Abstract
The traditional view of stream systems, exemplified by the river continuum concept, shows predictable shifts in species composition and ecosystem function based on changes in nutrients and energy. Usually headwaters have fewer species, lower nutrients and greater habitat variability than downstream sites. The modern view of stream systems is much more complex with disturbances altering expected environmental conditions and assemblages. Reset mechanisms function to change the expected downstream biotic assemblages so that they resemble what would be expected at an upstream location. The presence of a reset mechanism may be indicated by a variety of factors including decreases in biomass and diversity. To assess the potential impacts of a cave, which can impact a variety of hydrologic factors including temperature, fish assemblages and environmental conditions were sampled around a cave. Differences were seen both in assemblage and environmental conditions between sites located upstream and downstream of a cave; variation in assemblage and environment also occurred at a 10 m scale. Overall, species diversity and richness were lower downstream of the cave, resembling a headwater-like site. There was a shift from warmer water species upstream to an assemblage defined by high abundances of mottled sculpin and the presence of brown trout. These differences in assemblage were linked with environmental conditions, including temperature, that were likely influenced by the presence of the cave. Results of this study indicate that, in this case, a cave has impacted fish assemblages and the surrounding stream ecosystem and may be functioning as a reset mechanism, resulting in decreased species diversity and changes in assemblage composition over a short distance.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Variation in the environment is an important predictor in determining fish assemblages at a given location (e.g., Vannote et al. 1980; Hill and Grossman 1993; Petty and Grossman 2007; Rowe et al. 2009). Spatial variation in streams is usually examined longitudinally (e.g., Adams et al. 2004; Grubbs et al. 2007; Li et al. 2012) because of stream gradation (Kayde et al. 2008). Changes are evident both environmentally and biologically from a stream’s headwaters to its mouth as described in the river continuum concept (RCC; Vannote et al. 1980). For example, headwater sections of temperate streams tend to have lower temperatures due to more riparian vegetation and a greater groundwater component (Vannote et al. 1980; Mellina et al. 2002; Moore et al. 2005). The longitudinal gradation is altered when disturbance events result in patchy or discontinuous habitats (Poole 2002; Stanley et al. 2010; Winemiller et al. 2010) where disturbances are defined as in Resh et al. (1988) as discrete events that are outside the expected range in severity, frequency, or intensity that may alter the ecosystem. In addition to longitudinal alterations, patchy habitats can also contribute to finer spatial scale heterogeneity in environment and fish assemblages (Adams et al. 2004; Kayde et al. 2008; Winemiller et al. 2010). For example, daily fluctuations in dissolved oxygen or temperature can limit habitation by a species. Habitat diversity is considered responsible for much of the diversity seen in stream assemblages (Hynes 1970; Bain et al. 1988; Scheidegger and Bain 1995; Barbosa et al. 2003), and habitat diversity, in turn, is influenced by disturbance.
Physical disturbance is a commonly studied mechanism influencing local environmental conditions (Reice 1985; Robinson and Minshall 1986). Some disturbance events may disrupt the traditional pattern of longitudinal gradation and return environmental variables at a more downstream site to headwater-like conditions (Vannote et al. 1980; Poff and Allan 1995). For example, a strong spate or shifting of sediments has been found to alter the macroinvertebrate community and ‘reset’ the community to one more expected in headwaters (Reice 1985). However, not all reset mechanisms are disturbances. A cave, for example, may also function to alter downstream water temperature (Dare et al. 2001; Bichuette and Trajano 2003; Ferreira and Martins 2009), dissolved oxygen concentrations (Tobler et al. 2006), food web complexity (Fong and Culver 1994), and carbon availability (Poulson and Lavoie 2000; Simon et al. 2003). Most research concerning the impacts of caves on fish ecology has focused on stygobitic (cave-adapted) fishes, but there is a deficit in the literature regarding the influences of caves on epigean (surface) species (Poly 2001) and whether caves influence external stream ecosystems.
Caves, sinkholes, and disappearing streams are associated with karst landscapes. Karst is characterized by bedrock, usually near the earth’s surface, which has been dissolved by water to create formations such as sinkholes. Karst landscapes occur worldwide, but their associated hydrologic complexity, including the impacts of caves, requires additional study to enhance our understanding. While studies have examined the importance of caves and karst on cave-adapted fishes (e.g., Day et al. 2014), there is a gap in the research regarding stream and assemblage characteristics upstream and downstream of a cave. The goal of this study was to compare fish assemblages and environmental variables upstream and downstream of a cave as a means of assessing the impacts of the cave as a potential reset mechanism. To examine variation at a finer spatial scale, the upstream and downstream sites were divided into 10 m cells. Differences in fish assemblages were related to environmental variables, which could indicate whether the cave has influenced the downstream portion of the stream.
Methods
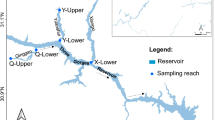
The south branch of the Root River runs through primarily agricultural land in the driftless zone of southeastern Minnesota, USA (Delong 2005). Groundwater creates complex stream systems (Delong 2005), and the area’s karst topography is characterized by numerous sinkholes, caves, and sinking or “disappearing” streams. Portions of the Root River sink through cracks in its limestone bed and flow through a cave, while the rest of the river continues to flow on the surface except at low flow. Water flows through the cave covering at least 2.4 km in 6–10 h, although the exact distance of water flow through the cave is unknown.
In summer 2009, there was no surface flow on the Root River to connect sections of the river upstream and downstream of the cave. Sites upstream (U) and downstream (D) of the cave were divided into five consecutive 10 m cells (e.g., Aparicio and De Sostoa 1999); the cells were numbered 1 through 5, with cell 1 being the most upstream (e.g., D1 = most upstream cell at the downstream site). Two additional cells (D6 and D7) were added after the first sampling event due to low water levels in cells D1 and D2. The upstream site was within one km of the beginning of the cave. The downstream site was approximately 8 km from the upstream site following the river channel and 2 km below where water exits the cave. Entrances and exits to the cave consisted of small cracks, making fish passage through the cave unlikely.
Fish were sampled in each 10 m cell with a backpack electrofishing unit (Smith-Root Model 12, Vancouver, WA, USA) in May–July 2009, which is post-spawn for most stream species. Each cell was sampled four times to assess variability and species diversity. Cells D1 and D2 were only sampled once due to low water conditions, while cells D6 and D7 were sampled three times. Block nets were placed at the upstream and downstream ends of a cell to prevent fish from escaping. Cells were electrofished from downstream to upstream, similar to Aparicio and De Sostoa (1999) and Skaski and Gilliam (2000). Each cell was electrofished using three-pass depletion for approximately 45 min. After three passes were conducted, all fish were identified, weighed (g), measured [total length (TL); mm], and returned to the cell. Block nets were maintained between cells during sampling to prevent fish from being sampled twice.
Abiotic data were collected at each cell on each sampling date. Width (m) was measured as wet width in the center of each cell. Depth (m) and velocity (m · s−1) were the average of three measurements across the center of each cell. Velocity data were collected with a Marsh-McBirney Flo-Mate 2000 (Marsh-McBirney, Inc., Fredrick, MD, USA) at 2/3 of the water depth at right, center and left of the channel. Percent cobble and percent canopy cover were visually estimated in three locations across the middle of each cell by two independent observers; final percent cobble and percent cover values represent an average of the six estimates per cell.
A principle component analysis (PCA) was used to condense the environmental variables into principal component axes (SAS 9.2 2002, SAS Institute Inc., Cary, NC, USA). Examination of eigenvectors and correlations between abiotic variables and the PCs determined which variables were most influential for each principle component (PC). Fish species richness, diversity, biomass, and density were calculated for each cell on each sampling date. Shannon-Weaver Diversity Index (Peet 1974) was calculated as: H’ = −∑ pi log pi, where p i is the number of individuals of one species divided by the total number of individuals (N). Mean richness, diversity, biomass and all environmental variables were compared between upstream and downstream but were not compared statistically due to lack of independence. Non-metric multidimensional scaling (NMDS) was performed on the fish assemblage data (proportion of catch) to identify and visualize assemblage similarities among cells. The NMDS was performed with the Sorenson (Bray-Curtis) distance measure; 400 maximum iterations were performed over 40 runs using the fish assemblage data and 50 runs with the Monte Carlo permutation procedure in PC-ORD (v. 4.0, MJM Software Design, Gleneden Beach, OR, USA). Upstream to downstream differences in fish assemblages were statistically compared using multi-response permutation procedure (MRPP). Principle components were correlated with the NMDS dimensions to evaluate relationships between assemblages and environmental variables (e.g., Uzarski et al. 2005).
Results
Environmental characteristics differed between the upstream and downstream sites. Over 90 % of the variation among cells was explained by three PCs. Principle component 1, which explained 44 % of the variation, was correlated with velocity, wet width, and percent cobble (Fig. 1; Table 1). Principal component 2 explained 32 % of the variation and was correlated with depth, velocity and percent cover. Principal component 3 explained 16 % of the variation and was correlated with water temperature. Overall, upstream cells were deeper and narrower with lower percent canopy cover than downstream cells. Upstream cells were closer together in ordination space than downstream cells, indicating the environmental variables and habitat were more consistent upstream of the cave (Fig. 1). Principal component 3 demonstrated that temperature varied among the 10 m cells. Cells D1 and U5 appeared different in environmental characteristics from other downstream or upstream cells, respectively.
Principle components (PCs) from principle components analysis (PCA). Percentages represent the variability explained by each PC. 10 m cell names are either upstream (U) or downstream (D) followed by the cell number. Cells are numbered in ascending order with cell 1 being the most downstream. Abiotic data include velocity (vel), percent cover (cov), depth (dep), width (wid), percent cobble (cobb), and temperature (temp)
Species richness was found to be greater upstream (18 species) than downstream (15 species) and species diversity was also greater (upstream =1.10, downstream =0.61). Average biomass was 291.2 g∙m−1 upstream and 247.9 g∙m−1 downstream. The upstream cells tended to be warmer (Table 2), deeper and have more canopy cover than downstream cells. Downstream biomass was influenced by high numbers of mottled sculpin (Cottus bairdii; Girard 1850) and four large brown trout (Salmo trutta; Linnaeus 1758) that were the largest fish captured during this study. Bigmouth shiner (Notropis dorsalis; Agassiz 1854), common shiner (N. cornutus; Mitchill 1817), johnny darter (Etheostoma nigrum; Rafinesque 1820), and sunfish spp. (Lepomis spp.) appeared in greater numbers at the upstream sites (Fig. 2).
Mean densities (individuals∙m−2, standard error bars) of all fish species captured at the site upstream (open bars; U) and downstream (black bars; D) during the study. Codes for species are as follows: ABL-American brook lamprey (Lampetra appendix; Dekay 1842), BMS-bigmouth shiner, BND-blacknose dace (Rhinichthys atratulus; Hermann 1804), BNM-bluntnose minnow (Pimephales notatus; Rafinesque 1820), BSB-brook stickleback (Culaea inconstans; Kirtland 1840), BT-brown trout, CSR-central stoneroller (Campostoma anomalum; Rafinesque 1820), CS-common shiner, CC-creek chub (Semotilus atromaculatus; Rafinesque 1820), FD-fantail darter (Etheostoma flabellare; Rafinesque 1819), HH- hornyhead chub (Nocomis biguttatus; Kirtland 1840), JD- johnny darter, LND-longnose dace (R. cataractae; Valenciennes 1842), MS-mottled sculpin, NHS-northern hog sucker (Hypentelium nigricans; Lesueur 1817), RD- rainbow darter (E. caeruleum; Storer 1845), SR-redhorse spp. (Moxostoma spp.), RB-rock bass (Ambloplites rupestris; Rafinesque 1817), SRD-Southern redbelly dace (Phoxinus erythrogaster; Rafinesque, 1820), PS-sunfish spp., and WS-white sucker (Catostomus commersonii; Lacepede 1803)
A two dimensional solution was used for NMDS because further dimensions did not improve stress (e.g., McCune et al. 2002). Dimension 1 explained 38 % of the variation in the fish assemblages while dimension 2 explained 49 % of the variation (Fig. 3). The NMDS grouped the cells by site (i.e., upstream or downstream of the cave), indicating differences in the fish assemblage between the sites (MRPP, P = 0.006). Upstream cells were characterized by northern hogsucker, white sucker and central stoneroller. Rock bass, rainbow darter and southern redbelly dace were also associated with upstream of the cave and were not found downstream of the cave. Downstream cells were characterized by the presence of cool water species, specifically brown trout and a high density of mottled sculpin as well as fantail darter and longnose dace. Overall, results of the NMDS indicated that the assemblages in downstream 10 m cells were more different from each other than upstream cells as indicated by their distribution across dimension 1. The presence of American brook lamprey separated D7 from other downstream cells. Greater prevalence of mottled sculpin separated D4 and D5 from other downstream cells.
Results of NMDS based on the average proportion of catch across sampling periods within a cell. a Groupings of cells based on fish assemblages. Sites upstream (U) and downstream (D) of the cave are enclosed by circles as grouped in the MRPP. b Groupings of species based on their appearances in assemblages. Fish species abbreviations are given in Fig. 2
Correlations among fish NMDS dimensions and the abiotic PCs provide some indication as to the possible environmental causes of differences in assemblages between sites upstream and downstream of the cave. Dimension 1 was correlated with PC3, indicating differences seen in mottled sculpin abundance may be related to temperature (Fig. 4; Table 1). Dimension 2 was correlated with PC2, indicating that assemblage differences between upstream and downstream sites may be related to differences in velocity, width and percent cobble.
Correlations between non-metric multidimensional scaling (NMDS) dimensions generated from fish community data and principle components (PCs) from the environmental data. a Correlation between dimension 1 from NMDS and PC3 (r = −0.79, P = 0.003). b Correlation between dimension 2 from NMDS and PC2 (r = −0.82, P = 0.001). Site 1 is the site upstream of the cave
Discussion
Differences in fish assemblages and environmental characteristics upstream and downstream of the cave demonstrate that caves may function as a reset mechanism. Sites that are located downstream should have increased diversity and be more homogeneous in environmental characteristics with increased flow, primary production, and autochthonous nutrient inputs (RCC; Vannote et al. 1980). In this study, the site downstream of the cave was cooler and greater variability in both environmental conditions and fish assemblage. Moreover, downstream sites within rivers will typically have higher numbers of species (Matthews 1998; Kayde et al. 2008), but the upstream site in this study a higher value. Fine-scale habitat variation is often overlooked, but the variability among the downstream cells highlights the influence small environmental differences can have on an assemblage. Contrary to what was found in this study, habitat may become more stable within a given area downstream (Ostrand and Wilde 2001; Mellina et al. 2002; Moore et al. 2005).
Disturbance contributes to variation in environmental conditions (Montgomery 1999; Poole 2002; Winemiller et al. 2010) just as reset mechanisms also impact environmental conditions. Variation was observed at a fine spatial scale in both fish assemblage and environmental variables upstream and downstream of the cave. However, the downstream of the cave exhibited greater variability among 10 m cells than at the upstream site. While information is limited regarding the impacts of caves on streams, disturbances as reset mechanisms are better understood. Disturbances, such as floods, are considered potential reset mechanisms (Cummins 1977; Reice 1985; Townsend 1989). Floods cause resets through removal of diversity as water scours away habitat, nutrients, and organisms leading to uniformity (Cummins 1977). Many forms of disturbance occur quickly and last for only a short time (Townsend 1989), resulting in relatively rapid shifts in assemblages and environmental conditions. However, storm-related floods occurring frequently cause a continual reset of fish assemblages when modeled over more than a decade (Strange et al. 1992), indicating that reset mechanisms can function at longer temporal scales. Such frequent disturbances can also function as reset mechanisms and have been shown to decrease species diversity (Robinson and Minshall 1986). Caves impact streams over geologic time and their long-term presence on the landscape could result in a permanent reset in environmental characteristics and fish assemblage.
Caves are known to influence multiple environmental variables, including temperature, within the cave itself and downstream (Tuttle and Stevenson 1978). Other streams and springs in southeastern Minnesota have exhibited temperature impacts from caves and karst (Luhmann et al. 2011). Another cave stream near the current study site demonstrated extremely stable temperatures but with some daily fluctuations caused largely by precipitation (Luhmann et al. 2011). The site downstream of the cave appeared impacted by the presence of the cave even though it was approximately 2 km from where water exited the cave. The downstream impacts of caves are not as well known, but the influence of groundwater input, which can exert the same temperature buffering influence as a cave, is better understood (Brunke and Gonser 1997; Mellina et al. 2002; O’Driscoll and DeWalle 2006). Temperature in a headwater stream can be controlled and moderated by groundwater input resulting in cooler temperatures during summer and warmer temperatures throughout winter (Mellina et al. 2002; Moore et al. 2005). Some of the downstream impacts observed in this study could have been caused by an alteration in the thermal regime or thermal buffering caused by the cave. Indeed, the downstream site water temperatures were significantly lower throughout the summer. A stream’s thermal regime is known to impact fish assemblage (Lyons 1996) as each species distributes based on individual behavior and temperature tolerance (Nielsen et al. 1994; Wootton 1998; Ebersole et al. 2001) and cave temperature is known to influence aquatic macroinvertebrate communities (Reid et al. 2012). Correlations between NMDS and PCA indicated that temperature may drive assemblage differences including the exclusive presences of American brook lamprey downstream of the cave. The site downstream of the cave was characterized by the presence of cooler water species such as brown trout and mottled sculpin. The presence of brown trout and mottled sculpin downstream of the cave and their absence upstream demonstrates that the cave may influence fish assemblage through its impact on water temperature similar to the effects of groundwater inputs.
Cave ecosystems and communities downstream of the cave may also be impacted by energy and carbon availability. Many caves are nutrient polluted because nitrogen and phosphorus are not filtered before entering the cave and are also not utilized within the cave itself because there is no photosynthesis occuring (Boyer and Pasquarell 1996; Simon and Buikema 1997; Simon and Benfield 2002). Cave ecosystems have been found to be limited in organic carbon (Simon and Benfield 2002) and energy (Huntsman et al. 2011). Therefore, organic matter and carbon entering the cave is utilized by cave organisms (Simon and Benfield 2002; Huntsman et al. 2011) and so organic material maybe decreased downstream. For example, organic carbon turnover rate in bethic cave communities was found to be extremely rapid (Simon and Benfield 2002). This usage of organic carbon by the cave ecosystem may increase the importance of allocthonous carbon inputs, which, therefore, more similar to a headwater stream.
The temperature and biotic characteristics of cave ecosystems are mediated by flow, which may also impact the stream ecosystem downstream of a cave. In southeastern Minnesota karst streams, the temperature of stream was impacted by the amount of stream flow (Luhmann et al. 2011). This is caused by changes in the paths of water flow, which may decrease the time water spends in the cave allowing for less time for the water to equilibrate to rock temperature (Luhmann et al. 2011). Water sources (i.e., precipitation trickling in from above vs. stream flow) can also impact cave temperature (Covington et al. 2012) resulting in warmer cave streams around major precipitation events (Luhmann et al. 2011). However, some caves may remain at consistent temperatures despite precipitation (Ravbar and Kosutnik 2014). Increased flow events can also allow the movement of organisms or wash organisms through a cave and alter fish assemblages (Romero 2011). For example, higher flows allowed an epigean form of a fish to invade a cave ecosystem and displace the hypogean variety of the same species (Romero et al. 2002). Increased flows can also increase available organism matter by washing debris into the cave. The impacts of flow on this study system may not have been detected in this case because the study encompassed relatively stable summer conditions although several precipitation events did occur.
A cave may function as a reset mechanism, with the stream downstream of a cave being more headwater-like. This study examined just one stream system and so examination of additional sites would be ideal; however, these results indicate that karst impacts on epigean fishes do exist. Cool water fish species were present and temperature was lower, similar to headwater streams where ground water inputs moderate stream temperature. There was also increased fine-scale variability downstream of the cave in both fish assemblage and habitat. This river and cave system provided an opportunity to study a unique stream allowing for direct comparison of fish assemblage and environmental characteristics upstream and downstream of a cave. This study demonstrated that a cave’s influence may be similar to groundwater inputs but may also reset the stream. Karst landscapes are present worldwide, but their ecological effects have yet to be fully realized.
References
Adams SB, Warren ML Jr, Haag WR (2004) Spatial and temporal patterns in fish assemblages of upper coastal plain streams, Mississippi, USA. Hydrobiologia 528:45–61
Aparicio E, De Sostoa A (1999) Pattern of movements of adult Barbus haasi in a small Mediterranean stream. J Fish Biol 55:1086–1095
Bain MB, Finn JT, Booke HE (1988) Streamflow regulation and fish community structure. Ecology 69:382–392
Barbosa RP, de Freitas CE, Santos SM (2003) The fish community of an upland stream in the Central Amazon (Presidente Figueiredo – Amazonas – Brazil). Acta Limnol Bras 15:37–41
Bichuette ME, Trajano E (2003) Epigean and subterranean ichthyofauna from the Sao Domingos karst area, Upper Tocantins River basin, Central Brazil. J Fish Biol 63:1100–1121
Boyer DG, Pasquarell GC (1996) Agricultural land use effects on nitrate concentrations in a mature karst aquifer. Am Water Resour Bull 32:565–573
Brunke M, Gonser T (1997) The ecological significance of exchange process between rivers and groundwater. Freshw Biol 37:1–33
Covington MD, Luhmann AJ, Wicks CM, Saar MO (2012) Process lengths scales and longitudinal damping in karst conduits. J Geophys Res 117 doi: 10.1029/2011JF002212
Cummins KW (1977) From headwater streams to rivers. Am Biol Teach 39:305–312
Dare MR, Hubert WA, Meyer JS (2001) Influence of stream flow on hydrogen sulfide concentrations and distributions of two trout species in a rocky mountains tailwater. N Am J Fish Manag 21:971–975
Day JL, Starkey DE, Adams G, Brummett S, Keeney D (2014) Population genetics of grotto sculpin (Cottus specus), a new cave-adapted fish species. Environ Biol Fish. doi:10.1007/s10641-013-0216-5
Delong MD (2005) Chapter 8: Upper Mississippi River Basin. In: Benke AC, Cushing CE (eds) Rivers of North America. Elsevier Academic Press, Amsterdam, pp 327–374
Ebersole JL, Liss WJ, Frissell CA (2001) Relationship between stream temperature, thermal refugia and rainbow trout Oncorhynchus mykiss abundance in arid-land streams in the northwestern United States. Ecol Freshw Fish 10:1–10
Ferreira RL, Martins RP (2009) Mapping subterranean resources: the cave invertebrates distribution as indicator of food availability. Rev Bras Farmacogn 11:119–127
Fong DW, Culver DC (1994) Fine-scale biogeographic differences in the crustacean fauna of a cave system in West Virginia, USA. Hydrobiologia 287:29–37
Grubbs SA, Meier OW, Meier AJ (2007) Longitudinal patterns of fish assemblage in small unregulated subbasins: evaluation reach- and watershed-scale parameters. Hydrobiologia 592:211–223
Hill J, Grossman GD (1993) An energetic model of microhabitat use for rainbow trout and rosyside dace. Ecology 74:685–698
Huntsman BM, Vernarsky MP, Benstead JP (2011) Relating carrion breakdown rates to ambient resource level and community structure in four cave stream ecosystems. J N Am Benthol Soc 30:882–892
Hynes HBN (1970) The ecology of running water. University of Toronto Press, Toronto
Kayde WT, Moyo NAG, Magadza CHD, Kativu S (2008) Stream fish assemblages in relation to environmental factors on a montane plateau (Nyika Plateau, Malawi). Environ Biol Fish 83:417–428
Li J, Huang L, Zou L, Kano Y, Sato T, Yahara T (2012) Spatial and temporal variation of fish assemblages and their associations to habitat variables in a mountian stream of north Tiaoxi River, China. Environ Biol Fish 93:403–417
Luhmann AJ, Covington MD, Peters AJ, Alexander SC, Anger CT, Green JA, Runkel AC, Alexander EC Jr (2011) Classification of thermal patterns at karst springs and cave streams. Ground Water 49:324–335
Lyons J (1996) Patterns in the species composition of fish assemblages among Wisconsin streams. Environ Biol Fish 45:329–341
Matthews WJ (1998) Patterns in freshwater fish ecology. Chapman and Hall, New York
McCune B, Grace JB, Urba DL (2002) Analysis of ecological communities. MJM Software Design, Glenden Beach
Mellina E, Moore RD, Hinch SG, Macdonald JS, Pearson G (2002) Stream temperature responses to clear-cut logging in British Columbia: the moderating influences of groundwater and headwater lakes. Can J Fish Aquat Sci 59:1886–1900
Montgomery DR (1999) Process domains and the river continuum. J Am Water Resour Assoc 35:397–410
Moore RD, Spittlehouse DL, Story A (2005) Riparian microclimate and stream temperature response to forest harvesting: a review. J Am Water Resour Assoc 41:813–834
Nielsen JL, Lisle TE, Ozaki A (1994) Thermally stratified pools and their use by steelhead in northern California streams. Trans Am Fish Soc 123:613–626
O’Driscoll MA, DeWalle DR (2006) Stream-sir temperature relations to classify stream-ground water interactions in a karst setting, central Pennsylvania, USA. J Hydrol 329:140–153
Ostrand KG, Wilde GR (2001) Temperature, dissolved oxygen, and salinity tolerance of five prairie stream fishes and their role in explaining fish assemblage patterns. Trans Am Fish Soc 130:742–749
Peet RK (1974) The measurement of species diversity. Annu Rev Ecol Syst 5:285–307
Petty JT, Grossman GD (2007) Size-dependent territoriality of mottled sculpin in a southern Appalachian stream. Trans Am Fish Soc 136:1750–1761
Poff NL, Allan JD (1995) Functional organization of stream fish assemblages in relation to hydrological variability. Ecology 76:606–627
Poly WJ (2001) Nontroglobitic fishes in Bruffey-Hills Creek Cave, West Virginia, and other caves worldwide. Environ Biol Fish 62:73–83
Poole GC (2002) Fluvial landscape ecology: addressing uniqueness with the river continuum. Freshw Biol 47:641–660
Poulson TL, Lavoie KH (2000) The trophic basis of subsurface ecosystems. In: Wilkens H, Culver DC, Humphreys WF (eds) Ecosystems of the world 30: subterranean ecosystems. Elsevier, Amsterdam, pp 231–249
Ravbar N, Kosutnik J (2014) Variation of karst underground air temperature induced by various factors (Cave of Zupanova jama, Central Slovenia). Theor Appl Climatol 116:327–341
Reice SR (1985) Experimental disturbance and the maintenance of species diversity in a stream community. Oecologia 76:90–97
Reid DJ, Scarsbrook MR, Wright-Stow AE, van Houte-Howes KSS, Joy K (2012) Water quality and benthic macroinvertebrate communities in karst landscapes of North Island, New Zealand: influences of water sources, habitat type and anthropogenic disturbances. New Zeal J Mar Fresh 46:263–277
Resh VH, Brown AV, Covich AP, Gurtz ME, Li HW, Minshall GW, Reice SR, Sheldon AL, Wallace JB, Wissmar RC (1988) The role of disturbance in stream ecology. J N Am Benthol Soc 7:433–455
Robinson CT, Minshall GW (1986) Effects of disturbance frequency on stream benthic community structure in relation to canopy cover and season. J N Am Benthol Soc 5:237–248
Romero A (2011) The evolution of cave life. Am Sci 99:144–151
Romero A, Singh A, McKie A, Manna M, Baker R, Paulson KM, Creswell JE (2002) Replacement of the troglomorphic population of Rhamdia quelen (Pisces: Pimelodidae) by an epigean population of the same species in the Cumaca Cave, Trinidad, West Indies. Copeia 2002:938–942
Rowe DC, Pierce CL, Wilton TF (2009) Fish assemblage relationships with physical habitat in wadeable Iowa streams. N Am J Fish Manag 29:1314–1332
Scheidegger KJ, Bain MB (1995) Larval fish distribution and microhabitat use in free-flowing and regulated rivers. Copeia 1995:125–135
Simon KS, Benfield EF (2002) Ammonium retention and whole-stream metabolism in cave streams. Hydrobiologia 482:31–39
Simon KS, Buikema AL Jr (1997) Effects of organic pollution on an Appalachian cave: changes in macroinvertebrate populations and food supplies. Am Midl Nat 138:387–401
Simon KS, Benfield EF, Macko SA (2003) Food web structure and the role of epilithic biofilms in cave streams. Ecology 69:279–284
Skaski GT, Gilliam JF (2000) Modeling diffusive spread in a heterogeneous population: a movement study with stream fish. Ecology 81:1685–1700
Stanley EH, Powers SM, Lottig NR (2010) The evolving legacy of disturbance in stream ecology: concepts, contributions, and coming challenges. J N Am Benthol Soc 29:67–83
Strange EM, Moyle PB, Foin TC (1992) Interactions between stochastic and deterministic processes in stream fish community assembly. Environ Biol Fish 36:1–15
Tobler M, Schlupp I, Heubel KU, Riesch R, de Leon FJG, Giere O, Plath M (2006) Life on the edge: hydrogen sulfide and the fish communities of a Mexican cave and surrounding waters. Extremophiles 10:577–585
Townsend CR (1989) The patch dynamics concept of stream community ecology. J N Am Benthol Soc 8:36–50
Tuttle MD, Stevenson DE (1978) Variation in the cave environment and its biological implications. Natl Cave Manag Symp Proc 1977:108–121
Uzarski DG, Burton TM, Cooper MJ, Ingram JW, Timmermans STA (2005) Fish habitat use within and across wetland classes in coastal wetlands of the five Great Lakes: development of a fish-based index of biotic integrity. J Great Lakes Res 31:171–187
Vannote RL, Minshall GW, Cummins KW, Sedell JR, Cushing CE (1980) The river continuum concept. Can J Fish Aquat Sci 37:130–137
Winemiller KO, Flecker AS, Hoeinghaus DJ (2010) Patch dynamics and environmental heterogeneity in lotic ecosystems. J N Am Benthol Soc 29:84–99
Wootton RJ (1998) Ecology of teleost fishes, 2nd edn. Kluwer Academic Publishers, Dorddrecht
Acknowledgments
We thank Central Michigan University and the Office of Research and Sponsored Programs for funding this project. Without the assistance of volunteers from Forestville/Mystery Cave State Park, this research would not have been possible. Thanks to Warren Netherton, cave specialist at Forestville/Mystery Cave State Park, for information regarding the Root River and Mystery Cave. Additional thanks go to Daelyn Woolnough and David Coulter for assistance leading to the improvement of this work. Thanks also go to the editors and anonymous reviewers of this work whose input is greatly appreciated. This work was completed with approval from the CMU Institutional Animal Care and Use Committee (IACUC).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Coulter, A.A., Galarowicz, T.L. Fish assemblage and environmental differences upstream and downstream of a cave: a potential reset mechanism. Environ Biol Fish 98, 1223–1231 (2015). https://doi.org/10.1007/s10641-014-0354-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10641-014-0354-4