes. Trends in Ecology and Evolution 8: 275–279.
Sioli, H., 1984. The Amazon: limnology and lands cape eco1ogy of a mighty tropica1 river and its basin. Junk Publications, Dordrecht.
Soares, M. C. S., V. L. M. Huszar & F. Roland, 2007. Phytoplankton dynamics in two tropical rivers with different degrees of human impact (Southeast Brazil). River Research and Applications 23: 698–714.
Souza, D. G., N. C. Bueno, J. C. Bortolini, L. C. Rodrigues, V. M. Bovo-Scomparin & G. M. de Souza Franco, 2016. Phytoplankton functional groups in a subtropical Brazilian reservoir: responses to impoundment. Hydrobiologia 779: 47–57.
StatSoft, 2005. STATISTICA (data analysis software system)., www.statsoft.com.
Sun, J. U. N. & D. Liu, 2003. Geometric models for calculating cell biovolume and surface area for phytoplankton. Journal of Plankton Research 25(11): 1331–1346.
Teixeira de Oliveira, M., O. Rocha & A. Peret, 2011. Structure of the phytoplankton community in the Cachoeira Dourada reservoir (GO/MG), Brazil. Brazilian Journal of Biology 71: 587–600.
Tilman, D., S. S. Kilham & P. Kilham, 1982. Phytoplankton community ecology: the role of limiting nutrients. Annual Review of Ecology and Systematics 13: 349–372.
Trinh, D. A., T. N. M. Luu, Q. H. Trinh, H. S. Tran, T. M. Tran, T. P. Q. Le, T. T. Duong, D. Orange, J. L. Janeau, T. Pommier & E. Rochelle-Newall, 2016. Impact of terrestrial runoff on organic matter, trophic state, and phytoplankton in a tropical, upland reservoir. Aquatic Sciences Springer Basel 78: 367–379.
Tundisi, J. G. & T. Matsumura-Tundisi, 2003. Integration of research and management in optimizing multiple uses of reservoirs: the experience in South America and Brazilian case studies. Hydrobiologia 500: 231–242.
Turgeon, K., C. Turpin & I. Gregory-Eaves, 2019. Dams have varying impacts on fish communities across latitudes: a quantitative synthesis. Ecology Letters 22(9): 1501–1516.
Utermöhl, H., 1958. Zur Vervollkommnung der quantitativen Phytoplankton-Methodik. Mitteilungen der Internationalen Vereinigung für Theoretische und Angewandte Limnologie 9: 1–38.
Violle, C., M. L. Navas, D. Vile, E. Kazakou, C. Fortunel, I. Hummel & E. Garnier, 2007. Let the concept of trait be functional! Oikos 116: 882–892.
Walker, B., A. Kinzig & J. Langridge, 1999. Adsorption behavior of phenanthrene onto treated natrural sorbents. Ecosystems 2: 95–113.
Winemiller, K. O., D. B. Fitzgerald, L. M. Bower & E. R. Pianka, 2015. Functional traits, convergent evolution, and periodic tables of niches. Ecology Letters. https://doi.org/10.1111/ele.12462.
Winemiller, K. O., P. B. McIntyre, L. Castello, E. Fluet-Chouinard, T. Giarrizzo, S. Nam, I. G. Baird, W. Darwall, N. K. Lujan, I. Harrison, M. L. J. Stiassny, R. A. M. Silvano, D. B. Fitzgerald, F. M. Pelicice, A. A. Agostinho, L. C. Gomes, J. S. Albert, E. Baran, M. Petere. Jr., C. Zarfl, M. Mulligan, J. P. Sullivan, C. C. Arantes, L. M. Sousa, A. A. Koning, D. J. Hoeinghaus, M. Sabaj, J. G. Lundberg, J. Armbruster, M. L. Thieme, P. Petry, J. Zuanon, G. T. Vilara, J. Snoeks, C. Ou, W. Rainboth, C. S. Pavanelli, A. Akama, A. Van Soesbergen & L. Sáenz, 2016. Balancing hydropower and biodiversity in the Amazon, Congo, and Mekong. Science 351: 128–129.
Acknowledgements
We thank Programa de Pós-Graduação em Biodiversidade, Ecologia e Conservação (old Ecologia de Ecótonos) for providing infrastructure and support. We also thank Núcleo Estadual de Metereologia e Recursos Hídricos (Universidade Estadual do Tocantins) for field and laboratory work. Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPEs) provided a scholarship for I.G.S. and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) provided research grants for F.M.P.
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling editor: Luigi Naselli-Flores
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
da Silva, I.G., Pelicice, F.M. & Rodrigues, L.C. Loss of phytoplankton functional and taxonomic diversity induced by river regulation in a large tropical river. Hydrobiologia 847, 3471–3485 (2020). https://doi.org/10.1007/s10750-020-04355-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-020-04355-2