Abstract
Activated hazelnut shell (HSAC), an organic waste, was utilized for the adsorptive removal of Congo red (CR) dye from aqueous solutions, and a modelling study was conducted using artificial neural networks (ANNs). The structure and characteristic functional groups of the material were examined by the FTIR method. The BET surface area of the synthesized material, named HSAC, was 812 m2/g. Conducted in a batch system, the adsorption experiments resulted in a notable removal efficiency of 87% under optimal conditions. The kinetic data for hazelnut shell activated carbon (HSAC) removal of CR were most accurately represented by the pseudo-second-order kinetic model (R2 = 0.998). Furthermore, the equilibrium data demonstrated a strong agreement with the Freundlich model. The maximum adsorption capacity of HSAC for CR was determined to be 34.8 mg/g. The optimum adsorption parameters were determined to be pH 6, contact time of 60 min, 10 g/L of HSAC, and a concentration of 400 mg/L for CR. Considering the various experimental parameters influencing CR adsorption, an artificial neural network (ANN) model was constructed. The analysis of the ANN model revealed a correlation of 98%, indicating that the output parameter could be reliably predicted. Thus, it was concluded that ANN could be employed for the removal of CR from water using HSAC.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Reactive dyes constitute one of the most extensive categories of organic chemicals utilized across various industries including leather, tannery, textiles, cosmetics, food, printing, and plastics (Balarak et al., 2020). The textile industry is characterized by its substantial water consumption and the use of various dyes to impart colour to its products (Das et al., 2023). This process results in the generation of a substantial volume of coloured wastewater. The direct discharge of this wastewater into natural water bodies such as rivers and streams has far-reaching environmental consequences. These include a high organic load, the introduction of toxic substances, colour pollution, diminished light penetration, and a detrimental impact on aquatic ecosystems due to its effect on photosynthesis (Chen et al., 2018). Furthermore, it should be noted that many dyes used in this industry exhibit either toxic, mutagenic, or carcinogenic properties (Patel et al., 2022). Long-term exposure to dye pollutants may lead to various adverse health effects in humans, such as eye irritation, dermatitis, and allergic diseases (Al-Musawi et al., 2021). The treatment of dye-containing wastewater poses a significant challenge due to the varied synthetic origins and complex aromatic structures of these dyes. Moreover, they exhibit resistance to biological degradation. One notable example is Congo red (CR), an anionic diazo dye commonly used for colouring materials like paper, cotton, hemp, and silk, which has been associated with allergic reactions (Patel & Vashi, 2012; Rendon-Castrillon et al., 2023).
Even at low concentrations, the presence of residual dyes in wastewater not only results in the undesirable production of visible colours but also exerts adverse effects on the health of both aquatic organisms and human populations when this wastewater is discharged into water bodies (Balarak et al., 2019; Almroth et al., 2021; Sudarshan et al., 2023). Numerous strategies have been applied to address the challenge of dye removal from wastewater. Photodegradation (Sun et al., 2009), coagulation and flocculation (Hassaan et al., 2017), ozonation (Khadhraoui et al., 2009), and adsorption (Munagapati & Kim, 2017; Zhang et al., 2015) represent diverse methods that have been investigated and implemented for the elimination of CR from wastewater (Balarak et al., 2021; Nguyen et al., 2021). Among these approaches, adsorption stands out as a particularly prominent method, garnering extensive research attention due to the abundance and versatility of raw materials and the notable efficiency of this technique (Ali et al., 2018). Recent investigations have emphasized that adsorption is a particularly promising technology for treating wastewater contaminated with dyes (Balarak et al., 2020; Jorge et al., 2023). Its appeal lies in its simplicity, cost-effectiveness, and the wide array of adsorbents available for use. Activated carbon–based adsorption, while highly effective, introduces challenges related to cost and resource availability. Therefore, there has been a shift towards seeking effective, economically viable, and readily available biosorbents as alternatives to activated carbon (Bhatia et al., 2017; Katheresan et al., 2018).
Consequently, several investigations have been undertaken to examine the efficacy of economical adsorbents in eliminating dyes from aqueous solutions. Agricultural waste or by-products have surfaced as viable contenders to mitigate production expenses (Singha et al., 2015). These materials are renewable, are easily obtainable, and enable the elimination of energy costs associated with thermal processes. In this context, hazelnut shell–based activated carbon (HSAC) has been prepared through activation with nitrogen gas in an oxygen-free environment to investigate its potential for CR removal. Batch adsorption experiments have been carried out to systematically analyze the impact of parameters such as contact time, pH, adsorbent dosage, CR concentration, and temperature on the adsorption of CR by HSAC.
Artificial neural networks (ANNs) represent advanced computational architectures specifically designed to replicate the cognitive capacities of the human mind, including learning, discovery, and the generation of knowledge. These networks find utility in scenarios where conventional computational paradigms prove intricate or unfeasible. ANNs constitute a specialized subfield of computer science dedicated to adaptive data processing and have demonstrated their applicability across diverse domains, spanning from engineering to healthcare and textiles. They are particularly favored for tasks involving prediction, diagnosis, interpretation, classification, and recognition (Souza et al., 2018; Yusuf et al., 2020).
In recent years, there has been a growing interest in employing artificial neural networks (ANN) for modelling the adsorption of organic dyes (Hasanzadeh et al., 2019; Dahlan et al., 2023; Ramesh et al., 2023). ANNs offer a powerful tool to capture intricate relationships in data, enabling more precise predictions of adsorption behaviours. Their flexibility allows the incorporation of various parameters, contributing to a comprehensive understanding of the adsorption process. This integration of ANN with experimental data holds promise for optimizing adsorption conditions and advancing wastewater treatment efficiency.
This study presents a novel activation approach for hazelnut shells to enhance their effectiveness as adsorptive agents for CR dye removal. Unlike previous works, which primarily focused on hazelnut shell waste, our research investigates the activation process to optimize adsorption capacity. Additionally, we employ artificial neural networks (ANNs) to predict the adsorption process, distinguishing our study from others. Furthermore, our findings contribute to a deeper understanding of adsorption kinetics and mechanisms, offering insights into effective wastewater treatment strategies. Overall, our study highlights the potential of hazelnut shell waste as a sustainable adsorbent and introduces an innovative approach to predictive modelling in adsorption research.
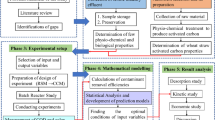
Materials and method
Adsorbent preparation
Before utilizing hazelnut shells as an adsorbent, they underwent a thorough cleaning process involving multiple washes with distilled water to eliminate surface contaminants. A portion of the cleaned hazelnut shells was immersed in 95% hexane for a duration of 2 h, using hexane of analytical grade from Merck. Subsequently, all hazelnut shells were subjected to drying in an oven at temperatures between 103 and 105 °C and then reduced to a finer powder by crushing them in a mortar. Further refinement to achieve a particle size of ≤ 0.30 mm was accomplished using a mechanical grinder. These finely powdered hazelnut shells were employed as adsorbents in both their natural state and following purification with hexane. Following this stage, an oxygen-free environment was established within an oven, and the hazelnut shells were activated at 800 °C for 90 min with nitrogen gas.
The surface area of the hazelnut shell has been increased through thermal modification. Consequently, the augmented porous structure has led to an increase in the active sites necessary for adsorption.
Subsequently, the adsorbent, referred to as HSAC (C6767 from Sigma-Aldrich), was collected in a sealed container for future use. The chemical structures of the CR dye are illustrated in Fig. 1.
Adsorption studies
The adsorption study was conducted using a thermostatic shaker (Nüve SL350) with a 1 L solution volume and a shaking speed of 150 rpm. In the investigation of the effect of contact time on the removal of CR, adsorption analyses were carried out at different contact times ranging from 2 to 240 min. When examining the impact of the adsorbent quantity on CR removal, varying amounts of HSAC ranging from 0.5 to 10 g/L were employed. To evaluate the influence of pH on CR adsorption, experiments were conducted using pH values within the range of 3 to 9. For pH adjustments, HCl (ACS reagent, 37%) and NaOH (ACS reagent, ≥ 97.0%, pellets) were procured from Sigma-Aldrich. Adsorption studies were also performed to determine the effect of the initial concentration of CR dye on removal efficiency, utilizing seven different initial dye concentrations ranging from 5 to 400 mg/L. Temperature’s effect on CR removal efficiency was investigated by conducting analyses at temperatures between 20 and 40 °C. In the studies on the effects of adsorbent quantity, pH, contact time, temperature, and initial CR concentration, samples were centrifuged at 3000 rpm for 4 min (using a Nüve CN180 centrifuge) after collection from the solutions. Subsequently, measurements were carried out using a UV-Vis spectrophotometer. Based on the data obtained from the experiments, Ce (mg/L), qe (mg/g), and percentage removal rates were calculated according to Eqs. (1) and (2).
C 0 and Ce (mg/L) are respectively the initial and final concentrations of CR. m (g) represents the mass of AAS. The volume of the solution is indicated with V (L), where qe (mg/g) is the amount of adsorbed CR ions by AAS.
Isotherm and kinetic study
The Langmuir adsorption isotherm (Langmuir, 1918) is specifically applicable to the monolayer adsorption of a solute from a solution onto a surface containing a defined number of identical sites. This model, based on the assumption of similarity in energy of the adsorbent surface, is utilized to describe monolayer homogeneous adsorption and enables the estimation of maximum adsorption capacity (Kostić et al., 2016). The model rests on fundamental assumptions: (i) adsorption transpires within distinct homogeneous regions within the adsorbent, (ii) a single dye molecule associates with a sole binding site, (iii) the adsorbent possesses a finite capacity for the pollutant at equilibrium, and (iv) all regions exhibit uniformity and energetic equivalence. Conversely, the Freundlich isotherm model (Freundlich, 1906) postulates a multi-layered manifestation of adsorption and acknowledges the heterogeneity of the adsorptive surface in terms of both adsorption sites and energy. The Freundlich model is an empirical equation used to determine the adsorption intensity that may occur on the adsorbent surface (Kaykıoğlu, 2016). In contrast, the Temkin isotherm model (El-Shafie, 2023) proposes a linear reduction in adsorption heat with the adsorbed molecules in the environment, indicative of a homogeneous binding energy. The Dubinin-Radushkevich (D-R) isotherm (Laskar & Hashisho, 2020) is rooted in the potential change theory, specifically applied to a heterogeneous surface. Adsorption kinetics is utilized to specify the type of mechanism involved during the adsorption of pollutants onto the surface of the adsorbent. This study provides information about the potential adsorption mechanism and the ultimate interaction between the pollutant and adsorbent, assisting in the development of appropriate mathematical models to characterize these interactions. For this purpose, the pseudo-first-order kinetic model, pseudo-second-order kinetic model, and intra-particle diffusion model have been utilized in the adsorption process (Ho et al., 2000; Ho & McKay, 1999; Kostić et al., 2017; Lagergren, 1898). Isotherm and kinetic equations along with their corresponding parameters are provided in Table 1.
Relative deviation in adsorption capacity, RD (%), and mean relative deviation, MRD (%), were calculated using Eqs. (3) and (4), respectively (Kostić et al., 2022).
where i is the experimental point and n is the number of experimental points.
ANN structure
ANN model exhibits an inherent proficiency in discerning and internalizing latent patterns that underlie complex processes by virtue of analyzing preceding datasets (Tanzifi et al., 2018). This assimilated knowledge subsequently extends itself to encompass the intricate mathematical relationships governing the dynamic interplay between input and output data. The ANN model, contingent upon its specific architectural instantiation, evinces the capacity to prognosticate intricate systems of diverse typologies, a phenomenon that finds articulation within the conceptual confines of its design (Mendoza-Castillo et al., 2018). Considering the intricate and multifarious attributes characterizing adsorption processes, an ANN model imbued with computational intelligence offers a heightened degree of adaptability vis-à-vis its conventional statistical counterparts. This is particularly salient when considering the latter’s limited efficacy in modelling intricate datasets imbued with nonlinear characteristics. Demonstrating its aptitude for sophisticated modelling endeavours, the ANN model instantiated through a hierarchical composition encompassing input, hidden, and output layers—individually characterized as neurons—constitutes a representation endowed with the acumen to anticipate the intricate interrelationships underpinning the input-output continuum (Pauletto et al., 2020). The visual exposé of this construct is vividly portrayed in Fig. 2, thereby underscoring its prospective utility in elucidating and prognosticating the interrelationships.
The entirety of the dataset has been meticulously partitioned into two distinct categories: a learning subset, encompassing 70% of the data, and two validation and testing subsets, each embracing 15% of the data volume.
The learning subset stands as the crucible for the training of the artificial neural network (ANN) model, while the testing subset furnishes the means for meticulously appraising the prognostic capabilities exhibited by the ANN model.
The quantitative evaluation of the ANN model’s prognostic outputs is judiciously undertaken by recourse to two pivotal performance metrics: the mean square error (MSE) and the coefficient of determination (R2). The efficacy and potency of the ANN model are profoundly interwoven with a constellation of pivotal variables. Among them, the count of neurons populating both the concealed and terminal strata commands significance, while the intrinsic attributes of the transfer function exert a pronounced bearing. Notably, in the context of this scholarly inquiry, the computational implementation of the ANN protocol is executed leveraging the MATLAB version R2023a Toolbox.
The architectural composition of the adopted ANN structure adheres to a tripartite arrangement, distinguished by a tangent sigmoid transfer function (tansig) adorning the hidden layer, a linear transfer function (purelin) adorning the output layer, all galvanized by the Levenberg-Marquardt backpropagation algorithm (trainLM), iterated across a course of 1000 cycles. The ingress layer, with discerning acumen, accommodates three constituent nodes that emblemize the elemental factors underpinning the adsorption process. These encompass the initial concentration, reaction time, and the quantum of adsorbent dosage. In the hidden layer, the neural ensemble varies across a span of 1 to 20, while the egress layer is graced by a solitary neuron, constituting the ultimate arbiter of the prognosticative endeavour.
Results and discussions
Characterization of the adsorbent
The FTIR spectra corresponding to HSAC are presented in Fig. 3. Within the hazelnut shell spectra, discernible spectral bands have been identified. Notably, a band appearing at 3360 cm−1 is attributed to the hydroxyl (–OH) group, while a prominent band observed at 3020 cm−1 indicates C–H stretching vibrations, suggesting the presence of constituents such as pectin, hemicellulose, cellulose, and lignin. The band observed at approximately 2700 cm−1 can be assigned to the aliphatic C–H group. A significant feature at 1670 cm−1 originates from C=C stretching, plausibly corresponding to the aromatic C–C bond. The presence of an aromatic ring within lignin is notably evident through bands occurring at 1460 cm−1. In the spectral range spanning from 1360 cm−1, the observed band likely corresponds to manifestations of O–H bending, O–H stretching, or C–H bending vibrations. The distinct C–O stretching vibration of carboxylic acids becomes perceptible at 1080 cm−1. It is remarkable to mention that carboxylic groups C–O have an absorbance in the region of 1650–1900 cm−1, while the bands of 400–650 cm−1 propose the formation of dye coupled with oxygen. These functional groups serve as indicative features within agricultural waste substrates. The surface area of the hazelnut shell, which served as the adsorbent material, was determined through BET analysis. Similarly, the surface analysis of the HSAC following the activation process was conducted using BET analysis. The BET-specific surface area of unmodified hazelnut shells and HSACs was 367 m2/g and 812 m2/g.
Effect of operating parameters on CR removal
In the adsorption study, parameters such as pH, adsorbent quantity, initial CR concentration, and contact time were investigated. Detailed results for each parameter are provided in Fig. 4. The adsorption behaviour of the examined systems is influenced by pH, with variations in adsorption capacity occurring because of the alteration in the ionization state of binding groups in both the adsorbate and adsorbent, as previously discussed. Consequently, it becomes imperative to identify the optimal pH for CR adsorption onto HSAC. To address this, experiments were conducted across a pH range of 3 to 9, incorporating different initial dye concentrations, solid/liquid ratios ranging from 0.5 to 10 g/L, and a temperature of 25 °C. The quantity of adsorbent is instrumental in elucidating the removal capacity within the adsorption mechanism. In this context, the amount of HSAC was incrementally varied from 0.5 to 10 g while maintaining a constant pH and a temperature of 25 °C. The removal efficiency of CR increased up to 87%. As the adsorbent quantity was increased, the efficiency of CR removal also increased naturally due to the increase in binding sites. The removal efficiency initially increases as the adsorbent quantity is raised from 0.5 to 10 g, reaching a balance thereafter. However, the adsorption capacity decreases. This is attributed to the lower dose of adsorbent having relatively more available reaction and binding sites on the surface area. In the study, the removal rate of CR occurs rapidly within the first 30 min of contact time. Subsequently, a slow increase leads to the attainment of equilibrium in approximately 60 min. The presence of a greater active surface area of the adsorbent in the initial minutes of adsorption has ensured a high removal efficiency of CR. With CR initial concentrations ranging from 5 to 400 mg/L, a 98% efficiency was achieved within the first 60 min.
Upon comprehensive examination of all results, the optimal adsorption parameters for CR adsorption with HSAC are determined as follows: a contact time of 60 min, HSAC quantity of 10 g/L, initial CR concentration of 400 mg/L, and a pH of 6. Additionally, the adsorption mechanism is conducted at a temperature of 25 °C, in a solution volume of 1 L, and with agitation at a speed of 150 rpm.
The adsorption process is subject to numerous influencing factors, among which the pHpzc (point of zero charge) plays a pivotal role in elucidating the mechanism of dye adsorption on the designated adsorbent. Understanding the pHpzc of the adsorbent is crucial for elucidating the electrostatic interactions between the adsorbent surface and the adsorbate at a specific pH level. Theoretically, when the pH is below the pHpzc, the adsorbent surface is positively charged, creating favourable conditions for the adsorption of anionic dyes like CR. According to these findings, effective removal of the anionic CR dye can be achieved by maintaining the solution pH below 6.5 (Fig. 5). This aligns perfectly with the experiment’s optimal pH result of 6, resulting in an 87% removal efficiency. Upon literature review, it is supported that in the study conducted by Ribeiro et al. (2018) (who used wood sawdust powder from eucalyptus Corymbia citriodora), CR adsorption is augmented with increasing removal percentages at low pH values. Additionally, Khan et al. (2014), who utilized chir pine sawdust, and Chanzu et al. (2019), who employed brewers’ spent grains as adsorbents, achieved maximum removal efficiency at low pH values in CR adsorption.
Isotherm and kinetic study
Equilibrium data, often referred to as adsorption isotherms, play a pivotal role in characterizing and designing adsorption systems. These data establish a mathematical relationship between the quantity of adsorbate per unit of adsorbent at equilibrium and the residual amount in the solution. Isothermal investigations were conducted by subjecting CR dye solutions with initial concentrations ranging from 5 to 400 mg/L to a constant temperature of 293 K for a duration of 30 min. Table 2 provides the constants of Langmuir, Freundlich, Temkin, and D-R isotherm models.
The examination of the data presented in Table 2 indicates that the regression coefficients (R2 values) obtained for the Freundlich and Langmuir isotherm models are in close proximity to 1. It is noteworthy that the Langmuir equation is based on the assumption that the adsorption process occurs on homogeneous surfaces within a single layer. The Langmuir adsorption model is commonly employed to characterize monolayer adsorption on homogeneous surfaces where there is no significant attraction between binding ions. The calculated Langmuir constant (RL) value for the adsorption on hazelnut shell activated carbon (HSAC) was 0.1322 for CR. The RL offers insights into the sorbent’s affinity for binding ions. This determined value affirms the occurrence of favourable adsorption concerning the reduction of dye ions by the organic adsorbents (RD was − 2.64%, and MRD was 1.04%).
Kinetic parameters play a pivotal role in deciphering the adsorption rate, thereby furnishing crucial insights for the modelling of the adsorption process. Consequently, in this context, three distinct kinetic models, specifically the pseudo-first-order, pseudo-second-order, and intra-particle diffusion models, were employed to comprehensively scrutinize the experimental data. Table 3 presents the pseudo-first-order, pseudo-second-order, and intra-particle diffusion models for the kinetic processes of adsorption, along with their respective parameter values. The correlation coefficients obtained for the pseudo-first-order and intra-particle diffusion models are compared, revealing a good fit to the pseudo-second-order model with higher R2 = 0.998 and lower values of RD and MRD. Similar kinetic results to this study have also been reported in the literature (Altintig et al., 2023; Kostić et al., 2013; Zabihi et al., 2022).
The results of the literature review conducted for the adsorption of CR dye are presented in Table 4. HSAC provides higher removal compared to other adsorbents.
ANN modelling
ANNs are employed to predict the behaviour and outcomes of systems, as well as to forecast existing processes. In this study, initial CR concentration, pH, contact time, and HSAC quantity are introduced as input parameters to the system. The removal efficiency of the adsorption mechanism is designated as the output parameter. An ANN network structure is constructed using these input and output parameters. The performance of the network is assessed using the mean squared error (MSE) and the regression analysis coefficient (R2). ANNs exhibit a notable sensitivity to the quantity of neurons in the hidden layer of the model. An excessive number of neurons may induce overfitting, while an insufficient number may lead to underfitting in the model system (Badkar et al., 2013). The study’s findings, aimed at identifying the optimal number of neurons for integration into the ANN model, are illustrated in Table 5. According to these results, the network structure with 8 neurons achieves the lowest MSE and the highest R2 values. The network (4-8-1) is trained using the Levenberg-Marquardt backpropagation algorithm (trainlm). The primary reasons for choosing this algorithm include its suitability for large datasets and its relatively faster training time.
In neural network studies, to achieve the best learning and closest results to reality, the constructed network structure iteratively operates consecutively. In each iteration, the network provides mean squared error and correlation coefficient. When the model yielding the lowest mean squared error and the highest correlation coefficient is obtained, the network operation is halted, and the network is saved. In this study, the regression results that yield the lowest MSE and the highest correlation coefficient for the network are presented in Fig. 6. These high correlation values obtained in the created ANN structure show that they best describe the relationship between input and output parameters, which consist of data of the adsorption mechanism.
As seen in Fig. 6, a correlation coefficient of 0.984 was obtained for both the training and test data, indicating a strong correlation. For the validation data, a correlation coefficient of 1 shows a perfect fit. Despite the limited amount of data used for model training, the reliable agreement between the model’s predictions and the unseen data suggests the credibility of the model’s predictions. According to the results of the ANN, the correlation coefficient values indicate a good fit of the output. This result demonstrates that the ANN model is sufficiently accurate in predicting the actual values. Additionally, the ANN prediction data generated has been thoroughly compared with the experimental data in Fig. 7. Moreover, these values are like the correlation results of many other studies that have used and succeeded with ANN (Dolatabadi et al., 2018; Kaya et al., 2022; Al-Musawi et al., 2023; Ilavenil et al., 2024).
Conclusions
In this study, an artificial neural network (ANN) model structure has been developed to predict the removal efficiency of the CR dye using HSAC. The obtained results demonstrate that ANN emerges as a promising predictive technique, exhibiting a high level of effectiveness and satisfactory accuracy in forecasting the removal of CR from aqueous solutions. Functional groups of HSAC were identified through FTIR analysis, confirming the binding of the CR dye onto the adsorbent after adsorption. The results revealed a maximum removal efficiency of 87% under optimized conditions (pH 6, HSAC dosage 10 g/L, CR concentration 400 mg/L, and adsorption time 60 min). A kinetic study demonstrated a good fit of the experimental data to a pseudo-second-order model. Furthermore, isothermal data were consistent with both Freundlich and Langmuir models. The maximum adsorption capacity of HSAC for CR was determined to be 34.8 mg/g. This finding suggests that activated hazelnut shells, as a natural waste material, can be effectively utilized for dye removal. The key finding of this study is the high applicability of ANN in modelling the CR adsorption process, as evidenced by a strong correlation between the results of ANN analyses and the experimental values of the CR adsorption process.
Data availability
No datasets were generated or analysed during the current study.
References
Ali, A., Mannan, A., Hussain, I., Hussain, I., & Zia, M. (2018). Effective removal of metal ions from aqueous solution by silver and zinc nanoparticles functionalized cellulose: Isotherm, kinetics and statistical supposition of process. Environmental Nanotechnology, Monitoring & Management, 9, 1–11. https://doi.org/10.1016/j.enmm.2017.11.003
Almroth, B. C., Cartine, J., Jonander, C., Karlsson, M., Langlois, J., Lindstrom, M., Lundin, J., Melander, N., Pesqueda, A., Rahmqvist, I., Renaux, J., Roos, J., Spilsbury, F., Svalin, J., Vestlund, H., Zhao, L., Aske, N., Asmonaite, G., Birgersson, L., et al. (2021). Assessing the effects of textile leachates in fish using multiple testing methods: From gene expression to behavior. Ecotoxicolgy and Environmental Safety, 207, 111523. https://doi.org/10.1016/j.ecoenv.2020.111523
Al-Musawi, T. J., Arghavan, S. M. A., Allahyari, E., Arghavan, F. S., Othmani, A., & Nasseh, N. (2023). Adsorption of malachite green dye onto almond peel waste: A study focusing on application of the ANN approach for optimization of the effect of environmental parameters. Biomass Conversion and Biorefinery, 13, 12073–12084. https://doi.org/10.1007/s13399-021-02174-6
Al-Musawi, T. J., Rajiv, P., Mengelizadeh, N., Mohammed, I. A., & Balarak, D. (2021). Development of sonophotocatalytic process for degradation of acid orange 7 dye by using titanium dioxide nanoparticles/graphene oxide nanocomposite as a catalyst. Journal of Environmental Management, 292, 112777. https://doi.org/10.1016/j.jenvman.2021.112777
Altintig, E., Sarıcı, B., & Karataş, Ş. (2023). Prepared activated carbon from hazelnut shell where coated nanocomposite with Ag+ used for antibacterial and adsorption properties. Environmental Science and Pollution Research, 30, 13671–13687. https://doi.org/10.1007/s11356-022-23004-w
Badkar, D. S., Pandey, K. S., & Buvanashekaran, G. (2013). Development of RSM- and ANN-based models to predict and analyze the effects of process parameters of laser-hardened commercially pure titanium on heat input and tensile strength. The International Journal of Advanced Manufacturing Technology, 65, 1319–1338. https://doi.org/10.1007/s00170-012-4259-0
Balarak, D., Abasizadeh, H., Yang, J. K., Shim, M. J., & Lee, S. M. (2020). Biosorption of acid orange 7 (AO7) dye by canola waste: Equilibrium, kinetic and thermodynamics studies. Desalination and Water Treatment, 190, 331–339. https://doi.org/10.5004/dwt.2020.25665
Balarak, D., Al-Musawi, T. J., Mohammed, I. A., & Abasizadeh, H. (2020). The eradication of reactive black 5 dye liquid wastes using Azolla filiculoides aquatic fern as a good and an economical biosorption agent. SN Applied Science, 2(6), 1015. https://doi.org/10.1007/s42452-020-2841-x
Balarak, D., Ganji, F., Choi, S. S., Lee, S. M., & Shim, M. J. (2019). Effects of operational parameters on the removal of acid blue 25 dye from aqueous solutions by electrocoagulation. Applied Chemical Engineering, 30(6), 742–748. https://doi.org/10.14478/ace.2019.1092
Balarak, D., Zafariyan, M., Igwegbe, C. A., Onyechi, K. K., & Ighalo, J. O. (2021). Adsorption of acid blue 92 dye from aqueous solutions by single-walled carbon nanotubes: Isothermal, kinetic, and thermodynamic studies. Environmental Processes, 8, 869–888. https://doi.org/10.1007/s40710-021-00505-3
Bhatia, D., Sharma, N. R., Singh, J., & Kanwar, R. S. (2017). Biological methods for textile dye removal from wastewater: A review. Critical Reviews in Environmental Science and Technology, 47(19), 1836–1876. https://doi.org/10.1080/10643389.2017.1393263
Burca, S., Indolean, C., & Maicaneanu, A. (2017). Isotherms study of Congo red biosorption equilibrium using FIR (Abies Nordmanniana) sawdust biomass. Revue Roumaine de Chimie, 62, 381–389.
Carletto, R. A., Chimirri, F., Bosco, F., & Ferrero, F. (2008). Adsorption of Congo red dye on hazelnut shells and degradation with Phanerochaete chrysosporium. Adsorption Journal of the International Adsorption Society, 3, 1146–1155.
Chanzu, H. A., Onyari, J. M., & Shiundu, P. M. (2019). Brewers’ spent grain in adsorption of aqueous Congo red and malachite green dyes: Batch and continuous flow systems. Journal of Hazardous Materials, 380, 120897. https://doi.org/10.1016/j.jhazmat.2019.120897
Chen, Y., Lin, Y.-C., Hoa, S.-H., Zhouc, Y., & Ren, N. (2018). Highly efficient adsorption of dyes by biochar derived from pigments-extracted macroalgae pyrolyzed at different temperature. Bioresearch Technology, 259, 104–110. https://doi.org/10.1016/j.biortech.2018.02.094
Dahlan, I., Obi, C. C., & Poopathi, V. (2023). Predicting the adsorption capacity of organic dye using zirconium-based metal–organic framework: A comparative analysis of RSM and ANN-based models. Biomass Conversion and Biorefinery. https://doi.org/10.1007/s13399-023-05012-z
Das, S., Cherwoo, L., & Singha, R. (2023). Decoding dye degradation: Microbial remediation of textile industry effluents. Biotechnology Notes, 4, 64–76. https://doi.org/10.1016/j.biotno.2023.10.001
Dolatabadi, M., Mehrabpour, M., Esfandyari, M., Alidadi, H., & Davoudi, M. (2018). Modeling of simultaneous adsorption of dye and metal ion by sawdust from aqueous solution using of ANN and ANFIS. Chemometrics and Intelligent Laboratory Systems, 181, 72–78. https://doi.org/10.1016/j.chemolab.2018.07.012
El-Shafie, M. (2023). A comprehensive assessment of ammonia synthesis reaction kinetics and rate equations. International Journal of Hydrogen Energy, 48, 35938–35952. https://doi.org/10.1016/j.ijhydene.2023.06.011
Freundlich, H. M. (1906). Over the adsorption in solution. Journal of Physical Chemistry A, 57, 385–470.
Hasanzadeh, M., Simchi, A., & Far, H. S. (2019). Kinetics and adsorptive study of organic dye removal using water-stable nanoscale metal organic frameworks. Material Chemistry and Physics, 233, 267–275. https://doi.org/10.1016/j.matchemphys.2019.05.050
Hassaan, M. A., El Nemr, A., & Madkour, F. F. (2017). Testing the advanced oxidation processes on the degradation of direct blue 86 dye in wastewater. The Egyptian Journal of Aquatic Research, 43(1), 11–19. https://doi.org/10.1016/j.ejar.2016.09.006
Ho, J. H. N., Kallstrom, G., & Johnson, A. W. (2000). Nmd3p is a Crm1p-dependent adapter protein for nuclear export of the large ribosomal subunit. Journal of Cell Biology, 151(5), 1057–1066. https://doi.org/10.1083/jcb.151.5.1057
Ho, Y. S., & McKay, G. (1999). Pseudo-second order model for sorption processes. Process Biochemistry, 34, 451–465. https://doi.org/10.1016/S0032-9592(98)00112-5
Ilavenil, K. K., Senthilkumar, V., & Pichailakshmi, R. (2024). Lantana trifolia activated carbon for removal of 2-phenoxyethanoic acid: neural network optimization approach. RASĀYAN Journal of Chemistry, 17(2), 363–371. https://doi.org/10.31788/RJC.2024.1728543
Jorge, A. M. S., Athira, K. K., Alves, M. B., Gardas, R. L., & Pereira, J. F. B. (2023). Textile dyes effluents: A current scenario and the use of aqueous biphasic systems for the recovery of dyes. Journal of Water Process Engineering, 55, 104125. https://doi.org/10.1016/j.jwpe.2023.104125
Katheresan, V., Kansedo, J., & Lau, S. Y. (2018). Efficiency of various recent wastewater dye removal methods: A review. Journal of Environmental Chemical Engineering, 6(4), 4676–4697. https://doi.org/10.1016/j.jece.2018.06.060
Kaya, N., Uzun, Z. Y., Altuncan, C., & Uzun, H. (2022). Adsorption of Congo red from aqueous solution onto KOH-activated biochar produced via pyrolysis of pine cone and modeling of the process using artificial neural network. Biomass Conversion and Biorefinery, 12, 5293–5315. https://doi.org/10.1007/s13399-021-01856-5
Kaykıoğlu, G. (2016). Kolemanit ve Üleksit Atığı ile Sulu Çözeltilerden Metilen Mavisi Giderimi: Kinetik ve İzoterm Değerlendirmesi. CBÜ Fen Bilimleri Dergisi, 12(3), 499–509.
Khadhraoui, M., Trabelsi, H., Ksibi, M., Bouguerra, S., & Elleuch, B. (2009). Discoloration and detoxification of a Congo red dye solution by means of ozone treatment for a possible water reuse. Journal of Hazardous Materials, 161, 974–981. https://doi.org/10.1016/j.jhazmat.2008.04.060
Khan, T. A., Sharma, S., Khan, E. A., & Mukhlif, A. A. (2014). Removal of Congo red and basic violet 1 by chir pine (Pinus roxburghii) sawdust, a saw mill waste: Batch and column studies. Toxicology & Environmental Chemistry, 96, 555–568. https://doi.org/10.1080/02772248.2014.959017
Kostić, M., Đorđević, M., Mitrović, J., Velinov, N., Bojić, D., Antonijević, M., & Bojić, A. (2017). Removal of cationic pollutants from water by xanthated corn cob: Optimization, kinetics, thermodynamics, and prediction of purification process. Environmental Science and Pollution Research, 24, 17790–17804. https://doi.org/10.1007/s11356-017-9419-1
Kostić, M., Mitrović, J., Radović, M., Đorđević, M., Petović, M., Bojić, D., & Bojić, A. (2016). Effects of power of ultrasound on removal of Cu(II) ions by xanthated Lagenaria vulgaris shell. Ecological Engineering, 90, 82–86. https://doi.org/10.1016/j.ecoleng.2016.01.063
Kostić, M., Najdanović, S., Velinov, N., Vučić, M. R., Petrović, M., Mitrović, J., & Bojić, A. (2022). Ultrasound-assisted synthesis of a new material based on MgCoAl-LDH: Characterization and optimization of sorption for progressive treatment of water. Environmental Technology & Innovation, 26, 102358. https://doi.org/10.1016/j.eti.2022.102358
Kostić, M. M., Radović, M. D., Mitrović, J. Z., Bojić, D. V., Milenković, D. D., & Bojić, A. L. (2013). Application of new biosorbent based on chemicaly modified Lagenaria vulgaris shell for the removal of copper(II) from aqueous solutions: Effects of operational parameters. Hemijska Industrija, 67(4), 559–567. https://doi.org/10.2298/HEMIND120703097K
Lagergren, S. (1898). About the theory of so-called adsorption of soluble substances. Kungliga Svenska Vetenskapsakademiens Handlingar, 24, 1–39.
Langmuir, I. (1918). The adsorptıon of gases on plane surfaces of glass, mica and platinum. Journal of the American Chemical Society, 40(9), 1361–1403.
Laskar, I. I., & Hashisho, Z. (2020). Insights into modeling adsorption equilibria of single and multicomponent systems of organic and water vapors. Separation and Purification Technology, 241, 116681. https://doi.org/10.1016/j.seppur.2020.116681
Litefti, K., Freire, M. S., Stitou, M., & González-Álvarez, J. (2019). Adsorption of an anionic dye (Congo red) from aqueous solutions by pine bark. Scientific Reports, 9, 16530. https://doi.org/10.1038/s41598-019-53046-z
Mendoza-Castillo, D. I., Reynel-Ávila, H. E., Sánchez-Ruiz, F. J., Trejo-Valencia, R. J., Jaime-Leal, E., & Bonilla-Petriciolet, A. (2018). Insights and pitfalls of artificial neural network modeling of competitive multi–metallic adsorption data. Journal of Molecular Liquids, 251, 15–27. https://doi.org/10.1016/j.molliq.2017.12.030
Mobarak, M. B., Pinky, N. S., Chowdhury, F., Hossain, M. S., Mahmud, M., Quddus, M. S., Jahan, S. A., & Ahmed, S. (2023). Environmental remediation by hydroxyapatite: Solid state synthesis utilizing waste chicken eggshell and adsorption experiment with Congo red dye. Journal of Saudi Chemical Society, 27, 101690. https://doi.org/10.1016/j.jscs.2023.101690
Mondal, N. K., & Kar, S. (2018). Potentiality of banana peel for removal of congo red dye from aqueous solution: isotherm, kinetics and thermodynamics studies. Applied Water Science, 8, 157. https://doi.org/10.1007/s13201-018-0811-x
Munagapati, V. S., & Kim, D. S. (2017). Equilibrium isotherms, kinetics, and thermodynamics studies for Congo red adsorption using calcium alginate beads impregnated with nano-goethite. Ecotoxicology and Environmental Safety, 141, 226–234. https://doi.org/10.1016/j.ecoenv.2017.03.036
Namasivayam, C., & Kavitha, D. (2002). Removal of Congo Red from water by adsorption onto activated carbon prepared from coir pith, an agricultural solid waste. Dyes and Pigments, 54(1), 47–58. https://doi.org/10.1016/S0143-7208(02)00025-6
Nguyen, V. D., Nguyen, H. T. H., Vranova, V., Nguyen, L. T. N., Bui, Q. M., & Khieu, T. T. (2021). Artificial neural network modeling for Congo red adsorption on microwave-synthesized akaganeite nanoparticles: Optimization, kinetics, mechanism, and thermodynamics. Environmental Science and Pollution Research, 28, 9133–9145. https://doi.org/10.1007/s11356-020-10633-2
Patel, H., & Vashi, R. T. (2012). Removal of Congo red dye from its aqueous solution using natural coagulants. Journal of Saudi Chemical Society, 16, 131–136. https://doi.org/10.1016/j.jscs.2010.12.003
Patel, H., Yadav, V. K., Yadav, K. K., Choudhary, N., Kalasariya, H., Alam, M. M., Gacem, A., Amanullah, M., Ibrahium, H. A., Park, J.-W., Park, S., & Jeon, B.-H. (2022). A recent and systemic approach towards microbial biodegradation of dyes from textile industries. Water, 14(19), 3163. https://doi.org/10.3390/w14193163
Pauletto, P. S., Gonçalves, J. O., Pinto, L. A. A., Dotto, G. L., & Salau, N. P. G. (2020). Single and competitive dye adsorption onto chitosan–based hybrid hydrogels using artificial neural network modeling. Journal of Colloid and Interface Science, 560, 722–729. https://doi.org/10.1016/j.jcis.2019.10.106
Ramesh, P., Padmanabhan, V., Saravanan, P., & Thillainayagam, B. P. (2023). Batch studies of turquoise blue dye (TB) adsorption onto activated carbon prepared from low-cost adsorbents: An ANN approach. Biomass Conversion and Biorefinery, 13, 3267–3280. https://doi.org/10.1007/s13399-021-01355-7
Rendon-Castrillon, L., Ramírez-Carona, M., Ocampo-Lopez, C., Gonzalez-Lopez, F., Cuartas-Uribe, B., & Mendoza-Roca, J. A. (2023). Treatment of water from the textile industry contaminated with indigo dye: A hybrid approach combining bioremediation and nanofiltration for sustainable reuse. Case Studies in Chemical and Environmental Engineering, 8, 100498. https://doi.org/10.1016/j.cscee.2023.100498
Ribeiro, A. V. F. N., Silva, A. R., Pereira, M. G., Licinio, M. V. V. J., & Ribeiro, J. N. (2018). Wood sawdust powder from Corimbya citriodora to congo red toxic dye adsorption. Indian Journal of Applied Research, 8, 29–31.
Singha, B., Bar, N., & Das, S. K. (2015). The use of artificial neural network (ANN) for modeling of Pb(II) adsorption in batch process. Journal of Molecular Liquids, 211(2015), 228–232. https://doi.org/10.1016/j.molliq.2015.07.002
Souza, P. R., Dotto, G. L., & Salau, N. P. G. (2018). Artificial neural network (ANN) and adaptive neuro-fuzzy interference T system (ANFIS) modelling for nickel adsorption onto agro-wastes and commercial activated carbon. Journal of Environmental Chemical Engineering, 6, 7152–7160. https://doi.org/10.1016/j.jece.2018.11.013
Stjepanović, M., Velić, N., Galić, A., Kosović, I., Jakovljević, T., & Habuda-Stanić, M. (2021). From waste to biosorbent: removal of congo red from water by waste wood biomass. Water, 13(3), 279. https://doi.org/10.3390/w13030279
Sudarshan, S., Harikrishnan, S., RathiBhuvaneswari, G., Alamelu, V., Aanand, S., Rajasekar, A., & Govarthanan, M. (2023). Impact of textile dyes on human health and bioremediation of textile industry effluent using microorganisms: current status and future prospects. Journal of Applied Microbiology, 134(2), 1–23. https://doi.org/10.1093/jambio/lxac064
Sun, J., Wang, Y., Sun, R., & Dong, S. (2009). Photodegradation of azo dye Congo red from aqueous solution by the WO3–TiO2/activated carbon (AC) photocatalyst under the UV irradiation. Materials Chemistry and Physics, 115, 303–308. https://doi.org/10.1016/j.matchemphys.2008.12.008
Tanzifi, M., Yaraki, M. T., Kiadehi, A. D., Hosseini, S. H., Olazar, M., Bharti, A. K., Agarwal, S., Gupta, V. K., & Kazemi, A. (2018). Adsorption of amido black 10B from aqueous solution using polyaniline/SiO2 nanocomposite: Experimental investigation and artificial neural network modeling. Journal of Colloid and Interface Science, 510, 246–261. https://doi.org/10.1016/j.jcis.2017.09.055
Yusuf, M., Song, K., & Li, L. (2020). Fixed bed column and artificial neural network model to predict heavy metals adsorption dynamic on surfactant decorated graphene. Colloids and Surfaces A, 585, 124076. https://doi.org/10.1016/j.colsurfa.2019.124076
Zabihi, M., Omidvar, M., Motavalizadehkakhky, A., & Zhiani, R. (2022). Competitive adsorption of arsenic and mercury on nano-magnetic activated carbons derived from hazelnut shell. Korean Journal of Chemical Engineering, 39(2), 367–376. https://doi.org/10.1007/s11814-021-0903-4
Zhang, E. L., Sun, X. J., Liu, X. T., & Wang, Q. D. (2015). Morphology controlled synthesis of α-FeOOH crystals and their shape-dependent adsorption for decontamination of Congo red dye. Materials Research Innovation, 19, 385–391. https://doi.org/10.1179/1433075X15Y.0000000019
Author information
Authors and Affiliations
Contributions
The article was written entirely by Özgül Çimen Mesutoğlu.
Corresponding author
Ethics declarations
Ethical approval
Author has read, understood, and has complied as applicable with the statement on “Ethical responsibilities of Authors” as found in the Instructions for Authors.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Çimen Mesutoğlu, Ö. The use of artificial neural network for modelling adsorption of Congo red onto activated hazelnut shell. Environ Monit Assess 196, 630 (2024). https://doi.org/10.1007/s10661-024-12797-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10661-024-12797-7