Abstract
The Fresno-Benavente Pepper (F-BP) Protected Geographical Indication (PGI) is a horticultural crop characterized by its great agronomic, economic and cultural importance in the region of Castilla y León (Spain). Field production is threatened by verticillium wilt caused by Verticillium dahliae and postharvest losses due to grey mould caused by Botrytis cinerea. Eight Bacillus spp. strains endophytically isolated from F-BP roots were used in the study. By conducting an in vitro antagonism study, we found that all Bacillus strains were effective against B. cinerea and five of them showed high antagonism against V. dahliae, with B. siamensis and B. proteolyticus strains being the most effective against both pathogens. Eight Bacillus strains were used for an infection test in F-BP fruits and plants to test their activity against both fungal pathogens. We report that Bacillus thuringiensis, B. siamensis and B. pumilus (SCFC 1–2) could control B. cinerea in pepper fruits through direct action and local activation of plant defences. In the case of V. dahliae root infection, plant roots inoculated with B. siamensis and B. proteolyticus were able to significantly decrease the occurrence of disease through direct action and local activation of jasmonic acid as a defence response. Therefore, we propose that B. siamensis could be used to control B. cinerea and V. dahliae in F-BP fruits and plants, respectively, through direct antagonism as well as the induction of local plant defence responses.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Sweet pepper (Capsicum annuum L.) is an annual herbaceous plant belonging to the Solanaceae family and originating from South America (Fratianni et al., 2020; Guevara et al., 2021). It is part of the diet especially in European countries (Fratianni et al., 2020). Sweet peppers represent an important nutritional resource of antioxidant compounds, such as carotenoids, ascorbic acid and polyphenols (Fratianni et al., 2020). The world production of this crop stands at 36 million tons per year, on 2 million hectares (FAO, 2020).
According to the European Union Council Regulation (EC) No 925/2012, the Fresno-Benavente Pepper (F-BP) (“Pimiento de Fresno-Benavente, in Spanish) is produced in three specific regions in Castilla y León (Spain), namely Benavente and Los Valles (Zamora province) and Fresno (León province), all of them watered by the Esla and Tera rivers. F-BP was granted a Protected Geographical Indication (PGI) in 2012 (EC 925/2012). The F-BP describes a type of pepper that has been specifically adapted to its production area, including 55 municipalities in the north of the province of Zamora, 33 municipalities in the southeast of the province of León, and a municipality in the province of Valladolid. It is a “cow’s nose” or “bell pepper” type, large (10 cm minimum width), red in colour, rectangular in shape and with thick and juicy flesh (EC 925/2012).
Wilt caused by Verticillium dahliae and grey mould caused by Botrytis cinerea are the two primary diseases impacting production. V. dahliae is a soilborne pathogen, causing serious economic losses in more than 150 different crops (Tubeileh & Stephenson, 2020). It is the most important root pathogen in pepper cultivation, causing a vascular wilt that leads to plant death, including symptoms such as a delay in plant growth, chlorosis, discoloration of the vascular bundles, and a whitish colour can be seen in the roots with serious damage that causes the loss of the outermost layers (Tyvaert et al., 2019). B. cinerea is a plant pathogenic fungus that causes serious diseases in more than 1400 different plant species. In peppers, B. cinerea can attack the leaves and fruits in the field, but the real damage occurs at post-harvest when fruits develop the characteristic grey mould that spreads rapidly and causes fruit rot (Mekawi et al., 2019; Poveda et al., 2020).
Plant endophytes are microorganisms that develop their entire life or part of it within plant tissues without causing visible damage or disease (Poveda et al., 2021). With regard to bacteria, they can be found inside different organs and plant tissues, such as seeds, tubers, roots, stems, leaves or fruits; the roots being the organ in which the greatest number and diversity of microorganisms can be found. The most commonly isolated bacterial genera are Pseudomonas, Bacillus, Enterobacter and Agrobacterium (Eljounaidi et al., 2016). Endophytic bacteria can be of great importance in the agricultural system, promoting plant growth, favouring tolerance to abiotic stresses or reducing the incidence of biotic stresses. Regarding the effects against phytopathogens, endophytic bacteria reduce the damage caused to plants due to the release of chemical compounds, such as antibiotics, a mechanism known as antibiosis, through direct parasitism, competition for space and/or nutrients, or the induction of specific plant defensive responses (Eljounaidi et al., 2016; Poveda & González-Andrés, 2021).
In a previous work, different endophytic bacterial strains were isolated locally from F-BP plants (in 2010, and in the municipalities of: San Cristóbal de Entreviñas, Micereces de Tera and Fresno de la Vega) and selected for their outstanding biocontrol capacity against the root pathogenic oomycete of pepper, Phytophthora capsici. Out of the 122 isolated strains, two strains of the Bacillus pumilus species were described as the ones that most reduced the P. capsici infection in pepper (Barquero et al., 2016). The bacterial strains with the greatest biocontrol potential were those used in the present work.
The objective of this work was to identify effective endophytic and locally isolated bacterial biological control agents (BCAs) against V. dahliae and B. cinerea in F-BP and to assess potential modes of action. We utilized a local bacterial strain collection (described in the following section), isolated from F-BP plants grown in their geographical demarcation, because they are the bacteria that are fully adapted to the host plant and the environmental conditions of the crop. Strains were assessed for biocontrol activity through antagonistic in vitro assays, plant inoculations and infections in vivo and in planta, and molecular and biochemical analysis to assess potential activation of plant defences.
Materials and methods
Microbiological material
Bacterial strains used in this study (Barquero et al., 2016) included: B. siamensis strains SCFB 2–2 and SCFB 3–4, B. pumilus strains SCFC 1–2 and FVA 1–5, B. proteolyticus strain SCFC 2–2, Bacillus safensis strain MTA 1–2, Bacillus megaterium strain MMM 3–6 and Bacillus thuringiensis strain FVA 2–3, all of them identified by 16S RNA gene sequencing. Strains are preserved in glycerol at −80 °C in the collection of the Chemical, Environmental and Bioprocess Engineering Group (IQUIMAB) of the University of León (Spain), accessible to any researchers that would like to acquire them for testing. Strains are routinely grown on tryptic-soy-agar (TSA, Sigma-Aldrich, Madrid, Spain).
The pathogenic fungi used were obtained from the Spanish Type Culture Collection (CECT) (Valencia, Spain): B. cinerea (CECT 20973) and V. dahliae (CECT 20246). Strains are routinely grown on potato-dextrose-agar (PDA, Sigma-Aldrich, Madrid, Spain), adding powdery tomato leaves to the medium (0.1% v/v) formulation in order to maintain the phytopathogen activity (Acosta-Morel et al., 2019).
In vitro antagonism assay
The evaluation of the antagonistic activity of Bacillus species against B. cinerea and V. dahliae, was assessed by the dual culture technique in vitro, based on the methodology described by Slama et al. (2019), with some modifications. Mycelium disks (6 mm diameter) of each pathogenic fungus, obtained from the edge of the colonies grown on PDA medium, were placed in the centre of the Petri plate with TSA medium (pathogenic fungi grow similarly on TSA medium and PDA medium: Fig. S1). From an overnight pure bacterial culture on TSA agar, a loopful of cells was suspended in 0.9% NaCl solution, applying two 5 μl drops of 108 CFU mL−1 at 30 mm distance from the fungus inoculum. Controls were performed with and without the application of 5 μl of 0.9% NaCl solution, and no difference was found between them. The inhibition rate (IR) was calculated according to the following formula (Lyousfi et al., 2021):
where AC means the colony area of the pathogenic fungus in the control treatment (TSA medium without bacterial treatments), and AT means the colony area of the pathogenic fungus in bacterial treatments. Fungal areas were measured using ImageJ photographic analysis software, at 3, 5 and 7 days for B. cinerea, and at 7, 10 and 14 days for V. dahliae.
The experimental unit is represented by each of the Petri dishes. Each treatment was performed on eight plates (replicates per treatment), and the experiment was repeated twice.
In vivo biocontrol of B. cinerea in fruits assay
The in vivo study against the pathogen B. cinerea with the different Bacillus species was carried out on pepper fruits of the F-BP. For this, the methodology previously described by Shi and Sun (2017) for tomatoes was carried out, with some modifications. The fruits were superficially sterilized by washing with 70% ethanol and 2% sodium hypochlorite. A 3 × 3 mm cross-shaped wound was made in the epidermis of the fruits where each of the bacteria was inoculated with 40 μl at a concentration of 108 CFU ml−1. Two hours after bacterial inoculation, the wounds were inoculated with 15 μl of B. cinerea spores at a concentration of 105 spores ml−1. Subsequently, the fruits were placed in plastic bags to maintain constant-high humidity and kept at 23 °C and in the dark for 7 days. Different controls were used: peppers without B. cinerea inoculation and without bacterial treatment, peppers without B. cinerea inoculation and with bacterial treatment, and peppers with B. cinerea inoculation and without bacterial treatment.
After one week of fungal infection, measurements were taken of the areas of lesions produced by B. cinerea in peppers, tissue viability and reactive oxygen species (ROS) accumulation. In addition, infected tissues from the fruits were collected and immediately frozen in liquid nitrogen to analyse levels of pathogen colonization and expression of defence-related genes.
The experimental unit is represented by each fruit. Each treatment was performed on eight fruits (replicates per treatment), and the experiment was repeated three times. Treatments and B. cinerea-inoculations on each of the peppers were performed randomly.
In planta biocontrol of V. dahliae assay
F-BP seeds were surface-sterilized by vigorous sequential shaking in 70% ethanol and 5% sodium hypochlorite solutions for 10 min each and then washed thoroughly four times in sterile, distilled water. Pepper seeds were individually transferred to 1 L pots, containing a sterilized mixture of field soil/vermiculite (3:1) that was previously prepared by autoclaving a local field soil twice (24 h apart). Seeded pots were maintained in a greenhouse.
Bacillus strains were applied to three-week-old pepper seedlings as described by Mena-Violante and Olalde-Portugal (2007), with some modifications. Pepper seedlings were treated by applying a 1 ml suspension (107 CFU ml−1) of each bacterium to individual roots. Inoculation with the pathogenic fungus V. dahliae was carried out as described by Mercado-Blanco et al. (2004), with some modifications. Two weeks after applying bacterial root treatments, the pepper plants were root inoculated with 3 ml of a V. dahliae spores suspension (107 spores ml−1).
Four weeks after inoculation with the fungus, the plants were collected and measurements were taken of the dry weight of the aerial part of the plant (after drying at 65 °C for 48 h), disease severity, root vitality and ROS accumulation in root tissues. In addition, roots were collected and immediately frozen in liquid nitrogen to analyse levels of pathogen colonization and defence gene expression. Disease severity was analysed by a 0–4 rating scale according to the percentage of affected leaves and twigs (0 = no symptoms, 1 = 1–33%, 2 = 34–66%, 3 = 67–100%, and 4 = dead plant) (Mercado-Blanco et al., 2004).
The experimental unit is represented by the individual plants. Each treatment was performed on eight plants (replicates per treatment), and the experiment was repeated three times. Treatments and V. dahliae-inoculations on each of the plants were performed randomly. In addition, plants were randomly arranged in the greenhouse.
Vitality test and indirect quantification of ROS in tissues
To analyse and quantify the damage produced by both pathogens on F-BP fruits and roots, the vitality test and ROS quantification were assayed, as reported by Poveda (2021a).
The vitality test is based on the reduction of triphenyltetrazolium chloride (TTC) by living tissues to red-coloured insoluble triphenylformazan (TF), due to the activity of the mitochondrial respiratory chain. Thus, only living tissues should reduce TTC to TF. Pools were formed from eight fruits (B. cinerea-infected plants) or roots (V. dahliae-infected) per condition. From these pools, 100 mg was transferred to 1 mL of 1% TTC in triplicate and incubated for 48 h at 37 °C. After incubation, 200 mg of Ballotini Glass Balls (0.15–0.25 mm diameter) was added to each sample in 1.5 mL Eppendorf tubes and shaken vigorously. After centrifuging the samples for 15 min at 10,000 rpm, the supernatant was removed, and 1 mL of isopropanol was added to each tube. The samples were again agitated and centrifuged in the same way, and the supernatant was used to quantify the absorbance at 620 nm; this acted as an indirect measure of the vitality of the F-BP tissues.
The indirect quantification of reactive oxygen species (ROS) in F-BP tissues was carried out by measuring electrolyte leakage, which is a measure of cellular oxidative damage related to the production of ROS, a sign of stress in plant tissues. This methodology of indirect analysis of cell damage caused by a plant pathogen has been used in many other works, with a direct relationship between the quantified damage and the indirect ROS data obtained (Poveda, 2020, 2021a, b, 2022a, b), demonstrating its success.
From each pool formed from fruits (B. cinerea-infected plants) or roots (V. dahliae-infected), 1 cm2 of fresh tissue was briefly mixed with water and floated on 5 mL of double-distilled water at room temperature for 6 h. The conductivity of the water was measured using a Crison™ Conductimeter GLP31 (Crison, Barcelona, Spain). This represented the electrolyte leakage from the tissues (Reading 1). Then, samples were boiled for 20 min at 90 °C. After the liquid cooled down, the conductivity of the water was measured again. This represented the total concentration of ions present in the tissues (Reading 2). Electrolyte leakage, an indirect measurement of ROS, was represented as the percentage of total ions released [(Reading 1/Reading 2) × 100].
Tissue-colonization analysis
In order to determine the differences in F-BP tissue colonization by B. cinerea and V. dahliae, they were quantified by qPCR. For the analysis of B. cinerea-fruit and V. dahliae-root colonization, pools formed from eight fruits (B. cinerea-infected) or roots (V. dahliae-infected) per treatment were used. All material was immediately frozen with liquid nitrogen and pulverized with a mortar.
Following the methodology described by Poveda (2022a), DNA was extracted using the cetyltrimethyl-ammonium bromide (CTAB) extraction method. A mix was prepared in a 10-μL volume using 5 μL of Brilliant SYBR Green qPCR Master Mix (Roche, Penzberg, Germany), 10 ng of DNA, the forward and reverse primers at a final concentration of 100 nM, and nuclease-free PCR-grade water to adjust the final volume. The endogenous genes of pathogens and pepper used for the quantification are represented in Table 1. Amplifications were performed in an ABI PRISM 7000 Sequence Detection System (Applied Biosystems, Foster City, CA, USA), programmed for 40 cycles under the following conditions: denaturation, 95 °C for 15 s; annealing, 60 °C for 1 min; extension, 72 °C for 1 min. Each PCR was performed in triplicate by using the DNA extracted from three tissue pools of eight plants each – one for each treatment. Cycle threshold values served to calculate the amount of pathogen DNA using standard curves. The values of pathogen DNA were referred to the amount of pepper DNA in every corresponding sample.
Defence gene expression analysis
To analyse the defence responses following F-BP infection with B. cinerea and V. dahliae, an expression analysis of defence genes was performed by RT-qPCR. For gene expression studies, the pools formed from eight fruits (B. cinerea-infected) or roots (V. dahliae-infected) per treatment were used for RNA extraction with TRIzol reagent (Invitrogen, Waltham, Massachusetts, USA). RNA was cleaned up with the Direct-zol RNA Miniprep Kit (Zymo Research, Irvine, CA, USA) including a DNase enzymatic treatment. cDNA was synthesized from RNA with Reverse Transcription System (Promega, Madison, WI, USA). Gene expression was analysed by RT-qPCR using an Agilent Mx3005P Real-Time PCR System (Agilent Technologies, Santa Clara, CA, USA) with Luna ® Universal qPCR Mastermix (New England Biolabs, Ipswich, MA, USA). All PCR reactions were performed in triplicate in a total volume of 10 μL for 40 cycles under the following conditions: denaturation at 95 °C for 15 s; and annealing and extension at 60 °C for 30 s; and melt curve. Threshold cycles (CT) were calculated using the pepper Actin gene as an endogenous control. The primers used are shown in Table 1, as follows: phenylalanine ammonia lyase 1 (PAL1), pathogenesis-related protein 1 (PR-1), lipoxygenase 1 (LOX1), and defensin 1 (DEF1) genes. Data are expressed using the 2−ΔΔCT method (Livak & Schmittgen, 2001).
Statistical analysis
Statistical analysis of the data was carried out with Statistix 8.0 software. One-way ANOVA using Tukey’s multiple range test at P < 0.05 was used for pairwise comparisons; the different letters indicate significant differences.
Results
In vitro antagonism
The in vitro antagonism test between the different Bacillus species and the pathogens B. cinerea and V. dahliae (Fig. 1) reported positive IRs in all confrontations, with the exception of B. megaterium strain MMM 3–6 against V. dahliae (up to −22% IR) (Fig. 2b).
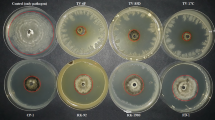
Photographs of the antagonism in vitro study between the pathogens B. cinerea (3, 5 and 7 days) and V. dahliae (7, 10 and 14 days) and Bacillus endophytic bacteria. SCFB 2–2: B. siamensis; SCFB 3–4: B. siamensis; SCFC 1–2: B. pumilus; SCFC 2–2: B. proteolyticus; MTA 1–2: B. safensis; MMM 3–6: B. megaterium; FVA 1–5: B. pumilus; FVA 2–3: B. thuringiensis
Inhibition rate (IR, %) in B. cinerea (a) and V. dahliae (b) in their antagonistic confrontation in vitro against B. siamensis (SCFB 2–2), B. siamensis (SCFB 3–4), B. pumilus (SCFC 1–2), B. proteolyticus (SCFC 2–2), B. safensis (MTA 1–2), B. megaterium (MMM 3–6), B. pumilus (FVA 1–5) and B. thuringiensis (FVA 2–3). Data are the mean of two biological replicates for each condition with eight plates in each one. One-way analysis of variance (ANOVA) was performed, followed by the Tukey’s test. Different letters represent significant differences (P < 0.05)
B. siamensis SCFB 3–4 and B. pumilus SCFC 1–2 produced a significantly higher IR against B. cinerea compared to other bacteria strains (between 16% and 28% higher inhibition). Furthermore, B. proteolyticus SCFC 2–2 and B. megaterium MMM 3–6 at day 7, also reduced the pathogen growth area in a significantly greater way, compared to the other bacteria strains (between 18% and 20% higher inhibition). On the other hand, B. pumilus FVA 1–5 produced a significantly lower IR compared to the other bacteria strains (Fig. 2a). In addition, in B. siamensis SCFB 2–2, B. pumilus SCFC 1–2, B. proteolyticus SCFC 2–2, B. megaterium MMM 3–6, B. pumilus FVA 1–5 and B. thuringiensis FVA 2–3 confrontations, B. cinerea growth had zones of inhibition. A higher mycelial density was also observed in the B. siamensis SCFB 3–4, B. pumilus SCFC 1–2, B. megaterium MMM 3–6 and B. thuringiensis FVA 2–3 confrontations, and a darker coloration was observed in the B. siamensis SCFB 3–4, B. proteolyticus SCFC 2–2 and B. thuringiensis FVA 2–3 confrontations (Fig. 2a).
For V. dahliae, B. megaterium MMM 3–6 promoted the growth of the pathogen in vitro. B. pumilus SCFC 1–2 and FVA 1–5 and B. safensis MTA 1–2 inhibited the pathogen growth. Moreover, B. siamensis (SCFB 2–2 and SCFB 3–4) and B. proteolyticus (SCFC 2–2) produced significantly higher inhibition rates (between 19% and 27% higher inhibition) (Fig. 2b). Furthermore, in the confrontations with B. siamensis SCFB 2–2, B. siamensis SCFB 3–4, B. pumilus SCFC 1–2, B. megaterium MMM 3–6, B. pumilus FVA 1–5 and B. thuringiensis FVA 2–3, the growth of V. dahliae had zones of inhibition. A lower mycelial density was also observed in the B. siamensis SCFB 3–4, B. proteolyticus SCFC 2–2 and B. pumilus FVA 2–3 confrontations, and a lighter coloration was observed in the confrontations against B. pumilus SCFB 2–2, B. siamensis SCFB 3–4, B. proteolyticus SCFC 2–2, B. megaterium MMM 3–6 and B. pumilus FVA 2–3 (Fig. 2b).
In vivo biocontrol of B. cinerea in fruits
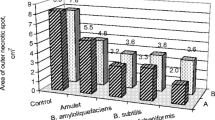
The treatment of pepper wounds with the Bacillus species caused a significant reduction in the lesions caused by B. cinerea (reductions of between 41% and 98%) (Fig. 3). The most efficient strain as BCA was B. safensis MTA 1–2, with a significantly smaller area of average lesions produced (reduction of 98%) by the pathogen than the following, B. thuringiensis FVA 2–3, B. pumilus SCFC 1–2 and B. siamensis SCFB 2–2 and SCFB 3–4 (Fig. 3b).
Lesions produced by B. cinerea in peppers from the F-BP PGI. Appearance of infections caused by B. cinerea and wounds inoculated only with bacteria (−) (a) and quantification of lesions area (cm2) (b). Control: non-treated, inoculated control; SCFB 2–2: B. siamensis; SCFB 3–4: B. siamensis; SCFC 1–2: B. pumilus; SCFC 2–2: B. proteolyticus; MTA 1–2: B. safensis; MMM 3–6: B. megaterium; FVA 1–5: B. pumilus; FVA 2–3: B. thuringiensis. The columns represent the average of three biological replicates with eight fruits in each one. One-way analysis of variance (ANOVA) was performed, followed by a Tukey test. The different letters signify significant differences (P < 0.05)
Six out of eight Bacillus strains significantly improved tissue viability based on relative TTC staining, compared to the non-treated control. From the effective strains, B. safensis MTA 1–2 produced the highest values of viability (0.078), significantly higher than B. siamensis SCFB 3–4 (0.039) and B. pumilus FVA1–5 (0.037); moreover B. siamensis SCFB 2–2 and SCFB 3–4, B. pumilus SCFC 1–2 and B. thuringiensis FVA 2–3 produced intermediate values (Fig. 4a). Concomitantly, only three strains reduced ROS levels compared with the non-treated control (4.23), namely B. safensis MTA 1–2 (1.22) followed by B. thuringiensis FVA 2–3 (2.34) and B. pumilus SCFC 1–2 (2.81) (Fig. 4b).
Tissue vitality by TTC test (a) and indirect quantification of ROS (b) in F-BPs PGI infected with the pathogen B. cinerea. Control: non-treated, inoculated control; SCFB 2–2: B. siamensis; SCFB 3–4: B. siamensis; SCFC 1–2: B. pumilus; SCFC 2–2: B. proteolyticus; MTA 1–2: B.s safensis; MMM 3–6: B. megaterium; FVA 1–5: B. pumilus; FVA 2–3: B. thuringiensis. The columns represent the average of three biological replicates with eight fruits in each one. One-way analysis of variance (ANOVA) was performed, followed by a Tukey test. The different letters signify significant differences (P < 0.05)
Except for strains B. proteolyticus SCFC 2–2 and B. pumilus FVA 1–5, six Bacillus strains reduced B. cinerea colonization relative to the non-treated control based on qPCR (Fig. 5). Strain B. safensis MTA1–2 was statistically the most effective of the Bacillus strains. Based on qPCR results, strain B. safensis MTA 1–2 reduced B. cinerea colonization by 92% compared to the control, whereas strains B. siamensis SCFB 2–2, B. siamensis SCFB 3–4, B. proteolyticus SCFC 2–2, B. megaterium MMM 3–6 and B. thuringiensis FVA 2–3 gave an intermediate reduction of 26% to 73%.
Quantification of fungal-plant tissue-colonization in F-BP fruits infected with B. cinerea. Control: non-treated, inoculated control; SCFB 2–2: B. siamensis; SCFB 3–4: B. siamensis; SCFC 1–2: B. pumilus; SCFC 2–2: B. proteolyticus; MTA 1–2: B. safensis; MMM 3–6: B. megaterium; FVA 1–5: B. pumilus; FVA 2–3: B. thuringiensis. The columns represent the average of three biological replicates with eight fruits in each one. One-way analysis of variance (ANOVA) was performed, followed by a Tukey test. The different letters signify significant differences (P < 0.05)
The expression analysis of several genes involved in the defensive responses of the plant was carried out in the tissues of pepper infected with B. cinerea (Fig. 6). Strains B. safensis MTA 1–2 and B. thuringiensis FVA 2–3 significantly reduced the expression of PAL1 and PR-1 (88% reduction) and significantly increased the expression of LOX1 and DEF1 (>400% increase). Similarly, inoculation with B. siamensis SCFB 2–2 and SCFB 3–4 and B. pumilus SCFC 1–2 caused a significant increase in the expression of LOX1 and DEF1, although the decrease in the expression of PAL1 and PR-1 was significantly less (except PAL1 expression for B. siamensis SCFB2–2 and PR-1 expression for B. siamensis SCFB3–4), compared to the non-treated control fruit. Fruit treated with B. proteolyticus SCFC 2–2, B. megaterium MMM 3–6 and B. pumilus FVA 1–5 showed a significant increase in the expression of PAL1 and PR-1 (except expression of PR-1 for B. proteolyticus SCFC2–2 and B. pumilus FVA1–5) and a significant decrease in LOX1 and DEF1 compared to the non-treated control (Fig. 6).
Quantitative reverse transcription polymerase chain reaction (RT-qPCR) analysis of the expression of some defence genes in F-BP fruits infected with B. cinerea. Control: non-treated, inoculated control; SCFB 2–2: B. siamensis; SCFB 3–4: B. siamensis; SCFC 1–2: B. pumilus; SCFC 2–2: B. proteolyticus; MTA 1–2: B. safensis; MMM 3–6: B. megaterium; FVA 1–5: B. pumilus; FVA 2–3: B. thuringiensis. The genes analysed were phenylalanine ammonium lyase 1 (PAL1), pathogenesis-related protein (PR-1), lipoxygenase 1 (LOX1) and defensin 1 (DEF1). The measurements were referenced to the value obtained for the plants without inoculation with Bacillus (2–ΔΔCt = 1). The pepper Actin gene was used as an internal reference gene. The columns represent the average of three biological replicates with 8 fruits in each one. One-way analysis of variance (ANOVA) was performed, followed by a Tukey test. The different letters signify significant differences (P < 0.05)
In planta biocontrol of V. dahliae
The inoculation with V. dahliae plus the following Bacillus strains produced significantly higher foliar biomass (up to 10 times greater) and lower disease severity than the non-treated control: B. siamensis SCFB 2–2 and SCFB 3–4, B. proteolyticus SCFC 2–2 and B. megaterium MMM 3–6 (Fig. 7). Only tissues from roots infected with V. dahliae and inoculated with B. siamensis SCFB 2–2 and SCFB 3–4 showed significantly higher viability (0.065–0.073) and lower ROS (1.22–1.28) values compared to the non-treated controls (0.021 and 3.85, respectively) (Fig. 8).
Dry weight of aerial part (a) and disease severity (b) of V. dahliae-infected F-BP plants. Control: non-treated, inoculated control; SCFB 2–2: B. siamensis; SCFB 3–4: B. siamensis; SCFC 1–2: B. pumilus; SCFC 2–2: B. proteolyticus; MTA 1–2: B. safensis; MMM 3–6: B. megaterium; FVA 1–5: B. pumilus; FVA 2–3: B. thuringiensis. The columns represent the average of three biological replicates with eight plants in each one. One-way analysis of variance (ANOVA) was performed, followed by a Tukey test. The different letters signify significant differences (P < 0.05)
Tissue vitality by TTC test (a) and indirect quantification of ROS (b) in F-BP plants infected with the pathogen V. dahliae. Control: non-treated, inoculated control; SCFB 2–2: B. siamensis; SCFB 3–4: B. siamensis; SCFC 1–2: B. pumilus; SCFC 2–2: B. proteolyticus; MTA 1–2: B. safensis; MMM 3–6: B. megaterium; FVA 1–5: B. pumilus; FVA 2–3: B.s thuringiensis. The columns represent the average of three biological replicates with eight plants in each one. One-way analysis of variance (ANOVA) was performed, followed by a Tukey test. The different letters signify significant differences (P < 0.05)
The quantification of the root colonization of pepper plants by the pathogen V. dahliae showed that roots treated with B. proteolyticus SCFC 2–2, B. siamensis SCFB 2–2 and SCFB 3–4, and B. megaterium MMM 3–6, significantly reduced the root colonization by the pathogen, compared to the non-treated control (reductions of between 69% and 83%) (Fig. 9).
Quantification of fungal-plant tissue-colonization in F-BP plants infected with V. dahliae. Control: non-treated, inoculated control; SCFB 2–2: B. siamensis; SCFB 3–4: B. siamensis; SCFC 1–2: B. pumilus; SCFC 2–2: B. proteolyticus; MTA 1–2: B. safensis; MMM 3–6: B. megaterium; FVA 1–5: B. pumilus; FVA 2–3: B. thuringiensis. The columns represent the average of three biological replicates with eight plants in each one. One-way analysis of variance (ANOVA) was performed, followed by a Tukey test. The different letters signify significant differences (P < 0.05)
Regarding defence-gene expression in pepper roots infected with V. dahliae, the inoculation with B. thuringiensis FVA 2–3 increased the expression of SA-related genes (24–44% increase), compared to plants without bacterial inoculation (Fig. 10). On the contrary, the inoculations of the bacteria B. proteolyticus SCFC 2–2 and B. megaterium MMM 3–6 produced a significant reduction in the expression of PR-1, with an even greater reduction with the inoculations of B. siamensis SCFB 2–2 and SCFB 3–4 (reduction of 80–98%) (Fig. 10). Regarding JA-related genes, root inoculation with B. siamensis SCFB 2–2 and SCFB 3–4, B. proteolyticus SCFC 2–2 and B. megaterium MMM 3–6 bacteria significantly increased their expression (even >400% increase), compared to the non-treated control. However, inoculation with B. pumilus SCFC 1–2 and FVA 1–5, B. safensis MTA 1–2 and B. thuringiensis FVA 2–3 bacteria significantly reduced the expression of JA-related genes (except LOX1 in B. pumilus FVA 1–5), compared to plants without bacterial inoculation (Fig. 10).
Quantitative reverse transcription polymerase chain reaction (RT-qPCR) analysis of the expression of some defence genes in F-BP plants infected with V. dahliae. Control: non-treated, inoculated control; SCFB 2–2: B. siamensis; SCFB 3–4: B. siamensis; SCFC 1–2: B. pumilus; SCFC 2–2: B. proteolyticus; MTA 1–2: B. safensis; MMM 3–6: B. megaterium; FVA 1–5: B. pumilus; FVA 2–3: B. thuringiensis. The genes analysed were phenylalanine ammonium lyase 1 (PAL1), pathogenesis-related protein (PR-1), lipoxygenase 1 (LOX1) and defensin 1 (DEF1). The measurements were referenced to the value obtained for the plants without inoculation with Bacillus (2–ΔΔCt = 1). The pepper Actin gene was used as an internal reference gene. The columns represent the average of three biological replicates with eight plants in each one. One-way analysis of variance (ANOVA) was performed, followed by a Tukey test. The different letters signify significant differences (P < 0.05)
Discussion
From the species included in our work, B. proteolyticus inhibited in vitro the growth of B. cinerea and V. dahliae, being the first description for both pathogens, although it has recently been described as an in vitro antagonist of pathogens Nigrospora sphaerica, Pestalotiopsis theae, Curvularia eragrostidis, Glomerella cingulata, Rhizoctonia solani and Fusarium oxysporum (Dutta & Thakur, 2021). On the other hand, B. siamensis is a bacterial species widely described as a BCA against pathogens, such as Alternaria alternata (Xie et al., 2021), Colletotrichum fructicola (Park et al., 2021), or fungi producing postharvest diseases, including B. cinerea (You et al., 2021). The two isolates from this study belonging to B. siamensis, inhibited the growth of B. cinerea. Moreover, they also inhibited the growth of the root pathogen V. dahliae, previously described (Gao et al., 2021). Similarly, B. safensis has been widely described as a potent BCA, including pathogens such as Aspergillus flavus (Einloft et al., 2021), A. alternata (Prakash & Arora, 2021), Magnaporthe oryzae (Rong et al., 2020), Plenodomus destruens (Mateus et al., 2021) and B. cinerea (Berrada et al., 2012; Hassan et al., 2021); the B. safensis strain from this study controlled the growth of B. cinerea in vitro as previously reported, but it also stood out as a BCA of V. dahliae, never reported before, to the best of our knowledge.
On the other hand, the potential of B. pumilus as a BCA has been described for R. solani and F. oxysporum (Agarwal et al., 2017). In our work, we have demonstrated in vitro effect against B. cinerea and V. dahliae. Interestingly, the inhibitory effect was strain-dependent in the case of B. cinerea, with B. siamensis SCFC1–2 being more effective than B. pumilus FVA 1–5, whilst the two strains were very effective against V. dahliae.
Finally, B. megaterium has previously been identified as an efficient BCA in vitro against A. flavus (Kong et al., 2010), Septoria tritici (Kildea et al., 2008) or Alternaria japonica (Vásconez et al., 2020) and B. cinerea, (Donmez et al., 2011). The B. megaterium strain from this work inhibited in vitro the growth of B. cinerea, but unexpectedly, it promoted V. dahliae growth, contrary to what other authors have observed for other strains (Abada et al., 2018). However, it has been previously described that B. megaterium promotes the growth of other fungi such as mycorrhizal ones (Marulanda et al., 2009). In this sense, Sarathambal et al. (2022) suggested that the reason for the positive interaction could be related to the bacteria zinc solubilizing capacity.
The results obtained in the in vivo infection of F-BP fruits with the pathogen B. cinerea after the different bacterial inoculations with Bacillus showed that the strains that best controlled the pathogen were B. safensis MTA 1–2, B. thuringiensis FVA 2–3, B. siamensis SCFB 3–4 and SCFC 1–2, and B. pumilus SCFC 1–2. Furthermore, it has been found that the bacterial strains B. proteolyticus SCFC 2–2, B megaterium MMM 3–6 and B. pumilus FVA 1–5 of do not induce the activation of defence genes in colonized pepper tissues.
B. safensis MTA 1–2 was able to reduce the disease caused by B. cinerea in pepper fruits. However, its in vitro antagonism was very low. Therefore, its ability as a BCA on fruits could only be a consequence of an activation of plant defence responses. The ability of B. safensis to induce plant defences has been determined in rice plants root inoculated with the bacteria and infected in the aerial part with the pathogen Bipolaris sorokiniana (Sarkar et al., 2018). In the specific case of B. cinerea, the present work could be the first description of B. safensis as an inducer of plant defences against the pathogen, although further tests are required to confirm this, e.g., by physically separating bacterial strain and pathogen.
Based on the results obtained in the study, B. thuringiensis FVA 2–3 would be able to act against B. cinerea in pepper fruits due to a direct antagonism and the activation of plant defensive responses. The ability of B. thuringiensis to activate plant defence responses has been previously described with tomato root treatments, against the fungus F. oxysporum f. sp. lycopersici (Akram et al., 2013) or against the bacteria Ralstonia solanacearum (Takahashi et al., 2014). Against B. cinerea, the ability of B. thuringiensis to activate plant defences and reduce the disease produced by the pathogen in tomato leaves has been described due to the induction of the expression of different defence genes (Yoshida et al., 2019).
Similarly, both strains of B. siamensis used would be capable of reducing the ability of B. cinerea to infect pepper fruit tissues through direct antagonism and activation of genes related to plant defence. In this sense, it has been previously described how the root inoculation of tobacco plants with B. siamensis significantly reduced the foliar lesions caused by the pathogenic fungus A. alternata, due to the activation of plant defence genes (Xie et al., 2021). In the specific case of B. cinerea, our study represents the first description of activation of plant defences against the pathogen mediated by B. siamensis.
Finally, regarding the two strains of B. pumilus, we have verified how the strain SCFC 1–2 would be able to reduce the damage caused by B. cinerea in pepper fruits thanks to the direct antagonism and activation of the plant defences, while the FVA 1–5 strain did not show plant defences activation. The ability of B. pumilus to activate plant defences has been previously described in tobacco and pepper plants against foliar infection by the bacteria Xanthomonas axonopodis, through the induction of SA-related genes (Li et al., 2020; Yi et al., 2013).
The results obtained in the in planta study showed that the bacterial species that best reduces the disease caused by V. dahliae was B. siamensis. In addition, B. proteolyticus and B. megaterium also reduced disease, to a lesser degree than B. siamensis.
In the control of V. dahliae in F-BP roots by B. siamensis mechanisms of both direct antagonism and activation of plant defences would be involved. In this sense, the ability of the bacterium to activate plant defences systemically when inoculated into roots has been previously described (Xie et al., 2021), its ability to activate defence genes locally in roots has not been previously described. Therefore, our study represents the first description of activation of root defences by B. siamensis inoculation and the first description of its effectiveness against the pathogen V. dahliae.
In the case of the bacteria B. proteolyticus, we have described its ability to control V. dahliae in pepper roots also through direct antagonism and activation of plant defences. These results represent a highly relevant finding since B. proteolyticus had never been described as a BCA against V. dahliae and our study places it as one of the most effective bacterial species.
Finally, B. megaterium would be able to reduce the disease caused by V. dahliae in pepper roots mainly through the activation of plant defences mediated by JA (specific response against necrotrophic pathogens, such as V. dahliae). The ability of B. megaterium to induce systemic resistance in plants has been previously described in cucumber roots against the oomycete Pythium aphanidermatum (Liang et al., 2011) and in Camellia sinensis roots against the fungus Fomes lamaoensis (Chakraborty et al., 2006). Against V. dahliae, the present work would be the first description of the ability of B. megaterium to control the pathogen due to the activation of plant defence responses, since in the direct in vitro interaction we found that the bacteria promoted the growth of the pathogenic fungus.
Our results show how the best protection effect against B. cinerea and V. dahliae occurs when SA-related genes (PAL1 and PR1) are downregulated, and JA-related genes (LOX1 and DEF1) are upregulated. The SA pathway activates plant defences against biotrophic pathogens, whereas the JA pathway activates plant defences against necrotrophic pathogens (Li et al., 2019; Thaler et al., 2012). Therefore, SA pathway activation is known to compromise plant defence against necrotrophes through JA pathway suppression (Li et al., 2019). Our results are consistent with those studies, by showing a clear antagonism between SA-related genes and JA-related genes activations. Those bacterial strains that downregulate SA-related genes and upregulate JA-related genes (B. siamensis SCFB 2–2, B. siamensis SCFB 3–4, B. thuringiensis FVA 2–3 and B. safensis MTA 1–2 in the case of B. cinerea infection; and B. siamensis SCFB 2–2, B. siamensis SCFB 3–4, B. proteolyticus SCFC 2–2 and B. megaterium MMM 3–6 in the case of V. dahliae infection) are the ones that show the best results in fruit injury area, aerial biomass, ion leakage and viability. JA-related genes activation explains this protective effect as B. cinerea and V. dahliae are necrotrophic pathogens. However, the mechanism used by the strains to activate the JA pathway has not been uncovered by our investigation, thus requiring further research in order to know how it is done. One possibility is the production of molecules that mimic JA by the bacteria (Thaler et al., 2012), but they have not been detected in the present study.
In conclusion, the endophytic species B. siamensis is the best choice as a biological control agent for the main diseases suffered by the F-BP. This choice is because B. siamensis can reduce the disease caused by B. cinerea in fruits and V. dahliae in roots, through direct antagonism and activation of plant defences. Although these results have been obtained under laboratory and greenhouse conditions (with local soil), the application and efficiency of the treatment in field and storage conditions can give very different results, as there are other variables, such as microbiota already present in the soils and fruits, or adverse weather conditions. Therefore, tests with B. siamensis should be carried out in real situations and its effectiveness in the agricultural system of the area should be determined.
Data availability
Not applicable.
Code availability
Not applicable.
References
Abada, K. A. M., Attia, A. M. F., & Zyton, M. A. L. (2018). Management of pepper Verticillium wilt by combinations of inducer chemicals for plant resistance, bacterial bioagents and compost. Journal of Applied Biotechnology & Bioengineering, 5, 117–127. https://doi.org/10.15406/jabb.2018.05.00126
Acosta-Morel, W., Marques-Costa, T. M., Santander-Gordón, D., Anta Fernández, F., Zabalgogeazcoa, I., Vázquez de Aldana, B. R., et al. (2019). Physiological and population genetic analysis of Botrytis field isolates from vineyards in Castilla y León, Spain. Plant Pathology, 68(3), 523–536. https://doi.org/10.1111/ppa.12967
Agarwal, M., Dheeman, S., Dubey, R. C., Kumar, P., Maheshwari, D. K., & Bajpai, V. K. (2017). Differential antagonistic responses of Bacillus pumilus MSUA3 against Rhizoctonia solani and Fusarium oxysporum causing fungal diseases in Fagopyrum esculentum Moench. Microbiological Research, 205, 40–47. https://doi.org/10.1016/j.micres.2017.08.012
Akram, W., Mahboob, A., & Javed, A. A. (2013). Bacillus thuringiensis strain 199 can induce systemic resistance in tomato against Fusarium wilt. European Journal of Microbiology and Immunology, 3(4), 275–280. https://doi.org/10.1556/EuJMI.3.2013.4.7
Barquero, M., Terrón, A., Velázquez, E., & González-Andrés, F. (2016). Biocontrol of fusarium oxysporum f. sp. phaseoli and Phytophthora capsici with autochthonous endophytes in common bean and pepper in Castilla y León (Spain). In F. González-Andrés & E. James (Eds.), Biological nitrogen fixation and beneficial plant-microbe interaction (pp. 221–235). Springer. https://doi.org/10.1007/978-3-319-32528-6_19
Berrada, I., Benkhemmar, O., Swings, J., Bendaou, N., & Amar, M. (2012). Selection of halophilic bacteria for biological control of tomato gray mould caused by Botrytis cinerea. Phytopathologia Mediterranea, 51(3), 625–630. https://doi.org/10.14601/Phytopathol_Mediterr-10627
Chakraborty, U., Chakraborty, B., & Basnet, M. (2006). Plant growth promotion and induction of resistance in Camellia sinensis by Bacillus megaterium. Journal of Basic Microbiology, 46(3), 186–195. https://doi.org/10.1002/jobm.200510050
Donmez, M. F., Esitken, A., Yildiz, H., & Ercisli, S. (2011). Biocontrol of Botrytis cinerea on strawberry fruit by plant growth promoting bacteria. Journal of Animal and Plant Sciences, 21, 758–763.
Dutta, J., & Thakur, D. (2021). Diversity of culturable bacteria endowed with antifungal metabolites biosynthetic characteristics associated with tea rhizosphere soil of Assam, India. BMC Microbiology, 21, 1–13. https://doi.org/10.1186/s12866-021-02278-z
EC 925/2012. (2012). Council regulation (EC) no 925/2012 of 8 October 2012, entering a name in the register of protected designations of origin and protected geographical indications (Pimiento de Fresno-Benavente (PGI)). Official Journal of the European Union, 275, 23–24 http://data.europa.eu/eli/reg_impl/2012/925/oj
Einloft, T. C., de Oliveira, P. B., Radünz, L. L., & Dionello, R. G. (2021). Biocontrol capabilities of three Bacillus isolates towards aflatoxin B1 producer a. flavus in vitro and on maize grains. Food Control, 125, 107978. https://doi.org/10.1016/j.foodcont.2021.107978
Eljounaidi, K., Lee, S. K., & Bae, H. (2016). Bacterial endophytes as potential biocontrol agents of vascular wilt diseases–review and future prospects. Biological Control, 103, 62–68. https://doi.org/10.1016/j.biocontrol.2016.07.013
Food and Agriculture Organization of the United Nations (FAO).(2020). FAOSTAT Database. Production/Yield quantities of Chillies and peppers, green in World. Retrieved December 21, 2021, from http://www.fao.org/faostat/en/#data/QC
Fratianni, F., d’Acierno, A., Cozzolino, A., Spigno, P., Riccardi, R., Raimo, F., Pane, C., Zaccardelli, M., Tranchida Lombardo, V., Tucci, M., Grillo, S., Coppola, R., & Nazzaro, F. (2020). Biochemical characterization of traditional varieties of sweet pepper (Capsicum annuum L.) of the Campania region, Southern Italy. Antioxidants, 9(6), 556. https://doi.org/10.3390/antiox9060556
Gao, L., Ma, J., Liu, Y., Huang, Y., Mohamad, O. A. A., Jiang, H., Egamberdieva, D., Li, W., & Li, L. (2021). Diversity and biocontrol potential of cultivable endophytic bacteria associated with halophytes from the West Aral Sea basin. Microorganisms, 9, 1448. https://doi.org/10.3390/microorganisms9071448
Guevara, L., Domínguez-Anaya, M. Á., Ortigosa, A., González-Gordo, S., Díaz, C., Vicente, F., Corpas, F. J., Pérez del Palacio, J., & Palma, J. M. (2021). Identification of compounds with potential therapeutic uses from sweet pepper (Capsicum annuum L.) fruits and their modulation by nitric oxide (NO). International Journal of Molecular Sciences, 22(9), 4476. https://doi.org/10.3390/ijms22094476
Hassan, E. A., Mostafa, Y. S., Alamri, S., Hashem, M., & Nafady, N. A. (2021). Biosafe Management of Botrytis Grey Mold of strawberry fruit by novel bioagents. Plants, 10(12), 2737. https://doi.org/10.3390/plants10122737
Hwang, I. S., & Hwang, B. K. (2010). The pepper 9-lipoxygenase gene CaLOX1 functions in defense and cell death responses to microbial pathogens. Plant Physiology, 152(2), 948–967. https://doi.org/10.1104/pp.109.147827
Kildea, S., Ransbotyn, V., Khan, M. R., Fagan, B., Leonard, G., Mullins, E., & Doohan, F. M. (2008). Bacillus megaterium shows potential for the biocontrol of Septoria tritici blotch of wheat. Biological Control, 47(1), 37–45. https://doi.org/10.1111/jam.13171
Kim, D. S., & Hwang, B. K. (2014). An important role of the pepper phenylalanine ammonia-lyase gene (PAL1) in salicylic acid-dependent signalling of the defence response to microbial pathogens. Journal of Experimental Botany, 65(9), 2295–2306. https://doi.org/10.1093/jxb/eru109
Klimes, A., Amyotte, S. G., Grant, S., Kang, S., & Dobinson, K. F. (2008). Microsclerotia development in Verticillium dahliae: Regulation and differential expression of the hydrophobin gene VDH1. Fungal Genetics and Biology, 45(12), 1525–1532. https://doi.org/10.1016/j.fgb.2008.09.014
Kong, Q., Shan, S., Liu, Q., Wang, X., & Yu, F. (2010). Biocontrol of Aspergillus flavus on peanut kernels by use of a strain of marine Bacillus megaterium. International Journal of Food Microbiology, 139(1–2), 31–35. https://doi.org/10.1016/j.ijfoodmicro.2010.01.036
Li, N., Han, X., Feng, D., Yuan, D., & Huang, & L.-J. (2019). Signaling crosstalk between salicylic acid and ethylene/jasmonate in plant defense: Do we understand what they are whispering? International Journal of Molecular Sciences, 20(3), 671. https://doi.org/10.3390/ijms20030671
Li, W., Lee, S. Y., Cho, Y. J., Ghim, S. Y., & Jung, H. Y. (2020). Mediation of induced systemic resistance by the plant growth-promoting rhizobacteria Bacillus pumilus S2-3-2. Molecular Biology Reports, 47(11), 8429–8438. https://doi.org/10.1007/s11033-020-05883-9
Liang, J. G., Taom, R. X., Hao, Z. N., Wang, L., & Zhang, X. (2011). Induction of resistance in cucumber against seedling damping-off by plant growth-promoting rhizobacteria (PGPR) Bacillus megaterium strain L8. African Journal of Biotechnology, 10(36), 6920–6927. https://doi.org/10.5897/AJB11.260
Livak, K. J., & Schmittgen, T. D. (2001). Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods, 25(4), 402–408. https://doi.org/10.1006/meth.2001.1262
Lyousfi, N., Lahlali, R., Letrib, C., Belabess, Z., Ouaabou, R., Ennahli, S., Blenzar, A., & Barka, E. A. (2021). Improving the biocontrol potential of bacterial antagonists with salicylic acid against brown rot disease and impact on nectarine fruits quality. Agronomy, 11(2), 209. https://doi.org/10.3390/agronomy11020209
Marulanda, A., Barea, J. M., & Azcón, R. (2009). Stimulation of plant growth and drought tolerance by native microorganisms (AM fungi and bacteria) from dry environments: Mechanisms related to bacterial effectiveness. Journal of Plant Growth Regulation, 28(2), 115–124. https://doi.org/10.1007/s00344-009-9079-6
Mateus, J. R., Dal’Rio, I., Jurelevicius, D., da Mota, F. F., Marques, J. M., Ramos, R. T. J., da Costa da Silva, A. L., Gagliardi, P. R., & Seldin, L. (2021). Bacillus velezensis T149-19 and Bacillus safensis T052-76 as potential biocontrol agents against foot rot disease in sweet potato. Agriculture, 11(11), 1046. https://doi.org/10.3390/agriculture11111046
Mekawi, E. M., Khafagi, E. Y., & Abdel-Rahman, F. A. (2019). Effect of pre-harvest application with some organic acids and plant oils on antioxidant properties and resistance to Botrytis cinerea in pepper fruits. Scientia Horticulturae, 257, 108736. https://doi.org/10.1016/j.scienta.2019.108736
Mena-Violante, H. G., & Olalde-Portugal, V. (2007). Alteration of tomato fruit quality by root inoculation with plant growth-promoting rhizobacteria (PGPR): Bacillus subtilis BEB-13bs. Scientia Horticulturae, 113(1), 103–106. https://doi.org/10.1016/j.scienta.2007.01.031
Mercado-Blanco, J., Rodrıguez-Jurado, D., Hervás, A., & Jiménez-Dıaz, R. M. (2004). Suppression of Verticillium wilt in olive planting stocks by root-associated fluorescent Pseudomonas spp. Biological Control, 30(2), 474–486. https://doi.org/10.1016/j.biocontrol.2004.02.002
Park, B. R., Son, H. J., Park, J. H., Kim, E. S., Heo, S. J., Youn, H. R., Koo, Y. M., Heo, A. Y., Choi, H. W., Sang, M. K., Lee, S. W., Choi, S. H., & Hong, J. K. (2021). Chemical fungicides and Bacillus siamensis H30-3 against fungal and oomycete pathogens causing soil-borne strawberry diseases. The Plant Pathology Journal, 37(1), 79–85. https://doi.org/10.5423/PPJ.NT.12.2020.0232
Poveda, J. (2020). Marchantia polymorpha subsp. ruderalis (Bischl. & Boissel.-dub.)-arbuscular mycorrhizal fungi interaction: Beneficial or harmful? Symbiosis, 82, 165–174. https://doi.org/10.1007/s13199-020-00708-6
Poveda, J. (2021a). Biological control of Fusarium oxysporum f. sp. ciceri and Ascochyta rabiei infecting protected geographical indication Fuentesaúco-Chickpea by Trichoderma species. European Journal of Plant Pathology, 160, 825–840. https://doi.org/10.1007/s10658-021-02286-9
Poveda, J. (2021b). Glucosinolates profile of Arabidopsis thaliana modified root colonization of Trichoderma species. Biological Control, 155, 104522. https://doi.org/10.1016/j.biocontrol.2020.104522
Poveda, J. (2022a). AtCube: Performing pathogen-root infection tests on Arabidopsis thaliana in a completely controlled way. Physiological and Molecular Plant Pathology, 117, 101780. https://doi.org/10.1016/j.pmpp.2021.101780
Poveda, J. (2022b). Effect of volatile and non-volatile metabolites from Leptosphaeria maculans on tomato calli under abiotic stresses. Plant Stress, 3, 100054. https://doi.org/10.1016/j.stress.2021.100054
Poveda, J., & González-Andrés, F. (2021). Bacillus as a source of phytohormones for use in agriculture. Applied Microbiology and Biotechnology, 105(23), 8629–8645. https://doi.org/10.1007/s00253-021-11492-8
Poveda, J., Barquero, M., & González-Andrés, F. (2020). Insight into the microbiological control strategies against Botrytis cinerea using systemic plant resistance activation. Agronomy, 10(11), 1822. https://doi.org/10.3390/agronomy10111822
Poveda, J., Eugui, D., Abril-Urías, P., & Velasco, P. (2021). Endophytic fungi as direct plant growth promoters for sustainable agricultural production. Symbiosis, 85(1), 1–19. https://doi.org/10.1007/s13199-021-00789-x
Prakash, J., & Arora, N. K. (2021). Novel metabolites from Bacillus safensis and their antifungal property against Alternaria alternata. Antonie Van Leeuwenhoek, 114, 1245–1258. https://doi.org/10.1007/s10482-021-01598-4
Ren, W., Liu, N., Sang, C., Shi, D., Zhou, M., Chen, C., Qin, Q., & Chen, W. (2018). The autophagy gene BcATG8 regulates the vegetative differentiation and pathogenicity of Botrytis cinerea. Applied Environmental Microbiology, 84(11), e02455–e02417. https://doi.org/10.1128/AEM.02455-17
Rong, S., Xu, H., Li, L., Chen, R., Gao, X., & Xu, Z. (2020). Antifungal activity of endophytic Bacillus safensis B21 and its potential application as a biopesticide to control rice blast. Pesticide Biochemistry and Physiology, 162, 69–77. https://doi.org/10.1016/j.pestbp.2019.09.003
Sarathambal, C., Dinesh, R., Srinivasan, V., Sheeja, T. E., Jeeva, V., & Manzoor, M. (2022). Changes in bacterial diversity and composition in response to co-inoculation of arbuscular mycorrhizae and zinc-solubilizing bacteria in turmeric rhizosphere. Current Microbiology, 79(1), 1–9. https://doi.org/10.1007/s00284-021-02682-8
Sarkar, J., Chakraborty, U., & Chakraborty, B. N. (2018). Induced defense response in wheat plants against Bipolaris sorokiniana following application of Bacillus safensis and Ochrobactrum pseudogrignonense. Indian Phytopathology, 71(1), 49–58. https://doi.org/10.1007/s42360-018-0006-2
Shi, J. F., & Sun, C. Q. (2017). Isolation, identification, and biocontrol of antagonistic bacterium against Botrytis cinerea after tomato harvest. Brazilian Journal of Microbiology, 48(4), 706–714. https://doi.org/10.1016/j.bjm.2017.03.002
Slama, H. B., Cherif-Silini, H., Chenari Bouket, A., Qader, M., Silini, A., Yahiaoui, B., Alenezi, F. N., Luptakova, L., Triki, M. A., Vallat, A., Oszako, T., Rateb, M. E., & Belbahri, L. (2019). Screening for Fusarium antagonistic bacteria from contrasting niches designated the endophyte Bacillus halotolerans as plant warden against Fusarium. Frontiers in Microbiology, 9, 3236. https://doi.org/10.3389/fmicb.2018.03236
Takahashi, H., Nakaho, K., Ishihara, T., Ando, S., Wada, T., Kanayama, Y., Asano, S., Yoshida, S., Tsushima, S., & Hyakumachi, M. (2014). Transcriptional profile of tomato roots exhibiting Bacillus thuringiensis-induced resistance to Ralstonia solanacearum. Plant Cell Reports, 33(1), 99–110. https://doi.org/10.1007/s00299-013-1515-1
Thaler, J. S., Humphrey, P. T., & Whiteman, N. K. (2012). Evolution of jasmonate and salicylate signal crosstalk. Trends in Plant Science, 17(5), 260–270. https://doi.org/10.1016/j.tplants.2012.02.010
Tubeileh, A. M., & Stephenson, G. T. (2020). Soil amendment by composted plant wastes reduces the Verticillium dahliae abundance and changes soil chemical properties in a bell pepper cropping system. Current Plant Biology, 22, 100148. https://doi.org/10.1016/j.cpb.2020.100150
Tyvaert, L., Everaert, E., Lippens, L., Cuijpers, W. J. M., França, S. C., & Höfte, M. (2019). Interaction of Colletotrichum coccodes and Verticillium dahliae in pepper plants. European Journal of Plant Pathology, 155(4), 1303–1317. https://doi.org/10.1007/s10658-019-01857-1
Vásconez, R. D. A., Moya, E. M. T., Yépez, L. A. C., Chiluisa-Utreras, V. P., & de los Ángeles Vaca-Suquillo, I. (2020). Evaluation of Bacillus megaterium strain AB4 as a potential biocontrol agent of Alternaria japonica, a mycopathogen of Brassica oleracea var. italica. Biotechnology Reports, 26, e00454. https://doi.org/10.1016/j.btre.2020.e00454
Xie, Z., Li, M., Wang, D., Wang, F., Shen, H., Sun, G., Feng, C., Wang, X., Chen, D., & Sun, X. (2021). Biocontrol efficacy of Bacillus siamensis LZ88 against brown spot disease of tobacco caused by Alternaria alternata. Biological Control, 154, 104508. https://doi.org/10.1016/j.biocontrol.2020.104508
Yi, H. S., Yang, J. W., & Ryu, C. M. (2013). ISR meets SAR outside: Additive action of the endophyte Bacillus pumilus INR7 and the chemical inducer, benzothiadiazole, on induced resistance against bacterial spot in field-grown pepper. Frontiers in Plant Science, 4, 122. https://doi.org/10.3389/fpls.2013.00122
Yoshida, S., Koitabashi, M., Yaginuma, D., Anzai, M., & Fukuda, M. (2019). Potential of bioinsecticidal Bacillus thuringiensis inoculum to suppress gray mold in tomato based on induced systemic resistance. Journal of Phytopathology, 167(11–12), 679–685. https://doi.org/10.1111/jph.12864
You, W., Ge, C., Jiang, Z., Chen, M., Li, W., & Shao, Y. (2021). Screening of a broad-spectrum antagonist-Bacillus siamensis, and its possible mechanisms to control postharvest disease in tropical fruits. Biological Control, 157, 104584. https://doi.org/10.1016/j.biocontrol.2021.104584
Funding
Open Access funding provided by Universidad Pública de Navarra. This work was supported by the Instituto de Estudios Zamoranos “Florián de Ocampo” (IEZFO) as part of the XXVIII Call for Research Grants in 2020.
Author information
Authors and Affiliations
Contributions
J.P. is the Principal Researcher of the research project. J.P., M.B. and F.G.-A. thought and designed the work. J.P. and J.C. performed the assays and analyzed the results. M.B. provided the endophytic bacteria used in the study. J.P. wrote the first version of the manuscript. J.C., M.B. and F.G.-A. contributed to the manuscript correction and critical reading, as well as to the knowledge on the bacteria field. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Supplementary Information
ESM 1
(PNG 1915 kb)
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Poveda, J., Calvo, J., Barquero, M. et al. Activation of sweet pepper defense responses by novel and known biocontrol agents of the genus Bacillus against Botrytis cinerea and Verticillium dahliae. Eur J Plant Pathol 164, 507–524 (2022). https://doi.org/10.1007/s10658-022-02575-x
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10658-022-02575-x