Abstract
Shallow lake managers seek strategies to improve water quality and ecological features of these habitats, but lake responses are unpredictable and factors responsible for changes are often unclear. We summarized results of eight whole-lake rehabilitation projects in Minnesota, USA, an area with many shallow waters highly impacted by anthropogenic activities. To assess lake responses, we compared characteristics of managed sites to those of other regional shallow lakes manifesting clear- or turbid-state conditions. Managed lakes showed modest similarity to clear-water reference lakes in terms of phytoplankton (as chlorophyll a), nutrients, and submerged aquatic plants. Responses of aquatic invertebrate communities were more equivocal, with relatively little similarity to clear-water sites following management. These patterns indicate that these lakes either failed to undergo transitions to clear-water states, or that clear-water conditions did not persist throughout the 2–3 year period following treatment and prior to our evaluation. We believe these results show responses of shallow lakes that have been pushed beyond boundaries where they maintain sufficient natural resilience to resist local stressors. This means that shallow lake rehabilitation efforts will not always succeed and that, when improvements occur, management may need to be repeated to maintain favorable ecological conditions in highly modified landscapes.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Shallow freshwater lakes are probably some of the most impaired aquatic ecosystems because they often occur in close proximity to human population centers, lie within watersheds that are intensively cultivated for agriculture, have limited capacity to handle nutrient loading, and often have undergone dramatic alterations to natural hydrology (Moss et al., 1996; Søndergaard et al., 2007). Water depth in shallow lakes is highly variable, but maximum depth is often <4 m (Zimmer et al., 2016); large areas with even less depth are typical and sediments are often well-lighted. As a result, these sites provide some of the best examples of complex transitions between contrasting ecological states; shallow lakes shift rapidly from clear-water conditions with abundant submerged aquatic vegetation (SAV), to turbid-water conditions with poor water quality, little SAV, marginal value for wildlife, and diminished ecosystem services (Scheffer et al., 2001; Scheffer, 2004; Zimmer et al., 2009). Shallow lake managers in North America, Europe, and elsewhere have been working for over 75 years to reverse trends toward declining water quality and loss of submerged plant communities. Early efforts in North America focused on removing common carp (Cyprinus carpio Linnaeus, 1758) to improve habitat suitability for migratory waterfowl (Cahoon, 1953; Robel, 1961). With broader understanding of roles of benthivorous and planktivorous fish, management efforts have expanded to include the elimination of undesirable fish communities such as those dominated by bullheads (Ameiurus spp.) and small cyprinids, rather than just eradicating carp (Crivelli, 1983; Shapiro & Wright, 1984; Northcote, 1988; Hanson & Butler, 1994; Moss et al., 1996; Zimmer et al., 2001; Scheffer, 2004, and many others). Still, manipulation of fish communities remains a core strategy of shallow lake managers worldwide (Søndergaard et al., 2007; Bernes et al., 2015) despite the knowledge that maintaining results of biomanipulations can be challenging (Shapiro, 1990; Meijer et al., 1999).
Water quality (here defined as water clarity, concentrations of nutrients and phytoplankton) in shallow lakes is also dramatically influenced by water-level dynamics, and especially by flooding frequency, depth, and duration (van der Valk & Davis, 1978; White et al., 2008). Because high water quality in shallow lakes depends on healthy aquatic plant communities, it follows that lake rehabilitation strategies would also focus on mimicking natural hydrologic fluctuations. Water-level fluctuations affect virtually all aquatic plants including emergent and submergent growth forms (Squires & van der Valk, 1992; Wilcox & Meeker, 1992; Murkin & Ross, 2000; Euliss et al., 2004; van der Valk, 2005, and many others). Periodic drawdowns are often essential for maintaining natural shallow lake processes and biotic communities (Coops & Hosper, 2003; Chow-Fraser, 2005; Leira & Cantonati, 2008). Shallow lakes with natural water-level dynamics support more diverse plant and animal communities than do those with modifications that maintain more stable water levels (Wilcox & Meeker, 1992; Hanson et al., 2005; van der Valk, 2005; Leira & Cantonati, 2008; Wantzen et al., 2008; McCauley et al., 2015). Sustained high water levels are almost certain to favor loss of plants and may induce transitions to turbid regimes, especially in lakes with relatively high productivity and populations of benthivorous and/or planktivorous fishes (Blindow et al., 1993; Coops et al., 2003; Hobbs et al., 2012). Alternatively, intermittent periods of lower water levels often increase abundance and diversity of submerged aquatic plants and may stabilize, or favor transitions to, clear-water regimes (but see Cobbaert et al., 2015 for a contrasting example).
Drawdown and fish removal treatments are often applied simultaneously (or sequentially) to improve water clarity and light availability for SAV, and to enhance zooplankton and macroinvertebrates, in hopes of improving water quality and often to benefit waterfowl and other wetland-dependent species (Meronek et al., 1996). Lake rehabilitation efforts combining fish removal with water-level drawdown are intended to maximize responses by greatly reducing fish populations, favoring re-establishment of SAV, and stimulating germination of emergent macrophytes in lake margins. Following drawdown, rotenone is often applied during fall-winter periods when lake volumes are reduced, and target fish populations may be concentrated and already stressed by under-ice hypoxia. In nearly all cases, shallow lake rehabilitation aims to improve lake water clarity and ultimately induce shifts to clear-regime conditions (sensu Moss et al., 1996; Scheffer, 2004; Zimmer et al., 2009) within 1 or 2 years following management.
Most shallow lake rehabilitation projects include drawdowns, removal of undesirable fishes, or combinations of both, thus the interpretation of lake responses is often complicated by the presence of multiple treatment factors required to achieve management goals. For example, inducing drawdown can be confounded by high precipitation, extensive surface water connectivity, and requirements of recreational lake users. Fish removal is logistically difficult, often requires use of chemical toxicants, and must be repeated because fish typically recolonize lakes within a few years after treatment. To maximize project results, managers often apply multiple types of treatments, but this makes it nearly impossible to identify cause and effect relationships that would inform future projects (Søndergaard et al., 2007; Noordhuis et al., 2015).
We evaluated characteristics of eight shallow lakes in Minnesota, USA, in 2010–2011, subsequent to management treatments during 2007–2010. In a modified comparative study design, we gathered post-treatment data from these managed lakes, comparing features of the managed sites to characteristics of unmanaged clear-state and turbid-state reference lakes. We used several analytical approaches to evaluate responses. First, we compared patterns among managed, clear-, and turbid-state reference lakes using vertical scatter plots to help visualize trends. Second, we applied ordination methods to compare annual mean nutrient and phytoplankton levels, SAV mass, and abundance of fish and aquatic invertebrates in managed sites with attributes of reference lakes. Finally, we used model fitting procedures to assess the extent to which lake management history and ecological regime status predicted limnological characteristics and fish communities in managed and reference sites. We summarize results of these comparisons, explore whether management was successful in inducing transitions to clear-water states, and discuss implications for future shallow lake management.
Methods
Study framework
Our study focused on evaluating limnological and community responses of eight managed shallow lakes. We were limited by a lack of pre-treatment data from these sites; thus, we gathered data from a group of similar, but unmanaged shallow lakes within the same ecoregion. We used a modified comparative design that substitutes space for time (Pickett, 1989) and has been shown to be effective for assessing responses to environmental gradients in aquatic systems (Gyllström et al., 2005; Baho et al., 2015). Going one step further, we assigned unmanaged reference lakes to clear-state or turbid-state categories (hereafter clear or turbid reference) using chlorophyll a (Chla) threshold values developed from other regional shallow lakes (following Zimmer et al., 2009). We assigned all lakes to one of three classes based on management histories and prevailing water clarity conditions (managed, and clear or turbid reference sites). This allowed us to assess the extent to which characteristics of managed lakes concurrently resembled clear or turbid reference counterparts. Overall then, we sampled the managed lakes, along with a total of 46 unmanaged reference sites during 2010–2011. All managed and most reference lakes were sampled during both study years. Unfortunately, in 2010, logistical problems prevented us from collecting limnological data from two reference lakes, and aquatic invertebrates were not gathered from four reference lakes. Sampling managed and reference lakes during 2 years produced a total of 106 and 104 lake-year combinations for among-lake comparisons of limnological variables and invertebrates, respectively. Overall, our lake set included 17 and 20 turbid-regime lakes during 2010 and 2011, with the remainder showing characteristics of clear-regime conditions.
Managed lakes were treated using combinations of water-level reduction (drawdown) and fish removal. In one lake, piscivorous walleye (Sander vitreus Mitchell, 1818) were also added (as fry) to suppress fathead minnows (Pimephales promelas Cope, 1868) and to increase large-bodied zooplankton populations (sensu Potthoff et al., 2008). In all cases, the objective was to improve wildlife habitat through re-establishment of clear-water conditions as described by Scheffer (2004), Zimmer et al. (2009), and many others. Managed lakes were chosen based on treatment histories so random selection of sites was not possible. We took several steps to select a homogeneous group of sites and to minimize temporal and geographic variability. We considered only lake management projects that had been completed within the 4 years prior to our data gathering. All study lakes lay within the Prairie Parkland Province (Omernik, 1987), a large area originally composed of grass and parkland now largely converted to agricultural land uses, and covering much of western-central and southern Minnesota. All managed lakes were partially drawn down for at least one growing season ( 2007 or 2008). Four of these sites also received fall or winter (under-ice) rotenone applications to remaining flooded areas to eliminate remaining fish populations. Management activities at each of the study lakes are summarized in Table 1 along with lake names, sizes, and other project information.
Community sampling
Zooplankton density estimates were derived from samples gathered on single dates in July 2010 and August 2011 concurrent with fish sampling by collecting two replicate vertical column samples (Swanson, 1978) at five locations in each lake. Abundance of macroinvertebrates was estimated from samples gathered concurrently with sweep nets (Murkin et al., 1983) at 0.75 m depth at five randomly selected locations. All invertebrate samples were preserved in the field by placing in 70% ethanol and transported back to the lab where they were enumerated using dissecting microscopes. Abundance counts were derived for all major taxa of zooplankton and macroinvertebrates.
Fish species composition and relative abundance (biomass per unit effort) were determined using a combination of gears deployed overnight. All fish sampling was done once each year, during July and August in 2010 or 2011, respectively. Three mini-fyke nets (6.5-mm bar mesh with 4 hoops, 1 throat, 7.62 m lead, and a 0.69 × 0.99 m rectangular frame opening into the trap) were set overnight in the littoral zone of each lake. One experimental gill net (2 m by 61 m multifilament net with 19.0, 25.0, 32.0, 38.0, and 51.0-mm bar meshes) was deployed along the deepest depth contour in lakes less than 2 m deep or along a 2 m contour in lakes with sufficient depth. The protocol outlined above has been effective in sampling fish assemblages in small lakes in Minnesota, surrounding states, and an adjacent Canadian province (Tonn & Magnuson, 1982; Rahel, 1984; Jackson & Harvey, 1989; Robinson & Tonn, 1989; Herwig et al., 2010). All fish sampled were sorted by species, and weighed in bulk. Data were summarized as the total biomass of each predominant feeding guild (piscivores, planktivores, and benthivores) collected in all four nets.
Limnological variables: nutrients, phytoplankton, and submerged aquatic vegetation
Surface (dip) water samples were taken from central locations of each lake once during July or August concurrent with other sampling. Water samples were frozen and transported to the University of St. Thomas (St. Paul, MN) for analysis of phytoplankton abundance (Chla), total nitrogen (TN), and total phosphorus (TP) using methods of APHA (1994).
We considered SAV in our analyses along with other limnological variables because previous work shows strong (usually reciprocal) abundance relationships between TP and Chla, and SAV in shallow lakes (Scheffer, 2004; Zimmer et al., 2009). Abundance of SAV was measured using modified techniques of Deppe & Lathrop (1992). During July (2010) or August (2011), SAV was sampled at 15 stations in each lake, equidistant along three transects crossing the width of each basin. A single cast with a weighted plant rake was made at each location, and a plant sample was gathered along 3 m of lake bottom. Plants collected on each rake cast were weighed using spring scales (wet weight). For each lake, SAV data were summarized as average total mass of all rake samples (mean mass across the total number of rake casts). Prior to our study, and as summarized in Fig. 4, Secchi disk transparency and extent of SAV were measured in managed lakes during one or 2 years immediately before treatment using a 30-cm-diameter disk and following techniques of Deppe & Lathrop (1992). Secchi disk transparency and SAV extent were also measured after lake management both prior to and during our study. However, values shown in Fig. 4 were not considered along with other data used in more formal comparisons.
Framework for evaluating responses
We used several approaches to compare patterns in limnological variables and communities among lake classes. First, we constructed vertical scatterplots of fish abundance and limnological variables to help visualize trends among our three lake classes. Scatterplots are useful for depicting data trends and are less likely to distort information about data characteristics than bar graphs or other common approaches, especially with small datasets (Weissgerber et al., 2015). Next, we performed nonmetric multidimensional scaling (NMS; McCune & Grace, 2002) to compare characteristics of our managed lakes with features of unmanaged clear and turbid reference sites. NMS is useful for exploring data patterns in cases like this because resulting plots maximize similarity relationships, in our case, depicting patterns among lakes (and lake classes). Additionally, the method relaxes requirements for normal data distributions and linear relationships to environmental gradients. A first NMS ordination was performed using a matrix of our limnological variables (TP, TN, Chla, and abundance of all SAV); a second NMS was done with a matrix composed of 22 taxa of zooplankton and macroinvertebrates. All NMS procedures were conducted in PC-ORD v. 5 (McCune & Mefford, 1999) and following guidelines suggested by McCune & Grace (2002). Values of limnological variables (TP, TN, Chla, and SAV mass) and invertebrates were log10(n + 1) transformed prior to analyses. Preliminary ordinations were done with the autopilot mode. We used Sorenson (Bray-Curtis) distance measures and preliminary models based on six-dimensional solutions, and significance was based on Monte Carlo permutation procedures (250 runs). Final ordination models were based on stress reduction and followed autopilot recommendations (McCune & Mefford, 1999; McCune & Grace, 2002). Following varimax rotation, we also assessed correlations between data matrices and final fitted axes and summarized R 2 values (McCune & Grace, 2002). To identify differences in community patterns among lake classes, we also performed an Indicator Species Analysis (ISA) using our matrix of zooplankton and macroinvertebrates. ISA results were then assessed with Monte Carlo procedures with significance testing based on 4,999 random permutations; for each taxon, differences among our three lake classes were inferred at P < 0.05. ISA and permutation tests were run in PC-ORD v. 5 (McCune & Mefford, 1999) according to recommendations of McCune & Grace (2002).
Finally, to further explore patterns among lake classes, we compared mass of major fish feeding guilds (benthivores, planktivores, and piscivores) and key limnological variables using linear mixed models. Invertebrate data were not included in this model fitting because our interest in these organisms focused on community-level patterns and we believed these were better assessed with NMS as described above. We started with a ‘base’ generalized linear mixed model (GLMM; Bolker et al., 2009) with lake entered as a random effect to account for repeated measures correlations. We then compared four GLMM models (again including Lake as random effect) to explore strength of evidence for models containing the following fixed effects: class (clear reference, turbid reference, and managed lakes), year (2010, 2011), class + year, and class * year. For each response variable, data were log10(n + 1) transformed and analyzed separately. Modeled responses included biomass of benthivores, planktivores, benthivores + planktivores, piscivores, black bullhead (Ameiurus melas Rafinesque, 1820), common carp, Chla, SAV, TP, and TN. We focused on black bullhead and common carp because they are often the most abundant benthivores in shallow Minnesota lakes and they are often associated with transitions to turbid-state conditions in regional lakes (Zimmer et al., 2009; Bajer et al., 2015). Use of the Bayesian Information Criterion (BIC) has been suggested for model selection when, as in our case, the goal is to find adequate descriptive models (Taper, 2004). Lower BIC scores indicate better parsimony, and difference of BIC scores between models (denoted as ΔBIC) gives a measure of the strength of evidence for one model over the other. A ΔBIC value of 2 or less implies models have essentially the same support, while a ΔBIC of greater than 6 implies strong evidence in support of the model with the lower BIC score (Raftery, 1995). If two models had similar support (i.e., ΔBIC < 2), we selected as best the model including the fewest predictors (Arnold, 2010). All linear models were run using version 3.1.0 of program R (R Development Core Team, 2014); the GLMM models were fit using the lmer function from the lme4 package (Bates & Maechler, 2012).
Results
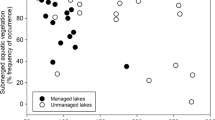
Limnological variables, SAV, and sometimes fish communities were highly variable among- and within-lake classes (Figs. 1, 2, 3). However, relative abundance of black bullhead was consistently low in managed lakes (Fig. 3e). TP was also lower in managed lakes than in most turbid reference counterparts, with observed values often even lower than those in clear reference sites (Fig. 1a). Chla levels in managed lakes were often comparable to values observed in regional clear reference sites, but usually lower than those in turbid reference lakes (Fig. 2a). SAV mass in managed lakes was similar to that in turbid reference lakes, but values were usually lower than in clear reference sites, perhaps indicating weak or latent responses following treatment (Fig. 2b). Overall, individual lake features following management were highly variable with these lakes most often showing characteristics intermediate between unmanaged clear and turbid reference sites (Fig. 4).
Total phosphorus, total nitrogen, and N:P ratios in managed, clear, and turbid study lakes. Symbols depict summer values during 2010 (circles) and 2011 (triangles); turbid- and clear-water regime status based on chlorophyll a thresholds of Zimmer et al. (2009)
Chlorophyll a and SAV mass in managed, clear, and turbid study lakes. Symbols depict summer values during 2010 (circles) and 2011 (triangles); turbid- and clear-water regime status based on chlorophyll a thresholds of Zimmer et al. (2009)
Relative abundance of planktivorous, benthivorous, and piscivorous fish in managed, clear, and turbid study lakes. Symbols depict total mass of fish captured during 24-h sampling during 2010 (circles) and 2011 (triangles); turbid- and clear-water regime status based on chlorophyll a thresholds of Zimmer et al. (2009)
Change in Secchi depth and percent vegetated sites in eight managed lakes relative to pre-treatment conditions. Pre-treatment values based on averages of available data gathered prior to our study (1 to 2 years). Post-treatment values were average measurements from a period prior to and during our study, and over a range of 1–5 years. More details are provided in text
Similarity patterns between managed and reference lakes
Two significant dimensions (axes) were identified by NMS and these explained approximately 97% of the variability in Chla, TP, TN, and SAV mass among all lakes (including managed, clear, and turbid reference sites, Table 2). Patterns showed that managed lakes clustered toward intermediate positions along ordination axes, and generally between clear and turbid reference counterparts (Fig. 5). Comparing correlations between Chla, TP, TN, SAV mass and ordination axes indicated that TP and Chla values were most strongly associated with both underlying axes (Table 2). Stress associated with our final 2-dimensional NMS model was 10.8% indicating relatively good fit to underlying data (McCune & Grace, 2002).
Recommended two-dimensional NMS model with varimax rotation based on matrix of chlorophyll a, total phosphorus, total nitrogen, and total SAV mass measured in shallow Minnesota lakes during 2010 and 2011 (details discussed in text). Symbols indicate lake status: managed lakes (solid circles), regional clear lakes (triangles), and regional turbid lakes (stars)
Our invertebrate NMS identified three significant axes based on 22 taxa and these axes cumulatively explained about 91% of the variability in zooplankton and macroinvertebrates among our study lakes (Table 3). However, this NMS showed little differentiation among managed, clear, and turbid reference lake scores, suggesting considerable similarity of zooplankton and macroinvertebrates among our three lake classes (Fig. 6). Correlations between invertebrate taxa and underlying axes were relatively low with the highest values evident for large cladocera (R 2 values = 0.542 for axis 1; Table 3). Despite weak separation of lake scores along NMS axes, ISA indicated that several groups of macroinvertebrates were more abundant in clear reference lakes, while no taxa were reliable indicators of managed or turbid reference sites (Table 4). Moderate stress (11.8%) for our final model again indicated good fit to data.
Recommended NMS model with varimax rotation based on matrix of zooplankton and macroinvertebrates measured in shallow Minnesota lakes during 2010 and 2011 (details discussed in text). Symbols indicate lake status: managed lakes (solid circles), regional clear lakes (triangles), and regional turbid lakes (stars). Only 2 of 3 recommended axes are shown
Trends in limnological features, SAV, and fish populations among lake classes
Our linear model containing lake class was best supported (lowest BIC values) for all limnological variables (TP, TN, Chla, SAV; Table 5). Comparisons among model intercepts, SD, and t values suggested that clear reference and managed lakes had lower TP levels than turbid reference lakes, but levels were similar between clear reference and managed sites (Table 5). Our best TN model included a class*year interaction, suggesting that patterns differed between years, but higher TN values were sometimes observed in turbid reference lakes, with similar TN levels in clear reference and managed sites. The best SAV model also included lake class; SAV abundance was the highest in clear reference lakes and lowest in turbid reference sites, with intermediate SAV in managed lakes (Table 6). The opposite pattern was observed for Chla as values were the highest in turbid reference lakes, lowest in clear reference sites, and intermediate in managed lakes. Intraclass correlations in our limnological variables were relatively low among study lakes, ranging from approximately 0.09 to 0.24 for SAV and TP, respectively, suggesting that Chla, nutrient concentrations, and SAV values during 2010 were not good predictors of levels in 2011. TP and TN in managed and clear lakes were generally lower than in turbid lakes during both study years.
Our best supported linear models for fish (benthivores, planktivore + benthivores, and black bullhead) were lake class models. In contrast, BIC values indicated that our base model performed best for planktivores and piscivores, and year was our best common carp model (Table 6). Comparisons of model intercepts, standard deviations (SD), and t values showed that turbid reference lakes had highest, and managed lakes lowest, biomass of benthivores and black bullhead, with intermediate abundance in clear lakes. Models indicated that piscivore and planktivore abundance was similar among the three lake classes. Abundance of common carp was also similar among lake classes, but differed between study years, with higher biomass during 2011 (Table 5). Overall, between-year intraclass correlations in fish community variables were high, ranging from 0.54 to 0.84 for bullheads and planktivores + benthivores, respectively, indicating that fish communities in individual lakes were generally similar between study years.
Discussion
Our analyses indicated that limnological attributes of managed shallow lakes generally placed these sites at intermediate positions between clear and turbid reference sites along gradients identified using NMS. There are at least two plausible explanations for these patterns. First, managed lakes might still have been transitioning from turbid- to clear-state conditions (2–4 years following management). Alternatively, biomanipulation and/or water-level drawdown might have failed to trigger transitions to clear states. In either case, patterns point to several generalizations that should be of interest to lake managers. First, postmanagement levels of Chla and TP were lower than those observed concurrently in turbid reference lakes and in some cases values were even lower than those in clear lakes. Lower Chla levels were consistent with expected trends if features of managed lakes were in transition from turbid- to clear-water states, but we are uncertain why relatively low TP levels were observed in the managed lakes during our study, a period 2–4 years following lake management. Second, SAV mass in managed lakes appeared to be relatively low during both years of our study. This may indicate that the extent of plant communities in managed lakes increased relatively slowly, or it may show that SAV failed to increase to levels observed in clear reference lakes. Other studies have also indicated that plant establishment can be critical to developing a more stable clear-water condition (Hanson & Butler, 1994; Hansel-Welch et al., 2003) and plant communities can take several years to develop after a management treatment (Hansel-Welch et al., 2003). Also, aquatic plant propagule densities can be highly variable in shallow lakes and impact restoration efforts (Baker et al., 2013). Third, fish populations were widespread among managed lakes and fish in some sites had already reached levels comparable to those observed in turbid reference lakes by 2010, just 2–4 years following treatment. However, benthivorous fish (especially black bullhead) were far less abundant in managed lakes, presumably in response to the lake management efforts employed. It is plausible that fish toxicants and/or water-level drawdowns were especially effective in reducing these benthivores, at least during the first few years following lake management. Common carp showed a slightly different pattern; two managed lakes developed relatively high common carp populations following management. With respect to common carp, we do not know whether individuals we sampled resulted from colonization following lake management, or if these are offspring of fish that survived lake manipulations.
Improving water quality and ecological characteristics of shallow lakes is a growing international priority as evidenced by the extent and scope of related literature and the large expenditures of jurisdictional authorities (Moss et al., 1996; Scheffer, 2004; Bernes et al., 2015). Water-level drawdowns and biomanipulation remain focal strategies for triggering improved water quality and habitat suitability for wildlife in shallow lakes across the US, Europe, and elsewhere. Unfortunately, despite initial improvements, resilience of turbid states often limits recovery and lake conditions frequently deteriorate again within 5–10 years (Søndergaard et al., 2007). Studies of lake sediments in Europe and US show evidence of increased phosphorus and nitrogen loading since settlement (Søndergaard et al., 2005; Ramstack et al., 2004; Hobbs et al., 2012, 2014) and it is likely that high rates of in-lake nutrient cycling from anthropogenic sediment both (a) favors transitions to turbid states and (b) contributes to resilience of turbid conditions (Søndergaard et al., 2005; Hobbs et al., 2012). While restoration of upland cover within lake watersheds has many beneficial effects, in-lake communities and characteristics are often difficult to link with watershed-scale land cover variables (Jeppesen et al., 2007; Hanson et al., 2012; Bayley et al., 2013; Cobbaert et al., 2015). Thus, lakes may be very slow to improve in response to reduced nonpoint loading of nutrients, sediments, and other constituents (Carpenter et al., 1998). In addition, upland restoration is limited by private landowners’ willingness to sell land or enroll in conservation programs as there are few incentives to participate given recent agricultural economics. In-lake management can often be done by public agencies and does not require as much private landowner cooperation.
We need to better understand the interplay between lakes’ responses to specific management activities, and factors causing resilience of turbid (and clear) lake states. Paleolimnological investigations of sediments in shallow lakes in conjunction with lake rehabilitation projects have provided powerful tools for interpreting modern lake characteristics and transitions in historical contexts (Ramstack Hobbs et al., in press). As one example, Hobbs et al. (2012) recently showed that sometime around 1950, likely due to dense fish populations and nutrient loading within the lake watershed, a large shallow Minnesota lake (Christina) had been pushed beyond a point where it’s intrinsic resilience (sensu Scheffer et al., 2001) allowed maintenance of clear-water conditions. Like historical patterns documented in Lake Christina (Hanson & Butler, 1994; Hansel-Welch et al., 2003; Hobbs et al., 2012), results from rehabilitation projects discussed here illustrate responses of shallow lakes which have been perturbed beyond their “safe operating space” (after Scheffer et al., 2015). While management “pushed” our managed lakes to transitory limnological conditions, efforts apparently were insufficient to induce shifts to characteristics similar to those observed in clear reference sites. This shows why resilience of turbid- (and clear-) water states in shallow lakes needs further study, both to improve understanding of these complex ecological systems and to facilitate more effective lake management.
Whole-lake management projects in shallow lakes not only offer unique research opportunities, but also pose extraordinary challenges for researchers. Combinations of rehabilitation measures are often applied to the same lakes, and projects quickly become multiple treatment experiments with small sample sizes. Lake management efforts have often focused on within-lake properties (fish communities, water levels) because these practices are sometimes effective and because economics usually limit the extent of watershed restoration. Investigators may be offered collaboration opportunities, but find few financial or logistical incentives for intensive pre-treatment data gathering. Gathering compatible data from multiple lake projects is extremely challenging, and requires innovative approaches to data synthesis and analyses. Nonetheless, we believe whole-lake projects afford unique and necessary learning opportunities that engage multiple stakeholders in ecosystem-scale experiments as they help advance restoration science.
We suggest that expectations of lake managers and project stakeholders need to be better aligned with a growing body of evidence highlighting needs for ongoing, often repeated, management to achieve effective rehabilitation of shallow eutrophic lakes (Scheffer, 2004; Søndergaard et al., 2007); satisfactory results may rarely be obtained by single management efforts. Often, ongoing management will certainly be necessary to mitigate—and possibly overcome—ecological resilience in many sites (Carpenter et al., 2001; Hobbs et al., 2012). Dramatic reductions in TP and more modest responses in SAV observed in some lakes in the present study are incentive for managers to continue to explore and pursue management on shallow lakes that previously had little to no habitat value for wildlife. We encourage continued management of shallow lakes even if such results are relatively short-lived, especially in landscapes that contain few if any other shallow lakes in better condition. The fact that our managed lakes often failed to make transitions to clear-water states also shows why it is important to preserve ecological resilience of existing clear-water state waterbodies (sensu Carpenter et al., 2001). Allowing eutrophication or other anthropogenic influences to surpass the limits of ecological resilience may have irreversible consequences (Carpenter et al., 1999). Managers and stakeholders must recognize that, at best, repeated manipulations will often be necessary to maintain (or even to induce) desired ecological characteristics in shallow lakes, especially in highly altered landscapes and in lakes with high levels of anthropogenic nutrient-laden sediments.
References
APHA, 1994. Standard Methods for the Examination of Waste and Wastewater. American Public Health Association, Washington, DC.
Arnold, T. W., 2010. Uninformative parameters and model selection using Akaike’s information criterion. Journal of Wildlife Management 74: 1175–1178.
Baho, D. L., U. N. Tavşanoğlu, M. Šorf, K. Stefanidis, S. Drakare, U. Scharfenberger, H. Agasild, M. Beklioğlu, J. Hejzlar, R. Adrian, E. Papastergiadou, P. Zingel, M. Søndergaard, E. Jeppesen & D. G. Angeler, 2015. Macroecological patterns of resilience inferred from a multinational, synchronized experiment. Sustainability 7: 1142–1160.
Bajer, P. G. & P. W. Sorenson, 2015. Effects of common carp on phosphorus concentrations, water clarity, and vegetation density: a whole system experiment in a thermally stratified lake. Hydrobiologia 746: 303–311.
Baker, E. S., J. M. Sarneel, R. D. Gulati, Z. Liu & E. van Donk, 2013. Restoring macrophyte diversity in shallow temperate lakes: biotic versus abiotic constraints. Hydrobiologia 710: 23–37.
Bates, D. & M. Maechler, 2012. lme4: Linear mixed-effects models using S4 classes. R package version 0.999375-42. Available at http://cran.r-project.org/web/packages/lme4/index.html.
Bayley, S. E., A. S. Wong & J. E. Thompson, 2013. Effects of agricultural encroachment and drought on wetlands and shallow lakes in the boreal transition zone of Canada. Wetlands 33: 17–28.
Bernes, C., S. R. Carpenter, A. Gardmark, P. Larsson, L. Persson, C. Skov, J. D. M. Speed & E. Van Donk, 2015. What is the influence of a reduction of planktivorous and benthivorous fish on water quality in temperate eutrophic lakes? A systematic review. Environmental Evidence 4: 7.
Blindow, I., G. Andersson, A. Hargeby & S. Johansson, 1993. Long-term pattern of alternative stable states in two shallow eutrophic lakes. Freshwater Biology 30: 159–167.
Bolker, B. M., M. E. Brooks, C. J. Clark, S. W. Geange, J. R. Poulsen, M. H. H. Stevens & J. S. White, 2009. Generalized linear mixed models: a practical guide for ecology and evolution. Trends in Ecology & Evolution 24: 127–135.
Cahoon, W. E., 1953. Commercial carp removal at Lake Mattamusket, North Carolina. Journal of Wildlife Management 17: 312–317.
Carpenter, S. R., N. F. Caraco, D. L. Correll, R. W. Howarth, A. N. Sharpley & V. H. Smith, 1998. Nonpoint pollution of surface waters with phosphorus and nitrogen. Ecological Applications 8: 559–568.
Carpenter, S. R., D. Ludwig & W. A. Brock, 1999. Management of eutrophication for lakes subject to potentially irreversible change. Ecological Applications 9: 751–771.
Carpenter, S., B. Walker, J. M. Anderies & N. Abel, 2001. From metaphor to measurement: resilience of what to what? Ecosystems 4: 765–781.
Cobbaert, D., A. S. Wong & S. E. Bayley, 2015. Resistance to drought affects persistence of alternative regimes in shallow lakes of the Boreal Plains (Alberta, Canada). Freshwater Biology 10: 2084–2099.
Chow-Fraser, P., 2005. Ecosystem response to changes in water level of Lake Ontario marshes: lessons from the restoration of Cootes Paradise Marsh. Hydrobiologia 539: 189–204.
Coops, H. & S. H. Hosper, 2003. Water-level management as a tool for the restoration of shallow lakes in the Netherlands. Lake and Reservoir Management 18: 292–297.
Coops, H., M. Beklioglu & T. L. Crisman, 2003. The role of water-level fluctuation in shallow lake ecosystems-workshop conclusions. Hydrobiologia 506–509: 23–27.
Crivelli, A. J., 1983. The destruction of aquatic vegetation by carp. Hydrobiologia 106: 37–41.
Deppe, E. R., & R. C. Lathrop, 1992. A comparison of two rake sampling techniques for sampling aquatic macrophytes. Wisconsin Department of Natural Resources Research and Management Findings No. 32.
Euliss, N. H., J. W. LaBaugh, L. H. Fredrickson, D. M. Mushet, M. K. Laubhan, G. A. Swanson, T. C. Winter, D. O. Rosenberry & R. D. Nelson, 2004. The wetland continuum: a conceptual framework for interpreting biological studies. Wetlands 24: 448–458.
Faraway, J. J., 2006. Extending the linear model with R: generalized linear, mixed effects and nonparametric regression models. Chapman & Hall, New York, NY.
Gyllström, M., L. A. Hansson, E. Jeppesen, F. Garcia-Criado, E. Gross, E. K. Irvine, K. Kairesalo, R. Kornijów, M. Miracle, M. Nykänen, T. Nõges, S. Romo, Susana D. Stephen, E. van Donk & B. Moss, 2005. The role of climate in shaping zooplankton communities of shallow lakes. Limnology & Oceanography 50: 2008–2021.
Hansel-Welch, N., M. G. Butler, T. J. Carlson & M. A. Hanson, 2003. Changes in macrophyte community structure in Lake Christina (Minnesota), a large shallow lake, following biomanipulation. Aquatic Botany 75: 323–337.
Hanson, M. A. & M. G. Butler, 1994. Responses of plankton, turbidity, and macrophytes to biomanipulation in a shallow prairie lake. Canadian Journal of Fisheries and Aquatic Sciences 51: 1180–1188.
Hanson, M. A., K. D. Zimmer, M. G. Butler, B. A. Tangen, B. R. Herwig & N. H. Euliss, 2005. Biotic interactions as determinants of ecosystem structure in prairie wetlands: an example using fish. Wetlands 25: 764–775.
Hanson, M. A., B. R. Herwig, K. D. Zimmer, J. Fieberg, S. R. Vaughn, R. G. Wright & J. A. Younk, 2012. Comparing effects of lake- and watershed-scale influences on communities of aquatic invertebrates in shallow lakes. PLoS One 7(9): e44644.
Herwig, B. R., K. D. Zimmer, M. A. Hanson, M. L. Konsti, J. A. Younk, R. W. Wright, S. R. Vaughn & M. D. Haustein, 2010. Factors influencing fish distributions in shallow lakes in prairie and prairie-parkland region of Minnesota, USA. Wetlands 30: 609–619.
Hobbs, W. O., J. M. Ramstack Hobbs, T. LaFrancois, K. D. Zimmer, K. M. Theissen, M. B. Edlund, N. Michelutti, M. G. Butler, M. A. Hanson & T. J. Carlson, 2012. A 200-year perspective on alternative stable state theory and lake management from a biomanipulated shallow lake. Ecological Applications 22: 1483–1496.
Hobbs, W. O., K. M. Theissen, S. M. Hagen, C. W. Bruchu, C. Ben Czeck, J. M. Hobbs & K. D. Zimmer, 2014. Persistence of clear-water, shallow-lake ecosystems: the role of protected areas and stable aquatic food webs. Journal of Paleolimnology 51: 405–420. doi:10.1007/s10933-013-9763-1.
Jackson, D. A. & H. H. Harvey, 1989. Biogeographic associations in fish assemblages: local vs. regional processes. Ecology 70: 1472–1484.
Jeppesen, E., M. Meerhoff, B. A. Jacobsen, R. S. Hansen, M. Søndergaard, J. P. Jensen, T. L. Lauridsen, N. Mazzeo & C. W. C. Branco, 2007. Restoration of shallow lakes by nutrient control and biomanipulation-the successful strategy varies with lake size and climate. Hydrobiologia 581: 269–285.
Leira, M. & M. Cantonati, 2008. Effects of water-level fluctuations on lakes: an annotated bibliography. Hydrobiologia 613: 171–184.
McCauley, L. A., M. J. Anteau, M. Post van der Burg & M. T. Wiltermuth, 2015. Land use and wetland drainage affect water levels and dynamics of remaining wetlands. Ecosphere 6: 92.
McCune, B. & J. B. Grace, 2002. Analysis of Ecological Communities. MJM Software Design, Gleneden Beach, CA.
McCune, B. & M. J. Mefford, 1999. PC-ORD. Multivariate Analysis of Ecological Data, version 4. MjM Software Design, Gleneden Beach, CA.
Meronek, T. G., P. M. Bouchard, E. R. Buckner, T. M. Burri, K. K. Demmerly, D. C. Hatleli, R. A. Klumba, S. H. Schmidt & D. W. Coble, 1996. A review of fish control projects. North American Journal of Fisheries Management 16: 63–74.
Meijer, M., I. de Boois, M. Scheffer, R. Portielje & H. Hosper, 1999. Biomanipulation in shallow lakes in The Netherlands: an evaluation of 18 case studies. Hyrdrobiologia 408(409): 13–30.
Moss, B., J. Madgwick & G. Phillips, 1996. Guide to the Restoration of Nutrient-Enriched Shallow Lakes. The Broads Authority, Norwich.
Murkin, H. R., P. G. Abbott & J. A. Kadlec, 1983. A comparison of activity traps and sweep nets for sampling nektonic invertebrates in wetlands. Freshwater Invertebrate Biology 2: 99–106.
Murkin, H. R. & L. C. M. Ross, 2000. Invertebrates in prairie wetlands. In Murkin, H. R., A. G. van der Valk & W. R. Clark (eds), Prairie Wetland Ecology. Iowa State University Press, Ames IA: 201–248.
Noordhuis, R., B. G. van Zuidam, E. T. Peeters & G. J. van Geest, 2015. Further improvements in water quality of the Dutch Borderlakes: two types of clear states at different nutrient levels. Aquatic Ecology. doi:10.1007/s10452-015-9521-8.
Northcote, T. G., 1988. Fish in the structure and function of freshwater ecosystems: a “op-down” view. Canadian Journal of Fisheries and Aquatic Sciences 45: 361–379.
Omernik, J. M., 1987. Ecoregions of the conterminous United States. Annals of the Association of American geographers 77: 118–125.
Picket, S. T., 1989. Space-for-time substitution as an alternative to long-term studies. In Likens, G. E. (ed.), Long-Term Studies in Ecology. Springer, New York, NY: 110–135.
Potthoff, A. J., B. R. Herwig, M. A. Hanson, K. D. Zimmer, M. G. Butler, J. R. Reed, B. G. Parsons & M. C. Ward, 2008. Cascading food-web effects of piscivore introductions in shallow lakes. Journal of Applied Ecology 45: 1170–1179.
R Development Core Team, 2014. The R Project for Statistical Computing. Available at http://www.R-project.org.
Ramstack, J. M., S. C. Fritz & D. R. Engstrom, 2004. Twentieth century water quality trends in Minnesota lakes compared with presettlement variability. Canadian Journal of Fisheries and Aquatic Sciences 61: 561–576.
Ramstack Hobbs, J. M., W. O. Hobbs, M. B. Edlund, K. D. Zimmer, K. M. Theissen, N. Hoidal, L. M. Domine, M. A. Hanson, B. R. Herwig, & J. B. Cotner. In Press. The legacy of large regime shifts in shallow lakes. Ecological Applications.
Raftery, A. E., 1995. Bayesian model selection in social research. Sociological Methodology 25: 111–163.
Rahel, F. J., 1984. Factors structuring fish assemblages along a bog lake successional gradient. Ecology 65: 1276–1289.
Robel, R. J., 1961. The effect of carp populations on the production of waterfowl food plants on a western waterfowl marsh. Transactions of the North American Wildlife Natural Resource Conference 26: 147–159.
Robinson, C. L. & W. M. Tonn, 1989. Influence of environmental factors and piscivory in structuring fish assemblages of small Alberta lakes. Canadian Journal of Fisheries and Aquatic Sciences 46: 81–89.
Scheffer, M., S. Carpenter, J. A. Foley, C. Folk & B. Walker, 2001. Catastrophic shifts in ecosystems. Nature 413: 591–596.
Scheffer, M., 2004. Ecology of Shallow Lakes. Kluver Academic Publishers, Dordrecht.
Scheffer, M., S. Barrett, S. R. Carpenter, C. Folke, A. J. Green, M. Holmgren, T. P. Hughes, S. Kosten, I. A. van de Leemput, D. C. Nepstad, E. H. van Nes, E. T. Peeters & B. Walker, 2015. Creating a safe operating space for iconic ecosystems. Science 347: 1317–1319.
Shapiro, J., 1990. Biomanipulation: the next phase – making it stable. Hydrobiologia 200(201): 13–27.
Shapiro, J. & D. I. Wright, 1984. Lake restoration by biomanipulation. Round Lake, Minnesota – the first two years. Freshwater Biology 14: 371–383.
Søndergaard, M., J. P. Jensen & E. Jeppesen, 2005. Role of sediment and internal loading of phosphorus in shallow lakes. Hydrobiologia 506/509: 135–145.
Søndergaard, M., E. Jeppesen, T. L. Lauridsen, C. Skov, E. H. van Nex, R. Roijackers, E. Lammens & R. Portielje, 2007. Lake restoration: successes, failures and long-term effects. Journal of Applied Ecology 44: 1095–1105.
Squires, L. & A. G. van der Valk, 1992. Water-depth tolerances of the dominant emergent macrophytes of the Delta Marsh, Manitoba. Canadian Journal of Botany 70: 1860–1867.
Swanson, G. A., 1978. A plankton sampling device for shallow wetlands. The Journal of Wildlife Management 78: 670–672.
Taper, M. L., 2004. Model selection from many candidates. In Taper, M. L. & S. R. Lele (eds), The Nature of Scientific Evidence: Statistical, Philosophical, and Empirical Considerations. University of Chicago Press, Chicaco, IL: 488–524.
Tonn, W. M. & J. J. Magnuson, 1982. Patterns in the species composition and richness of fish assemblages in northern Wisconsin lakes. Ecology 63: 1149–1166.
Van der Valk, A. G. & C. B. Davis, 1978. The role of the seed bank in the vegetation dynamics of prairie glacial marshes. Ecology 59: 322–335.
Van der Valk, A. G., 2005. Water-level fluctuations in North American prairie wetlands. Hydrobiologia 539: 171–188.
Wantzen, K. M., K. O. Rothhaupt, M. Mörtl, M. Cantonati, L. G. Tóth & P. Fisher, 2008. Ecological effects of water-level fluctuations in lakes: an urgent issue. Hydrobiologia 613: 1–4.
White, M. S., M. A. Xenopoulos, K. Hogsden, R. A. Metcalfe & P. J. Dillon, 2008. Natural lake level fluctuation and associated concordance with water quality and aquatic communities within small lakes of the Laurentian Great Lake region. Hydrobiologia 613: 21–31.
Weissgerber, T. L., N. M. Milic, S. J. Winham & V. D. Garovic, 2015. Beyond bar and line graphs: time for a new data presentation paradigm. PLoS Biol 13(4): e1002128. doi:10.1371/journal.pbio.1002128.
Wilcox, D. A. & J. E. Meeker, 1992. Implications for faunal habitat related to altered macrophyte structure in regulated lakes in northern Minnesota. Wetlands 12: 192–203.
Zimmer, K. D., M. A. Hanson & M. G. Butler, 2001. Effects of fathead minnow colonization and removal on a prairie wetland ecosystem. Ecosystems 4: 346–357.
Zimmer, K. D., M. A. Hanson, B. R. Herwig & M. L. Konsti, 2009. Thresholds and stability of alternative regimes in shallow Prairie-Parkland lakes of central North America. Ecosystems 12: 843–852.
Zimmer, K. D., M. A. Hanson & D. A. Wrubleski, 2016. Invertebrates in permanent wetlands (long-hydroperiod marshes and shallow lakes). In Batzer, D. & D. Boix (eds), Invertebrates in Freshwater Wetlands. Springer International Publishing, Switzerland: 251–286.
Acknowledgments
Many graduate and undergraduate students assisted with field and laboratory activities and tasks were often completed under difficult conditions. We especially thank Winston Allen, Cynthia Kuettel, Stefan Bischof, Douglas Galvas, Luke Ginger, Ariel Gittens, Jordan Goetting, Adam Johannsen, LaToya Kissoon, Kristine Maurer, Luke Nolby, Josh Norenberg, Jonathon Gustafson, Thomas Langer, Elisabeth McHale, Dustin Potter, Brandon Palesh, and Lauren Reuss. Research funding was provided by the Wildlife and Fisheries Research Units, Minnesota Department of Natural Resources, the Minnesota Environment and Natural Resources Trust Fund, and the University of St. Thomas. We also appreciate the cooperation of landowners who granted access to private property. Earlier drafts of this paper were improved considerably by review and comments from two anonymous reviewers.
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling editor: Chris Joyce
Rights and permissions
About this article
Cite this article
Hanson, M.A., Herwig, B.R., Zimmer, K.D. et al. Rehabilitation of shallow lakes: time to adjust expectations?. Hydrobiologia 787, 45–59 (2017). https://doi.org/10.1007/s10750-016-2865-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-016-2865-9