Abstract
Mating systems in fishes are extremely diverse, ranging from monogamy to multiple polygamous forms, some of which include alternative male mating tactics within species. The major reproductive mode across approximately 33,000 bony fishes is pelagic-spawning with external fertilization. Despite the dominance of this mode, it is relatively little studied in the field because the greater mobility of larger species typically makes individuals difficult to follow for extensive periods, or to catch and mark. However, smaller, more sedentary pelagic spawners provide excellent opportunities to examine mating system variability in response to environmental conditions. The mandarinfish, Synchiropus splendidus, is a small sedentary pelagic spawner which is accessible to divers. Its complex body markings are distinctive and variable allowing individuals to be identified and followed repeatedly without the need for capturing and marking fish. In over 400 h of underwater surveys, we recorded movements, inter- and intra-sexual behaviors, sex ratios, and the timing and duration of spawning activity of focal fish in predetermined study areas. We concluded that the mandarinfish has a polygamous mating system with pair-spawning. Females are sequentially polyandrous, spawning once nightly or not at all, while males are polygynous, spawning up to 8 times a night. Males and females use loosely defined spawning sites on a daily basis and show flexibility in their tactics through differential movements according to dispersion of individuals, distances between spawning sites and temporally limited daily female receptivity. These results reveal the mandarinfish to be an excellent model species for studying small pelagic spawners in the field.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Mating systems are fundamental components of the lives of plants and animals, shaped by multiple biotic and abiotic factors (Shuster and Wade 2003). They result from the behavior of males and females attempting to maximize their own reproductive success in the context of interactions with members of both sexes and under the influence of environmental conditions. Mating systems are determined by many factors, including spatial and temporal distribution of sexually receptive individuals, resource availability, pre- and post-copulatory behaviors, defensibility of resources (such as females or space), operational sex ratios, risk of predation, timing and duration of the reproductive season, and phenotypic and genotypic factors (Emlen and Oring 1977; Shuster and Wade, 2003; Shuster 2009; Kuijper et al. 2012).
Mating systems are usually defined in terms of numbers of mates per male or per female (Shuster and Wade 2003). In monogamy, a single male and single female have a pair bond and mate exclusively with one another over extended periods or multiple mating seasons (Wickler and Seibt, 1983). In polygamous or polygynandrous species (Shuster and Wade 2003), males and/or females mate with multiple individuals, being often classified as polygynous, polyandrous, or promiscuous according to the number of partners that one or both sexes have. The mating system may vary between populations of a species, or among years within a given population, according to resource availability, environmental conditions, and individual abundance. For example, in several bird species a shift from monogamy to polygamy may occur in response to changes in food or nesting site availability (Carranza 2000; Shuster and Wade 2003). Moreover, mating system definitions do not necessarily account for the mating behavior adopted by all individuals of each sex. In some species, where resources crucial to reproduction are limited or clumped, competition for mates may lead to the evolution of alternative reproductive tactics in either sex. This results in male and/or female phenotypes varying in mating behavior and often in morphology, physiology, and life history traits, within the same population or over time (Oliveira et al. 2008; Taborsky and Brockmann 2010). In species with male alternative reproductive tactics, males often compete among themselves to fertilize the same groups of eggs (Birkhead and Møller 1998; Taborsky and Brockmann 2010). In such matings, as in all promiscuous matings, individual male mating success does not necessarily correspond to fertilization success (Birkhead and Møller 1998). Indeed, processes such as sperm competition, i.e., the contest between sperm of different males to access females’ eggs, and cryptic female choice, i.e., the ability of females to favor the sperm of one male over another, can influence fertilization outcomes (Birkhead and Pizzari 2002).
Fishes, which comprise the majority of vertebrate species, exhibit a particularly diverse array of mating systems and reproductive modes. Of 33,000 species of bony fishes, the majority spawn pelagically (Fitzpatrick 2020). Pelagic spawners tend to be larger, typically exceeding 10-cm total length or so, and more mobile than demersal spawners or internal fertilizers (Thresher 1984). This mobility presents opportunities for different types of male–female interactions and mating tactics. For example, many larger, otherwise solitary, pelagic spawners form temporary mating gatherings (spawning aggregations) of small to large numbers of females and males, by migrating between residential and mating sites (Domeier and Colin 1997). Some aggregating species may spawn in pairs, while others spawn promiscuously in clusters of a single female and multiple males (Sadovy de Mitcheson and Colin 2012). Extensive movements to and from familiar residential areas and aggregation sites, and pelagic egg release, however, both bring challenges. For example, individuals are vulnerable to predation during migrations, and both adults and eggs are particularly exposed during spawning rises to release eggs high in the water column (Sancho et al. 2000a, b; Graham and Castellanos 2012).
As in many other animal taxa, the mating systems of pelagic spawners appear to vary within or among populations and/or can involve alternative reproductive tactics (e.g., Taborsky et al. 2008). For example, parrotfishes and wrasses shift from lek-like polygyny to haremic polygyny in response to changes in population density (Donaldson 2015). Alternative male tactics, depending on opportunities for resource (female or space) monopolization, can include both haremic polygyny with either pair- or promiscuous-spawning (van Rooij et al. 1996; de Girolamo et al. 1999; Wernerus and Tessari 2008). In such cases, smaller/subdominant males spawn in groups involving a single female and multiple males, or parasitize a pair-spawn by a dominant male (Robertson and Warner 1978; Warner and Robertson 1978; Thresher 1984; Marconato and Shapiro 1996; Taborsky 2008; Fitzpatrick 2020). However, most studies on the mating system of pelagic spawners have primarily focused on male behavior, neglecting the female contribution, and many studies do not consider the influence of ecological factors.
The limited information on the mating systems of pelagic spawners is due, in large part, to practical difficulties in studying them in the field. For example, it is challenging to mark, follow and observe individual fish in their natural environment (Amundsen 2003), particularly because most pelagic spawners are larger and/or more mobile compared to demersal species where male territoriality or nest defense is common (Thresher 1984). While studies on fishes can be conducted in aquaria, these are unlikely to fully replicate natural conditions in a way that allows for a comprehensive evaluation of the environmental factors shaping their mating systems and is often challenging with larger species. A few groups of pelagic spawners, however, contain multiple small and relatively sedentary species, such as labrids, serranids, chaetodontids, and cirrhitids and are well suited to detailed field study. Among those, the dragonets (family Callionymidae) exhibit a marked sexual dimorphism and may be readily observed in the field. The gemmous dragonet, Callionymous lyra, was the earliest example of sexual selection identified in marine fishes as expressed in the dramatic extension of the anterior male dorsal fin (Darwin 1871).
Dragonets have been studied in the field and laboratory for 150 years, yet details of their reproductive biology are sparse. Studies variously describe spawning season, diel timing of spawning, mating behaviors, sexual maturation, female preference, sex ratio, age and growth, egg number, and spawning frequency (Holt 1898; Akazaki 1957; Johnson 1972, 1973a, b; Wilson 1978; Gibson and Ezzi 1979; Takita and Okamoto 1979; Fricke and Zaiser 1982; Takita et al. 1983; Zhu et al. 1989, 1991; King et al. 1994; Harrington 1996, 1997; Ikejima and Shimizu 1998; Gonzales et al. 1996, 1997; Narita et al. 2006; Awata et al. 2010; Wittenrich and Ho 2013). The form, relative size, or use of the male anal fin during the spawning rise, or at the time of gamete release, may be important in callionymids according to aquarium studies (Holt 1898; Gonzales et al 1996). Holt (1898), for example, comments that in Callionymus lyra the male anal fin points towards the female on spawning, apparently forming a funnel around the female genital opening during gamete release, that may aid fertilization. It could limit the opportunity for release of sperm by other males (sneak spawning). Despite this long history of research, however, little experimental work and few comprehensive field studies have been conducted on their mating systems, and none consider individual behaviors or interrelationships among known individuals under natural conditions. Hence mating systems in this taxon remain poorly known.
The mandarinfish, Synchiropus splendidus (Herre 1927), is a particularly promising dragonet species for field studies on mating systems. Experimental work on female choice in captive fish showed that females prefer males larger than themselves, and gamete collections in the field showed that the mean number of gametes produced positively correlates with body size in both sexes (Rasotto et al. 2010). There is no male care or other direct benefits to females from mating with large males, and hence females may select these for reasons related to fertilization efficiency, such as sperm number and/or anal fin size. The relatively sedentary nature and small size of the mandarinfish, its ready accessibility, approachability, close substrate association, and abundance in shallow, quiet waters are characteristics that collectively provide an excellent opportunity for detailed examination of the mating system of a small pelagic-spawning fish in the field.
Despite the popularity of this colorful and attractive fish with diver-photographers and aquarists and experimental studies conducted to date, the species has not been studied in detail under natural conditions. It lives in sheltered, slow-moving waters of the tropical Indo-Pacific down to 18 m and ranges from southern Japan to New Caledonia, across Indonesia, and the Philippines, New Guinea and Australia, and east to the Caroline Islands in the western Pacific (Myers 1999). The species can reach 90 mm in total length, and feeds on tiny crustaceans (Debelius and Baensch 1994). The species has a short larval duration among pelagic-spawning species, of about 14 days (Sadovy et al. 2001). The color pattern is the same in both sexes with males reported to attain larger maximum sizes than females and exhibiting sexual dimorphism in their anterior dorsal fin (Myers 1999). It mates, in pairs, at consistently used sites on the reef following elaborate fin displays by courting males (Sadovy et al. 2001; Rasotto et al. 2010).
We conducted a field study in Palau, western Pacific, to determine the mating system of the mandarinfish. Based on current knowledge of its life history, we predicted the species to be polygamous, with both males and females having more than one partner, and to move only locally, hence not forming large spawning aggregations. We expected that the female preference for males larger than themselves, as observed in captivity, would also occur under field conditions, with larger males gaining a higher mating success than smaller ones. We also predicted, given that pair-spawning appears typical of the species and considering indications from other fish species, including callionymids, that the anal fin may be active in males during spawning and could preclude the opportunity for small males to parasitize the spawnings of larger males. We expected that an understanding of reproductive ecology, including activity patterns, mating behavior, density, sex ratios, location, and timing and duration of spawning activity at the individual level, would allow us to define the mating system of a small pelagic spawner in detail, and to determine whether the species exhibits alternative mating tactics.
Methods
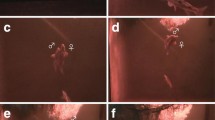
Underwater surveys were conducted in shallow inshore areas of intact coral habitat off Malakal Island, Palau, where mandarinfish are common. A 27 × 27-m2 area of 1–9-m depth was initially selected to determine diel activity patterns. A smaller, 1–3 m deep 12 × 12 m2, area was demarcated from within the larger area for more detailed repeated observations of focal fish. Surveys were conducted on SCUBA. Mature males and females were distinguishable by dorsal fin morphology (Rasotto et al. 2010). Focal fish were individually identifiable using variations in markings on body sides, operculum and dorsal fins, and were each seen on 3 or more days (Fig. 1a–f).
a Individually distinctive markings occur on operculum, the sides of the fish, and on the dorsal fin. Extended fins shown in large male (upper fish) as he courts female, lower fish. (b) Two similarly sized small males displaying at each other, the one on the left chasing the other away. (c) After spawning fish move to nearby sleeping areas, often communally shared, where they became motionless and pale, settling on horizontal coral plates. (d) Spawning pair with medium female above and large male positioned below in horizontal orientation; male anal fin is splayed towards female. (e) Male (left) and female in vertical spawning rise showing extended anal fin of male that shades the area of gamete release. (f) Following spawning the small cloud of egg and sperm (visible to the mid-lower right of the photo) remains intact for at least a few minutes
The study ran from 1997 to 2001 over five survey periods, including winter and summer months, all lunar phases, and involving the same three observers. The five survey periods covered 24.7.97–19.8.97, 4.8.98–25.8.98, 7.12.98–17.1.99, 4.8.99–3.9.99, and 10.8.01–16.9.01, for more than 400 h of underwater observations. Each of the five periods focused on a different set of focal fish, focal spawning sites, and study objectives. Hence sample sizes vary according to each survey period, and numbers of focal spawning sites and fish observed. Inter- and intra-sexual interactions were noted. Observations determined adult sex ratio, individual spawning frequencies, mating associations, patterns of movement, and spawning site use in time and space. Body sizes (total lengths) were visually estimated in-water following work by Rasotto et al. (2010), which determined the relationship between actual measured and estimated lengths to be correlated (Spearman’s correlation: rS = 0.90, P = 0.001, n = 9) with no significant deviation from zero (one sample test, t8 = 2.09, P = 0.070). While we acknowledge that visual length estimations may reduce the accuracy of length determination, fish capture and measuring would have disrupted natural behavior. To increase estimation accuracy, the body size of each individual was determined on the basis of at least 3 visual estimates, performed by different observers (all co-authors). Water temperature at the study site ranged from 26 to 30 °C and was typically 28–29 °C. Lower temperatures occurred during periods of heavy rainfall and the highest was during the El Niño period in the summer of 1998. Adult fish surveyed were between 25 and 60.5 mm TL (females 25–39 mm TL; males 32.5–60.5 mm TL).
Temporal patterns: diel activity and timing of spawning
To determine diel patterns of activity and spawning, observations were initially conducted throughout daylight hours, from dawn to dusk. The 27 × 27-m2 area was surveyed hourly from 6 am to 7 pm for 30 min each hour with each hourly slot repeated 5 times. Three different observers covered the site and all hour slots. The number and sex of individuals observed in each 30-min observation period during swims over the site were noted.
The species is readily detectable when it spawns as fish rise well above the substrate to release gametes. The initial diel surveys of the 27 × 27-m2 area determined that all spawning occurs at dusk; hence, daily observations of spawning behavior focused on this latter part of the day. Spawning events were documented in the smaller 12 × 12-m2 study area to the nearest minute, and the identity of spawners, when known, or their size and sex, were noted. Spawning times were referenced against the “official” time of sunset according to a Garmin GPS model 55. A daily “spawning window” was identified as the time from the first courtship-related display to the time of the last spawn at the spawning site.
Spatial patterns: spawning and sleeping sites and use of space
Substrate surveys were conducted to determine whether particular substrates were preferred for spawning, and, if so, whether these substrates in the study area might be limiting. The 27 × 27-m2 study area was used to characterize substrate availability and the habitat associated with spawning sites. Specific spawning sites where fish regularly gathered in the late afternoon were identified in pilot studies. We analyzed 27 randomly selected quadrats (1 m2), and 20 spawning sites, each demarcated with a removable numbered buoy. We quantified, by percentage occurrence, the 5 predominant substrate types (sand, rubble, Porites rus, forms of “finger” Porites which we assigned to P. cylindrica, and an “other” category consisting mainly of sponges and soft corals) using PVC frames sub-divided by lines into 10-cm2 units. We also surveyed the smaller 12 × 12-m2 area in detail to identify the substrate type at identified spawning sites.
Use of space (i.e., areas of activity, AoAs) by focal males and females was determined by regularly plotting their positions on a map, printed on underwater paper, in the latter daylight hours (spawning window), the time during which we had determined spawning to occur, i.e., 5 pm until shortly after dark. To plot and measure the AoAs, a line was drawn to encompass the outer edges of all locations indicated on the map for each focal individual and the areas calculated manually on fine graph paper. Open habitat such as sand patches, regularly avoided by fish moving across the substrate (presumably due to lack of shelter from predation), were excluded from calculations of AoAs. Only focal animals for which 20 or more locations were marked over multiple days were used to generate AoAs. Fish were followed to sleeping areas whenever possible after spawning had finished. These and spawning sites were mapped. To examine whether or how male and female AoAs related to each other, these were plotted together as different layers in a graphics programme and visually inspected to examine (a) AoAs of males relative to each other, (b) AoAs of females relative to each other, and (c) areas of overlap among spawning partners.
Inter- and intra-sexual behaviors, spawning frequencies, locations, and male mating success
Inter- and intra-sexual behaviors were noted (i.e., chases, displays, spawning, unsuccessful spawning rises, and disrupted spawns). Data were collected for 1–1.5 h prior to sunset until animals retired into the coral for the night. Data were collected on fish identity, if known, or on sex and estimated size of fish, focal site identity, and time(s) of spawning.
Selected fish were followed to collect information on spawning frequency and spawning site use. Selected focal females were followed for at least 7 consecutive days and the presence or absence of spawning noted daily. For focal males, daily spawning frequency was determined only for males followed for at least three nights. Numbers of sites used daily by focal females and males were noted. Spawning frequency was used as a measure of male mating success and to calculate reproductive success (i.e., number of eggs acquired according to size of female partner times mating success) in relation to body size and to the number of spawning sites visited (“Statistical analysis” section).
Adult sex ratios and sizes of spawning partners
To determine adult sex ratios, the numbers of adult males and females at focal sites were recorded daily. Since some individuals move among several sites in a single daily survey period, a fish was considered to be present at a focal spawning site (and hence counted) on a particular day if it stopped at some point to interact with other fish at the site. To establish the sizes of fish participating as spawning partners, focal fish were followed and the identity or size (for non-focal fish) of spawning partner(s) noted.
Large male removal experiment
A brief removal experiment was conducted to investigate the impact of removing selected large dominant males on female mate selection with the aim of determining whether females preferred to spawn with males larger than themselves in the field, as previously demonstrated under laboratory conditions (Rasotto et al. 2010). An area for this brief experiment was selected distant from the main 12 × 12-m2 study area to avoid any possible impact on the latter. The experimental area contained four spawning sites with 10 males (32.5–43 mm TL) and 15 females (25–37.5 mm TL) before removals were conducted. After an initial survey period, three of the largest four males in a study area were removed and maintained in captivity; they were returned to the same site at the end of the experiment. The fourth largest male was left as a control. The identities of partners for all spawns were noted for 18 days prior to removals and 11 days following removals.
Statistical analysis
Standard statistical tests were applied when possible. Descriptive assessments were used as relevant; tests for between-group comparisons (t-test, chi-square test) were two-tailed with unequal variances. The relationships between the mean daily male reproductive success, male body size, and total number of sites visited by each male were analyzed with a Generalized Linear Model (GLM) assuming a quasi-Poisson error distribution (log link) to account for over-dispersion. Male body size and total number of sites were included as continuous predictors. The number of spawns was included as the response count variable, and the total number of nights of observation (log) was added as an offset to model the number of spawns per observation unit (total spawns/total nights). The relationship between the maximum number of spawns per night (response count variable) and male body size and total number of sites visited (continuous predictors) was analyzed with a GLM assuming Poisson error distribution (log link). A Poisson regression was also used to test the relationship between the two predictors, i.e., male body size and number of sites visited.
Average female fecundity in relation to body size, evaluated from previous work on the gamete collection of 67 mandarinfish spawning events (Rasotto et al. 2010), was used to estimate male reproductive success (i.e., daily mating success × total number of eggs acquired from females of known size; unpublished fecundity data are provided in Online Resource 1). The relationships between the mean daily male reproductive success, male body size, and total number of sites visited by each male were analyzed with a GLM assuming a quasi-Poisson error distribution (log link) to account for over-dispersion.
Generalized linear models were applied with the Software: R Studio Version 1.1.463 using the “glm” function of the package “MASS.” The fit of Poisson regressions was assessed with a deviance goodness of fit test and over-dispersion was checked with the “overdisp” function (package “AER”). In the case of over-dispersed data quasi-Poisson models were used to account for over-dispersion.
Results
Temporal patterns: diel activity and timing of spawning
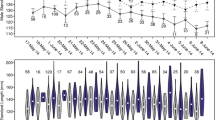
Diel activity: Fish were variously visible during diel surveys, but infrequently seen towards the middle of the day unless there was heavy cloud cover, with more fish typically visible early and late in the day (Fig. 2). Although light levels were not quantified all three observers independently noted more fish at lower light levels. Fish were observed grazing on live coral surfaces, but spent most time hidden from view until 1–1.5 h before sunset when fish were increasingly seen in the vicinity of spawning sites, and inter- and intra-sexual displays became frequent. Displays were sometimes observed at dawn, but spawning was only ever seen at dusk.
Timing and duration of spawning: Spawning was only observed within 20 min of official sunset. Out of 198 timed spawns, all took place between 16 min before and 18 min after sunset (Fig. 3). While our studies only covered the months of July, August, September, December, and January, conversations with local dive guides who take tourists on evening dives to observe this species confirmed that spawning occurs throughout the year and was widely anticipated to be daily. The mean daily duration of the spawning window, taken as the time between the first courtship display and the last recorded spawn time, was 28.18 min (s.d. = 11.64; n = 80). The mean time of the first daily spawn relative to the end of the spawning window on the same day was 4.50 min (s.d. = 5.12; n = 80).
Spatial patterns: spawning and sleeping sites and use of space
Spawning site substrate: The 20 spawning sites analyzed within the 27 × 27-m2 study area for 5 predominant substrates (sand, rubble, Porites cylindrica, P. rus, other) occupied 8.25 m2, representing 1.13% of the area surveyed for substrate type. The most common substrate in the area comprised Porites spp. (42.18%, s.d. = 17, P. cylindrica; 8.78%, s.d. = 6.90, P. rus). These hard corals were also the most common substrate associated with spawning sites (64.95%, s.d. = P. cylindrica; s.d. = 40.80, 21.95%; s.d. = 37.40, P. rus). Comparison of the availability of the different substrates with their occurrence at spawning sites showed that P. cyclindrica (chi-square: p < 0.01; df = 4) predominated at spawning sites. Since P. cylindrica covered more than 300 m2 of the surveyed area, this substrate was probably not a limiting factor for mandarinfish spawning sites (Fig. 4).
Map of 12 × 12-m2 study area (each grid square measures 3 × 3 m.2) showing locations of spawning sites (n = 18, black dots) and regularly used sleeping areas (n = 13, black stars). Observations were made after 5 pm until after dark. Substrate types: scalloped shading is “head” Porites, dark gray is Porites rus, light gray is “Finger” Porites (cylindrica)
Spawning and sleeping site locations: Fourteen spawning sites in the 12 × 12-m2 study area were the basis for detailed daily focal fish and site studies; a spawning site was confirmed for this study if it had hosted at least 3 days of spawning events (Fig. 4). The mean distance between spawning sites was 4.18 m (s.d. = 1.82; n = 14). After spawning, females immediately moved to a regularly used sleeping area nearby, often shared with conspecifics (Fig. 4). Males usually remained visible until after dark, continuing to seek or court females, or interacting with other males, then moved to sleeping areas where they also settled for the night. Typically, sleeping areas were located within sheltered cavities containing horizontal coral plates close to spawning sites (Fig. 1c). Despite repeated observations after dark, there was no evidence of later spawning.
Space and site use by males and females: Focal males and females regularly used one to several spawning sites daily or over multiple days. The mean number of sites used by males (n = 14) over multiple days (x = 2.40; s.d. = 1.62; range 1–5 sites) was weakly but significantly greater than the mean number of sites used by females (n = 9) (x = 1.64; s.d. = 0.55; range 1–3 sites) (t = − 2.18; p = 0.037; df = 31). On a daily basis, males were more mobile among spawning sites than females.
The areas of activity (AoAs) used by 9 focal females and 14 focal males were plotted over multiple nights in December 1998 and January 1999 in the 12 × 12-m2 survey area. AoAs of individual fish ranged from approximately 1 to 8 m2 and encompassed one to several spawning sites and sleeping areas. Males had significantly larger AoAs than females (t = 2.38; p = 0.029). There was a significant and positive relationship between body size and AoA area (r = 0.48; p = 0.021; n = 23). Since females are smaller than males it could not be determined whether AoA area is influenced by sex.
There was considerable overlap in AoAs between males and females and no indication that these areas were defended by either sex; hence, they were not considered to be territories (Fig. 5). It is unknown whether individuals remained within these areas earlier in the day, when they were typically hidden from view, since observations of AoAs were only conducted within 1–1.5 h prior to sunset. Using an interactive, layered, model, multiple combinations of AoAs by focal fish and substrate type were possible by visual inspection. AoAs overlapped either fully or partially between one to several individuals of both sexes, irrespective of spawning partner associations (Fig. 5; additional information are given in Online Resource 1).
Four examples of AoA overlap for combinations of 12 focal fish with their spawning partners. (a) One female (solid line) and her three male partners, (b) one female (solid line) and her two male partners, (c) one male (solid line) and his two partners, and (d) one male (solid line) and his three female partners. Two fish, one male, and one female appear in different combinations (b and d). Each grid square is 3 × 3 m.2 and observations were made after 5 pm. Each AoA encompasses at least 20 point locations per individual
Inter- and intra-sexual interactions
Aggressive interactions: Dominance relationships occur within both sexes according to body size (TL), with aggressive interactions most quickly resolved the larger the size difference; the most intense such interactions lasted up to 8 min, but these were not common. Males displayed to other males by extending dorsal, caudal, anal, and pelvic fins (Fig. 1a, b; video in Online Resource 2) followed by combinations of circling/sidling, lunging, biting, or chasing, depending on the extent of body size difference and response. Submissive behavior was indicated whereby the first dorsal fin was crossed diagonally across the back with the second dorsal fin lowered, body curved, operculae flared, and, if interactions were intense, body color became pale. Paling of the body also occurred when fish settled to sleep (Fig. 1c).
Chasing over short distances (m) was the most common interaction between males and between females. Relatively larger males dominated and typically gained access to nearby females. Small males were able to spawn in the vicinity of larger males only when the latter were otherwise occupied. Larger females often chased away smaller females at spawning sites.
Courtship interactions: Males courted females by extending all fins, generally at a higher intensity on initial female encounter (Online Resource 3). As spawning time approached males would begin to “half-circle” a female in a jerky (stop-start) fashion and become increasingly agitated, sometimes briefly chasing females. Towards the end of the spawning window, females paid more attention to males and were more ready to align to rise to spawn.
Spawning interactions: Male and female pairs rose slowly into a successful spawn shortly before the end of the spawning window although rises were often aborted during earlier spawning attempts (Online Resource 3) possibly due to poor body alignment or to perceived predation threat. Prior to successful spawning, the female adjusted herself to be in close proximity to the expanded male anal fin which extended towards the female forming a partial funnel next to her genital opening (Fig. 1d, e). Once alignment occurred the pair slowly rose (7–12 s) about 1 m up into the water column, released gametes, and quickly returned to the substrate (Fig. 1d, f; Online resource 3). Larger females only spawned with males larger than themselves, despite being intensely courted by males of different sizes, and showed little interest to spawn with males similar in size or smaller than themselves (Rasotto et al. 2010; this study). On just three occasions, of hundreds of spawns observed, spawning rises were disrupted by a nearby male or female closely approaching the rising pair and chasing the male. The original pair would return rapidly to the substrate before realigning to rise and spawn. The disruptor was never seen to release gametes with a rising pair.
Spawning frequencies and male mating success: Mean daily spawning frequencies did not differ significantly between males and females (t = 8.59E − 10; p > 0.05; n = 240 focal fish samples; 98 female days; 142 male days) although variability was greater in males. Male daily spawning frequency (number of daily partners) was highly variable, from 0 to a maximum of 8 spawns per night, but for most (75%) nights males only spawned between 0 and 2 times (x = 1.71; s.d. = 1.61; n = 142). Females spawned either once daily or not at all (x = 0.79; s.d. = 0.408; n = 98).
Females showed no evidence of spawning synchrony across days. However, considering the very short nightly spawning period of less than 10 min, males were constrained to a very short time window for spawning with multiple females. On any particular night whether or not a particular female would spawn could not be predicted by observers unless the female was heavily gravid, in which case she did spawn. Observations on the spawning occurrence of 12 females (25–36 mm TL) for 7–11 consecutive days per female (a total of 102 days) showed females spawned on 77% of the days surveyed. Spawning occurred consecutively for 1 to at least 6 days interspersed by 1 to 3 zero spawn days consecutively and frequency did not appear to be associated with body size (Online Resource 1).
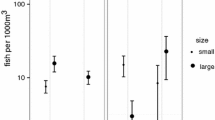
Larger males had a higher mating success than smaller ones in terms of both mean number of spawns per night (estimate = 0.100 ± 0.019 SE; z-value = 5.144; 95% CI = 0.063 to 0.140; p < 0.001) and maximum number of spawns per night (estimate = 0.082 ± 0.027 SE; z-value = 3.087; 95% CI = 0.033 to 0.138; p < 0.01) (Fig. 6). Seven out of 20 focal males regularly visited a single site, 2 males visited 2 sites, and 11 males regularly visited from 3 to 5 sites. The number of sites visited was not influenced by male body size (estimate = − 0.025 ± 0.018 SE; z-value = − 1.384; 95% CI = − 0.055 to 0.006; p = 0.166; n = 20).
Mating success in relation to male body size (TL mm) (n = 20). Maximum daily spawns observed (right axis; open circle) and average daily spawns (total spawns observed/total observation nights) (left axis; closed circle). Data from 3 to 14 days of observations. Days of no spawning are included. Overlapping points are offset from each other to present all data
Male spawning frequency was negatively but weakly influenced by the number of sites visited, suggesting that males staying in one or two sites have a higher or similar mating success compared to those moving among multiple sites (mean nightly spawns: estimate = − 0.170 ± 0.079 SE; z-value = − 2.168; 95% CI = − 0.329 to − 0.020; p = 0.045; maximum nightly spawns: estimate = − 0.246 ± 0.125 SE; z-value = − 1.967; 95% CI = − 0.506 to − 0.014; p = 0.049) (Fig. 7). The 9 focal males regularly staying in one or two sites were noted at the sites with the largest number of females (on average more than 3.2 female/night) or at sites somewhat isolated from the majority of other spawning sites in the survey area.
Male mating success in relation to the number of spawning sites visited (males; n = 20; sites: 1–5) showing maximum daily spawns (right axis; open circle) and average daily spawns (total spawns observed/total observation nights) (left axis; closed circle). Data from 3 to 14 days of observations. Days of no spawning are included. Overlapping points are offset from each other to present all data
When factoring in female fecundity with mating success, larger males obtained a higher reproductive success (as determined by number of eggs × mating success) per night compared to smaller males (estimate = 0.126 ± 0.028 SE; z-value = 4.506; 95% CI = 0.074 to 0.184; p < 0.001; n = 17 males). On the other hand, the mean number of eggs per night was not influenced by the number of sites visited (estimate = − 0.121 ± 0.103 SE; z-value = − 1.179; 95% CI = − 0.335 to 0.072; p = 0.258).
Adult sex ratios and sizes of spawning partners
Sex ratios: The mean numbers of adult males (x = 2.68; s.d. = 1.036) to females (x = 3.32; s.d. = 1.44) at 34 spawning sites noted over 131 nights and in 4 survey periods did not differ significantly (t = 5.31E − 05; p > 0.05; n = 131). Spawning sites contained 1–8 females and 1–5 males (Fig. 8). Females were present at sites even on nights they did not spawn. The mean sex ratio for the 131 nights was 1:1.24 M:F. Given that the likelihood was 77% that a female would spawn on any one night, the overall nightly OSR was equal (1:0.95).
Sizes of spawning partners: The sizes of focal males and females in successful spawning pairings were correlated (r = 0.28; p = 0.01; n = 83 spawns; 13 different males and 19 different females) with females always spawning with males larger than themselves despite the frequent presence of males at spawning sites of equal or smaller size that courted them vigorously on multiple occasions.
Large male removal experiment
A brief experiment designed to test whether females prefer to mate with males larger than themselves in the field, as demonstrated by an earlier laboratory-based experiment (Rasotto et al. 2010), confirmed this preference under natural conditions. Four focal spawning sites were surveyed for 18 days before (25 focal fish; 10 males, 15 females) and 11 days after (the remaining 22 focal fish; 7 males, 15 females) removals of three of the four largest males in the area; the fourth male was left as a control. Before removals the four largest males gained 98 percent of all spawnings. After removals the three medium-sized males that had not spawned pre-removal, despite vigorously courting females, gained 61% of spawnings and the large control male 18%; hence the largest 4 males post-removal gained 79% of spawnings (Fig. 9).
Mean (and standard deviation) number of daily spawns (y-axis) by 10 different males (x-axis shown by TL mm) before (black; 18 days, 44 spawns) and after (grey; 10 days, 33 spawns) removals of 3 large (41, 42.5, 43 mm TL) at 4 adjacent spawning sites. One large male (42.5 mm TL) was left as a control. Total 15 females: 25–37.5 mm TL. Note that 5 of the smallest males (33–38 cm TL) did not spawn at all prior to removals
Post-removal male-male interactions among three intermediate-sized fish that had not spawned before removals (35, 36, 38 mm TL) intensified markedly according to qualitative observation; interactions were more frequent and lasted much longer among these fish than pre-removal, and they more frequently moved among the 4 spawning sites. Females were clearly hesitant to spawn with intermediate-sized males, as determined by the often-slow rises of paired fish, and frequent aborting of rises by females. The largest two females stopped spawning after removals, and no female spawned with a male of similar size to itself or smaller.
Discussion
The mandarinfish exhibits a polygamous mating system. Males are polygynous, either spawning daily with one or more different partners or they may not spawn at all. In contrast, females are sequentially polyandrous, typically spawning once daily with one male, and occasionally not spawning at all. Often a female will spawn with different males on successive days. Spawning occurs in pairs, around sunset, at consistently used sites where small clusters of fish gather for the brief daily courtship and spawning period (spawning window). Male and female densities vary both among sites and within the same site on different days. Promiscuous mating, such as group-spawning or intrusion on the spawning pair by other individuals releasing gametes, was never observed. Hence the possibility of sperm competition and/or of cryptic female choice is excluded.
Overall, mating decisions by both sexes appear to be shaped by temporal and spatial factors that constrain the potential for resource monopolization by males, and for mating opportunities by both sexes. Female variability in their spatial distribution, their short and predictable daily spawning period, and the asynchrony of their ripeness appear to influence male behavior. Indeed, males may vary in their behavior either demonstrating a form of defense polygyny, whereby they consistently visit and attempt to dominate the same spawning site over successive days, or engaging in scramble competition, whereby they move among spawning sites within or between days. Both mating tactics allow the larger males, those preferred by females (which prefer males larger than themselves), to gain high mating success. Females, too, are flexible in their daily choice of spawning site(s), but most do not move among sites during the same daily spawning window. The lack of potential for mate monopolization is further reflected in the sexual pattern of this species which was determined to be gonochoristic based on histological examination of 58 gonads (Rasotto and Sadovy de Mitcheson, unpublished data).
In the mandarinfish, courtship and spawning occurred during a narrow timeframe each day and was generally characteristic of dusk-spawning pelagic-egg producers. Mean duration of the daily spawning window was 28 min, with egg and sperm release confined to a period of less than 10 min. The short window was consistent with other dusk-spawning pelagic-egg producing fishes (Thresher 1984) with egg release evidently influenced by light intensity (Myrberg et al. 1989). Timing might be linked to the risk of predation generally being lower at dusk than during daytime hours (Sancho et al. 2000b). Spawning typically involved a slow rise of 7–12 s of a paired male and female, a similar duration to other pair-spawning pelagic-egg releasing reef fishes (Habrun and Sancho 2011) . Work on other dragonets similarly reports short spawning periods in the latter daylight hours with spawning being seasonal at higher latitudes (Takita and Okamoto 1979; Takita et al. 1983; Fricke and Zaiser 1982; Zhu et al. 1991; Gonzales et al. 1996; Harrington 1996, 1997).
Spawning occurred at spawning sites where small clusters of fish gathered only during the daily spawning window. At other times of the day, fish dispersed over the reef feeding and were mostly hidden from view. The spawning sites and areas of activity (AoAs) of both sexes were largely confined to Porites cylindrica, despite other coral species being present. The P. cylindrica habitat, however, was not limiting in our study area so there was plenty of potentially suitable substrate available that was not used for spawning. It was not clear what determined the location of individual spawning sites. These locations were consistent within survey periods of multiple weeks each, but showed some variation among different survey periods. Hence, sites may not persist over extensive time periods (multiple months to years); our longest survey period had consistent site use over 42 consecutive days. Male and female AoAs overlapped with several other individuals of the same and opposite sex, and with both spawning partners and non-partners. The number of spawning sites visited by individual males was influenced by the varying number of females present at the sites, and in response to the degree of spatial separation of spawning sites and the relative sizes of other males present.
AoAs were not defended and hence were not considered territories. Fish clearly avoided moving across open areas of sand between different parts of their AoAs, likely to minimize risk from predation. Despite the protective mucus of the species, predation evidently remains a risk for the species (Sadovy et al. 2005). Three predation attempts were noted in over 400 h of observations, two unsuccessful ones by lizardfish and a successful grab of a spawning pair by a trumpetfish. This latter occurred in the post-removal period of the removal experiment when females appeared generally more reluctant to spawn. AoAs ranged from 1 to 8 m2 with larger individuals using larger areas; distances between spawning sites within the survey area ranged from 1 to 9 m with a mean separation of 4.18 m.
Movements among spawning sites differed between males and females. Small groups of females clustered at focal spawning sites with most remaining for the daily spawning window and just a few moving among adjacent sites. In the case of males, medium and larger individuals either remained at a single site or moved among several sites seeking mating opportunities; few males visited more than 4 sites in a single night. Data indicated males that visited fewer sites on average had a higher or similar mating success than those that moved among multiple sites. This suggests that lower mobility could be advantageous, maybe allowing males to focus time and effort on procuring all or most spawnings at a few select sites during the short spawning window. Limiting movements might also minimize the risk of predation. Data indicated that male movement was reduced when distances between mating sites were particularly great; hence males were more likely to remain at a site distant from others.
Daily spawning frequency and inter-daily variability in spawning success were both positively correlated with male size, while individual females spawned either once or not at all each day. Larger males often spawned multiple times each day and smaller males typically spawned either once or not at all. Relatively smaller males usually did not procure a mating at a site where one or several large males were present unless the larger fish was distracted, for example, by male-male fighting, courtship, or spawning. In females the chance of an individual spawning on a given day was 77% and this was not according to any discernible temporal pattern. Males did not appear to know whether a particular female would spawn on a given day since they courted all females encountered. Females were not synchronized among themselves in their spawning patterns across days, except for the fact that all spawning occurred within a very limited, mean of 4.5 min, timeframe each day.
Overall the behavioral ecology of mandarinfish reproduction confirms that male density and female dispersion and receptive timing are crucial determinants of male polygyny (Andersson 1994). Theoretically, the temporally limited female receptivity, the unpredictability of their readiness to spawn, and their distribution among spawning sites are expected to favor male polygyny by scramble behavior (Thornhill and Alcock 1983; Dobson 1984; Schwagmeyer 1988; Andersson 1994; Evans 2019), with males roaming in competitive searching to maximize encounters with potential mates. In the mandarinfish, however, the variability in female density among spawning sites and the spatial distribution of those sites might give insights into the equally successful mating decision of mandarinfish males to remain at just one site, rather than roaming among sites. For example, a high female density at a spawning site could increase the opportunity for males to take advantage of distractions faced by other, possibly larger, males at the same site. Similarly, remote spawning sites increase the cost of mate searching and might, therefore, result in a lower local male density and reduced male-male competition, thereby favoring a switch from a scramble tactic to a resident/territorial one (Dobson 1984; Sandell and Liberg 1992).
In the mandarinfish, females too appear to exhibit flexible mating decisions with respect to the number and frequency of spawning sites visited, with some of them spawning at different sites with different larger (than themselves) males on different nights. This is an active sequential polyandry, with females choosing their partners and, hence, having some control over their offspring paternity (Immler and Taborsky 2009). A similar sequential polyandry occurs in several species of demersal spawners in which females either sequentially mate with different males, or parcel out egg clutches among different males, thereby increasing offspring variability (Giacomello et al. 2008; Li et al. 2015; Lobel et al. 2019).
In pelagic spawners with pair-spawning, information on female mate choice is sparse, while polyandry is well-documented in species with group spawning or parasitic spawning (e.g., Shapiro et al. 1993; Colin 1992; Marconato and Shapiro 1996; Petersen and Warner 1998). However, in the latter cases the polyandry could be considered passive, since females seem to have a limited influence on who is fathering their offspring (Taborsky 1994). It is unclear whether mandarinfish polyandry is indicative of a real difference between pelagic compared to demersal spawners or, alternatively, reflects the difficulty of studying the behavioral ecology of such species at the individual level. Further field studies, specifically focused on female behavior, would shed further light on the details of this mating system.
A question remains regarding the possible role of the anal fin as the basis for mandarinfish female preference for males larger than themselves, as has been suggested in several other dragonets (Holt 1898; Gonzales et al 1996) and in at least one other taxon (Koseki et al. 2001). In both the field (this study) and in laboratory experiments, female mandarinfish spawned exclusively with males larger than themselves and were very reluctant to engage with males of similar size or those smaller than themselves. Rasotto et al. (2010) concluded from the laboratory study (a) that the mean number of gametes produced positively correlates with body size in both sexes and (b) that fertilization success is significantly related to the mean sperm number released but not directly to male body size.
Relative or absolute male size, or a correlate of male size, might be important for alignment of paired fish during the spawning rise, possibly limiting the rapid dilution of gametes. Indeed, larger males have larger anal fins and, in the mandarinfish, as in several dragonet species, the male anal fin appears to closely frame the female genital opening during spawning rises funneling the release of eggs and sperm (Fig. 1d, e). While small males may successfully fertilize the egg clutch of a small/similar-sized female, despite absolutely smaller anal fins than larger males and producing fewer sperm, they might not be able to do the same with females larger than themselves (Rasotto et al. 2010).
A close and efficient alignment of the spawning pair, in addition to favoring gamete contact, might limit the risk of predation during the spawning event (Lima and Dill 1990; Sih 2005). In the medaka (family Adrianichthyidae), the anal fin is larger in males than in females and experimental work involving the alteration of anal fin sizes in the medaka demonstrated that the size of anal fin in males is important for efficient fertilization, which the authors suggest could account for selection for male anal fin size (Koseki et al. 2001). Moreover, the role played by the anal fin in mandarinfish mating dynamics might account for the lack of sneaking interference during spawning.
This study expands on prior knowledge of the mating systems of dragonets and advances our understanding of the mating system of the mandarinfish as an example of a small pelagic-spawner. The data enable a good understanding of the temporal and spatial aspects of their natural environment that shape the behaviors and interactions of males and females during the daily spawning window, and allow for the development of novel hypotheses for future research. This species is excellently suited to in-depth field study of the mating system of a small pelagic-spawning fish, and of particular interest because the pelagic-spawning mode, in general, is difficult to study at the individual level under natural conditions.
Data availability
The datasets analyzed during the current study are available in the Figshare repository, https://doi.org/10.6084/m9.figshare.19780153.v1.
References
Akazaki M (1957) Biological studies on a dragonet, Synchiropus altivelis (Temminck et Schlegel). Jpn J Ichthyol 5:146–152 (In Japanese)
Amundsen T (2003) Fishes as models of sexual selection and parental care. J Fish Biol 63:17–52. https://doi.org/10.1111/j.1095-8649.2003.00219.x
Andersson M (1994) Sexual selection. Princeton University Press
Awata S, Kimura MR, Sato N, Sakai K, Abe T, Munehara H (2010) Breeding season, spawning time, and description of spawning behaviour in the Japanese ornate dragonet, Callionymus ornatipinnis: a preliminary field study at the northern limit of its range. Ichthyol Res 57:16–23. https://doi.org/10.1007/s10228-009-0116-z
Birkhead TR, Møller AP (1998) Sperm competition and sexual selection. Academic Press, San Diego
Birkhead TR, Pizzari T (2002) Postcopulatory sexual selection. Nat Rev Genet 3:262–273. https://doi.org/10.1038/nrg774
Carranza J (2000) Environmental effects on the evolution of mating systems in endotherms. In: Apollonio M, Festa-Bianchet M, Mainardi D (eds) Vertebrate Mating Systems. World Scientific, pp 106–139
Colin PL (1992) Reproduction of the Nassau grouper, Epinephelus striatus (Pisces: Serranidae), and its relationship to environmental conditions. Environ Biol Fish 34:357–377. https://doi.org/10.1007/BF00004740
Darwin C (1871) The descent of man, and selection in relation to sex. Murray, London
De Girolamo M, Scaggiante M, Rasotto MB (1999) Social organization and sexual pattern in the Mediterranean parrotfish Sparisoma cretense (Teleostei: Scaridae). Mar Biol 135:353–360. https://doi.org/10.1007/s002270050634
Debelius H, Baensch HA (1994) Marine Atlas. Mergus, USA
Dobson FS (1984) Environmental influences on sciurid mating systems. In: Murie JO, Michener GR (eds) The biology of ground-dwelling squirrels. Lincoln, University of Nebraska Press, pp 229–249
Domeier ML, Colin PL (1997) Tropical reef fish spawning aggregations: defined and reviewed. Bull Mar Sci 60:698–726
Donaldson TJ (2015) Variation in the mating systems of wrasses (Labridae) at a spawning aggregation site on Guam. Mariana Islands Front Mar Sci Conference Abstract: XV European Congress of Ichthyology. https://doi.org/10.3389/conf.fmars.2015.03.00054
Emlen ST, Oring LW (1977) Ecology, sexual selection, and the evolution of mating systems. Science 197:215–223. https://doi.org/10.1126/science.327542
Evans KO (2019) Scramble defense polygyny. In: Vonk J, Shackelford T (eds) Encyclopedia of Animal Cognition and Behavior. Springer, Cham. https://doi.org/10.1007/978-3-319-47829-6_1955-1
Fitzpatrick JL (2020) Sperm competition and fertilization mode in fishes. Phil Trans R Soc B 375:20200074. https://doi.org/10.1098/rstb.2020.0074
Fricke R, Zaiser MJ (1982) Redescription of Diplogrammus xenicus (Teleostei: Callionymidae) from Miyake-jima, Japan, with ecological notes. Jpn J Ichthyol 29:253–259
Giacomello E, Neat FC, Rasotto MB (2008) Mechanisms enabling sperm economy in blenniid fishes. Behav Ecol Sociobiol 62:671–680. https://www.jstor.org/stable/i25511735
Gibson RN, Ezzi IA (1979) Aspects of the biology of the spotted dragonet Callionymus maculatus Rafinesque-Schmaltz from the west coast of Scotland. J Fish Biol 15:555–569
Gonzales BJ, Okamura O, Taniguchi N (1996) Spawning behavior of laboratory-reared dragonet, Repomucenus huguenini, and development of its eggs and prolarvae. Suisanzoshoku 44:7–15
Gonzales BJ, Taniguchi N, Okamura O (1997) Spawning cycle of two dragonet species, Calliurichthys japonicus and Repomucenus huguenini, in Tosa Bay, Southern Japan. Fish Sci 63:15–21
Graham RT, Castellanos D (2012) Apex predators target mutton snapper spawning aggregation. Coral Reefs 31:1017. https://doi.org/10.1007/s00338-012-0921-0
Habrun CA, Sancho G (2011) Spawning ascent durations of pelagic spawning reef fishes. Curr Zool 58:95–102. https://doi.org/10.1093/czoolo/58.1.95
Harrington ME (1996) Sexual selection in the spotted dragonet, Diplogrammus pauciradiatus (Pisces: Callionymidae). University of Miami, Florida, USA, p 152
Harrington ME (1997) Behavior patterns and sexual dimorphism in the spotted dragonet, Diplogrammus pauciradiatus (Pisces: Callionymidae). Bull Mar Sci 60:872–893
Herre AWCT (1927) A new genus and three new species of Philippine fishes. Philippine Journal of Science 32(3):413–419, Pls. 1–2
Holt EWL (1898) On the breeding of the dragonet (Callionymus lyra) in the Marine Biological Association’s aquarium at Plymouth; with a preliminary account of the elements, and some remarks on the significance of sexual dimorphism. Proc Zool Soc Lond 66:281–315
Ikejima K, Shimizu M (1998) Annual reproductive cycle and sexual dimorphism in the dragonet, Repomucenus valenciennei, in Tokyo bay, Japan. Ichthyol Res 45:157–164. https://doi.org/10.1007/BF02678558
Immler S, Taborsky M (2009) Sequential polyandry affords post-mating sexual selection in the mouths of cichlid females. Behav Ecol Sociobiol 63:1219–1230. Springer-Verlag. https://doi.org/10.1007/s00265-009-0744-3
Johnson CR (1972) Biology and ecology of Callionymus belcheri (Pisces: Callionymidae). Copeia 1972:461–470
Johnson CR (1973) Biology and ecology of three Australian dragonets (Pisces: Callionymidae). Zool J Linn Soc 52:231–261
Johnson CR (1973) Biology of the dragonet, Callionymus kaianus moretonensis Johnson (Pisces: Callionymidae). Zool J Linn Soc 52:217–230
King PA, Fives JM, McGrath D (1994) Reproduction, growth and feeding of the dragonet, Callionymus lyra (Teleostei: Callionymidae), in Galway Bay, Ireland. J Mar Biol Ass UK 74:513–526
Koseki Y, Takata K, Maekawa K (2001) The role of the anal fin in fertilization success in male medaka. Oryzias latipes Fish Sci 66:633–635. https://doi.org/10.1046/j.1444-2906.2000.00103.x
Kuijper B, Pen I, Weissing FJ (2012) A guide to sexual selection theory. Ann Rev Ecol 43:287–311. https://doi.org/10.1146/annurev-ecolsys-110411-160245
Li N, Takeyama T, Jordan LA, Kohda M (2015) Female control of paternity by spawning site choice in a cooperatively polyandrous cichlid. Behav 152:231–245. https://doi.org/10.1163/1568539X-00003242
Lima SL, Dill LM (1990) Behavioral decisions made under the risk of predation — a review and prospectus. Can J Zool 68:619–640. https://doi.org/10.1139/z90-092
Lobel LK, Drown DM, Barber PH, Lobel PS (2019) A genetic assessment of parentage in the blackspot sergeant damselfish, Abudefduf sordidus (Pisces: Pomacentridae). Fishes 53:15. https://doi.org/10.3390/fishes4040053
Marconato A, Shapiro DY (1996) Sperm allocation, sperm production and fertilization rates in the bucktooth parrotfish. Anim Behav 52:971–980. https://doi.org/10.1006/anbe.1996.0245
Myers RF (1999) Micronesian reef fishes. 3rd edn. Coral Graphics, Guam
Myrberg AA, Montgomery WL, Fishelson L (1989) The reproductive behavior of Acanthurus nigrofuscus (Forskal) and other surgeonfishes (Fam. Acanthuridae) off Eilat, Israel, (Gulf of Aqaba, Red Sea). Ethol 79:31–61. https://doi.org/10.1111/j.1439-0310.1988.tb00698.x
Narita T, Ganmanee M, Sekiguchi H (2006) Population dynamics of a dragonet Repomucenus valenciennei in Ise Bay, central Japan. Nippon Suisan Gakkaishi 72:860–872. https://doi.org/10.2331/suisan.72.860
Oliveira RF, Taborsky M, Brockmann HJ (2008) Alternative reproductive tactics: an integrative approach. Cambridge University Press, UK, pp 251–299
Petersen CW, Warner RR (1998) Sperm competition in fishes. In: Competition S, Selection S (eds) Birkhead TR, Møller AP. Academic Press, London, pp 435–463
Rasotto MB, Sadovy de Mitcheson Y (2010) Male body size predicts sperm number in the mandarinfish. J Zool (Lond) 281:161–167. https://doi.org/10.1111/j.1469-7998.2009.00688.x
Robertson DR, Warner RR (1978) Sexual patterns in the labroid fishes of the western Caribbean: II. The parrotfishes (Scaridae). Smithson Contr Zool 255:1–26. https://doi.org/10.5479/si.00810282.255
Sadovy Y, Mitcheson G, Rasotto MB (2001) Early development of the mandarinfish, Synchiropus splendidus Callionymidae, with notes on its fishery and potential for culture. Aquar Sci Conserv 3:253–263. https://doi.org/10.1023/A:1013168029479
Sadovy Y, Randall JE, Rasotto MB (2005) Skin structure in six dragonet species (Gobiesociformes; Callionymidae) interspecific differences in glandular cell types and mucus secretion. J Fish Biol 66:1411–1418. https://doi.org/10.1111/j.0022-1112.2005.00692.x
Sadovy de Mitcheson Y, Colin PL eds (2012) Reef fish spawning aggregations: biology, research and management, Fish & Fisheries Series 35: pp. 644 Springer Science+Business Media B.V. https://doi.org/10.1007/978-94-007-1980-4_3
Sancho G, Petersen CW, Lobel PS (2000) Predator-prey relations at a spawning aggregation site of coral reef fishes. Mar Ecol Prog Ser 203:275–288. https://doi.org/10.3354/meps206193
Sancho G, Solow AR, Lobel PS (2000) Environmental influences on the diel timing of spawning in coral reef fishes. RIES Mar Ecol Prog Ser 206:193–212. https://doi.org/10.3354/meps206193
Sandell M, Liberg O (1992) Roamers and stayers: a model on male mating tactics and mating systems. Am Nat 139:177–189. https://doi.org/10.1086/285319
Schwagmeyer PL (1988) Scramble-competition polygyny in an asocial mammal: male mobility and mating success. Am Nat 131:885–892. https://www.jstor.org/stable/2461819
Shapiro DY, Sadovy Y, McGehee MA (1993) Size, composition, and spatial structure of the annual spawning aggregation of the red hind, Epinephelus guttatus (Pisces: Serranidae). Copeia 2:399–406. https://doi.org/10.2307/1447138
Shuster SM (2009) Sexual selection and mating systems. PNAS 106(Supplement 1):10009–10016. https://doi.org/10.1073/pnas.0901132106
Shuster SM, Wade MJ (2003) Mating system and strategies Princeton University Press. Princeton, New Jersey, pp 553
Sih A (2005) Predation risk and the evolutionary ecology of reproductive behaviour. J Fish Biol 45:111–130. https://doi.org/10.1111/j.1095-8649.1994.tb01087.x
Taborsky M (1994) Sneakers, satellites and helpers: parasitic and cooperative behaviour in fish reproduction. Adv Stud Behav 23:1–100
Taborsky M (2008) Alternative reproductive tactics in fish. In: Oliveira R, Taborsky M, Brockmann HJ (eds) Alternative reproductive tactics: an integrative approach. Cambridge University Press, Cambridge, UK, pp 251–299
Taborsky M, Brockmann HJ (2010) Alternative reproductive tactics and life history phenotypes. In: Kappeler P (ed) Animal behaviour: evolution and mechanisms, pp 1–707
Taborsky M, Oliveira RF, Brockmann H. J. (2008) The evolution of alternative reproductive tactics: concepts and questions pp. 21. In: Oliveira RF, Taborsky M, Brockmann (eds) Alternative Reproductive Tactics. Cambridge University Press
Takita T, Okamoto E (1979) Spawning behavior of the two dragonets, Callionymus flagris and C. richardsoni, in the aquarium. Jpn J Ichthyol 26:282–288
Takita T, Iwamoto T, Kai S, Sogabe I (1983) Maturation and spawning of the dragonet, Callionymus enneactis, in an aquarium. Jpn J Ichthyol 30:221–226
Thornhill R, Alcock J (1983) The evolution of insect mating systems. Harvard University Press, Cambridge, Mass
Thresher RE (1984) Reproduction in reef fishes. Inc., Ltd., Neptune City, New Jersey, TFH Publications, p 399
van Rooij JM, Kroon FJ, Videler JJ (1996) The social and mating system of the herbivorous reef fish Sparisoma viride: one-male versus multi-male groups. Envir Biol Fish 47:353–378
Warner RR, Robertson DR (1978) Sexual patterns in the labroid fishes of the western Caribbean, I: The wrasses (Labridae). Smithson Contrib Zool 254:1–27. https://doi.org/10.5479/si.00810282.254
Wernerus F, Tessari V (2008) The influence of population density on the mating system of Thalassoma pavo, a protogynous Mediterranean labrid fish. Mar Ecol 12:361–368. https://doi.org/10.1111/j.1439-0485.1991.tb00264.x
Wickler W, Seibt U (1983) Monogamy: an ambiguous concept. In Mate Choice (ed. P. P. G. Bateson). Cambridge University Press, Cambridge, pp. 33–50
Wilson DP (1978) Territorial behaviour of male dragonets (Callionymus lyra). J Mar Biol Assoc United Kingdom 58:731–734
Wittenrich M, Ho A (2013) Social structure and reproductive behavior of the callionymid fish Callionymus bairdi (Gobiesociformes: Callionymidae): with notes on male alternative reproductive tactics. Aqua, Int J Ichthyol 19:17–28
Zhu Y, Furukawa K, Aida K, Hanyu I (1989) Annual reproductive rhythm of the Tobinumeri-dragonet Repomucenus beniteguri (Callionymidae) in Lake Hamana. Nippon Suisan Gakkaishi 55:591–599
Zhu Y, Furukawa K, Aida K, Hanyu I (1991) Daily spawning rhythm during spring and autumn spawning seasons in the Tobinumeri dragonet Repomucenus beniteguri. Nippon Suisan Gakkaishi 57:1865–1870
Acknowledgements
We are very grateful for the invaluable help of Lisa Locatello, Zoological Station “Anton Dohrn” of Naples, with statistical analyses and of Stefano Gueraldi and Giuliano Bocchi, University of Padova, for the artwork of mandarinfish movement data. We thank the staff of the Coral Reef Research Foundation in Palau and particularly Lori Bell-Colin and Patrick Colin for their generous support and guidance. Rachel Wong and Loby Hau assisted with graphics. Carlotta Mazzoldi assisted with field work. We thank the Palau National Government and Koror State Government for permission to do the research.
Funding
Research was funded by the National Geographic Society grant number 6295–98; by the Division of Ecology & Biodiversity, University of Hong Kong (YSM); and the Department of Biology, University of Padova, Italy (MBR).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics approval
Not applicable for that section.
Consent to participate
All authors have agreed to participate.
Consent for publication
All authors have read and agreed to the published version of the manuscript.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Supplementary file2 (MP4 24963 KB)
Supplementary file3 (MP4 369800 KB)
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Sadovy de Mitcheson, Y.J., Mitcheson, G.R. & Rasotto, M.B. Mating system in a small pelagic spawner: field case study of the mandarinfish, Synchiropus splendidus. Environ Biol Fish 105, 699–716 (2022). https://doi.org/10.1007/s10641-022-01281-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10641-022-01281-1