Abstract
Background
Organ Procurement and Transplantation Network and United Network for Organ Sharing (OPTN/UNOS) implemented the Share 35 policy in June 2013 to prioritize the sickest patients awaiting liver transplantation (LT). However, Model for End-Stage Liver Disease (MELD) score does not incorporate hepatic encephalopathy (HE), an independent predictor of waitlist mortality.
Aim
To evaluate the impact of severe HE (grade 3–4) on waitlist outcomes in MELD ≥ 30 patients.
Methods
Using the OPTN/UNOS database, we evaluated LT waitlist registrants from 2005–2014. Demographics, comorbidities, and waitlist survival were compared between four cohorts: MELD 30–34 with severe HE, MELD 30–34 without severe HE, MELD ≥ 35 with severe HE, and MELD ≥ 35 without severe HE.
Results
Among 10,003 waitlist registrants studied, 41.6% had MELD score 30–34 and 58.4% had MELD ≥ 35. Patients with severe HE had a higher 90-day waitlist mortality in both MELD 30–34 (severe HE 71.1% vs. no HE 56.6%; p < 0.001) and MELD ≥ 35 subgroups (severe HE 85% versus no HE 74.2%; p < 0.001). MELD 30–34 patients with severe HE had similar 90-day waitlist mortality as MELD ≥ 35 patients without severe HE (71.1 vs. 74.2%, respectively; p = 0.35). On multivariate Cox proportional hazards modeling, MELD ≥ 30 patients had 58% greater risk of 90-day waitlist mortality than those without severe HE (HR 1.58, 95% CI 1.53–1.62; p < 0.001).
Conclusion
Patients awaiting LT with MELD score of 30–34 and severe HE should receive priority status for organ allocation with exception MELD ≥ 35.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Due to the geographic disparities in the liver donor organ supply, the Organ Procurement and Transplantation Network and United Network for Organ Sharing (OPTN/UNOS) identified medical urgency as the governing principle in defining liver donor allocation within the USA. In 2002, the OPTN/UNOS adopted the Model for End-Stage Liver Disease (MELD) scoring system, a validated objective predictor for short-term mortality in patients on liver transplant (LT) waitlist. Since the implementation of MELD scoring system, the geographic disparities became increasingly evident as the MELD score at transplant was widely variable between Donor Service Areas (DSAs), suggesting that the sickest patients are not served in an equitable fashion [1, 2]. Over the past decade, several modifications to the MELD-based allocation policy have been adopted in an effort to provide egalitarian access to LT and to reduce the growing geographic disparity within the OPTN/UNOS regions. In 2013, the Share 35 policy was implemented to prioritize sicker regional patients with a MELD score 35 and above over local patients with a MELD score under 35. The early benefits of the Share 35 were immediately evident; there was a 30.3% increase in the proportion of LT for MELD ≥ 35 patients and 60.8% increase in the proportion of regional sharing with no overall negative impact on the waitlist survival [3]. Additionally, despite the increase in donor organ travel distance and cold ischemia time (CIT), the liver discard rate remained stable. Edwards et al. [4] evaluated the impact of Share 35 policy 2 years after its implementation and noted that the proportion of LT for MELD ≥ 35 increased from 18.5 to 26.5%, 90-day probability of LT increased from 59 to 66%, and more importantly the waitlist mortality decreased from 32 to 25%.
Although the MELD-based liver allocation is successful in many ways, it underestimates the severity of liver disease and the risk of death in subpopulations of patients with specific complications of end-stage liver disease. For example, hepatic encephalopathy (HE), a marker of hepatic decompensation and an independent predictor of mortality, is not represented in the MELD score. On contrary, Child–Turcotte–Pugh (CTP) score incorporates HE and therefore more accurately reflects the severity of liver disease in these patients. Several studies have demonstrated that HE negatively impacts the survival in patients with cirrhosis [5,6,7,8]. Furthermore, with the increasing severity of HE reflected by higher grades (West Haven grade 3–4), the risk of mortality increases exponentially. A retrospective study by Stewart et al. [9] showed that grade 3 or higher HE was associated with 3.7-fold (95% CI 1.9–7.3; p < 0.01) increase in the risk of death in patients with cirrhosis undergoing transjugular intrahepatic portosystemic shunt (TIPS), and in hospitalized patients with grade 2–4 HE the risk was 3.9-fold (95% CI 2.6–5.7; p < 0.01) higher. Wong et al. [10] demonstrated that HE was associated with worsening short-term waitlist survival in patients awaiting LT. While the access to donor organs improved in sicker patients with MELD ≥ 35 following the implementation of Share 35 policy, waitlist outcomes in patients with MELD < 35 and severe HE remain unknown. In this study, we evaluated the impact of severe HE on waitlist outcomes in patients with a MELD score 30–34 in comparison with MELD ≥ 35. Additionally, we evaluated waitlist outcomes in these patients with and without severe HE before and after implementation of the Share 35 policy.
Methods
Data Source
Our study utilized data from the OPTN/UNOS database. The OPTN/UNOS registry includes national data on all liver transplant waitlist registrants and recipients in the USA.
Study Population
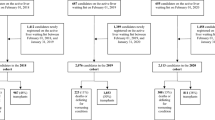
We performed a retrospective cohort study comparing adult (18 years of age or older) liver transplant waitlist registrants with a laboratory MELD score at listing of 30 and above (MELD ≥ 30) from January 1, 2005 to December 31, 2014. The study population with a MELD ≥ 30 was selected because these patients demonstrated a relatively higher waitlist mortality risk. Our primary objective was to evaluate the impact of severe (grade 3–4) HE on waitlist outcomes including (1) death and (2) LT among registrants with an MELD score ≥ 30 at waitlist registration. Waitlist registrants were categorized as MELD score 30–34 and MELD ≥ 35 and further sub-categorized into four cohorts: (1) initial listing MELD score 30–34 with severe HE, (2) initial listing MELD score 30–34 without HE, (3) initial listing MELD score ≥ 35 with severe HE, and (4) initial listing MELD score ≥ 35 without HE. Waitlist registrants who were listed emergently as Status 1, with hepatocellular carcinoma, received MELD exception points, or who were listed for liver re-transplantation were excluded from the analysis. The etiology of chronic liver disease leading to waitlist registration and LT was determined based on primary diagnosis code in the OPTN/UNOS registry. Clinical and demographic data collected at the time of waitlist registration were as follows: mean age ± standard deviation (SD), gender, ethnicity, etiology of liver disease including alcoholic liver disease (ALD), chronic hepatitis C virus (HCV) infection, and nonalcoholic steatohepatitis (NASH), diabetes mellitus, ascites, spontaneous bacterial peritonitis (SBP), and OPTN/UNOS region of listing. Patients with missing data on waitlist outcomes were excluded from our analysis.
Statistical Analysis
Clinical and demographic characteristics of the study cohort were presented as frequencies and proportions for categorical variables and mean ± SD for continuous variables. Comparisons between groups utilized Chi-square test for categorical variables and Student’s t test for continuous variables. To reduce the bias created by geographic OPTN/UNOS regional variation in waitlist time to transplant, rates for waitlist removal reason (death or LT) were assessed at 90-day follow-up from initial listing date. Ninety-day waitlist removal due to death was censored for LT. Ninety-day overall mortality was uncensored for LT and post-transplant survival. Using Cox hazards regression analyses, 90-day rate for waitlist mortality was determined within our cohorts and was adjusted for age, gender, ethnicity, etiology of liver disease, ascites, SBP, and OPTN/UNOS region of listing. In our sub-analysis, we assessed waitlist outcomes among our cohorts, 18 months prior to the implementation of the Share 35 policy (June 18, 2013), denoted ‘pre-Share 35 era’ (December 17, 2011–June 17, 2013), to an equivalent time period after, denoted ‘Share 35 era’ (June 19, 2013–December 19, 2014). To evaluate the impact of Share 35 policy on waitlist outcomes within our 4 cohorts, 90-day overall mortality (uncensored for LT) from waitlist registration date was analyzed among initial waitlist registrants. Initial registrants with 90-day follow-up that overlapped into Share 35 era were censored from waitlist. In addition, a cumulative waitlist mortality incidence curve with LT as the competing risk was constructed using a Cox regression model [11]. All statistical analyses were performed using the SAS statistical package (version 9.4, Cary, NC). Statistical significance was met with a p value < 0.05.
Results
Demographics and Clinical Characteristics
During the study period, there were 10,003 patients listed nationally for LT who met the inclusion criteria (MELD ≥ 30). Among those, 41.6% (n = 4166) had an initial listing MELD score 30–34 and 58.4% (n = 5837) had an initial listing MELD score ≥ 35. Out of 4166 patients with MELD score 30–34, 13.7% (n = 571) patients were noted to have grade 3–4 HE (severe HE) at registration. In contrast, nearly one-fifth of patients with MELD ≥ 35 were noted to have severe HE at registration (n = 1049, 17.9%). Baseline clinical demographics and characteristics of patients with severe HE who presented with an initial MELD 30–34 versus MELD ≥ 35 are summarized in Table 1. Within the four cohorts, there was a higher prevalence of male gender and Caucasian ethnicity/race with ALD as the leading etiology of liver disease. Comorbidities including diabetes, ascites, and SBP were evenly distributed across all cohorts. In addition, nearly one-fifth of patients in all four cohorts resided in UNOS region 5 at the time of listing.
Waitlist Outcomes
Patients with a MELD score ≥ 35 had a higher 90-day waitlist mortality rate than those with a MELD score of 30–34 (MELD 30–34, 65.5% vs. MELD ≥ 35, 82.8%; p < 0.001). Patients with severe HE had a significantly higher 90-day waitlist mortality compared to patients without severe HE among those with MELD scores of 30–34 (severe HE 71.1% vs. no HE 56.6%; p < 0.001) and MELD scores ≥ 35 (severe HE 85% vs. no HE 74.2%; p < 0.001) as tabulated in Table 2. Similar trends were noted in 30-, 60-day, and 1-year waitlist mortality (Fig. 1). Median waitlist time to death was shorter among patients with severe HE in both MELD score of 30–34 and MELD score ≥ 35 sub-cohorts compared to patients without HE (Table 2). Compared to patients with a MELD score ≥ 35 without HE, patients with MELD scores of 30–34 with severe HE had comparable 90-day waitlist mortality (MELD 30–34 with severe HE 71.1% vs. MELD ≥ 35 without HE 74.2%; p = 0.35). This is further clarified in the Kaplan–Meier survival analysis curve (Fig. 1) comparing MELD score 30–34 with severe HE subgroup versus MELD score ≥ 35 without severe HE subgroup (p = 0.12). On multivariate Cox proportional hazards modeling of 90-day waitlist mortality among patients with a MELD score ≥ 30, the presence of severe HE was the highest independent predictor of waitlist mortality (HR 1.58, 95% CI 1.53–1.62; p < 0.001) (Supplementary Table 1). Compared to patients with a MELD score of 30–34 with severe HE (reference), patients with a MELD score of 30–34 without HE had a significantly lower risk of death (HR 0.63, 95% CI 0.47–0.79; p < 0.001), while patients with a MELD score ≥ 35 with severe HE had the higher risk of death (HR 1.81, 95% CI 1.69–1.91; p < 0.001) as outlined in Table 3. However, when compared to patients with a MELD score ≥ 35 without severe HE, no significant difference in waitlist mortality was noted (HR 1.15, 95% CI 0.99–1.31; p = 0.37). Demographic and clinical characteristics of patients classified into cohorts based on MELD scores 30–34 and > 35 are included in Supplementary Table 2.
Effects of Share 35
After the implementation of Share 35 (Share 35 era) policy, patients with a MELD score 30–34 with or without HE did not experience a significant change in 90-day probability of undergoing LT. However, compared to pre-Share 35 era, patients with a MELD score ≥ 35 had increased 90-day probability of undergoing LT in Share 35 era, in both severe HE (pre-Share 35 era 47.6% vs. Share 35 era 56.3%; p = 0.04) and non-severe HE (pre-Share 35 era 59.6% vs. Share 35 era 69.6%; p < 0.001) groups. In terms of waitlist mortality, MELD 30–34 patients with or without severe HE did not experience change in 90-day overall mortality in Share 35 era (pre-Share 35 era 18.4% vs. Share 35 era 19.4%; p = 0.22). Patients with a MELD score ≥ 35 and severe HE had statistically insignificant change in 90-day mortality during the Share 35 era (pre-Share 35 era 37.5% vs. Share 35 era 44.8%; p = 0.09), whereas MELD ≥ 35 patients without severe HE had improved 90-day overall mortality (pre-Share 35 era 21.3% vs. Share 35 era 30.5%; p < 0.001) (Table 4).
Discussion
The principle objective of the regional Share 35 policy is to reduce waitlist mortality in sicker regional patients (MELD ≥ 35) by increasing their likelihood of receiving a donor liver and without negatively impacting the overall waitlist mortality in all registrants awaiting LT. Results from the studies that have evaluated 1- and 2-year post-Share 35 policy outcomes demonstrated an increase in regional sharing and improved probability of LT in patients with a MELD score ≥ 35 with the corresponding reduction in overall waitlist mortality [3, 4]. While the proportion of liver transplant recipients with an allocation MELD score ≥ 35 has increased from 22.3 to 30.5% in the Share 35 era, the probability of LT decreased for patients with a MELD score of 30–34 [3]. Although the overall waitlist mortality did not change for MELD 30–34 cohort, survival of patients with severe HE within this cohort is unknown. In the current study, we evaluated the impact of severe HE on 90-day waitlist mortality in all patients with a MELD score ≥ 30 and their sub-cohorts, MELD 30–34 with severe HE and MELD ≥ 35 with severe HE. We noted that patients with a MELD score of 30–34 and severe HE demonstrated a similar 90-day waitlist mortality as those with a MELD score ≥ 35 without severe HE. Severe HE is associated with 58% increased risk of death in patients with a MELD ≥ 30 and was noted to be the strongest independent predictor of waitlist mortality in these patients. Despite the comparable risk of waitlist mortality to patients with a MELD ≥ 35 and without severe HE, patients with MELD 30–34 and severe HE remain at a disadvantage to receive a donor offer for LT under Share 35 policy—a weakness of MELD-based score to recognize severe HE as a predictor of waitlist mortality. Accurate prioritization for LT is crucial to reduce the waitlist mortality in patients with end-stage liver disease. Current MELD-based allocation system does not incorporate the presence of HE, a marker of hepatic decompensation which has shown to independently impact waitlist mortality irrespective of MELD score. Several studies have shown that severity of underlying liver disease and related mortality are underestimated by excluding HE from the LT allocation criteria [8, 9, 12,13,14]. In a large multicenter center study, any grade of HE, especially higher grades, during the hospitalization was independently associated with increased risk of in-hospital and 30-day mortality regardless of multi-organ failure [15]. Wong et al. [10] utilizing UNOS registry demonstrated that severe HE at the time of registration was associated with 66% increase in 90-day waitlist mortality. In another study, patients with severe HE prior to LT were noted to have poor post-transplant outcomes including increased rates of infections [16]. HE is an important landmark in the natural history of cirrhosis-related hepatic decompensation, and its presence influences the patient survival independent of MELD score [9]. The results of our study show that both MELD 30–34 and MELD ≥ 35 patients with severe HE are predictors of 30-, 60-day, and 1-year waitlist mortality compared to patients without HE. During Share 35 era, patients with MELD ≥ 35 demonstrated an increased probability of undergoing LT, whereas patients with MELD 30–34 with or without severe HE experienced no change in the likelihood of undergoing LT. The MELD scoring system utilizes objective clinical data and is calculated by using verifiable laboratory results, and therefore inclusion of any subjective parameter such as HE is less desirable. In addition to the risk of subjective bias, unpredictable disease course with waxing and waning symptoms, effects of medications like sedatives and anxiolytics, difficulty in differentiating from other causes of mental status changes, and relative contribution of underlying cognitive disorders are considered barriers for incorporating HE in the current allocation model. The subjective variability can be minimized by using severe (grade 3–4) HE which is more reproducible with less inter-observer variability [15]. HE is a component of the CTP scoring system. Therefore, the presence of severe HE increases the CTP score in a patient and accurately reflects the severity of underlying liver disease. In a retrospective study comparing the predictive ability of the MELD and the CTP scores for waitlist mortality, area under curve analysis showed stronger trend toward the CTP than the MELD (AUROC, 0.73 vs. 0.68, p = 0.091) [17]. Utilizing the CTP score in addition to the MELD score or assigning MELD exception points for patients with severe HE awaiting LT can further reduce waitlist mortality. Results of our study which utilized the large national liver transplant registry emphasizes that the patients with a MELD score of 30–34 with severe HE should receive equal priority as regional waitlist registrants with a MELD score ≥ 35.
While the ways to incorporate severe HE in current liver allocation model are being explored, continued efforts for prompt diagnosis and effective management of HE in earlier stages must be taken to prevent the progression to more severe HE. Recently, with a relative reduction in other complications of cirrhosis such as variceal hemorrhage and availability of newer drugs for HE, there has been an increased emphasis on early recognition and management of HE [15]. HE has multidimensional burden on patients, caregivers, and health care resource utilization [15, 18]. Patient and family education, emphasis on compliance, correction of underlying electrolyte abnormalities, and treatment of infections and other precipitating factors of HE such as variceal hemorrhage are crucial in improving waitlist survival of these patients.
The strengths of our study include the 10-year data of all adult liver transplant registrants in the USA. Consistency of data reporting by OPTN/UNOS is expected, and therefore the results can be generalizable. However, our study has several limitations. Data from databases are subject to errors of data entry and coding. Diagnosing and grading the severity of HE are vulnerable to subjective bias and variability. It is unclear and cannot be verified if all centers used identical grading system for HE. We studied the presence and severity of HE at listing in our analysis; however, the duration and variation in the grades of HE are not available in the OPTN/UNOS registry, and thus its impact cannot be assessed. Also, we are not able to identify and analyze the underlying etiology or precipitating factor of HE.
In conclusion, our study demonstrates that HE is a serious complication of end-stage liver disease and is an independent predictor of mortality in patients with cirrhosis on the liver transplant waitlist. HE has emerged as a leading cause of hospitalizations and readmissions in this patient population [19, 20]. While higher proportion of critically ill patients with a MELD ≥ 35 have received LT after implementation of Share 35, patients with a MELD score of 30–34 and severe HE who have similar waitlist mortality risk continue to be at disadvantage. Our results indicate that these patients should receive an equal priority in current liver transplant allocation system as those with a MELD score ≥ 35.
Abbreviations
- ALD:
-
Alcoholic liver disease
- CIT:
-
Cold ischemia time
- CTP:
-
Child–Turcotte–Pugh
- DSA:
-
Donor Service Area
- HE:
-
Hepatic encephalopathy
- HCV:
-
Hepatitis C virus
- LT:
-
Liver transplantation
- MELD:
-
Model for End-Stage Liver Disease
- NASH:
-
Nonalcoholic steatohepatitis
- OPTN:
-
Organ Procurement and Network
- UNOS:
-
United Network for Organ Sharing
References
Dzebisashvili N, Massie AB, Lentine KL, et al. Following the organ supply: Assessing the benefit of inter-DSA travel in liver transplantation. Transplantation. 2013;95:361–371.
Roberts JP, Dykstra DM, Goodrich NP, et al. Geographic differences in event rates by Model for End-Stage Liver Disease score. American Journal of Transplantation. 2006;6:2470–2475.
Massie AB, Chow EK, Wickliffe CE, et al. Early changes in liver distribution following implementation of Share 35. American Journal of Transplantation. 2015;15:659–667.
Edwards EB, Harper AM, Hirose R, et al. The impact of broader regional sharing of livers: 2-year results of “Share 35”. Liver Transplatation. 2016;22:399–409.
D’Amico G, Garcia-Tsao G, Pagliaro L. Natural history and prognostic indicators of survival in cirrhosis: a systematic review of 118 studies. Journal of Hepatology. 2006;44:217–231.
Gines P, Quintero E, Arroyo V, et al. Compensated cirrhosis: natural history and prognostic factors. Hepatology. 1987;7:122–128.
Merkel C, Bolognesi M, Angeli P, et al. Prognostic indicators of survival in patients with cirrhosis and esophageal varices, without previous bleeding. American Journal of Gastroenterology. 1989;84:717–722.
Milani A, Marra L, Siciliano M, et al. Prognostic significance of clinical and laboratory parameters in liver cirrhosis. A multivariate statistical approach. Hepatogastroenterology. 1985;32:270–272.
Stewart CA, Malinchoc M, Kim WR, et al. Hepatic encephalopathy as a predictor of survival in patients with end-stage liver disease. Liver Transplatation. 2007;13:1366–1371.
Wong RJ, Gish RG, Ahmed A. Hepatic encephalopathy is associated with significantly increased mortality among patients awaiting liver transplantation. Liver Transplantation. 2014;20:1454–1461.
Kim WR, Therneau TM, Benson JT, et al. Deaths on the liver transplant waiting list: an analysis of competing risks. Hepatology. 2006;43:345–351.
Said A, Williams J, Holden J, et al. Model for end stage liver disease score predicts mortality across a broad spectrum of liver disease. Journal of Hepatology. 2004;40:897–903.
Yoo HY, Edwin D, Thuluvath PJ. Relationship of the Model for End-Stage Liver Disease (MELD) scale to hepatic encephalopathy, as defined by electroencephalography and neuropsychometric testing, and ascites. American Journal of Gastroenterology. 2003;98:1395–1399.
Ham J, Gish RG, Mullen K. Model for End-Stage Liver Disease (MELD) exception for hepatic encephalopathy. Liver Transplantation. 2006;12:S102–S104.
Bajaj JS, Wade JB, Gibson DP, et al. The multi-dimensional burden of cirrhosis and hepatic encephalopathy on patients and caregivers. American Journal of Gastroenterology. 2011;106:1646–1653.
Wong RJ, Aguilar M, Gish RG, et al. The impact of pretransplant hepatic encephalopathy on survival following liver transplantation. Liver Transplantation. 2015;21:873–880.
Gotthardt D, Weiss KH, Baumgartner M, et al. Limitations of the MELD score in predicting mortality or need for removal from waiting list in patients awaiting liver transplantation. BMC Gastroenterology. 2009;9:72.
Tapper EB. Challenge accepted: Confronting readmissions for our patients with cirrhosis. Hepatology. 2016;64:26–28.
Bajaj JS, Reddy KR, Tandon P, et al. The 3-month readmission rate remains unacceptably high in a large North American cohort of patients with cirrhosis. Hepatology. 2016;64:200–208.
Cordoba J, Ventura-Cots M, Simon-Talero M, et al. Characteristics, risk factors, and mortality of cirrhotic patients hospitalized for hepatic encephalopathy with and without acute-on-chronic liver failure (ACLF). Journal of Hepatology. 2014;60:275–281.
Author information
Authors and Affiliations
Contributions
CG and GC were responsible for study concept and design, acquisition of the data, analysis and interpretation of the data, and drafting and approval of the final manuscript. MH was responsible for acquisition of the data, statistical analyses, and drafting and approval of the final manuscript. ERY was responsible for drafting, critical revision, and approval of the final manuscript. RJW and AA were responsible for the interpretation of the data, study supervision, drafting, critical revision, and approval of the final manuscript. All authors were involved in the final approval of the manuscript submitted and have agreed to be accountable for all aspects of the work.
Corresponding author
Ethics declarations
Conflict of interest
All authors declare no conflict of interest in the preparation of this manuscript, including financial, consultant, institutional, and other relationships that might lead to bias.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Gadiparthi, C., Cholankeril, G., Yoo, E.R. et al. Waitlist Outcomes in Liver Transplant Candidates with High MELD and Severe Hepatic Encephalopathy. Dig Dis Sci 63, 1647–1653 (2018). https://doi.org/10.1007/s10620-018-5032-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-018-5032-5