Abstract
The presence of contaminants of emerging concern (CECs) in wastewater treatment plant effluents is a significant underlying health risk and environmental concern. CECs consist of a wide variety of contaminants, including pharmaceuticals and personal care products, hormones, steroids, alkyl-phenols, flame retardants and pesticides. Their impact is of particular relevance to agricultural settings due to CEC uptake and accumulation in food crops and consequent diffusion into the food-chain. Meanwhile, marijuana reform is accelerating in the US, based on the scope and pace of legalization efforts and on wider acceptance in polls of voters. In this review, the effectiveness of industrial hemp (Cannabis sativa L.) in phytoremediation and hyperaccumulation of organic contaminants (e.g., benzo(a)pyrene, Naphthalene, and Chrysene) and heavy metal (e.g., Selenium and Cobalt) from either aqueous solutions or contaminated soils has been reviewed. The potential of industrial hemp as a renewable resource to biodegrade and/or decontaminate CECs is explored. Disposal strategies of this new phytoremediation crop that promote circular economy are also discussed. According to this current review, we believe the use of industrial hemp for phytoremediation is promising to have a sustainable, environmentally friendly and economically viable future.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
Considerable attention has been attributed to contaminants of emerging concern (CECs) due to their dramatic blossom in wastewater treatment plants (WWTPs) in recent years. CECs are those synthetic or naturally occurring pollutants detected in water bodies which typically fall outside current environmental regulations (Sauve and Desrosiers 2014). The CECs include, but are not limited to pharmaceuticals and personal care products (PPCPs), hormones, flame retardants, pesticide and disinfection byproducts. CECs along with their precursor compounds and metabolites, are discharged into the WWTPs during manufacturing processes and/or through disposal of used and unwanted products. Due to their continuous input and lack of appropriate removal equipment, they maintain pseudo-persistence during wastewater treatment (Gulkowska et al. 2008). The occurrences of CECs in WWTPs effluent and biosolid/sludge have been repeatedly reported with the detected concentrations at ng L−1 to µg L−1 and at ng g−1 to µg g−1 (Alvarez et al. 2014). However, because of the low concentrations of CECs, related regulations in the US have not been well established until there is more firm scientific data (Barbosa et al. 2016). A previous study demonstrated that CECs can cause an extremely wide range of adverse effects to human-beings and various organisms (e.g., chronic, reproductive damage, behavioral changes and accumulation in tissues) even at very low-level exposures (Phillips et al. 2010). For example, veterinary antibiotics exhibited oxidative damage to liver cells of rainbow trout (Oncorhynchus mykiss) (Gagné et al. 2006). The exposure of fish and benthic invertebrates to psychoactive drugs altered their behavioral responses (Rosi-Marshall et al. 2015). Therefore, it is urgent to study the fate and transport of CECs and correspondingly, the removal protocols of CECs.
CECs from the WWTPs effluents have been reported to contaminate surrounding agricultural land via different ways. Reuse of wastewater for irrigation purposes contribute significant amounts of CECs to agricultural systems (Becerra-Castro et al. 2015). Sewage sludge, which was recycled as the soil amendment, also brought a portion of hydrophilic CECs to agricultural soil (Kirchmann et al. 2017). In the agricultural subsoil system, the likelihood of CECs transport was affected by several factors. Among them the most important are: its physicochemical properties (e.g., half-life and polarity), the soil properties and climate conditions (e.g., precipitation, seasons). A study showed that the specific surface area and cation exchange capacity of the soil correlated with the adsorption affinities of a reproductive hormone, 17β-estradiol (Casey et al. 2003). A high correlation between specific surface area and sorption was found (r2 = 0.92) in the experiment. While cationic exchange capacity only partially affect the adsorption affinity because of soil organic matters and clay mineral. Nonylphenol polyethoxylates had fast and complete degradation (initial half time 0.3–5 days). But nonylphenol, the degraded byproduct of nonylphenol polyethoxylates, depredated rapidly in the beginning. 26–35% of them remained in the soil till the end (Sjöström et al. 2008). Those hydrophobic residues were found to be accumulated in the contaminated environments and unable to be further consumed by any plants (Soares et al. 2008). Because of higher precipitation and larger herbicide/pesticide applications, CECs' concentrations in agricultural catchments were found to be significantly higher in summer seasons (Fairbairn et al. 2016). These factors altered the transport of CECs in the subsoil system, which makes the detection and removal even more difficult to conduct.
Among different CECs' decontamination strategies, phytoremediation has received great attention because of its efficiency and cost-effectiveness. It employs plants, crops, and grasses to extract, sequester and eradicate those potentially toxic chemicals in the soil, water and other environments (Kumar et al. 2017; Jiang et al. 2015). Rather than chasing the limited number of CECs, plants effect and control chemical activities including contaminant transport and metabolisms in soil biota, since its intense root systems and continuous exudes. Industrial hemp (Cannabis sativa L.) is one of the widely investigated plants for phytoremediation. The feasibility of in-situ contaminants' removal is mainly credited due to its porous and hydrophilic surface structure, as well as the strong recalcitrance on levels of toxicity. For example, hemp fibers were chosen as the remediator of heavy metal ions (i.e., lead (II), zinc (II) and cadmium (II)). The metal removal efficiencies of hemp that were persuasive ranging from 17.5 to 39% in single/ternary ion metal(s) solutions (Pejic et al. 2011). Campbell et al. (2002) observed large reductions of benzo(a)pyrene (~ 33.5%), but inconsistent results on chrysene from -50% to 64% in the contaminated soil. It was also estimated that contaminant accumulation was highly selective on hemp parts. For example, the accumulation of nickel, lead and cadmium in hemp leaves were 4–12 times larger than the metal in other parts like fibers, seeds and herbs (Linger et al. 2002). It makes the potential remanufacture/reuse of less contaminated hemp parts possible, which aligns with the circular economy. In addition, the short maturation period and high biomass yield of industrial hemp also extended the utilization to other non-food manufacturing in the rest of a year (Kumar et al. 2017).
However, these benefits were not fully realized until the recent marijuana reform policies. According to search results returned by keywords of “industrial hemp” or " Cannabis sativa L." plus “phytoremediation” using the Web of Science database, countries that have most prominently addressed these topics over the last 20 years include Italy, USA, Canada, China and Germany (Fig. 1). The historical importance of hemp in Europe and China leads to the continuous research input in these areas (Salentijn et al. 2015). While in North America, published studies on phytoremediation of industrial hemp evidently exploded until gradual legalization of hemp production in 1990s (Fike 2016). Therefore, industrial hemp is expected to be applied to wider scaled applications with this trend, in which phytoremediation is believed to be a promising answer for CECs removal.
With increasing concerns of CEC contamination, the realization of a low-cost and efficient in-situ CECs removal via hemp is a feasible solution. But it is often not lucrative enough to make it appealing for groups of people because of limited data and large uncertainty. Therefore, it is essential to shed light on the viability and great economic benefit of this strategy before action. This review aims to provide more information about the efficacies of CECs remediation by industrial hemp. The detoxification performance of plants reported in previous studies are collected, estimated, compared and the underlying mechanisms of phytoremediation are revealed in the following sections. Major factors such as soil properties and physicochemical properties of contaminants are investigated in order to optimize the decontamination process. Nonetheless, the application of industrial hemp cannot achieve ambitious goals without addressing the issue of residue management after implantation. Therefore, the potential disposal strategies of phytoremediation via industrial hemp under the framework of the circular economy are rigorously advised in this review.
2 Mechanism of CEC decontamination
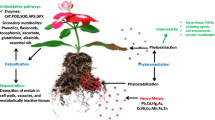
Numerous CEC phytoremediation studies interpreted its mechanism under the guide of the 'green liver model' theory (Burken 2003). This theory successfully brought the concept of mammalian liver function into plants. Plants and mammals have very similar responses to the xenobiotic molecules, except excretion stages only appear in mammals. Under this theory, the functionality of plants' phytoremediation has been considered as the effort of the whole-plant system instead of sole compartment of plants. The overall detoxification can be divided into three stages: translocation, transformation and/or conjugation and sequestration (Fig. 2). However, this theory seems to only envisage the overall trend of organic pollutant decontaminations. In recent experimental and field studies, the observations do not always follow such theoretical steps, sometimes they only partially align with the theory. Therefore, the theory was updated with new findings collected in recent studies (2005 ~ 2020) in order to have a clearer idea of the phytoremediation process, specifically the external CEC-plant-soil interaction.
2.1 Stage I—translocation and ex planta biodegradation
Theoretically, contaminants enter plant tissues by active transport, namely translocation, in the direction opposite of the chemical potential gradient. Such translocation utilizes soil water with a transpiration stream to reach the aerial plant tissues. Besides active transport, passive diffusion was also largely observed in studies which utilized the macrophytes vegetation and/or which were conducted in a hydroponic environment (Tai et al. 2019). In addition, those CECs with semi-volatile characteristics and low molecular weights (MW) were partitioned or adsorbed from the atmospheric air (Gawrońska and Bakera 2015). Volatile organic matter are reported to have two major transports solely or combinedly: partition into waxy cuticle and convey through the surface stomata, followed by the translocation through the phloem (Ferro et al. 2013). A significant portion of CECs has high vapor pressures such as benzene, toluene, ethylbenzene, xylene and methyl tert-butyl ether, which indicates such translocation is very possible. However, the translocation of volatile organic matters is reported less because of current technological difficulties to capture and quantify the contaminants and their metabolites in the air. After plants-CEC interaction, CECs can either be further translocated/transformed or simply adsorbed on plants' waxy cuticles, it is largely dependent on the plants' adsorption capacity and existing enzymatic system.
In recent years, plants were found to be functional externally, namely ex planta phytoremediation. Plant exudes of organic acid are helpful to mobilize those highly hydrophobic contaminants by competing binding sites in a soil matrix. At the same time, plants exude acts as carbon-rich source for the rhizobacteria, which could further detoxify the CECs retained in the soil. Huesemann et al. estimated that highly hydrophobic polynuclear aromatic hydrocarbons were effectively removed (73% decrease) by the eelgrass (Zostera marina) phytoremediation, but only 25% removed in unplanted controls (Huesemann et al. 2009). Since only 0.35% of contaminants were detected in the roots and shoots in the initial 60 weeks, the results suggested that plant-enhanced biodegradation in rhizosphere was the major contributor within the plant-amendment environments. Besides the acceleration of in-situ phytoremediation, this 'defense' system of plants was also reported to prevent further phytotoxicity. To protect plants, plant exudes could further inhibit translocation into aerial tissues and increase defense enzymes activity in affected regions. Such uptake inhibition was documented to relate to specific enzymes and metabolite inhibitor, such as protoporphyrinogen oxidase, 2,4-dinitrophenol (2,4-DNP), and iodo-acetate. It was reported that these inhibitors targeted protoporphyrinogen in cytoplasm (Madalão et al. 2012), proton-couple fluxes through the plasma membrane or endomembrane system (Kong et al. 2007). Specifically, hemp was found to produce a significant amount of salicylic acid, which efficiently induced polycyclic aromatic hydrocarbon (PAH) bacteria and increased PAH mineralization (Liste and Prutz 2006).
2.2 Stage II—internal transformation and conjugation
Plants' internal phytodegradation is similar to xenophobic metabolism of mammalian living organisms. The initial metabolic step is transformation, which includes a series of contaminants' mobilization, such as oxidations, reductions, methylation, dehalogenation, hydroxylation and photolysis (Boonsaner and Hawker 2010). Both mammal and plants rely largely on cytochrome P450 monooxygenases to metabolize exotic chemicals (Nebert et al. 2013). This step increases the hydrophilicity, which serves as the pre-treatment for the following conjugation. Transformation also allows contaminants to be hydrophilic enough to diffuse into the cytoplasm through apoplast pathway (Tanoue et al. 2012). Conjugation is the predominant step in the detoxification, which also serves as a protection system against high oxidative stress caused by xenobiotics. It utilizes enzymes like glycosyltransferase, glutathione S-transferases, peroxidases, and hydrolases to combine the transformed metabolites with natural molecules like sugar, amino acid and malonate. The detected metabolites and conjugates showed the same order with theoretical metabolisms. Since the rapid metabolism and associated conjugation normally happen in a relatively short-time period, these two steps sometimes are investigated together in previous studies (Huber et al. 2009). For example, diclofenac metabolism has been extensively studied, which includes a rapid hydroxylation, followed by the conjugation with glucuronide and/or sulfate. Bartha et al. (2014) found that 4′–OH diclofenac, the product of transformation, were detected only after one day in even higher concentration than diclofenac itself. 4′–O–glucopyranosyl–oxydiclofenac, product of conjugation, was found to have increase concentration after 3 days. However, the frequency and concentration of the detected conjugates are still very limited compared to the transformed metabolites. In the previous example, only 20% of the metabolites have been conjugated in the first 3 days and remained unchanged afterward.
So far, there is still limited information on the contaminant’s metabolism. Most of metabolism investigations refer to the similar detoxification conducted in mammalian liver more or less. The difficulty falls in the technological detection of low-concentration, unknown and complexed metabolites as well as resulted conjugates. Additionally, the lack of halogen on these polar contaminants make it even more difficult to detect due to the lack of isotope in mass spectra (Fu et al. 2018). As stated in the green liver model theory, the metabolism is a whole-plant activity. But degrees of metabolisms, involved enzymes, and products can vary. For example, metabolic pathways of carbamazepine (CB) are well-documented, which constitutes epoxidation by cytochrome P450 enzyme, hydroxylation by epoxide hydrolase enzyme, and conjugates with glucuronide (Dordio et al. 2011). Dordio et al. (2011) found that only one of the metabolites, epoxide–CB, was detected in the leaf of Typha spp. after 21 days. Mordechay et al. (2018) demonstrated epoxide-CB, which substituted ~ 60% of parent molecule, was detected in wheat ear after 155 days without the detected dihydroxy-CB. In tomato fruit, dihydroxy–CB replaced ~ 50% of parent molecule without detected epoxide-CB after 98 days. Only lettuce leaf showed a comprehensive metabolites' distribution which includes epoxide–CB (20%), dihydroxy-CB (20%), and CB itself (60%) after 42 days. None of them have detected the CB-conjugates. Even the same plant would carry out several metabolisms at the same time. For example, Macherius et al. (2012) found that triclosan was rapidly adsorbed up to 95% into the carrots tissue in the first 2 h, and subsequently, followed by a quick metabolism with a 9-h half-life. Eight different metabolites were detected during the triclosan detoxification, which continuously conjugated with saccharides, disaccharides, malonic acid, amino acid and sulfate. Tai et al. (2019) detected a total of 15 metabolites in the root of Iris pseudacorus., 9 of them in shoot, and a confirmed acetyl-conjugate in exposure of sulfonamides. These results proved the metabolisms are largely plant specific. It also revealed the importance of selecting an appropriate plant for phytoremediation.
Xenophobic metabolisms have some similarities among plants. Besides the similarity of involved enzymes (e.g., cytochrome P450 enzyme, glutathione S-transferases and glycosyltransferase), some studies indicated that those contaminants bearing polar functional groups are easier to be detoxified, such as phenolic group, amines and carboxylic acids (Macherius et al. 2012). Contaminants with lower molecular weight contaminants are easily detoxified than higher molecular weight. But moieties such as chloride, nitro and methyl groups, which have stronger steric hindrance than others, are harder to be transformed (Fu et al. 2017). Amino acid conjugates are consistently detected in quantities contaminants, but sometimes as a side reaction (He et al. 2017).
2.3 Stage III—CECs sequestrations and/or accumulation
The major difference between mammalian and plant metabolism is in the ultimate fate-storage, which is opposite to excretion in mammals. Due to the lack of excretion pathway, most of the metabolites and conjugates are sequenced in the plant tissues, which is also termed as 'excretion storage'. There are at least three terminal fates within the plant tissues for sequestration: storage in cell vacuole, storage in the apoplast, or covalent binding to cell walls (Bartha et al. 2014). And few compounds are reported to be volatized through stomatal pores on the leaves (Dordio et al. 2009). It was also found that released conjugates and plant natural exudes could be further re-uptaken to enhance the bioavailability of residual contaminants in soil environment (Tai et al. 2019). Some contaminants that are not metabolized or sequestered are released to the atmosphere through stoma on leaves surface (Barbour et al. 2005). Some are partitioned into plant lipids during the translocation without further metabolism (Tai et al. 2019).
3 Phytoremediation potential of industrial hemp
3.1 Performance of industrial hemp for the decontamination of CECs
So far, the fiber production and medical value of industrial hemp have been dominantly recognized. However, because of the long-term illegalization for planting industrial hemp in the North America, its phytoremediation potential has been largely unknown. In the limited industrial hemp phytoremediation studies available, the effectiveness of hemp’s ability to remove heavy metals has been repeatedly proven (Tofan et al. 2013; Stonehouse et al. 2020; Praspaliauskas et al. 2020). Compared to other popular phytoremediation species, such as mustard and sunflower, hemp exhibited excellent removal efficiency with heavy metals (Meers et al. 2005). Industrial hemp is ideal for heavy metal removal owing to its large and porous surface structure, low nutrient requirements and high contaminants tolerance. Studies demonstrated that hemp can survive in highly contaminated sites such as landfill leachate, mine area, where very few crop plants could survive (Mihoc et al. 2012). Accumulated heavy metal can be further digested, metabolized and even be exuded as dietary form of heavy metal (Stonehouse et al. 2020). Industrial hemp also performed good clean-up of CECs. CECs with a wide range of molecular weights and solubilities were remediated with hemp (Table 1). Several studies reported that the soil remediation and microbial activity were also enhanced by the hemp cultivation. During the growth of hemp, powerful allelopathic chemicals through its root has been secreted, accelerating the soil remediation (Linger et al. 2002). Similarly, a study conducted on Zea mays proved the biogenetic, rather than anthropogenic sources of hydrocarbon contents in soil (Grifoni et al. 2020). Bacteria including Achromobacter sp., Pseudomonas sp., and Alcaligenes sp., which were isolated from hemp, completely degraded the phenol and benzene, indicating great phytoremediation potential of hemp (Iqbal et al. 2018). Co-contaminations of heavy metal and CECs are even more challenging. The phytoremediation results are debatable because of the different functions of heavy metals and the interactions between heavy metal and organic contaminants. Pyrene was reported to facilitate copper penetration into the plant cell, increasing copper accumulation (Chigbo et al. 2013). The competition between heavy metal and organic contaminants, on the other hand, resulted in the limited phytoextraction of residual contaminants. For example, the accumulation of norfloxacin in rice roots was inhibited with the increasing dosage of Fe (II) (0–50 mg Kg−1) (Yan et al. 2017). However, a handful of studies have investigated the contaminants with halogenated functional groups or with larger molecular weights (> 400), which are commonly found as CECs. Studies investigating the phytoremediation potential of these CECs are urgently needed as the increasing contamination is found globally (Marsik et al. 2017; Mattes et al. 2018).
3.2 Factors influencing the performance
3.2.1 Physicochemical properties of contaminants
Physicochemical properties (i.e., log Kow, molecular weight and numbers of carbon) of the contaminants are dominant factors to consider during the uptake of xenobiotics. As discussed in Sect. 2, plant uptake of contaminants occurs by passive and/or active transport. Compounds characterized as semi-volatile or volatile can be partitioned or adsorbed from the atmospheric air. These quite different decontamination approaches are resulted from the physicochemical properties, collectively described as octanol-water partitioning coefficient (Kow or Dow) and octanol-air partitioning coefficient (Koa) (Lin et al. 2007). Predictions have been made that nonionic compounds, which log Kow in a range of 0.5–3.5 are lipophilic enough to move through the lipid bilayer of membrane, yet water-soluble enough to transfer into the cell fluid. For passive transport of poorly water-soluble organic compounds, it has been noted that the single and most important plant characteristic is the plant lipid content (Barbour et al. 2005). This might indicate that multiple uptake mechanisms occurred. It has also found that log Kow smaller than 2 exhibited a more diverse bioaccumulation factor (BCF), which represents the uptake efficiency of contaminants from the studied medium (Fig. 3). Plants exhibited complex preferences on remediation of compounds with different molecular weights (MW), especially when MW > 300. In some cases, contaminants with higher molecular weights have more resistance to biodegradation, resulting in decreased accumulations to plants (Liste and Prutz 2006). Some studies showed high BCF (> 3) of compounds with large molecular weight. This result, however, was contributed by multiple factors including optimal growth medium (e.g., commercial potting soil and Hoagland nutrient solution), smaller logKow and longer study duration (Cui et al. 2017; Boonsaner and Hawker 2010; Yan et al. 2017). And the lower transformation efficiencies ranging from 0 to 0.06 (from root to shoot) were found with these compounds (Cui et al. 2016; Yan et al. 2017). As for weak electrolytes, parameters have different reflections on the plant uptake. Dow is the normalized parameter to Kow considering the effect of ionized functional groups. Dow is largely dependent on the factors such as the acid–base coefficient (pKa) of the compound and the medium pH. It can be estimated with the following equation: \(D_{\text{OW}}= K_{\text{ow}}{\text{(1 + }{10}^{\text{pH} - {\text{pK}}_{\text{a}}}\text{)}}^{-1}\)(Halling-Sørensen et al. 1998). Contaminants transported in phloem was found to be optimal for compounds of intermediate hydrophobicity (Log Kow 1–3) and weak acidity (pKa 3–6) (Trapp 2004). This indicates the contaminants with these properties have larger chances to be further metabolized. However, in general, there are still very limited studies investigating the weak electrolyte's phytoremediation.
3.2.2 Plant type and growing medium
Selection of plants as phytoremediators depends on the phytoremediation efficiency. Higher efficiency and more attention have been found with herbs (e.g., mustard and industrial hemp) and grains (e.g., soybean and corn) (Fig. 3). Common characteristics they share are high tolerance to on-site pollution, short life cycles and handling ease. Fibrous root systems are also preferred because of the large rhizoplane surface area, which enriches the microbial population (Escalante-Espinosa et al. 2005). For example, Alfalfa increased the number of both culturable, aerobic heterotrophic and PAH degrading bacteria in the rhizosphere after 7 weeks compared to bulk soil (Kirk et al. 2005). Uncontaminated soil also showed the increasing amount of large molecular hydrocarbon (C > 12) with the growth of maize (Zea mays), indicating the biogenic sources of the compounds (Grifoni et al. 2020). Yellow medick (Medicago falcata L.) and alfalfa were both effective at on-site remediation of petroleum contaminants, but the number of hydrocarbon-oxidizing microorganisms were larger in the rhizosphere zone of alfalfa (Panchenko et al. 2017). Soil pH and pKa values of contaminants combinedly decided the neutral or ionic form in the study medium, determining the availability to plants. However, uptake efficiencies and forms of contaminants are not always closely related in studies. For example, in soil (pH = 6.5–6.7) fortified with pharmaceuticals, uptake efficiencies were reported the greatest of carbamazepine (neutral) and sulfamethoxazole (ionic) in plant tissue, suggesting that factors other than soil pH affect the uptake (Holling et al. 2012). Similarly, Herklotz et al. (2010) found the largest accumulation in cabbage tissue was correlated to the compound with lowest pKa, sulfamethoxazole (ionic form in studied medium). Some studies found the metabolism could be independent from environmental factors, such as soil type, carrier medium and solely controlled by the total amount taken up by the plant (Mordechay et al. 2018). Therefore, comparing to medium properties, characteristics of target compounds seem to play larger roles in uptake efficiencies.
4 Circular economy of industrial hemp applications and management
4.1 Hemp potential disposal strategies
It is essential to discuss the disposal strategies of industrial hemp when considering the economic benefits. Industrial hemp is a particularly vigorous annual crop, which can be seeded from late spring when the soil just starts to warm. It has a remarkable growth rate, which has been recorded with heights from 1 to 5 m by August (Ip and Miller 2012). High tolerance of contaminants make hemp valuable in terms of its further application after phytoremediation (Kumar et al. 2017). Estimating from previous studies, contaminants only showed limited effect on hemp quality. For example, Linger et al. (2002) indicated that heavy metal contamination did not show negative effect on the hemp fiber quality (i.e., fineness or strength) or quantity. Revealed by greenhouse experiments, dry biomass of hemp cultivated in a moderately contaminanted area decreased up to 40% compared to those in uncontaminated soil (Pietrini et al. 2019). While chronolyII content and other photochemistry parameter only slightly reduced, indicating the good physiological status of plants. Husain et al. (2019) also demonstrated that the height of hemp in contaminated soil and commercial soil exhibited no significant difference when heavy metal accumulation were detected in all parts of hemp.
So far, industrial hemp, serving as an economical crop, has been extensively applied to the fields including bioenergy, paper, construction etc. (Table 2). Hemp can either be utilized as the whole plant or each individual part, such as hemp fibers, seeds and inflorescences (Table 2). These non-food applications can also be applied to the disposal strategies with appropriate pretreatments and investigations. In order to optimize socioeconomic benefits and limited environmental impact of hemp disposal after phytoremediation, strategies that target each individual part instead of whole plant are suggested.
4.1.1 Hemp fibers
Hemp fibers have been largely employed in bioenergy, paper construction sectors (Bowyer 2001). In terms of bioenergy production, it has been recognized as an economic option with low environmental impacts. High cellulose content (~ 44%) as well as high biomass yield ensure hemp a suitable crop for bioenergy production (Kumar et al. 2017). Nowadays, 36% of the global energy consumption is in the form of conventional liquid fuels including petrol and diesel (Staples et al. 2017). Therefore, the utilization of bioenergy (e.g., bioethanol, biodiesel, biobutanol, and biogas) instead of conventional energy resources exhibits environmental, energy-saving and socioeconomic advantages (González-García et al. 2012). Bioethanol, as recorded, can be produced from the cellulose and lignocellulosic biomass, such as agricultural residues, herbaceous crops or forestry residues (Hou et al. 2020). These materials are abundant and inexpensive, as promising bioenergy production resources. Hemp fiber can also be utilized for paper production. However, there is no record of 100% hemp-based paper production (Naithani et al. 2020). It was also not economically or environmentally competitive comparing to conventional tree plantation (Bowyer 2001).
4.1.2 Hemp hurds
During hemp fiber separation, hemp crops also produce large amount of byproducts: hemp and dust (González-García et al. 2012). Hurds are non-fiber components obtained by retting hemp stem (Scrucca et al. 2020). The chemical contents of hemp hurds are very close to that of wood species, with a high portion of cellulose and hemicellulose. Therefore, it has mainly contributed to the animal bedding production and construction sector. Several studies also estimated the feasibility of converting hemp hurds into bioethanol (Barta et al. 2010; González-García et al. 2012). But hemp hurds-related technologies are relatively new and still under development.
4.1.3 Hemp shiv
Hemp bales are shopped and decertified. The bales are then separated to hemp shiv, hemp fiber and hemp dust. Hemp shiv have been implemented in hemp-lime wall construction. At the same time, hemp fiber and hemp dust are recycled in use of other products. The dust can be utilized as filler in plastics, lime render or compressed for use as fuel logs (Ip and Miller 2012). Hemp-lime wall has been used in Europe early from 1990s, but not commonly accepted (Evrard and De Herde 2010). Nowadays, due to increasing efforts being made on environmental protection and greenhouse gas (GHG) emission reduction, hemp-lime construction is striking globally. Hemp, serving a part of construction material, significantly enhances construction status from different aspects, such as high levels of airtightness, improved air quality and lower energy consumption because of latent thermal capacities of hemp-lime wall (Shea et al. 2012). It was estimated that hemp-lime wall would be able to last over 100 years, or at least as durable as traditional wall (Cripps and Fovargue 2004).
4.1.4 Hemp seeds
Biodiesel is clean production as an alternative to petroleum-based diesel fuel. Biodiesel production has already been commercialized by utilizing crop oils such as sunflower, palm tree and soybean (Meher et al. 2006). Although not largely commercialized, industrial hemp is one of the most promising sources. It has a high yield of oil and biomass, indicating potential production of both biodiesel and bioethanol simultaneously (Li et al. 2010). Li et al. successfully convert the oil of hemp seed to biodiesel through the base-catalyzed transesterification with the conversion yield of 97% (Li et al. 2010). Yang et al. (2010) used a one-pot process, which combined transesterification and selective hydrogenation, converted hemp seed oil to biodiesel with a yield of 96%.
4.1.5 Hemp inflorescences
Industrial hemp has the potential to be employed as insecticides because of its inflorescences. Glandular hair, which exuded oleoresin (a barrier entrapping plant enemies), are accumulated on its inflorescences (G. Benelli et al. 2018b, a). Benelli et al. estimated that the essential oil extracted from hemp inflorescences via hydrodistillation is efficient as natural insecticides for the control and management of mosquito vectors, houseflies and moth pests (Giovanni Benelli et al. 2018a, b). Additionally, the essential oil exhibited protection for earthworms, which are major contributor to the consumption of biodegradable materials and organic waste, which could be converted to vermicast, unlike synthetic insecticides that kill almost all earthworms instantly (Pavela 2018).
4.2 Outlook: implications for industrial hemp based on circular management
In the last few years, the circular economy (CE) has emerged as an initiative facing resource depletion and global climate change (Manriquez-Altamirano et al. 2020). Protocols (e.g., alternatives, technologies and practices) that allow substituting non-renewable natural resources to resources recovered from 'waste' thereby achieve socioeconomic and environmental goals of the CE concept (Lieder and Rashid 2016). In addition, CE system would also positively affect local by-product demand if managed properly. For example, using locally produced and homogenous feedstock can alleviate the economic and environmental concerns caused by long-range transportation and complicated materials supply, subsequently assisting local businesses and employment (Bolognesi et al. 2019). Therefore, industrial hemp after phytoremediation should be re-considered as a 'resource' instead of 'waste' by implanting a more holistic circular economy model (Fig. 4). Before the conduction of such system, pot trial and field trial are required in order to examine the toxicity of hemp/hemp parts and feasibility of the proposed strategy. According to the characterization of contaminants and meso/macro level tests, appropriate amounts of industrial hemp would be applied for the phytoremediation purpose. After 120 days of growth, mature hemp would be qualified by the amount of biomass, volume, height and level of phytoxicity. Hemp, with higher quality will be continuously processed for further recycling/remanufacturing/reusing processes. While hemp with lower quality would be pyrolysis/digested on-site depending on the treatment efficiency and local conditions. It can also serve as an energy supply to local industries. Nonetheless, the CE system cannot achieve its ambitious goals without addressing residue management after all the steps (Peng and Pivato 2019). Those residues (e.g., ashes and dust) and contaminant-concentrated hemp could be sent back to the domestic boilers for heating purposes with the goal of reducing treatment costs as well as energy consumption.
4.3 Challenges
Recent studies discussed previously shed light on the potential disposal strategies of hemp crops after phytoremediation. But the challenges of industrial hemp utilization remain unsolved. Studies investigating disposal strategies of industrial hemp achieved different results on aspects of the economic and environmental performance mainly due to the lack of a commercial production line. Most of the study methods are limited to the literature review, life-cycle analysis (LCA) and lab-scaled experiments. In addition, the estimations such as LCA are largely based on the literature instead of experimental data, reasonable assumptions, and results of similar crops, making the estimates less accurate.
The application of industrial hemp might negatively affect the environment during crop growth. It was demonstrated that bioenergy production significantly increases greenhouse gas emissions, acidification, and eutrophication because of the feedstock cultivation (Martin et al. 2014). González-García et al. (2012) indicated that 85% blending of ethanol with fossil fuels contributed more than 2 times of GHG to global warming than conventional fuel energy. The study, which investigated the environmental impacts of hemp and flax for non-wood pulp mills showed 80% of greenhouse gas emissions are contributed by fertilizer usage during hemp cultivation (Gonzalez-Garcia et al. 2010).
Not all the hemp parts are feasible for recycling. For example, the recycling strategy of hemp ashes as mineral fertilizer is still under discussion because ash can only replace a minor portion of fertilizer and requires a similar amount of energy. However, ash recycling is an important tool for closing nutrient cycles considering phosphorus deposit depletion (Prade et al. 2012). In addition, it is uncertain whether the ashes are non-toxic serving as mineral fertilizer. In fact, some studies have determined that the heavy metal contaminated biomass are not only reduced in weight, but also concentrate the heavy metals in ash/char fraction. Nevertheless, their individual pyrolysis fraction, i.e., bio-oil/tar and gas, are both heavy metal free (Liu et al. 2012). Therefore, the utilization of ashes are doubtable, but the volorization of metal might be feasible.
Last but not least, some strategies are commercially uncompetitive. For example, biofuels are less acceptable worldwide because the technology is still being developed. Even though the biomass is relatively abundant and inexpensive, not all of them can be utilized for biofuel production, mainly due to the difficulty of effective hydrolysis in terms of cost and energy consumption (González-García et al. 2012).
5 Conclusions
With increasing attention and legalization policies on industrial hemp, comprehensive utilizations of this precious crop have been studied in recent years. This review estimated the phytoremediation potential of CECs and the underlying mechanism of industrial hemp. The Green Liver Model precisely described CECs uptake and detoxification with several steps: transformation, translocation, conjugations and final storage. Studies in recent years (2005–2020) were found to further complete this theory by adding several transformation pathways, final storage terminals and ex planta biodegradation. Contaminants physicochemical properties were found to play a more significant role than soil or plant properties in regards of the phytoremediation performances. However, there are still limited studies available that employ industrial hemp for organic compound decontamination. The circular economy system has been applied in the disposal strategies, in order to optimize the socioeconomic as well as environmental benefits of industrial hemp. In addition, circular economy protocol can also promote local business development and employment if well managed. Future research is urgently required on performance aspects of hemp on phytoremediation and realization of its circular disposal/recycling managements.
References
Alvarez, D. A., Maruya, K. A., Dodder, N. G., Lao, W. J., Furlong, E. T., & Smalling, K. L. (2014). Occurrence of contaminants of emerging concern along the California coast (2009–10) using passive sampling devices. Marine Pollution Bulletin, 81(2), 347–354. https://doi.org/10.1016/j.marpolbul.2013.04.022.
Barbosa, B., Costa, J., Fernando, A. L., & Papazoglou, E. G. (2015). Wastewater reuse for fiber crops cultivation as a strategy to mitigate desertification. Industrial Crops and Products, 68, 17–23. https://doi.org/10.1016/j.indcrop.2014.07.007.
Barbosa, M. O., Moreira, N. F. F., Ribeiro, A. R., Pereira, M. F. R., & Silva, A. M. T. (2016). Occurrence and removal of organic micropollutants: An overview of the watch list of EU Decision 2015/495. Water Research, 94, 257–279. https://doi.org/10.1016/j.watres.2016.02.047.
Barbour, J. P., Smith, J. A., & Chiou, C. T. (2005). Sorption of aromatic organic pollutants to grasses from water. Environmental Science & Technology, 39(21), 8369–8373. https://doi.org/10.1021/es0504946.
Barta, Z., Oliva, J. M., Ballesteros, I., Dienes, D., Ballesteros, M., & Reczey, K. (2010). Refining hemp hurds into fermentable sugars or ethanol. Chemical and Biochemical Engineering Quarterly, 24(3), 331–339.
Bartha, B., Huber, C., Harpaintner, R., & Schröder, P. (2010). Effects of acetaminophen in Brassica juncea L. Czern.: investigation of uptake, translocation, detoxification, and the induced defense pathways. Environmental Science and Pollution Research, 17(9), 1553–1562. https://doi.org/10.1007/s11356-010-0342-y.
Bartha, B., Huber, C., & Schroder, P. (2014). Uptake and metabolism of diclofenac in Typha latifolia—how plants cope with human pharmaceutical pollution. Plant Science, 227, 12–20. https://doi.org/10.1016/j.plantsci.2014.06.001.
Becerra-Castro, C., Lopes, A. R., Vaz-Moreira, I., Silva, E. F., Manaia, C. M., & Nunes, O. C. (2015). Wastewater reuse in irrigation: A microbiological perspective on implications in soil fertility and human and environmental health. Environment International, 75, 117–135. https://doi.org/10.1016/j.envint.2014.11.001.
Benelli, G., Pavela, R., Lupidi, G., Nabissi, M., Petrelli, R., Kamte, S. L. N., et al. (2018a). The crop-residue of fiber hemp cv. Futura 75: From a waste product to a source of botanical insecticides. Environmental Science and Pollution Research, 25(11), 10515–10525. https://doi.org/10.1007/s11356-017-0635-5.
Benelli, G., Pavela, R., Petrelli, R., Cappellacci, L., Santini, G., Fiorini, D., et al. (2018b). The essential oil from industrial hemp (Cannabis sativa L.) by-products as an effective tool for insect pest management in organic crops. Industrial Crops and Products, 122, 308–315. https://doi.org/10.1016/j.indcrop.2018.05.032.
Bolognesi, S., Bernardi, G., Callegari, A., Dondi, D., & Capodaglio, A. G. (2019). Biochar production from sewage sludge and microalgae mixtures: Properties, sustainability and possible role in circular economy. Biomass Conversion and Biorefinery. https://doi.org/10.1007/s13399-019-00572-5.
Boonsaner, M., & Hawker, D. (2010). Accumulation of oxytetracycline and norfloxacin from saline soil by soybeans. Science of the Total Environment, 408(7), 1731–1737. https://doi.org/10.1016/j.scitotenv.2009.12.032.
Bourmaud, A., Le Duigou, A., & Baley, C. (2011). What is the technical and environmental interest in reusing a recycled polypropylene–hemp fiber composite? Polymer Degradation and Stability, 96(10), 1732–1739. https://doi.org/10.1016/j.polymdegradstab.2011.08.003.
Bowyer, J. L. (2001). Industrial hemp (Cannabis sativa L.) as a papermaking raw material in Minnesota: Technical, economic, and environmental considerations. Department of Wood & Paper Science Report Series.
Burken, J. G. (2003). Uptake and metabolism of organic compounds: Green-liver model. In S. C. McCurtcheon & J. L. Schnoor (Eds.), Phytoremediation: Transformation and control of contaminants. Hoboken, NJ: Wiley. https://doi.org/10.1002/047127304X.ch2.
Calderón-Preciado, D., Renault, Q., Matamoros, V., Cañameras, N., & Bayona, J. M. (2012). Uptake of organic emergent contaminants in spath and lettuce: An in vitro experiment. Journal of Agricultural and Food Chemistry, 60(8), 2000–2007. https://doi.org/10.1021/jf2046224.
Campbell, S., Paquin, D., Awaya, J. D., & Li, Q. X. (2002). Remediation of benzo [a] pyrene and chrysene-contaminated soil with industrial hemp (Cannabis sativa). International Journal of Phytoremediation, 4(2), 157–168. https://doi.org/10.1080/15226510208500080.
Casey, F. X. M., Larsen, G. L., Hakk, H., & Simunek, J. (2003). Fate and transport of 17 beta-estradiol in soil-water systems. Environmental Science & Technology, 37(11), 2400–2409. https://doi.org/10.1021/es026153z.
Chigbo, C., Batty, L., & Bartlett, R. (2013). Interactions of copper and pyrene on phytoremediation potential of Brassica juncea in copper–pyrene co-contaminated soil. Chemosphere, 90(10), 2542–2548. https://doi.org/10.1016/j.chemosphere.2012.11.007.
Cripps, A., & Fovargue, J. (2004). Crops in construction handbook. London: CIRIA.
Cui, H., de Angelis, M. H., & Schröder, P. (2017). Iopromide exposure in Typha latifolia L.: Evaluation of uptake, translocation and different transformation mechanisms in planta. Water Research, 122, 290–298. https://doi.org/10.1016/j.watres.2017.06.004.
Cui, H. B., Fan, Y. C., Yang, J., Xu, L., Zhou, J., & Zhu, Z. Q. (2016). In situ phytoextraction of copper and cadmium and its biological impacts in acidic soil. Chemosphere, 161, 233–241. https://doi.org/10.1016/j.chemosphere.2016.07.022.
Dordio, A. V., Belo, M., Teixeira, D. M., Carvalho, A. J. P., Dias, C. M. B., Pico, Y., et al. (2011). Evaluation of carbamazepine uptake and metabolization by Typha spp., a plant with potential use in phytotreatment. Bioresource Technology, 102(17), 7827–7834. https://doi.org/10.1016/j.biortech.2011.06.050.
Dordio, A. V., Duarte, C., Barreiros, M., Carvalho, A. J. P., Pinto, A. P., & da Costa, C. T. (2009). Toxicity and removal efficiency of pharmaceutical metabolite clofibric acid by Typha spp.—potential use for phytoremediation? Bioresource Technology, 100(3), 1156–1161. https://doi.org/10.1016/j.biortech.2008.08.034.
Escalante-Espinosa, E., Gallegos-Martínez, M. E., Favela-Torres, E., & Gutiérrez-Rojas, M. (2005). Improvement of the hydrocarbon phytoremediation rate by Cyperus laxus Lam. inoculated with a microbial consortium in a model system. Chemosphere, 59(3), 405–413. https://doi.org/10.1016/j.chemosphere.2004.10.034.
Evrard, A., & De Herde, A. (2010). Hygrothermal performance of lime-hemp wall assemblies. Journal of Building Physics, 34(1), 5–25. https://doi.org/10.1177/1744259109355730.
Fairbairn, D. J., Karpuzcu, M. E., Arnold, W. A., Barber, B. L., Kaufenberg, E. F., Koskinen, W. C., et al. (2016). Sources and transport of contaminants of emerging concern: A two-year study of occurrence and spatiotemporal variation in a mixed land use watershed. Science of the Total Environment, 551, 605–613. https://doi.org/10.1016/j.scitotenv.2016.02.056.
Ferro, A. M., Kennedy, J., & LaRue, J. C. (2013). Phytoremediation of 1, 4-dioxane-containing recovered groundwater. International Journal of Phytoremediation, 15(10), 911–923. https://doi.org/10.1080/15226514.2012.687018.
Fike, J. (2016). Industrial hemp: Renewed opportunities for an ancient crop. Critical Reviews in Plant Sciences, 35(5–6), 406–424. https://doi.org/10.1080/07352689.2016.1257842.
Fu, Q., Dudley, S., Sun, C., Schlenk, D., & Gan, J. (2018). Stable isotope labeling-assisted metabolite probing for emerging contaminants in plants. Analytical Chemistry, 90(18), 11040–11047. https://doi.org/10.1021/acs.analchem.8b02807.
Fu, Q., Zhang, J., Borchardt, D., Schlenk, D., & Gan, J. (2017). Direct conjugation of emerging contaminants in Arabidopsis: Indication for an overlooked risk in plants? Environmental Science & Technology, 51(11), 6071–6081. https://doi.org/10.1021/acs.est.6b06266.
Gagné, F., Blaise, C., & André, C. (2006). Occurrence of pharmaceutical products in a municipal effluent and toxicity to rainbow trout (Oncorhynchus mykiss) hepatocytes. Ecotoxicology and Environmental Safety, 64(3), 329–336. https://doi.org/10.1016/j.ecoenv.2005.04.004.
Gawrońska, H., & Bakera, B. (2015). Phytoremediation of particulate matter from indoor air by Chlorophytum comosum L plants. Air Quality, Atmosphere & Health, 8(3), 265–272. https://doi.org/10.1007/s11869-014-0285-4.
Gonzalez-Garcia, S., Hospido, A., Feijoo, G., & Moreira, M. T. (2010). Life cycle assessment of raw materials for non-wood pulp mills: Hemp and flax. Resources Conservation and Recycling, 54(11), 923–930. https://doi.org/10.1016/j.resconrec.2010.01.011.
González-García, S., Luo, L., Moreira, M. T., Feijoo, G., & Huppes, G. (2012). Life cycle assessment of hemp hurds use in second generation ethanol production. Biomass and Bioenergy, 36, 268–279. https://doi.org/10.1016/j.biombioe.2011.10.041.
Grifoni, M., Rosellini, I., Angelini, P., Petruzzelli, G., & Pezzarossa, B. (2020). The effect of residual hydrocarbons in soil following oil spillages on the growth of Zea mays plants. Environmental Pollution, 265, 114950. https://doi.org/10.1016/j.envpol.2020.114950.
Gulkowska, A., Leung, H. W., So, M. K., Taniyasu, S., Yamashita, N., Yeung, L. W. Y., et al. (2008). Removal of antibiotics from wastewater by sewage treatment facilities in Hong Kong and Shenzhen China. Water Research, 42(1), 395–403. https://doi.org/10.1016/j.watres.2007.07.031.
Halling-Sørensen, B., Nors Nielsen, S., Lanzky, P. F., Ingerslev, F., Holten Lützhøft, H. C., & Jørgensen, S. E. (1998). Occurrence, fate and effects of pharmaceutical substances in the environment- a review. Chemosphere, 36(2), 357–393. https://doi.org/10.1016/S0045-6535(97)00354-8.
He, Y. J., Langenhoff, A. A. M., Sutton, N. B., Rijnaarts, H. H. M., Blokland, M. H., Chen, F. R., et al. (2017). Metabolism of ibuprofen by phragmites australis: Uptake and phytodegradation. Environmental Science & Technology, 51(8), 4576–4584. https://doi.org/10.1021/acs.est.7b00458.
Herklotz, P. A., Gurung, P., Vanden Heuvel, B., & Kinney, C. A. (2010). Uptake of human pharmaceuticals by plants grown under hydroponic conditions. Chemosphere, 78(11), 1416–1421. https://doi.org/10.1016/j.chemosphere.2009.12.048.
Holling, C. S., Bailey, J. L., Heuvel, B. V., & Kinney, C. A. (2012). Uptake of human pharmaceuticals and personal care products by cabbage (Brassica campestris) from fortified and biosolids-amended soils. Journal of Environmental Monitoring, 14(11), 3029–3036. https://doi.org/10.1039/c2em30456b.
Hou, J. J., Zhang, X. T., Liu, S. J., Zhang, S. D., & Zhang, Q. Z. (2020). A critical review on bioethanol and biochar production from lignocellulosic biomass and their combined application in generation of high-value byproducts. Energy Technology. https://doi.org/10.1002/ente.202000025.
Huber, C., Bartha, B., Harpaintner, R., & Schröder, P. (2009). Metabolism of acetaminophen (paracetamol) in plants—two independent pathways result in the formation of a glutathione and a glucose conjugate. Environmental Science and Pollution Research, 16(2), 206. https://doi.org/10.1007/s11356-008-0095-z.
Huesemann, M. H., Hausmann, T. S., Fortman, T. J., Thom, R. M., & Cullinan, V. (2009). In situ phytoremediation of PAH-and PCB-contaminated marine sediments with eelgrass (Zostera marina). Ecological Engineering, 35(10), 1395–1404. https://doi.org/10.1016/j.ecoleng.2009.05.011.
Husain, R., Weeden, H., Bogush, D., Deguchi, M., Soliman, M., Potlakayala, S., et al. (2019). Enhanced tolerance of industrial hemp (Cannabis sativa L.) plants on abandoned mine land soil leads to overexpression of cannabinoids. PLoS ONE, 14(8), e0221570. https://doi.org/10.1371/journal.pone.0221570.
Ip, K., & Miller, A. (2012). Life cycle greenhouse gas emissions of hemp–lime wall constructions in the UK. Resources, Conservation and Recycling, 69, 1–9. https://doi.org/10.1016/j.resconrec.2012.09.001.
Iqbal, A., Arshad, M., Hashmi, I., Karthikeyan, R., Gentry, T. J., & Schwab, A. P. (2018). Biodegradation of phenol and benzene by endophytic bacterial strains isolated from refinery wastewater-fed Cannabis sativa. Environmental Technology, 39(13), 1705–1714. https://doi.org/10.1080/09593330.2017.1337232.
Jiang, Y., Lei, M., Duan, L. B., & Longhurst, P. (2015). Integrating phytoremediation with biomass valorisation and critical element recovery: A UK contaminated land perspective. Biomass & Bioenergy, 83, 328–339. https://doi.org/10.1016/j.biombioe.2015.10.013.
PubChem 2019 update: improved access to chemical data. (2019). https://www.nlm.nih.gov/toxnet/index.html.
Kirchmann, H., Borjesson, G., Katterer, T., & Cohen, Y. (2017). From agricultural use of sewage sludge to nutrient extraction: A soil science outlook. Ambio, 46(2), 143–154. https://doi.org/10.1007/s13280-016-0816-3.
Kirk, J. L., Klironomos, J. N., Lee, H., & Trevors, J. T. (2005). The effects of perennial ryegrass and alfalfa on microbial abundance and diversity in petroleum contaminated soil. Environmental Pollution, 133(3), 455–465. https://doi.org/10.1016/j.envpol.2004.06.002.
Kong, W., Zhu, Y., Liang, Y., Zhang, J., Smith, F., & Yang, M. (2007). Uptake of oxytetracycline and its phytotoxicity to alfalfa (Medicago sativa L.). Environmental Pollution, 147(1), 187–193. https://doi.org/10.1016/j.envpol.2006.08.016.
Kumar, S., Singh, R., Kumar, V., Rani, A., & Jain, R. (2017). Cannabis sativa: A plant suitable for phytoremediation and bioenergy production. In Phytoremediation potential of bioenergy plants. Singapore: Springer. https://doi.org/10.1007/978-981-10-3084-0_10.
Latif, E., Wijeyesekera, D. C., Newport, D., & Tucker, S. (2010). Potential for research on hemp insulation in the UK construction sector. In Proceedings of Advances in Computing and Technology,(AC&T) The School of Computing and Technology 5th Annual Conference, University of East London, pp, 2010
Li, S. Y., Stuart, J. D., Li, Y., & Parnas, R. S. (2010). The feasibility of converting Cannabis sativa L. oil into biodiesel. Bioresource Technology, 101(21), 8457–8460. https://doi.org/10.1016/j.biortech.2010.05.064.
Lieder, M., & Rashid, A. (2016). Towards circular economy implementation: A comprehensive review in context of manufacturing industry. Journal of Cleaner Production, 115, 36–51. https://doi.org/10.1016/j.jclepro.2015.12.042.
Lin, H., Tao, S., Zuo, Q., & Coveney, R. (2007). Uptake of polycyclic aromatic hydrocarbons by maize plants. Environmental Pollution, 148(2), 614–619. https://doi.org/10.1016/j.envpol.2006.11.026.
Linger, P., Müssig, J., Fischer, H., & Kobert, J. (2002). Industrial hemp (Cannabis sativa L.) growing on heavy metal contaminated soil: Fibre quality and phytoremediation potential. Industrial Crops and Products, 16(1), 33–42. https://doi.org/10.1016/S0926-6690(02)00005-5.
Liste, H.-H., & Prutz, I. (2006). Plant performance, dioxygenase-expressing rhizosphere bacteria, and biodegradation of weathered hydrocarbons in contaminated soil. Chemosphere, 62(9), 1411–1420. https://doi.org/10.1016/j.chemosphere.2005.05.018.
Liu, C. F., Li, Y. H., & Shi, G. R. (2012). Utilize heavy metal-contaminated farmland to develop bioenergy. Advanced Materials Research, 414, 254–261. https://doi.org/10.4028/www.scientific.net/AMR.414.254.
Macherius, A., Eggen, T., Lorenz, W., Moeder, M., Ondruschka, J., & Reemtsma, T. (2012). Metabolization of the bacteriostatic agent triclosan in edible plants and its consequences for plant uptake assessment. Environmental Science & Technology, 46(19), 10797–10804. https://doi.org/10.1021/es3028378.
Madalão, J. C., Pires, F. R., Cargnelutti Filho, A., Nascimento, A. F., Chagas, K., Procópio, S. O., et al. (2012). Selection of species tolerant to the herbicide sulfentrazone with potential for phytoremediation of contaminated soils. Semina: Ciências Agrárias, 33(6), 2199–2213. https://doi.org/10.5433/1679-0359.2012v33n6p2199.
Manriquez-Altamirano, A., Sierra-Perez, J., Munoz, P., & Gabarrell, X. (2020). Analysis of urban agriculture solid waste in the frame of circular economy: Case study of tomato crop in integrated rooftop greenhouse. Science of the Total Environment. https://doi.org/10.1016/j.scitotenv.2020.139375.
Marsik, P., Rezek, J., Židková, M., Kramulová, B., Tauchen, J., & Vaněk, T. (2017). Non-steroidal anti-inflammatory drugs in the watercourses of Elbe basin in Czech Republic. Chemosphere, 171, 97–105. https://doi.org/10.1016/j.chemosphere.2016.12.055.
Martin, M., Svensson, N., Fonseca, J., & Eklund, M. (2014). Quantifying the environmental performance of integrated bioethanol and biogas production. Renewable Energy, 61, 109–116. https://doi.org/10.1016/j.renene.2012.09.058.
Mattes, T. E., Ewald, J. M., Liang, Y., Martinez, A., Awad, A., Richards, P., et al. (2018). PCB dechlorination hotspots and reductive dehalogenase genes in sediments from a contaminated wastewater lagoon. Environmental Science and Pollution Research, 25(17), 16376–16388. https://doi.org/10.1007/s11356-017-9872-x.
Meers, E., Ruttens, A., Hopgood, M., Lesage, E., & Tack, F. M. G. (2005). Potential of Brassic rapa, Cannabis sativa, Helianthus annuus and Zea mays for phytoextraction of heavy metals from calcareous dredged sediment derived soils. Chemosphere, 61(4), 561–572. https://doi.org/10.1016/j.chemosphere.2005.02.026.
Meher, L. C., Sagar, D. V., & Naik, S. N. (2006). Technical aspects of biodiesel production by transesterification—a review. Renewable & Sustainable Energy Reviews, 10(3), 248–268. https://doi.org/10.1016/j.rser.2004.09.002.
Mihoc, M., Pop, G., Alexa, E., & Radulov, I. (2012). Nutritive quality of romanian hemp varieties (Cannabis sativa L.) with special focus on oil and metal contents of seeds. Chemistry Central Journal, 6(1), 122. https://doi.org/10.1186/1752-153X-6-122.
Mordechay, B. E., Tarchitzky, J., Chen, Y., Shenker, M., & Chefetz, B. (2018). Composted biosolids and treated wastewater as sources of pharmaceuticals and personal care products for plant uptake: A case study with carbamazepine. Environmental Pollution, 232, 164–172. https://doi.org/10.1016/j.envpol.2017.09.029.
Naithani, V., Tyagi, P., Jameel, H., Lucia, L. A., & Pal, L. (2020). Ecofriendly and innovative processing of hemp hurds fibers for tissue and towel paper. BioResources, 15(1), 706–720. https://doi.org/10.15376/biores.15.1.706-720.
Nebert, D. W., Wikvall, K., & Miller, W. L. (2013). Human cytochromes P450 in health and disease. Philosophical transactions of the Royal Society of London. Series B, Biological Sciences, 368(1612), 20120431–20120431. https://doi.org/10.1098/rstb.2012.0431.
Panchenko, L., Muratova, A., & Turkovskaya, O. (2017). Comparison of the phytoremediation potentials of Medicago falcata L. and Medicago sativa L. in aged oil-sludge-contaminated soil. Environmental Science and Pollution Research, 24(3), 3117–3130. https://doi.org/10.1007/s11356-016-8025-y.
Pavela, R. (2018). Essential oils from Foeniculum vulgare Miller as a safe environmental insecticide against the aphid Myzus persicae Sulzer. Environmental Science and Pollution Research, 25(11), 10904–10910. https://doi.org/10.1007/s11356-018-1398-3.
Pejic, B. M., Vukcevic, M. M., Pajic-Lijakovic, I. D., Lausevic, M. D., & Kostic, M. M. (2011). Mathematical modeling of heavy metal ions (Cd2+, Zn2+ and Pb2+) biosorption by chemically modified short hemp fibers. Chemical Engineering Journal, 172(1), 354–360. https://doi.org/10.1016/j.cej.2011.06.016.
Peng, W., & Pivato, A. (2019). Sustainable management of digestate from the organic fraction of municipal solid waste and food waste under the concepts of back to earth alternatives and circular economy. Waste and Biomass Valorization, 10(2), 465–481. https://doi.org/10.1007/s12649-017-0071-2.
Phillips, P. J., Smith, S. G., Kolpin, D. W., Zaugg, S. D., Buxton, H. T., Furlong, E. T., et al. (2010). Pharmaceutical formulation facilities as sources of opioids and other pharmaceuticals to wastewater treatment plant effluents. Environmental Science & Technology, 44(13), 4910–4916. https://doi.org/10.1021/es100356f.
Pietrini, F., Passatore, L., Patti, V., Francocci, F., Giovannozzi, A., & Zacchini, M. (2019). Morpho-physiological and metal accumulation responses of hemp plants (Cannabis Sativa L.) grown on soil from an agro-industrial contaminated area. Water, 11(4), 808.
Prade, T., Svensson, S.-E., & Mattsson, J. E. (2012). Energy balances for biogas and solid biofuel production from industrial hemp. Biomass and Bioenergy, 40, 36–52. https://doi.org/10.1016/j.biombioe.2012.01.045.
Praspaliauskas, M., Žaltauskaitė, J., Pedišius, N., & Striūgas, N. (2020). Comprehensive evaluation of sewage sludge and sewage sludge char soil amendment impact on the industrial hemp growth performance and heavy metal accumulation. Industrial Crops and Products, 150, 112396. https://doi.org/10.1016/j.indcrop.2020.112396.
Rosi-Marshall, E. J., Snow, D., Bartelt-Hunt, S. L., Paspalof, A., & Tank, J. L. (2015). A review of ecological effects and environmental fate of illicit drugs in aquatic ecosystems. Journal of Hazardous Materials, 282, 18–25. https://doi.org/10.1016/j.jhazmat.2014.06.062.
Salentijn, E. M. J., Zhang, Q., Amaducci, S., Yang, M., & Trindade, L. M. (2015). New developments in fiber hemp (Cannabis sativa L.) breeding. Industrial Crops and Products, 68, 32–41. https://doi.org/10.1016/j.indcrop.2014.08.011.
Sauve, S., & Desrosiers, M. (2014). A review of what is an emerging contaminant. Chemistry Central Journal. https://doi.org/10.1186/1752-153x-8-15.
Schettini, E., Santagata, G., Malinconico, M., Immirzi, B., Scarascia Mugnozza, G., & Vox, G. (2013). Recycled wastes of tomato and hemp fibres for biodegradable pots: Physico-chemical characterization and field performance. Resources, Conservation and Recycling, 70, 9–19. https://doi.org/10.1016/j.resconrec.2012.11.002.
Scrucca, F., Ingrao, C., Maalouf, C., Moussa, T., Polidori, G., Messineo, A., et al. (2020). Energy and carbon footprint assessment of production of hemp hurds for application in buildings. Environmental Impact Assessment Review,. https://doi.org/10.1016/j.eiar.2020.106417.
Shea, A., Lawrence, M., & Walker, P. (2012). Hygrothermal performance of an experimental hemp–lime building. Construction and Building Materials, 36, 270–275. https://doi.org/10.1016/j.conbuildmat.2012.04.123.
Sjöström, A. E., Collins, C. D., Smith, S. R., & Shaw, G. (2008). Degradation and plant uptake of nonylphenol (NP) and nonylphenol-12-ethoxylate (NP12EO) in four contrasting agricultural soils. Environmental Pollution, 156(3), 1284–1289. https://doi.org/10.1016/j.envpol.2008.03.005.
Soares, A., Guieysse, B., Jefferson, B., Cartmell, E., & Lester, J. N. (2008). Nonylphenol in the environment: A critical review on occurrence, fate, toxicity and treatment in wastewaters. Environment International, 34(7), 1033–1049. https://doi.org/10.1016/j.envint.2008.01.004.
Staples, M. D., Malina, R., & Barrett, S. R. H. (2017). The limits of bioenergy for mitigating global life-cycle greenhouse gas emissions from fossil fuels. Nature Energy, 2(2), 16202. https://doi.org/10.1038/nenergy.2016.202.
Stonehouse, G. C., McCarron, B. J., Guignardi, Z. S., El Mehdawi, A. F., Lima, L. W., Fakra, S. C., et al. (2020). Selenium metabolism in hemp (Cannabis sativa L.)—potential for phytoremediation and biofortification. Environmental Science & Technology, 54(7), 4221–4230. https://doi.org/10.1021/acs.est.9b07747.
Tai, Y., Tam, N.F.-Y., Ruan, W., Yang, Y., Yang, Y., Tao, R., et al. (2019). Specific metabolism related to sulfonamide tolerance and uptake in wetland plants. Chemosphere, 227, 496–504. https://doi.org/10.1016/j.chemosphere.2019.04.069.
Tanoue, R., Sato, Y., Motoyama, M., Nakagawa, S., Shinohara, R., & Nomiyama, K. (2012). Plant uptake of pharmaceutical chemicals detected in recycled organic manure and reclaimed wastewater. Journal of Agricultural and Food Chemistry, 60(41), 10203–10211. https://doi.org/10.1021/jf303142t.
Tofan, L., Teodosiu, C., Paduraru, C., & Wenkert, R. (2013). Cobalt (II) removal from aqueous solutions by natural hemp fibers: Batch and fixed-bed column studies. Applied Surface Science, 285, 33–39. https://doi.org/10.1016/j.apsusc.2013.06.151.
Trapp, S. (2004). Plant uptake and transport models for neutral and ionic chemicals. Environmental Science and Pollution Research, 11(1), 33. https://doi.org/10.1065/espr2003.08.169.
Yan, D., Ma, W., Song, X., & Bao, Y. (2017). The effect of iron plaque on uptake and translocation of norfloxacin in rice seedlings grown in paddy soil. Environmental Science and Pollution Research, 24(8), 7544–7554. https://doi.org/10.1007/s11356-017-8368-z.
Yang, R., Su, M. X., Li, M., Zhang, J. C., Hao, X. M., & Zhang, H. (2010). One-pot process combining transesterification and selective hydrogenation for biodiesel production from starting material of high degree of unsaturation. Bioresource Technology, 101(24), 9829–9829. https://doi.org/10.1016/j.biortech.2010.02.095.
Funding
This work is supported by the USDA National Institute of Food and Agriculture through grant No. 2020-38422-32253 to California State Polytechnic University Pomona (Cal Poly Pomona). This work is also supported by the USDA National Institute of Food and Agriculture through grant No. 2018-68002-27920 to Florida A&M University and the National Science Foundation through grant No. 1735235 as part of the National Science Foundation Research Traineeship.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest in relation to this work.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wu, Y., Trejo, H.X., Chen, G. et al. Phytoremediation of contaminants of emerging concern from soil with industrial hemp (Cannabis sativa L.): a review. Environ Dev Sustain 23, 14405–14435 (2021). https://doi.org/10.1007/s10668-021-01289-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10668-021-01289-0