Abstract
Antifungal activity of plant-derived compounds can be exploited in disease management systems to improve sustainability and replace synthetic molecules. In this study, four crude hydroethanolic extracts of leaves, collected from Italian Solanum spp. landraces, were selected to evaluate their ability to suppress Sclerotinia minor Jagger, a great threat to lettuce production worldwide. In vitro fungal development was inhibited by Solanum melongena L. and S. aethiopicum L. extracts showing a dose-dependent correlation. At the highest concentration tested in the current experiments (45 mg mL−1) antifungal activity caused up to 90% growth reduction. The exposure of the fungus to S. aethiopicum extracts incited pronounced changes in the hyphal morphology as observed under light microscopy. Consistently, under laboratory conditions, in planta application of the active extracts on lettuce significantly reduced Sclerotinia drop disease in comparison to non-treated controls. Phytochemical composition was determined by liquid chromatography/mass spectrometry (LC/MS) analyses. Four secondary metabolites differentially present in the extracts, identified as n-caffeoylputrescine, chlorogenic acid, isoquercitrin and solasodoside A, are hypothesized to play a crucial role in mechanisms underlying biological effects of extracts. PCA analysis showed positive correlations of these compounds with the overall control ability of the extracts. The results indicated that foliar material from cultivated eggplant could be suitable to produce biological-based remedies for controlling plant diseases.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Sclerotinia minor Jagger is the causal agent of Sclerotinia lettuce drop disease that is common in temperate agricultural areas worldwide, producing severe economic losses subsequent to significant quantitative yield abatements. This ascomycete is able to attack all organs of the plant; generally, the infections start from lower leaves and collar that are most exposed to overwintering inoculum in soil. Under favorable environmental conditions, the disease spreads through the remaining healthy parts of the plants resulting in watery rots, plantlet damping-off or lettuce head collapse in later outbreaks. Infected plants initially exhibit hydropic parts and, successively, are covered by mycelium and numerous small-sized sclerotia; finally, they become necrotic and following become completely desiccated.
The effective management of Sclerotinia lettuce drop disease has been achieved mainly through reiterated fungicide applications both for soil disinfection and for plant protection. While, some non-chemical methods to counteract this disease, including the use of the biocontrol agent Coniothyrium minitans and/or the adoption of cultural strategies (i.e. rotations, soil solarisation, organic amendments, optimized irrigation systems), have been proposed (Jones et al. 2014; Pane et al. 2012; Subbarao et al. 1997). However, Sclerotinia lettuce drop disease remains difficult to control, and new tools for its sustainable management are desirable.
Alternatives to the current strategies adopted for controlling plant pathogens are also leading to consider the relevant potential of plant-derived natural antifungals (Pane et al. 2013). Simple solvent soaking methods allow extraction from plant materials of a cocktail of bioactive compounds exhibiting antimicrobial properties, known as phytochemicals, belonging to different chemical classes such as polyphenols, saponins and alkaloids (Yun et al. 2015; Pusztahelyi et al. 2015; Ono et al. 2006; Roddick et al. 1990). Bioactive plant compounds have potential in disease control applications because they are biodegradable, refractory to the occurrence of resistance in fungi and may have a broad spectrum of activity (da Cruz Cabral et al. 2013). Microscopy studies have shown general disorders and deviations from the normal morphology of fungal structures as detrimental and direct effects of plant extracts (Hashem 2011) as in the case of Aspergillus niger under the effect of Anethum graveolens essential oil (Tian et al. 2011), and Rhizoctonia solani treated with active compounds from Platycladus orientalis extracts (Wang et al. 2014). Due to their antifungal activity, plant extracts were proposed for the effective management of several plant diseases, including potato Verticillium wilt (Uppal et al. 2008), rice brown spot caused by Bipolaris oryzae (Nguefack et al. 2013), chilli damping-off due to Pythium aphanidermatum (Muthukumar et al. 2010) and lupine Fusarium wilt (Abdel-Monaim et al. 2011).
Currently, the preparation of antifungal extracts makes large use of biomaterials from medicinal plants, because of their particular richness in bioactive constituents (Gahukar 2012). It also has gained increasing attention for phytochemistry of agro-industrial wastes to be valorised as sources of antimicrobial compounds (Dias et al. 2015). Elsherbiny et al. (2016) provided evidence for the potential application of pomegranate peel methanolic extracts in reducing incidence and severity of dry rot of potato caused by Fusarium sambucinum. Rosado-Álvarez et al. (2014) characterized flavonoid-enriched extracts from asparagus agricultural by-products for their significant in vitro anti-Fusarium activity. Additionally, glucosinolate-containing vegetal wastes from the biofuel chain can find a new life in sustainable plant disease management due to their isothiocyanate-based biofumigating properties (De Corato et al. 2015).
The current study aims to investigate the antifungal properties of extracts produced from leaves of Italian traditional eggplants, including Solanum melongena and S. aethiopicum landraces cultivated in two agricultural areas of Southern Italy, such as Campania and Basilicata regions, respectively. Research proposal concerns the valorisation of residues from these cropping systems through the individuation of a potential anti-Sclerotinia tool.
The antifungal potential of Solanum melongena-sourced foliar extracts against human pathogenic dermatophytes has been evidenced (Das et al. 2010). Whereas, the antifungal properties of S. aethiopicum were confirmed by some authors who have reported high levels of steroidal glycoalkaloids in the fruit (Mennella et al. 2010). Moreover, this allied species was also used to carry out the successful incorporation of fungi resistance/tolerance traits into the eggplant gene pool and the introgression lines developed showed a wide range of variation in their phytochemical content (Toppino et al. 2008).
However, application of eggplant extracts against crop pathogens must be further investigated.
Therefore, the objectives of this work were the following: (i) to study the effects of hydroethanolic foliar extracts from cultivated eggplant materials against mycelial growth of S. minor; (ii) to evaluate the efficacy of plant-derived extracts in controlling Sclerotinia lettuce drop disease under laboratory conditions; and (iii) to assess the phytochemical profile of extracts as well as evaluate the correlation with the observed biological activity.
Materials and methods
Preparation of plant extracts
The plant materials consisted of five foliar samples of local accessions of S. melongena Campania’s landraces, including “Lunga Napoletana” (LN), three accessions of “Violetta Tonda” (VT1, VT2 and VT3) and “A Grappolo” (AG) eggplants, and two leaf samples of scarlet eggplant (S. aethiopicum) collected at Rotonda (Basilicata, Southern Italy Region), at two sites: Cassaneto (RC) and Piano dell’Incoronata (RI). A fruit sample (RF) of this last accession was also included. Extraction protocol was based on those previously described (Uppal et al. 2008; Sayago et al. 2012; Pane et al. 2016). Dried (70 °C) and powdered plant materials (10 g) were extracted by soaking in 100 mL 50% vol. ethanol-water solution for 24 h at room temperature. Then, extracts were filtered (Whatman No. 1 filter paper), centrifuged (10,000×g, 2 min.) and vacuum-dried (rotary evaporator at 30 °C) to remove the solvent. The residues were re-suspended in distilled water at a final concentration of 150 mg mL−1 to prepare the extract standard batches which, after filter-sterilization (0.22 μm filter pore size), were stored at −20 °C until use.
Fungal pathogen
Plant fungal pathogen used in this study was Sclerotinia minor, isolated from a symptomatic lettuce (Lactuca sativa L.) plant. The fungus was maintained on slants of PDA (potato dexstrose agar, Difco) at 20 °C at CREA-Centro di Ricerca per l’Orticoltura (Pontecagnano, Italy).
In vitro assessment of antifungal activity
To assess the inhibitory activity of foliar eggplant extracts on in vitro growth of S. minor, each filtrate was added into Czapek Dox medium (0.1×), to reach the final concentration of 20 mg mL−1, and poured into Petri dishes (3 cm diameter). A fungal plug (0.5 cm diameter) from a Czapek sub-culture was placed mycelia side against the agar on the centre of each plate; while, Czapek Dox medium without amendments was used as control. Plates were inoculated in triplicate and incubated in the dark at 25 °C. The radius of each colony was measured at the end of the experiment when the fungus in the control plates reached the edge. The percentage of fungal growth inhibition was calculated with the formula: 100 × [(radius on the control - radius on the treatment)/radius on the control]. Afterwards, further in vitro experiments was made by testing fewer extracts, chosen on the basis of the initial screening, at final concentrations of 7.5, 22.5 and 45 mg mL−1. The experiment was carried out two times.
Samples of mycelia, taken from the edge of the expanding fungal colony in the plates treated with 22.5 mg mL−1 of each extract, were stained with trypan blue and examined under light microscopy to record any structural abnormalities. The observations were compared with those made on the control sample lacking amendments.
Control assay of Sclerotinia lettuce drop disease
Entire one-month old nursery lettuce plants (cv. Penelope), grown and maintained in a cell-growing tray on peat-based media, were used to test the ability of the chosen extracts in the control of Sclerotinia drop disease. Two separate assays were carried out making infection points over the petiole or the leaf as described by Smith et al. (2008) and Chen et al. (2014), respectively. One inoculation point per plant was pre-treated by pipetting 10 μL of extract dissolved in distilled water at concentrations of 22.5 and 45 mg mL−1 while, sterile distilled water was used as the control. After air-drying, a 5-mm mycelial plug from two-weeks-old S. minor Czapek culture was laid over the treated area, on petiole or on leaf, to achieve fungal inoculation. Then, the cell-growing tray carrying the plants was placed in a plastic chamber to maintain a high relative humidity and incubated in a climatic chamber at 25 ± 2 °C. The experimental design was a randomized complete block with ten replicates (plants) per treatment and two factors (extract type and concentration). One week after the inoculation, disease severity was assessed by measuring the lesion length on the petiole, or the diameter of the lesion area around each infected point on the leaf surface. The experiments were carried out two times.
Phytochemical analysis of the extracts
All the extracts were analyzed in triplicate using reversed phase liquid chromatography coupled to a photodiode array detector and to an ion trap mass spectrometry (LC–PDA–MS) system. Such system consisted of an ultra-performance liquid chromatography (UPLC) Dionex Ultimate 3000 model coupled to an LTQ XL mass spectrometer (Thermo Fisher Scientific, Sunnyvale, CA, USA). A 100 μL aliquot of sample was diluted with Water LC/MS grade (1:10) and then 4 μL were injected onto a Luna C18 (100 × 2.0 mm, 2.5 μm particle size) column equipped with a SecurityGuard guard column (3.0 × 4.0 mm) (Phenomenex, Torrance, CA, USA). The separations were carried out using a binary gradient of ultrapure water (A) and acetonitrile (B), both acidified with 0.1% (v/v) formic acid, with a flow rate of 0.22 mL min−1. The initial solvent composition consisted of 95% (v/v) of A and 5% (v/v) of B; increased linearly to 25% A and 75% B in 25 min and maintained for 1 min; returned to 95% of A in 1 min. The column was equilibrated to 95% A and 5% B for 11 min before the next injection. The analysis lasted for 38 min and the column temperature was set to 40 °C. Mass spectra were obtained in positive ion mode over the range m/z 80–1400. The capillary voltages were set at 9.95 V and the source temperature was 34 °C. The putative identification of some metabolites was confirmed by m/z data from authentic, distinct standards such as chlorogenic acid (CA) and isoquercitrin (IS) (Sigma-Aldrich, St. Louis, MO, USA), solamargine (SM) and solasonine (SS) (kindly provided by Prof. Adelia Emilia de Almeida). Solasodoside A (SDA), n-caffeoylputrescine (CP) and malonyl-solamargine (MSM) were tentatively identified based on MS/MS. fragments identity and retention time (r.t.) by matching the resulting spectra with those reported in Solanum melongena secondary metabolite data-bases (Wu et al. 2013; Delledonne et al. 2014). Xcalibur software (Thermo Fisher Scientific, Sunnyvale, CA, USA) was used to control all instruments and for data acquisition and data analysis.
Data analysis
Probit analysis was performed to calculate the effective concentrations causing 50% (EC50) and 90% (EC90) inhibition of in vitro pathogen growth in comparison with the control. The Chi-square test was performed to compare observed with expected dose-response dataset and the resulting p level (> 0.05), associated to each EC value, indicates the goodness of fit between the distributions; analysis of variance (ANOVA) on the means of probit and Tukey’s HSD test (p ≤ 0.05), instead, were used for further statistical evaluations. Data concerning the effects of the extracts on the control of Sclerotinia lettuce drop disease, were subjected to ANOVA and means were separated by Tukey’s multiple range test (p ≤ 0.05). The percentage data were arcsine transformed before undergoing statistical analysis; non-transformed means are presented. In order to determine the extent of differentiation among the extracts in relation to their antifungal activity, all analysed characteristics were subjected to principal component analysis (PCA). Pearson correlation coefficients were calculated for biochemical parameters from a regression of genotype mean values using the statistical software JMP v7.0 (SAS Institute 2007).
Results
In vitro antifungal activity of plant extracts
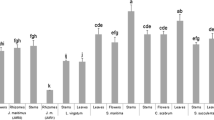
Results of the initial screening for the in vitro antifungal activity of all eggplant extracts against S. minor are presented in Fig. 1. RI, RC, AG and VT3 foliar samples were found the most inhibitive extracts reducing by 65.9–76.5% the development of the fungus compared to the untreated control, while, LN, VT1 and VT2 showed moderate activity producing 36–51% of inhibition. The extract from scarlet eggplant fruit proved weak. Afterwards, the three extracts that have showed the best performances in the initial screening, AG, RC and RI, and the least antifungal foliar extract LN, were chosen for further investigations.
Effect of extracts from leaves of Lunga Napoletana (LN), three accessions of Violetta Tonda (VT1, VT2 and VT3), A Grappolo (AG), and Rotonda’s leaf (RC and RI) and fruit (RF) samples of eggplant landraces, applied at dose of 20 mg mL−1 on mycelial growth of Sclerotinia minor. Bars are the mean values ± standard deviation; different lowercase letters indicate significant differences among bars according to Tukey HSD test (p ≤ 0.05)
These four extracts showed a significant dose-dependent pattern of activity in further in vitro experiments (Fig. 2). RI, RC and AG extracts at 45 mg mL−1 in media determined a significant inhibition of Sclerotinia’s radial growth. RI extract proved to be the most effective at levels of 22.5 mg mL−1. Extracts, when used at lower dose, proved ineffective. The values of EC50 and EC90 (Table 1) confirmed the effectiveness of the RI, RC and AG extracts to inhibit the pathogen.
Effect of foliar extracts of Lunga Napoletana (LN), A Grappolo (AG), and Rotonda’s (RC and RI) eggplant landraces, applied at doses of 7.5, 22.5 and 45 mg mL−1 on mycelial growth of Sclerotinia minor. Bars are the mean values ± standard deviation; different lowercase letters indicate significant differences among bars according to Tukey HSD test (p ≤ 0.05)
Light microscopic observations revealed morphological abnormalities in hyphae of the fungus exposed to the extracts (Fig. 3). Alterations, including swelling of hyphae and, subsequently, formation of irregular shapes, were observed under exposure to all extracts; in addition, shrinking and branching were more marked in RI and RC treated samples.
Assessment of control of Sclerotinia lettuce drop disease
The four hydroethanolic eggplant extracts showed significant control ability of Sclerotinia lettuce drop disease in the bioassays performed by artificially infecting both the petioles and the leaves of plants. The lesion lengths on treated petioles was reduced, on average, from 32 to 52%, when compared to the water control (Fig. 4). On leaves, mean lesion size was reduced less consistently, in the wide range 19–75% of the control (Fig. 5). Extracts RI and RC, when tested at the highest concentration, consistently showed the greatest reduction of disease severity. A general positive dose-effect trend on activity of the extract was also observed.
Sclerotinia lettuce drop severity on lettuce petiole treated with Lunga Napoletana (LN), A Grappolo (AG), and Rotonda’s (RC and RI) eggplant foliar extracts, applied at doses of 22.5 and 45 mg mL−1, to untreated control (CTRL). Experiments I and II are presented separately. Bars are the mean values ± standard deviation; different lower-case letters indicate significant differences between bars according to Tukey HSD test (p ≤ 0.05)
Sclerotinia lettuce drop severity on lettuce leaves treated with Lunga Napoletana (LN), A Grappolo (AG), and Rotonda’s (RC and RI) eggplant foliar extracts, applied at doses of 22.5 and 45 mg mL−1, to untreated control (CTRL). Experiments I and II are presented separately. Bars are the mean values ± standard deviation; different lower-case letters indicate significant differences between bars according to Tukey HSD test (p ≤ 0.05)
However, since a significant experimental effect was found (P < 0.01), data of repeated experiments were not pooled; thus, they are presented separately.
Metabolomic analysis of extracts
As expected, the analysis of chromatograms evidenced different elution profiles of the two Rotonda’s materials compared to LN and AG ones; a typical HPLC profile is reported in Fig. 6. Differentially present semi-polar metabolites were revealed by LC-MS analysis in these extracts (Table 2) and tentatively identified as: i. CP, a conjugated polyamine (r. t. 3.05, m/z = 251); ii. CA, a monomeric phenol (r. t. 7.97, m/z = 355); iii. IS, a flavonoid (r. t. 10.9, m/z = 465); iv. SS, SM and MSM, three glycoalkaloids (r. t. 11.92 and m/z = 884, r. t. 12.21 and m/z = 868, r. t. 12.83 and m/z = 954, respectively); and v. SDA, a saponin (r. t. 14.05 and m/z = 1031). The semi-quantitative analysis showed for Rotonda ecotypes a higher amount of CP, CA and SDA with respect to the other genotypes. IS was only detected in LN, RI and RC extracts, but not in AG. On the other hand, the three glycoalkaloids were only detected in LN, whereas CP was only found in the foliar extracts of Rotonda ecotypes.
Regression and principal component analysis
The statistical relationships between biochemical compound levels in leaf material of the different Solanum genotypes studied are reported in a correlation matrix (Table 3). Most biochemical traits were positively correlated with each other, with the exception of CP, CA and SDA, which were negatively correlated to SS, SM and MSM (p < 0.05). The strongest positive and negative correlations were found respectively between the glycoalkaloids and CA vs SDA (1.00) and between the glycoalkaloids and CP (−0.58). Positive correlations, albeit not significant, were detected between glycoalkaloids and IS (0.26).
An ordination biplot for PCA analysis of data is shown in Fig. 7. The first variable (PC1) accounted for 60.53%, whereas the second variable (PC2) accounted for 32.54% of the total variance, respectively. Along the PC1 axis, the foliar extracts of Rotonda eggplant clustered together and were distant from AG and LN. PC1-variable correlation resulted significant (R > 0.60) and positive for in vitro fungal growth, for in planta disease severity (with two exceptions) and for the presence of glycoalkaloids; conversely, was negative for concentration of chlorogenic acid, isoquercitrin, solasodoside A and n-caffeoylputrescine. Instead, only five out of eight cases of in planta disease severity resulted significantly (R > 0.60) and negatively correlated with PC2.
Discussion
Results indicated that the hydroethanolic foliar extracts from landraces of S. melongena and S. aethiopicum could be promising to develop eco-friendly antifungal tools for counteracting Sclerotinia lettuce drop disease. To the best of our knowledge, this is the first report showing the efficacy of extracts from cultivated eggplant biomaterials against one of the most deleterious diseases of lettuce.
All eggplant foliar extracts were found to be highly active in reducing the in vitro growth of S. minor with a dose-dependent behaviour, gaining a strong inhibition upon exposure of the fungus at the maximum dosage tested (45 mg mL−1). The values of EC50 and EC90 indicate better effectiveness of AG, RC and RI extracts in inhibiting the pathogen growth. Moreover, the visualizations by light microscopy of the expanding hyphae exposed to extracts evidenced the occurrence of morphological abnormalities likely due to a direct toxicity exerted by the phytocomplexes extracted from foliar materials (Gatto et al. 2011). Extracts from scarlet eggplant proved the most detrimental to the fungus, followed by AG and LN extracts. In a recent study, disorders to S. minor hyphal structures were also documented after treatment with a suppressive wild Capsicum annuum foliar extract enriched in polyphenolic compounds, including gallic, chlorogenic, caffeic, and ferulic acids, catechin, rutin and hyperoside (Pane et al. 2015), suggesting a particular sensitivity of the fungus versus plant-extracted constituents. These findings agree with Soylu et al. (2007) who observed similar effects on Sclerotinia sclerotiorum upon treatments with oregano and fennel essential oils and with Plodpai et al. (2013) who observed Rhizoctonia solani completely collapsed following exposure to Desmos chinensis extracts.
In a previous report, fungicidal effects of Ballota undulata ethanol extract against the dermatophyte Paecilomyces lilacinus have been ascribed to the potential involvement of 35 compounds, singly or in combination with others, in alterations of hyphal structures such as swelling and decrease in cytoplasmic content (Hashem 2011). Recently, Jasso de Rodríguez et al. (2015) identified eight antimicrobial compounds in Rhus muelleri ethanol extract putatively involved in in vitro growth inhibition of Fusarium oxysporum f. sp. lycopersici.
Phytochemical composition of extracts produced in this study was analysed through LC-MS. PCA analysis suggested that the observed anti-Sclerotinia activity might be correlated to four secondary metabolites differentially present in the extracts, identified as n-caffeoylputrescine, chlorogenic acid, isoquercitrin and solasodoside A. Biological activity of these compounds has been described and their direct involvement in the mechanisms underlying the antifungal effects of treatments could be hypothesized. The steroidal glycosidic saponin, solasodoside A, has been previously isolated from fractioned methanolic extract of underground parts of Solanum sodomaeum and showed a slight cell anti-proliferative activity (Ono et al. 2006). Cruz-Cruz et al. (2010) purified a similar steroidal saponin from water infusion of banana leaves, which was strongly active against the phytopathogenic fungus Mycosphaerella fijiensis. The polyphenol chlorogenic acid is an ester between caffeic acid and L-quinic acid, ubiquitariuos in many vegetal species, including solanaceous plants (Ahmad et al. 2013). It has been described as an important antifungal secondary metabolite from tomatoes (Ruelas et al. 2006). Isoquercitrin is a flavonoid found in Aster yomena that has been reported to explicate antimicrobial effects through membrane disturbance mechanisms (Yun et al. 2015). According to the current findings, the role of antimicrobial activity of plant-derived polyphenolic compounds has been widely investigated (Martins et al. 2015) and, in some cases, linked to the ability of extracts in controlling plant diseases (Sayago et al. 2012; Trigui et al. 2013; Pane et al. 2016). N-caffeoylputrescine, which was found only in Rotonda’s samples, is a conjugated polyamine belonging to the sub-group of hydroxycinnamic acid amides indicated to be involved in plant defense responses to pathogens (Walters 2003) through jasmonate signalling (Chen et al. 2006). Additionally, in the present work, other compounds identified as malonyl-solamargine, solamargine, and solasonine, were found only in LN sample. Solamargine and Solasonine are the major eggplant steroidal glycoalkaloids and may have the role to protect eggplant from attack by fungi, bacteria, insects, nematodes, slugs, etc. (Cipollini and Levey 1997). They consist of two structural components – a hydrophilic carbohydrate chain attached to a hydrophobic steroidal skeleton (aglycone) (Schreiber 1968). Although, antifungal activity of glycoalkaloids has been previously reported (Roddick et al. 1990), the presence of these detected compounds did not incite any increase in extract suppressiveness.
However, the role of all detected molecules in conferring antifungal properties to the extracts must be verified in further studies.
Overall, findings could suggest the hypotheses that the control effectiveness may be attributed to: i) differences concerning the presence/absence and the distinct amount of each individual compound, and/or ii) synergic/additive work of the antifungal chemical constituents (Gatto et al. 2011; Sayago et al. 2012).
Results concerning a significant reduction of symptoms obtained by treatments with extracts are promising for the development of plant-derived remedies to diseases as a new frontier among non-synthetic plant protectants and use of waste materials. This work contributes to the ongoing research in the field of plant natural antifungals. It demonstrated that cultivated eggplant foliar materials could be used as a natural feedstock to easily produce crude extracts with antifungal phytochemical composition. The extracted phytochemical complex carrying previously identified antimicrobial compounds was likely to contribute to the activity against Sclerotinia lettuce drop disease. Moreover, the individuation of extract-marker compounds linked to the biological activity may be useful to follow fluctuations in the plant accumulation of active molecules due to environmental factors. Eggplant extracts are promising to meet the requirements for sustainability of agricultural systems through the purpose of low impactful control methods and the use of recycling biowastes.
References
Abdel-Monaim, M. F., Abo-Elyousr, K. A. M., & Morsy, K. M. (2011). Effectiveness of plant extracts on suppression of damping-off and wilt diseases of lupine (Lupinus termis Forsik). Crop Protection, 30, 185–191.
Ahmad, A., Shafique, S., & Shafique, S. (2013). Cytological and physiological basis for tomato varietal resistance against Alternaria alternata. Journal of the Science of Food and Agriculture, 93, 2315–2322.
Chen, H., Jones, A. D., & Howe, G. A. (2006). Constitutive activation of the jasmonate signalling pathway enhances the production of secondary metabolites in tomato. FEBS Letters, 580, 2540–2546.
Chen, Y., Gao, X., Chen, Y., Qin, H., Huang, L., & Han, Q. (2014). Inhibitory efficacy of endophytic Bacillus subtilis EDR4 against Sclerotinia sclerotiorum on rapeseed. Biological Control, 78, 67–76.
Cipollini, M. L., & Levey, D. J. (1997). Antifungal activity of Solanum fruit glycoalkaloids: implications for frugivory and seed dispersal. Ecology, 78, 799–809.
Cruz-Cruz, C. A., Ramírez-Tec, G., García-Sosa, K., Escalante-Erosa, F., Hill, L., Osbourn, A. E., & Peña-Rodríguez, L. M. (2010). Phytoanticipins from banana (Musa acuminate cv. Grande Naine) plants, with antifungal activity against Mycosphaerella fijiensis, the causal agent of black Sigatoka. European Journal of Plant Pathology, 126, 459–463.
da Cruz Cabral, L., Fernández Pinto, V., & Patriarca, A. (2013). Application of plant derived compounds to control fungal spoilage and mycotoxin production in foods. International Journal of Food Microbiology, 166, 1–14.
Das, J., Prasad Lahan, J., & Srivastava, R. B. (2010). Solanum melongena: a potential source of antifungal agent. Indian Journal of Microbiology, 50, 62–69.
De Corato, U., Pane, C., Bruno, G. L., Cancellara, F. A., & Zaccardelli, M. (2015). Co-products from a biofuel production chain in crop disease management: a review. Crop Protection, 68, 12–26.
Delledonne, M., Dal Molin, A., Minio, A., Ferrarini, A., Tononi, P., Zamperin, G., Toppino, L., Sala, T., Barchi, L., Comino, C., Acquadro, A., Portis, E., Rinaldi, R., Scaglione, D., Francese, G., D’Alessandro, A., Mennella, G., Fantini, E., Pietrella, M., Sulli, M., Lanteri, S., Rotino, G. L., & Giuliano, G. (2014). A high quality eggplant (Solanum melongena L.) genome draft and its use for mapping metabolic QTLs. Proceedings of the 11th Solanaceae Conference, SOL 2014. November 2–6, 2014 Porto Seguro–Bahia, Brazil. pp 99
Dias, C., Domínguez-Pereles, R., Aires, A., Teixeira, A., Rosa, E., Barros, A., & Saavedra, M. J. (2015). Phytochemistry and activity against digestive pathogens of grape (Vitis vinifera L.) stem’s (poly)phenolic extracts. LWT–Food Science and Technology, 61, 25–32.
Elsherbiny, E. A., Amin, B. H., & Baka, Z. A. (2016). Efficiency of pomegranate (Punica granatum L.) peels extract as a high potential natural tool towards Fusarium dry rot on potato tubers. Postharvest Biology and Technology, 111, 256–263.
Gahukar, R. T. (2012). Evaluation of plant-derived products against pests and diseases of medicinal plants: a review. Crop Protection, 42, 202–209.
Gatto, M. A., Ippolito, A., Linsalata, V., Cascarano, N. A., Nigro, F., Vanadia, S., & Di Venere, D. (2011). Activity of extracts from wild edible herbs against postharvest fungal diseases of fruit and vegetables. Postharvest Biology and Technology, 61, 72–82.
Hashem, M. (2011). Antifungal properties of crude extracts of five egyptian medicinal plants against dermatophytes and emerging fungi. Mycopathologia, 172, 37–46.
Jasso de Rodríguez, D., Trejo-González, F. A., Rodríguez-García, R., Díaz-Jimenez, M. L. V., Sáenz-Galindo, A., Hernández-Castillo, F. D., Villareal-Quintanilla, J. A., & Peña-Ramos, F. M. (2015). Antifungal activity in vitro of Rhus muelleri against Fusarium oxysporum f. Sp. lycopersici. Industrial Crops and Products, 75, 150–158.
Jones, E. E., Mead, A., & Whipps, J. M. (2014). Effect of inoculum type and timing of application of Coniothyrium minitans on Sclerotinia sclerotiorum: control of sclerotinia disease in glasshouse lettuce. Plant Pathology, 53, 611–620.
Martins, N., Barros, L., Henriques, M., Silva, S., & Ferreira, I. C. F. R. (2015). Activity of phenolic compounds from plant origin against Candida species. Industrial Crops and Products, 74, 648–670.
Mennella, G., Rotino, G. L., Fibiani, M., D’Alessandro, A., Francese, G., Toppino, L., Cavallanti, F., Acciarri, N., & Lo Scalzo, R. (2010). Characterization of health-related compounds in eggplant (Solanum melongena L.,) lines derived from introgression of allied species. Journal of Agricultural and Food Chemistry, 58, 7597–7603.
Muthukumar, A., Eswaran, A., Nakkeeran, S., & Sangeetha, G. (2010). Efficacy of plant extracts and biocontrol agents against Pythium aphanidermatum inciting chili damping-off. Crop Protection, 29, 1483–1488.
Nguefack, J., Wulff, G. E., Lekagne Dongmo, J. B., Fouelefack, F. R., Fotio, D., Mbo, J., & Torp, J. (2013). Effect of plant extracts and an essential oil on the control of brown spot disease, tillering, number of panicles and yield increase in rice. European Journal of Plant Pathology, 137, 871–882.
Ono, M., Nishimura, K., Suzuki, K., Fukushima, T., Igoshi, K., Yoshimitsu, H., Ikeda, T., & Nohara, T. (2006). Steroidal glycosides from the underground parts of Solanum sodomaeum. Chemical and Pharmaceutical Bulletin, 54, 230–233.
Pane, C., Villecco, D., Pentangelo, A., Lahoz, E., & Zaccardelli, M. (2012). Integration of soil solarisation with Brassica carinata seed meals amendment in a greenhouse lettuce production system. Acta Agriculturae Scandinavica-Section B, Soil Plant Science, 62, 291–299.
Pane, C., Villecco, D., Roscigno, G., De Falco, E., & Zaccardelli, M. (2013). Screening of plant derived antifungal substances useful for the control of seedborne pathogens. Archives of Phytopathology and Plant Protection, 46, 1533–1539.
Pane, C., Fratianni, F., Caputo, M., Parisi, M., Nazzaro, F., & Zaccardelli, M. (2015). Antifungal activity of wild Capsicum foliar extracts containing polyphenols against the phytopathogens Alternaria alternata, Rhizoctonia solani, Sclerotinia minor and Verticillium dahliae. In A. Mendez-Vilas (Ed.), Multidisciplinary approach for studying and combating microbial pathogens (pp. 34–38). Boca Raton: BrownWalker Press.
Pane, C., Fratianni, F., Parisi, M., Nazzaro, F., & Zaccardelli, M. (2016). Control of Alternaria post-harvest infections on cherry tomato fruits by wild pepper phenolic-rich extracts. Crop Protection, 84, 81–87.
Plodpai, P., Chuenchitt, S., Petcharat, V., Chakthong, S., & Voravuthikunchai, S. P. (2013). Anti-Rhizoctonia solani activity by Desmos chinensis extracts and its mechanism of action. Crop Protection, 43, 65–71.
Pusztahelyi, T., Holb, I. J., & Pócsi, I. (2015). Secondary metabolites in fungus-plant interactions. Frontiers in Plant Science, 6, 573. doi:10.3389/fpls.2015.00573.
Roddick, J. G., Rijnenberg, A. L., & Weissenberg, M. (1990). Membrane-disrupting properties of the steroidal glycoalkaloids solasonine and solamargine. Phytochemistry, 29, 1513–1518.
Rosado-Álvarez, C., Molinero-Ruiz, L., Rodríguez-Arcos, R., & Basallote-Ureba, M. J. (2014). Antifungal activity of asparagus extracts against phytopathogenic Fusarium oxysporum. Scientia Horticulturae, 171, 51–57.
Ruelas, C., Tiznado-Hernández, M. E., Sánchez-Estrada, A., Robles-Burgueno, M. R., & Troncoso-Rojas, R. (2006). Changes in phenolic acid content during Alternaria alternate infection in tomato fruit. Journal Phytopathological, 154, 236–244.
SAS Institute (2007). JMP statistics and graphics guide. Cary: SAS Institute.
Sayago, J. E., Ordoñez, R. M., Negrillo Kovacevich, L., Torres, S., & Isla, M. I. (2012). Antifungal activity of extracts of extremophile plants from the argentine puma to control citrus postharvest pathogens and green mold. Postharvest Biology and Technology, 67, 19–24.
Schreiber, K. (1968). Steroid alkaloids: the Solanum group. In R. H. F. Manske (Ed.), The alkaloids–chemistry and physiology (Vol. X, pp. 1–192). New York: Academic Press.
Smith, D. L., Garrison, M. C., Hollowell, J. E., Isleib, T. G., & Shew, B. B. (2008). Evaluation of application timing and efficacy of the fungicides fluazinam and boscalid for control of sclerotinia blight of peanut. Crop Protection, 27, 823–833.
Soylu, S., Yigitbas, H., Soylu, E. M., & Kurt, S. (2007). Antifungal effects of essential oils from oregano and fennel on Sclerotinia sclerotiorum. Journal of Applied Microbiology, 103, 1021–1030.
Subbarao, K. V., Hubbard, J. C., & Schulbach, K. F. (1997). Comparison of lettuce diseases and yield under subsurface drip and furrow irrigation. Phytopathology, 87, 877–883.
Tian, J., Ban, X., Zeng, H., Huang, B., He, J., & Wang, Y. (2011). In vitro and in vivo activity of essential oil from dill (Anethum graveolens) against fungal spoilage of cherry tomatoes. Food Control, 22, 1992–1999.
Toppino, L., Vale’, G., & Rotino, G. L. (2008). Inheritance of Fusarium wilt resistance introgressed from Solanum aethiopicum Gilo and Aculeatum groups into cultivated eggplant (S. melongena) and development of associated PCR-based markers. Molecular Breeding, 22, 237–250.
Trigui, M., Hsouna, A. B., Hammami, I., Culioli, G., Ksantini, M., Tounsi, S., & Jaoua, S. (2013). Efficacy of Lawsonia inermis leaves extract and its phenolic compounds against olive knot and crown gall diseases. Crop Protection, 45, 83–88.
Uppal, A. K., El Hadrami, A., Adam, L. R., Tenuta, M., & Daayf, F. (2008). Biological control of potato Verticillium wilt under controlled and field conditions using selected bacterial antagonists and plant extracts. Biological Control, 44, 90–100.
Walters, D. R. (2003). Polyamines and plant disease. Phytochemistry, 64, 97–107.
Wang, H., Wang, J., Peng, X., Zhou, P., Bai, N., Meng, J., & Deng, X. (2014). Control efficacy against rice sheath blight of Platycladus orientalis extract and its antifungal active compounds. European Journal of Plant Pathology, 140, 515–525.
Wu, S.-B., Meyer, R. S., Whitaker, B. D., Litt, A., & Kennelly, E. J. (2013). A new liquid chromatography-mass spectrometry-based strategy to integrate chemistry, morphology, and evolution of eggplant (Solanum) species. Journal of Chromatography A, 1314, 154–172.
Yun, J. E., Lee, H., Ko, H. J., Woo, E. R., & Lee, D. G. (2015). Fungicidal effect of isoquercitrin via inducing membrane disturbance. Biochimica et Biophysica Acta, 1848, 695–701.
Acknowledgements
This work was carried-out within the Project: “Valorizzazione di produzioni ortive campane di eccellenza con strumenti di genomica avanzata (GenHort)”, funded by Italian Ministry of Education, University and Research - MIUR through the EU program P.O.N. Research and Competitiveness 2007-2013 (PON_02_00395_3215002). The authors thank dr. Rocco Sileo from ALSIA (Basilicata Region), for providing Rotonda’s eggplant samples, and Prof. Adelia Emilia de Almeida of the Universidade Estadual Paulista, São Paulo, Brazil, for the purified solamargine and solasonine supply.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pane, C., Francese, G., Raimo, F. et al. Activity of foliar extracts of cultivated eggplants against sclerotinia lettuce drop disease and their phytochemical profiles. Eur J Plant Pathol 148, 687–697 (2017). https://doi.org/10.1007/s10658-016-1126-0
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10658-016-1126-0