Abstract
Plants suffer with combined stress of sulfur (S) deficiency and hexavalent chromium [Cr(VI)] in soils. There are a few reports on the interactive effects of S-deficiency and Cr(VI) stress. Therefore, the interactions between S nutrition and Cr(VI) stress were investigated in hydroponically grown canola (Brassica napus L.) cultivars differing in Cr(VI) tolerance. The relatively Cr(VI)-tolerant (NK Petrol) and Cr(VI)-susceptible (Sary) cultivars were grown in S-sufficient nutrient solution and then exposed to variable S concentrations [deficient (0 mM S, −S) and sufficient (1 mM S, +S)]. The seedlings were then exposed to 100 μM Cr(VI) for 3 days. S-deficiency (−S/−Cr) and combined stress (−S/+Cr) caused a significant decrease in growth parameters of Sary than NK Petrol (P < 0.05). In −S/+Cr treatment, Cr accumulation in Sary was significantly higher than NK Petrol. The higher level of Cr in Sary increased lipid peroxidation and decreased chlorophyll content. The activities of antioxidant enzymes and cysteine content were significantly higher in NK Petrol than in Sary under combined stress. The levels of ascorbate (AsA) and glutathione (GSH) were significantly decreased by S deficiency. The expression level of metallothionein gene (BnMP1) in the tolerant NK Petrol was increased by −S/+Cr treatment. However, expression level of BnMP1 gene in the susceptible Sary was enhanced by +S/+Cr treatment. This result suggests metallothionein (MT) may be involved in Cr(VI) tolerance under S-deficient condition. In conclusion, S nutrition influenced Cr accumulation and enhanced tolerance caused by a positive effect on defense systems and gene expression.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Chromium (Cr) is the seventh most abundant element on earth, and found in both trivalent [Cr(III)] and hexavalent [Cr(VI)] forms (Shanker et al. 2005). Cr(VI) species are released into the environment from a wide range of industries such as electroplating, cement plants, steel production works, manufacture of dyes, paints and pigments, leather and wood preservation, metal finishing and metal plating (Zayed and Terry 2003). However, Cr levels in agricultural soil may be strongly enhanced as a consequence of anthropogenic activities, and it’s regarded as one of the most harmful pollutants that pose a risk to human health (Shanker et al. 2005; Zhang et al. 2014). Therefore, phytoremediation approaches are currently being used to remediate metal-contaminated soils (Chang et al. 2014; Ho et al. 2014). Cr phytotoxicity can result in the generation of reactive oxygen species (ROS) such as superoxide anion (O ·−2 ), hydroxyl radical (·OH) and hydrogen peroxide (H2O2) (Shanker et al. 2009). The capability of plants to cope with the oxidative damage depends on the efficiency of antioxidative defence system. Cr(VI) stress induced the enzymatic antioxidative system [superoxide dismutase (SOD), catalase (CAT) and guaiacol peroxidase (GPX)] and increased the levels of low molecular weight non-enzymatic antioxidants such as ascorbate (AsA) and glutathione (GSH) (Pandey et al. 2009). The AsA–GSH pathway is a key part of the network of reactions involving important enzymes [ascorbate peroxidase (APX) and glutathione reductase (GR)] and metabolites with redox properties for the efficient elimination of the removal of H2O2 in different cellular compartments (Noctor and Foyer 1998). Besides it’s one of the major forms of organic sulfur translocated in the phloem (Mendoza-Cózatl et al. 2008), GSH is considered as the most important intracellular defense against ROS induced oxidative damage in plants (Gill and Tuteja 2010; Anjum et al. 2012). Glutathione S-transferases (GSTs) are catalyzing the nucleophilic conjugation of GSH to a wide variety of hydrophobic and electrophilic organic molecules to yield often inactive, water soluble, lesser non-toxic peptide derivatives than the parent compounds (Marrs 1996).
In order to tolerate high levels of metals, plants possess several sulphur-containing compounds, including phytochelatins (PCs) and metallothioneins (MTs) to transport and sequester the accumulated metals (Cobbett and Goldsbrough 2002; Gonzalez-Mendoza et al. 2007). MTs are low-molecular-weight proteins that have the capacity to bind toxic heavy metals through the thiol groups of their cysteine residues, thus clearly increasing the resistance of the cells to heavy-metal toxicity (Cobbett and Goldsbrough 2002). The effect of heavy metals on the expression level of MT genes has been studied in different plant species, which can vary with the plant species, tissue, and MT type (Ahn et al. 2012; Schiller et al. 2014). Our previous study revealed that the transcript abundance of Brassica napus, metallothionein protein (BnMP1) was up-regulated by Cr(VI) treatment for 1 day, but was repressed after 7 days (Yıldız et al. 2013). Currently, no literature is available on the effects of combined stress of sulfur deficiency and Cr(VI) toxicity on the expression levels of the MT genes in canola.
Sulfur (S) is a component of the amino acids cysteine and methionine, cofactors, metal clusters, and a diverse range of primary and secondary metabolites, such as glutathione, phytochelatins, and glucosinolates that protect plants from oxidative and environmental stresses (Rausch and Wachter 2005). Adaptation of the sulfur metabolic circuit is a crucial component of plant survival under heavy metal stress (Nocito et al. 2007). A number of reports are available regarding the S metabolism and Cr(VI) stress (Schiavon et al. 2007, 2008, 2012). In these studies, Cr(VI) can influence not only sulfate transport, but also S assimilation. Chromium caused the reduction in sulfate uptake capacity and transcript abundance of the low affinity sulfate transporters in both S sufficient and S-starved Brassica juncea plants (Schiavon et al. 2012). Moreover, genes coding for enzymes of the S assimilatory pathway were up-regulated in Zea mays and B. juncea plants in response to Cr (Schiavon et al. 2007, 2008). In addition, the over-production of those enzymes has been found to increase resistance to various trace elements (Wangeline et al. 2004; Ivanova et al. 2011). Therefore, S-containing compounds are thought to primarily play an important role in the detoxification of heavy metals in plants (Na and Salt 2011).
Chromium tolerance differs greatly among plant species and among genotypes within a species. Canola has been studied for its potential use in phytoremediation due to its characteristics of fast-growing, high biomass and considerable tolerance to heavy metals (Shams et al. 2010; Yu et al. 2012). However, a wide range of tolerance has been observed among canola cultivars. Cr(VI) tolerant cultivars display less oxidative stress compared to Cr(VI) susceptible cultivars of canola (Terzi and Yıldız 2014). In addition, Brassica species are characterized by a high sulfur requirement for growth. According to our knowledge, there is no existing work on the effect of different S levels on Cr(VI) toxicity in canola plants. Thus, the current research was conducted in order to study the effect of S levels on Cr(VI) toxicity in two canola cultivars differing in Cr(VI) tolerance. Sulfur-deficiency experiments were conducted to observe the impact of Cr(VI) on the plants when no S is supplied. Thus, to determine the importance of the sulfur availability in Cr(VI)-stressed canola plants, we analyzed the changes in plant growth, Cr accumulation, chlorophyll content, thiol contents, lipid peroxidation, antioxidant enzymes, key components of AsA–GSH pathway, and expression pattern of BnMP1 mRNA.
Materials and methods
Plant material and treatments
Two canola (B. napus L.) cultivars were selected: NK Petrol, a relatively Cr(VI)-tolerant cultivar, and Sary, a relatively Cr(VI)-susceptible cultivar (Terzi and Yıldız 2014). The seeds were germinated on moisture filter paper in dark at 23 °C. Two-day-old seedlings were selected, and ten seedlings per pot were transplanted into 1 L plastic pots containing nutrient solution with the following composition (μM): 200 KNO3, 200 Ca(NO3)2, 40 KH2PO4, 1000 MgSO4, 10 FeNa2EDTA, 4.6 H3BO3, 0.16 CuCl2, 0.9 MnCl2·2H2O, 0.09 ZnCl2 0.01 NaMoO4·2H2O (Schiavon et al. 2008). The seedlings were then grown hydroponically in a growth chamber (Climacell 404, MMM Medcenter Einrichtungen GmbH, Germany) under controlled environmental conditions (constant temperature of 23 °C, a 16 h photoperiod with photosynthetically active radiation at photon flux density of 260 μmol m−2 s−1, and relative humidity of 60 %) for 1 week. After this period, the seedlings were transferred to nutrient solutions supplied with two levels of sulfur supply: deficient (0 mM S; −S) and sufficient (1 mM S; +S). After keeping the plants in different sets of S supply for 1 week, they were exposed to 100 μM Cr(VI) (supplied as potassium dichromate) under above-mentioned conditions for 3 days. Preliminary experiments with Cr(VI) at 0–300 μM was carried out to determine the appropriate test concentration and duration. The exposure duration was limited to 3 days, because −S plants suffered too much toxicity and damage. Also, S concentrations were selected according to Schiavon et al. (2008). The following four experimental treatments were formed: (1) control, 1 mM sulfate (+S/−Cr), this treatment used as reference to compare treatments; (2) Cr(VI) stress, 1 mM sulfate and 100 μM Cr(VI) (+S/+Cr); (3) S-deficiency, 0 mM sulfate (−S/−Cr); and (4) combined stress, 0 mM sulfate and 100 μM Cr(VI) (−S/+Cr). In the S-deficient treatments, MgCl2 was used instead of MgSO4. The pH of the nutrient solution was adjusted to 6.0 ± 0.1 using 0.05 M KOH, and the nutrient solution was renewed every alternate day. The seedlings were harvested at 3 days after treatments. The second and third leaves from seedlings were sampled for the physiological and molecular analysis.
Plant growth parameters and Cr accumulation
The aerial parts of seedlings were used for the determination of dry weight (DW) and Cr accumulation. For DW, ten seedlings per treatment were weighed and dried in a hot air oven at 80 ± 2 °C for 48 h. A reduction in growth was calculated as a percent of control (+S/−Cr). For Cr accumulation, dried samples (1 g) were acid digested in HNO3. The Cr accumulation (μg g−1 DW) was determined by an inductively coupled plasma-optical emission spectroscopy (ICP-OES, Perkin Elmer Optimal 2100DV). Chromium standard solutions were prepared by diluting 1000 ppm standard (Inorganic Ventures, Christiansburg, Virginia, USA; CGCR(3)1-1, Lot # A2-CR03007), and they were utilized to calibration and quality assurance. Analytical data quality of metal was ensured with repeated analysis of quality control samples and the results were within the certified values. Analytical limit of detection was 0.05 mg L−1 for Cr. The analytical recovery range of Cr was 96.9 %, which was measured up to the analytical demand (QA/QC).
Oxidative damage and chlorophyll content
Lipid peroxidation in leaf tissues was measured in terms of malondialdehyde (MDA) determined by thiobarbituric acid (TBA) reactions as described by Heath and Packer (1968). Leaf tissues (250 mg) were frozen in liquid nitrogen, ground in a pre-chilled mortar and homogenized in 5 mL of 0.1 % trichloroacetic acid. The homogenate was centrifuged at 10,000×g for 10 min. Four volume of 0.5 % thiobarbituric acid in 20 % trichloroacetic acid was added to one volume of the supernatant. After heating for 30 min at 95 °C in a water bath, the mixture was cooled and centrifuged. The amount of MDA was calculated by difference in absorbance at 532 and 600 nm using the extinction coefficient of 155 mM−1 cm−1. Total chlorophylls (Chl a + b) in leaf tissues (100 mg) were extracted and estimated according to Wellburn (1994).
Cysteine and non-protein thiol content
The cysteine content was determined according to the standard protocol as described by Gaitonde (1967). The leaf tissues (500 mg) were extracted in 5 % chilled perchloric acid and the homogenate was centrifuged at 10,000×g for 10 min at 4 °C. The supernatant was treated with equal volumes of acid ninhydrin reagent and glacial acetic acid. The absorbance was read at 560 nm and the cysteine content was determined against the standard curve of l-cysteine. The cysteine content was expressed as nmol g−1 FW.
Non-protein thiols (NPT) were estimated according to Nagalakshmi and Prasad (2001) with some modifications described in Zhang et al. (2009). The leaf tissues (500 mg) were homogenized in 20 mM EDTA with 20 mM ascorbate and the homogenates were centrifuged at 12,000×g for 20 min at 4 °C. Aliquots of the supernatant were mixed with 10 % (w/v) sulfosalicylic acid. The contents were mixed, incubated for 15 min, and centrifuged at 3000×g for 15 min. Then the resulting supernatant fraction was mixed with 400 mM Tris–HCl (pH 8.9) and 10 mM Ellman’s reagent. The absorbance at 412 nm was measured within 5 min of mixing.
Assays of enzymes and metabolites of antioxidant system
Fresh leaves (5 mg) were ground in 5 mL of 50 mM cold phosphate buffer (pH 7.0) containing 1 % (w/v) soluble polyvinylpyrrolidone and centrifuged at 15,000×g for 20 min at 4 °C. The supernatant was used for the determination of the activities of antioxidant enzymes. The protein concentration of the supernatant was determined according to Bradford (1976) using bovine serum albumin as a standard. SOD activity was assayed on the basis of its ability to inhibit the photochemical reduction of nitro blue tetrazolium (NBT) (Beauchamp and Fridovich 1971). Total SOD activity was expressed as unit mg−1 protein. APX activity was measured following the method of Nakano and Asada (1981). Enzyme activity was expressed as amount of AsA oxidized per min per mg protein. The GPX activity was measured following Chance and Maehly (1955). One unit of enzyme activity was defined as the amount of H2O2 decomposed per min per mg protein. The activity of CAT was assayed following H2O2 decomposition at 240 nm for 3 min according to Aebi (1984). Glutathione reductase (GR) assay was carried out according to Carlberg and Mannervik (1985). A unit of activity is the amount of enzyme that catalyzes the reduction of 1 μmol of GSSG min−1 mg−1 protein. Glutathione S-transferase (GST) activity was determined according to Habig and Jacoby (1981). The enzyme activity was calculated from the absorption coefficient of 9.6 mM−1 cm−1 and expressed as units min−1 mg−1 protein.
The measurement of total AsA (AsA + DHA) and reduced ascorbate (ASC) contents were determined by the modified method of Hodges et al. (1996). The fresh leaf tissues (300 mg) were homogenized in 5 % m-phosphoric acid. The homogenate was centrifuged at 12,000×g for 20 min. Total ascorbate was determined in a reaction mixture consisting of 100 μL of supernatant, 500 μL of 150 mM potassium phosphate containing 5 mM EDTA and 100 μL of 10 mM dithiothreitol (DTT) to reduce DHA to AsA. After 50 min at room temperature, 100 μL of 0.5 % N-ethylmaleimide was added to remove excess DTT. Then 400 µL of 0.61 M TCA, 400 µL of 0.8 M orthophosphoric acid, 400 µL of α,α′-bipyridyl in 70 % ethanol, and 200 μL of 30 g L−1 were added and the mixture was incubated at 40 °C for 1 h and the absorbance was detected at 525 nm. AsA content was determined by adding distilled water instead of DTT and N-ethylmaleimide and then followed the same method as above. Total AsA and AsA contents were estimated from the standard curve. DHA contents were calculated by subtraction of AsA from total AsA.
Reduced glutathione (GSH) and GSSG levels were measured according to the method of Griffith (1980). 500 mg of fresh leaf was homogenized in 5 % sulfosalicylic acid at 4 °C. The homogenate was centrifuged at 12,000×g for 10 min at 4 °C. A 300 μL aliquot of the supernatant was neutralized by the addition of 18 μL 7.5 M triethanolamine. For the determination of GSSG alone, a 150 μL sample was treated with 3 μL 2-vinylpyridine for 60 min at 20 °C to mask GSH by derivatization. Another 150 mL sample was used to determination of total glutathione (GSH + GSSG). Fifty microliters aliquots of both types of samples were mixed with 700 μL of 0.3 mM NADPH, 100 μL of 10 mM DTNB and 150 μL of 125 mM NaPO4–6.3 mM EDTA buffer pH 6.5. Reaction was started by adding 10 µL of GR (5 U mL−1) and the change in absorbance at 412 nm was monitored. A standard curve prepared by using GSH and GSSG was used in calculating the amounts of GSH and GSSG.
Quantification of BnMP1 mRNA expression
The total RNA of the leaf tissues were extracted using RNeasy Plant Mini Kit (Qiagen, Hilden, Germany) according to the manufacturer’s instructions. The first strand of cDNA was synthesized using 1.0 μg of total RNA, random primers, and reverse transcription system (QuantiTect® Reverse Transcription Kit, Qiagen) in a total volume of 20 μL. Reverse transcription was performed in a thermal cycler (Techne TC3000G) using the following program: 42 °C for 30 min, 95 °C for 3 min and 4 °C as a final hold.
Primers used for the amplification of target cDNAs were designated according to B. napus genes available in the databank (http://www.ncbi.nlm.nih.gov/). The B. napus complete metallothionein mRNA sequence (GenBank accession No. GU189581.1) was used to design primers for RT-PCR. The sense and antisense primers were 5′-taccaccacccagactcttg-3′ and 5′-gcagacagatccgcacttg-3′, respectively. Brassica napus β-actin gene (GenBank accession No. AF111812.1) was used as an internal control to verify real-time quantitative PCR, and primers were 5′-cagaccgtatgagcaaagagat-3′ and 5′-ctgagggaagcaagaatggaa-3′. Real-time quantitative PCR amplification was performed with Rotor-Gene Q (Qiagen) using the QuantiTect® Probe RT-PCR Master Mix (Qiagen) according to the manufacturer’s instructions. PCR conditions consisted of a 95 °C for 15 min, 40 cycles of 95 °C for 15 s, 60 °C for 60 s. The abundance of targeted gene transcripts was normalized to β-actin mRNA and fold induction in BnMP1 mRNA expression relative to the control was determined by the standard 2−ΔΔCT method of Livak and Schmittgen (2001).
Statistical analysis
All experiments were done on a completely randomized design. Each data was a mean of the two independent experiments with six replicates. All data were compared by the analysis of variance (ANOVA) using SPSS.17 (SPSS Inc., Chicago, IL, USA). Duncan’s multiple range test (DMRT) at 5 % probability was used to test the significant differences among the mean values.
Results and discussion
Plant growth and Cr accumulation
The effects of different treatments of S and Cr(VI) on plant growth in terms of dry weight (DW) of Cr(VI)-tolerant NK Petrol and Cr(VI)-susceptible Sary canola cultivars are shown in Table 1. S-deficiency treatment (−S/−Cr) resulted into a significant decrease in DW as compared to the control (+S/−Cr), but this reduction was significantly higher in susceptible Sary than in tolerant NK Petrol canola cultivar (P < 0.05). Cr(VI) stress significantly reduced DW only in susceptible cultivar Sary. The maximum decline in DW was observed under combined stress (−S/+Cr). The inhibition of DW accumulation in the absence of S may be related to the impaired chlorophyll biosynthesis and down-regulation of photosynthetic enzymes (Nikiforova et al. 2003; Lunde et al. 2008). Also increased photosynthetic process was found to be the basis for the increased DW under heavy metal stress in general and Cr in particular (Bishnoi et al. 1993). Furthermore, Cr ions are known also to compete with S for carrier binding resulting in reduced plant growth (Shanker et al. 2005). However, a significant decrease of transcript accumulation for the low affinity sulfate transporter accompanied by the reduction in sulfate uptake rates was observed in B. juncea plants under Cr(VI) stress (Schiavon et al. 2008). In our study, Cr accumulation in the aerial parts of two canola cultivars is shown in Table 1. After 3 days of Cr(VI) exposure, the accumulation of Cr in both canola cultivars was significantly higher in S-deficient seedlings (P < 0.05). Cr accumulation was found significantly higher in Sary than in NK Petrol in the presence or absence of S. These results showed that S and Cr(VI) seemed to compete for uptake because Cr levels were higher in the S-deficient seedlings than in the S-sufficient seedlings which is in agreement with many previous findings (Schiavon et al. 2008, 2012). Between the two cultivars of canola, the more reduction in seedling growth of susceptible cultivar Sary might be due to more accumulation of Cr.
Oxidative stress and chlorophyll content
Compared to the control, Cr(VI) stress significantly increased MDA content by 1.2–1.3 fold in Cr(VI)-tolerant cultivar NK Petrol while increased by 1.3–1.7 fold in Cr(VI)-susceptible cultivar Sary in the absence and presence of S (P < 0.05) (Fig. 1a). This result suggests that high level of endogenous Cr induced production of ROS leading to an increase in lipid peroxidation of the susceptible cultivar Sary more than the tolerant cultivar NK Petrol. In contrast to previous studies on Brassica campestris (Anjum et al. 2008) and Arabidopsis thaliana (Bashir et al. 2013), S-deficiency treatment (−S/−Cr) alone did not affect MDA content in both canola cultivars. Leaf chlorosis is one of the earliest and most distinct symptoms of S deficiency in plants (Lunde et al. 2008); it was also observed under Cr(VI) stress (Schiavon et al. 2012). In the present study, the effects of different S concentrations and Cr(VI) stress on chlorophyll content of two canola cultivars are depicted in Fig. 1b. As compared to the control (+S/−Cr), S-deficiency treatment (−S/−Cr) caused a significant reduction (23.6 %) in chlorophyll content of susceptible cultivar Sary (P < 0.05). However, Cr(VI) stress had a significant decreasing effect on chlorophyll content of Sary both in the presence and absence of S, whereas only combined stress (−S/+Cr) decreased chlorophyll content of NK Petrol. In both canola cultivars, the depression of biomass production upon exposure to Cr(VI) stress and S-deficiency may be related with the decrease in chlorophyll content as well as with the increase of MDA content.
Cysteine and non-protein thiol content
As a precursor of GSH and PCs, cysteine (Cys) has essential roles in various biological functions, including maintaining protein structure and redox control (Droux 2004). The Cys contents of both canola cultivars were significantly increased by +S/+Cr (P < 0.05) (Fig. 2a). However, Cys content showed no significant changes in −S/−Cr treatment compared to the control. As compared to the control, the Cys content of Cr(VI)-tolerant cultivar NK Petrol was significantly increased by −S/+Cr treatment, while the Cys content of Cr(VI)-susceptible cultivar Sary was not affected. These results may indicate that the pathway for Cys biosynthesis in plants is a critically important metabolic process and it plays a role in Cr tolerance.
In plants, homeostasis and detoxification of toxic metals are related to the metal binding capacity of peptides and proteins, which have a high affinity to thiol groups (Ernst et al. 2008). As compared to the control, S-deficiency treatment (−S/−Cr) caused a significant decrease in the NPT contents of both canola cultivars (P < 0.05) (Fig. 2b). Cr(VI) stress under the absence (−S/+Cr) and presence (+S/+Cr) of S resulted into a significant increase in the NPT content, but this increase was significantly higher in susceptible Sary than in tolerant NK Petrol. Increased NPT content accompanied with increased Cr accumulation suggests that thiol-containing chelators may play an important role in the sequestration of Cr in canola seedlings.
Enzymes and metabolites of antioxidative defense system
Defense mechanism of plants produces antioxidant enzymes to combat the stress by reducing ROS to harmless products. In the present study, Cr(VI) stress significantly increased SOD activity in two S treatments, but this increase was significantly higher in Cr(VI)-tolerant NK Petrol than in Cr(VI)-susceptible Sary canola cultivar (P < 0.05) (Fig. 3a). However, a combined stress (−S/+Cr) led to more induction of SOD activity in both canola cultivars as compared to the control. S deficiency caused no significant changes in SOD activity of both canola cultivars. This might indicate that the capacity of the tolerant cultivar NK Petrol under S deficiency for the O ·−2 dismutation was also high and hence it experienced lower oxidative stress. In −S and +S treatments, GPX activity showed an increase in response to Cr(VI) as compared to the control (Fig. 3b). However, GPX activity of the susceptible cultivar Sary showed significantly greater increase than that of the tolerant cultivar NK Petrol under Cr(VI) stress (P < 0.05). Similar to SOD activity, a combined stress (−S/+Cr) led to more increase in GPX activity, and this may be either due to more accumulation of Cr in canola plants (Table 1). Asada (1992) also reported that the potential cellular mechanisms in detoxification of heavy metals include their sequestration in the cell wall and vacuole where the GPX is especially localized (Asada 1992).
Effects of Cr(VI) stress (+S/+Cr), S-deficiency (−S/−Cr) and combined stress of S-deficiency and Cr(VI) stress (−S/+Cr) on the activities of SOD (a), GPX (b), APX (c), CAT (d), GR (e) and GST (f) in the leaves of two canola cultivars. Columns marked with the different letters are significantly different (DMRT, P < 0.05)
H2O2, a product of SOD activity, is also toxic to cells and has to be further detoxified by CAT and the ascorbate–glutathione cycle. APX has a higher affinity for H2O2, allowing for the scavenging of small amount of H2O2 in more specific locations (Mittler 2002). In −S treatment, APX activity of Cr(VI)-tolerant NK Petrol was significantly increased than that of Cr(VI)-susceptible Sary (P < 0.05) (Fig. 3c). Application of Cr(VI) stress tended to increase APX activity in −S and +S treatments, but this increase was significantly higher in tolerant NK Petrol than in susceptible Sary. The increase in APX activity of NK Petrol under S-deficiency contradicted the findings of Bashir et al. (2013) who showed the decrease in APX activity in S-deficient Arabidopsis plants. This may be related to the ability of canola seedlings to tolerate the S deficiency. In contrast to SOD, GPX and APX enzymes, a decreasing trend was observed in CAT activity in response to +S/+Cr and −S/+Cr treatments (Fig. 3d) which may indicate the loss of function in degradation of H2O2. A reduction in CAT activity was significantly higher in susceptible Sary than in tolerant NK Petrol under −S/+Cr treatment (P < 0.05). The decrease in the activity of the enzyme may be probably attributed to an affinity of Cr ions to thiol compounds disturbing protein synthesis and enzyme activity. Antioxidative enzymes work in a cooperative or synergistic way to protect against oxidative stress (Liang et al. 2008). Between the two cultivars of canola, the tolerant cultivar NK Petrol showed higher values of SOD, APX and CAT activities than the susceptible cultivar Sary in response to Cr(VI) stress and S deficiency. Therefore, greater increases in the activities of antioxidant enzymes result in the detoxification of ROS which may be beneficial to plant growth under combined stress.
Glutathione reductase is one of the key components of the cells’ scavenging system for ROS in plants (Anjum et al. 2012). In the present study, GR activity was significantly decreased in −S/−Cr and +S/+Cr treatments compared to the control (P < 0.05) (Fig. 3e). GR activity in −S/+Cr treatment was significantly increased when compared to the –S/−Cr treatment, but this increase was significantly higher in tolerant NK Petrol than in susceptible Sary. It was interesting to see an increase in GR activity in response to S-deficiency and Cr(VI) stress which might be due to early use up of available S. A similar trend was also observed in Arabidopsis plants under Cd stress (Bashir et al. 2013). Heavy metal induced increase in GR activity firstly allows AsA–GSH pathway to operate at a high rate to detoxify ROS and secondly, provides GSH for the synthesis of PCs and to be used by peroxide-metabolizing enzymes such as GSTs (Anjum et al. 2012). Also induction of PCs in response to Cr stress has been reported in B. juncea (Diwan et al. 2010). However, the decrease in GR activity in response to Cr(VI) may be the result of a direct reaction of metal with sulfhydryl groups interfering with glutathione cycle since Cr is known to bind to the thiol group and thereby inactivate the thiol-containing enzymes (Diwan et al. 2010). Under such circumstances, the alterations in GR activity indicated that maintenance of a relative redox balance of GSH pool was favored under conditions of abiotic stresses (Noctor et al. 2002).
S-deficiency treatment (−S/−Cr) caused a significant increase in GST activity in Cr(VI)-tolerant cultivar NK Petrol, whereas it decreased the activity in Cr(VI)-susceptible cultivar Sary (P < 0.05) (Fig. 3f). A combined stress (−S/+Cr) resulted in a further significant increase of GST activity in NK Petrol, while it caused no significant changes in Sary. In S availability, Cr(VI) stress decreased the GST activity in both canola cultivars. GSTs are GSH-dependent ROS detoxifying enzymes catalyzing the nucleophilic conjugation of GSH to electrophilic xenobiotic substrates to yield often less- or non-toxic peptide (Marrs 1996). An increase in GST activity was also found in Cr(VI)-stressed B. juncea plants (Pandey et al. 2005). Moreover, an overexpression of a rice GST gene (OsGSTL2) in Arabidopsis provides tolerance to different abiotic stresses including heavy metals (Kumar et al. 2013). Our results showed that the increase in GST activity under S-deficiency contradict the findings of Bashir et al. (2013) who showed the decrease in GST activity in S-deficient Arabidopsis plants. In our study, the activity of GST showed a similar trend to that of GR. Glutathione reductase and GSTs, in conjunction with SODs form an efficient system to protect the plants against ROS and/or their reaction products effects during various stress conditions in plants (Halušková et al. 2009). GSH-related enzyme activities in plants may be related to higher resistance or higher susceptibility where a plant can be regarded as tolerant to heavy metals which efficiently maintains a strong coordination between the oxidation and reduction of GSH pool with its regenerating and metabolism enzymes (Anjum et al. 2012). Overall, it is suggested that higher SOD, APX, CAT, GR and GST activities in Cr(VI)-tolerant cultivar NK Petrol under the S-deficiency and Cr(VI) stress might confer plant tolerance by moderating ROS levels and alleviating Cr(VI)-induced lipid peroxidation.
As compared to control (+S/−Cr), S-deficiency (−S/−Cr) significantly decreased AsA content in both cultivars (P < 0.05) (Fig. 4a). A combined stress (−S/+Cr) caused a further significant reduction in AsA content. In +S/+Cr treatment, AsA content was decreased more significantly in NK Petrol than in Sary. The level of DHA also showed significant changes in various treatments (data not shown). The ratio of AsA/DHA was decreased upon exposure to both S-deficiency and Cr(VI) stress as compared to the control (Fig. 4b). AsA eliminates ROS through coordination with the production of GSH in AsA–GSH cycle (Noctor et al. 2002). In the present study, canola cultivars under S-deficiency and Cr(VI) stress experienced extensive oxidation of the AsA pool. This was evident from decreasing AsA/DHA ratios in plants exposed to S-deficiency and Cr(VI). Moreover, our results are contrary to the findings of Srivastava and D’Souza (2010) who found the increase in AsA level in S-deficient Hydrilla verticillata plants.
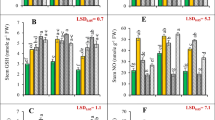
As a significant component of AsA–GSH pathway, GSH is involved in both direct and indirect control of ROS, and thus protects plants against heavy metal induced oxidative stress (Anjum et al. 2012). As compared to the control (+S/−Cr), S-deficiency (−S/−Cr) caused a significant decrease in the GSH content of both canola cultivars (P < 0.05) (Fig. 5a). A number of reports are available regarding the depletion of GSH pool in Cr(VI)-exposed and S-deficient plants (Schiavon et al. 2007, 2008; Bashir et al. 2013). In +S availability, Cr(VI) stress significantly decreased GSH content only in NK Petrol. In −S/+Cr treatment, GSH contents of both canola cultivars were significantly increased, but this increase was significantly higher in Cr(VI)-susceptible Sary than in Cr(VI)-tolerant NK Petrol canola cultivar. Similar result was observed by Schiavon et al. (2008) in B. juncea. In our study, the exceptionally high GSH levels in −S/+Cr-treated seedlings may also be due to elevated level of GR activity. On the other hand, high ratios of GSH-Cd in the phloem sap of canola suggested that GSH can function as long-distance carriers for Cd (Mendoza-Cozatl et al. 2008). In this context, high levels of GSH in the leaves of −S/+Cr-treated seedlings may be related to translocation of GSH-Cr complex from roots to leaves. Maintaining a high ratio of GSH/GSSG that functions as a redox couple was shown to play an important role in heavy metal tolerance (Anjum et al. 2012). In our study, GSH/GSSG ratio was significantly increased by S deficiency as well as by Cr(VI) stress in both canola cultivars, but this increase was significantly higher in the susceptible Sary than in tolerant NK Petrol (Fig. 5b). The lower GSH concentration and GSH/GSSG ratio in the tolerant cultivar NK Petrol may be due to the higher activity of GST which utilizes GSH in cellular detoxification or to maintain high levels of PCs which are produced from GSH. These results showed that stimulated GSH synthesis may be responsible for the improved Cr accumulation but not Cr(VI) tolerance in canola plants.
Expression of BnMP1 gene
As the plant MTs efficiently bind with metals (Huang et al. 2011) and some MT genes are positively regulated by metals, MTs are thought to be involved in cellular metal homeostasis and tolerance mechanisms (Cobbett and Goldsbrough 2002). To date, several studies have analyzed the expression pattern of MT mRNAs in plant species in response to Cr(VI) stress (Goupil et al. 2009; Yıldız et al. 2013). In the present study, to test the effects of different S concentrations and Cr(VI) stress on BnMP1 gene, the expression level of BnMP1 mRNA was investigated in leaves of Cr(VI)-tolerant NK Petrol and Cr(V)-susceptible Sary canola cultivars. The expression level of BnMP1 gene showed different responses between two canola cultivars in response to Cr(VI) stress in the presence or absence of S (Fig. 6). As compared to the control (+S/−Cr), S-deficiency (−S/−Cr) caused no significant changes in the expression levels of both canola cultivars. A combined stress of S deficiency and Cr(VI) stress (−S/+Cr) resulted into an increase in expression level of BnMP1 gene only in NK Petrol. However, Cr(VI) stress increased the expression level only in Sary under S-sufficient condition (+S/+Cr). These results indicate that MTs may be involved in Cr(VI) tolerance but only under S deficiency not during availability of S. The induction of the expression of BnMP1 gene in susceptible cultivar Sary under Cr(VI) stress suggests that its encoded protein may not function in S-dependent Cr(VI) tolerance.
Effects of Cr(VI) stress (+S/+Cr), S-deficiency (−S/−Cr) and combined stress of S-deficiency and Cr(VI) stress (−S/+Cr) on the expression level of BnMP1 gene in the leaves of two canola cultivars. The relative BnMP1 gene expression level as expressed by 2−ΔΔCT was determined for each group and normalized with housekeeping β-actin transcripts. Columns marked with the different letters are significantly different (DMRT, P < 0.05)
Conclusion
The results of the present work show that a combined stress of S deficiency and Cr(VI) stress results in an increase of oxidative stress leading to membrane destruction which, in turn, affects the antioxidative defense systems in two canola cultivars differing in Cr(VI) tolerance. The alterations in dry matter, lipid peroxidation and chlorophyll contents indicated that Sary seems to be more susceptible to S deficiency and Cr(VI) treatments than NK Petrol. The levels of Cr in the aerial parts of the tolerant cultivar NK Petrol were lower than that of the susceptible cultivar Sary suggesting that NK Petrol could effectively exclude Cr ions from photosynthetic tissues. The higher activities of antioxidant enzymes such as SOD, APX, CAT, GR and GST may probably lead to greater levels of detoxification mechanisms of ROS in Cr(VI)-tolerant cultivar NK Petrol than in Cr(VI)-susceptible Sary in response to S-deficiency and Cr(VI) stress treatments. Since Cr(VI) stress led to the accumulation of Cys, our results suggest reduced S compounds may play a crucial role in Cr(VI) tolerance under S-deficient conditions. Our findings also show that MT may be involved in Cr(VI) tolerance under S-deficiency. On the other hand, Cr(VI) and sulfate seemed to compete for uptake because Cr accumulation was higher in the absence of S than in the presence of S. Under field conditions, supplementary S-fertilization could enhance S-containing defense compounds which are crucial for the survival of plants exposed to abiotic stresses. Therefore, the S availability in the soil must be taken into consideration when canola plants are going to be used for Cr phytoremediation.
References
Aebi H (1984) Catalase in vitro. Method Enzymol 105:121–126
Ahn YO, Kim HK, Lee J, Kim H, Lee HS, Kwak SS (2012) Three Brassica rapa methallothionein genes are differentially regulated under various stress conditions. Mol Biol Rep 39:2059–2067
Anjum N, Umar S, Ahmad A, Iqbal M, Khan N (2008) Sulphur protects mustard (Brassica campestris L.) from cadmium toxicity by improving leaf ascorbate and glutathione. Plant Growth Regul 54:271–279
Anjum NA, Ahmad I, Mohmood I, Pacheco M, Duatre AC, Pereira E, Umar S, Ahmad A, Khan NA, Iqbal M, Prasad MNV (2012) Modulation of glutathione and its related enzymes in plants’ responses to toxic metals and metalloids-A review. Environ Exp Bot 75:307–324
Asada K (1992) Ascorbate peroxidase-a hydrogen peroxide scavenging enzyme in plants. Physiol Plant 85:235–241
Bashir H, Ahmad J, Bagheri R, Nauman M, Qureshi MI (2013) Limited sulfur resource forces Arabidopsis thaliana to shift towards non-sulfur tolerance under cadmium stress. Environ Exp Bot 94:19–32
Beauchamp C, Fridovich I (1971) Superoxide dismutase: improved assays and an assay applicable to acrylamide gels. Anal Biochem 44:276–287
Bishnoi NR, Chugh LK, Sawhney SK (1993) Effect of chromium on photosynthesis, respiration and nitrogen fixation in pea (Pisum sativum L.) seedlings. J Plant Physiol 142:25–30
Bradford MN (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Carlberg C, Mannervik B (1985) Glutathione reductase. In: Meister A (ed) Methods in enzymology, vol 113. Academic Press, New York, pp 488–495
Chance B, Maehly AC (1955) Assay of catalase and peroxidase. In: Colowick SP, Kaplan NO (eds) Methods in enzymology, 2nd edn. Academic Press, New York, pp 764–775
Chang F-C, Ko C-H, Tsai M-J, Wang Y-N, Chung C-Y (2014) Phytoremediation of heavy metal contaminated soil by Jatropha curcas. Ecotoxicology 23:1969–1978
Cobbett C, Goldsbrough P (2002) Phytochelatins and metallothioneins: roles in heavy metal detoxification and homeostasis. Annu Rev Plant Biol 53:159–182
Diwan H, Khan I, Ahmad A, Iqbal M (2010) Induction of phytochelatins and antioxidant defence system in Brassica juncea and Vigna radiata in response to chromium treatments. Plant Growth Regul 61:97–107
Droux M (2004) Sulfur assimilation and the role of sulfur in plant metabolism: a survey. Photosynth Res 79:331–348
Ernst WHO, Krauss GJ, Verkleij JAC, Wesenberg D (2008) Interaction of heavy metals with the sulphur metabolism in angiosperms from an ecological point of view. Plant Cell Environ 31:123–143
Gaitonde MK (1967) A spectrophotometric method for the direct measurement of cysteine in the presence of other naturally occurring amino acids. Biochem J 104:627–633
Gill SS, Tuteja N (2010) Reactive oxygen species and antioxidant machinery in abiotic stress tolerance in crop plants. Plant Physiol Biochem 48:909–930
Gonzalez-Mendoza D, Morenob AQ, Zapata-Perez O (2007) Coordinated responses of phytochelatin synthase and metallothionein genes in black mangrove, Avicennia germinans, exposed to cadmium and copper. Aquat Toxicol 83:306–314
Goupil P, Souguir D, Ferjani E, Faure O (2009) Expression of stress related genes in tomato plants exposed to arsenic and chromium in nutrient solution. J Plant Physiol 166:1446–1452
Griffith OW (1980) Determination of glutathione and glutathione disulfide using glutathione reductase and 2-vinylpyridine. Anal Biochem 106:207–212
Habig WH, Jakoby WB (1981) Assay for differentiation of glutathione S-transferases. In: Jakoby WB (ed) Methods in enzymology, vol 77. Academic Press, New York, pp 398–405
Halušková L, Valentovičová K, Huttová J, Mistrĭk I, Tamás L (2009) Effect of abiotic stresses on glutathione peroxidase and glutathione S-transferase activity in barley root tips. Plant Physiol Biochem 47:1069–1074
Heath RL, Packer L (1968) Photoperoxidation in isolated chloroplasts I. Kinetics and stoichiometry of fatty acid peroxidation. Arch Biochem Biophys 125:189–198
Ho J-R, Ma H-W, Wang Y-C, Ko C-H, Chang F-C, Feng F-L, Wang Y-N (2014) Extraction of heavy metals from contaminated soil by Cinnamomum camphora. Ecotoxicology 23:1987–1995
Hodges DM, Andrews JC, Johnson DA, Hamilton RI (1996) Antioxidant compound responses to chilling stress in different sensitive inbred maize lines. Physiol Plant 98:685–692
Huang GY, Wang YS, Ying GG (2011) Cadmium-inducible BgMT2, a type 2 metallothionein gene from mangrove species (Bruguiera gymnorrhiza), its encoding protein shows metal-binding ability. J Exp Mar Biol Ecol 405:128–132
Ivanova LA, Ronzhina DA, Ivanov LA, Stroukova LV, Peuke AD, Rennenberg H (2011) Overexpression of gsh1 in the cytosol affects the photosynthetic apparatus and improves the performance of transgenic poplars on contaminated soil. Plant Biol 13:649–659
Kumar S, Asif MH, Chakrabarty D, Tripathi RD, Dubey RS, Trivedi PK (2013) Expression of a rice Lambda class of glutathione S-transferase, OsGSTL2, in Arabidopsis provides tolerance to heavy metal and other abiotic stresses. J Hazard Mater 248–249:228–237
Liang YC, Zhu J, Li ZJ, Chu GX, Ding YF, Zhang J, Sun WC (2008) Role of silicon in enhancing resistance to freezing stress in two contrasting winter wheat cultivars. Environ Exp Bot 64:286–294
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real time quantitative PCR and the 2−ΔΔCT method. Methods 25:402–408
Lunde C, Zygadlo A, Simonsen HT, Nielsen PL, Blennow A, Haldrup A (2008) Sulfur starvation in rice: the effect on photosynthesis, carbohydrate metabolism, and oxidative stress protective pathways. Physiol Plant 134:508–521
Marrs KA (1996) The functions and regulation of glutathione S-transferases in plants. Annu Rev Plant Physiol Plant Mol Biol 47:127–158
Mendoza-Cózatl DG, Butko E, Springer F, Torpey JW, Komives EA, Kehr J, Schroeder JI (2008) Identification of high levels of phytochelatins, glutathione and cadmium in the phloem sap of Brassica napus. A role for thiol-peptides in the long-distance transport of cadmium and the effect of cadmium on iron translocation. Plant J 54:249–259
Mittler R (2002) Oxidative stress, antioxidants and stress tolerance. Trends Plant Sci 7:405–441
Na G, Salt DE (2011) The role of sulfur assimilation and sulfur-containing compounds in trace element homeostasis in plants. Environ Exp Bot 72:18–25
Nagalakshmi N, Prasad MNV (2001) Responses of glutathione cycle enzymes and glutathione metabolism to copper stress in Scenedesmus bijugatus. Plant Sci 160:291–299
Nakano Y, Asada K (1981) Hydrogen peroxide is scavenged by ascorbate-specific peroxidase in spinach chloroplasts. Plant Cell Physiol 22:867–880
Nikiforova V, Freitag J, Kempa S, Adamik M, Hesse H, Hoefgen R (2003) Transcriptome analysis of sulfur depletion in Arabidopsis thaliana: interlacing of biosynthetic pathways provides response specificity. Plant J 33:633–650
Nocito FF, Lancilli C, Giacomini B, Sacchi GA (2007) Sulfur metabolism and cadmium stress in higher plants. Plant Stress 1:142–156
Noctor G, Foyer CH (1998) Ascorbate and glutathione: keeping active oxygen under control. Annu Rev Plant Physiol Plant Mol Biol 49:249–279
Noctor G, Gomez L, Vanacker H, Foyer CH (2002) Interactions between biosynthesis, compartmentation and transport in the control of glutathione homeostasis and signaling. J Exp Bot 53:1283–1304
Pandey V, Dixit V, Shyam R (2005) Antioxidative responses in relation to growth of mustard (Brassica juncea cv. Pusa Jaikisan) plants exposed to hexavalent chromium. Chemosphere 61:40–47
Pandey V, Dixit V, Shyam R (2009) Chromium effect on ROS generation and detoxification in pea (Pisum sativum) leaf chloroplasts. Protoplasma 236:85–95
Rausch T, Wachter A (2005) Sulfur metabolism: a versatile platform for launching defence operations. Trends Plant Sci 10:503–509
Schiavon M, Wirtz M, Borsa P, Quaggiotti S, Hell R, Malagoli M (2007) Chromate differentially affects the expression of a high-affinity sulfate transporter and isoforms of components of the sulfate assimilatory pathway in Zea mays (L.). Plant Biol 9:662–671
Schiavon M, Pilon-Smits EAH, Wirtz M, Hell R, Malagoli M (2008) Interactions between chromium and sulfur metabolism in Brassica juncea. J Environ Qual 37:1536–1545
Schiavon M, Galla G, Wirtz M, Pilon-Smits EAH, Telatin V, Quaggiotti S, Hell R, Barcaccia G, Malagoli M (2012) Transcriptome profiling of genes differentially modulated by sulfur and chromium identifies potential targets for phytoremediation and reveals a complex S-Cr interplay. J Hazard Mater 239–240:192–205
Schiller M, Hegelund JN, Pedas P, Kichey T, Laursen KH, Husted S, Schjoerring JK (2014) Barley metallothioneins differ in ontogenetic pattern and response to metals. Plant Cell Environ 37:353–367
Shams KM, Tichy G, Fischer A, Sager M, Peer T, Bashar A, Filip K (2010) Aspects of phytoremediation for chromium contaminated sites using common plants Urtica dioica, Brassica napus and Zea mays. Plant Soil 328:175–189
Shanker AK, Cervantes C, Loza-Tavera H, Avudainayagam S (2005) Chromium toxicity in plants. Environ Int 31:739–753
Shanker AK, Djanaguiraman M, Venkateswarlu B (2009) Chromium interactions in plants: current status and future strategies. Metallomics 1:375–383
Srivastava S, D’Souza SF (2010) Effect of variable sulfur supply on arsenic tolerance and antioxidant responses in Hydrilla verticillata (L.f.) Royle. Ecotoxicol Environ Saf 73:1314–1322
Terzi H, Yıldız M (2014) Variations in chromium tolerance and accumulation among canola (Brassica napus L.) cultivars. Bull Environ Contam Toxicol 93:113–119
Wangeline AL, Burkhead JL, Hale KL, Lindblom SD, Terry N, Pilon M, Pilon-Smits EAH (2004) Overexpression of ATP sulfurylase in Indian mustard: effects on tolerance and accumulation of twelve metals. J Environ Qual 33:54–60
Wellburn AR (1994) The spectral determination of chlorophylls a and b, as well as total carotenoids, using various solvents with spectrophotometers of different resolution. J Plant Physiol 144:307–313
Yıldız M, Terzi H, Bingül N (2013) Protective role of hydrogen peroxide pretreatment on defense systems and BnMP1 gene expression in Cr(VI)-stressed canola seedlings. Ecotoxicology 22:1303–1312
Yu R, Ji J, Yuan X, Song Y, Wang C (2012) Accumulation and translocation of heavy metals in the canola (Brassica napus L.)—soil system in Yangtze River Delta, China. Plant Soil 353:33–45
Zayed AM, Terry N (2003) Chromium in the environment: factors affecting biological remediation. Plant Soil 249:139–156
Zhang H, Lian C, Shen Z (2009) Proteomic identification of small, copper-responsive proteins in germinating embryos of Oryza sativa. Ann Bot 103:923–930
Zhang N, Zang S, Sun Q (2014) Health risk assessment of heavy metals in the water environment of Zhalong Wetland, China. Ecotoxicology 23:518–526
Acknowledgments
The authors wish to thank Afyon Kocatepe University Research Fund (Project No. 12.FEN.BIL.01) for financial support. The authors also wish to thank Afyon Kocatepe University’s Foreign Language Support Unit for language editing.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Terzi, H., Yıldız, M. Interactive effects of sulfur and chromium on antioxidative defense systems and BnMP1 gene expression in canola (Brassica napus L.) cultivars differing in Cr(VI) tolerance. Ecotoxicology 24, 1171–1182 (2015). https://doi.org/10.1007/s10646-015-1468-y
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10646-015-1468-y