Abstract
Our study aimed to incorporate 60 newly acquired spring durum wheat genotypes into the Prague Gene Bank collection, focusing on 54 experimental breeding research genotypes and their technological quality contributions for conservation and future use. We analyzed a total of 11 field and 6 technological parameters of grain quality. Additionally, we identified the composition of high molecular weight glutenin subunits (HMW-GSs) in 13 accessions with contrasting technological characteristics. Two different extraction procedures were employed to detect HMW-GSs: one based on total protein extraction and the other on gliadin pre-extraction. The tested parameters exhibited a significant range of variability, with the relative standard deviation ranging from 2.1% for starch content to 96.7% for the gluten index. Additionally, six breeding research genotypes exhibited high technological grain quality comparable to the two modern durum wheat cultivars. Two accessions: M90-99–2 and IG 142076 showed above-average Zeleny sedimentation values, probably positively influenced by the presence of HMW-GSs A1: (2*). Genotypes PI 675012 and IG 142039 displayed the highest grain stability in technological parameters. Both extraction methods also detected the specific subunit B1: (6 + 8*) in the genotype IG 142039. Preserving these genotypes thus represents a substantial expansion of genetic resources in the genebank.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Durum wheat (Triticum durum, Desf.) is a traditional Mediterranean crop that falls under the category of tetraploid wheat (BBAA). It originated in the Fertile Crescent (10,000 BP) and spread over the northern side of the Mediterranean, reaching the Iberian Peninsula in about 7000 BP from both Italy and North Africa (Soriano et al. 2016).
In 2018, the European Union was the largest producer of durum wheat, with a total production of 9 million tonnes. Other top durum wheat producing countries included Canada, Turkey, United States, Algeria, Mexico, Kazakhstan, Syria, and India. The leading countries in durum wheat production within the European Union, based on average production in the last decade, are Italy, France, Greece, and Spain. Italy is considered the leader in durum wheat production, with an average production of 4.26 million tonnes in the last decade, covering 1.28 million hectares of growing area. France follows with an average production of 1.89 million tonnes and 0.37 million hectares of growing area. Greece with 0.98 million tonnes and Spain with 0.98 million tonnes also contribute significantly to durum wheat production within the European Union (Xynias et al. 2020).
Interestingly, a dynamic development in the cultivation of durum wheat has been observed in Slovakia, where the acreage has significantly increased to the current 65,000 hectares in recent years. With a production of 315,000 tons per year, Slovakia is already the fifth-largest producer of durum wheat in the EU (Babincová 2023). The increase in acreage is also being observed in other countries such as Austria and Germany.
The primary use of durum wheat is mainly for the production of various types of pasta. For less traditional uses of durum wheat, examples include two esteemed breads: “Pane di Altamura” (Altamura bread) and “Pagnotta del Dittaino” (Dittaino bread), both of which are crafted from durum wheat re-milled semolina, following a well-established bread-making tradition in Southern Italy (Giannone et al. 2018). In Algeria, a major consumer of durum wheat, the crop is often utilized in the production of couscous and various types of traditional flatbreads (Yesli et al. 2017).
In general, pasta quality is determined by three main factors: the raw materials, the production recipe, and the production process. Aspects of primary grain quality include predominantly visual appearance, test weight, weight of 1000 kernels, physical defects, vitreousness, moisture content, weather damage and grain protein percentage (Sissons 2008).
Modern Italian and Spanish durum wheat cultivars typically have a gluten index ranging from 55 to 87%, and SDS sedimentation is 30% higher compared to old durum wheat landraces. This indicates that modern cultivars have a stronger and more tenacious gluten, which is essential for producing high-quality pasta (Mefleh et al. 2019). There is much evidence of the influence of the allelic gluten protein composition (gliadins and glutenins) on the technological quality of durum wheat, since gluten quality strongly affects the firmness of pasta after cooking.
Glutenins are associated with the viscoelastic properties of gluten, while gliadins are responsible for gluten viscosity and extensibility. The two groups of single monomeric glutenins that have been separated based on their mobility in SDS-PAGE are high- and low-molecular-weight glutenin subunits (HMW-GSs) and (LMW-GSs), respectively. Both groups of glutenins are coded by orthologous genes located on the long and short arms of the group 1 chromosomes, respectively. LMW-GS are encoded by Glu-2 and Glu-3 genes, whereas HMW-GSs are encoded by Glu-1 genes. Gliadins are encoded by two loci (Gli-1 and Gli-2) located on the short arms of group 1 (-γ and -ω types) and 6 (-α and -β types) chromosomes (Roncallo et al. 2021; Aguiriano et al. 2008; Güleç et al. 2019).
Grain hardness is further a key determinant of milling performance for semolina production. Proteins responsible for determining grain texture (hardness) of wheat grain are called puroindolines. The higher level of puroindolines, the lower the hardness is. Absence of the genes coding for puroindolines in durum wheat is the reason for the very hard texture of this crop (Pasha et al. 2010).
There are several gene banks around the world where durum wheat landraces, wild accessions, breeding lines from crosses, obsolete cultivars and modern cultivars are preserved. Among the most important banks (based on the number and origin of accessions) are those hosted at CIMMYT (MEX), ICARDA (LBN), USDA-ARS (USA), and Australian Seed Bank Partnership. Durum wheat is represented with 20,779 accessions in CIMMYT collections. The ICARDA gene bank further preserves 18,276 accessions. The NSGC (National Small Grains Collection) at the USDA-ARS in Aberdeen holds 10,690 accessions of durum wheat and the AGG (Australian Grains GeneBank) in Canberra (Australia) includes 9,966 accessions of durum wheat. Significant numbers of durum wheat genetic resources can also be found in the gene banks of other countries such as Italy, Russia, India, Canada and Spain (Martínez-Moreno et al.2020).
The Global Durum Panel (GDP) was established in 2015 by the Expert Working Group “Durum wheat Genomics and Breeding” of the Wheat Initiative. Its purpose is to explore the wild relatives of tetraploid wheat to harness novel beneficial allelic diversity. The GDP with 2,500 accessions includes 2,130 worldwide durum wheat accessions and accessions of other related tetraploid species. Six sub-panels comprising 1,000 accessions are currently used for genome-wide association study (Martínez-Moreno et al. 2020).
One of the most extensive diversity analyses of tetraploid wheat species were carried out by Sansaloni et al. (2020) using DArTseq™ technology. The study analyzed 80 000 accessions including 18,946 domesticated tetraploid samples (in which 77.6% was T. durum). The analysis of tetraploid accessions showed, that the elite lines are distributed across almost the entire genetic diversity space of the landraces. However, there is a large subset of accessions collected in Ethiopia that forms an isolated cluster, whose genetic content is largely unexplored but definitely distinct from the other elite genotypes (Sansaloni et al. 2020).
Compared to the above mentioned collections and research, the collection of the Czech Gene Bank in Prague is more modest and contains about 1319 samples of Triticum durum, of which 1103 are spring forms (see GRIN CZECH 2023). About 79% of the total number of accessions was already part of this collection before the year 2000. The genotypes are interpreted in the current collection in 4 basic groups (1st group—Improved/commercial cultivars, 2nd group—Breeding research genotypes, 3rd group—Traditional landraces and the 4th group—Wild genotypes) with the current percentages: 1st group: 56%; 2nd group: 23%; 3rd group: 18% and 4th group: 3%. The significant prevalence of spring accessions at the Prague Gene Bank is due to the higher risk of overwintering associated with the weather conditions at the location of the gene bank.
According to Leonova et al. (2013), the strategy for preserving wheat collections involves seeking promising genotypes associated with enhanced resistance to lodging, leaf, yellow, and stem rusts, drought tolerance, as well as genes linked to higher protein concentration. This strategy aims to enhance biodiversity and considers the suitability of the newly obtained accessions for the climatic conditions of the Czech Republic. Simultaneously, the suitability of the newly obtained accessions for the cooler and precipitation-varying climatic conditions of the Czech Republic, which are traditionally perceived as riskier for durum wheat, is also considered.
From the above perspective of conserving valuable genetic resources for breeding, our study mainly focused on evaluating the impact of recently introduced experimental breeding genotypes of spring durum wheat. The objective was to assess their variability in selected field and technological parameters in the context of the currently modern cultivars of spring durum wheat, including the HMW-GSs composition of selected genotypes, which exhibited the highest technological grain quality. Simultaneously, the suitability of these genotypes for the climatic conditions of the Czech Republic, which are traditionally perceived as riskier for durum wheat, was also considered.
Materials and methods
Durum wheat accessions and cultivation
The selected set of 60 accessions of spring forms of durum wheat (Triticum turgidum subsp. Durum, Desf., van Slageren), preserved in the collections of the Gene Bank in Prague, included five modern cultivars classified into the 1st group -Improved/commercial cultivars (Toscadou—France, Casteldoux—France, Durofinus—Austria, Tamadur—Austria, Durafox—Austria), one non-traditional wild accession classified into the 4th group - Wild genotypes (Tan Blue—USA) and 54 experimental breeding research genotypes classified into the 2nd group - Breeding research genotypes. The whole set originated from 10 countries (Austria, Japan, Israel, Great Britain, Mexico, Syria, Czech Republic, Germany, USA, and France), with a predominance of Syrian “IG…” (23 accessions) and Japanese “ANDW…” (22 accessions) research breeding genotypes. The additional evaluated research genotypes included four accessions originating from GB designated as “PI…”, two accessions D-3 and D-9 originating from Israel, one genotype KM 35–16 originating from Germany, one genotype M90-99–2 durum originating from CR, and one genotype Joric-21 originating from Mexico.
The specific genetic background was more defined only for 2 groups of breeding research genotypes. ANDW (Analog of durum wheat), involved near-isogenic lines incorporating selected genes such as for black glume (Bg), hairy glume (Hg), glaucous leaf character (w1, lw1), spherical grain (S2, S3), or brittle rachis (Br2, Br3) into the genetic background of the spring hard wheat genotype LD222. The mutation in the gw2-A1 allele of the four PI… genotypes significantly increases grain weight, including its width and length. The Gene Bank in Aleppo, from which the collection of Syrian genotypes designated as (IG…) was obtained, unfortunately no longer exists. Owing to the origin of these genotypes, it is reasonable to expect a high degree of adaptability to arid environmental conditions. These genotypes were progressively incorporated into the genetic resource collections of the Gene Bank in Prague in recent years, particularly from 2015 to 2018, through systematic and ongoing inclusion processes.
The list of all 60 evaluated genotypes is presented in Fig. 1 and is readily accessible in the public genetic resource database, including individual passport data (see GRIN CZECH 2023). Subsequent to their acquisition, these accessions were gradually planted in non-replicated, three-year field trials on a 4.5 m2 plot in Prague-Ruzyně between 2016 and 2021. The genotypes were evaluated in 3 different three-year annual periods designated as A: 2016–2018, B: 2018–2020 and C: 2019–2021. 2 genotypes (cultivars) were evaluated in the case of period A, 22 genotypes in the case of period B and a total of 36 durum wheat genotypes in period C. The individual distribution of each genotype into the respective evaluation period is shown in Figs. 1 and 2. A total of 58 genotypes were overlapped annually in the two-year period 2019—2020. In each year, peas were cultivated as the preceding crop. The crop management included the necessary application of herbicides and a single nitrogen dose of 30 kg.ha−1. In accordance with standard genetic resource assessment procedures, no insecticidal or fungicidal control was applied.
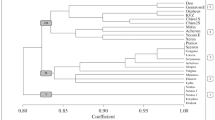
The heat map, in conjunction with cluster analysis, depicts the interrelationships among durum wheat accessions based on 17 monitored parameters (rows), as well as the simultaneous associations among the parameters themselves (columns). PH plant height, DH days to heading, ST starch content, CP crude protein content, ZS Zeleny sedimentation test, HI Harvest index, GWS grain weight per spike, PM presence of powdery mildew, GY grain yield, PSI Particle Size Index, SL spike length, CL crop lodging, GI gluten index, GFP grain filling period, TGW thousand grain weight, GN grain number, WG wet gluten content, Cl. cluster
Locality and agronomic characteristics
Prague–Ruzyně is a sugar beet-growing region (altitude: 340 m; yearly average precipitation: 472 mm; annual mean air temperature: 8.4 °C; soil type: Orthic Luvisol).
The agronomic characteristics corresponding with standard wheat genetic resources evaluation of Gene bank of Prague were analyzed in all samples and included following morphological and phenological parameters: Plant height (PH), Spike length (SL), Thousand grain weight (TGW), Grain number (GN), Grain weight per spike (GWS), Harvest index (HI), Days to heading (DH), Grain filling period (GFP) and recalculated Grain yield (GY). The occurrence of grass mildew was also evaluated for resistance on a scale from 1 (low resistance) to 9 (high resistance). The presence of powdery mildew (Blumeria graminis)—(PM) and crop lodging (CL) was assessed using a rating scale 1–9, where 1 indicated high susceptibility to PM or CL and 9 indicated low susceptibility to CL or high resistance to PM.
Grain quality parameters
The assessment of grain quality parameters involved the determination of various factors such as crude protein content (CP) using the Kjeldahl method (ČSN EN ISO 20483), wet gluten content (WG) and gluten index (GI) based on the Curic et al. (2001) method, sedimentation index using the Zeleny sedimentation test (ZS) (ČSN ISO 5529), starch content (ST) measured by Ewers polarimetric methods in accordance with ČSN EN ISO 10520, and Particle Size Index (PSI) according to AACC Method 55–30 for assessing wheat hardness.
Electrophoretic analyses of high-molecular-weight glutenin subunits
Two extraction procedures were employed to enhance the reliable identification of high-molecular-weight glutenin subunits (HMW-GSs) in prepared flour samples derived from a blend of seeds from selected genotypes exhibiting above-average technological parameters. Whole storage protein extraction, followed by electrophoresis, was performed using SDS-PAGE, according to the protocol outlined by Gao et al. (2018) with minor modifications. Briefly, one hundred individual grains from each accession were pulverized using an IKA mill, and 40 mg of the resulting sample was used for whole storage protein extraction. For each sample, 0.3 ml of extraction solution (composed of SDS-PAGE sample buffer, 2% SDS, 10% glycerol, 5% 2-mercaptoethanol, 0.002% bromophenol blue, and 62.5mM Tris-HCl at pH 6.8) was used. The samples were subjected to a 2-h extraction at refrigerated conditions and subsequently heated at 100 °C for 2 min, followed by centrifugation at 15,000 RPM for 4 min. The sequential procedure performed in accordance with the method of Singh et al. (1991) involved pre-extraction with 50% propan-1-ol in identically pulverized samples as mentioned above.
Electrophoretic separation and HMW-GSs identification were conducted in a manner identical to that used for the separation of whole storage proteins.
Electrophoresis was carried out at a constant current of 30 mA per gel (dimensions: 13 × 15 × 0.175 cm) at 10 °C. The HMW-GSs at Glu-A1 and Glu-B1 loci were identified following the nomenclature established by Payne and Lawrence (1983).
Statistical analysis
The software Statistica 7.1 CZ was used for basic statistical parameters: minimum (Min.), maximum (Max.), mean, standard deviation (SD), relative standard deviation (RSD), Spearman‘s correlation index and Fisher LSD post-hoc comparison test. The heat map was constructed using a freely available web server: “Heatmapper” which was developed by Babicki et al. (2016). The created clusters of durum wheat accessions according to the variability of 17 monitored parameters were obtained using the Complete Linkage method and similarity calculation according to Euclidean distance.
Results
Basic statistical characteristics of monitored parameters and mutual comparison of durum wheat research breeding genotypes with other accessions
The three-year average, minimum, and maximum values of the parameters, as well as their inter-variety variability within the durum wheat set, are presented in Table 1. The range of variability (RSD) across monitored field parameters varied, ranging from 5.1% (days to heading) to over 20% for parameters such as plant height, spike length, grain yield, particle size index, and susceptibility to powdery mildew. The RSD range for grain technology parameters was even more extensive, spanning from 2.1% (starch content) to 45.6% (Zeleny sedimentation) or even up to 96.7% (gluten index). The typical characteristics of durum wheat were also affirmed by the observed higher grain hardness (PSI = 8.5%) and elevated protein content (15.5%).
In Fig. 1, the individual differences and similarities among the evaluated durum wheat genotypes are visually represented based on 3-year averages of 17 parameters transformed into the Z-score system followed by the heat map method. This method allows for a visual representation of the relationships and patterns among the durum wheat genotypes based on the 17 parameters. Additionally, a cluster analysis (Euclidean distance) was applied, which grouped the durum wheat genotypes horizontally and the monitored parameters vertically into clusters of varying similarity. According to this horizontal cluster analysis, the set of 60 genotypes was categorized into five distinct clusters (Cl. I—Cl. V) based on the values of parameters achieved.
The analysis confirmed that the breeding research genotypes labelled as IG (Cl. III and IV) and ADNV (Cl. V) exhibited very similar characteristics and were grouped into common clusters. The main difference between these clusters (Cl. III—V) was primarily related to the varying plant height in the ADNV group of genotypes and the earliness of the IG series of Syrian genotypes (observed in Cl. V: PH > 100 cm) (seen in Cl. IV: DH < 58 days). Only the ANDW 11A, ANDW 4 K, and ANDW 3C/4A accessions displayed even closer relationships with the Syrian items, leading to their assignment to the common Cl. IV.
The four modern commercial durum wheat cultivars (Toscadou, Tamadur, Durofinus, and Durafox) located in Cl. I confirmed the best field and technological parameters. These mentioned standard cultivars exhibited higher resistance to powdery mildew (scoring 7.0—8.7), increased spike productivity (GWS = 1.5—1.7 g) and higher total yield (6.3—7.0 t.ha−1).
In this cluster (Cl. I), there were six other experimental breeding research genotypes designated as PI…, D-3, and D-9. When compared to the commercial cultivars, these breeding research genotypes (Cl. I) showed similar grain quality, a reduced tendency to lodging, but lower spike productivity and yield. Common characteristics of the genotypes in Cluster I, compared to other clusters, included shorter plant height, earlier heading time, lower protein content (CP < 14.2%), but higher technological quality of protein (WG > 28%; GI > 66%).
In a more detailed comparison, especially focusing on technological parameters, it's worth noting the breeding research genotype M90-99–2, being of Czech origin, which constituted a distinct cluster, designated as Cl. II. This particular genotype stood out due to its exceptionally high Zeleny sedimentation (ZS) value of 63.8 ml. Also, it displayed above-average contents of crude protein (CP) at 17.7%, wet gluten (WG) at 39%, and gluten index (GI) at 75.9%. It also exhibited higher sensitivity to powdery mildew (3 points).
Interrelationships among monitored parameters based on cluster and correlation analysis
The cluster analysis grouped the 17 observed parameters into three similar clusters (Cl. 1–3, as depicted in Fig. 1), uncovering several close relationships among some of the observed parameters. For instance, in Cl. 2, production parameters such as harvest index (HI), grain yield (GY), and spike length (SL) exhibited expected close associations with parameters related to higher resistance against common leaf rust (CL) or powdery mildew (PM). Similarly, closer relationships between thousand grain weight (TGW), grain number (GN), and grain weight per spike (WG) with the grain filling period (GFP) can be observed in cluster 3 (Cl. 3).
The correlation analysis presented in Table 2 reveals individual relationships between pairs of parameters. There are several significant and very strong correlations worth highlighting. For instance, there is a strong negative correlation between plant height (PH) and lodging (CL) (r = − 0.76), as well as a negative relationship between days to heading (DH) and grain filling period (GFP) (r = − 0.80). A highly negative correlation was also observed between starch content (ST) and crude protein content (CP) (r = − 0.87). Interestingly, a significant positive correlation exists between grain weight per spike (GWS) and crude protein content (CP) (r = 0.77). Furthermore, a positive correlation is observed between the parameters of the Zeleny sedimentation test (ZS) and the values of gluten index (GI) (r = 0.51).
The correlation coefficients further revealed that shorter plant height, more compact spikes, higher TGW, and greater grain weight per spike (GWS) were significant factors contributing to intensified yields.
The relationship between the days to heading (DH) and grain-filling period (GFP) parameters, and how they affect the total yield, was also fascinating. The results of the correlation analysis suggested that higher yields were achieved with earlier-maturing genotypes (r = − 0.58) while maintaining a longer grain-filling period (r = 0.57).
Variability, clustering, and identification of promising breeding research genotypes based on high technological quality of gluten
The variability of the tested genotypes in relation to the achieved technological grain parameters influenced by protein properties is depicted in Fig. 2 using a heat map combined with cluster analysis. There are 4 main clusters. Clusters I and II closely correspond to the distribution of the set when evaluating all 17 parameters. This indicates that these parameters had the most significant impact on their collective clustering.
As mentioned above, the breeding research genotype M90-99–2, which exhibits above-average levels of protein (17.7%), wet gluten (39.8%), GI (75.9%), and the previously mentioned extremely high ZS content (63.8 ml), is isolated within Cluster I. Cluster II comprises 10 genotypes, including 4 modern registered cultivars. The predominant characteristic of these genotypes is the high firmness of gluten, as indicated by their above-average GI values within the tested cluster (ranging from 71.2 to 86.4%). Three accessions, known as PI 675010, PI 675011, and PI 675012, even surpassed the GI levels of the commercial cultivars, albeit not significantly (Table 3).
From a technological perspective focused on achieving higher grain quality, it's noteworthy that two major research breeding genotypes, IG 14276 and IG 14239, have above-average ZS values. Additionally, IG 14239 demonstrated a higher GI of 55%. Therefore, this particular genotype can be considered almost equivalent to high-quality commercial cultivars from a technological point of view (Table 3).
Given the greater variability in weather conditions in the central European region compared to southern Europe, it is also advisable to evaluate the stability of these key parameters for genetic resources selected for their higher technological quality (see Table 3). The level of inter-annual variability (RSD) among the selected genotypes varied individually, ranging from single digits to low tens of percentages. Furthermore, by summing the RSD values for the four chosen parameters, we identified the two most technologically stable genotypes (PI 675012 and IG 142039), which exceeded the performance of the modern cultivars included in the study (Table 3).
Composition of high molecular weight glutenin subunits in durum wheat accessions with promising gluten quality
The polymorphism of high molecular weight glutenin subunits (HMW-GSs) in a selection of technologically superior genotypes (see Tab. 3) is illustrated in Fig. 3A and B. Two procedures, total protein extraction and a sequential method involving gliadin pre-extraction (currently more frequently applied), were utilized. The most prevalent combinations of HMW-GSs, A1: (0) and B1: (6 + 8), were consistently identified in 8 out of the 13 chosen genotypes using both methods. Furthermore, both approaches confirmed the presence of the A1: (0) and B1 (7 + 8) combination in the commercial cultivars Durofinus and Toscadou.
HMW-GS polymorphism in 13 selected durum wheat accessions with the highest gluten technological quality. A: Whole storage proteins B: Sequential procedure after removal of gliadins. *T. Aestivum with declared HMW-GSs composition A1: 0; B1: (6 + 8); D1: (2 + 12). Arrows between band positions B1: (6 + 8) identify presences of other HMW-GSs. Presence of ω gliadins below position of HMW-GS D1: (12) are also marked with arrows
Additional distinct HMW-GSs were identified using both methods for IG 142076 and M90-99–2. IG 142076 exhibited an HMW-GS composition of A1: (2*) and a double-line composition for B1: (7 + 8)/(7 + 9). Genotype M90-99–2 featured a multi-line composition for both A1: (1)/(2*) and B1: (7 + 8)/(7 + 9). Both extraction methods further confirmed a specific combination B1: (6 + 8*)in IG 142039. Precise identification of the 8* band position is without a defined standard challenging.
The analysis of HMW-GS polymorphisms, based on two different extraction procedures, revealed slight variations. In the (6 + 8) subunit, a weaker band was frequently observed at the B1: (7) position of HMW-GSs in Durafox, Tamadur, and D-9, and two weaker bands were observed at the B1: (7) and (an unidentified band) above the B1: (8*) band position, respectively, in all four PI-labeled British accessions and the D-3 genotype. However, the multi-line composition of the HMW-GSs was confirmed through sequence analysis for the PI 675012 accession only. On the other hand, this method revealed the double-line composition of HMW-GSs B1: (6 + 8*)/(7 + 8*) in IG 142039.
Another distinction between the two methods can be observed in the presence of one or two high molecular weight bands located below the D1:(12) band position, which were solely identified in the total protein extraction procedure (Fig. 3A).
Additionally, it is noteworthy that the presence of the A1: 2* allele with the double-line combination B1: (7 + 8)/(7 + 9) in the IG 142076 and M90-99–2 genotypes is associated with a high Zeleny sedimentation rate (IG 142076 = 37.3 ml and M90-99–2 = 63.8 ml) compared to the other selected accessions.
Discussion
Indeed, conducting a comprehensive comparison of the monitored parameters and their variability with other durum wheat accessions stored in the Prague Gene Bank is challenging. Up until 2013, the cereal collections of the Gene Bank were only evaluated for a period of 2 years. In previous years, certain technological parameters (e.g. Zeleny sedimentation, PSI) were also not determined. As a result, the obtained results are mainly compared with the published findings that declare the complexity of traits or a 3-year evaluation.
So, it can be concluded that the 3-year means and variability (RSD) of morphological parameters (e.g. plant height, thousand kernel weight, number of kernels and kernel weight per spike) were significantly lower in our tested durum wheat population (Table 1) compared to a large selection of durum wheat genetic resources from the Italian gene bank (Pignone et al. 2015). This is likely attributed not only to the low number of samples evaluated but also to the significant presence of breeding research genotypes with similar genetic backgrounds in this dataset. Conversely, when comparing these results to the variability observed for selected field parameters and protein content of winter wheat cultivars (T. aestivum L.) grown at the same location (Prague—Ruzyně), a similar level of variability was confirmed (Dotlačil et al. 2010). The very high variability in the observed grain quality parameters (GI and ZS, see Table 1) is likely primarily associated with the genetic variability of the selected genotypes. Furthermore, extensive environmental experiments in durum wheat indicate that factors such as thousand grain weight (TGW), vitreousness, falling number, or protein content are influenced more by the interaction of environmental conditions and the specific year rather than by genotype (Moayedi et al. 2021). Hence, the less stable weather conditions in Central Europe during the summer months, compared to regions like the Mediterranean, may also significantly contribute to the higher variability of these parameters. It is worth mentioning the mean value of Zeleny sedimentation test (ZS = 16.4 ml) and gluten index (GI = 27.1%) of the entire set (Table 1) did not meet the acceptable purchasing standards for durum wheat processors. Nevertheless, due to the extensive variability in both traits, we have identified several promising breeding research genotypes (refer to Figs. 1 and 2) with corresponding gluten strength, alongside modern cultivars (Mefleh et al. 2019).
The relationships obtained between the tested parameters (Fig. 1 and Table 2) cannot be fully generalized due to the limited and somewhat randomly selected set of genotypes evaluated in this study. Simultaneously, it is important to acknowledge that the Spearman correlations of the three-year averages of the monitored parameters, calculated for genotypes with only partial agreement in the growing period, are somewhat inaccurate. This inaccuracy is expected to be more significant for parameters that heavily rely on environmental conditions, such as phenological parameters, crude protein, or starch content. Therefore, only the strongest correlations and relationships between ZS or GI parameters are discussed here, with the anticipated stronger influence of genotype. Nevertheless, the results from the cluster and correlation analysis of the relationships between the observed parameters can serve as a certain indicator of breeding strategy in the development and selection of durum wheat cultivars. The identified relationships also enable predictions about the traits of future cultivars that will be incorporated into genetic resource collections.
Emphasized high positive or negative correlations among certain pairs of parameters (plant height vs. lodging, grain filling period and days to heading, protein content vs. starch content) are commonly confirmed as noted in previous research. The breeding strategy for durum wheat faces uncertainty regarding the moderately strong relationship between Zeleny sedimentation and gluten index, a relationship that is also commonly observed in common wheat (Najafian 2012). At the same time, the observed correlations between wet gluten and GI (r = 0.03) or ZS (r = 0.09) are very low, indicating a small effect of gluten content on the baking quality of durum wheat grain compared to the importance of gluten content for common wheat (Dvořáček et al. 2008).
The breeding benefits for durum wheat are well evident in a separate group of 10 entries (4 commercial cultivars and 6 breeding research genotypes) in Cl. I (Fig. 1). The integration of rht (dwarfing) genes (Isidro et al. 2011) is apparent, resulting in increased material productivity. Additionally, there is a clear focus on effective selection for appropriate grain hardness and superior gluten quality, as demonstrated here (Mefleh et al. 2019).
In terms of promising breeding research genotypes, in addition to the 6 items listed in Cl. 1, there were three other breeding research genotypes with above-average values in some of the ZS or GI sub-technological parameters (Table 3). Among the 54 evaluated breeding research genotypes, one-sixth can be identified as potential donors, particularly for significant technological parameters. It is essential to note that if these accessions are included, comprehensive molecular analyses should run parallel with their phenotypic evaluation. By combining both approaches, we can ensure the optimal inclusion of these genotypes and avoid potential duplications that could unnecessarily increase the cost of genetic resource conservation.
The most common HMW-GS combinations A1: (0) and B1: (6 + 8) detected in our sample collection (see Fig. 3A and B) were also found to be prevalent in a large collection of 196 durum wheat samples, as reported by Argentine and Spanish authors (Roncallo et al. 2021; Aguiriano et al. 2008).
A similar position of the 8* band was reported by Aguiriano et al. (2008) in Dawbull common wheat with the B1: (6* + 8*) combination. According to the combination scheme of HMW-GSs for durum wheat in Güleç et al. (2019), this band’s position would most closely correspond to subunit B1: (18). Roncallo et al. (2021) detected a unique HMW-GS B1: (14 + 22) configuration in Italian T. durum genotypes, and the position of band 22 notably aligns with the position marked as 8* in our study.
The disappearance of certain faint bands on the gel may be due to the flexible boundary between the solubility of different protein fractions. Hamer (2003) described this phenomenon with the statement: “nothing is absolute.” Furthermore, they explained that even the pre-extracted ethanol fraction may contain some HMW glutenins. This transition of HMW glutenins was also affirmed by Cinco-Moroyoqui and MacRitchie (2008) when employing 50% propanol. Consequently, low concentrations of HMW-GSs from under-represented sister lines could potentially be excluded during sequence analysis when gliadins are removed, leading to a loss of information regarding the multi-line composition of HMW-GSs in the tested genotypes. On the other hand, the different findings in the case of IG 142039 using sequential analysis can be attributed to the non-uniformity of the prepared sample.
These specific band positions do not have standard nomenclature within the glutenin subunit classification (Payne and Lawrence 1983). Margiotta et al. (1996) exclusively noted similar band positions in wild-type T. urartum and T. dicoccoides materials and referred to them as HMW-GSs A1: y-type. The absence of these bands in the case of the sequential extraction method strongly suggests that they belong to a group of high molecular weight ɷ gliadins, where the largest peptides under SDS-PAGE conditions are typically found near HMW-GSs D1: (10 or 12), as reported by Žilić (2013). The high solubility of ɷ gliadins in an aqueous propanol solution was further confirmed by Cinco-Moroyoqui and MacRitchie (2008).
The documented positive impact of the 2* allele, particularly on the micro-sedimentation test in durum wheat, is also suggested by the research of Roncallo et al. (2021). In contrast, both breeding genotypes studied exhibited a tendency toward softer grains (M90-99–2: PSI = 14.2%, IG 142076: 16.6%), which implies they may be better suited for standard white flour production (compared to semolina) and for the development of new durum wheat cultivars intended for bakery production.
The yearly routine characterization of HMW-GS polymorphisms in a large and genetically diverse collection of durum wheat genetic resources in gene banks requires reliable methods that are both cost-effective and time-efficient. Both methods based on the total or sequential extraction of HMW-GS only partially fulfil this requirement. The method involving total protein extraction from a grain mixture appears to be more sensitive in HMW-GS identification and is also more time and cost-efficient compared to the sequential method. On the other hand, sequence analysis allows for a detailed description of LMW-GS composition, which is particularly crucial for the comprehensive prediction of the technological quality of durum wheat proteins. However, in genetically diverse genotypes, identifying primarily LMW-GSs in a mixed sample can be very complex and time-consuming. Hence, the utilization of DNA markers for identifying HMW-GSs (see Nucia et al. 2019), coupled with the development of reliable DNA markers for more complex LMW-GSs, could offer a promising solution. Nevertheless, developing these molecular markers for durum wheat will not be feasible without parallel identification of both HMW- and LMW-GSs at the protein level.
Conclusion
The evaluation of the current durum wheat genetic resources has revealed a wide range of variability in both field and, more significantly, technological grain parameters. Several perspective parameters were detected in the tested breeding research genotypes. First, the Syrian accessions labelled as IG exhibited the highest earliness of heading. Additionally, we identified six promising genotypes with technological grain quality comparable to modern sown wheat cultivars. Simultaneously, PI 675012 and IG 142039 demonstrated higher annual stability in four crucial technological grain parameters compared to modern cultivars. Furthermore, accessions M90-99–2 and IG 142076 displayed above-average ZS values, potentially influenced positively by the presence of HMW-GSs A1: (2*).
Selected breeding research genotypes with higher technological quality confirmed the multi-line composition of HMW-GSs, particularly in the case of the total protein extraction procedure. The total protein extraction method appeared to be more sensitive in detecting durum wheat genotypes with multiple-line composition of HMW-GSs compared to the sequential method. In contrast, both methods were consistent in detecting the presence of the specific subunit B1: (6 + 8*) in genotype IG 142039.
Conversely, these results confirmed the high similarity of the 17 basic parameters within the groups of jointly derived breeding research genotypes. On the other hand, the variability of primarily technological grain parameters across these groups was very wide. The identification of 9 promising research breeding genotypes with comparable, or even superior in some aspects, technological grain quality compared to current commercial cultivars, indicates that these genetic resources may offer a range of valuable basic parameters. Their preservation in durum wheat collections therefore remains essential.
References:
Aguiriano LE, Ruiz VM, Fité SM, Carrillo BJM (2008) Genetic variation for glutenin and gliadins associated with quality in durum wheat (Triticum turgidum L. ssp. turgidum) landraces from Spain. Span J Agric Res 6:599–609. https://doi.org/10.5424/sjar/2008064-353
Babicki S, Arndt D, Marcu A, Liang Y, Grant JR, Maciejewski A, Wishart DS (2016) Heatmapper: web-enabled heat mapping for all. Nucleic Acids Res 44:W147–W153
Babincová Z (2023). Súpis plôch osiatych poľnohospodárskymi plodinami k 20. 5. 2023 (in Slovak). Statistical office of the Slovak Republic, 2023. https://slovak.statistics.sk/wps/portal/ext/products/publikacie/. Accessed 15th October 2023.
Cinco-Moroyoqui FJ, MacRitchie F (2008) Quantitation of LMW-GS to HMW-GS ratio in wheat flours. Cereal Chem 85:824–829. https://doi.org/10.1094/CCHEM-85-6-0824
Curic D, Karlovic D, Tusak D, Petrovic B, Dugum J (2001) Gluten as a standard of wheat flour quality. Food Technol Biotechnol 39:353–362
Dotlačil L, Hermuth J, Stehno Z, Dvořáček V, Bradová J, Leišová L (2010) How can wheat landraces contribute to present breeding? Czech J Genet Plant Breed 46:S70–S74. https://doi.org/10.17221/1519-CJGPB
Dvořáček V, Kodeš A, Stehno Z, Hučko B, Mudřík Z (2008) Nutritive effect of protein composition and other grain properties of doubled haploid wheat lines with/without translocation 1B/1R in a model feeding test. Czech J Anim Sci 53:487–498. https://doi.org/10.17221/343-CJAS
Gao Z, Tian G, Wang Y, Li Y, Cao Q, Han M, Shi Z (2018) Allelic variation of high molecular weight glutenin subunits of bread wheat in Hebei province of China. J Genet 97:905–910. https://doi.org/10.1007/s12041-018-0985-x
GRIN CZECH (2023) Germplasm Resources Information Network, Czech database, constantly updated. https://grinczech.vurv.cz/gringlobal/search.aspx. Accessed 25th October 2023
Giannone V, Giarnetti M, Spina A, Todaro A, Pecorino B, Summo C et al (2018) Physico-chemical properties and sensory profile of durum wheat Dittaino PDO (protected designation of origin) bread and quality of re-milled semolina used for its production. Food Chem 241:242–249. https://doi.org/10.1016/j.foodchem.2017.08.096
Güleç TE, Yildrim A, Koyuncu M, Sayaslan A (2019) Analysis of some Turkish durum wheat cultivars and landraces for HMW-glutenin subunits. J Agric Fac Gaziosmanpaşa Univ (JAFAG) 36:74–80
Hamer RJ (2003) Fractionation Techniques. In: Shewry, P.R. Lookhart, G.L. (eds.) Wheat gluten protein analysis. American Association of Cereal Chemists, pp 19–29
Isidro J, Alvaro F, Royo C, Villegas D, Miralles DJ, García del Moral LF (2011) Changes in duration of developmental phases of durum wheat caused by breeding in Spain and Italy during the 20th century and its impact on yield. Ann Bot 107:1355–1366. https://doi.org/10.1093/aob/mcr063
Leonova IN, Badaeva ED, Orlovskaya OA, Röder MS, Khotyleva LV, Salina EA, Shumny VK (2013) Comparative characteristic of Triticum aestivum/Triticum durum and Triticum aestivum/Triticum dicoccum hybrid lines by genomic composition and resistance to fungal diseases under different environmental conditions. Russ J Genet 49:1112–1118. https://doi.org/10.1134/S1022795413110136
Margiotta B, Urbano M, Colaprico G, Johansson E, Buonocore F, D’Ovidio R, Lafiandra D (1996) Detection of y-type subunit at the Glu-A1 locus in some Swedish bread wheat lines. J Cereal Sci 23:203–212. https://doi.org/10.1006/jcrs.1996.0021
Martínez-Moreno F, Solís I, Noguero D, Blanco A, Özberk İ, Nsarellah N et al (2020) Durum wheat in the mediterranean rim: historical evolution and genetic resources. Genet Resour Crop Evol 67:1415–1436. https://doi.org/10.1007/s10722-020-00913-8
Mefleh M, Conte P, Fadda C, Giunta F, Piga A, Hassoun G, Motzo R (2019) From ancient to old and modern durum wheat varieties: Interaction among cultivar traits, management, and technological quality. J Sci Food Agric 99:2059–2067. https://doi.org/10.1002/jsfa.9388
Moayedi S, Elias EM, Manthey FA (2021) Effect of weather on grain quality traits of durum wheat grown in the Northern Plains of USA. Am J Plant Sci 12:1894–1911. https://doi.org/10.4236/ajps.2021.1212131
Najafian G (2012) Study of relationship among several bread making quality assessment indices in hexaploid wheat (Triticum aestivum L.) using correlation analysis. Qual Assur Saf Crops Foods 4:148–148. https://doi.org/10.1111/j.1757-837X.2012.00156.x
Nucia A, Okoń S, Tomczyńska-Mleko M (2019) Characterization of HMW glutenin subunits in European spring common wheat (Triticum aestivum L.). Genet Resour Crop Evol 66:579–588. https://doi.org/10.1007/s10722-018-00733-x
Pasha I, Anjum FM, Morris CF (2010) Grain hardness: a major determinant of wheat quality. Food Sci Technol Int 16:511–522. https://doi.org/10.1177/1082013210379691
Payne PI, Lawrence GJ (1983) Catalogue of alleles for the complex gene loci, Glu-A1, Glu-B1, and Glu-D1 which code for high-molecular-weight subunits of glutenin in hexaploid wheat. Cereal Research Communications 29–35.
Pignone D, De Paola D, Rapanà N, Janni M (2015) Single seed descent: a tool to exploit durum wheat (Triticum durum Desf.) genetic resources. Genet Resour Crop Evol 62:1029–1035. https://doi.org/10.1007/s10722-014-0206-2
Roncallo PF, Guzmán C, Larsen AO, Achilli AL, Dreisigacker S, Molfese E et al (2021) Allelic variation at glutenin loci (Glu-1, Glu-2 and Glu-3) in a worldwide durum wheat collection and its effect on quality attributes. Foods 10:2845. https://doi.org/10.3390/foods10112845
Sansaloni C, Franco J, Santos B, Percival-Alwyn L, Singh S, Petroli C et al (2020) Diversity analysis of 80,000 wheat accessions reveals consequences and opportunities of selection footprints. Nat Commun 11:4572. https://doi.org/10.1038/s41467-020-18404-w
Singh N, Shepherd K, Cornish G (1991) A simplified SDS-PAGE procedure for separating LMW subunits of glutenin. J Cereal Sci 14:203–208. https://doi.org/10.1016/S0733-5210(09)80039-8
Sissons M (2008) Role of durum wheat composition on the quality of pasta and bread. Food 2:75–90
Soriano JM, Villegas D, Aranzana MJ, García del Moral LF, Royo C (2016) Genetic structure of modern durum wheat cultivars and Mediterranean landraces matches with their agronomic performance. PLoS ONE 11:e0160983. https://doi.org/10.1371/journal.pone.0160983
Xynias IN, Mylonas I, Korpetis EG, Ninou E, Tsaballa A, Avdikos ID, Mavromatis AG (2020) Durum wheat breeding in the Mediterranean region: current status and future prospects. Agronomy 10:432. https://doi.org/10.3390/agronomy10030432
Yesli A, Latati M, Tellah S, Abdellaoui Z, Ounane G (2017) Physicochemical and rheological properties and bread-making potential of durum flour and semolina. J Food Agric Environ 15:14–20
Žilić S (2013) Wheat gluten: Composition and health effects. In: Walter DB (ed.) Gluten. Nova Science Publishers, Inc, pp 71–86
Acknowledgements
We are grateful to the Gene Bank team in Prague for providing comprehensive access to the archived data of durum wheat genetic resources.
Funding
Open access publishing supported by the National Technical Library in Prague. This research study was funded by the Ministry of Agriculture of the Czech Republic via the project number QK22010029 and via institutional support under project number MZE-RO0423.
Author information
Authors and Affiliations
Contributions
VD: Experimental planning, statistical processing and data design. Manuscript writing. JH Conducting field experiments, processing and evaluation of field parameters. MJ (Jelínek): Laboratory evaluation of technological parameters and participation in isolation of grain storage proteins for electrophoretic analysis of HMW-GSs polymorphism. MJ (Jágr): Performance and evaluation of HMW-GSs polymorphism using vertical electrophoresis.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Dvořáček, V., Jelínek, M., Jágr, M. et al. The benefits of experimental breeding research genotypes for the genetic resources of spring durum wheat. Genet Resour Crop Evol (2024). https://doi.org/10.1007/s10722-024-01946-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10722-024-01946-z