Abstract
The use of organisms like macroinvertebrates in developing bioassessment tools, such as multimetric indices (MMIs), is gaining global recognition in monitoring the health status of lakes. The transition from traditional methods of physico-chemical parameters is due to the financial and time costs involved in their analysis while failing to provide accurate early warning signals on ecosystem conditions. Currently, there is scanty information on the use of MMIs in the conservation and management of lakes in Nigeria. This study aimed at developing a macroinvertebrate-based MMI to assess the ecological status of lakes in North Central Nigeria. The study was conducted on Tagwai and Old Gawu Lakes, from April to October 2022. Sampling sites were clustered based on organic pollution and categorized into reference (four) and impaired (four) sites. Out of 54 macroinvertebrate-based candidate metrics, only five were selected after discriminatory, stability, and redundancy tests (performed using R software). The final metrics were abundance of Ephemeroptera + Trichoptera; abundance of Coleoptera + Ephemeroptera; Gastropoda richness; Shannon Wiener index; and percentages of shredders + predators + scrapers, hereafter referred to as North Central Nigeria-Lakes Multimetric Index (NCN-LMMI). The NCN-LMMI values ranged as follows: 21–25, 16–20, 11–15, and 5–10 corresponding to categories I, II, III, and IV for water quality, as indications of good, fair, poor, and very poor ecological status of the lake, respectively. The developed NCN-LMMI will be a useful tool for aquatic resource managers and environmentalists to assess the ecological condition of lakes, mainly the North Central Nigeria municipal lakes.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Biological indicators are increasingly used in aquatic ecosystem health monitoring due to their potential over other evaluation methods like physico-chemical parameters (Lopez-Lopez & Sedeno-Diaz, 2015; Moog et al., 2018). Bioindicators can reflect long-term aquatic ecosystem conditions (Uherek & Gouveia, 2014; Young et al., 2014). Biomonitoring is a fast and cost-effective method, since it requires less field and laboratory equipment compared to physico-chemical parameters (Asif et al., 2018; Fontanetti et al., 2012; Kumari & Paul, 2021). This approach has gained recognition and has been used in freshwater assessment worldwide (Karmakar et al., 2021; Mahaut et al., 2013; Parmar et al., 2016; Poulíčková et al., 2004; Riato & Leira, 2020). In particular, a technique of grouping biological metrics into a single index, known as the multimetric index (MMI), is being used in health monitoring of both lentic and lotic ecosystems. Multimetric index represents various aspects of ecological information, such as abundance, diversity, composition, richness, and function (Arman et al., 2019; Gabriels et al., 2010; Huang et al., 2015; Zhang et al., 2019). Since the first attempt of MMI by Karr (1981) using fish, other organisms such as diatoms (Reavie et al., 2008; Schowe & Harding, 2014; Yu et al., 2021), macrophytes (Aguiar et al., 2011; Beck et al., 2010; Penning et al., 2008), and macroinvertebrates (Edegbene et al., 2019; Menetrey et al., 2011; Utami & Fajar, 2022) have been used worldwide. The use of MMIs as freshwater monitoring tools has been reported in various regions including North America (Haugerud, 2006; Karr, 1981; Wesolek et al., 2010), South America (Baptista et al., 2007; Carayon et al., 2020; Touron-poncet et al., 2014), Europe (McGoff et al., 2013; Miler et al., 2013; Solimini et al., 2008; Trigal et al., 2006), Asia (Ding et al., 2021; Zhang et al., 2019), and Africa (Edegbene et al., 2019; Wondmagegn & Mengistou, 2023).
Macroinvertebrates have for long been used as bioindicators of water quality, owing to their ability to respond differently to environmental stressors, mainly of anthropogenic origin (Assefa et al., 2020; Ntitslidou et al., 2021). Most macroinvertebrates are easy to collect/sample and have relatively long life cycle, thus representing seasonal changes and functional traits that enable them to indicate disturbance in the habitats (Bêche et al., 2006; Bonada et al., 2006; Young et al., 2014). Unlike relatively big-sized organisms such as fish with higher habitat restrictions (Kramer & Chapman, 1999), macroinvertebrates can easily be found in almost all aquatic ecosystems (Feio et al., 2023). Their responses to the environment are guided by their sensitivity/tolerance levels to pollution. For instance, Ephemeroptera, Plecoptera, and Trichoptera (EPT) have been reported to be very sensitive to organic pollution (Akamagwuna et al., 2021; Menetrey et al., 2008). As such, the abundance of EPT is often very low in an ecosystem or site with high organic pollution, often reflecting the status of impairment. Considering the same stressor (organic pollution), some chironomids are considered to be very tolerant, i.e., their abundance is not much affected by organic pollution (Auta et al., 2016 ; Cornette et al., 2015). The variation in sensitivity of organisms prompted studies to illustrate some macroinvertebrate taxa with their respective sensitivity/tolerance levels (Bonada et al., 2006). Indeed, the macroinvertebrate-based sensitivity/tolerance metrics have been used in a number of biomonitoring studies (Baptista et al., 2007; Edegbene et al., 2022; Macedo et al., 2016; Shah et al., 2011). In addition, macroinvertebrates have been considered in MMI development, an approach that has been widely applied in lotic environments (rivers/streams) in comparison to lentic (lakes/reservoirs) (Assefa et al., 2020; Ndatimana et al., 2023; Odountan et al., 2018).
The development and application of macroinvertebrate-based MMI in lakes are drastically increasing in developed countries (Ding et al., 2021; McGoff et al., 2013; Miler et al., 2013; Zhang et al., 2019) and a few developing countries, including African countries (Wondmagegn & Mengistou, 2023). In developed countries, various metrics are assessed in response to the main stressors of lake integrity such as eutrophication, acidification, and geomorphological changes (Young et al., 2014). The profundal-based macroinvertebrate metrics (Jyväsjärvi et al., 2010, 2012), littoral-based metrics (Johnson, 2003; McFarland et al., 2010; Schartau et al., 2008), and assessment of geomorphological alteration (Brauns et al., 2007; Miler et al., 2013) were applied to handle these main disturbances of lake integrity. In addition, collaboration and the implementation of water-related policies such as the European Union Water Framework Directive (WFD) and the United States Environmental Protection Agency (USEPA) have helped the development and application of MMIs in developed countries (Gabriels et al., 2010; Poikane et al., 2011; Sreeja, 2018).
In Africa, lakes are supporting the life of riparian communities, both directly and indirectly, for instance, through provision of fishery resources (Abo-Taleb et al., 2023; Ogutu-Ohwayo et al., 2016), water for irrigation (Agodzo, 2013; Salomonsson & Thoresson, 2010; Zamdayu & Japhet, 2021), municipal and domestic use (Salomonsson & Thoresson, 2010; Shishaye, 2018; Yonnana et al., 2017), as well as recreation services (Kolawole et al., 2020; Nakirya et al., 2015). However, these activities are affecting water quality and the health status of inhabiting organisms (Cohen et al., 2019; Onywere et al., 2012). Indeed, laundry activities, agricultural wastes, direct dumping of wastes, oil spillage, and automobile washing, among others, cause organic and heavy metal pollution, which alter the ecological functioning of lakes (Gwenzi & Chaukura, 2018; Obinnaa & Ebere, 2019; Redwan & Elhaddad, 2022). The effective response of macroinvertebrates to such disturbances makes them good indicators of aquatic health (Gezie et al., 2017; Wondmagegn & Mengistou, 2020). Therefore, the application of MMI is reliable especially when the exact stressors affecting the lake are not known (Schoolmaster et al., 2012). Thus, macroinvertebrate-based MMI is a good option in monitoring African lakes which are being affected by multiple stressors. Indeed, macroinvertebrates have been recommended in MMI development to improve the use of biomonitoring approaches on African lakes (Ndatimana et al., 2023; Odountan et al., 2018). Macroinvertebrates showed potentials of application in MMI for African rivers (Alemneh et al., 2019; Aura et al., 2010; Edegbene et al., 2019), but their development and application in lakes are still at infancy.
Old Gawu and Tagwai Lakes in North Central Nigeria are currently threatened by agricultural and municipal wastes from their catchments (Musa & Saba, 2011), which increase organic pollutants in the lakes. In addition, non-point source pollution mainly from human activities in and around the lakes such as laundry, agricultural runoff, fishing, and washing automobiles portends the continuous pollution of these municipal lakes. This hinders the ecological functioning of the lakes and affects the inhabiting organisms (Egwu et al., 2020). Thus, there is a need for consistent water quality monitoring to ensure desirable ecological integrity. Unfortunately, the existing water quality assessment of lakes in the region is mainly based on physico-chemical parameters (Ibrahim et al., 2019), which are relatively expensive, time-consuming, and do not provide early warning signals compared to biological indicators. Currently, there is no documented macroinvertebrate-based MMI developed for lakes in Nigeria. Therefore, the use of metrics of macroinvertebrates from Old Gawu and Tagwai Lakes to develop an MMI will provide a baseline for successful biomonitoring of lakes in Nigeria, particularly, North Central region. It is anticipated that macroinvertebrate-based MMI will be able to discriminate highly impaired from least impaired sites. In addition, the developed MMI will help in rapid assessment of lake health status, as well as effective and evidence-based policy development for lake water quality management in the region.
Methods
Study area description
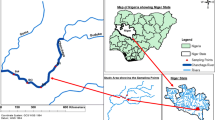
The study was conducted in two municipal lakes in North Central Nigeria: Old Gawu, located in the Federal Capital Territory (FCT) Abuja, and Tagwai Lake, located in Minna, Niger state Capital. Old Gawu Lake is situated on longitude 6°35′55.6″E and latitude 9°30′41.7″N (Fig. 1), located in Abaji local government area. Old Gawu is surrounded by agricultural farms dominated by subsistence crops such as yams, maize, beans, and white-seed melon locally known as Egusi. The main activities from this lake are livestock watering, laundry, and water abstraction for irrigation especially during dry season, recreation, and fishing. Meanwhile, Tagwai Lake is located in Minna, situated on longitude 6°39′40.95″E and latitude 9°33′59.958″N (Fig. 1). The lake is surrounded by agricultural farms dominated by maize, okra, and yams. This lake supports community in the vicinity through irrigation of agricultural farms, fishing, livestock watering, and recreation. Ecologically, the lakes are habited by a diversity of organisms such as fish, invertebrates (micro and macro), reptiles, and amphibians. The North Central Nigeria region has a tropical climate with a mean annual temperature of 30 °C, relative humidity of 61%, and rainfall of 1334 mm.
Macroinvertebrate sampling and laboratory analysis
Macroinvertebrates were sampled from eight sites, i.e., four sites per lake. For each site, three replicates from randomly selected sampling points were collected using a D-shaped hand net with a mesh size of 0.5 mm (Jones et al., 2004). Macroinvertebrates were collected within 25 to 40 cm depth, with a distance between sampling points approximately 3 to 5 m. The three replicate samples from each site were then pooled together into a single composite sample. Sampling was done monthly from April to August and October 2022, covering the dry period (April–May), wet period (June–August), and transition/before the dry season (BDS) (October). From the field, samples were fixed in 80% ethanol and transported to the Animal Biology Laboratory, Federal University of Technology, Minna (FUTMINNA).
In the laboratory, samples were sorted using a Digital microscope (YIZHAN, 38 MP FHD camera V6) connected to a computer and identified to family level using taxonomic keys by Arimoro and James (2008), de Moor et al. (2003), Gooderham and Tsyrlin (2002), and Thorp and Rogers (2011). Macroinvertebrates were assigned to functional feeding groups (FFG) using trait-based fuzzy coding by Tachet et al. (2010) and the aid of specialists from the Hydrobiology Unit, FUTMINNA.
Environmental variables
At each site, water temperature, dissolved oxygen (DO), hydrogen ion concentration (pH), and electric conductivity (EC) were determined using HACH multi-meter probe (HQ40d, USA). The laboratory analysis consisted of nitrate (NO3), calcium (Ca), phosphate (PO4), magnesium (Mg), BOD5, total alkalinity (TA), chlorine (Cl), total hardness (TH), chemical oxygen demand (COD), and chlorophyll-a (Chl-a), which were analysed based on standard methods by Gilcreas (1966).
Site categorization
The sampled sites were classified based on observed disturbance in the littoral zones, mainly the inlets of sewage/effluents, automobile washing, and recreational activities associated with point source pollution, as well as vegetation assemblage. Indeed, observation of riparian zone is an important aspect during site classification (Wondmagegn & Mengistou, 2023). The study area is mainly threatened by organic pollution, and the site clustering was emphasized by the level of impairment detected using selected environmental variables indicating organic pollution. The selected variables were DO, BOD, TDS, EC, and nutrients: NO3 and PO4. Thus, the Bray-Curtis clustering revealed four least impacted sites/reference and four impacted/impaired sites and was further confirmed using the Mann–Whitney U test (p < 0.05). The reference condition was given to sites with the least disturbance since it is not practical to find sites with undisturbed conditions (Zhang et al., 2019). The rest were classified as impaired/highly disturbed. The reference sites were Old Gawu Site 3 (OGS3), Old Gawu Site 4 (OGS4), Tagwai Site 2 (TS2), and Tagwai Site 3 (TS3), whereas impaired sites were Old Gawu Site 1 (OGS1), Old Gawu Site 2 (OGS2), Tagwai Site 1 (TS1), and Tagwai Site 4 (TS4). The reference sites were characterised by high DO and low BOD, EC, TDS, and nutrients, unlike impaired sites with low DO and high BOD, EC, TDS, and nutrients.
Candidate metrics selection
The macroinvertebrate metrics to be considered in MMI development are presumed to be available in the environment, with the potential ability to discriminate the sites effectively (Zhang et al., 2019). Since MMI combines several metrics into a single value (Gabriels et al., 2010), the constituent metrics are assumed to be representatives of various ecological information. Indeed, variation in metric groups creates diversity of response to disturbances, hence increasing the ecological information to be represented in an index (Baptista et al., 2007; Barbour et al., 1995, 1996).
To assess the response of the macroinvertebrate community to disturbances, 54 macroinvertebrate-based metrics were selected. The metrics selection consisted of measuring various aspects of macroinvertebrate communities gathered in five groups. The metric groups include (i) taxa abundance, i.e., the absolute number of macroinvertebrate individuals; (ii) taxa composition, i.e., relative abundance; (iii) taxa richness metrics; (iv) diversity metrics; and (v) functional feeding group (FFG). Of the 54 selected metrics, 15 were abundance metrics, 14 composition, 12 richness, six diversity indices, and seven FFG (Table 1).
Metrics computation and screening tests
To ensure better discrimination of references and impaired sites by candidate metrics, a series of tests were used. The screening tests included (i) sensitivity/discriminatory, (ii) stability/seasonality, (iii) redundancy, and (iv) correlation with environmental variables (abiotic factors) resulting into core metrics (Baptista et al., 2007; Jun et al., 2012; Wang et al., 2015; Wondmagegn & Mengistou, 2023). The metrics’ ranges (minimum-maximum) were considered in selection, whereby the metrics whose median value of 0 in both sites (reference and impaired) were excluded to avoid inefficient indication of the difference between these sites (Hering et al., 2006; Zhang et al., 2019). After this test, the normality of candidate metrics was performed using Shapiro-Wilk normality test. The metrics with normal and non-normal distribution were tested with parametric and parametric tests, respectively.
Discriminatory analysis, which consists of testing whether the metrics were able to differentiate the reference from impaired sites, was performed using box and whisker plots, accompanied with the Mann–Whitney U test. The box and whisker plots were used to visualize the variation of metrics range between reference and impaired sites, according to Barbour et al. (1996). The degree of interquartile (IQ) overlap helped to define the sensitivity value, by assigning the score to each metric. The IQ scores were 3 to 0, indicating no overlap to highly overlapped ranges. The IQ scores were assigned as IQ = 3, IQ = 2, and IQ = 1 when there was no overlap between two groups, overlap with all medians outside the IQ range, and moderate overlap with one median outside the IQ range, respectively. Meanwhile, IQ = 0 was assigned when the ranges were completely overlapping each other with all medians inside the IQ ranges. The metrics selected for further tests were those that showed higher sensitivity/discriminatory power (IQ ≥ 2), with a significant difference between the reference and impaired sites (p < 0.05).
The metrics which successfully met the previous criteria were tested for seasonal stability using the Kruskal-Wallis test. This was done by testing whether the metrics can withstand the seasonal variations. The metrics with stability power can withstand the seasonal variation, indicated by the absence of significant difference (p > 0.05) among seasons (Baptista et al., 2007). The redundancy test was applied to stable metrics using Spearman’s rank correlation to eliminate more redundant metrics, which showed a significantly high correlation (Spearman r ≥ 0.7 and p < 0.05). These metrics are considered not to represent new information in the index. In this case, one or more metrics were eliminated to ease the index application, reduce the analysis cost, and avoid redundant information (Barbour et al., 1996; Hering et al., 2006; Wondmagegn et al., 2019). The metrics that met redundancy tests were tested for correlation with environmental variables. Indeed, the potential metrics to be integrated in MMI should be correlated with environmental variables to be able to indicate the changes associated with disturbances. The retained metrics had significant correlation with environmental variables as indicated by Spearman’s correlation. The final metrics were expected to be negatively or positively correlated depending on explanatory factors and the prediction response to stressor/disturbance. Therefore, the metrics that passed all the aforementioned criteria were considered core/final metrics and were integrated in NCN-LMMI.
Development of North central Nigeria Lake multimetric index (NCN-LMMI)
The metrics which successfully met all the three criteria were integrated into NCN-LMMI as core metrics. The integration of core metrics into the final MMI required standardization of metrics since they had different numerical scales (numbers or percentages). Therefore, discrete scoring systems were performed to enable the collection of their values into single metrics (Baptista et al., 2007). For each metric, the range of values was sectioned into three possible scores, corresponding to the upper (75%), median (50%), and lower quartile (25%). The metrics’ quartile was used as a threshold, referring to the responses of metrics to the increase of disturbance or pollution. For metrics anticipated to decrease in response to the rise in disturbance/pollution, the values above the lower quartile of the reference distribution were given a score 5. Nonetheless, for metrics anticipated to increase with disturbance, each value below the upper quartile of the reference distribution was assigned a score 5. The scores 5, 3, and 1 represent the reference, intermediate, and highly degraded condition, respectively (Wang et al., 2015; Wondmagegn & Mengistou, 2023). Thus, the values of each metric, including the minimum, lower quartile, median, upper quartile, and maximum at reference sites, were used as the threshold to split score ranges for the establishment of the MMI.
NCN-LMMI performance validation
To evaluate the performance of the developed MMI, we estimated precision and compare the coefficient of variation (CV) of MMI values (Chen et al., 2014, 2017; Hawkins et al., 2010; Macedo et al., 2016). The estimation of precision was obtained using the correct classification percentage (CCP). The CCP of the reference cluster was computed as the percentage of sites appraised as good and fair from sites in the entire cluster, whereas the CCP of the impaired cluster was computed as the percentage of sites appraised as poor and very poor from sites in the entire group. Furthermore, to estimate the MMI precision, the CV of MMI values of the two clusters were compared, whereby the more precise the MMI, the lower the CV (Chen et al., 2014).
Data analysis
The metrics were organized, and arranged in Microsoft Excel (Microsoft Excel, 2019 Office), and further analyses were performed in R environment (RCore Team, 2022). The cluster analysis was performed using the Factoshiny package (Vaissie et al., 2021), for site classification. The Shapiro test was used to check for the normality of data sets. Mann–Whitney U test was used to assess the discriminatory power of metrics between reference and impaired sites, along with box and whisker plots visualization. Kruskal-Wallis test was used to check for seasonal differences of metrics across the study period. Spearman’s correlation was applied to determine the relationship between the metrics themselves and with environmental variables for core metrics selection, using the corrplot package (Taiyun & Simko, 2021).
Results
Macroinvertebrate metrics selection
Out of 54 metrics tested for discriminatory/sensitivity test, only 16 metrics showed discriminatory potential between reference and impaired sites (Table 2). This was revealed by the discriminatory power/sensitivity score (IQ ≥ 2) and significant difference (p < 0.05, Mann–Whitney test) expressed by the metrics between impaired and reference sites. Among these 16 metrics, five were abundance metrics, three composition, two richness, four FFG, and two diversity metrics. However, Per_lee, abun_Tric, and Tric_rich were not found in the reference sites (Median = 0) and thus were eliminated in further analysis. Therefore, 13 metrics were considered sensitive with their ability to discriminate reference from impaired sites, as indicated by the Mann–Whitney test and box–whisker plot (Table 2).
The sensitive metrics were tested for their ability to withstand seasonal changes. Macroinvertebrate assemblages often depend on the temporal changes in ecosystems (Bêche et al., 2006). The selected metrics for this test did not show significant difference between seasons, i.e., stable metrics. Therefore, only seven out of 13 metrics were able to show stability in various seasons (Table 3). The selected metrics, i.e., seasonally stable metrics were Eph_abun, ET_abun; CE_abun; Gast_rich; Shan_H, Marg_Index, Per_filt_feeder, and Per_Shre+Pred+Scrap.
The redundancy test was applied to seven stable metrics selected for seasonality test using Spearman’s rank correlation. Metrics were considered redundant when expressed a significant correlation coefficient greater than or equal to 0.7 (r ≥ 0.7; p < 0.05) (Hering et al., 2006; Wondmagegn & Mengistou, 2023; Fig. 2). For two or more redundant metrics, we selected one based on either knowledge of its potential indicator, i.e., ecological information derived from the metrics comparably to other(s) or its simplicity in calculation, to ease the index. Based on the aforementioned criteria, Shan_H and Marg_Index were redundant. However, Shan_H was chosen, ascribed to its ability to inform macroinvertebrate diversity aspect in the site and is often applied in diversity measurements, particularly during MMI development (Miler et al., 2013; Timm & Möls, 2012). Likewise, Per_filt_feeder was selected comparably to Per_Shre + Pred + Scrap, owing to its relatively easy identification cost, thus contributing to the simplicity of the index. Indeed, the main target of MMI is to compile the ecological information from various metrics response to disturbance into a single, simple, effective, and robust index (Chowdhury et al., 2016). The five metrics that succeeded redundancy test were ET_abun, CE_abun, Gast_rich, Shan_H, and Per_filt_feeder.
While assessing the relationship between metrics and environmental variables, the five selected metrics that passed through sensitivity, stability, and redundancy were considered. The Spearman’s rank correlation revealed that all selected metrics were correlated with at least two environmental variables (Table 4). Therefore, the selected metrics at this stage were considered core metrics and were finally integrated into the final NCN-LMMI.
Development of North central Nigeria Lake multimetric index (NCN-LMMI)
From all the 54-candidate metrics tested, 9.25% were integrated into the index. The final metrics integrated into the NCN-LMMI were ET_abun, CE_abun, Gast_rich, Shan_H, and Per_Filt-feeder (Fig. 3). For each of the five-core metrics integrated into the NCN-LMMI, the values for apt quartile at reference sites were used as a threshold for splitting high score from the lower score (Table 5). To standardize the metrics values, the discrete scoring was applied following the response of final metrics (either increase or decrease with increase in disturbance) as described in Table 5. NCN-LMMI was attained by summing the scores of five metrics, thus generating the range of 5 to 25 as the possible range of the index. Thereafter, the NCN-LMMI range was quadrisected to provide four water quality categories I, II, III, and IV portending to good, fair, poor, and very poor ecological condition of the lake (site), respectively (Table 6).
NCN-MMI validation
The CCP values of the reference cluster and the impaired cluster were 100% and 75%. respectively, an indication of a very good precision of MMI. Indeed, the scores of the newly developed NCN-LMMI were applied to the impaired sites, OG1, OG2, TS1, and TS4 data, and revealed that all were in poor ecological condition (Table 7). This indicates the NCN-LMMI’s ability to differentiate the impaired from reference sites. This was emphasized by the CVs of the reference cluster and impaired cluster which were 0.14 and 0.15, respectively, proving that NCN-LMMI was more precise with both reference and impaired sites.
Discussion
Site classification
Based on the nature of Tagwai and Old Gawu Lakes, with common stressors including agricultural activities and the domestic and municipal wastes (Oladosu et al., 2019), the NCN-LMMI was developed for organic pollution. Indeed, these stressors are the primary sources of organic pollution that lead to nutrient enrichment of the lakes (Zhang et al., 2019), thus affecting aquatic ecosystem health. Dissolved oxygen (DO), BOD, EC, and nutrients (nitrate and phosphate) were used, since their concentration can indicate the levels of organic pollution in a water body (Menetrey et al., 2011; Šidagyte et al., 2013). The impairment of sites can also be linked to physical degradation such as water abstraction and clearing of the vegetation buffering the lakes leading to direct entry of pollutants. The reference sites should be able to express the natural patterns and processes basically with no anthropogenic disturbances, to be able to differentiate them from disturbed sites (Whittier et al., 2007). However, it is hard to find the reference sites as described (Stoddard et al., 2006; Whittier et al., 2007); instead, the least-disturbed sites were considered as reference in this study. Generally, site classification depends on water body stressor/disturbance; thus, various parameters act as indicators, for instance, trophic level (Ding et al., 2021; Zhang et al., 2019), degree of acidity (e.g., Schartau et al., 2008), or combination of multiple factors (Menetrey et al., 2011).
The site classification resulted in two final clusters namely reference sites, i.e., less disturbed, and impaired sites, i.e., highly degraded/disturbed. Two clusters were also obtained by Ruse (2010) and Shah et al. (2011), during MMI development. However, various MMIs resulted in the use of three categories: reference/no-impacted/less impacted, moderately impaired, and disturbed/impaired group (Chowdhury et al., 2016; Ding et al., 2021; Wondmagegn & Mengistou, 2023). Therefore, the number of clusters depends on degradation gradients and site classification methods.
Metrics selection and NCN_MMI development
The choice of candidate metrics for NCN-LMMI reflects metrics used locally and in the bioassessment of the water quality of lake ecosystems. Metrics selection can be done based on taxa indicators used in the local/regional area (Edegbene et al., 2019; Wondmagegn & Mengistou, 2023) or the successful metrics used in the same ecosystem/type of water body, either lake or river (Hering et al., 2006; Zhang et al., 2021). Indeed, a similar approach was applied while selecting metrics for MMI Flanders, where metrics which were successfully used throughout the targeted area were considered (Gabriels et al., 2010).
A total of five-core metrics from four metrics groups: two abundance, one composition, one diversity, and one FFG were integrated into the final MMI. The metrics from various groups indicate the complexity (diversity of information/responses to disturbance) by macroinvertebrate assemblages, hence improving the robustness of MMI (Hering et al., 2006; Stoddard et al., 2008). The screening tests helped eliminate the metrics from 54 to only five with intention of obtaining the simple, effective, and more informative metrics governing the index. Through this process, some MMIs end up screening process with different numbers of core/final metrics. Final metrics can be four (Ding et al., 2021), often between five and ten final metrics (McGoff et al., 2013; Shah et al., 2011; Wesolek et al., 2010; Zhang et al., 2019), or even from ten and above (Keizer-Vlek, 2014; Trigal et al., 2006). The variation in the number of final metrics is mostly based on the nature of water body, metrics computation and analysis, and target of MMI as well.
The abundance metrics were used in water quality monitoring, specifically in MMI development (Šidagyte et al., 2013; Timm & Möls, 2012; Zhang et al., 2019). Indeed, the absolute abundance of macroinvertebrate provides a response to the environment, owing to their affinity to stressors (Camargo et al., 2011). For instance, the abundance of macroinvertebrates showcases water chemistry accruing to pollution that may be occurring in the environment (Arimoro et al., 2015). Composition metrics have been used in many MMIs (Hering et al., 2006; McGoff et al., 2013; Schartau et al., 2008; Shah et al., 2011; Taowu et al., 2008), due to their details in indicating the organisms’ responses to stressors. Meanwhile, diversity metrics are widely used in water quality monitoring including MMI (Ding et al., 2021; Hering et al., 2006; Timm & Möls, 2012; Zhang et al., 2019) and other biological approaches of water quality assessment such as surveys and multivariate analysis (Djamai et al., 2019; Ghosh & Biswas, 2015; Latha & Thanga, 2010). Shannon–Wiener index has a strong influence on indicating the status of macroinvertebrates (Clews et al., 2014), hence predicting their response to stressors. The FFG was considered as a group of metrics while developing NCN-LMMI, a similar approach that has previously been applied (Chowdhury et al., 2016; Ding et al., 2021; Huang et al., 2015; Wesolek et al., 2010). The selection of FFG metrics was associated with their sensitivity to nutrient enrichment, thus acting as a good indicator of organic pollution, compared to other taxonomic metrics (Menezes et al., 2010; Xu et al., 2014). In addition, FFG encompasses various taxa that could respond differently to stressors (Newbold et al., 2020); however, as single feeding behaviour, assemblage provides efficient information to the final MMI. Furthermore, this metric (Per_filter feeders) was able to successfully pass through all screening tests, mainly due to its sensitivity to stressors, stability among seasons, and ability to correlate to environmental variables (Figs. 2 and 3).
The core metrics correlated significantly with environmental variables, justifying the robustness of NCN-LMMI. For instance, Shan_H positively correlated with DO and negatively correlated with phosphate, TA, Ca, and Mg. Indeed, Shan_H decreases with the increase in pollution (Moiseenko et al., 2009). The pollution-sensitive metrics such as ET_abun and EC_abun, which decrease with the increasing pollution negatively correlated with variables like BOD, TDS, EC, and nutrients. This expresses the ability of NCN-LMMI to notice the change, and status of pollution, and provide ideal signals/early warning. Similar findings, i.e., the correlation of the metrics with environmental parameters, often used as indicators of pollution (Gabriels et al., 2010; Shah et al., 2011; Šidagyte et al., 2013). On the other hand, a relationship was revealed among the two pollution tolerant metrics: the Gast_rich and Per_filt_feeder with environmental variables such as TDS, EC, Cl, and TH. This is congruent with the reported studies by Schartau et al. (2008) and Zhang et al. (2019), where a positive correlation was found between metrics that respond positively to the disturbance.
Ephemeroptera and Trichoptera taxa have been reported to be highly sensitive to organic pollution (Akamagwuna et al., 2019; Chowdhury et al., 2016; Mohammed et al., 2022). Similarly, these taxa are part of EPT, the commonly used metric in both biological assessment and MMIs (Baptista et al., 2007; Gabriels et al., 2010; McGoff et al., 2013; Menetrey et al., 2011). However, EPT did not show stability by discriminating reference from impaired sites. The ET_abun is considered sensitive, its abundance decreased with increasing pollution, and the trend was emphasized in the data collected at Old Gawu and Tagwai Lakes. On the contrary, ET can respond positively to pollution when the sample is dominated by the less sensitive taxa such as Caenidae and Heptageniidae (Brown, 2009), which could alter the response to disturbance. Ephemeroptera appeared in two final metrics (ET_abun and CE_abun), not only to efficiently represent their information in the MMI but also to contemplate its interactions with Trichoptera and Coleoptera. Individually, Coleoptera and Trichoptera did not show discriminatory power; however, when combined with Ephemeroptera, they were able to differentiate reference from impaired and withstood the seasonal variation. Indeed, Coleoptera were recorded in high abundance from reference sites compared to impaired sites. In addition, studies reported metrics comprising Coleoptera as sensitive metrics (Dong et al., 2014; McGoff et al., 2013; Menetrey et al., 2011).
Gastropoda richness was integrated into MMI, owing to its response to disturbances. The most sampled family was Thiaridae, a pollution-tolerant taxa (Akindele & Liadi, 2013; Weir & Salice, 2012). Nevertheless, some gastropods are sensitive, e.g., Caenogastropoda, also known as gilled snails (Puglisi, 2021). Caenogastropoda’s breathing mechanism requires absorption of DO through gills, making them highly dependent on good water quality with sufficient DO concentration for survival, hence indicating high sensitivity to pollution (Anyanwu et al., 2019). Gastropoda richness has been integrated into various MMIs as a core metric (Schartau et al., 2008; Taowu et al., 2008; Zhang et al., 2019), owing to their ability to discriminate efficiently reference from impaired sites.
In NCN-LMMI development, Shan_H was able to differentiate reference from impaired sites, owing to its response to environmental degradation. Shannon–Wiener index is the most common metric used in macroinvertebrate-based MMI (Burton et al., 1999; Gabriels et al., 2010; Trigal et al., 2006; Wesolek et al., 2010). Its reliable measure and indicator help in understanding community diversity (Ghosh & Biswas, 2015), hence indicating the diversity of taxa in the targeted sites. In addition, Shannon–Wiener index is a pollution indicator, which is expected to be lower in impaired areas compared to reference sites (Kartikasari et al., 2013). Apart from being used in MMI development as diversity metrics, Shannon–Wiener is often used in biomonitoring and management of various water bodies (Djamai et al., 2019; Ghosh & Biswas, 2015; Latha & Thanga, 2010).
Macroinvertebrates express various feeding habits, which give them the ability to respond differently to disturbance. The percentage of filter feeders was part of NCN-LMMI after successful completion of all metrics screening. Filter feeding is common among macroinvertebrates like bivalves, using gills (ctenidia) (Ward et al., 2003), and Ephemeroptera, Trichoptera, and Diptera using setae, mouth brushes, and silk secretions, acting as sieves (Yee & Kaufman, 2019). The Per_filt_feeders was an increasing metric, probably due to the abundance of Culicidae, a pollution-tolerant taxa (Jeanrenraud et al., 2023). This metric was also considered by O’Toole et al. (2008) in testing the relationship between nutrients and selected benthic macroinvertebrates in lakes of Europe.
Precision through CCP and CV was estimated to prove whether NCN-LMMI reacts appropriately to environmental condition, and it has been revealed accordingly. The validation of metrics is one step to ensure the MMIs developed are indeed reflecting the expectations. During validation, some developers consider the same dataset used during MMI development (Alemneh et al., 2019; Zhang et al., 2019) or validate using separate dataset collected from the same sites (e.g., Huang et al. (2015) and Wondmagegn & Mengistou, (2023)). A similar validation method was applied by Chen et al. (2014), Krynak and Yates (2018), and Zhang et al. (2019).
Conclusion
NCN-LMMI is the first attempt as a lake-based multimetric index, using macroinvertebrates in North Central Nigeria and Nigeria at large to the best of our knowledge. It is anticipated to be helpful in the monitoring of lake water quality especially municipal lakes in this region, while contributing to addressing public health issues associated with pollution, such as waterborne diseases. Policy makers and water resource managers can use this tool to develop effective management strategies. Indeed, this can be a better alternative in water quality monitoring of lakes over traditional methods of physico-chemical parameters, owing to its cost-effectiveness, both in terms of finance and time.
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Abo-Taleb, H. A., El-Tabakh, M. A., & Hendy, D. M. (2023). Plankton and fish nutrition in African lakes. In M. El-Sheekh & H. E. Elsaied (Eds.), Lakes of Africa: Microbial diversity and sustainability (pp. 139–172). Elsevier. https://doi.org/10.1016/C2021-0-02119-2
Agodzo, B. D. (2013). Changes in the ecosystem services of the Volta Lake and their impacts on local livelihoods along its catchment areas in Ghana. Norwegian University of Life Sciences.
Aguiar, F. C., João, M., & Teresa, M. (2011). Choosing the best method for stream bioassessment using macrophyte communities: Indices and predictive models. Ecological Indicators, 11(2), 379–388. https://doi.org/10.1016/j.ecolind.2010.06.006
Akamagwuna, F. C., Mensah, P. K., Nnadozie, C. F., & Odume, O. N. (2019). Evaluating the responses of taxa in the orders Ephemeroptera, Plecoptera and Trichoptera (EPT) to sediment stress in the Tsitsa River and its tributaries, Eastern Cape, South Africa. Environmental Monitoring and Assessment, 191(11), 664. https://doi.org/10.1007/s10661-019-7846-9
Akamagwuna, F. C., Ntloko, P., Edegbene, A. O., & Odume, O. N. (2021). Are Ephemeroptera, Plecoptera and Trichoptera traits reliable indicators of semi-urban pollution in the Tsitsa River, Eastern Cape Province of South Africa? Environmental Monitoring and Assessment, 193(5), 309. https://doi.org/10.1007/s10661-021-09093-z
Akindele, E. O., & Liadi, A. A. (2013). Diversity and response of benthic macroinvertebrates to natural and induced environmental stresses in Aiba Stream, Iwo, Southwestern Nigeria. West African Journal of Applied Ecology, 22(1), 101–111.
Alemneh, T., Ambelu, A., Zaitchik, B. F., Bahrndorff, S., Mereta, S. T., & Pertoldi, C. (2019). A macroinvertebrate multi-metric index for Ethiopian highland streams. Hydrobiologia, 843(1), 125–141. https://doi.org/10.1007/s10750-019-04042-x
Anyanwu, E. D., Okorie, M. C., & Odo, S. N. (2019). Macroinvertebrates as bioindicators of water quality of effluent-receiving Ossah River, Umuahia, Southeast Nigeria. ZANCO Journal of Pure and Applied Sciences, 31(5), 9–17. https://doi.org/10.21271/zjpas.31.5.2
Arimoro, F. O., & James, H. (2008). Preliminary pictorial guide to the macroinvertebrates of Delta State Rivers. Southern Nigeria.
Arimoro, F. O., Odume, O. N., Uhunoma, S. I., & Edegbene, A. O. (2015). Anthropogenic impact on water chemistry and benthic macroinvertebrate associated changes in a southern Nigeria stream. Environmental Monitoring and Assessment, 187(2), 14. https://doi.org/10.1007/s10661-014-4251-2
Arman, N. Z., Salmiati, S., Said, M. I. M., & Aris, A. (2019). Development of macroinvertebrate-based multimetric index and establishment of biocriteria for river health assessment in Malaysia. Ecological Indicators, 104, 449–458. https://doi.org/10.1016/j.ecolind.2019.04.060
Asif, N., Malik, M., & Chaudhury, F. N. (2018). A review of on environmental pollution bioindicators. Pollution, 4(1), 111–118. https://doi.org/10.22059/poll.2017.237440.296
Assefa, W. W., Beneberu, G., Sitotaw, B., & Wondie, A. (2020). Biological monitoring of freshwater ecosystem health in Ethiopia: A review of current efforts, challenges, and future developments. Ethiopian Journal of Science and Technology, 13(3), 229–264. https://doi.org/10.4314/ejst.v13i3.5
Aura, C. M., Raburu, P. O., & Herrmann, J. (2010). A preliminary macroinvertebrate Index of Biotic Integrity for bioassessment of the Kipkaren and Sosiani Rivers, Nzoia River basin, Kenya. Lakes and Reservoirs: Research and Management, 15(2), 119–128. https://doi.org/10.1111/j.1440-1770.2010.00432.x
Auta, Y. I., Arimoro, F. O., Mohammed, A. Z., & Ayanwale, A. V. (2016). Monitoring seasonal changes in environmental variables and chironomid diversity at Shiroro Lake, Niger State, Nigeria. International Journal of Applied Biological Research, 7(1), 36–47.
Baptista, D. F., Buss, D. F., Egler, M., Giovanelli, A., Silveira, M. P., & Nessimian, J. L. (2007). A multimetric index based on benthic macroinvertebrates for evaluation of Atlantic Forest streams at Rio de Janeiro State, Brazil. Hydrobiologia, 575(1), 83–94. https://doi.org/10.1007/s10750-006-0286-x
Barbour, M. T., Gerritsen, J., Griffith, G. E., Frydenborg, R., Mccarron, E., White, J. S., & Bastian, M. L. (1996). A framework for biological criteria for Florida streams using benthic macroinvertebrates. Journal of the North American Benthological Society, 15(2), 185–211. https://doi.org/10.2307/1467948
Barbour, M. T., James, S. B., & James, K. R. (1995). The multimetric approach for establishing biocriteria and measuring biological condition. In W. S. Davis & T. P. Simon (Eds.), Biological assessment and criteria: Tools for water resource planning and decision making (pp. 63–80). Lewis Publishers.
Bêche, L. A., McElravy, E. P., & Resh, V. H. (2006). Long-term seasonal variation in the biological traits of benthic-macroinvertebrates in two Mediterranean-climate streams in California, U.S.A. Freshwater Biology, 51(1), 56–75. https://doi.org/10.1111/j.1365-2427.2005.01473.x
Beck, M. W., Hatch, L. K., Vondracek, B., & Valley, R. D. (2010). Development of a macrophyte-based index of biotic integrity for Minnesota lakes. Ecological Indicators, 10, 968–979. https://doi.org/10.1016/j.ecolind.2010.02.006
Bonada, N., Prat, N., Resh, V. H., & Statzner, B. (2006). Developments in aquatic insect biomonitoring: A comparative analysis of recent approaches. Annual Review of Entomology, 51, 495–523. https://doi.org/10.1146/annurev.ento.51.110104.151124
Brauns, M., Garcia, X. F., Pusch, M. T., & Walz, N. (2007). Eulittoral macroinvertebrate communities of lowland lakes: Discrimination among trophic states. Freshwater Biology, 52(6), 1022–1032. https://doi.org/10.1111/j.1365-2427.2007.01750.x
Brown, B. L. (2009). Using field experimentation to evaluate benthic macroinvertebrates recovery in highly disturbed Lake (Onondaga Lake, NY). State University of New York.
Burton, T. M., Uzarski, D. G., Gathman, J. P., Genet, J. A., Keas, B. E., & Stricker, C. (1999). Development of preliminary invertebrate index of biotic integrity for Lake Huron Coastal wetlands. Wetlands, 19, 869–882 papers://9813c380-4a5a-426e-ab3b-10e227534cf8/Paper/p660.
Camargo, J. A., Gonzalo, C., & Alonso, Á. (2011). Assessing trout farm pollution by biological metrics and indices based on aquatic macrophytes and benthic macroinvertebrates: A case study. Ecological Indicators, 11(3), 911–917. https://doi.org/10.1016/j.ecolind.2010.10.001
Carayon, D., Eulin-Garrigue, A., Vigouroux, R., & Delmas, F. (2020). A new multimetric index for the evaluation of water ecological quality of French Guiana streams based on benthic diatoms. Ecological Indicators, 113, 106248. https://doi.org/10.1016/j.ecolind.2020.106248
Chen, K., Hughes, R. M., Brito, J. G., Leal, C. G., Leitão, R. P., de Oliveira-Júnior, J. M. B., de Oliveira, V. C., Dias-Silva, K., Ferraz, S. F. B., Ferreira, J., Hamada, N., Juen, L., Nessimian, J., Pompeu, P. S., & Zuanon, J. (2017). A multi-assemblage, multi-metric biological condition index for eastern Amazonia streams. Ecological Indicators, 78, 48–61. https://doi.org/10.1016/j.ecolind.2017.03.003
Chen, K., Hughes, R. M., Xu, S., Zhang, J., Cai, D., & Wang, B. (2014). Evaluating performance of macroinvertebrate-based adjusted and unadjusted multi-metric indices (MMI) using multi-season and multi-year samples. Ecological Indicators, 36, 142–151. https://doi.org/10.1016/j.ecolind.2013.07.006
Chowdhury, G. W., Gallardo, B., & Aldridge, D. C. (2016). Development and testing of a biotic index to assess the ecological quality of lakes in Bangladesh. Hydrobiologia, 765, 55–69. https://doi.org/10.1007/s10750-015-2399-6
Clews, E., Low, E. W., Belle, C. C., Todd, P. A., Eikaas, H. S., & Ng, P. K. L. (2014). A pilot macroinvertebrate index of the water quality of Singapore’s reservoirs. Ecological Indicators, 38, 90–103. https://doi.org/10.1016/j.ecolind.2013.10.030
Cohen, A. S., Kaufman, L., & Ogutu-Ohwayo, R. (2019). Anthropogenic threats, impacts and conservation strategies in the African Great Lakes: A review. The Limnology, Climatology and Paleoclimatology of the East African Lakes, 13, 575–624.
Cornette, R., Gusev, O., Nakahara, Y., Shimura, S., Kikawada, T., & Okuda, T. (2015). Chironomid midges (Diptera, Chironomidae) show extremely small genome sizes. Zoological Science, 32(3), 248–254. https://doi.org/10.2108/zs140166
de Moor, I., Day, J., & de Moor, F. (2003). Guides to the freshwater invertebrates of Southern Africa (Vol. 9). Water Research Commission. https://doi.org/10.2989/ajas.2009.34.3.12.988
Ding, Y. F., Sun, S. J., Feng, J., Cui, P., Zhang, D., & Long, Z. Y. (2021). An assessment of lake ecology on the basis of the macrobenthos multi-metric index (MMI) in 11 lakes in the western region of Jilin, China. Water, 13(2), 235. https://doi.org/10.3390/w13020235
Djamai, S., Mimeche, F., Bensaci, E., & Oliva-Paterna, F. J. (2019). Diversity of macro-invertebrates in Lake Tonga (northeast Algeria). Biharean Biologist, 13(1), 8–11.
Dong, B., Geng, C., Cai, Y., & Ji, L. (2014). Aquatic Coleoptera response to environmental factors of freshwater ecosystems in Changbai Mountain, northeast China. Aquatic Ecosystem Health & Management, 17(2), 171–178. https://doi.org/10.1080/14634988.2014.910441
Edegbene, A. O., Akamagwuna, F. C., Odume, O. N., Arimoro, F. O., Ovie, T. T. E., Akumabor, E. C., Ogidiaka, E., Kaine, E. A., & Nwaka, K. H. (2022). A macroinvertebrate-based multimetric index for assessing ecological condition of forested stream sites draining Nigerian urbanizing landscapes. Sustainability, 14(18), 11289. https://doi.org/10.3390/su141811289
Edegbene, A. O., Elakhame, L. A., Arimoro, F. O., Osimen, E. C., & Odume, O. N. (2019). Development of macroinvertebrate multimetric index for ecological evaluation of a river in North Central Nigeria. Environmental Monitoring and Assessment, 191(5), 274. https://doi.org/10.1007/s10661-019-7438-8
Egwu, O. C., Akudo, O. C., Chinecherem, B. E., Victor, U. C., & Diskson, M. A. (2020). Health risk assessment of some toxic elements in aquatic bioindicator (Clarias gariepinus and Oreochromis niloticus) from Tagwai Reservoir Dam Minna, Niger State Nigeria. Scholars International Journal of Biochemistry, 3(02), 12–21. https://doi.org/10.36348/sijb.2020.v03i02.001
Feio, M. J., Hughes, R. M., Serra, S. R. Q., Nichols, S. J., Kefford, B. J., Lintermans, M., Robinson, W., Odume, O. N., Callisto, M., Macedo, D. R., Harding, J. S., Yates, A. G., Monk, W., Nakamura, K., Mori, T., Sueyoshi, M., Mercado-Silva, N., Chen, K., Baek, M. J., et al. (2023). Fish and macroinvertebrate assemblages reveal extensive degradation of the world’s rivers. Global Change Biology, 29(2), 355–374. https://doi.org/10.1111/gcb.16439
Fontanetti, C. S., da Souza, T. S., & Christofoletti, C. A. (2012). The role of biomonitoring in the quality assessment of water. In C. Bibilio, O. Hensel, & J. F. Selbach (Eds.), Sustainable water management in the tropics and subtropics and cases study in Brazil (pp. 975–1005). Fundação Universidade Federal do Pampa, UNIKASSEL, PGCult-UFMA.
Gabriels, W., Lock, K., De Pauw, N., & Goethals, P. L. M. (2010). Multimetric Macroinvertebrate Index Flanders (MMIF) for biological assessment of rivers and lakes in Flanders (Belgium). Limnologica, 40(3), 199–207. https://doi.org/10.1016/j.limno.2009.10.001
Gezie, A., Anteneh, W., Dejen, E., & Mereta, S. T. (2017). Effects of human-induced environmental changes on benthic macroinvertebrate assemblages of wetlands in Lake Tana Watershed, Northwest Ethiopia. Environmental Monitoring and Assessment, 189(4), 152. https://doi.org/10.1007/s10661-017-5853-2
Ghosh, D., & Biswas, J. K. (2015). Macroinvertebrate diversity indices: A quantitative bioassessment of ecological health status of an oxbow lake in Eastern India. Journal of Advances in Environmental Health Research, 3(2), 78–90.
Gilcreas, F. W. (1966). Standard methods for the examination of water and waste water. American Journal of Public Health and the Nation’s Health, 56(3), 387–388. https://doi.org/10.2105/AJPH.56.3.387
Gooderham, J., & Tsyrlin, E. (2002). A guide to the freshwater macroinvertebrates of template Australia: The Waterbug book. CSIRO Publishing.
Gwenzi, W., & Chaukura, N. (2018). Organic contaminants in African aquatic systems: Current knowledge, health risks, and future research directions. Science of the Total Environment, 619, 1493–1514. https://doi.org/10.1016/j.scitotenv.2017.11.121
Haugerud, N. J. (2006). Macroinvertebrate index of biotic integrity for the Lake Agassiz plain ecoregion (48) of North Dakota. North Dakota Department of Health Division of Water Quality. Bismarck, North Dakota
Hawkins, C. P., Cao, Y., & Roper, B. (2010). Method of predicting reference condition biota affects the performance and interpretation of ecological indices. Freshwater Biology, 55(5), 1066–1085. https://doi.org/10.1111/j.1365-2427.2009.02357.x
Hering, D., Feld, C. K., Moog, O., & Ofenbo, T. (2006). Cook book for the development of a multimetric index for biological condition of aquatic ecosystems: Experiences from the European AQEM and STAR projects and related initiatives. Hydrobiologia, 566, 311–324. https://doi.org/10.1007/978-1-4020-5493-8
Huang, Q. I., Gao, J., Cai, Y., Yin, H., Gao, Y., Zhao, J., Liu, L., & Huang, J. (2015). Development and application of benthic macroinvertebrate-based multimetric indices for the assessment of streams and rivers in the Taihu Basin, China. Ecological Indicators, 48, 649–659. https://doi.org/10.1016/j.ecolind.2014.09.014
Ibrahim, S., Umar, F., Yakubu, U., Adama, B., & Olubobokun, A. E. (2019). Assessment of some physico-chemical parameters of Tagwai Reservoir in Minna, Niger State, Nigeria. In Proceedings of the 2nd Conference and Annual General Meeting: Association of Nigerian Fisheries Scientists.
Jeanrenraud, A. C., Letinić, B. D., Mollett, J., Brooke, B. D., & Oliver, S. V. (2023). The effect of pollution on the competitive dynamics of Anopheles arabiensis Patton, 1905 and Culex quinquefasciatus Say, 1823 (Diptera: Culicidae). African Entomology, 31, e10656. https://doi.org/10.17159/2254-8854/2023/a10656
Johnson, R. K. (2003). Development of a prediction system for lake stony-bottom littoral macroinvertebrate communities. Archiv Fur Hydrobiologie, 158(4), 517–540. https://doi.org/10.1127/0003-9136/2003/0158-0517
Jones, C., Somers, K. M., Craig, B., & Reynoldson, T. (2004). Ontario Benthos Biomonitoring Network (OBBN) protocol manual; Version 1.0 (Vol. 108). Dorset Environmental Science Centre, Ontario Ministry of Environment.
Jun, Y. C., Won, D. H., Lee, S. H., Kong, D. S., & Hwang, S. J. (2012). A multimetric benthic macroinvertebrate index for the assessment of stream biotic integrity in Korea. International Journal of Environmental Research and Public Health, 9(10), 3599–3628. https://doi.org/10.3390/ijerph9103599
Jyväsjärvi, J., Aroviita, J., & Hämäläinen, H. (2012). Performance of profundal macroinvertebrate assessment in boreal lakes depends on lake depth. Fundamental and Applied Limnology, 180(2), 91–100. https://doi.org/10.1127/1863-9135/2012/0205
Jyväsjärvi, J., Nyblom, J., & Hämäläinen, H. (2010). Palaeolimnological validation of estimated reference values for a lake profundal macroinvertebrate metric (Benthic Quality Index). Journal of Paleolimnology, 44(1), 253–264. https://doi.org/10.1007/s10933-009-9401-0
Karmakar, P., Pal, S., & Mishra, M. (2021). Arthropods: An important bio-indicator to decipher the health of the water of South Asian rivers. In B. C. Patra, P. K. Shit, G. S. Bhunia, & M. Bhattacharya (Eds.), River health and ecology in South Asia: Pollution, restoration, and conservation (pp. 9–38). Springer Nature. https://doi.org/10.1007/978-3-030-83553-8_2
Karr, J. R. (1981). Assessment of biotic integrity using fish communities. Fisheries, 6(6), 21–27.
Kartikasari, D., Retnaningdyah, C., & Arisoesilaningsih, E. (2013). Application of water quality and ecology indices of benthic macroinvertebrate to evaluate water quality of tertiary irrigation in Malang District. Journal of Tropical Life Science, 3(3), 193–201. https://doi.org/10.11594/jtls.03.03.09
Keizer-Vlek, H. E. (2014). Fifty shades of grey: Variability in metric-based assessment of surface waters using macroinvertebrates. In Alterra Scientific Contributions 44, Alterra, part of Wageningen UR. Wageningen.
Kolawole, G. T., Omotoyo, O. L., Adedeji, E. O., Chikezie, J., & Attahiru, H. (2020). Environmental management and recreational facilities usage: A study of Kainji Lake National Park. Technium Social Sciences Journal, 7(29), 276. https://doi.org/10.37745/ejhtr/vol8.no2.pp18-24.2020
Kramer, D. L., & Chapman, C. R. (1999). Implication of fish home range size and relocation for marine reserve function. Environmental Biology of Fishes, 55, 65–79.
Krynak, E. M., & Yates, A. G. (2018). Benthic invertebrate taxonomic and trait associations with land use in an intensively managed watershed: Implications for indicator identification. Ecological Indicators, 93, 1050–1059. https://doi.org/10.1016/j.ecolind.2018.06.002
Kumari, D., & Paul, D. K. (2021). Assessing the role of bioindicators in freshwater ecosystem. Journal of Interdisciplinary Cycle Research, 7(9), 58–74.
Latha, C., & Thanga, G. S. V. (2010). Macroinvertebrate diversity of Veli and Kadinamkulam lakes, South Kerala, India. Journal of Environmental Biology, 31, 543–547.
Lopez-Lopez, E., & Sedeno-Diaz, J. E. (2015). Biological indicators of water quality: The role of fish and macroinvertebrates as indicators of water quality. In O. Hanninen & R. H. Armon (Eds.), Environmental indicators. Springer Science and Business Media. https://doi.org/10.1007/978-94-017-9499-2
Macedo, D. R., Hughes, R. M., Ferreira, W. R., Firmiano, K. R., Silva, D. R. O., Ligeiro, R., Kaufmann, P. R., & Callisto, M. (2016). Development of a benthic macroinvertebrate multimetric index (MMI) for Neotropical Savanna headwater streams. Ecological Indicators, 64, 132–141. https://doi.org/10.1016/j.ecolind.2015.12.019
Mahaut, M., Basuyaux, O., Baudiniere, E., Chataignier, C., Pain, J., & Caplat, C. (2013). The porifera Hymeniacidon perlevis (Montagu, 1818) as a bioindicator for water quality monitoring. Environmental Science and Pollution Research, 20, 2984–2992. https://doi.org/10.1007/s11356-012-1211-7
McFarland, B., Carse, F., & Sandin, L. (2010). Littoral macroinvertebrates as indicators of lake acidification within the UK. Aquatic Conservation: Marine and Freshwater Ecosystems, 20, S105–S116. https://doi.org/10.1002/aqc.1064
McGoff, E., Aroviita, J., Pilotto, F., Miler, O., Solimini, A. G., Porst, G., Jurca, T., Donohue, L., & Sandin, L. (2013). Assessing the relationship between the Lake Habitat Survey and littoral macroinvertebrate communities in European lakes. Ecological Indicators, 25, 205–214. https://doi.org/10.1016/j.ecolind.2012.09.018
Menetrey, N., Oertli, B., & Lachavanne, J. B. (2011). The CIEPT: A macroinvertebrate-based multimetric index for assessing the ecological quality of Swiss lowland ponds. Ecological Indicators, 11(2), 590–600. https://doi.org/10.1016/j.ecolind.2010.08.005
Menetrey, N., Oertli, B., Sartori, M., Wagner, A., & Lachavanne, J. B. (2008). Eutrophication: Are mayflies (Ephemeroptera) good bioindicators for ponds ? Hydrobiologia, 597, 125–126. https://doi.org/10.1007/s10750-007-9223-x
Menezes, S., Baird, D. J., & Soares, A. M. V. M. (2010). Beyond taxonomy: A review of macroinvertebrate trait-based community descriptors as tools for freshwater biomonitoring. Journal of Applied Ecology, 47, 711–719. https://doi.org/10.1111/j.1365-2664.2010.01819.x
Miler, O., Porst, G., McGoff, E., Pilotto, F., Donohue, L., Jurca, T., Solimini, A., Sandin, L., Irvine, K., Aroviita, J., Clarke, R., & Pusch, M. T. (2013). Morphological alterations of lake shores in Europe: A multimetric ecological assessment approach using benthic macroinvertebrates. Ecological Indicators, 34, 398–410. https://doi.org/10.1016/j.ecolind.2013.06.002
Mohammed, I., Wondie, A., & Mengist, M. (2022). The environmental quality and macroinvertebrate community structures of wetlands found in the Lake Tana Watershed, Ethiopia. Journal of the Cameroon Aademy of Sciences, 17(3), 183–203.
Moiseenko, T. I., Sharov, A. N., Vandish, O. I., Kudryavtseva, L. P., Gashkina, N. A., & Rose, C. (2009). Long-term modification of Arctic lake ecosystems: Reference condition, degradation under toxic impacts and recovery (case study Imandra Lakes, Russia). Limnologica, 39(1), 1–13. https://doi.org/10.1016/j.limno.2008.03.003
Moog, O., Schmutz, S., & Schwarzinger, I. (2018). Biomonitoring and bioassessment. In S. Schmutz & J. Sendzimir (Eds.), Riverine ecosystem management: Science for governing towards a sustainable future (pp. 371–390). Springer Nature. https://doi.org/10.1007/978-3-319-73250-3_19
Musa, J. J., & Saba, V. (2011). Water transfer methods in response to water demand for agricultural practice at Chanchaga River and Tagwai dam in Niger State, Nigeria. Journal of Environmental Issues and Agriculture in Developing Countries, 3(3), 84–89.
Nakirya, D., Okot-Okumu, J., & Muyodi, F. J. (2015). Microbial safety assessment of recreation water at Lake Nabugabo, Uganda. African Journal of Environmental Science and Technology, 9(10), 773–782. https://doi.org/10.5897/ajest2015.1979
Ndatimana, G., Nantege, D., & Arimoro, F. O. (2023). A review of the application of the macroinvertebrate-based multimetric index (MMI) for water quality monitoring in lakes. Environmental Science and Pollution Research, 30, 73098–73115. https://doi.org/10.1007/s11356-023-27559-0
Newbold, T., Bentley, L. F., Hill, S. L. L., Edgar, M. J., Horton, M., Su, G., Şekercioğlu, Ç. H., Collen, B., Purvis, A., & Newbold, T. (2020). Global effects of land use on biodiversity differ among functional groups. Functional Ecology, 34(3), 684–693. https://doi.org/10.1111/1365-2435.13500
Ntitslidou, C., Rossaro, B., Lazaridou, M., & Bobori, D. C. (2021). What drives benthic macroinvertebrate dispersal in different lake substrata? The case of three Mediterranean lakes. Aquatic Ecology, 55(3), 1033–1050. https://doi.org/10.1007/s10452-021-09880-3
O’Toole, C., Donohue, I., Moe, S. J., & Irvine, K. (2008). Nutrient optima and tolerances of benthic invertebrates, the effects of taxonomic resolution and testing of selected metrics in lakes using an extensive European data base. Aquatic Ecology, 42(2), 277–291. https://doi.org/10.1007/s10452-008-9185-8
Obinnaa, I. B., & Ebere, E. C. (2019). A review: Water pollution by heavy metal and organic pollutants: Brief review of sources, effects and progress on remediation with aquatic plants. Analytical Methods in Environmental Chemistry Journal, 2(3), 5–38. https://doi.org/10.24200/amecj.v2.i03.66
Odountan, O. H., de Bisthoven, L. J., Abou, Y., & Eggermont, H. (2018). Biomonitoring of lakes using macroinvertebrates: Recommended indices and metrics for use in West Africa and developing countries. Hydrobiologia, 826(1), 1–23. https://doi.org/10.1007/s10750-018-3745-2
Ogutu-Ohwayo, R., Natugonza, V., Musinguzi, L., Olokotum, M., & Naigaga, S. (2016). Implications of climate variability and change for African lake ecosystems, fisheries productivity, and livelihoods. Journal of Great Lakes Research, 42(3), 498–510. https://doi.org/10.1016/j.jglr.2016.03.004
Oladosu, S. O., Ojigi, L. M., Aturuocha, V. E., Anekwe, C. O., & Tanko, R. (2019). An investigative study on the volume of sediment accumulation in Tagwai dam reservoir using bathymetric and geostatistical analysis techniques. SN Applied Sciences, 1(5), 1–13. https://doi.org/10.1007/s42452-019-0393-8
Onywere, S. M., Mironga, J. M., & Simiyu, I. (2012). Use of remote sensing data in evaluating the extent of anthropogenic activities and their impact on Lake Naivasha, Kenya. The Open Environmental Engineering Journal, 5, 9–18.
Parmar, T. K., Rawtani, D., & Agrawal, Y. K. (2016). Bioindicators: The natural indicator of environmental pollution. Frontiers in Life Science, 9(2), 110–118. https://doi.org/10.1080/21553769.2016.1162753
Penning, E. W., Dudley, B., Mjelde, M., Hellsten, S., Jenica, H., Kolada, A., den Berg, V. M., Poikane, S., Phillips, G., Willby, N., & Ecke, F. (2008). Using aquatic macrophyte community indices to define the ecological status of European lakes. Aquatic Ecology, 42, 253–264. https://doi.org/10.1007/s10452-008-9183-x
Poikane, S., van den Berg, M., Hellsten, S., de Hoyos, C., Ortiz-Casas, J., Pall, K., Portielje, R., Phillips, G., Solheim, A. L., Tierney, D., & Wolfram, G. (2011). Lake ecological assessment systems and intercalibration for the European Water Framework Directive: Aims achievements and further challenges. Procedia Environmental Sciences, 9, 153–168. https://doi.org/10.1016/j.proenv.2011.11.024
Poulíčková, A., Duchoslav, M., & Dokulil, M. (2004). Littoral diatom assemblages as bioindicators of lake trophic status: A case study from perialpine lakes in Austria. European Journal of Phycology, 39(2), 143–152. https://doi.org/10.1080/0967026042000201876
Puglisi, M. V. (2021). Effects of anthropogenic pollution on gilled snail abundance in Stroubles Creek in Montgomery County, .
RCore Team. (2022). R: A language and environment for statistical computing. R Foundation for Statistical. https://www.r-project.org/
Reavie, E. D., Kireta, A. R., Kingston, J. C., Sgro, G. V., Danz, N. P., Axler, R. P., & Hollenhorst, T. P. (2008). Comparison of simple and multimetric diatom-based indices for Great Lakes coastline disturbance. Journal of Phycology, 44(3), 787–802. https://doi.org/10.1111/j.1529-8817.2008.00523.x
Redwan, M., & Elhaddad, E. (2022). Heavy metal pollution in Manzala Lake sediments, Egypt: Sources, variability, and assessment. Environmental Monitoring and Assessment, 194(6), 436. https://doi.org/10.1007/s10661-022-10081-0
Riato, L., & Leira, M. (2020). Heterogeneity of epiphytic diatoms in shallow lakes: Implications for lake monitoring. Ecological Indicators, 111, 105988.
Ruse, L. (2010). Classification of nutrient impact on lakes using the chironomid pupal exuvial technique. Ecological Indicators, 10(3), 594–601. https://doi.org/10.1016/j.ecolind.2009.10.002
Salomonsson, S., & Thoresson, H. (2010). Windmill driven water pump for small-scale irrigation and domestic use in Lake Victoria Basin. University of Skovde http://www.diva-portal.org/smash/get/diva2:327897/FULLTEXT01.pdfBachelor
Schartau, A. K., Moe, S. J., Sandin, L., McFarland, B., & Raddum, G. G. (2008). Macroinvertebrate indicators of lake acidification: Analysis of monitoring data from UK, Norway and Sweden. Aquatic Ecology, 42(2), 293–305. https://doi.org/10.1007/s10452-008-9186-7
Schoolmaster, D. R., Grace, J. B., & Schweiger, E. W. (2012). A general theory of multimetric indices and their properties. Methods in Ecology and Evolution, 3(4), 773–781. https://doi.org/10.1111/j.2041-210X.2012.00200.x
Schowe, K. A., & Harding, J. S. (2014). Development of two diatom-based indices: A biotic and a multimetric index for assessing mine impacts in New Zealand streams. New Zealand Journal of Marine and Freshwater Research, 48(2), 163–176. https://doi.org/10.1080/00288330.2013.852113
Shah, R. D. T., Shah, D. N., & Nesemann, H. (2011). Development of a macroinvertebrate-based Nepal Lake Biotic Index (NLBI): An applied method for assessing the ecological quality of lakes and reservoirs in Nepal. International Journal of Hydrology Science and Technology, 1(1–2), 125–146. https://doi.org/10.1504/IJHST.2011.040744
Shishaye, H. A. (2018). Diagnosing the suitability of lake water for domestic and agricultural uses: A case study in Eastern Ethiopia. East African Journal of Sciences, 12(2), 101–110.
Šidagyte, E., Višinskiene, G., & Arbačiauskas, K. (2013). Macroinvertebrate metrics and their integration for assessing the ecological status and biocontamination of Lithuanian lakes. Limnologica, 43(4), 308–318. https://doi.org/10.1016/j.limno.2013.01.003
Solimini, A. G., Bazzanti, M., Ruggiero, A., & Carchini, G. (2008). Developing a multimetric index of ecological integrity based on macroinvertebrates of mountain ponds in central Italy. Hydrobiologia, 597(1), 109–123. https://doi.org/10.1007/s10750-007-9226-7
Sreeja, J. (2018). Biomonitoring of Paravur Lake in Kerala using macro-invertebrates. In V. Singh, S. Yadav, & R. Yadava (Eds.), Environmental pollution (Vol. 77). Springer.
Stoddard, J. L., Herlihy, A. T., Peck, D. V., Hughes, R. M., Whittier, T. R., & Tarquinio, E. (2008). A process for creating multimetric indices for large-scale aquatic surveys. Journal of the North American Benthological Society, 27(4), 878–891. https://doi.org/10.1899/08-053.1
Stoddard, J. L., Larsen, D. P., Hawkins, C. P., Johnson, R. K., & Norris, R. H. (2006). Setting expectations for the ecological condition of streams: The concept of reference condition. Ecological Applications, 16(4), 1267–1276. https://doi.org/10.1890/1051-0761(2006)016[1267:SEFTEC]2.0.CO;2
Tachet, H., Richoux, P., Bournaud, M., & Usseglio-Polatera, P. (2010). Usseglio-Polatera, P., & Invertébrés d’eau Douce: Systématique, biologie, écologie (Vol. 15). CNRS Editions.
Taiyun, W., & Simko, V. (2021). R package “corrplot”: Visualization of a Correlation Matrix (Version 0.92). https://github.com/taiyun/corrplot
Taowu, M., Qinghui, H., Hai, W., Zijian, W., Chunxia, W., & Shengbiao, H. (2008). Selection of benthic macroinvertebrate-based multimetrics and preliminary establishment of biocriteria for the bioassessment of the water quality of Taihu Lake, China. Acta Ecologica Sinica, 28(3), 1192–1200. https://doi.org/10.1016/S1872-2032(08)60038-4
Thorp, J. H., & Rogers, C. D. (2011). Freshwater invertebrates of North America. Academic Press.
Timm, H., & Möls, T. (2012). Littoral macroinvertebrates in Estonian lowland lakes: The effects of habitat, season, eutrophication and land use on some metrics of biological quality. Fundamental and Applied Limnology, 180(2), 145–156. https://doi.org/10.1127/1863-9135/2012/0203
Touron-Poncet, H., Bernadet, C., Compin, A., Nicolas, B., & Céréghino, R. (2014). Implementing the water Framework Directive in overseas Europe: A multimetric macroinvertebrate index for river bioassessment in Caribbean islands. Limnologica, 1–38. https://doi.org/10.1016/j.limno.2014.04.002
Trigal, C., García-Criado, F., & Fernández-Aláez, C. (2006). Among-habitat and temporal variability of selected macroinvertebrate based metrics in a Mediterranean shallow lake (NW Spain). Hydrobiologia, 563(1), 371–384. https://doi.org/10.1007/s10750-006-0031-5
Uherek, C. B., & Gouveia, F. B. P. (2014). Biological monitoring using macroinvertebrates as bioindicators of water quality of Maroaga Stream in the Maroaga Cave System, Presidente Figueiredo, Amazon, Brazil. International Journal of Ecology, 2014, 308149. https://doi.org/10.1155/2014/308149
Utami, S., & Fajar, E. (2022). Exploration of aquatic macroinvertebrates as a bioindicator of water quality in Nogosari River , Pacitan Regency. 2nd International Conference on Education and Technology, 630, 91–98.
Vaissie, P., Monge, A., & Husson, F. (2021). Factoshiny: Perform factorial analysis from “FactoMineR” with a Shiny application. R Package Version, 2, 4 https://cran.r-project.org/package=Factoshiny
Wang, X., Zheng, B., Liu, L., & Wang, L. (2015). Development and evaluation of the lake multi-biotic integrity index for Dongting Lake, China. Journal of Limnology, 74(3), 1–5. https://doi.org/10.4081/jlimnol.2015.1186
Ward, J. E., Levinton, J. S., & Shumway, S. E. (2003). Influence of diet on pre-ingestive particle processing in bivalves I: Transport velocities on the ctenidium. Journal of Experimental Marine Biology and Ecology, 293(2), 129–149. https://doi.org/10.1016/S0022-0981(03)00218-1
Weir, S. M., & Salice, C. J. (2012). High tolerance to abiotic stressors and invasion success of the slow growing freshwater snail, Melanoides tuberculatus. Biological Invasion, 14, 385–394. https://doi.org/10.1007/s10530-011-0084-x
Wesolek, B. E., Genrich, E. K., Gunn, J. M., & Somers, K. M. (2010). Use of littoral benthic invertebrates to assess factors affecting biological recovery of acid- and metal-damaged lakes. Journal of the North American Benthological Society, 29(2), 572–585. https://doi.org/10.1899/09-123.1
Whittier, T. R., Stoddard, J. L., Larsen, D. P., & Herlihy, A. T. (2007). Selecting reference sites for stream biological assessments: Best professional judgment or objective criteria. Journal of the North American Benthological Society, 26(2), 349–360. https://doi.org/10.1899/0887-3593(2007)26[349:SRSFSB]2.0.CO;2
Wondmagegn, T., & Mengistou, S. (2020). Effects of anthropogenic activities on macroinvertebrate assemblages in the littoral zone of Lake Hawassa, a tropical rift valley lake in Ethiopia. Lakes & Reservoirs: Research & Management, 25(1), 61–71. https://doi.org/10.1111/lre.12303
Wondmagegn, T., & Mengistou, S. (2023). Development of macroinvertebrate based multimetric index for ecological health monitoring in Lake Hawassa, Ethiopia. Environmental and Sustainability Indicators, 18, 100242. https://doi.org/10.1016/j.indic.2023.100242
Wondmagegn, T., Mengistou, S., & Barker, P. A. (2019). Testing of the applicability of European diatom indices in the tropical rift valley lake, Lake Hawassa, in Ethiopia. African Journal of Aquatic Science, 44(3), 209–217. https://doi.org/10.2989/16085914.2019.1645640
Xu, M., Wang, Z., Duan, X., & Pan, B. (2014). Effects of pollution on macroinvertebrates and water quality. Hydrobiologia, 729, 247–259. https://doi.org/10.1007/s10750-013-1504-y
Yee, D. A., & Kaufman, M. G. (2019). Suspension and filter feeding in aquatic insects. In D. A. Yee & M. G. Kaufman (Eds.), Insect mouthparts: Form, function, development and performance (Vol. 5, pp. 101–125). Springer.
Yonnana, E., Yamta, S., Kaigama, I., & Bedeson, A. (2017). Assessment of water quality for Goro Dong (Lake) and its suitability for consumption and domestic use by the immediate lake communities in Numan, Adamawa State Nigeria. Journal of Geography, Environment and Earth Science International, 12(4), 1–8. https://doi.org/10.9734/jgeesi/2017/37517
Young, S., Yang, H.-N., Huang, D.-J., Liu, S.-M., Huang, Y.-H., Chiang, C.-T., & Liu, J.-W. (2014). Using benthic macroinvertebrate and fish communities as bioindicators of the Tanshui river basin around the greater Taipei area - Multivariate analysis of spatial variation related to levels of water pollution. International Journal of Environmental Research and Public Health, 11(7), 7116–7143. https://doi.org/10.3390/ijerph110707116
Yu, P., You, Q., Pang, W., Cao, Y., Bi, Y., & Wang, Q. (2021). Development of a periphytic diatom-based comprehensive diatom index for assessing the trophic status of lakes in the lower reaches of the Yangtze River, China. Water, 13(24), 3570. https://doi.org/10.3390/w13243570
Zamdayu, W. J., & Japhet, M. (2021). Evaluation of soil and water quality of Lake Gerio irrigation project of Upper Benue River Basin Development Authority Yola, Nigeria. International Research Journal of Advanced Engineering and Science, 6(1), 266–268.
Zhang, Y., Cheng, L., Kong, M., Li, W., Gong, Z., Zhang, L., Wang, X., Cai, Y., & Li, K. (2019). Utility of a macroinvertebrate-based multimetric index in subtropical shallow lakes. Ecological Indicators, 106, 105527. https://doi.org/10.1016/j.ecolind.2019.105527
Zhang, Y., Leung, J., Zhang, Y., Cai, Y., Zhang, Z., & Li, K. (2021). Agricultural activities compromise ecosystem health and functioning of rivers: Insights from multivariate and multimetric analyses of macroinvertebrate assemblages. Environmental Pollution, 275, 116655.
Acknowledgements
We thank the Intra-African Academic Mobility Scheme of the European Union which funded this research through the African Water Resources Mobility Network (AWaRMN). We acknowledge members of Hydrobiology Unit of the Federal University of Technology, Minna, for their contribution to laboratory works.
Funding
This paper emanates from the African Water Resources Mobility Network (AWaRMN) Grant No. 2019-973/004-001, which has received funding from the Intra-African Academic Mobility Scheme of the European Union.
Author information
Authors and Affiliations
Contributions
Gilbert Ndatimana: conceptualization and implementation, data collection, formal analysis, visualization, writing the original draft, reviewing, and editing the manuscript; Francis O. Arimoro: conceptualization, supervision, reviewing the manuscript; Victoria I. Chukwuemeka: conceptualization, supervision, reviewing the manuscript; Fulbert AGJ Assie: investigation, and reviewing manuscript; Simon Action: investigation, and reviewing the manuscript; Diana Nantege: investigation, reviewing and editing the manuscript. The authors approved the version to be published.
Corresponding author
Ethics declarations
Ethical approval
Not applicable
Consent to participate
Not applicable
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ndatimana, G., Arimoro, F.O., Chukwuemeka, V.I. et al. Development of lake macroinvertebrate-based multimetric index for monitoring ecological health in North Central Nigeria. Environ Monit Assess 195, 1429 (2023). https://doi.org/10.1007/s10661-023-12036-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10661-023-12036-5