Abstract
Historically, the Brazilian coast has been impacted by urban, industrial, and port activities that have increased the input of chemical contaminants, such as heavy metals, to the ecosystem. The Paranaguá estuarine complex (PEC), Cananéia-Iguape estuarine-lagoon complex (CIELC), and Santos-São Vicente estuarine complex (SSVEC) (S-SE Brazil) are surrounded by urbanized cities and port areas characterized by various anthropogenic discharges comprising several potential pollutants, including heavy metals. Concerns about such contamination are paramount because these estuaries are important for traditional fishing communities and are categorized as World Heritage sites and biodiversity hotspots by UNESCO. In this study, metals (Cu, Cr, Ni, Pb, Zn, and Hg) and metalloids (As and Se) known to affect the health of marine life were evaluated in regional fishes. Muscle and liver tissues from three demersal teleosts (Stellifer rastrifer, Paralonchurus brasiliensis, and Isopisthus parvipinnis) were analyzed by inductively coupled plasma optical emission spectrometry (ICP-OES), with a coupled vapor generated accessory (VGA). Irrespective of species, metal bioconcentration was significantly greater in fishes from the PEC and CIELC, which had higher As, Cu, and Zn concentrations, while Se levels were higher in fish from the PEC and SSVEC estuaries. Seasonality, fish species and maturation stage affected the accumulation of metals. Some metal levels, including As, Cr, Pb, and Se in all species across all estuaries, and Zn in the PEC, exceeded the maximum permitted level for seafood and might present a risk for daily human consumption. The results provide reference points for existing chemical contamination and should be used to guide monitoring programs and the sustainable development of these coastal regions, within a broader objective of maintaining public health.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Tropical and subtropical estuaries are highly productive ecosystems that have an important role in human history and the life histories of aquatic organisms (Chapman and Wang 2001). Nevertheless, in the last century, these regions have been highly modified by deleterious anthropogenic activities (e.g., as urban and industrial developments) that contaminate aquatic ecosystems (Salgado-Ramírez et al. 2017).
In south and southeastern Brazil, the Paranaguá estuarine complex (PEC), Cananéia-Iguape estuarine-lagoon complex (CIELC), and Santos-São Vicente estuarine complex (SSVEC) are characterized by different levels of contaminants throughout their trophic chains (Marone et al. 2005; Azevedo et al. 2009). These estuaries and their coastal zones comprise densely population areas, where industrial, agricultural, and port activities coexist with the largest Atlantic Forest remnants and mangroves supporting high fisheries productivity and biodiversity. Such characteristics have qualified the areas as World Heritage sites and biodiversity hotspots (UNESCO 1999). Therefore, understanding and evaluating anthropogenic impacts in these environments is crucial, not only for their conservation but also sustainable development and human wellbeing (Chapman and Wang 2001).
Metals are among the most frequently detected pollutants in estuaries and represent a problem owing to their toxicity, persistence and stability in the environment, and accumulation capacity in biota, including fish (Islam and Tanaka 2004). High levels of metals in water and sediments do not evoke direct toxicological risks to trophic chain. Rather, risk only occurs when such elements are bioavailable. Metal bioconcentration includes complex processes that are regulated by exogenous (i.e., abiotic) and endogenous (i.e., biotic) factors (Moiseenko and Kudryavtseva 2001). Exogenous factors are reflected in environmental parameters such as metal bioavailability, and the temperature and salinity of the aquatic environment, while endogenous factors encompass the age, sex, body size (i.e., length or weight), physiological status, feeding habits, habitats, and migrations of species (Moiseenko and Kudryavtseva 2001; Al-Yousuf et al. 2000; Angeli et al. 2013).
Fish are considered good indicators of environmental quality, mostly because (i) their taxonomies and life cycles usually are sufficiently known, (ii) their taxonomic groups occupy various trophic levels and habitats, and (iii) many are economically important (Gusso-Choueri et al. 2018; Santana et al. 2017; Storelli 2008). Fishes are consumers that generally are located at higher trophic levels, and for this reason, they may accumulate large amounts of metals (Jaric et al. 2011). Moreover, fish are among those aquatic organisms most susceptible to toxic substances present in the water (Jaric et al. 2011). Therefore, because they are sensitive to short-term environmental changes (e.g., dissolved oxygen levels, dissolved and solid-phase spills), have a relatively long life (among many aquatic organisms), and can accumulate metals, fish frequently are used for evaluating acute and/or chronic pollution (Kasper et al. 2007; Vernenberg 1981). Using fish in monitoring programs informs the environmental quality and the contamination status of animals consumed by higher trophic level species, including humans. Consequently, evaluating fish health condition is a precursor to ecological, socioeconomic, and human health risk assessments related to fish consumption (Azevedo et al. 2009).
Currently, there remains a dearth of knowledge regarding metal levels in the tissues of the most commonly harvested fish species, including Stellifer rastrifer (cangoá, Bloch, 1790), Isopisthus parvipinnis (pescada tortinha, Cuvier, 1830), and Paralonchurus brasiliensis (maria-luiza, Steindachner, 1875) in the SSVEC, CIELC, and PEC. All three species are demersal, inhabiting regional estuarine and coastal environments, are among the species identified in fishing landings in the study areas, and serve as food for local fishing community and prey for top predators in the trophic web (Branco et al. 2005; Mendonça and Miranda 2008; Queiroz et al. 2006). Consequently these species are vectors of the metal contamination to consuming animals and humans.
Considering the above, the objective of this study was to first quantify metal and metalloid concentrations (As, Cu, Cr, Ni, Pb, Se, Zn, and Hg) among key fish species (S. rastrifer, I. parvipinnis, and P. brasiliensis) inhabiting the SSVEC, CIELC, and PEC. These data were then used to address the hypotheses that any regional bioconcentration of metals is (i) affected by various exogenous or endogenous factors, (ii) related to anthropic pressures within each estuary, and (iii) likely to pose risk to human health via consumption (via contamination assessment using the target hazard quotient (THQ)). Ultimately, addressing these hypotheses provides the required baseline information that will contribute to future monitoring programs and coastal management guidelines.
Material and methods
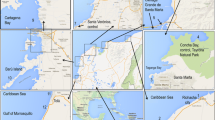
The study area comprises the stated three estuarine systems located along the southeastern and southern Brazilian coast (Fig. 1). The SSVEC is formed by the Santos and São Vicente bays and is on the central coast of São Paulo state, with a total area of 2.4 km2 and an extension of 40 km. Surrounded by the cities of Santos, São Vicente, and Cubatão, the total population is approximately 870,565 inhabitants (IBGE 2010). Santos Bay is highly urbanized. Further, the region is a petrochemical and metallurgical industrial complex (Cubatão Industrial Complex), and where the port of Santos is placed, which is the largest commercial port in Latin America (Azevedo et al. 2009; CETESB 2008).
The CIELC is located to the south on the coast of São Paulo state and has an extension of ~ 110.0 km2 (Besnard 1950). The estimated population in the surrounding area is 50,092 inhabitants (IBGE 2010). The estuary is characterized by the presence of mangroves, and the main human activities are artisanal fisheries. Thus, the CIELC is considered a non-polluted site in biomonitoring studies. Furthermore, this complex has also been recognized as a World Heritage site by UNESCO (1999) and was recently included in the RAMSAR List as the Environmental Preservation Area of Cananéia-Iguape-Peruíbe (MMA 2017).
Further south, the PEC is located in the adjacent state of Paraná and covers an area of ~ 551.8 km2 (Noernberg et al. 2006). The PEC surroundings encompass the largest populated area on the coast of Paraná, with a population of approximately 197,231 inhabitants (IBGE 2010). The PEC is surrounded by one of the last remnants of Atlantic rainforest, which is an important characteristic that contributes to its status as a World Heritage site and Biosphere Reserve (UNESCO 1999). It also has great economic importance owing to the port areas of Paranaguá and Antonina, with the former considered the largest port for grain exports in South America (Soares and Barcelos 1995; Choueri et al. 2009).
The fish sampling occurred in the summer and winter of 2015. The fish species were collected using bottom trawls deployed in standardized 5-min tows. Captured fish were kept frozen for further analyses. Despite the sampling effort, the only site where fish were not captured was at S3 (Fig. 1). This site is located in the Santos upper estuary, which is a zone with high anthropogenic impacts and perceived by different groups of researchers as an area with poor fauna diversity and abundance. These conditions might be a consequence of the human activities and associated stressors.
In the laboratory, 775 captured fish specimens were identified to species, sex, and maturation stage after they were measured and weighed according to the identification manual elaborated by Menezes and Figueiredo (1980), and were analyzed in composite samples according to the characteristics described above. Muscle and liver tissues were extracted, lyophilized, and macerated for further analysis.
The metal extraction from the biota was performed following the method described by Trevizani et al. (2016). Specifically, 0.35 g of each dried sample was digested with 4 mL of nitric acid, followed by the addition of 1 mL of hydrogen peroxide. After 18 h, the treated analytes were placed into a heated digester block for 3 h, and the final solution was filtered and diluted to 35 mL. An adapted version of method 7471 (USEPA 1994) was used for Hg analysis. The determination of heavy metals was performed using inductively coupled plasma optical emission spectrometry (ICP-OES – Varian, model 710ES), with a coupled vapor generated accessory (VGA) for Hg analysis.
The trueness of the method was checked using the certified reference materials SRM 2976 (mussel tissue, trace elements and methylmercury, freeze-dried) of the National Institute of Standards and Technology and DORM-2 of the NRCC (National Research Council Canada). All recoveries were between 70 and 110% (Supplementary material).
The results were evaluated by statistical analyses, the normality premises were verified using the Shapiro-Wilk test, and the homogeneity of the data was verified using the Bartlett test. When data did not present normality and homogeneity, a logarithmic transformation was performed. Analysis of variance (ANOVA) followed by Tukey’s HSD test was used to test for differences related to the sampling station, seasonality, fish species (S. rastrifer, P. brasiliensis, and I. parvipinnis), sex (male and female), and maturation stage (1 to 4) on the metal concentration in the muscle tissue. The ANOVA was also used to verify the metal accumulation in the different tissues (muscle and liver) of the studied fish.
Fishes are important indicators of contamination in marine organisms due to their high consumption (Abdel-Baki et al. 2011; Afonso et al. 2017); thus, it is necessary to follow the specific legislation to identify levels that are safe for human consumption. Worldwide, these legislative limits are established by the Food and Agriculture Organization of the United Nations (FAO 1983), and nationally the limits are established by Agência Nacional de Vigilância Sanitária (ANVISA 1965, 2013). The metal concentration values were compared with the available legislation to evaluate potential toxically levels and the availability of consumption by human and the health status of fish from the studied regions.
The target hazard quotient (THQ, below) was estimated only for those metals that presented values above the limits established by the legislation. Considering the exposure of adults and children, different scenarios of fish consumption were simulated: daily (365 days per year), weekly (52 days per year), and monthly (12 days per year). These calculations facilitated estimating the number of safe meals for the consumption of these fish.
The THQ considers the ratio between the exposure and the reference dose and was calculated as recommended by USEPA (2000), according to Eq. 1:
In the equation, EF is the exposure frequency (365 days year−1, 52 days year−1, and 12 days year−1); ED is the exposure duration (6 years for children and 70 years for adults); MS is the food meal size (0.114 kg for children and 0.227 kg for adults) (USEPA 1989; USEPA 2000); C is the annual mean concentration of the metal in fish (mg kg−1); BW is the body weight (16 kg for children and 70 kg for adults); and AT is the average time of exposure (days) to the chemical (365 days year−1 × ED) (USEPA 1989; USEPA 2000). The RfD is the oral reference dose (i.e., Cr 3 × 10−3 mg kg−1 day−1; Se 5 × 10−3 mg kg−1 day−1; Zn 3 × 10−1 mg kg−1 day−1 (USEPA 2018); and Pb 4 × 10−3 mg kg−1 day−1 (Storelli and Barone 2013)). Note that the As consumption limits were calculated as inorganic arsenic contents (3% of the total arsenic) (FSA 2004), whereby the RfD for inorganic arsenic is 3 × 10−4 mg kg−1 day−1 (USEPA 2018). Values of exposure higher than the reference dose (THQ > 1) suggest that chronic systemic effects may occur (USEPA 1989).
Results
Heavy metal concentrations in different tissues
Significant differences were observed in the accumulation of As (p = 0.00), Cr (p = 0.00), Cu (p = 0.00), Ni (p = 0.02), Se (p = 0.00), Zn (p = 0.00), and Hg (p = 0.01) between the concentration of analyzed tissues. The highest concentrations were found in the liver (Fig. 2).
Biotic and abiotic factors affecting heavy metal concentrations in muscle
The sampling station was a significant variable in the accumulation of As (p = 0.0003). The highest levels were found at sampling stations P1 and C3 in the PEC and CIELC. The levels of Cu, Se, and Zn differed significantly between the studied sites (p = 0.00) and seasons (p = 0.00). Higher levels were found during the summer. The sampling stations P3 and C3 in the PEC and CIELC showed the highest levels of Cu. The highest levels of Se were found in fish from the sampling stations P1 and S2 in the PEC and SSVEC. Additionally, all PEC sampling stations showed higher levels of Zn. Neither sample station nor season significantly affected the concentrations of Cr, Ni, Pb, and Hg in the studied fish (Fig. 3).
The concentrations of As (p = 0.00), Cr (p = 0.04), and Ni (p = 0.00) varied significantly among the studied species (Fig. 4), with higher levels found in P. brasiliensis. Zinc showed the highest concentrations in fish, but differed significantly between species (p = 0.00), with higher levels in I. parvipinnis and S. rastrifer and lower levels in P. brasiliensis. The concentrations of Cu, Se, and Hg did not differ among fish species.
Variation of mean (± SD) metal concentrations (mg kg−1) among the studied fish species (Stellifer rastrifer, Isopisthus parvipinnis, and Paralonchurus brasiliensis), sampled in the Santos-São Vicente estuarine complex, Cananéia-Iguape estuarine-lagoon complex, and Paranaguá estuarine complex, Brazil
The variables of sex and maturation stage did not influence the concentrations of As, Cr, Ni, Se, and Hg in the studied fish (Fig. 5). However, of these two factors, the maturation stage influenced the concentration of Cu (p = 0.00) and Zn (p = 0.00), with higher levels in juveniles.
Comparison with legislation and food security
The concentrations of metals concentrated in muscle tissues in the fishes species (P. brasiliensis, S. rastrifer, and I. parvipinnis) collected from SSVEC, CIELC, and PEC, as well as those obtained during other studies along the Brazilian coast are presented in Table 1 along with the minimum levels legislated for human consumption (ANVISA 1965, 2013; FAO 1983).
The THQ of inorganic As, Cr, Pb, Se, and Zn in P. brasiliensis, S. rastrifer, and I. parvipinnis from SSVEC, CIELC, and PEC are presented in Table 2. It is assumed that THQ values greater than 1 are of concern because there is a high risk of developing chronic systemic effects (USEPA 2000). In general, for those adults and children that consume these fish species daily (365 days year−1), THQs were greater than 1 for inorganic As and Se in all species and sites, for Cr only in P. brasiliensis from the PEC, and for Pb only in S. rastrifer from the CIELC and SSVEC. Only the THQ for Zn did not exceed the reference values.
Discussion
All fish are exposed to metals via prey and, to a lesser extent, by direct absorption from the water column (Rejomon et al. 2010). Because metals sink and accumulate in sediments, demersal fish species, such as the P. brasiliensis, S. rastrifer, and I. parvipinnis, assessed here generally are more affected by bioconcentration than pelagic species (Rejomon et al. 2010). Nevertheless, despite greater bioconcentration among demersal species, like all fish, the rates are influenced by several factors, including sex, maturation stage, length/weight, tissue type, habitats, feeding strategies, and, perhaps most importantly, the duration of exposure to contaminants (Azevedo et al. 2009; Storelli 2008). These various factors can be discussed separately for the assessed species, and used to propose the likely risks to regional populations that rely on their exploitation.
Metals and metalloids in different tissues
Previous studies on a plethora of fish species around the world clearly demonstrate that metals have the tendency to bioconcentrate at different levels in various tissues, but with the greatest short-term rates in the liver (Afonso et al. 2017; Alamdar et al. 2017; Dikanovic et al. 2016; El-Ghazaly et al. 2016; Kehrig et al. 2009; Salgado-Ramírez et al. 2017). The data here support this trend with the highest metal concentrations in the livers of all analyzed species. The liver has high metabolic activity, and great capacity for the assimilation and accumulation of metals, being perhaps the most appropriate organ to evaluate in studies focused on environmental pollution (Alamdar et al. 2017; Dikanovic et al. 2016; Salgado-Ramírez et al. 2017).
Fish usually regulate the concentrations of metals in their muscle tissue and preferentially accumulate them in the liver due to the different contents of proteins and amino acids, such as metallothionein and cysteine that are present in different organs. Metallothioneins are proteins found in large amounts in the liver and are responsible for the storage and detoxification of metals, particularly Cu and Zn (D’ Costa et al. 2017; Eisler 1993; Fernandez et al. 2014; Dikanovic et al. 2016). This latter characteristic means that for assessing longer-term bioaccumulation of metals, it is also appropriate to assess muscle tissue owing to its high consumption. Specifically, the ingestion route absorbs more than 90% of the metals that further accumulate in different parts of an organism and act as final reservoirs, such as the muscles (Afonso et al. 2017; Alamdar et al. 2017; Moreno-Sierra et al. 2016; Salgado-Ramírez et al. 2017).
Biotic and abiotic factors affecting heavy metal concentrations
Even though fishes are considered good indicators for environmental pollution, it is well established there are species-specific metabolic and biological accumulation capacities. The results here reiterate that metal accumulation may vary according to the fish species and the metal type; with the highest concentrations of As, Cr, and Ni observed in P. brasiliensis, while Zn showed higher concentrations in S. rastrifer and I. parvipinnis.
Other biotic factors were less influential. For example, irrespective of the metal, the sex of the fish did not influence metal concentrations, and the maturation stage influenced only the accumulation of Cu and Zn, which were present in relatively greater concentrations among juveniles (maturation stage 1). During a study conducted on several fish species in the Caspian Sea, Dadar et al. (2016) also failed to detect sex-specific differences in bioconcentration. But, such differences have been reported elsewhere and attributed to distinct sex-specific physiological and biochemical processes (Al-Yousuf et al. 2000; El-Ghazaly et al. 2016).
By comparison, the observed variation in the metal concentrations with the maturation stage of the fish here might have been influenced by metabolic rates and the dilution of these elements because the ingestion rate is reduced based on the development of the individual. Such an outcome would justify an often observed reduction in metal accumulation with fish growth (Farkas et al. 2003; Anan et al. 2005).
Like biotic factors, abiotic influences were also clearly variable. The concentrations of Cu, Se, and Zn were influenced by seasonality and were greater in summer. Such increases in the concentrations of these essential metals may also be related to an increase in food availability during summer, leading to increased intake and prey sizes for the fish species studied (Branco et al. 2005). However, the result may also be related to the availability of these elements in sewage discharges, which are substantially higher during the summer. For example, the seasonal variability of Hg levels in Cathorops spixii from the Santos estuary (São Paulo state) was associated with the occurrence of different inputs, especially from the industrial area, and a deactivated dump and the mouth of a sewer (Azevedo et al. 2011). Further, the Cu, Se, and Zn concentrations in this study were greater than those in fish from many other environments around the world (Chouvelon et al. 2017; Dadar et al. 2016; Moreno-Sierra et al. 2016; Salgado-Ramírez et al. 2017), implying at least some sewage discharge contamination of three estuaries.
Bioconcentration was also different among the estuaries, and in general was greater in the PEC and CIELC than the SSVEC. Specifically the concentrations of As, Cu, and Zn were greater in fish from the PEC and CIELC, while levels of Se were greater in fish from the PEC and comparable to those observed at SSVEC. Furthermore, the greatest levels of Cr, Ni, and Hg were observed in fish from the PEC, and the highest levels of Pb were found in fish from CIELC and SSVEC. These results raise concerns, and not only for resident human populations, but also because these regions are globally recognized as hotspots of biodiversity and are considered priority areas for conservation (MMA 2017).
Although subject to many anthropogenic pressures, the SSVEC had a lower bioconcentration of metals. The lower bioavailability of these contaminants maybe related to hydrographic conditions of the region, which transports the contaminated sediments out of the Bay (Magalhães et al. 2017). Nonetheless, the presence of compounds such as persistent organic pollutants (POPs) and metals imply that urban activities in the region affect regional water and sediments quality (Lamparelli et al. 2001), and consequently could affect the health of biota across both short- and long-term scales (Azevedo et al. 2011; Magalhães et al. 2017).
Heavy metals, metalloids, and human health
It is clear from the obtained results that Cu, Ni, and Hg in muscle tissues were below the allowed limits established by the legislation for fish (FAO 1983; ANVISA 1965, 2013). But, the concentrations of As and Se were greater in all species from the studied sites and are very likely to be affecting human health, including adults and children that consume these fish species daily. These results were compared with other regions in terms of spatial variation, and with C. spixii, in the same study area. This demersal species was chosen for comparison because it is also regionally abundant, and it is considered a bioindicator of metal pollution (Angeli et al. 2013; Azevedo et al. 2011; Gusso-Choueri et al. 2018).
The results for As were similar to those previously obtained in C. spixii from the PEC (Angeli et al. 2013) and CIELC (Gusso-Choueri et al. 2018). The presence of As in the environment and biota might reflect of anthropogenic activities, but it also can be released through the natural weathering of rocks. For example, previous studies have implied strong natural As enrichment in the PEC, CIELC, and SSVEC (Luiz-Silva et al. 2008; Sá et al. 2006, 2015).
By comparison, the levels of Se in the present study were lower than those measured in fish from the coast of Rio de Janeiro (Kehrig et al. 2013), but greater than those found in P. brasiliensis and I. parvipinnis from the Parnaíba do Sul River (Rio de Janeiro) (Kehrig et al. 2009). Selenium is absorbed by fish through ingestion, and it presents a bioconcentration tendency at the base of a trophic web (Kehrig et al. 2009; Seixas et al. 2012). Water, sediment, and organic debris can be potential routes of exposure to Se in benthic organisms (Kehrig et al. 2009, 2013; Seixas et al. 2012).
With respect to Cr, while the concentrations here were lower than those obtained by other studies conducted in Brazil, the levels were above those allowed by legislation in all species and sites and daily consumption of P. brasiliensis from the PEC could affect human health. Chromium is an essential element, but when present in excess (especially, chromium VI), cause adverse health effects in humans (Demirezen and Uruç 2006).
Similarly, the concentrations of Pb were above the established limits in all species from the studied sites and present a clear risk for daily human consumption of S. rastrifer from the CIELC and SSVEC. Further, the Pb levels in fish from the studied regions were greater than those in fish many other fish species from around the world (Alamdar et al. 2017; Chouvelon et al. 2017; Dadar et al. 2016; Moreno-Sierra et al. 2016). One hypothesis for the high observed Pb may be related to the intense historical mining activity carried out in Cananéia (Mahiques et al. 2013). This metal is related to a reducing fraction in the sediments of the region, which are then available in the environment for the absorption of the biota (Tramonte et al. 2016).
The Zn levels were above the established limits in S. rastrifer and I. parvipinnis from the PEC, but the consumption of these species might not pose a risk to human health. In the fishes from the PEC, the Zn concentrations were similar to C. spixii (Angeli et al. 2013) but at the CIELC, the Zn levels found here were greater than those in C. spixii (Gusso-Choueri et al. 2018). Zinc is an important element involved in metabolic activities and reproductive processes (Salgado-Ramírez et al. 2017); due to its essentiality, its incorporation appears to be independent of environmental conditions (Abdallah 2008).
Considering the calculation of THQ, it was possible to estimate the number of days of consumption of fish that might be considered safe. This value varies according to the evaluated element and the type of consumer (i.e., adult or child). Considering all of the metals and metalloids studied, food safety for fish consumption in these regions is 118 and 41 days a year for adults and children, respectively.
Human risk, impacts, and conclusions
This study is one of the first to quantify the bioconcentration of metals in fishes and key contributing biotic and abiotic factors across three divergent, but proximal, estuaries that are among the most important in southern Brazil. Considering that the main anthropogenic sources of metals for the marine environment include mining activities, burning of fossil fuels, use of pesticides, domestic and industrial sewage, and atmospheric emissions from industries (Abdallah 2008; Baird 2002; Eisler 1988, 2010) and that these sources are present in the regions studied, measures should be taken to mitigate contamination. Doing so will avoid further consequences to ecosystem and human health.
The THQ results of the current study reveal the levels of As, Cr, Pb, and Se in the muscle of P. brasiliensis, S. rastrifer, and I. parvipinnis from PEC, CIELC, and SSVEC have a potential risk of affecting human health through the daily consumption of contaminated fish. Despite the essentiality of some metals, excesses of inorganic As, Cr, Pb, and Se can cause acute intoxication, tissue hypoxia, and inhibit hemoglobin production, which ultimately causes the death of fishes (Eisler 1993, 2010). For humans, such impacts extend to food poisoning, allergies, lung irritation, liver and kidney damage, and cancer (Demirezen and Uruç 2006). These data reinforce the need for monitoring, to accompany ecological and socioeconomic damages resulting from the pollution of these environments.
The data from this study should provide an alert against excessive consumption of fish from the studied estuaries because they are natural metal routes for the human population (Copat et al. 2012; Gusso-Choueri et al. 2018). Quantifying the local sources of pollutants is an essential task for the conservation, recovery, and remediation of the ecosystem; moreover, this information provides security related to food consumption and human health (Rabitto et al. 2011; Gusso-Choueri et al. 2018).
The need for such data is reiterative by the excessive level of some compounds. The concentrations of As, Cr, Se, and Pb were above the legislation limits for fish (FAO 1983; ANVISA 1965), while the level of Zn was above the allowed limits for fish consumption in PEC, representing a health concern, particularly for daily consumers of the fish species studied. The current findings will be useful for environmental monitoring and management of the SSVEC, CIELC, and PEC, contributing to the conservation and sustainable development of coastal regions that include a World Heritage site and a Biosphere Reserve (UNESCO), a RAMSAR site, and environmental preservation areas.
References
Abdallah, M. A. M. (2008). Trace element levels in some commercially valuable fish species from coastal waters of Mediterranean Sea, Egypt. Journal of Marine Systems, 73, 114–122.
Abdel-Baki, A. S., Dkhil, M. A., & Al-Quraishy, S. (2011). Bioaccumulation of some heavy metals in tilapia fish relevant to their concentration in water and sediment of Wadi Hanifah, Saudi Arabia. African Journal of Biotechnology, 10, 2541–2547.
Afonso, A., Gutiérrez, A. J., Lozano, G., González-Weller, D., Rubio, C., Caballero, J. M., Hardisson, A., & Revert, C. (2017). Determination of toxic metals, trace and essentials, and macronutrients in Sarpa salpa and Chelon labrosus: risk assessment for the consumers. Environmental Science and Pollution Research, 24, 10557–10569.
Agência Nacional de Vigilância Sanitária (ANVISA). (1965). Decreto n° 55871 de Março de 1965. http://www.anvisa.gov.br/legis/decretos/55871_65.htm. Accessed 16 August 2016.
Agência Nacional de Vigilância Sanitária (ANVISA). (2013). Resolução n° 42, de 29 de Agosto de 2013. http://portal.anvisa.gov.br/documents/33916/393845/RDC%2Bn%C2%BA%2B42_2013_final.pdf/eec629cf-8d17-422b-a362-366b275c1a00?version=1.0. Accessed 06 February 2019.
Alamdar, A., Eqani, S. A. M. A. S., Hanif, N., Ali, S. M., Fasola, M., Bokhari, H., Katsoyiannis, I. A., & Shen, H. (2017). Human exposure to trace metals and arsenic via consumption of fish from river Chenab, Pakistan and associated health risks. Chemosphere, 168, 1004–1012.
Al-Yousuf, M. H., El-Shahawi, M. S., & Al-Ghais, S. M. (2000). Trace metals in liver, skin and muscle of Lethrinus lentjan fish species in relation to body length and sex. The Science of the Total Environment, 256(2–3), 87–94.
Anan, Y., Kunito, T., Tanabe, S., Mitrofanov, I., & Aubrey, D. G. (2005). Trace element accumulation in fishes collected from coastal waters of the Caspian Sea. Marine Pollution Bulletin, 51, 882–888.
Angeli, J. L. F., Trevizani, T. H., Ribeiro, A. P., Machado, E. C., Figueira, R. C. L., Market, B., Fraenzle, S., & Wuenschmann, S. (2013). Arsenic and other trace elements in two catfish species from Paranaguá Estuarine Complex, Paraná, Brazil. Environmental Monitoring and Assessment, 185, 8333–8342.
Azevedo, J. S., Fernandez, W. S., Farias, L. A., Fávaro, D. T. I., & Braga, E. S. (2009). Use of Cathorops spixii as bioindicator of pollution of trace metals in the Santos Bay, Brazil. Ecotoxicology., 18, 577–586.
Azevedo, J. S., Braga, E. S., Favaro, D. T., Perretti, A. R., Rezende, C. E., & Souza, C. M. M. (2011). Total mercury in sediments and in Brazilian Ariidae catfish from two estuaries under different anthropogenic influence. Marine Pollution Bulletin, 62, 2724–2731.
Baird, C. (2002). Química Ambiental (2a ed.). Porto Alegre: Bookman.
Besnard, W. (1950). Considerações gerais em torno da região lagunar de Cananéia e Iguape I. Boletim do Instituto Paulista de Oceanografia 1(1), 9:26.
Branco, J. O., Lunardon-Branco, M. J., & Verani, J. R. (2005). Aspectos biológicos e pesqueiros de Paralonchurus brasiliensis Stelndachner, (Pisces, Sciaenidae) na Armação do Itapocoroy, Penha, Santa Catarina, Brasil. Revista Brasileira de Zoologia, 22(4), 1063–1071.
CETESB (Companhia de Tecnologia e Saneamento ambiental do Estado de São Paulo). (2008). Qualidade das águas litorâneas no estado de São Paulo: relatório técnico – balneabilidade das praias. http://www.cetesb.sp.gov.br/Agua/praias/publicacoes.asp. Accessed 05 October 2016.
Chapman, P. M., & Wang, F. (2001). Assessing sediment contamination in estuaries. Environmental Toxicology and Chemistry, 20, 3–22.
Choueri, R. B., Cesar, A., Torres, R. J., Abessa, D. M. S., Morais, R. D., Pereira, C. D. S., Nascimento, M. R. L., Mozeto, A. A., Riba, I., & DelValls, T. A. (2009). Integrated sediment quality assessment in Paranaguá Estuarine System, Southern Brazil. Ecotoxicology and Environmental Safety, 72, 1824–1831.
Chouvelon, T., Brach-Papa, C., Auger, D., Bodin, N., Bruzac, S., Crochet, S., Degroote, M., Hollanda, S. J., Hubert, C., Knoery, J., Munschy, C., Puech, A., Rozuel, E., Thomas, B., West, W., Bourjea, J., & Nikolic, N. (2017). Chemical contaminants (trace metals, persistent organic pollutants) in albacore tuna from western Indian and south-eastern Atlantic Oceans: trophic influence and potential as tracers of populations. Science of the Total Environment, 596–597, 481–495.
Copat, C., Bella, F., Castaing, M., Fallico, R., Sciacca, S., & Ferrante, M. (2012). Heavy metals concentrations in fish from Sicily (Mediterranean Sea) and evaluation of possible health risks to consumers. Bulletin of Environmental Contamination and Toxicology, 88, 78–83.
D’ Costa, A., Shyama, S. K., & Praveen Kumar, M. K. (2017). Bioaccumulation of trace metals and total petroleum and genotoxicity responses in an edible fish population as indicators of marine pollution. Ecotoxicology and Environmental Safety, 142, 22–28.
Dadar, M., Adel, M., Saravi, H. N., & Dadar, M. (2016). A comparative study of trace metals in male and female Caspian kutum (Rutilus frisii kutum) from the southern basin of Caspian Sea. Environmental Science and Pollution Research, 23, 24540–24546.
Demirezen, D., & Uruç, K. (2006). Comparative study of trace elements in certain fish, meat and meat products. Meat Science, 74, 255–260.
Dikanovic, V., Skorić, S., & Gačić, Z. (2016). Concentrations of metals and trace elements in different tissues of nine fish species from the Međuvršje reservoir (West Morava River Basin, Serbia). Archives of Biological Sciences, 68(4), 811–819.
Eisler, R. (1988). Arsenic hazards to fish, wildlife, and invertebrates: a synoptic review. Contaminant Hazard Reviews Report No. 12. Laurel, MD: Patuxent Wildlife Research Center.
Eisler, R., (2010). Compendium of Trace Metals and Marine Biota. Volume 2: Vertebrates. Oxford, UK: Elsevier Publishing.
Eisler, R. (1993). Zinc hazards to fish, wildlife, and invertebrates: a synoptic review. Contaminant Hazard Reviews Report No. 26. Laurel, MD: Patuxent Wildlife Research Center.
El-Ghazaly, N. A., Wahbi, O. M., Abou –Shabana, N. M., Hafez, A. M., & Ghanem, S. F. (2016). Influence of sex on accumulation and distribution of some trace metals in Sardinella aurita from three sites in Alexandria and North Coast of Egypt. Journal of Experimental Biology (Zoology), 12(1), 31–38.
FAO (Food and Agriculture Organization). (1983). Compilation of legal limits for hazardous substances in fish and fishery products. FAO Fishery Circular, 464, 5–100.
Farkas, A., Salánki, J., & Specziár, A. (2003). Age and size specific patterns of heavy metals in the organs of freshwater fish Abramis brama L. populating a low-contaminated site. Water Research, 37, 959–964.
Fernandez, W. S., Dias, J. F., Boufleur, L. A., Amaral, L., Yoneama, M. L., & Dias, J. F. (2014). Bioacumulation of trace elements in hepatic and renal tissues of the white mullet Mugil curema Valenciennes, 1836 (Actinopterygii, Mugilidae) in two coastal systems in southeastern Brazil. Nuclear Instruments and Methods in Physics Research B, 318, 94–98.
FSA (Food Standards Agency). (2004). Total diet study: total and inorganic arsenic in food. In: Agency, F.S. (Ed.), Food surveillanca information sheet.
Gusso-Choueri, P. K., Araújo, G. S., Cruz, A. C. F., Stremel, T. R. O., Sandro Xavier de Campos, S. X., Abessa, D. S., Ribeiro, C. A. O., & Choueri, R. B. (2018). Metals and arsenic in fish from a Ramsar site under past and present human pressures: consumption risk factors to the local population. Science of the Total Environment, 628–629, 621–630.
IBGE (Instituto Brasileiro de Geografia e Estatística). (2010). Conheça Cidades e Estados do Brasil. https://cidades.ibge.gov.br/. Accessed 18 January 2017.
Islam, M. S., & Tanaka, M. (2004). Impacts of pollution on coastal and marine ecosystems including coastal and marine fisheries and approach for management: a review and synthesis. Marine Pollution Bulletin, 48, 624–649.
Jaric, I., Visnjic-Jeftic, Z., Cvijanovic, G., Gacic, Z., Jovanovic, L., Skoric, S., & Lenhardt, M. (2011). Determination of differential heavy metal and trace element accumulation in liver, gills, intestine and muscle of sterlet (Acipenser ruthenus) from the Danube River in Serbia by ICP-OES. Microchemical Journal, 98, 77–81.
Kasper, D., Botaro, D., Palermo, E. F. A., & Malm, O. (2007). Mercúrio em peixes - fontes e contaminação. Oecologia Brasiliensis., 11(228–239), 2007.
Kehrig, H. A., Seixas, T. G., Palermo, E. A., Baeta, A. P., Branco, C. W. C., Malm, O., & Moreira, I. (2009). The relationships between mercury and selenium in plankton and fish from a tropical food web. Environmental Science and Pollution Research, 16, 10–24.
Kehrig, H. A., Seixas, T. G., Malm, O., Di Beneditto, A. P. M., & Rezende, C. E. (2013). Mercury and selenium biomagnification in a Brazilian coastal food web using nitrogen stable isotope analysis: a case study in an area under the influence of the Paraiba do Sul River plume. Marine Pollution Bulletin, 75, 283–290.
Lamparelli, M.C.; Costa, M.P.; Prósperi, V.A.; Bevilacqua, J.E.; Araujo, R.P.A.; Eysink, G.G.J.; Pompeia, S. (2001). Sistema estuarino de Santos e São Vicente. In: Relatório Técnico CETESB, Vol. 183. São Paulo.
Luiz-Silva, W., Machado, W., & Matos, R. H. R. (2008). Multi-elemental contamination and historical record in sediments from the Santos-Cubatão Estuarine System, Brazil. Journal of the Brazilian Chemical Society, 19(18), 1490–1500.
Magalhães, C. A., Taniguchi, S., Lourenço, R. A., & Montone, R. C. (2017). Organochlorine pesticides, PCBs, and PBDEs in liver and muscle tissues of Paralonchurus brasiliensis, Trichiurus lepturus and Cathorops spixii in Santos Bay and surrounding area, São Paulo, Brazil. Regional Studies in Marine Science, 16, 42–48.
Mahiques, M. M., Figueira, R. C. L., Salaroli, A. B., Alves, D. P. V., & Gonçalves, C. (2013). 150 years of anthropogenic metal input in a biosphere reserve: the case study of the Cananéia–Iguape coastal system, Southeastern Brazil. Environment and Earth Science, 68, 1073–1087.
Marone, E., Machado, E. C., Lopes, R. M., & Da Silva, E. T. (2005). Land-ocean fluxes in the Paranaguá Bay estuarine system, southern Brazil. Brazilian Journal of Oceanography, 53, 169–181.
Mendonça, J. T., & Miranda, L. V. (2008). Estatística pesqueira do litoral sul do estado de São Paulo: subsídios para gestão compartilhada. Pan-American Journal of Aquatic Sciences, 3(3), 152–173.
Menezes, N. A.; Figueiredo, J. L. (1980). Manual de Peixes Marinhos do sudeste do Brasil: IV Teleostei (3). Museu de Zoologia – Universidade de São Paulo: São Paulo.
MMA. (2017). Ministério do Meio Ambiente – Sítios Ramsar. http://www.mma.gov.br/areas-protegidas/instrumentos-de-gestao/s%C3%ADtiosramsar. Accessed 17 August 2016.
Moiseenko, T. I., & Kudryavtseva, L. P. (2001). Trace metal accumulation and fish pathologies in areas affected by mining and metallurgical enterprises in the Kola Region, Russia. Environmental Pollution, 114, 285–297.
Moreno-Sierra, D., Bergés-Tiznado, M. E., Márquez-Farías, F., Torres-Rojas, Y. E., Ruelas-Inzunza, J. R., & Páez-Osuna, F. (2016). Trace metals in target tissues and stomach contents of the top predator sailfish Istiophorus platypterus from the Eastern Pacific: concentrations and contrasting behavior of biomagnification. Environmental Science and Pollution Research, 23, 23791–23803.
Muto, E. Y., Soares, L. S. H., Sarkis, J. E. S., Hortellani, M. A., Petti, M. A. V., & Corbisier, T. N. (2014). Biomagnification of mercury through the food web of the Santos continental shelf, subtropical Brasil. Marine Ecology Progress Series, 512, 55–69.
Noernberg, M. A., Lautert, L. F. C., Araújo, A. D., Marone, E., Angelotti, J. P. B., Jr, N., & Krug, L. A. (2006). Remote sensing and GIS integration for modelling the Paranaguá estuarine complex – Brazil. Journal of Coastal Research Special Issue, 39, 1627–1631.
Queiroz, G. M. N., Spach, H. L., Sobolewski-Morelos, M., Santos, L. O., & Schwarz Junior, R. (2006). Caracterização da ictiofauna demersal de duas áreas do Complexo Estuarino de Paranaguá, Paraná, Brazil. Biociências, 14(2), 112–124.
Rabitto, I., Bastos, W. R., Almeida, R., Anjos, A., de Holanda, I. B., Galvão, R. C., Neto, F. F., de Menezes, M. L., Dos Santos, C. A., & Oliveira Ribeiro, C. A. (2011). Mercury and DDT exposure risk to fish-eating human populations in Amazon. Environment International, 37(1), 56–65.
Rejomon, G., Mair, M., & Joseph, T. (2010). Trace metal dynamics in fishes from the southwest coast of India. Environmental Monitoring and Assessment, 167, 243–255.
Sá, F., Machado, E. C., Angulo, R. J., Veiga, F. A., & Brandini, N. (2006). Arsenic and heavy metals in sediments near Paranaguá Port, Southern Brazil. Journal of Coastal Research, 39, 1066–1068.
Sá, F., Sanders, C. J., Patchineelam, S. R., Machado, E. C., & Lombardi, A. T. (2015). Arsenic fractionation in estuarine sediments: does coastal eutrophication influence As behavior? Marine Pollution Bulletin, 96, 496–501.
Salgado-Ramírez, C. A., Mansilla-Rivera, I., & Rodríguez-Sierra, C. J. (2017). Comparison of trace metals in different fish tissues of Scomberomorus spp. (“sierra”) and Lutjanus synagris (“arrayado”) from Jobos Bay and La Parguera coastal areas in Southern Puerto Rico. Regional Studies in Marine Science, 13, 1–11.
Santana, C. O., de Jesus, T. B., Aguiar, W. M., Franca-Rocha, W. d. J. S., & Soares, C. A. C. (2017). Assessment of health risk related to the ingestion of trace metals through fish consumption in Todos os Santos Bay. Environmental Monitoring and Assessment, 189, 204.
Seixas, T. G., Moreira, I., Malm, O., & Kehrig, H. A. (2012). Mercury and selenium in a toppredator fish, Trichiurus lepturus (Lanjosinnaeus, 1758), from the Tropical Brazilian Coast, Rio de Janeiro. Bulletin of Environmental Contamination and Toxicology, 89, 434–438.
Soares, C. R., & Barcelos, J. H. (1995). Considerações sobre os sedimentos de fundo das baías de Laranjeiras e de Guaraqueçaba, Complexo estuarino da Baía de Paranaguá (Paraná, Brasil). Boletim Paranaense de Geociências, 43, 41–60.
Storelli, M. M. (2008). Potential human risks from metals (Hg, Cd and Pb) and polychlorinated biphenyls (PCBs) via seafood consumption: estimation of target hazard quotients (THQs) and toxic equivalents (TEQs). Food and Chemical Toxicology, 46, 2782–2788.
Storelli, M. M., & Barone, G. (2013). Toxic metals (Hg, Pb, and Cd) in commercially important demersal fish from Mediterranean sea: contamination levels and dietary exposure assessment. Journal of Food Science, 78, T362–T366.
Tramonte, K. M., Figueria, R. C. L., de Lima Ferreira, P. A., Ribeiro, A. P., Batista, M. F., & de Mahiques, M. M. (2016). Environmental availability of potentially toxic elements in estuarine sediments of the Cananéia–Iguape coastal system, Southeastern Brazil. Marine Pollution Bulletin, 103, 260–269.
Trevizani, T. H., Figueira, R. C. L., Ribeiro, A. P., Theophilo, C. Y. S., Majer, A. P., Petti, M. A. V., Corbisier, T. N., & Montone, R. C. (2016). Bioaccumulation of heavy metals in marine organisms and sediments from Admiralty Bay, King George Island, Antarctica. Marine Pollution Bulletin, 106, 366–371.
UNESCO - United Nations Educational, Scientific and Cultural Organization. (1999). Atlantic forest south-east reserves. World Heritage List. http://whc.unesco.org/en/list/893. Accessed 15 January 2016.
USEPA (Unitec States Environmental Protection Agency). (1994). Method 7471A mercury in solid or semisolid waste (manual cold-vapor technique). Revision, 1.
USEPA (United States Environmental Protection Agency). (2000). Risk-based concentration table. Philadelphia: United States Environmental Protection Agency.
USEPA- (United States Environmental Protection Agency). (1989). Risk assessment guidance for superfund. Human Health Evaluation Manual Part A, Interim Final (Vol. I). Washington (DC): United States Environmental Protection Agency (1989 EPA/540/ 1–89/002).
USEPA, IRIS Assessments. (United States Environmental Protection Agency). (2018). Integrated risk information system. https://cfpub.epa.gov/ncea/iris2/atoz.cfm. Accessed 22 February 2018.
Vernenberg, F. J. (1981). Biological mosnitoring of marine pollutants. New York: Academic Press.
Acknowledgements
This work was supported by the Conselho Nacional de Desenvolvimento Científico e Tecnológico [CNPq; grant numbers 162446/2014-4]. We thank PhD. Matt Broadhurst for the contributions with the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 23 kb)
Rights and permissions
About this article
Cite this article
Trevizani, T.H., Domit, C., Vedolin, M.C. et al. Assessment of metal contamination in fish from estuaries of southern and southeastern Brazil. Environ Monit Assess 191, 308 (2019). https://doi.org/10.1007/s10661-019-7477-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10661-019-7477-1