Abstract
In the mid-2000s a survey was conducted to evaluate fish mercury in lakes across New York State. Approximately 10 years later a second survey examining adult sportfish from 103 lakes and reservoirs was conducted to evaluate the response of fish mercury to recent declines in US mercury emissions. Of those lakes, 43 were part of the earlier survey and were examined to determine if mercury concentrations in four popular sport species, Yellow Perch, Walleye, and Small- and Largemouth Bass, declined in response to decreasing emissions. Water samples were also collected at 35 of these lakes and analyzed for mercury, methylmercury and other analytes. The Adirondack and Catskill regions remain biological mercury hotspots with elevated concentrations in fish. The most widely sampled species, Yellow Perch, showed significant increases in mercury in the Northeast and West regions of New York State over the past decade. The increases in Yellow Perch mercury is not consistent with significant reductions in water concentrations of both total and methylmercury observed corresponding in lake water samples. This discrepancy suggests watershed and in-lake processes beyond mercury emissions, such as recovery from acid deposition, impacts from climate change, or changes in food web structure may be controlling fish mercury concentrations. These results demonstrate a need for a consistent, long-term program to monitor fish mercury to inform the status of mercury contamination in New York State.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The bioaccumulation and biomagnification of mercury (Hg) is a major concern across New York State (NYS; Dittman et al. 2010) and globally. In reducing environments, Hg can be converted to methylmercury (MeHg) by groups like sulfate- or iron-reducing bacteria or archaea (Gilmour et al. 1998; Kerin et al. 2006). This organic form of Hg strongly bioaccumulates and biomagnifies in aquatic ecosystems (Benoit et al. 2002). Concentrations of MeHg biomagnify along food chains resulting in levels that can pose neurotoxicological risks to humans and fish-eating birds and mammals (Driscoll et al. 2007b, 2013; Evers et al. 2008). The consequences of Hg exposure on human health and the environment are globally recognized through international efforts to reduce Hg emission and exposure via the Minimata Convention (Minimata Convention on Mercury 2017).
The Mercury and Air Toxics Standard (MATS) was promulgated in 2012 and implemented in 2015, addressing the need for further emissions reductions by requiring a 90% reduction in Hg emissions from energy generation facilities (US EPA 2017). Coal-fired power plants are the largest source of US Hg emissions accounting for ~48% of Hg emissions in 2015 (Streets et al. 2019). MATS and earlier air quality management regulations have resulted in decreases in Hg emissions (Zhang et al. 2016; Streets et al. 2019). These decreases in regional emissions have coincided with atmospheric Hg concentrations of gaseous elemental Hg and reactive gaseous Hg (Zhou et al. 2017) and atmospheric wet and litter Hg deposition(Gerson et al. 2017; Mao et al. 2017; Zhou et al. 2018; Ye et al. 2019) in New York State and the northeastern US Mercury inputs to terrestrial and aquatic ecosystems largely occur due to atmospheric deposition (Fitzgerald et al. 1998). Concentrations of Hg in precipitation and wet Hg deposition have been declining in response to reductions in North American emissions (Prestbo and Gay 2009; Zhang et al. 2016; Mao et al. 2017). However it has recently been demonstrated that global emissions and long-range transport are becoming increasingly important to Hg deposition in North America (Weiss-Penzias et al. 2016; Streets et al. 2019).
Once deposited, transport of Hg and MeHg through the terrestrial to aquatic environments occurs largely in association with dissolved organic carbon (DOC) or suspended particulate matter (Dittman et al. 2010). The relationship of DOC with MeHg is complex. Natural DOC has been shown to facilitate the transport and formation of MeHg (Graham et al. 2012; Bravo et al. 2017; Herrero Ortega et al. 2018; Jiang et al. 2018), but also reduces the extent of bioaccumulation (Gorski et al. 2008; Chiasson-Gould et al. 2014; Jeremiason et al. 2016).
With over 4000 lakes in NYS, measurement of fish Hg concentrations is important to track environmental, economic and health impacts. A survey in the mid-2000s of 131 lakes divided NYS into three broad regions (Southeast, Southeast and West) finding 53 lakes with fish Hg concentrations high enough for the NYS Department of Health to issue consumption advisories while considering USA FDA and USA EPA guidelines (1 µg/g and 0.3 µg/g, respectively; Simonin et al. 2008b). Fish Hg concentrations were negatively correlated with pH, conductivity, and ANC, and positively correlated with water-Hg concentrations, wetland area, and the presence of dams. Spatially, the Adirondack and Catskill regions of NYS were found to have the highest fish Hg (Simonin et al. 2008b).
In the Northeastern US, concentrations of DOC and fish Hg have been increasing possibly in response to recovery from historical acid deposition (Monteith et al. 2007; Driscoll et al. 2016). This pattern is particularly important in regions like the Adirondack park where decades of sulfate deposition have depleted available calcium (Ca) and other base cations from soils (Warby et al. 2005), delaying recovery from acidification (Driscoll et al. 2001, 2003, 2007a; Chen and Driscoll 2005). A lengthy recovery could result in an extended period of contamination as fish Hg concentrations have been shown to increase with decreases in pH and increases in DOC (Driscoll et al. 2007b).
While recovery from acid deposition appears to be an important factor contributing to elevated fish Hg, anthropogenic forcing of climate can also affect bioaccumulation (Evans et al. 2006; Sebestyen et al. 2009). In the northeastern US, climate change is expected to generate higher annual temperatures, earlier peak streamflow from snowmelt, a longer growing season, increased drought frequency and increased winter precipitation (Hayhoe et al. 2007). Effects of climate change have already been observed in the Northeast with increases in air temperature, precipitation and streamflow (McCabe and Wolock 2002; Hodgkins et al. 2003; Huntington et al. 2004; Climate Science Special Report 2017). These changes could increase lake productivity (Watson et al. 2016), causing eutrophication and extending lake stratification, which would promote Hg methylation due to increases temperature and/or an increase in the anoxic period in lake sediments. An amplified hydrological cycle could also increase wet-dry cycles stimulating methylation rates (Coleman Wasik et al. 2015). While the long term impact of climate change remains unclear (Laudon et al. 2012), there has been some speculation of changing climate in driving increases in fish Hg (Bodaly et al. 1993; Monson et al. 2011).
To evaluate changes in NYS fish Hg concentrations over the last decade, we resurveyed 43 lakes for fish and 35 lakes for water chemistry that were previously sampled in the mid-2000s. In addition, fish were collected from 65 previously unsampled lakes. Using the information from both surveys, we also develope recommendations for a long-term monitoring program to assess the efficacy of Hg emission reduction efforts in New York State. These include locations where fish Hg and ancillary observations might be monitored, the number of waters monitored and frequency of collections.
Methods
Fish sampling and processing
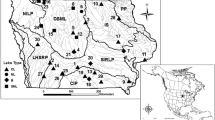
A subset of lakes across New York State previously sampled by Simonin et al. 2008a, b and lakes with multiple years of historical sampling were identified for study. Collections of fish samples began in 2014 and were concluded in 2016. Walleye (Sander vitreus; WE), Smallmouth Bass (Micropterus dolomieu; SMB), Largemouth Bass (Micropterus salmoides; LMB) and Yellow Perch (Perca flavescens; YP) were targeted for collection because of their popularity among sport fishers and the relative abundance of previous Hg observations. Three alternative species, Brook Trout (Salvelinus fontinalis; ST), Chain Pickerel (Esox niger; CP) and Lake Trout (Salvelinus namaycush; LT), were also collected from lakes where the target species were not available or abundant during the present sampling. Working with partners at the New York State Department of Environmental Conservation (DEC), Cornell University and SUNY-Oneonta, fish samples were collected from 108 lakes spread across all regions of the state to evaluate spatial patterns and temporal trends (Fig. 1).
Personnel from the DEC, Cornell University, SUNY Oneonta and Syracuse University collected fish using standard sampling methods including gill netting, trap netting, minnow trapping, angling and electrofishing (when necessary), to obtain a minimum of 10 adult fish samples from targeted species. Collected fish were measured, weighed, individually placed in labeled, food grade plastic bags and kept on ice until analyzed at Syracuse University. Upon arrival, samples were prepared according to DEC methods (standard fillet; skin-on, bone-in, scales removed) and 524 subset fish plugs were also collected according to previously established procedures (Baker et al. 2004; Peterson et al. 2004). All samples were frozen at a temperature below −18 °C. Sample were freeze dried and milled and % wetness was recorded for each sample, before being analyzed on a Milestone Hg analyzer utilizing thermal decomposition, catalytic reduction, amalgamation, desorption, and atomic absorption spectroscopy following US EPA method 7473 (US EPA 2007a). The instrument was primed and the calibration was verified with standard reference material at the beginning of each analysis. Quality control samples (QCS) samples were included at the beginning and end of each run, with Continuing Calibration Verification (CCV) and Continuing Calibration Blank (CCB) samples analyzed every ten samples. Every twenty samples analyzed included a duplicate, matrix spike and matrix spike duplicate with a triplicate every 40 samples (Table S1). The recorded % wetness was used to convert the dry THg concentrations to wet-weight THg concentrations.
Water sampling and processing
In addition to fish, surface water samples were collected from 35 lakes from 2014 to 2016, using Teflon bottles and trace metal collection methods (US EPA method 1669, US EPA 1995) for analysis of total Hg (THg), MeHg, and in plastic bottles using standard collection methods for SO42−, NO3−, Cl−, monomeric Al, ANC, pH, DOC, UV254 absorbance and chlorophyll a. These samples were placed on ice in the field and shipped to Syracuse University for analysis. Mercury samples were filtered using 0.45 µm Millipore filters prior to analysis. Total Hg was analyzed by oxidation, reduction, purge and trap, desorption and cold-vapor atomic fluorescence spectrometry (CVAFS) according to US EPA method 1631, revision E (US EPA 2002). The detection limit for this method was 0.2 ng/L and quality control results and standards for THg analysis can be found in Table S2. MeHg was analyzed by direct ethylation, purge and trap, desorption, and CVAFS following a modified US EPA method 1630 (US EPA 2007b). The detection limit for this method was 0.02 ng/L and quality control results and standards can be found in Table S3.
Acid neutralizing capacity (ANC) and pH were analyzed in the laboratory using a Brinkmann Metrohm 716 DMS Titrino and 760 sample changer with a Ross General Purpose Sure-Flow pH electrode. Ion chromatography was used for analysis of SO42−, NO3−, and Cl−. Water samples were prepared for DOC analysis by filtering through a 1.5 µm glass microfiber filter and analyzed using infrared detection following persulfate oxidation. UV254 was analyzed by measuring absorbance at 254 nm in a standard spectrophotometer. Colorimetric methods were used to analyze for monomeric aluminum (Alm) following chelation with pyrocatechol violet. This same method was used to analyze for organic monomeric aluminum (Alo) after the sample was passed through an ion exchange column. Inorganic monomeric aluminum (Ali) was then calculated by subtracting Alo from Alm (McAvoy et al. 1992). Chlorophyll a samples were sent to the Upstate Freshwater Institute for analysis by concentrating chlorophyll containing particulate matter on 0.45 µm cellulose nitrate filters. The pigment was then extracted from the filter using acetone and measured using a standard spectrophotometer.
Standardizing fish THg concentrations
Note that there is no statistically significant difference in Hg concentrations between standard fillet and plugs (Knight et al. 2019) from fish tissue, but DEC fillet usually has concentrations 15% lower than those in plugs. We measured paired fillet and plugs for 513 samples across the 7 fish species we studied. A conversion formula (R2 = 0.9923, p < 0.001) was developed for converting DEC fillet Hg results to plug Hg results:
The statistical software R (R Core Team 2018) was used to standardize THg concentrations at previously determined lengths (Yellow perch—229 mm, Smallmouth bass—356 mm, Largemouth bass—356 mm, Walleye—457 mm; Simonin et al. 2008a). Length standardization was achieved by fitting a linear model of THg concentration and length for each lake per year. Model residuals were then used to estimate the THg concentration of each sample at the standardized length for the species. We used these fitted values even in cases where the regression relationship was not significant because this standardized value was the best estimate of fish Hg concentrations comparable across all the lakes. The length adjusted data were used to evaluate temporal trends, correlations with chemical and physical parameters, and conduct spatial analysis.
Statistical analysis on water chemistry and fish Hg concentrations in 2010s
Surface water chemistry data were evaluated for 18 lakes in the Northeast, 15 lakes in the Southeast and 2 lakes in the West region of New York State. A simple t test was applied to compare water chemistry data among regions (i.e., Northeast vs. the Southeast) using lakes as replicates. Data for this and the analyses described below were log-transformed to meet the assumption of residual normality.
Because multiple fish samples were collected within each lake, we took the average of the values to represent the mean concentration for individual lakes for below analyses. To study differences in Hg concentrations among the seven fish species in this study, a one-way ANOVA was applied to the standardized fish THg concentrations with lakes as replicates. ArcMap (ESRI 2017) was used to examine the spatial variation of the four most commonly sampled species across New York State.
A Pearson correlation test was used to examine the relationship between water chemical variables and fish Hg concentrations of the four common fish species. Linear regressions were used to investigate the effects of lake characteristics on water chemistry and fish Hg concentrations. Lake characteristics were acquired from Simonin et al. (2008a), including elevation (m), watershed area (ha), lake area (ha), shoreline (km), contiguous wetlands (ha), watershed wetland area (ha) and watershed wetland area (%). We also tested whether an existing outlet dam would impact water chemical variables and fish Hg of the four common species using one-way ANOVA with lakes as replicates.
Changes in water chemistry and fish Hg concentrations between 2000s and 2010s
The majority of the lakes in this study only have data from two sampling events. Paired t tests were used to examine changes in water chemical variables and fish THg concentrations of each of the four common species state-wide and by region using lakes as replicates.
To examine if lake characteristics and changes in water chemistry (as a %) from 2000s to 2010s would explain the changes in fish THg concentrations in each of the four common species state-wide, linear regressions were used with lakes as replicates. Stepwise regression was also used to determine if any models would predict changes in fish THg concentrations for each species. ArcMap v10.6 was used to examine the percent change of THg concentrations in each species between the two collection periods. Kriging was again used for all species, and Inverse Distance Weighting (IDW) was used when kriging showed no variation across New York State (Smallmouth bass, Largemouth bass).
To determine the number of lakes required to detect a change in fish THg concentrations after a decade, we calculated the sample size needed to detect a difference of 2–10% of the mean THg concentrations for each commonly species using paired t test, assuming the mean values calculated from replicates within lakes were representative:
where N is the sample size (number of lakes per sampling period), s2 is the pooled variance, d is the detectable difference on an original scale, Z is the critical value from the standardized normal distribution, α = 0.05, and β = 0.2 (i.e., power is 0.8). Pooled variance can be calculated as:
To investigate if a change in sampling intensity by region would impact the ability to detect trends, we calculated and compared the number of lakes required for each common species among the three regions to detect a 10% change in fish THg concentrations over a decade. Because our dataset only allowed us to detect a temporal change between two points of time, it was not possible to examine the impact of sampling frequency in time. Smaller sample size would be needed if the calculation was based on a log-scale. These statistical analyses were conducted with SAS 9.4 (SAS Institute Inc. 2013).
Results
General lake chemistry and standard-size fish Hg concentrations in 2010s
The 18 Northeast, 15 Southeast and 2 West lakes in NYS had water column THg (mean ± SD) of 0.86 ± 0.70 ng L−1, MeHg of 0.06 ± 0.08 ng L−1 and %MeHg ((MeHg/THg) × 100) of 7.4 ± 5.6. Additional statistics for individual lakes can be found in Table S2. The Northeast lakes had a relatively lower ANC, pH and chlorophyll a, and a higher DOC, MeHg and THg than the Southeast lakes (p ≤ 0.05). The two lakes in West region had similar water chemistry compared with Southeast lakes (Table S2).
The standard-size wet weight Hg concentration (mean ± SD) in 2010s was 0.43 ± 0.59 µg g−1 for Yellow perch (57 lakes), 0.22 ± 0.13 µg g−1 for Brook trout (29 lakes), 0.58 ± 0.44 µg g−1 for Smallmouth bass (22 lakes), 0.43 ± 0.21 µg g−1 for Largemouth bass (21 lakes), 0.67 ± 0.39 µg g−1 for Walleye (14 lakes), 0.81 ± 0.31 µg g−1 for Chain pickerel (9 lakes) and 0.60 ± 0.61 µg g−1 for Lake trout (8 lakes) in NYS (Fig. 2). Chain Pickerel had higher Hg concentrations than Walleye, Lake trout, Smallmouth bass, Largemouth bass and Yellow perch; Brook trout had the lowest Hg concentration among all species (p < 0.001).
For the 43 resurveyed lakes, concentrations of Hg in Yellow perch muscle samples exceeded the US EPA Fish Tissue Residue Criterion for MeHg of 0.30 μg g−1 in 71% of Northeast lakes, 8% of Southeast lakes and 0% of West lakes. Most lakes had Hg concentrations exceeding 0.30 μg g−1 for Walleye (10 out of 11 lakes), Smallmouth bass (9 out of 12 lakes) and Largemouth bass (6 out of 10 lakes). Three Northeast lakes (North Lake for Yellow perch, Red Lake for Walleye and Hinckley Reservoir for Smallmouth bass), one Southeast lake (Swinging Bridge Reservoir for Walleye) and one West lake (Rushford Lake for Walleye) had fish Hg concentrations that exceeded the US Food and Drug Administration action level of 1.0 μg g−1 (Fig. 3; Figs. S1–S3).
Yellow perch concentrations were highest in the Northeast region on NYS. Fish Hg increased between 2000s and 2010s surveys in both the Northeast and West regions with modest reductions in the Southeast. Resurveyed lakes have a percent change and were sampled at least twice; during the Simonin et al. survey and our recent survey
Relationships between lake characteristics, lake chemistry and standard-size fish Hg concentrations in resurveyed lakes
Lake MeHg and THg concentrations were positively correlated with DOC, total monomeric Al and organic monomeric Al, and negatively correlated with pH and SO42− (Table S3). Lakes with higher organic monomeric Al or higher MeHg had higher %MeHg (p < 0.01 for both).
Lake pH was negatively correlated with total monomeric Al (r2 = −0.63, p < 0.01), DOC (r2 = −0.35, p = 0.05), and positively correlated with ANC (r2 = 0.39, p = 0.02) using a Pearson correlation test for all 35 lakes (Table S3).
Total Hg concentrations in lake water increased with increases in lake elevation (r2 = 0.41, p = 0.03) (Fig. 4). Two lakes (Rock Pond - 2.37 ng-Hg L−1; Sunday Lake 3.64 ng-Hg L−1) had relatively higher THg concentrations than the other lakes studied (0.22 – 1.55 ng L−1). With data from those two lakes removed, the relationship of higher aqueous THg concentration at higher elevation remained significant (r2 = 0.47, p = 0.01). %MeHg was positively correlated with shoreline length (r2 = 0.44, p = 0.03) and % contiguous wetland relative to lake area (r2 = 0.49, p = 0.04). Other lake characteristics such as watershed area, lake area and contiguous wetland area did not appear to influence the lake chemistry (p ≥ 0.12).
Mercury concentrations in standard-size Yellow perch (r2 = 0.64, p < 0.001) increased with lake THg concentration, but this pattern was not evident for Walleye, Largemouth bass and Smallmouth bass (p ≥ 0.29). Lake MeHg concentration and % MeHg was not correlated with standard-size Hg concentrations for any of the four fish species (p ≥ 0.20). Total monomeric Al, ANC, pH, and SO42− were correlated with standard-size Hg concentrations for Yellow perch and Smallmouth bass, but not for Walleye and Largemouth bass (Table S3).
As with water column THg, Hg concentrations in Yellow perch increased with lake elevation (r2 = 0.60, p < 0.001), but not for Walleye, Smallmouth bass and Largemouth bass (p ≥ 0.59). Lakes with a larger percentage of contiguous wetland relative to lake area had higher standard-size Hg concentrations for only Walleye (r2 = 0.88, p = 0.02), but not for Yellow perch, Smallmouth bass and Largemouth bass (p ≥ 0.27). Other lake characteristics such as watershed area, lake area, shoreline length and the presence of an outlet dam did not influence fish Hg concentrations (p ≥ 0.14) in the studied lakes.
Changes in resurveyed lake chemistry between 2000s and 2010s
For all resurvey lakes, concentrations of THg decreased 42% (p < 0.01), MeHg decreased 74% (p < 0.01), SO42− decreased 77% (p < 0.01), NO3− decreased 39% (p = 0.02), and total monomeric Al decreased 25% (p = 0.01) from 2000s to 2010s. Lake ANC increased 6% (p = 0.02), and DOC increased 22% (p = 0.04). The pH (p = 0.13), chlorophyll a (p = 0.9) and Cl (p = 0.18) were similar between the two sampling periods. There was no statistically significant change in the %MeHg values between the two sampling periods.
In both Northeast and South regions, lake THg (p < 0.001 for both), MeHg (p = 0.004 and 0.01) and SO42− (p < 0.001 for both) decreased from 2000s to 2010s. While, lake pH (p = 0.02) and NO3− (p = 0.05) decreased. DOC (p = 0.002) increased only in Northeast lakes. Total monomeric Al (p = 0.003) decreased in only Southeast lakes.
Changes in standard-size fish Hg concentrations between 2000s and 2010s
Concentrations of Hg in Smallmouth bass decreased 21% from 2000s to 2010s (p = 0.07). Standard-size Hg concentrations were similar between 2000s and 2010s for Yellow perch, Walleye and Largemouth bass (p ≥ 0.49) using all paired lakes. Temporal changes in fish Hg concentrations varied by lake and species (Table S4). In lakes sampled for Yellow perch (n = 36) and Walleye (n = 12), fish Hg concentrations increased in about as many study lakes (53% and 58%, respectively) as lakes in which values decreased (47% and 42%, respectively) from 2000s to 2010s. In contrast, lakes sampled for Smallmouth bass (n = 13) and Largemouth bass (n = 10) decreased in a much larger fraction of the lakes surveyed (77% and 70%, respectively).
For lakes with ≥2 fish species sampled, seven out of 18 lakes showed the same temporal patterns of change in Hg concentrations collected fish species. Two lakes (Hinckley Reservoir in the Northeast and Goodyear Lake in Southeast) had increases in fish Hg concentrations and five (Red Lake in the Northeast; East Sidney Reservoir, Fort Pond, Onteora Lake and Rio Reservoir in the Southeast) showed decreases in fish Hg concentrations.
Dividing the lakes into the three regions (Northeast, Southeast, West), Hg concentrations in Yellow perch from lakes in the West increased 29% from 2000s to 2010s (average of five lakes, p = 0.04). Concentrations of Hg in Smallmouth bass from Southeast lakes decreased 41% from 2000s to 2010s (average of six lakes, p = 0.02). Concentrations of Hg in other species from other regions did not change significantly (p ≥ 0.15).
Changes in standard-size fish Hg concentrations between 2000s and 2010s explained by lake characteristics and water chemistry
Changes in Hg concentration in Walleye over time were positively correlated with lake elevation (r2 = 0.67, p = 0.04). Changes in Hg concentration in Largemouth bass were positively correlated with contiguous wetland area (r2 = 0.86, p = 0.01). No empirical models could predict the changes in fish Hg concentrations from the 2000s to the 2010s using a stepwise regression of lake characteristics and changes in water chemistry.
The changes in fish Hg concentrations from 2000s to 2010s might be explained by the changes in lake chemistry over the same sampling period. In a Spearman rank test between percent changes in fish Hg concentrations for each species and the differences in chemical variables (ratio strength for difference in pH and percent difference for other variables), we found that changes in lake ANC (r2 = 0.87, p < 0.001), and pH (r2 = 0.74, p = 0.02) were positively correlated with changes in Hg concentrations in Walleye. Lake chlorophyll a was negatively correlated with changes in Hg concentrations in Largemouth bass (r2 = −0.58, p = 0.04). Changes in water THg concentration (r2 = −0.93, p < 0.001) was negatively correlated with changes in Hg concentrations in Largemouth bass.
Discussion
Variation in Hg in NYS lakes
Freshwater resources in NYS are sensitive to atmospheric Hg deposition (Driscoll et al. 2007b). All regions of the state show lakes with elevated concentrations of Hg in fish, with the highest concentrations generally found in samples collected from the Adirondack or Catskill park regions (Simonin et al. 2008b). Observations of the most widely sampled fish species, Yellow perch indicate that the lakes within the Adirondack Park contain fish with the highest concentrations of Hg (Fig. 3). Indeed, of the 106 lakes with specific fish consumption advisories for Hg in New York State, 62 occur in the Adirondack region. Moreover, there are regional advisories on fish consumption for the Adirondacks and Catskills (NYS DOH 2018). Even in regions of the state where fish Hg concentrations are generally lower, many of the popular gamefish remain above the 0.3 µg g−1 guideline (Fig. 2).
With still more than half (53%; Fig. 2) of standard-length fish Hg above the 0.3 µg g−1 criterion, additional efforts are needed to reduce fish Hg concentrations. Our analysis shows that despite decreases in emissions and atmospheric Hg deposition, there has not been a systematic decrease in fish Hg in NYS over the past decade. Rather, Walleye and Yellow perch Hg in many lakes have remained the same or increased relative to measurements from the survey of Simonin et al. 2008a, b. This pattern is consistent with findings from the Laurentian Great Lakes where decreases in atmospheric Hg were significantly correlated with decreases in fish Hg for all the lakes until 2010, when the patterns for Michigan, Erie and Ontario changed from decreasing to no change or increasing concentrations of fish Hg (Zhou et al. 2017). Like lakes Michigan, Erie and Ontario, the major driver of fish Hg trends in NYS inland waters may be shifting away from regional Hg emissions towards the effects of legacy Hg inputs or increasing global Hg emissions, changes in nutrient status, invasive species, climate change and/or global Hg emissions.
We observed marked decreases in water column THg and MeHg concentrations between the two surveys. Decreases in atmospheric Hg deposition (Gerson et al. 2017; Mao et al. 2017) could explain this pattern of decreases in water column concentrations. Concentrations of THg and SO42− alone were unable to explain the spatial variation in MeHg and %MeHg. While THg and SO42− are likely important drivers of MeHg, we found that only length of shoreline and wetland area were significantly correlated with MeHg concentrations in study lakes. This pattern suggests that landscape features where reducing conditions occur control the extent and rate of methylation and MeHg concentrations, similar to findings from other studies (Mitchell and Gilmour 2008; Skyllberg 2008; Burns et al. 2012).
Many of the spatial correlations of biophysical and chemical factors with fish Hg found by Simonin et al. 2008a, b were not evident in this resurvey. In our resurvey, only concentrations of THg in lake water were significantly correlated with Hg in Yellow perch, but not other fish species. The response of fish to decreases in atmospheric Hg deposition may be influenced by recovery from the impacts of acid deposition, particularly in the Adirondacks, (Driscoll et al. 2001; Jeffries et al. 2003). Increases in fish Hg and aquatic concentrations of DOC have been associated with recovery from acidification in northeastern North America and northern Europe (Monteith et al. 2007; Hongve et al. 2012; Åkerblom et al. 2012; Driscoll et al. 2013; Millard et al. 2018). This pattern is consistent with our findings where regions more highly impacted by acid deposition (Northeast region) have exhibited an increase in fish Hg which is coincident with increases in DOC.
Not only could chemical recovery from acid deposition alter fish Hg, but food webs could also be changing as acid-sensitive species recolonize impacted aquatic ecosystems. A longer food chain could result in a shift to higher trophic positions for sportfish and a subsequent increase in bioaccumulation (Ward et al. 2010). Conversely, increased nutrient inputs could result in biodilution or growth dilution of fish Hg through increases in aquatic productivity or accelerated individual growth (Riva-Murray et al. 2011; Kolka et al. 2019). The negative relation we observed between Hg in Largemouth bass and chlorophyll a may be evidence of this effect. Additional alterations to aquatic food webs such as stocking, or the introduction and/or management of invasive species (Taylor et al. 2020) can also have large impacts on food web dynamics and the biological cycling of Hg and may help explain why similar trends in Hg concentrations were observed for different fish species within the same lake for only 7 of 18 lakes where two or more species were collected. Community composition and structure play an important role for individual lake responses (Todorova et al. 2015) and further complicate statewide and regional trends.
The impacts of climate change contribute an additional level of uncertainty. Past and projected future climate for the Northeast have and will result in increases in air temperature and precipitation quantity (Climate Science Special Report 2017). A warmer and wetter climate in NYS could enhance primary production, lengthen the growing season, increase drought frequency, intensify precipitation and alter foodwebs (Hayhoe et al. 2007), which could impact deposition, transport, methylation and bioaccumulation of Hg. Within this survey, Yellow perch in the West region showed almost a 30% increase from previous observations, while no change was detected for Walleye and Largemouth bass (potentially due to lower power to detect changes). These changes in the West region may be a result of climate driven impacts evidenced by an increased incidence of algal blooms in this region (Halfman 2017). This could result in a shift in autochthonous vs. allochthonous organic matter input which recent studies in boreal (ca. 59–60 °N; Bravo et al. 2017) as well as moist subtropical (ca. 26–28 °N; Jiang et al. 2018) lakes have shown a positive correlation between autochtonous organic matter and Hg methylation in sediment. Conversely, this could also result in lower fish Hg as a result of growth dilution (Essington and Houser 2003; Kolka et al. 2019) or biodilution, as has been reported in phytoplankton and zooplankton (Pickhardt et al. 2002; Chen and Folt 2005).
Beyond these ecosystem complexities, an important consideration in the lack of fish Hg response to decreases in domestic Hg emission and local deposition may simply be a lag of Hg transport in the lake-watersheds. Although we would anticipate that the marked decreases in water column Hg species observed would eventually contribute to declines in fish Hg, a decade may be too short a period for adult fish to respond to decreases in atmospheric Hg deposition (Harris et al. 2007).
Number of lakes required to detect changes in standard-size fish Hg concentrations after a decade
The number of lakes required to detect a change between two sampling points would decrease if the rate of change in fish Hg concentration per decade was greater. Comparison among species of the number of lakes required to detect a given change in fish Hg showed that Largemouth bass and Walleye would require the greatest number of lakes monitored followed by Smallmouth bass and Yellow perch (Fig. 5). For example, it would require 18 lakes for Largemouth bass, 12 lakes for Walleye, 10 lakes for Smallmouth bass and only 5 lakes for Yellow perch to detect a 4% change after a decade.
In order to detect changes in fish Hg, lakes in the West (n = 5–46, varied by species) will require greater sampling efforts in terms of number of lakes monitored to detect a change of 10% in fish Hg per decade than lakes in Northeast (n = 2–15) and Southeast (n = 4–7) (Fig. 6). The lakes in Southeast will required a similar sampling efforts to detect a 10% change in fish Hg per decade in terms in number of lakes for each of the four fish species considered. In contrast the number of lakes required to detect a 10% change in fish Hg varied greatly for different fish species in Northeast and West regions, with Yellow perch needing little additional sampling effort (n = 2,5) while targeting Walleye would require significant increases (n = 15,46) in sampling effort in both regions. Largemouth and Smallmouth bass would also need significantly increased sampling efforts in the Northeast and West respectively.
Implications for continued monitoring of Fish Hg in NYS
Given the major pathway of human exposure to MeHg is by consumption of contaminated fish (Driscoll et al. 2013) and elevated and increasing concentrations of Hg in NYS freshwater fish, monitoring of Hg in NYS fish and waters should continue. With over 4000 lakes and many kilometers of rivers in the state (Simonin et al. 2008b) it is not possible to monitor all surface waters. However, a well-designed program could be used to monitor the regions defined by Simonin et al. 2008a, b or potentially use the ecological zones (ecozones) of New York State. For the current three regions, our power analysis shows that continuing previous sampling efforts of Yellow perch could detect a 10% change over a decade without increasing the monitoring effort.
Our analysis indicates that under the current three-region design, a lake monitoring program targeting 30 lakes (15 in the Adirondack State Park and 15 in the remainder of the state) would have a reasonable power to detect change in fish Hg concentrations (Fig. 5). Sites outside the Adirondacks should likely include (1) lakes in the Catskills; (2) lakes of high economic, recreational and/or cultural importance such as the Finger Lakes, Oneida Lake, and Lake Chautauqua; and (3) reservoirs (Rushford). Lakes should be selected based on the length of existing data records and those with recent fish Hg concentrations that exceed consumption guidelines.
In order to maintain a high statistical power, the majority of these lakes should be sampled at a 5-year time interval, with a few lakes sampled more intensely in order to assess interannual variability. These more frequently sampled lakes may also be good candidates for more intensive analysis of nutrients and Hg concentrations and lower trophic position species such as invertivorous fish species (e.g., dace species) or young of the year Yellow perch. Collecting smaller invertivorous species and/or young of the year would provide an opportunity to evaluate at interannual variability and should exhibit a more rapid response to future controls on anthropogenic Hg emissions.
An alternative to this three-region model, would be sampling within the 12 ecozones within NYS (Fig S4; Will et al. 1982, Dickinson 1983). While this approach would likely require a significant increase in sampling effort, it would provide a much better understanding of fish Hg response to policy decisions. If a similar program for stream and river species were to be adopted, it might be possible to cover the majority of ecozones within NYS without much additional effort.
References
Åkerblom S, Nilsson M, Yu J et al. (2012) Temporal change estimation of mercury concentrations in northern pike (Esox lucius L.) in Swedish lakes. Chemosphere 86:439–445. https://doi.org/10.1016/j.chemosphere.2011.09.037
Baker RF, Blanchfield PJ, Paterson MJ et al. (2004) Evaluation of nonlethal methods for the analysis of mercury in fish tissue. Trans Am Fish Soc 133:568–576. https://doi.org/10.1577/T03-012.1
Benoit JMM, Gilmour CCC, Heyes A, et al (2002) Geochemical and biological controls over methylmercury production and degradation in aquatic ecosystems. In: ACS symposium. p 262–297
Bodaly RA, Rudd JWM, Fudge RJP, Kelly CA (1993) Mercury concentrations in fish related to size of remote Canadian shield lakes. Can J Fish Aquat Sci 50:980–987. https://doi.org/10.1139/f93-113
Bravo AG, Bouchet S, Tolu J et al. (2017) Molecular composition of organic matter controls methylmercury formation in boreal lakes. Nat Commun 8:14255. https://doi.org/10.1038/ncomms14255
Burns DA, Riva-Murray K, Bradley PM et al. (2012) Landscape controls on total and methyl Hg in the upper Hudson River basin, New York, USA. J Geophys Res Biogeosciences 117:n/a–n/a. https://doi.org/10.1029/2011JG001812
Chen CY, Folt CL (2005) High plankton densities reduce mercury biomagnification. Environ Sci Technol 39:115–121. https://doi.org/10.1021/es0403007
Chen L, Driscoll CT (2005) Regional assessment of the response of the acid−base status of lake watersheds in the adirondack region of New York to changes in atmospheric deposition using PnET-BGC. Environ Sci Technol 39:787–794. https://doi.org/10.1021/es049583t
Chiasson-Gould SA, Blais JM, Poulain AJ (2014) Dissolved organic matter kinetically controls mercury bioavailability to bacteria. Environ Sci Technol 48:3153–3161. https://doi.org/10.1021/es4038484
Climate Science Special Report (2017) Climate science special report: fourth national climate assessment, Vol I. Climate Science Special Report, Washington, DC
Coleman Wasik JK, Engstrom DR, Mitchell CPJ et al. (2015) The effects of hydrologic fluctuation and sulfate regeneration on mercury cycling in an experimental peatland. J Geophys Res Biogeosciences 120:1697–1715. https://doi.org/10.1002/2015JG002993
Dickinson NR (1983) A division of southern and western New York State into ecological zones. NYSDEC. Albany, NY
Dittman JA, Shanley JB, Driscoll CT et al. (2010) Mercury dynamics in relation to dissolved organic carbon concentration and quality during high flow events in three northeastern US streams. Water Resour Res 46:W07522. https://doi.org/10.1029/2009WR008351
Driscoll CT, Driscoll KM, Fakhraei H, Civerolo K (2016) Long-term temporal trends and spatial patterns in the acid-base chemistry of lakes in the Adirondack region of New York in response to decreases in acidic deposition. Atmos Environ 146:5–14. https://doi.org/10.1016/j.atmosenv.2016.08.034
Driscoll CT, Driscoll KM, Mitchell MJ, Raynal DJ (2003) Effects of acidic deposition on forest and aquatic ecosystems in New York State. Environ Pollut 123:327–336. https://doi.org/10.1016/S0269-7491(03)00019-8
Driscoll CT, Driscoll KM, Roy KM, Dukett J (2007a) Changes in the chemistry of lakes in the Adirondack region of New York following declines in acidic deposition. Appl Geochem 22:1181–1188. https://doi.org/10.1016/j.apgeochem.2007.03.009
Driscoll CT, Han Y, Chen CY, Evers DC et al. (2007b) Mercury contamination in forest and freshwater ecosystems in the northeastern United States. Bioscience 57:17. https://doi.org/10.1641/B570106
Driscoll CT, Lawrence GB, Bulger AJ et al. (2001) Acidic deposition in the northeastern United States: sources and inputs, ecosystem effects, and management strategies. Bioscience 51:180
Driscoll CT, Mason RP, Chan HM et al. (2013) Mercury as a global pollutant: sources, pathways, and effects. Environ Sci Technol 47:4967–4983. https://doi.org/10.1021/es305071v
ESRI (2017) ArcGIS Desktop. Redlands, CA: Environmental Systems Research Institute
Essington TE, Houser JN (2003) The effect of whole-lake nutrient enrichment on mercury concentration in age-1 yellow perch. Trans Am Fish Soc 132:57–68. 10.1577/1548-8659(2003)132<0057:TEOWLN>2.0.CO;2
Evans CD, Chapman PJ, Clark JM et al. (2006) Alternative explanations for rising dissolved organic carbon export from organic soils. Glob Chang Biol 12:2044–2053. https://doi.org/10.1111/j.1365-2486.2006.01241.x
Evers DC, Savoy LJ, Desorbo CR et al. (2008) Adverse effects from environmental mercury loads on breeding common loons. Ecotoxicology 17:69–81. https://doi.org/10.1007/s10646-007-0168-7
Fitzgerald WF, Engstrom DR, Mason RP, Nater EA (1998) The case for atmospheric mercury contamination in remote areas. Environ Sci Technol 32:1–7. https://doi.org/10.1021/es970284w
Gerson JR, Driscoll CT, Demers JD et al. (2017) Deposition of mercury in forests across a montane elevation gradient: Elevational and seasonal patterns in methylmercury inputs and production. J Geophys Res Biogeosciences 122:1922–1939. https://doi.org/10.1002/2016JG003721
Gilmour CC, Riedel GS, Ederington MC et al. (1998) Methylmercury concentrations and production rates across a trophic gradient in the northern Everglades. Biogeochemistry 40:327–345. https://doi.org/10.1023/A:1005972708616
Gorski PR, Armstrong DE, Hurley JP, Krabbenhoft DP (2008) Influence of natural dissolved organic carbon on the bioavailability of mercury to a freshwater alga. Environ Pollut 154:116–123. https://doi.org/10.1016/j.envpol.2007.12.004
Graham AM, Aiken GR, Gilmour CC (2012) Dissolved organic matter enhances microbial mercury methylation under sulfidic conditions. Environ Sci Technol 46:2715–2723. https://doi.org/10.1021/es203658f
Halfman JD (2017) Decade-scale water quality variability in the eastern finger lakes New York. NYWEA. Clearwaters 47:20–33
Harris RC, Rudd JWM, Amyot M et al. (2007) Whole-ecosystem study shows rapid fish-mercury response to changes in mercury deposition. Proc Natl Acad Sci USA 104:16586–16591. https://doi.org/10.1073/pnas.0704186104
Hayhoe K, Wake CP, Huntington TG et al. (2007) Past and future changes in climate and hydrological indicators in the US Northeast. Clim Dyn 28:381–407. https://doi.org/10.1007/s00382-006-0187-8
Herrero Ortega S, Catalán N, Björn E et al. (2018) High methylmercury formation in ponds fueled by fresh humic and algal derived organic matter. Limnol Oceanogr 63:S44–S53. https://doi.org/10.1002/lno.10722
Hodgkins GA, Dudley RW, Huntington TG (2003) Changes in the timing of high river flows in New England over the 20th Century. J Hydrol 278:244–252. https://doi.org/10.1016/S0022-1694(03)00155-0
Hongve D, Haaland S, Riise G et al. (2012) Decline of acid rain enhances mercury concentrations in fish. Environ Sci Technol 46:2490–2491. https://doi.org/10.1021/es3002629
Huntington TG, Hodgkins GA, Keim BD, Dudley RW (2004) Changes in the proportion of precipitation occurring as snow in New England (1949–2000). J Clim 17:2626–2636. 10.1175/1520-0442(2004)017<2626:CITPOP>2.0.CO;2
Jeffries DS, Brydges TG, Dillon PJ, Keller W (2003) Monitoring thE Results of Canada/USA acid rain control programs: some lake responses. Environ Monit Assess 88:3–19. https://doi.org/10.1023/A:1025563400336
Jeremiason JD, Reiser TK, Weitz RA et al. (2016) Aeshnid dragonfly larvae as bioindicators of methylmercury contamination in aquatic systems impacted by elevated sulfate loading. Ecotoxicology 25:456–468. https://doi.org/10.1007/s10646-015-1603-9
Jiang T, Bravo AG, Skyllberg U et al. (2018) Influence of dissolved organic matter (DOM) characteristics on dissolved mercury (Hg) species composition in sediment porewater of lakes from southwest China. Water Res 146:146–158. https://doi.org/10.1016/j.watres.2018.08.054
Kerin EJ, Gilmour CC, Roden E et al. (2006) Mercury methylation by dissimilatory iron-reducing bacteria. Appl Environ Microbiol 72:7919–7921. https://doi.org/10.1128/AEM.01602-06
Knight A, Bhavsar SP, Branfireun BA et al. (2019) A comparison of fish tissue mercury concentrations from homogenized fillet and nonlethal biopsy plugs. J Environ Sci 80:137–145. https://doi.org/10.1016/j.jes.2018.12.004
Kolka RK, Riggs CE, Nater EA et al. (2019) Temporal fluctuations in young-of-the-year yellow perch mercury bioaccumulation in lakes of northeastern Minnesota. Sci Total Environ 656:475–481. https://doi.org/10.1016/j.scitotenv.2018.11.280
Laudon H, Buttle J, Carey SK et al. (2012) Cross-regional prediction of long-term trajectory of stream water DOC response to climate change. Geophys Res Lett 39:4–9. https://doi.org/10.1029/2012GL053033
Mao H, Ye Z, Driscoll C (2017) Meteorological effects on Hg wet deposition in a forested site in the Adirondack region of New York during 2000–2015. Atmos Environ 168:90–100. https://doi.org/10.1016/j.atmosenv.2017.08.058
McAvoy DC, Santore RC, Shosa JD, Driscoll CT (1992) Comparison between pyrocatechol violet and 8-hydroxyquinonline procedures for determining aluminum fractions. Soil Sci. Soc. Am. J. 56:449–455
McCabe GJ, Wolock DM (2002) A step increase in streamflow in the conterminous United States. Geophys Res Lett 29:38–1-38–4. https://doi.org/10.1029/2002GL015999
Millard GD, Driscoll CT, Burns DA et al. (2018) Response of mercury in an Adirondack (NY, USA) forest stream to watershed lime application. Environ Sci Process Impacts 20:607–620. https://doi.org/10.1039/c7em00520b
Minimata Convention on Mercury (2017) Minimata convention. Minimata Convention on Mercury, http://www.mercuryconvention.org/Convention/tabid/3426/language/en-US/Default.aspx
Mitchell CPJ, Gilmour CC (2008) Methylmercury production in a Chesapeake Bay salt marsh. J Geophys Res 113:1–14. https://doi.org/10.1029/2008JG000765
Monson BA, Staples DF, Bhavsar SP et al. (2011) Spatiotemporal trends of mercury in walleye and largemouth bass from the laurentian great lakes region. Ecotoxicology 20:1555–1567. https://doi.org/10.1007/s10646-011-0715-0
Monteith DT, Stoddard JL, Evans CD et al. (2007) Dissolved organic carbon trends resulting from changes in atmospheric deposition chemistry. Nature 450:537–540. https://doi.org/10.1038/nature06316
NYS DOH (2018) Health advice on eating fish you catch. https://www.health.ny.gov/environmental/outdoors/fish/health_advisories/. Accessed 16 Feb 2019
Peterson SA, Van Sickle J, Hughes RM et al. (2004) A biopsy procedure for determining filet and predicting whole-fish mercury concentration. Arch Environ Contam Toxicol 48:99–107. https://doi.org/10.1007/s00244-004-0260-4
Pickhardt PC, Folt CL, Chen CY et al. (2002) Algal blooms reduce the uptake of toxic methylmercury in freshwater food webs. Proc Natl Acad Sci USA 99:4419–4423. https://doi.org/10.1073/pnas.072531099
Prestbo EM, Gay DA (2009) Wet deposition of mercury in the US and Canada, 1996-2005: Results and analysis of the NADP mercury deposition network (MDN). Atmos Environ 43:4223–4233. https://doi.org/10.1016/j.atmosenv.2009.05.028
R Core Team (2018) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://Rproject.org/
Riva-Murray K, Chasar LC, Bradley PM et al. (2011) Spatial patterns of mercury in macroinvertebrates and fishes from streams of two contrasting forested landscapes in the eastern United States. Ecotoxicology 20:1530–1542. https://doi.org/10.1007/s10646-011-0719-9
Sebestyen SD, Boyer EW, Shanley JB (2009) Responses of stream nitrate and DOC loadings to hydrological forcing and climate change in an upland forest of the northeastern United States. J Geophys Res. Biogeosciences 114:1–11. https://doi.org/10.1029/2008JG000778
Simonin H, Loukmas J, Skinner L, Roy K (2008a) Strategic monitoring of Mercury in New York state fish. http://www.dec.ny.gov/docs/wildlife_pdf/hgfish.pdf. Accessed 3 May 2016
Simonin HA, Loukmas JJ, Skinner LC, Roy KM (2008b) Lake variability: Key factors controlling mercury concentrations in New York State fish. Environ Pollut 154:107–115. https://doi.org/10.1016/j.envpol.2007.12.032
Skyllberg U (2008) Competition among thiols and inorganic sulfides and polysulfides for Hg and MeHg in wetland soils and sediments under suboxic conditions: Illumination of controversies and implications for MeHg net production. J Geophys Res Biogeosciences 113:n/a–n/a. https://doi.org/10.1029/2008JG000745
Streets DG, Horowitz HM, Lu Z et al. (2019) Global and regional trends in mercury emissions and concentrations, 2010–2015. Atmos Environ 201:417–427. https://doi.org/10.1016/j.atmosenv.2018.12.031
Taylor MS, Driscoll CT, Lepak JM, et al (2020) Temporal trends in fish mercury concentrations in an Adirondack Lake managed with a persistent predator removal program. Ecotoxicology
Todorova S, Driscoll CT, Matthews DA, Steven WE (2015) Zooplankton community changes confound the biodilution theory of methylmercury accumulation in a recovering mercury- contaminated lake. 8–13. https://doi.org/10.1021/es5044084
US EPA (2017) Mercury and air toxics standards: clean power plants. In: Mercur. Air Toxics Stand. https://www.epa.gov/mats/cleaner-power-plants#controls. Accessed 4 Feb 2019
US EPA (2007a) Method 7473: mercury in solids and solutions by thermal decomposition, amalgamation, and atomic absorption spectrophotometry. Washington, DC: United States Environmental Protection Agency
US EPA (1995) Method 1669: sampling ambient water for trace metals at EPA water quality criteria levels. Washington, DC: United States Environmental Protection Agency, p 1–42
US EPA (2002) Method 1631, revision E: mercury in water by oxidation, purge and trap, and cold vapor atomic fluorescence spectrometry. Washington, DC: United States Environmental Protection Agency, p 1–46
US EPA (2007b) Method 1630 methyl mercury in water by distillation, aqueous ethylation, purge and trap, and cold vapor atomic fluorescence spectrometry. Washington, DC: United States Environmental Protection Agency, p 1–55
Warby RAF, Johnson CE, Driscoll CT (2005) Chemical recovery of surface waters across the northeastern United States from reduced inputs of acidic deposition: 1984-2001. Environ Sci Technol 39:6548–6554. https://doi.org/10.1021/es048553n
Ward DM, Nislow KH, Folt CL (2010) Bioaccumulation syndrome: identifying factors that make some stream food webs prone to elevated mercury bioaccumulation. Ann N. Y Acad Sci 1195:62–83. https://doi.org/10.1111/j.1749-6632.2010.05456.x
Watson SB, Miller C, Arhonditsis G et al. (2016) The re-eutrophication of lake Erie: harmful algal blooms and hypoxia. Harmful Algae 56:44–66. https://doi.org/10.1016/j.hal.2016.04.010
Weiss-Penzias PS, Gay DA, Brigham ME et al. (2016) Trends in mercury wet deposition and mercury air concentrations across the US and Canada. Sci Total Environ 568:546–556. https://doi.org/10.1016/j.scitotenv.2016.01.061
Will G, Stumvoll R, Gotie R, Smith E (1982) The ecological zones of northern New York. N. Y Fish Game J 29:1–25
Ye Z, Mao H, Driscoll CT (2019) Primary effects of changes in meteorology vs. anthropogenic emissions on mercury wet deposition: a modeling study. Atmos Environ 198:215–225. https://doi.org/10.1016/j.atmosenv.2018.10.052
Zhang Y, Jacob DJ, Horowitz HM et al. (2016) Observed decrease in atmospheric mercury explained by global decline in anthropogenic emissions. Proc Natl Acad Sci 113:526–531. https://doi.org/10.1073/pnas.1516312113
Zhou C, Cohen MD, Crimmins BA et al. (2017) Mercury temporal trends in top predator fish of the Laurentian Great Lakes from 2004 to 2015: are concentrations still decreasing? Environ Sci Technol 51:7386–7394. https://doi.org/10.1021/acs.est.7b00982
Zhou H, Zhou C, Hopke PK, Holsen TM (2018) Mercury wet deposition and speciated mercury air concentrations at rural and urban sites across New York state: temporal patterns, sources and scavenging coefficients. Sci Total Environ 637–638:943–953. https://doi.org/10.1016/j.scitotenv.2018.05.047
Acknowledgements
Support for this study was provided by the New York State Energy Research and Development Authority (NYSERDA), and the National Science Foundation through the National Science Foundation Research Training program through the Educational Model Program of Water Energy Research (EMPOWER). NYSERDA has not reviewed the information herein, and the opinions expressed in this report to not necessarily reflect those of NYSERDA or the State of New York. This research does not reflect the official positions and policies of the US EPA. Any use of trade, firm, or product names is for descriptive purposes only and does not imply endorsement by the US Government.
Funding
This material is based upon work supported by the National Science Foundation under Grant No. DGE-1449617 and by the New York State Energy Research and Development Authority (NYSERDA) under Award No. 034357. NYSERDA has not reviewed the information contained herein, and the opinions expressed in this report do not necessarily reflect those of NYSERDA or the State of New York.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Millard, G., Driscoll, C., Montesdeoca, M. et al. Patterns and trends of fish mercury in New York State. Ecotoxicology 29, 1709–1720 (2020). https://doi.org/10.1007/s10646-020-02163-x
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10646-020-02163-x