Abstract
Stroke was regarded as a severe disorder with high morbidity and high mortality worldwide, ischemic stroke (IS) accounts for 85 to 90 % of new increased stroke cases. Partial mechanisms were elucidated by genetic factors including genomic instability such as single nucleotide polymorphism (SNP). Previous reports demonstrated that inflammation was involved in IS, NLRP3 [nucleotide-binding domain (NOD)-like receptor protein 3], acting as a specific inflammatory gene, however, its function and influence on IS was not well clarified. In this study, a case–control study including 1102 IS patients and 1610 healthy controls was conducted to investigate the association between IS susceptibility with a SNP (rs10754558) in 3′UTR of NLRP3. Logistic regression analysis showed that the heterozygote and the homozygote GG confer a significantly increased risk of CRC after controlling for other covariates (adjusted OR = 1.52, 95 % C.I. 1.19–1.97, P = 0.002; adjusted OR = 2.22, 95 % C.I. 2.18–3.67, P < 0.001, respectively). Carriage of G allele was associated with a greatly increased risk of developing the disease (OR = 1.69, 95 % C.I. 1.31–1.83, P < 0.001). Stratification analysis found that hypertension had interaction with rs10754558 to modulate IS risk. Further in vitro assay revealed that rs10754558 can affect mRNA level of NLRP3, suggesting its possible functional significance. Our data suggested that genetic polymorphisms in NLRP3 may influence IS risk in Chinese population. Replication of our studies in other populations and further functional studies are required for complete comprehension of the roles of NLRP3 polymorphisms in IS risk.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Stroke was regarded as a severe disorder with high morbidity and high mortality, it claims over 6 million deaths each year worldwide, while developing countries such as China contributes the majority of death toll (Donnan et al. 2008; Matarin et al. 2010). As far as we know, the prevalence rate has been growing in China, ischemic stroke (IS) accounts for 85–90 % of new increased stroke cases (Adams et al. 2007; Feigin et al. 2014). Multiple elements are correlated with its pathogenesis, which include environmental factors and genetic profiles (Donnan et al. 2008; Dichgans 2007). Although Genome-wide association study (GWAS) was widely conducted in IS susceptibility-associated SNPs identification during a decade or two, coverage of the human genome with genome-wide studies is neither equal across regions of the genome nor comprehensive (Kubo et al. 2007; Yamada et al. 2009; Hata et al. 2007; Matarin et al. 2008). Many previously associated candidate genes such as ACE are not adequately covered by the currently commercially available GWAS platforms, which leads to incomplete understanding of the exact genetic susceptibility for IS.
Recently, the correlation between inflammation and IS has attracted much attention (Ahmad et al. 2014; Fann et al. 2013). Inflammation contributes to ischemic events through the promotion of atherosclerosis, Inflammatory genes may thereby influence the incidence and outcome of IS (Tong et al. 2015). Inflammasome pattern recognition receptors, which belong to the family of multimeric proteins, play an important role in innate immunity, including NLRPs, NLRC, and NAIP. Among these receptors, NLRP3 (nucleotide-binding domain (NOD)-like receptor protein 3) inflammasome may activate the inflammation and participate in atherosclerosis (Kristiina et al. Kristiina et al. 2010), pathophysiology of myocardial infarction (Takahashi 2014), result in cardiovascular diseases such as IS. The gene NLRP3 was involved in human acute immune and inflammatory response, inadequate or excessive reaction will cause damage to the human body. Innate immunity plays an important role in inflammation-related neuronal injury, which is associated with IS. Several investigations showed that the polymorphism within the 3′UTR of NLRP3 rs10754558 was associated with multiple diseases, and these diseases mainly resulted from inflammation (Zhang et al. 2014; Tan et al. 2013; Kamada et al. 2014). Furthermore, inflammation-associated genes including NLRP3 were known to influence IS. The polymorphism rs10754558 was located in potentially functional region-3′UTR of NLRP3, which may indicate a possible manner for NLRP3 expression modulation in IS. Thus, previous reports refereed above encouraged us to speculate that the polymorphism modulates IS risk, its function may be based on influence of NLRP3 expression.
To our knowledge, there were no well-conducted studies on the relationship between NLRP3 rs10754558 and IS, although the evidence indicated that they may have an association. In our study, polymerase chain reaction with sequence-specific primers (PCR-SSP) was used for NLRP3 rs10754558 genotyping and that whether the gene variant was associated with IS in a Chinese population was also validated, furthermore, qPCR was conducted in PBMCs of enrolled cases to demonstrate the genotype-phenotype connection. Here, our research group reported that NLRP3 rs10754558 was associated with the risk of IS, genotyping subgroups also showed dose-dependent effect in transcription level of blood and it may be a potentially promising marker for IS susceptibility. Our investigation may serve to high-risk populations identification and early prevention for IS patients.
Materials and Methods
All subjects recruited were unrelated ethnic Han Chinese. The case series consisted of 597 IS patients diagnosed, hospitalized and treated in ward and outpatient department of Emergency of the Suzhou Kowloon Hospital Shanghai Jiao Tong University School of Medicine from 2013 to 2015, and 505 IS patients diagnosed, hospitalized and treated in ward and outpatient department of Neurology of the Beijing 302 Military Hospital of China. All subjects underwent a standardized clinical evaluation. We included stroke patients from outpatients department as well as from neurology ward (where severely disable patients are more in number) to deal with survival bias. Stroke was classified as per TOAST classification (Adams et al. 1993). A total of 1610 controls were stroke-free individuals selected from community nutritional surveys that were conducted in the same regions during the same period as recruitment of IS patients, which consisted of 843 healthy controls from Suzhou Kowloon Hospital and 767 healthy controls from Beijing 302 Military Hospital of China. Informed consent was obtained from all individual participants each patient/control or from the relatives or a legal representative in the case of critically disabled patients. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Selection of Cases and Controls
Inclusion criteria for stroke patients were (a) diagnosis of stroke as defined by WHO (rapidly developing clinical symptoms and/or signs of focal (at times global) disturbance of cerebral function, with symptoms lasting more than 24 h with no apparent cause other than that of vascular origin; (b) age (18–85 years); (c) NCCT-head consistent with ischemic stroke; (d) birth place of subjects and their ancestors (2 generation) from mainland China. For this study patients and controls were recruited from Xuzhou and Beijing; (e) no evidence of trauma and brain tumor/metastases; and (f) willingness to provide informed consent by self or legal representative. Selection criteria for controls were same as cases except that they were stroke-free determined by means of the questionnaire for verifying stroke-free status (QVSS) (Meschia et al. 2000).
Questionnaire and Definition of Variables
Demographic characteristics and risk factors were collected in standardized data collection form using a structural questionnaire. Risk factors included hypertension, diabetes, dyslipidemia, smoking, first generation family history of stroke, body mass index (BMI).Definitions of variables were modified from the study (Feigin et al. 1998) and are as follows: Hypertension: subjects were considered to have hypertension if they either had the diagnosis of hypertension or were treated for hypertension before the stroke or reference date. Diabetes: if a subject had the diagnosis documented by a physician on the medical record or if fasting blood sugar level was ≥126 mg/dl. Dyslipidemia: if they either had the diagnosis of dyslipidemia or were treated for dyslipidemia before the stroke or reference date. Smoker: a person was defined as regular smoker if smoking ≥1 cigarette(s) daily, and biris and cigar for preceding ≥3 months. Body Mass Index: BMI were calculated by weight in kilograms divided by the square of height in meters (Table 1).
DNA Extraction and Genotyping
A Chelex method was used for extracting genomic DNA of human peripheral blood samples (Walsh et al. 1991). Genotyping was applied by polymerase chain reaction with sequence-specific primers (PCR-SSP). All primers for the PCR-SSP were designed using the genomic sequences in the GenBank (http://www.ncbi.nlm.nih.gov). The primer sequences of the polymorphism are listed in Table 2. PCR amplification was performed with a total of 30 μL reaction mixture containing 20 ng DNA, 0.5 μM of each primer, 2.5 μM dNTP, 2.0 Mm MgCl2 and 1.25 U Taq DNA polymerase with 1 × reaction buffer (Takara). The PCR protocol included an initial denaturation step at 95 °C for 5 min; 30 cycles of 1 min denaturation step at 95 °C, 45 s annealing step at appropriate temperatures and 1 min extension step at 72 °C, and with 10 min final extension step at 72 °C. The amplified products were assessed for the presence/absence of PCR specific to the particular alleles using a standard 2 % agarose gel electrophoresis followed by ethidium bromide staining. 10 % samples were then confirmed by sequencing, and the reproducibility was 100 %.
RNA Extraction and qPCR
PBMCs were separated from human peripheral blood samples by Ficoll/Histopaque (Sigma–Aldrich). Total RNA was extracted using the TaKaRa RNAiso Reagent (Takara) from PBMCs and cDNA was obtained using the PrimeScript II 1st strand cDNA synthesis kit(Takara). qPCR assay was performed to detect the expression of PBMCs in three genotype groups for the relative quantitative analysis, primers for qPCR are listed in Table 2.
Statistical Analysis
Allele frequencies and genotype distributions of NLRP3 rs10754558 in different groups were compared using Chi-square test. Conditional logistic regression model was used to estimate OR and 95 % confidence intervals (CI) for association of NLRP3 genotype with ischemic stroke. Multivariate logistic regression analysis was used to exclude the effect of possible confounding factors. Comparison between the mean value of NLRP3 level and NLRP3 genotypes was analyzed using the ANOVA test. Odds ratios with 95 % CI were taken to estimate the association of the specific genotype with stroke and were considered significant at P < 0.05. Chi-square test was used to determine the frequency distribution of genotypes in accordance with Hardy–Weinberg equilibrium (HWE). Data were analyzed using the SPSS statistical package, version 17.0 (SPSS, Chicago, IL, USA).
Results
Demographic Characteristics of Enrolled Subjects
The statistical analysis of demographic characteristics of the 1102 IS patients and 1610 controls is summarized in Table 1. Subjects of our case–control study were matched with age and sex, there were marked differences between cases and controls in terms of the frequency distribution of hypertension, diabetes, dyslipidemia, smoking status, and family history of stroke in both centers, the trend was similar with recruited subjects from previous reports. Beyond expected, BMI was not shown as a significant risk factor for IS in recruited subjects of Suzhou group.
Correlation Between NLRP3 rs10754558 and IS Risk
Statistical analysis of NLRP3 rs10754558 and IS risk in two centers is summarized in Table 3. Genotype distributions had no deviation from Hardy–Weinberg equilibrium in Suzhou control group, Beijing control group or pooled control group (P > 0.05, data not shown). Our results showed that rs10754558 was significantly associated with IS susceptibility, at both the allele and genotype levels (Table 3). As depicted in the co-dominant model of Suzhou case–control group, compared with the reference (CC), we found that the heterozygote and homozygote GG of rs10754558 were associated with a significantly increased risk of is after controlling for other covariates selected in our study (adjusted OR = 1.52, 95 % C.I. 1.19–1.97, P = 0.002; adjusted OR = 2.22, 95 % C.I. 2.18–3.67, P < 0.001, respectively). In the dominant model (CG + GG vs CC), significant associations were also observed between cases and controls (adjusted OR = 1.82, 95 % C.I. 1.82(1.47–2.81), P < 0.001), data from recessive model displayed similar trend. Meanwhile, frequencies of C or G allele were significantly different between IS and control groups. In the additive model, each additional copy of G allele was associated with a 69 % increased risk of developing cancer (OR = 1.69, 95 % C.I. 1.31–1.83, P < 0.001). Analysis of Beijing group and pooled group validated our findings in Suzhou group.
Stratification Analysis of rs10754558 with Risk Factors in IS Cases
NLRP3 rs10754558 and was confirmed as a risk factor for IS risk, we then had stratified analysis in IS cases to detect potential interactions between the polymorphism and candidate risk factors enrolled in present study. After stratification, only one risk factor hypertension status showed positive interaction with polymorphism (Table 4). Other candidate risk factors such as age, sex, BMI, diabetes, dyslipidemia, smoking status, and family history of stroke showed no significantly statistical difference (data not shown).
The Influence of rs10754558 on NLRP3 Expression
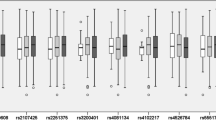
To further investigate whether rs10754558 would likely to affect the level of transcriptional activity of NLRP3, qPCR was conducted in PBMCs of enrolled cases to demonstrate the genotype-phenotype connection. As shown in Fig. 1, we found that IS cases carried with G allele drove a 1.36–1.88-fold increased transcription compared with the CC genotyped counterparts. Control genotyped groups showed similar propensity, but expression activities were slightly lower when compared with IS cases counterparts, the expression comparison showed no obvious difference (P > 0.05, data not shown).
Discussion
As far as we knew, this was the first molecular epidemiological report to investigate the correlation of NLRP3 polymorphism with risk of IS in Chinese Han population. The genotyping of 1102 IS patients and 1610 healthy control individuals showed significant associations with the SNP variant within 3′UTR of NLRP3 with IS susceptibility. Stratified analysis found that hypertension could interact with the polymorphism to upregulate IS risk in the present study. Furthermore, NLRP3 transcription in blood analysis showed that the G variant allele significantly upregulated the transcription activity of the NLRP3 gene compared with the C allele; control groups also showed similar propensity.
Polymorphisms within 3′UTR are possibly the most potential factors for gene expression inhibition/enhancement. The presence of the rs10754558 G allele was previously reported to lead to conformational change and increase the stability of the 3′UTR structure of NLRP3 mRNA, resulting in a higher expression level (Hitomi et al. 2009), our results also demonstrated this trend in Chinese Han population. NLRP3 was known to serve as a key mediator of the innate immune response, and inadequate or excessive reaction will cause damage to the human body. Enhanced NLRP3 expression may activate the inflammasome through the recognition of several cell stress molecules (Gram et al. 2012). Inflammasome has been found play a key role in the innate immune response reduced inflammation, innate immunity plays a pivotal role in inflammation-related neuronal injury, which is associated with IS.
An interesting find in present study was the interaction of NLRP3 rs10754558 and hypertension status in IS cases. At least one report supported the correlation between NLRP3 polymorphism and hypertension (Suárez and Buelvas 2015), but the possible association NLRP3 rs10754558 and hypertension risk was not validated to our knowledge. Known as a stroke prediction marker, hypertension was also one of general prevention strategies (Esenwa and Gutierrez 2015), if the possible association NLRP3 rs10754558 and hypertension risk was identified, NLRP3 rs10754558 would be a promising marker to guide both stroke and hypertension therapy and prevention.
We are aware that our study had several limitations, specifically the replication study in other populations, more primary data from in vivo analysis and further functional analysis. Considering location of the locus and previous report (Zhao et al. 2015), we speculated that G allele may disrupt the primary binding site, which in turn influences the potential miRNA-MicroRNA-223 recruitment and binding, thus NLRP3 transcription was upregulated.
Taken together, our data indicated that genetic polymorphisms in NLRP3 may influence IS risk in Chinese Han population. However, this IS-associated polymorphism rs10754558 showed none direct demonstration that rs150376137 is causative due to a genetic conception that another genetic variation may link to rs10754558 is the true mutation driving the association. Finally, the replication of our studies in other populations and further in vivo analysis are required for complete elucidation of the roles of NLRP3 polymorphisms in predisposition for IS.
References
Adams HPJ, Bendixen BH, Kappelle LJ et al (1993) Classification of subtype of acute ischemic stroke. Definitions for use in a multicenter clinical trial. TOAST. Trial of Org 10172 in Acute Stroke Treatment. Stroke 24(1):35–41
Adams HP Jr, Zoppo GD, Alberts MJ et al (2007) Guidelines for the early management of adults with ischemic stroke: a guideline from the American Heart Association/American Stroke Association Stroke Council, Clinical Cardiology Council, Cardiovascular Radiology and Intervention Council, and the Atherosclerotic Peripheral Vascular Disease and Quality of Care Outcomes in Research Interdisciplinary Working Groups. Stroke 2:29–36
Ahmad M, Dar NJ, Bhat ZS et al (2014) Inflammation in ischemic stroke: mechanisms, consequences and possible drug targets. CNS Neurol Disord Drug Targets 13(8):1378–1396
Dichgans M (2007) Genetics of ischaemic stroke. Lancet Neurol 6:149–161
Donnan GA, Fisher M, Macleod M, Davis SM (2008) Stroke. Lancet 371:1612–1623. doi:10.1016/S0140-6736(08)60694-7
Esenwa C, Gutierrez J (2015) Secondary stroke prevention: challenges and solutions. Vasc Health Risk Manag 7(11):437–450. doi:10.2147/VHRM.S63791
Fann DY, Lee SY, Manzanero S et al (2013) Pathogenesis of acute stroke and the role of inflammasomes. Ageing Res Rev 12(4):941–966
Feigin VL, Wiebers DO, Nikitin YP et al (1998) Risk factors for ischemic stroke in a Russian community: a population-based case-control study. Stroke 29(1):34–39
Feigin VL, Forouzanfar MH, Krishnamurthi R et al (2014) Global and regional burden of stroke during 1990–2010: findings from the Global Burden of Disease Study 2010. Lancet 383(9913):245–255. doi:10.1016/S2214-109X(13)70089-5
Gram AM, Frenkel J, Ressing ME (2012) Inflammasomes and viruses: cellular defence versus viral offence. J Gen Virol 93(10):2063–2075
Hata J, Matsuda K, Ninomiya T et al (2007) Functional SNP in an Sp1-binding site of AGTRL1 gene is associated with susceptibility to brain infarction. Hum Mol Genet 16:630–639
Hitomi Y, Ebisawa M, Tomikawa M et al (2009) Associations of functional NLRP3 polymorphisms with susceptibility to food-induced anaphylaxis and aspirin-induced asthma. J Allergy Clin Immunol 124(4):779–785.e6
Kamada AJ, Pontillo A, Guimarães RL et al (2014) NLRP3 polymorphism is associated with protection against human T-lymphotropic virus 1 infection. Mem Inst Oswaldo Cruz 109(7):960–963
Kristiina R, Jani L, Katariina Ö et al (2010) Cholesterol crystals activate the NLRP3 inflammasome in human macrophages: a novel link between cholesterol metabolism and inflammation. PLoS One 5(7):e11765. doi:10.1371/journal.pone.0011765
Kubo M, Hata J, Ninomiya T et al (2007) A nonsynonymous SNP in PRKCH (proteinkinase C eta) increases the risk of cerebral infarction. Nat Genet 39:212–217
Matarin M, Brown WM, Singleton A et al (2008) Whole genome analyses suggest ischemic stroke and heart disease share an association with polymorphisms on chromosome 9p21. Stroke 39:1586–1589. doi:10.1161/STROKEAHA.107.502963
Matarin M, Singleton A, Hardy J et al (2010) The genetics of ischaemic stroke. J Intern Med 267:139–155. doi:10.1111/j.1365-2796.2009.02202.x
Meschia JF, Brott TG, Chukwudelunzu FE et al (2000) Verifying the stroke-free phenotype by structured telephone interview. Stroke 31(5):1076–1080
Suárez R, Buelvas N (2015) Inflammasome: activation mechanisms. Invest Clin 56(1):74–99
Takahashi M (2014) Nlrp3 inflammasome as a novel player in myocardial infarction. Int Heart J 55(2):101–105
Tan MS, Yu JT, Jiang T et al (2013) NLRP3 polymorphisms are associated with late-onset Alzheimer’s disease in Han Chinese. J Neuroimmunol 265(1–2):91–95
Tong Y, Ding ZH, Zhan FX et al (2015) The NLRP3 inflammasome and stroke. Int J Clin Exp Med 8(4):4787–4794
Walsh PS, Metzger DA, Higushi R (1991) Chelex 100 as a medium for simple extraction of DNA for PCR-based typing from forensic material. Biotechniques 10(4):506–513
Yamada Y, Fuku N, Tanaka M et al (2009) Identification of CELSR1 as a susceptibility gene for ischemic stroke in Japanese individuals by a genome-wide association study. Atherosclerosis 207:144–149
Zhang HX, Wang ZT, Lu XX et al (2014) NLRP3 gene is associated with ulcerative colitis (UC), but not Crohn’s disease (CD), in Chinese Han population. Inflamm Res 63(12):979–985. doi:10.1007/s00011-014-0774-9
Zhao Y, Lina Z, Ronghua X et al (2015) MicroRNA-223 regulates inflammation and brain injury via feedback to NLRP3 inflammasome after intracerebral hemorrhage. Mol Immunol 65:267–276. doi:10.1016/j.molimm.2014.12.018
Acknowledgments
This study is supported by grants from Natural Science Foundation of China (No. 81502428), Natural Science Foundation of Jiangsu Province (No. BK20140222, No. 15KJB310024, No. BK20150220), Xuzhou Medical College scientific research fund for talents (No. D2015018 to Zhansheng Zhu, and No. D2015019 to Huiping Wang).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Zhu, Z., Yan, J., Geng, C. et al. A Polymorphism Within the 3′UTR of NLRP3 is Associated with Susceptibility for Ischemic Stroke in Chinese Population. Cell Mol Neurobiol 36, 981–988 (2016). https://doi.org/10.1007/s10571-015-0288-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-015-0288-1