Abstract
Compromised skin integrity of farmed Atlantic salmon, commonly occurring under low temperature and stressful conditions, has major impacts on animal welfare and economic productivity. Even fish with minimal scale loss and minor wounds can suffer from secondary infections, causing downgrading and mortalities. Wound healing is a complex process, where water temperature and nutrition play key roles. In this study, Atlantic salmon (260 g) were held at different water temperatures (4 or 12 °C) and fed three different diets for 10 weeks, before artificial wounds were inflicted and the wound healing process monitored for 2 weeks. The fish were fed either a control diet, a diet supplemented with zinc (Zn) or a diet containing a combination of functional ingredients in addition to Zn. The effect of diet was assessed through subjective and quantitative skin histology and the transcription of skin-associated chemokines. Histology confirmed that wound healing was faster at 12 °C. The epidermis was more organised, and image analyses of digitised skin slides showed that fish fed diets with added Zn had a significantly larger area of the epidermis covered by mucous cells in the deeper layers after 2 weeks, representing more advanced healing progression. Constitutive levels of the newly described chemokines, herein named CK 11A, B and C, confirmed their preferential expression in skin compared to other tissues. Contrasting modulation profiles at 4 and 12 °C were seen for all three chemokines during the wound healing time course, while the Zn-supplemented diets significantly increased the expression of CK 11A and B during the first 24 h of the healing phase.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Impaired skin integrity is common in Atlantic salmon (Salmo salar L.) farming, especially during transfer to seawater and at low water temperatures. In addition to alterations in environmental conditions, intensively farmed fish are inevitably exposed to a variety of stressors including transportation, grading, vaccination and infectious agents that can potentially traumatise the integument. This can have a negative impact on fish welfare and the economical aspect of farming. At low temperatures, salmon are more susceptible to skin lesions, such as winter ulcers associated with infection of Moritella viscosa (Benediktsdóttir et al. 1998; Løvoll et al. 2009) and other pathogens. Red mark syndrome, also known as cold water strawberry disease, causes skin problems in rainbow trout (Oncorhynchus mykiss) at low temperatures (Metselaar et al. 2010; Verner-Jeffreys et al. 2008).
Open wounds can act as portals of entry for pathogens, with fast closure being of importance to avoid secondary infections. Detachment of scales further impairs the integrity of the epidermal and the superficial dermal layers. Small wounds and scale loss are common in fish skin and can heal rapidly under good conditions. It has been proposed that constant exposure to water has necessitated teleosts to develop a more efficient wound healing process than terrestrial animals (Quilhac and Sire 1999). Small wounds are quickly covered by a layer of mucus, while re-epithelisation originating from the wound margins occurs within a few hours in warm water species (Quilhac and Sire 1999). Temperature has a fundamental effect on animal physiology, especially in poikilothermic animals such as fish. Low temperatures adversely affect both cellular and humoral specific immune response, and the non-permissive lower water temperature in salmonids has been established at 4 °C (Bly and Clem 1992). Low temperatures adversely affect both cellular and humoral specific immune responses, with negative consequences on the healing rate (Bowden 2008). A rapid wound closure, even at low temperatures, confers major advantages in the aqueous environment because the sealing of the integument helps prevent secondary infections and allows the fish to maintain homoeostasis.
Skin wound healing can be divided into a sequence of overlapping events; haemostasis, inflammation, proliferation and remodelling (Diegelmann and Evans 2004). In higher vertebrates, the innate immune system is fundamental in the initial phase of the wound response, but information on wound healing mechanisms of salmon is scarce.
Optimal nutrition plays a major role in maintaining normal skin physiology and repair mechanisms following injury. Micro-nutrients such as vitamin C and zinc (Zn) are known to fulfil roles as structural components, enzyme co-factors or physiological mediators in human nutrition (Lansdown 2004). As vitamin C cannot be synthesised by fish (Mæland and Waagbø 1998), tissue levels depend exclusively on dietary supplementation. It is essential for the synthesis of collagen by the hydroxylation of pro-collagen (Tsao 1997), as also verified in Atlantic salmon with vitamin C deficiency (Sandnes et al. 1992). Vitamin C also acts as a strong reducing agent and is involved in several physiological processes including growth, reproduction, wound healing, immune function and response to stress and infectious agents (Wahli et al. 2003, Lall 2000; Waagbø 2006).
Trace elements are required as co-enzymes and are vital for maintenance of the immune system in higher vertebrates (Lall 2000). Zn is an important trace element in fish nutrition (Fountoulaki et al. 2010), and it is known to exert a beneficial effect on wound healing by modulating inflammation and speeding up the re-epithelialisation process. It stimulates the proliferation of keratinocytes and fibroblasts in wounds and increases collagen synthesis (Tenaud et al. 2000). Zn deficiency has been associated with delayed wound healing and erosion of fins and skin (Ogino and Yang 1979, Hughes 1985). Yeast-sourced β-glucans are the mostly used immunostimulants in aquaculture, and their immunomodulatory effects have been described in numerous fish species (Ringø et al. 2012; Dalmo and Bøgwald 2008). β-Glucans have also been shown to accelerate the wound healing process in mammals (Berdal et al. 2007; Vetvicka and Vetvickova 2011) and fish (Przybylska-Diaz et al. 2013).
Inflammatory cytokines and a number of chemokines are up-regulated in the early stages of wound healing and play important roles in attracting leucocytes and regulating cell proliferation and differentiation (Gharaee-Kermani and Phan 2001). The expression profiles of specific cytokines and their receptors in the skin of several fish species have been reviewed by Esteban (2012), having shown that some of them are up-regulated by infection or mechanical injury to the skin. Among these cytokines, an up-regulation of the CXCa chemokine (also referred to as interleukin 8, IL8) and its receptor, CXCR1 was demonstrated in response to wounding in carp (Gonzalez et al. 2007). Chemokines mediate a rapid recruitment of leucocytes, not only as immunological effector cells, but also as an important source of inflammatory and growth promoting cytokines. Although the ascription of true orthologues between fish and mammalian CC chemokines has been a difficult task due to the expansion that this chemokine family has experienced in many fish species, Peatman and Liu (2007) through an extensive phylogenetic analysis classified fish CC chemokines into seven groups according to their relation to mammalian chemokines. These groups are the CCL19/21/25 group, the CCL20 group, the CCL27/28 group, the CCL17/22 group, the MIP group, and the MCP group and a fish-specific group. In mammals, CCL27, a skin-associated CC chemokine, is a major regulator in wound healing involved in the migration of keratinocyte precursor cells from bone marrow into the skin (Inokuma et al. 2006). According to the classification made by Peatman and Liu (2007), rainbow trout CK11 and four Atlantic salmon chemokine genes with GenBank accession numbers DY728991 (referred as CK11A in this paper), EG818960 (referred as CK11B in this paper), EG940598 (referred as CK11C in this paper) and EG766286 (referred as CK11D in this paper) can also be ascribed within the CCL27/28 group. In rainbow trout, CK11 also seems to be preferentially expressed in skin, as its mammalian homologue, and also in gills (Montero et al. 2011).
In the current work we first determined if salmon CK11A–D were preferentially expressed in salmon skin, and then determined if their transcription levels were modulated by mechanically inflicted wounds, temperature and diet. The effect of temperature and functional ingredients on the healing process was also assessed through subjective and quantitative histology.
Materials and methods
Feed and fish
Three diets were produced at Skretting Feed Technology Plant, Stavanger, Norway as 4.5 mm extruded sinking pellets, one control diet (Control), a diet with additional organic zinc (Zn) and a diet with a mix of additives (Zn+). All diets were formulated with the control diet (Control) as a base (Table 1), and functional ingredients were added on top. The Zn and the Zn+ diets were supplemented with organic Zn (Optimin Zn, Selko Feed Additives©, Tilburg, The Netherlands) to reach a total diet level of 250 mg Zn/kg. The Zn+ diet was in addition supplemented with a 1.5 g mixture of natural identical compounds (Lucta©, Barcelona, Spain), 1.0 g β-glucans (MacroGard®, Biorigin©, São Paulo, Brasil) and vitamin C and E (Trouw Nutrition, Putten, The Netherlands), both to reach target levels of 700 mg/kg feed.
The experiment was performed at Lerang Research Station (Stavanger, Norway). Atlantic salmon eggs from Erfjord stamfisk were hatched and offspring reared in house to an average fish weight of 262 g. At the start of the experiment, 120 fish were randomly distributed into twelve 1-m tanks. All tanks were supplied with flow through seawater at 8 °C and 33 ‰ salinity. The fish were adapted to the tanks for 2 weeks. During this period, all fish were fed a standard diet. After the adaptation period, six tanks were switched to 4 °C and the other six tanks to 12 °C overnight. Within each temperature three diets were randomly assigned to duplicate tanks. Fish in all tanks were fed the experimental diets for 10 weeks after changing temperature, before punch biopsies of the skin were performed. Dissolved oxygen levels and water temperature were measured daily, and feed intake was monitored by recording feeding and collecting waste feed. After 8 weeks, the fish were weighed to control growth and then allowed to recover from handling for 2 weeks before the biopsy.
Skin biopsy and sampling
Before sampling all fish were anaesthetised with Finquel MS-222 (Sigma). A skin biopsy punch was taken from both sides of the fish, between the dorsal fin and the lateral line (Fig. 1), of twelve fish per tank. Disposable 5-mm-diameter biopsy punches (Miltex, Inc., USA) were used to mechanically remove standardised pieces of skin and muscle. After wound infliction, the fish were returned to their respective tanks.
Control samples (time point 0) were taken from two fish in each tank at the same time as the biopsy was performed. The fish were then sampled at 6 h, 24 h, 72 h, 7 days and 16 days after the biopsies. A total of 24 fish were sampled per time point (two fish per tank or four per treatment). From 6 h onward, an area of 1.5 × 1.5 cm around the left side wound was dissected out with a scalpel and stored in 4 % buffered formalin for histological assessment. Around the wound on the right side an area of 1 × 1 cm was dissected out and the muscle removed from the skin before the sample was fixed in RNAlater (Sigma-Aldrich). These samples were stored overnight at 4 °C and then kept frozen at −80 °C until further analyses of chemokine expression. The experiment was conducted according to Norwegian legislation and approved by The Norwegian Animal Research Authority (NARA/FDU). All samplings were performed by scientists accredited by the Federation of Laboratory Animal Science Associations (FELASA).
Histology processing
The fixed skin samples were carefully dissected, orientated and placed in histo-cassettes (ChemiTeknikk, Norway). To decalcify the skin samples, each was incubated in EDTA (ChemiTeknikk, Norway) solution, pH 7, for 2 days. EDTA was dissolved in buffered 4 % formalin and pH adjusted with sodium hydroxide (ChemiTeknikk, Norway). Tissue processing was performed using an automated Tissue Processor (Leica, Shandon Excelsior), fat program. The tissue samples were dehydrated through 100 % alcohol and then a clearent Xylene bath before infiltration in melted 60 °C paraffin (Histowax, Sweden). Paraffin-embedded tissue samples were cut by a semi-automated Microtome (Leica). Paraffin sections of 3 µm thickness were mounted onto Superfrost Plus slides (Menzel, Braunschweig, Germany) and dried overnight at 37 °C. The sections were deparaffinised, rehydrated and special staining was performed using Bench Mark automated special stainer (Roche, France). Sections were stained by ABPAS ready-to-use special staining kit (Roche, France). Acidic mucosubstances was stained blue by incubating sections in PAS Alcian Blue for 8 min, while neutral mucosubstances stained magenta red by incubating sections in PAS Period Acid for 4 min following by PAS Schiff’s for 12 min. Cell nuclei were stained by PAS haematoxylin. Sections were dehydrated and cover slipped by using automated stainer Symphony (Roche, France), choosing program Special Stains Coverslip.
Subjective and quantitative histology assessments
Stained sections were digitally scanned, employing a Zeiss Mirax scanner (Carl Zeiss, GmbH, Germany). Subjective assessments were made of all fish from the 6-, 24- and 72-h samplings. After 6 h, there was minimal epidermal regrowth, and these slides were assessed to describe different type of wounds. All samples from 24 and 72 h were subjectively assessed for closure of the wound, epidermal growth, development of the basal layer and the infiltration of inflammatory cells. At these time points, it was not possible to separate dietary treatments, so only temperature differences were assessed. For the wound healing after 1 and 2 weeks, brief assessments were also made for each temperature, with the quantitative assessments only comparing dietary treatments.
The scanned images from first and second weeks post-wounding were quantitatively assessed by image analysis using Visiomorph module (VIS 4.6 software, Visiopharm, Denmark). Regions of interest (ROI’s) were manually drawn around the epidermis in the wound area, covering as much epidermis as possible, avoiding only the tip of the scales and areas where tissue structure had been compromised during processing. Two protocols were performed in Visiomorph (200× magnification) in order to assess mucous cells and epidermal thickness.
In the first protocol, custom-made colour de-convolutions were used to pre-process the image, before applying a Bayesian classifier. A series of post-processing steps were then performed to remove noise and to individually separate the mucous cells in the epidermis according to their distance to the skin surface. The mucous cell area percentage, of either all mucous cells or only mucous cells within a certain threshold distance from the surface, could then be calculated as the epidermal area comprising mucous cells relative to the entire ROI epidermal area.
The second protocol for calculation of epidermal thickness was based on the classified image after the post-processing from the first protocol. Using further post-processing steps, the mucous cells were unified with the remainder of the epidermal area, thus creating an interface shared only by the epidermal area and the skin outside. The length of this interface estimated the length of the epidermal ROI. The average epidermal thickness was then calculated by dividing the epidermal area by its length.
Identification of skin chemokines
Primers for real-time analysis were designed from GenBank sequences available of salmon chemokines ascribed in the CCL27/28 group and referred to as CK11A-D because of their close homology with rainbow trout CK11 (Table 2). As a first step, we analysed the transcription of these chemokines in different tissues obtained from naïve fish. For this, salmon were killed by an overdose of anaesthetics (Tricaine, Pharmaq AS), and skin, muscle, gill, spleen, head kidney and intestine removed and placed in RNAlater at −80 °C until used.
Analysis of chemokine transcription
Total RNA was extracted from tissue samples using a combination of Trizol (Invitrogen) and RNAeasy Mini kit (Qiagen). In summary, samples were transferred from RNAlater to 1 ml of Trizol and mechanically disrupted using a disruption pestle. Then, 200 µl of chloroform were added and the suspension centrifuged at 12,000×g for 15 min. The clear upper phase was recovered, mixed with an equal volume of 100 % ethanol and immediately transferred to RNeasy Mini kit columns. The procedure was then continued following manufacturer’s instructions, performing on-column DNase treatment. Finally, RNA pellets were eluted from the columns in RNase-free water and stored at −80 °C until used. Two micrograms of RNA was used to obtain cDNA in each sample using the Bioscript reverse transcriptase (Bioline Reagents Ltd) and oligo (dT)12–18 (0.5 µg/ml) following manufacturer’s instructions. The resulting cDNA was diluted in a 1:5 proportion with water and stored at −20 °C.
To evaluate the levels of transcription of the different chemokine genes, real-time PCR was performed in a LightCycler® 96 System instrument (Roche) using SYBR Green PCR core Reagents (Applied Biosystems) and specific primers (Table 2). The efficiency of the amplification was determined for each primer pair using serial tenfold dilutions of pooled cDNA, and only primer pairs with efficiencies between 1.95 and 2 were used. Each sample was measured in duplicate under the following conditions: 10 min at 95 °C, followed by 45 amplification cycles (15 s at 95 °C and 1 min at 60 °C). The expression of individual genes was normalised to relative expression of salmon EF-1α, and the expression levels were calculated using the 2−ΔCt method, where ΔCt is determined by subtracting the EF-1α value from the target Ct. Negative controls with no template were included in all the experiments. A melting curve for each PCR was determined by reading fluorescence every degree between 60 and 95 °C to ensure only a single product had been amplified.
Statistical analysis
Differences in initial weight and growth were analysed using the general linear model procedure in Unistat versus 6.0 (UNISTAT Ltd, London, UK), and Student–Newman–Keuls multiple comparison test was used to determine significance between treatments (UNISTAT Ltd, London, UK). Statistical analysis of quantitative histology measurements were performed in MATLAB and Statistics Toolbox, Release 2013a (The MathWorks Inc., Natick, Massachusetts, US), using two-way ANOVA with interaction at significance level of 0.05. Data handling, analyses and graphic representation of chemokine transcription levels were performed using Microsoft Office Excel 2010. Statistical analyses were performed using two-tailed unpaired t Student’s tests. Means ± standard deviation of the mean (SD) for each group were calculated.
Results
Feed and fish
All diets were produced in accordance with nutritional formulation, without any significant deviations in anticipated proximate composition (Table 3). Levels of Zn and vitamin C in the control feed were 92 and 38 mg kg−1, respectively, originating from the premix and the raw materials (Table 3).
There was no difference in initial weight (p > 0.05). After 8 weeks of feeding the experimental diets, the fish grew from 263 ± 5 g to 352 ± 4 g at 4 °C and 420 ± 7 g at 12°. Final weight, specific growth rate (SGR) and food conversion ratio (FCR) were all higher in the fish kept 12 °C than fish kept at 4 °C (p < 0.01) (Table 4). There were no growth differences between the different dietary groups within each temperature.
Histology
General description of wounds
The biopsy punch created a sharp cut through the dermis, giving a clear definition of the wound area in all fish. None of the wounds expanded after the biopsy. In the underlying tissue, extended haemorrhages and oedema were present. Non-structured material and myolytic fractions showed that the muscle tissue had been affected.
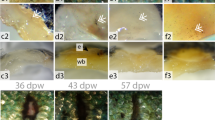
The biopsy punch method gave a consistent diameter of wounds in all fish, but there was still some variation in the depth of the wounds. The different types of wounds are shown in Fig. 2. The most common type was where all the epidermis and the dermis was removed, leaving a smooth muscle surface for the regrowth of the epidermis (Fig. 2a). In some wounds, the dermis was lifted up with the biopsy punch, leaving a bulging appearance of the wound area (Fig. 2b). In Fig. 2c, d, all the tissue from the biopsy was completely removed, leaving an opening in the epidermis and the dermis to be covered by new growth. Within some wounds debris of the epidermal material like scales made it more difficult for the wound to be closed (Fig. 2d).
In some fish, the epidermis, dermis and a part of the underlying muscle tissue were removed. In others, only the epidermis was removed, while the underlying tissue was cut through with the biopsy punch, and then lifted up into the wound, resulting in new epidermis growing directly on top of the muscle. Most fish had also suffered by the pressure exerted through the biopsy punch, which contributed to the haemorrhages and myolytic fractions.
Subjective assessment of wound healing at 4 and 12 °C
6 h post-wounding
At 4 °C there was no evidence for initiation of recovery 6 h post-wounding. This may be due to processing, as any growth of new epidermal cells would have been delicate and possibly destroyed during processing. At 12 °C small areas of new epithelial cells could already be seen at the edge of the wound site (Fig. 3).
24 h post-wounding
At 4 °C there was a noticeable onset of epidermal growth, but only visible on the empty scale pockets close to the wound. Epidermal growth was less than at 12 °C, and no inflammation was seen in the tissue. At 12 °C a new epidermis with rounded epithelial cells was under development (Fig. 3). On the empty scale pockets close to the wound, the epidermis was new. Epidermal cells started to cover the wound, and the first signs of inflammation could be seen.
72 h post-wounding
In fish kept at 4 °C the wound was closed, but the epidermis showed more vacuolisation, less organisation and had a less pronounced basal layer compared to fish kept at 12 °C, suggesting delay of healing processes. The basal lamina was only visible towards the edges of the wound, where the general organisation of the epidermis was most advanced. Some inflammatory cells were visible in the epidermis, but less than at 12 °C.
At 12 °C, the wound was closed after 72 h, and the epidermis constituted several cell layers, relatively good structure and a well-organised basal cell layer. In wounds where the wound bed was fluid, the surface of the epidermis and basal layer was uneven. Some inflammation was present, and the basal lamina could be recognised (Fig. 4).
1 week post-wounding
After 1 week there were still more haemorrhages in the wound area, and considerably less inflammatory cells in fish kept at 4 °C than at 12 °C. The new tissue was highly vacuolated, and the superficial cell layer was weak. Mucous cells were present in the superficial layer, mainly in the wound edges. Organisation of the basal layer was more developed along the wound boundaries, and less organised in the wound centre and where the foundation was fluid. The development at both temperatures at 72 h, 1 and 2 weeks are shown in Fig. 4.
At 12 °C less haemorrhage could be seen in the tissue, and more inflammatory cells were present. There were also more vacuoles in the new epidermis than what could be seen at 72 h. The most superficial cell layers mainly consisted of mucous cells, but no mucous cells were present in the deeper layer. In areas where the epidermis were most structured and well organised, the outer layers of epidermal cells showed a flattened and more elongated shape. In thicker and more vascularised areas, the cells were more rounded and became elongated at a later stage. There were no signs of any fibrous tissue at this sampling point.
2 weeks post-wounding
At 4 °C there was still a high level of inflammation after 2 weeks. The epidermis lacked organisation and a lot of vacuolisations was seen. The epidermal cells were more rounded than elongated, and the basal line was less defined. Mucous cells were mainly present in superficial layers. No fibrous tissue could be seen in any of the samples from 4 °C.
At 12 °C there was less inflammation, but the epidermis was still far from normalised. In areas where there was no support from muscle tissue underneath, the epidermis was very thick and the basal line was uneven. In areas with sufficient support under the new tissue, the organisation started normalising. Mucous cells were visible throughout the epidermis and not just in the superficial layers. There was also a clear start of fibrous tissue production in the deep area of the wound and along the wound edges.
Quantitative assessment of wound healing at 4 and 12 °C
Epidermal thickness was significantly lower at 4 °C than at 12 °C 1 week post-wounding when comparing the average of all fish within each temperature; 98 ± 21 and 135 ± 25 µm, respectively (ANOVA, p < 0.01) (Table 5). In the fish fed, the control diet the epidermal thickness varied from 85 ± 24 µm at 4 °C to 150 ± 33 µm at 12 °C (p < 0.05), while the Zn and Zn+ fed groups showed less variation (Table 5). After 2 weeks the epidermal thickness had increased at both temperatures, to 135 ± 40 µm at 4 °C and 165 ± 51 °C at 12° (ANOVA, p < 0.05) (Table 5). Total area of epidermis comprising mucous cells was not significantly different (p > 0.05) and constituted in average 5.0 and 5.3 % after 1 week, and more than 97 % of the mucous area was dispersed in the most superficial epidermal region, from 0 to 30 µm. Two weeks into the healing process the total mucous area was 3.4 % in average at 4 °C and 3.6 % at 12 °C. At 4 °C the outermost epidermal region comprised 98.1 ± 1.8 % of the mucous cell area, while at 12 °C only 84.4 ± 8.9 % of the mucous area was found in the outer region from 0 to 30 µm (Fig. 5). From 30 to 70 µm and >70 µm the mucous cell area was 7.9 ± 3.8 and 7.7 ± 7.2 %, respectively. In the epidermal layers deeper than 70 µm, the Zn and Zn+ diets had a relatively larger mucous area than the control group, 12 and 8 % respectively, compared to 4 % in the control group. These results were not significant (p > 0.05), due to the large individual variation.
Constitutive expression of salmon chemokines ascribed to the CCL27/28 group
As a first step towards the characterisation of the role of salmon CC chemokines with homology to mammalian CCL27 in wound repair, we compared the constitutive levels of transcription of these chemokines in different tissues. In the case of CK11A and CK11B, constitutive transcription levels were higher in the skin, followed by the muscle, the gill and the intestine (Fig. 6). Lower CK11A and CK11B transcription levels were detected in spleen and head kidney. CK11C transcription levels were also higher in skin than in the other tissues examined, but in this case intermediate mRNA levels were observed in the muscle, gill, spleen and head kidney, while lower mRNA levels were observed in the intestine. CK11D transcription levels always remained undetected; thus, we did not further study these chemokine genes. The results demonstrated that CK11A, CK11B and CK11C are preferentially expressed in the skin under normal physiological conditions.
Transcription of salmon chemokines ascribed to the CCL27/28 group after skin wounding
Having established that salmon CK11A, CK11B and CK11C are strongly expressed in the skin, we wanted to determine the levels of transcription of these chemokines after wounding in fish kept at different temperatures and having received different diets. At 4 °C, CK11A transcription levels significantly increased after 6 h when compared to unwounded skin when fish were fed with the Zn and Zn+ diets, whereas it was not until 24 h that these transcription levels were significantly increased in fish fed the control diets (Fig. 7a). At 12 °C, the levels of CK11A transcription after 6 h decreased in comparison with unwounded animals in fish fed the control and the Zn+ diets. After 24 h, the transcription levels increased in comparison with unwounded animals in fish fed with control and Zn diets. In the case of CK11B, we observed a significant up-regulation of its transcription levels at 6 h for fish fed with all three diets, when compared to the levels obtained in unwounded fish at 4 °C (Fig. 7b). These up-regulations were further increased after 24 h in fish fed the Zn and the Zn+ diets. On the other hand, at 12 °C, only a significant down-regulation of CK11B transcription levels after 6 h was detected in response to wounding. No significant modulations of CK11C transcription were observed during the first 24 h in any of the fish studied (Fig. 7c). From 72 h post-wounding, the levels of transcription of all chemokines remained significantly lower than those from unwounded fish for all diets at both 4 and 12 °C.
Effect of the different diets on the levels of transcription of the different chemokines in the skin of unhandled fish or wounded fish after different times post-wounding. Fish were kept at either 4 or 12 °C and within each temperature groups of fish were fed with different diets for 10 weeks, before a punch biopsy of the skin was performed. RNA was extracted from the wounded area at different times after the biopsy to analyse the levels of transcription of the different chemokine genes. Data are shown as the mean gene expression relative to the expression of endogenous control EF-1α ± SD (N = 4 in each group). *Transcription levels significantly different than those observed at time 0 (p < 0.05) for each diet (different colour code)
Discussion
In the current study, the impact of temperature and diet were investigated through subjective and quantitative skin histology and expression of skin-associated chemokines following mechanical skin damage in Atlantic salmon. Overall, a clear progression in the wound healing process was seen related to both temperature and diet.
As subtle changes in wound pallor constituted the sole gross sign of wound healing, other methods were necessary to investigate the healing processes and potentially discriminate between temperature and dietary treatments. Due to the severity of the wounds inflicted by the biopsy punch, and resultant variances between individual wounds, it was difficult to assess measurements by quantitative histology during the first 72 h, necessitation subjective morphological assessments during this phase. At 12 °C visible onset of epidermal growth was evident even within 6 h post-wounding. The delay of the first signs of healing at 4 °C as indicated by new epidermal cells at the wound edge after 24 h demonstrated a clear temperature effect. After 3 days, all of the examined wounds were closed, but the epidermis was more structured at high temperature, showing greater organisation of the epidermal basal layer cells. These results correlate to a previous study where rainbow trout at 15 °C were inflicted with similarly sized wounds; all of which showed closure at the first sampling after 3 days (Wahli et al. 2003). As poikilotherms, fish are dictated by the surrounding water temperature, with the metabolic rate decreasing rapidly at lower temperature (Claireaux and Lagardère 1999). Low temperature also affects the immune response (Bowden 2008; Bly and Clem 1992), and temperature has been shown to impact wound healing in both tropical and temperate teleosts (Anderson and Roberts 1975). The healing rate of small wounds on Atlantic salmon and white mountain cloud minnow (Tanichthyes albonubes) was significantly faster at high temperatures (23 vs 5 °C) (Anderson and Roberts 1975). At 23 °C the wounds were already closed after 4 h in both species, while the process took 24 h at 5 °C.
Wounds heal through a highly regulated, self-limited inflammatory response; however, precise inflammatory mediators have not been fully delineated (Ishida et al. 2008). Subjective and quantitative histology both confirmed that temperature had a large impact on the complex process of wound healing. After 24 h, no sign of inflammation was present at 4 °C, while the first inflammatory cells could be seen in fish at 12 °C. From 1 week post-wounding the subjective data could be supported by quantitative analyses, confirming that the epidermis was thicker at 12 °C both one and 2 weeks after wounding. This may be related to the faster healing process at high temperature and that a higher level of inflammation is reached more rapidly. Wound healing is always accompanied by an inflammatory reaction, which does not subside with epithelialisation, but persists until the tissue is remodelled (Gillitzer and Goebeler 2001). The grade and cells in the inflammation changes through the healing process, being responsible for combating pathogens and cleaning the wound for cell debris and foreign material (Medzhitov 2008).
The most significant quantifiable difference between the two temperatures was the dispersal of mucous cells. Total area of epidermis comprising mucous cells was similar at 4 °C and 12 °C, but their position in the epidermis differed. After 1 week, 98 % of the mucous cells were seen in the outer 30 µm of the epidermis, and a similar result was seen after 2 weeks at low temperature. However, mucous cells were seen in the deeper layers of the epidermis, after 2 weeks in fish reared at 12 °C, with approximately 15 % of the mucous cells located deeper than 30 µm from the surface. While epidermal cells are known to cover wounds by migration from the surrounding tissue, followed by proliferation, increases in intracellular spaces, leading to epidermal thickening and hyperplasia (Iger and Abraham 1990; Bereiter-Hahn 1986; Fast 2014), less is known about the mucous cell dynamics. It is known that stress leads to changes in mucous cell production in teleost skin (Tacchi et al. 2015; Esteban 2012; Vatsos et al. 2010), and a variation in mucous cell coverage in the epidermis related to temperature has been described for salmon (Jensen et al. 2014). Increased production and hypertrophy of mucous cells is also described as a response to ecto-parasitism attacks in fish (Fast et al. 2002a; Nolan et al. 1999). In mammals, mucous cell differentiation and function are known to be affected by mucosal immunity. In particular, growth factors, antimicrobial peptides, cytokines, chemokines and mucins play an important role in mucosal healing (Neurath and Travis 2012). Mucins from carp has also been described (Przybylska-Diaz et al. 2013; Marel et al. 2012) and putatively found in salmon (Micallef et al. 2012). Although the regulation of mucous cells in fish warrants further investigation, the potential range of roles and effect of mucus has been described by many authors (Fast et al. 2002b; Shephard 1994; Mustafa et al. 2005; Nigam et al. 2012; Subramanian et al. 2007), and accelerated reinstatement of mucous cells would strengthen teleost natural defence mechanisms.
In addition to temperature, diet was also shown to affect the wound healing process. Zn is known to exert a beneficial effect on wound healing by modulating inflammation and speeding up the re-epithelialisation process (Tenaud et al. 2000). It stimulates the proliferation of keratinocytes and fibroblasts in wounds and increase collagen synthesis in humans. Although the nature of the feedback mechanism regulating epidermal cell mitosis in humans is still not understood, there are suggestions that gradients in calcium and/or zinc may be contributory (Lansdown 2002). An inverse relationship seems to exist whereby zinc promotes mitosis and calcium suppresses it. In the current study, due to individual variation, it was not possible to determine any significant differences between diets the first 72 h post-wounding by subjective histology. After one and 2 weeks, quantitative measurements of epidermal indices allowed discrimination between diets. The finding of thicker epidermis at low temperature after 1 week in fish fed Zn-supplemented diets could represent enhanced recruitment of inflammatory cells and increased proliferation of epithelial cells.
Relative distribution of mucous cells within the epidermis was identified as a marker of healing progression with a greater proportion in the deeper strata related to advanced healing. The enhancement of healing at higher temperature and further potentiation by supplemented dietary Zn as assessed early during healing by chemokine expression was also borne out by greater presence of mucous cells in the deeper epidermal zones later in the process. As previously mentioned, the regulation of mucous cell differentiation needs more research, and it is likely that Zn could play a role in the regulation. Zn is among the most important trace elements in fish nutrition and is involved in various metabolic pathways (Watanabe et al. 1997). Although the mechanisms are not fully understood, Zn deficiency has been associated with delayed wound healing, eye lens cataract and erosions of the fins and skin in fish (Fountoulaki et al. 2010; Waagbø 2008; Ogino and Yang 1979; Hughes 1985). The importance of Zn in the protection against auto oxidation has been demonstrated in salmonids (Kaushik 2001). Zn is an essential component of several enzymes that are involved in the wound healing process such as collagen synthesis and cell division. The parameters studied in the current paper revealed no clear benefit from the additional functional ingredients in the Zn+ diet. Levels both of vitamin C and Zn in the skin correlated with dietary levels (results not shown), as previously described by Wahli et al. (2003) and Fountoulaki et al. (2010). Vitamin C has been shown to impact on wound healing in salmonids, and fish presented diets with no vitamin C completely failed to repair wounds (Halver 1972). It is involved in several physiological processes and is essential for collagen formation as a cofactor for hydroxylation of proline. Many of the parameters shown to be affected by the vitamin C concentration have been related to the formation of fibrous tissue, particularly in the dermis (Wahli et al. 2003; Halver 1972). The lack of observed effect of vitamin C in the current study may therefore be related to the fact that epidermis was the main focus, and differences in the dermis were not assessed by quantitative measures. Dermal and collagen-related indices should be assessed in future studies. The Zn+ diet was also supplemented with higher levels of vitamin E, but the level in the control diet was probably high enough to maintain antioxidative capacity and immune function (Hamre 2011; Lall 2000; NRC 2011).
The Zn+ diet was also supplemented with β-glucans. Yeast ß-glucans are probably the most extensively investigated immunostimulants in fish feed, and immunomodulatory activity has been reported for a range of species (Kiron 2012; Dalmo and Bøgwald 2008; Vetvicka and Vetvickova 2011). Their effect has also been studied on the wound healing process in common carp, describing a positive impact on wound closure and cytokine expression (Przybylska-Diaz et al. 2013). Chemokines, a superfamily of cytokines, are known to stimulate the recruitment, activation and adhesion of cells to the site of infections or injury (Whyte 2007). Although several genes coding for salmon CC chemokines have been reported in the databases, not many studies have focused on establishing a role for each of these genes or on studying their transcriptional regulation. In the current work, an evaluation of the constitutive expression levels of several chemokine genes that previously have been ascribed to the CCL27/28 group (Peatman and Liu 2007) was evaluated. CCL27 is a skin-associated chemokine in mammals, and it was considered of importance to determine whether these chemokines were also expressed in salmon skin and implicated in wound healing as CCL27. The results demonstrated that CK11A, CK11B and CK11C were preferentially expressed in the skin, in comparison with other tissues, as its homologue in rainbow trout, CK11 (Montero et al. 2011). Transcription of CK11D was not detected, which might suggest that this is actually a non-transcribed pseudogene. The analysed levels of chemokine expression in the wound area, demonstrated that CK11A, CK11B and CK11C were modulated in response to wounding. In general, there was an up-regulation at 6 h post-wounding, followed by significant down-modulations from 72 h post-wounding. The levels were still not recovered back to constitutive levels after 2 weeks. Different modulation profiles were seen between 4 and 12 °C for all three chemokines, where the profiles at 4 °C showed a clearer up-regulation at both 6 and 24 h, while the profiles at 12 °C dropped at 6 h and increased again at 24 h. Differences were also seen between diets. Zn-supplemented diets generally led to increased chemokine expression during the first 24 h following wounding. This could have affected early recruitment of immune cells to the traumatised area during the critical first phase of recovery, or influenced the initiation of cell proliferation. Other potential roles of these described chemokines warrants further investigation.
In conclusion, both temperature and dietary Zn had an effect on the healing of mechanically inflicted wounds in salmon. The work also demonstrated that CK 11A-C were preferentially expressed in skin and also modulated in reaction to skin damage. Quantitative histology was demonstrated to be a valuable tool in assessing changes in the epidermis, revealing an effect of dietary Zn levels on the dispersal of mucous cells 2 weeks post-wounding. The positive effects of Zn seen with histology, correlated with the increase in chemokine expression which was modulated during the acute phase of wound closure. Future studies focusing on optimising controlled skin trauma approaches could employ the newly described quantitative methodologies to complement traditional subjective appraisals and should also include gene expression of established inflammatory markers, to give more insight into healing processes of fish skin.
References
Anderson CD, Roberts RJ (1975) A comparison of the effects of temperature on wound healing in a tropical and a temperate teleost. J Fish Biol 7:173–182
Benediktsdóttir, Helgason, Sigurjónsdóttir (1998) Vibrio spp. isolated from salmonids with shallow skin lesions and reared at low temperature. J Fish Dis 21:19–28
Berdal M, Appelbom HI, Eikrem JH, Lund Å, Zykova S, Busund L-T, Seljelid R, Jenssen T (2007) Aminated β-1,3-d-glucan improves wound healing in diabetic db/db mice. Wound Repair Regen 15:825–832
Bereiter-Hahn J (1986) Epidermal cell migration and wound repair. In: Bereiter-Hahn J, Matoltsy AG, Richards KS (eds) Biology of the integument. Springer, Berlin, pp 443–471
Bly JE, Clem LW (1992) Temperature and teleost immune functions. Fish Shellfish Immunol 2:159–171
Bowden TJ (2008) Modulation of the immune system of fish by their environment. Fish Shellfish Immunol 25:373–383
Claireaux G, Lagardère JP (1999) Influence of temperature, oxygen and salinity on the metabolism of the European sea bass. J Sea Res 42:157–168
Dalmo RA, Bøgwald J (2008) ß-glucans as conductors of immune symphonies. Fish Shellfish Immunol 25:384–396
Diegelmann RF, Evans MC (2004) Wound healing: an overview of acute, fibrotic and delayed healing. Front Biosci 9:283–289
Esteban MA (2012) An overview of the immunological defenses in fish skin. ISRN Immunol 2012:29
Fast MD (2014) Fish immune responses to parasitic copepod (namely sea lice) infection. Dev Comp Immunol 43:300–312
Fast MD, Ross NW, Mustafa A, Sims DE, Johnson SC, Conboy GA, Speare DJ, Johnson G, Burka JF (2002a) Susceptibility of rainbow trout Oncorhynchus mykiss, Atlantic salmon Salmo salar and coho salmon Oncorhynchus kisutch to experimental infection with sea lice Lepeophtheirus salmonis. Dis Aquat Org 52:57–68
Fast MD, Sims DE, Burka JF, Mustafa A, Ross NW (2002b) Skin morphology and humoral non-specific defence parameters of mucus and plasma in rainbow trout, coho and Atlantic salmon. Comp Biochem Physiol A 132:645–657
Fountoulaki E, Morgane H, Rigos G, Antigoni V, Mente E, Sweetman J, Nengas I (2010) Evaluation of zinc supplementation in European sea bass (Dicentrarchus labrax) juvenile diets. Aquacult Res 41:e208–e216
Gharaee-Kermani M, Phan SH (2001) Role of cytokines and cytokine therapy in wound healing and fibrotic diseases. Curr Pharm Des 7:1083–1103
Gillitzer R, Goebeler M (2001) Chemokines in cutaneous wound healing. J Leukoc Biol 69:513–521
Gonzalez SF, Huising MO, Stakauskas R, Forlenza M, Lidy Verburg-Van Kemenade BM, Buchmann K, Nielsen ME, Wiegertjes GF (2007) Real-time gene expression analysis in carp (Cyprinus carpio L.) skin: inflammatory responses to injury mimicking infection with ectoparasites. Dev Comp Immunol 31:244–254
Halver J (1972) The role of Ascorbic acid in fish disease and tissue repair. Bull Jpn Soc Sci Fish 38:79–89
Hamre K (2011) Metabolism, interactions, requirements and functions of vitamin E in fish. Aquacult Nutr 17:98–115. doi:10.1111/j.1365-2095.2010.00806.x
Hughes SG (1985) Nutritional eye diseases in salmonids: a review. Prog Fish Cult 47:81–85
Iger Y, Abraham M (1990) The process of skin healing in experimentally wounded carp. J Fish Biol 36:421–437
Inokuma D, Abe R, Fujita Y, Sasaki M, Shibaki A, Nakamura H, Mcmillan JR, Shimizu T, Shimizu H (2006) CTACK/CCL27 accelerates skin regeneration via accumulation of bone marrow-derived keratinocytes. Stem Cells 24:2810–2816
Ishida Y, Gao J-L, Murphy PM (2008) Chemokine receptor CX3CR1 mediates skin wound healing by promoting macrophage and fibroblast accumulation and function. J Immunol 180:569–579
Jensen LB, Boltana S, Obach A, Mcgurk C, Waagbø R, Mackenzie S (2014) Investigating the underlying mechanisms of temperature-related skin diseases in Atlantic salmon, Salmo salar L., as measured by quantitative histology, skin transcriptomics and composition. J Fish Dis. doi:10.1111/jfd.12314
Kaushik SJ (2001) Mineral nutrition. In: Guillaume J, Kaushik S, Bergot P, Metailler R (eds) Nutrition and feeding of fish and crustaceans. Springer-Praxis, Publishing Ltd, Chichester, pp 169–181
Kiron V (2012) Fish immune system and its nutritional modulation for preventive health care. Anim Feed Sci Technol 173:111–133
Lall SP (2000) Nutrition and health of fish. In: Cruz-Suárez LE, Ricque-Marie D, Tapia-Salazar M, Olvera-Novoa MA, Civera-Cerecedo R (eds) Avances en Nutrición Acuícola V. Memorias del V Simposium Internacional de Nutrición Acuícola, Mérida, Yucatán, Mexico, pp 13–23
Lansdown ABG (2002) Calcium: a potential central regulator in wound healing in the skin. Wound Repair Regen 10:271–285
Lansdown AB (2004) Nutrition 2: a vital consideration in the management of skin wounds. Br J Nurs 13:1199–1210
Løvoll M, Wiik-Nielsen CR, Tunsjø HS, Colquhoun D, Lunder T, Sørum H, Grove S (2009) Atlantic salmon bath challenged with Moritella viscosa—pathogen invasion and host response. Fish Shellfish Immunol 26:877–884
Mæland A, Waagbø R (1998) Examination of the qualitative ability of some cold water marine teleosts to synthesise ascorbic acid. Comp Biochem Physiol A 121:249–255
Marel MVD, Adamek M, Gonzalez SF, Frost P, Rombout JHWM, Wiegertjes GF, Savelkoul HFJ, Steinhagen D (2012) Molecular cloning and expression of two β-defensin and two mucin genes in common carp (Cyprinus carpio L.) and their up-regulation after β-glucan feeding. Fish Shellfish Immunol 32:494–501
Medzhitov R (2008) Origin and physiological roles of inflammation. Nature 454:428–435
Metselaar M, Thompson KD, Gratacap RML, Kik MJL, Lapatra SE, Lloyd SJ, Call DR, Smith PD, Adams A (2010) Association of red-mark syndrome with a Rickettsia-like organism and its connection with strawberry disease in the USA. J Fish Dis 33:849–858
Micallef G, Bickerdike R, Reiff C, Fernandes JO, Bowman A, Martin SM (2012) Exploring the transcriptome of Atlantic salmon (Salmo salar) skin, a major defense organ. Mar Biotechnol 14:559–569
Montero J, Garcia J, Ordas MC, Casanova I, Gonzalez A, Villena A, Coll J, Tafalla C (2011) Specific regulation of the chemokine response to viral hemorrhagic septicemia virus at the entry site. J Virol 85:4046–4056
Mustafa A, Mackinnon BM, Piasecki W (2005) Interspecific differences between Atlantic salmon and Artic charr in susceptibility to infection with larval and adult Caligus elongatus: effect of skin mucus protein profiles and epidermal histological differences. Acta Ichthyol Pisc 35:7–13
Neurath MF, Travis SPL (2012) Mucosal healing in inflammatory bowel diseases: a systematic review. Gut 61:1619–1635
Nigam A, Kumari U, Mittal S, Mittal A (2012) Comparative analysis of innate immune parameters of the skin mucous secretions from certain freshwater teleosts, inhabiting different ecological niches. Fish Physiol Biochem 38:1245–1256
Nolan DT, Reilly P, Bonga SEW (1999) Infection with low numbers of the sea louse Lepeophtheirus salmonis induces stress-related effects in postsmolt Atlantic salmon (Salmo salar). Can J Fish Aquat Sci 56:947–959
NRC (2011) Nutrient requirements of fish and shrimp. The National Academies Press, Washington, DC
Ogino C, Yang G (1979) Requirement of carp for dietary zinc. Bull Jpn Soc Sci Fish 45:967–969
Peatman E, Liu Z (2007) Evolution of CC chemokines in teleost fish: a case study in gene duplication and implications for immune diversity. Immunogenetics 59:613–623
Przybylska-Diaz DA, Schmidt JG, Vera-Jiménez NI, Steinhagen D, Nielsen ME (2013) β-glucan enriched bath directly stimulates the wound healing process in common carp (Cyprinus carpio L.). Fish Shellfish Immunol 35:998–1006
Quilhac A, Sire J-Y (1999) Spreading, proliferation, and differentiation of the epidermis after wounding a Cichlid fish, Hemichromis bimaculatus. Anat Rec 254:435–451
Ringø E, Olsen RE, Vecino JLG, Wadsworth S, Song SK (2012) Use of immunostimulants and nucleotides in aquaculture: a review. J Mar Sci Res Dev 1:104. doi:10.4172/2155-9910.1000104
Sandnes K, Torrissen O, Waagbø R (1992) The minimum dietary requirement of vitamin C in Atlantic salmon (Salmo salar) using ascorbate-2-monophosphate as dietary source. Fish Physiol Biochem 10:315–319
Shephard KL (1994) Functions for fish mucus. Rev Fish Biol Fish 4:401–429
Subramanian S, Mackinnon SL, Ross NW (2007) A comparative study on innate immune parameters in the epidermal mucus of various fish species. Comp Biochem Physiol B 148:256–263
Tacchi L, Lowrey L, Musharrafieh R, Crossey K, Larragoite ET, Salinas I (2015) Effects of transportation stress and addition of salt to transport water on the skin mucosal homeostasis of rainbow trout (Oncorhynchus mykiss). Aquaculture 435:120–127
Tenaud I, Leroy S, Chebassier N, Drèno B (2000) Zinc, copper and manganese enhanced keratinocyte migration through a functional modulation of keratinocyte integrins. Exp Dermatol 9:407–416
Tsao CS (1997) An overview of ascorbic acid chemistry and biochemistry. In: Packer L (ed) Vitamin C in health and diseases. Marcel Dekker, New York, pp 25–58
Vatsos I, Kotzamanis Y, Henry M, Angelidis P, Alexis M (2010) Monitoring stress in fish by applying image analysis to their skin mucous cells. Eur J Histochem 54:107–111
Verner-Jeffreys DW, Pond MJ, Peeler EJ, Rimmer GS, Oidtmann B, Way K, Mewett J, Jeffrey K, Bateman K, Reese RA, Feist SW (2008) Emergence of cold water strawberry disease of rainbow trout Oncorhynchus mykiss in England and Wales: outbreak investigations and transmission studies. Dis Aquat Org 79:207–218
Vetvicka V, Vetvickova J (2011) β(1-3)-D-glucan affects adipogenesis, wound healing and inflammation. Orient Pharm Exp Med 11:169–175
Waagbø R (2006) Feeding and disease resistance in fish. In: Mosenthin R, Zentek J, Zebrowska T (eds) Biology of growing animal. Elsevier Limited, London, pp 387–415
Waagbø R (2008) Reducing production related diseases in farmed fish. In: Lie Ø (ed) Improving farmed fish quality and safety. VS Woodhead Publishing, Cambridge, pp 363–398
Wahli T, Verlhac V, Girling P, Gabaudan J, Aebischer C (2003) Influence of dietary vitamin C on the wound healing process in rainbow trout (Oncorhynchus mykiss). Aquaculture 225:371–386
Watanabe T, Kiron V, Satoh S (1997) Trace minerals in fish nutrition. Aquaculture 151:185–207
Whyte SK (2007) The innate immune response of finfish—a review of current knowledge. Fish Shellfish Immunol 23:1127–1151
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jensen, L.B., Wahli, T., McGurk, C. et al. Effect of temperature and diet on wound healing in Atlantic salmon (Salmo salar L.). Fish Physiol Biochem 41, 1527–1543 (2015). https://doi.org/10.1007/s10695-015-0105-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10695-015-0105-2