Abstract
To explore the value of right ventricular (RV) parameters detected by three-dimensional echocardiography (3DE) in risk stratification in pulmonary arterial hypertension (PAH) patients. We prospectively recruited 130 pulmonary hypertension patients from National Center for Cardiovascular Diseases, Fuwai Hospital. Each participant was performed a transthoracic echocardiography and 3DE parameters were measured using an off-line software (4D RV Function 2.0, TomTec). Patients were classified into low, intermediate-high risk group based on 2015 ESC Guidelines. A total of 91 PAH patients (34 ± 12 years old, 25 males) were enrolled, among which, 42 were classified into low risk group, while 49 were intermediate-high risk group. Compared with low-risk patients, those with intermediate-high risk had significantly larger 3DE-RV volumes, worse ejection fraction (EF) and tricuspid annular plane systolic excursion, and decreased longitudinal strain (LS). Receive operating characteristic curves illustrated all the 3DE parameters were able to predict intermediate-high risk stratification, especially 3D-RVEF (area under curve, 0.82, 95% CI 0.73–0.91, P < 0.001). And 3D-RVEF < 26.39% had a 81.6% sensibility and 73.8% specificity to predict intermediate-high risk stratification. Univariate and multivariate Logistic regression analyses identified 3D-RV end-diastolic (OR 1.02, 95% CI 1.01–1.03, P = 0.002) and end-systolic (OR 1.03, 95% CI 1.01–1.04, P < 0.001) volumes, 3D-RVEF (OR 0.82, 95% CI 0.75–0.90, P < 0.001) and LS of free wall (OR 1.17, 95% CI 1.05–1.31, P = 0.005) as independent predictors of intermediate-high risk stratification. In conclusion, RV volumes, EF and free wall strain detected by 3DE were independent predictors of intermediate-high risk stratification in PAH patients, among which, RVEF showed the best predictive capacity.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Pulmonary hypertension (PH) describes a group of disease characterized as an increased pressure of pulmonary circulation, and they often have poor prognosis because the volume and pressure overload inevitably result in right heart dysfunction [1]. According to 2015 European Society of Cardiology (ESC) Guidelines for the diagnosis and treatment of pulmonary hypertension, pulmonary arterial hypertension (PAH) is a subgroup of PH sharing similar hemodynamic characteristics [2]. Comprehensive risk stratification is recommended to evaluate disease severity at baseline and each follow-up, through which, PAH patients are divided into low, intermediate and high risk, corresponding to an estimated 1-year mortality of < 5%, 5–10%, > 10%, respectively. Based on the risk assessment, we consider low risk as therapeutic goal. However, the recommended risk stratification shares some deficits, it includes indices of symptoms, clinical signs, exercise capacity, imaging and haemodynamics, which are complex and some of them are obtained from invasive examination, such as right heart catheterization (RHC). Therefore, further investigation is still needed to explore simple and convenient ways to evaluate disease severity of PAH patients.
As a recent developed technique, three-dimensional echocardiography (3DE) is able to determine cardiac chamber anatomy and function rapidly and accurately, independent of angle and without making geometric assumption [3, 4]. Right ventricular (RV) volumes and ejection fraction (EF) detected by 3DE have been reported to be associated with clinical indices, such as WHO functional class (WHO-FC) [5], hemodynamic status [6] and clinical outcomes [7,8,9] in PH patients. Consider that most of these clinical indices are included in the comprehensive risk stratification, we hypothesized that 3DE parameters approaching RV function maybe associated with risk stratification in PAH patients. Accordingly, we aimed to explore the value of 3DE RV parameters in risk stratification based on 2015 ESC Guidelines in PAH patients.
Methods
Study population
From March 2017 to May 2018, we prospectively recruited 130 PH patients from National Center for Cardiovascular Diseases, Fuwai Hospital. The exclusion criteria included intra-cardiac shunts, arrhythmia, acute heart failure, renal or hepatic failure, and under these conditions, 5 patients were excluded. Moreover, 12 patients were excluded due to poor image quality, and 22 patients diagnosed as chronic thromboembolic PH (CTEPH) or PH due to hypoxia were also excluded. Finally, 91 patients diagnosed as PAH by RHC were recruited in our study (Fig. 1).
Written informed consents were obtained from all the participants or their legal representatives. The present study was approved by the Ethics Committee of Fuwai Hospital (No. 2018-1063). All procedures performed in our study involving human participants were in accordance with the 1964 Helsinki declaration and its later amendments.
Acquisition and analysis of three-dimensional echocardiography
Every participant recruited in the present study was performed a transthoracic echocardiography. At least three cine-loops of 3DE images were acquired from the apical RV-focused four-chamber view, using a Vivid S6 equipment (GE Medical Systems). We used the off-line software (4D RV Function 2.0, TomTec Imaging Systems GmbH, Germany) to analyze 3DE parameters. As showed in Fig. 2, three orthogonal planes and various landmarks were selected to define the end-diastolic and end-systolic frames. On the basis of the initial view adjustment, the program automatically supplied four-chamber, sagittal, and coronal RV views. RV end-diastolic volume (EDV), end-systolic volume (ESV), RVEF, RV longitudinal strain (RVLS) of septum (S) and free wall (FW) and tricuspid annular plane systolic excursion (TAPSE) were automatically generated by the software. All the measurements were performed by a trained technician blinded to clinical data, according to the guidelines of the American Association of Echocardiography [10]. Inter-operator variability was assessed in a randomly selected group of 15 PAH patients, respectively.
Clinical data
Clinical data were obtained by reviewing medical records. Demographic characteristics (age, gender and body mass index, BMI, calculated as weight/height2), N-terminal pro-brain natriuretic peptide (NT-proBNP) levels, WHO-FC, 6-min walk distance (6MWD), peak oxygen consumption (PVO2) detected by cardiopulmonary exercise testing (CPET), as well as targeted drugs were obtained from all participants. In addition, hemodynamic parameters including right atrial pressure (RAP), mean pulmonary arterial pressure (mPAP), pulmonary artery wedge pressure (PAWP) and mixed venous oxygen saturation (SvO2) were recorded during RHC. Cardiac output (CO) was measured by the thermodilution method and body surface area (BSA) was calculated according to the DuBois formula; Cardiac index (Ci) was calculated as CO/BSA. Pulmonary vascular resistance (PVR) was calculated as (mPAP–PAWP)/CO. The intervals between 3DE and RHC were less than 24 h in 77 out of 91 participants. For other 14 patients, the RHC were not performed simultaneously with 3DE, therefore, their hemodynamic indices were not used for analysis in the present study.
Risk stratification
Patients were classified into low, intermediate and high risk group based on 2015 ESC Guidelines [2] and other two previous reports [11, 12]. WHO-FC, NT-pro BNP, exercise capacity (6MWD or PVO2) and hemodynamic parameters detected by RHC (RAP, Ci, SvO2) were used to assess risk stratification, as described in Table 1. Each variable was scored from 1 to 3, corresponding to low risk, intermediate risk and high risk, respectively. We divided the sum of all scores by the number of available variables for each patient, and the mean grade was rounded off to the nearest integer, which was used to define the patient’s risk group [12]. We classified patients into low and intermediate-high risk groups for analysis in the present study.
Statistical analysis
Continuous variables were described as mean ± standard deviation (SD) and compared using two-tailed t tests if they were normally distributed, while those with skewed distribution were described as median (interquartile range, IQR) and compared using Mann–Whitney U tests; categorical data were expressed as counts and percentages, and compared using Chi-squared tests. Additionally, receive operating characteristic (ROC) curves were used to investigate and compare the predictive capability of 3DE parameters for the detection of intermediate-high risk. Finally, univariate and multivariate Logistic regression analyses were performed to further identify independent variables associated with the intermediate-high risk stratification, and the results were expressed as odd ratio (OR) and 95% confidence interval (CI). Intra-class Correlation Coefficient (ICC) was used to assess inter-observer variability of 3DE measurements.
P < 0.05 was considered as statistically significant. SPSS software (version 19.0, IBM) and GraphPad Prim software (version 6.01) were used for analysis.
Results
As showed in Table 2, a total of 91 PAH patients (34 ± 12 years old, 25 males) were finally enrolled in our study. Approximately 98% of the patients shared WHO-FCI-III. Additionally, hemodynamic characteristics detected by RHC were available in 77 patients, and exercise capacity parameters were available in 81 patients. All participants received treatments as 2015ESC Guidelines recommended, and 74 (81.3%) of them took phosphodiesterase-5 inhibitors.
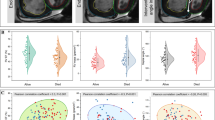
Based on the risk stratification mentioned above, 42 PAH patients were classified into low risk group, while 49 were into intermediate-high risk group. Table 3 described the demographics and 3DE characteristics of PAH patients in different risk groups. There were no significant differences in demographic characteristics between two risk groups. Compared with patients from low risk group, those from intermediate-high risk group shared significantly larger RV end-diastolic [117.6 ml (89.0, 136.5) ml vs 155.7 ml (108.5, 186.1) ml, P < 0.001] and end-systolic [85.3 ml (59.5, 94.2) ml vs 119.0 ml (80.1, 150.9) ml, P < 0.001] volumes, worse EF (30.35 ± 6.78% vs 22.45 ± 6.01, P < 0.001), decreased longitudinal strain either in septum [− 6.91% (− 8.97, − 5.56)% vs − 5.87% (− 6.94, − 4.50)%, P = 0.03] or in free wall [− 12.67% (− 15.78, − 9.56)% vs − 9.2% (− 11.22, − 6.43)%, P < 0.001], and its TAPSE tended to be worse [11.8 mm (9.5, 14.6) mm vs 10.3 mm (6.9, 12.8) mm, P = 0.05].
ROC curves (Fig. 3) illustrated that all the six 3DE parameters had the potential to predict intermediate-high risk stratification, especially the 3DE-RVEF (area under curve, AUC 0.82, 95% CI 0.73–0.91, P < 0.001), which showed the best predictive capacity. To be specific, EF < 26.39% had a 81.6% sensibility and 73.8% specificity to predict intermediate-high risk stratification.
Receiver operating characteristics curves and area under curves of 3DE-RV indices for prediction of intermediate-high risk stratification in PAH patients. PAH pulmonary arterial hypertension, RV right ventricular, EDV end-diastolic volume, ESV end-systolic volume, EF ejection fraction, LS longitudinal strain, S septum, FW free wall, TAPSE tricuspid annular plane systolic excursion, AUC area under curves
Table 4 showed the results of univariate and multivariate Logistic regression analyses for risk stratification. 3D-RV-EDV (OR 1.02, 95% CI 1.01–1.03, P = 0.002), 3D-RV-ESV (OR 1.03, 95% CI 1.01–1.04, P ≤ 0.001) and 3D-RVEF (OR 0.82, 95% CI 0.75–0.90, P < 0.001) were identified as independent predictors of intermediate-high risk stratification in PAH patients. After adjusted by age, gender and BMI, they still had the capacity to predict risk stratification independently. However, the predictive value of 3D-TAPSE (OR 1.00, 95% CI 0.95–1.05, P = 0.90) was not observed in our study, even after adjusted by age, gender, and BMI. With respect to strain analysis, LS(FW) (OR 1.17, 95% CI 1.05–1.31, P = 0.005) had the potential to predict intermediate-high risk stratification, while LS(S) (OR 1.05, 95% CI 0.94–1.18, P = 0.39) did not; the results remained the same after adjustment.
ICC for inter-observer variability were 0.88 (95% CI 0.70–0.94) for 3DE-RV-EDV, 0.91 (95% CI 0.74–0.97) for 3DE-RV-ESV, 0.91 (95% CI 0.75–0.97) for 3DE-RVEF, 0.72 (95% CI 0.55–0.94) for 3DE-RVLS(S), 0.70 (95% CI 0.52–0.92) for 3DE-RVLS(FW), 0.71 (95% CI 0.50–0.95) for 3DE-TAPSE, respectively.
Discussion
Several researches have focused on the value of 3DE in PH patients previously, however, to the best of our knowledge, this is the first time to investigate the value of 3DE parameters for predicting the comprehensive risk stratification in PAH patients. We found that RV volumes, EF and free wall strain detected by 3DE had the potential to independently predict intermediate-high risk stratification in PAH patients, and the predictive capacity retained after adjusted by demographic data. However, the predictive value of 3DE detected TAPSE and septum strain were not observed in our study.
The current risk assessment strategy is recommended by 2015 ESC Guidelines [2]. The most obvious problem is its complexity, it includes indices of symptoms, clinical signs, exercise capacity, imaging and haemodynamics. The symptoms and WHO-FC have certain subjectivity; both 6WMT and CPET have some contraindications, such as leg disease, or intolerance in critically ill patients; moreover, haemodynamics detected by RHC is an invasive examanition which may expose patients to operational risks. Hence, we have dedicated to explore easier and safer ways to evaluate disease severity in PH patients. We previously illustrated that 2DE-RV global strain and strain rate had predictive capacity for high risk assessed using PVO2 in pre-capillary PH patients [13]. In the present study, we focused on the 3DE RV parameters, and found their predictive capacity for intermediate-high risk stratification in PAH patients.
It is well established that RV remodeling due to pressure and volume overload includes RV dilatation and hypertrophy [14], which subsequently result in RV failure. Ultimately, RV failure is the primary cause of death in PAH patients [15]. RVEF is one of the most important indices to evaluate RV function, but it is very difficult to measure by traditional two-dimensional echocardiography (2DE). A previous study reported that 3DE-RVEF moderately correlated (Pearson correlation coefficient ranged from − 0.51 to − 0.64) with hemodynamic indices detected by RHC, moreover, compared with other 3DE parameters, 3D-RVEF had the highest area under the curve (0.89, P < 0.05) in detecting hemodynamic signs of RV failure (a composite of Ci < 2 L/min/m2 and RA P > 15 mmHg) [16]. Another study suggested that RVEF measured by cardiac magnetic resonance imaging (CMRI) was a better predictor of mortality than PVR (area under the ROC curve: 0.749 vs 0.628) [15]. Similar results were reported by Murata, in whose study, receiver operating characteristic analysis revealed that 3DE-RVEF had a greater AUC (0.78) than other hemodynamic measurements, such as mPAP, in predicting cardiac events including death, hospitalization, and intervention [17]. In the present study, we first illustrated that 3DE-RVEF had predictive value in detecting intermediate-high risk stratification in PAH patients (OR 1.02, 95% CI 1.01–1.03, P = 0.002). Furthermore, 3DE-RVEF showed the best (AUC = 0.82, 95% CI 0.73–0.91, P < 0.001) predictive capacity among all of the 3DE parameters.
The accurate measurement of RV volumes by 3DE in PH patients has been validated by CMRI [18, 19], which was considered as the “gold-standard” technique for quantification of RV volume. A previous study showed that PH patients with RV mass/volume ratio > 0.46 had more suitable systolic function and mechanical efficiency than those with RV mass/volume ratio ≤ 0.46, suggesting that an increased RV volume might be a symptom of maladaptation. This is one of the possible explanations for that the increased RV volume was closely associated with disease severity in PH patients in our study. In the report by Keiko Ryo, PH patients were divided into RV adapted, adapted-remodeled and adverse-remodeled groups by the cutoffs of RV-3D-ESV index (ESVi), and Cox proportional hazards regression analysis identified RV adverse-remodeled patients (ESVi ≥ 114 mL/m2) as an independent predictor for clinical outcome (hazard ratio, HR 2.2; 95% CI, 0.91 to 5.39; P = 0.04) [8]. Similar results were reported by Pei-Ni Jone recently [20].
As an angle-independent sensitive measure of global and regional myocardial contractility, strain analysis has the advantage of distinguishing true contractility of RV better than translational motion of tricuspid annulus [21]. With regard to 3D longitudinal strain, we found that rather than RVLS(S) (OR 1.05, 95% CI 0.94–1.18, P = 0.39) and TAPSE (OR 1.00, 95% CI 0.95–1.05, P = 0.90), 3D-RVLS(FW) (OR 1.17, 95% CI 1.05–1.31, P = 0.005) had the potential to independently predict intermediate-high risk stratification in PAH patients. Several previous studies focusing on 2DE RV global or free wall strain supported our results, illustrating correlations between RV strain and 6MWD, PVO2, NT-proBNP, hemodynamic characteristics detected by RHC, clinical deterioration and mortality in PH patients [13, 22, 23]. Study by Benjamin Smith also showed that 3DE-RVLS of free wall (HR 7.63, 95% CI 1.76 -10.27, P < 0.001) was a significant determinant of all-cause mortality [7].
In clinical practice, as a safe and convenient approach, 3DE may contribute to risk assessment in PAH patients, which solved the limitations and use difficulties of current risk assessment strategy recommended by 2015 ESC Guidelines [2]. In the future, we are looking forward that 3DE could be a routine examination to evaluate disease severity of PH patients in clinical practice, especially for patients with contraindications to RHC or intolerance to CPET.
Limitations
We acknowledged several limitations of our study. First, as this is a single center study, a referral bias is difficult to avoid. However, we shared a relatively larger sample size among similar studies. Second, for part of the participants, the hemodynamic parameters were not used for risk stratification because RHC were not performed simultaneously with 3DE. But this may have little influence on our results because we used a mean score of available indices for risk stratification. Additionally, imaging parameters, such as RA area, were not used in the risk stratification in our study, because there are limitations to the quantification of RA size by 2-dimensional echocardiography [10], and the simplified risk assessment tools without RA area were shown to have prognostic value [11, 12]. Finally, in the future, it still needs further investigation to compare the predictive capacity between 2 and 3DE parameters for disease severity in PH patients.
Conclusions
In conclusion, RV volumes, EF and free wall strain detected by 3DE were independent predictors for intermediate-high risk stratification in PAH patients, among which, RVEF showed the best predictive capacity.
References
Humbert M, Sitbon O, Chaouat A, Bertocchi M, Habib G, Gressin V, Yaici A, Weitzenblum E, Cordier JF, Chabot F, Dromer C, Pison C, Reynaud-Gaubert M, Haloun A, Laurent M, Hachulla E, Cottin V, Degano B, Jais X, Montani D, Souza R, Simonneau G (2010) Survival in patients with idiopathic, familial, and anorexigen-associated pulmonary arterial hypertension in the modern management era. Circulation 122:156–163. https://doi.org/10.1161/CIRCULATIONAHA.109.911818
Galie N, Humbert M, Vachiery JL, Gibbs S, Lang I, Torbicki A, Simonneau G, Peacock A, Vonk NA, Beghetti M, Ghofrani A, Gomez SM, Hansmann G, Klepetko W, Lancellotti P, Matucci M, McDonagh T, Pierard LA, Trindade PT, Zompatori M, Hoeper M (2015) 2015 ESC/ERS Guidelines for the diagnosis and treatment of pulmonary hypertension: The Joint Task Force for the Diagnosis and Treatment of Pulmonary Hypertension of the European Society of Cardiology (ESC) and the European Respiratory Society (ERS): Endorsed by: Association for European Paediatric and Congenital Cardiology (AEPC), International Society for Heart and Lung Transplantation (ISHLT). Eur Respir J 46:903–975. https://doi.org/10.1183/13993003.01032-2015
Shimada E, Zhu M, Kimura S, Streiff C, Houle H, Datta S, Sahn DJ, Ashraf M (2015) Quantitative assessment of mitral inflow and aortic outflow stroke volumes by 3-dimensional real-time full-volume color flow doppler transthoracic echocardiography: an in vivo study. J Ultrasound Med 34:95–103. https://doi.org/10.7863/ultra.34.1.95
Marsan NA, Maffessanti F, Tamborini G, Gripari P, Caiani E, Fusini L, Muratori M, Zanobini M, Alamanni F, Pepi M (2011) Left atrial reverse remodeling and functional improvement after mitral valve repair in degenerative mitral regurgitation: a real-time 3-dimensional echocardiography study. Am Heart J 161:314–321. https://doi.org/10.1016/j.ahj.2010.10.029
Leary PJ, Kurtz CE, Hough CL, Waiss MP, Ralph DD, Sheehan FH (2012) Three-dimensional analysis of right ventricular shape and function in pulmonary hypertension. Pulm Circ 2:34–40. https://doi.org/10.4103/2045-8932.94828
Kong D, Shu X, Pan C, Cheng L, Dong L, Yao H, Zhou D (2012) Evaluation of right ventricular regional volume and systolic function in patients with pulmonary arterial hypertension using three-dimensional echocardiography. Echocardiography 29:706–712. https://doi.org/10.1111/j.1540-8175.2012.01681.x
Smith BC, Dobson G, Dawson D, Charalampopoulos A, Grapsa J, Nihoyannopoulos P (2014) Three-dimensional speckle tracking of the right ventricle: toward optimal quantification of right ventricular dysfunction in pulmonary hypertension. J Am Coll Cardiol 64:41–51. https://doi.org/10.1016/j.jacc.2014.01.084
Ryo K, Goda A, Onishi T, Delgado-Montero A, Tayal B, Champion HC, Simon MA, Mathier MA, Gladwin MT, Gorcsan JR (2015) Characterization of right ventricular remodeling in pulmonary hypertension associated with patient outcomes by 3-dimensional wall motion tracking echocardiography. Circ Cardiovasc Imaging. https://doi.org/10.1161/CIRCIMAGING.114.003176
Moceri P, Duchateau N, Baudouy D, Schouver ED, Leroy S, Squara F, Ferrari E, Sermesant M (2018) Three-dimensional right-ventricular regional deformation and survival in pulmonary hypertension. Eur Heart J Cardiovasc Imaging 19:450–458. https://doi.org/10.1093/ehjci/jex163
Rudski LG, Lai WW, Afilalo J, Hua L, Handschumacher MD, Chandrasekaran K, Solomon SD, Louie EK, Schiller NB (2010) Guidelines for the echocardiographic assessment of the right heart in adults: a report from the American Society of Echocardiography endorsed by the European Association of Echocardiography, a registered branch of the European Society of Cardiology, and the Canadian Society of Echocardiography. J Am Soc Echocardiogr 23(685–713):786–788. https://doi.org/10.1016/j.echo.2010.05.010
Hoeper MM, Kramer T, Pan Z, Eichstaedt CA, Spiesshoefer J, Benjamin N, Olsson KM, Meyer K, Vizza CD, Vonk-Noordegraaf A, Distler O, Opitz C, Gibbs J, Delcroix M, Ghofrani HA, Huscher D, Pittrow D, Rosenkranz S, Grunig E (2017) Mortality in pulmonary arterial hypertension: prediction by the 2015 European pulmonary hypertension guidelines risk stratification model. Eur Respir J. https://doi.org/10.1183/13993003.00740-2017
Kylhammar D, Kjellstrom B, Hjalmarsson C, Jansson K, Nisell M, Soderberg S, Wikstrom G, Radegran G (2018) A comprehensive risk stratification at early follow-up determines prognosis in pulmonary arterial hypertension. Eur Heart J 39:4175–4181. https://doi.org/10.1093/eurheartj/ehx257
Liu BY, Wu WC, Zeng QX, Liu ZH, Niu LL, Tian Y, Cheng XL, Luo Q, Zhao ZH, Huang L, Wang H, He JG, Xiong CM (2019) Two-dimensional speckle tracking echocardiography assessed right ventricular function and exercise capacity in pre-capillary pulmonary hypertension. Int J Cardiovasc Imaging. 35:1499–1508. https://doi.org/10.1007/s10554-019-01605-w
Mertens LL, Friedberg MK (2010) Imaging the right ventricle–current state of the art. Nat Rev Cardiol 7:551–563. https://doi.org/10.1038/nrcardio.2010.118
van de Veerdonk MC, Kind T, Marcus JT, Mauritz GJ, Heymans MW, Bogaard HJ, Boonstra A, Marques KM, Westerhof N, Vonk-Noordegraaf A (2011) Progressive right ventricular dysfunction in patients with pulmonary arterial hypertension responding to therapy. J Am Coll Cardiol 58:2511–2519. https://doi.org/10.1016/j.jacc.2011.06.068
Vitarelli A, Mangieri E, Terzano C, Gaudio C, Salsano F, Rosato E, Capotosto L, D'Orazio S, Azzano A, Truscelli G, Cocco N, Ashurov R (2015) Three-dimensional echocardiography and 2D–3D speckle-tracking imaging in chronic pulmonary hypertension: diagnostic accuracy in detecting hemodynamic signs of right ventricular (RV) failure. J Am Heart Assoc 4:e1584. https://doi.org/10.1161/JAHA.114.001584
Murata M, Tsugu T, Kawakami T, Kataoka M, Minakata Y, Endo J, Tsuruta H, Itabashi Y, Maekawa Y, Murata M, Fukuda K (2016) Prognostic value of three-dimensional echocardiographic right ventricular ejection fraction in patients with pulmonary arterial hypertension. Oncotarget 7:86781–86790. https://doi.org/10.18632/oncotarget.13505
Zhang QB, Sun JP, Gao RF, Lee AP, Feng YL, Liu XR, Sheng W, Liu F, Yang XS, Fang F, Yu CM (2013) Feasibility of single-beat full-volume capture real-time three-dimensional echocardiography for quantification of right ventricular volume: validation by cardiac magnetic resonance imaging. Int J Cardiol 168:3991–3995. https://doi.org/10.1016/j.ijcard.2013.06.088
Badagliacca R, Poscia R, Pezzuto B, Nocioni M, Mezzapesa M, Francone M, Giannetta E, Papa S, Gambardella C, Sciomer S, Volterrani M, Fedele F, Dario VC (2015) Right ventricular remodeling in idiopathic pulmonary arterial hypertension: adaptive versus maladaptive morphology. J Heart Lung Transplant 34:395–403. https://doi.org/10.1016/j.healun.2014.11.002
Jone PN, Schafer M, Pan Z, Bremen C, Ivy DD (2018) 3D echocardiographic evaluation of right ventricular function and strain: a prognostic study in paediatric pulmonary hypertension. Eur Heart J Cardiovasc Imaging 19:1026–1033. https://doi.org/10.1093/ehjci/jex205
Giusca S, Dambrauskaite V, Scheurwegs C, D'Hooge J, Claus P, Herbots L, Magro M, Rademakers F, Meyns B, Delcroix M, Voigt JU (2010) Deformation imaging describes right ventricular function better than longitudinal displacement of the tricuspid ring. Heart 96:281–288. https://doi.org/10.1136/hrt.2009.171728
Hardegree EL, Sachdev A, Villarraga HR, Frantz RP, McGoon MD, Kushwaha SS, Hsiao JF, McCully RB, Oh JK, Pellikka PA, Kane GC (2013) Role of serial quantitative assessment of right ventricular function by strain in pulmonary arterial hypertension. Am J Cardiol 111:143–148. https://doi.org/10.1016/j.amjcard.2012.08.061
Sachdev A, Villarraga HR, Frantz RP, McGoon MD, Hsiao JF, Maalouf JF, Ammash NM, McCully RB, Miller FA, Pellikka PA, Oh JK, Kane GC (2011) Right ventricular strain for prediction of survival in patients with pulmonary arterial hypertension. Chest 139:1299–1309. https://doi.org/10.1378/chest.10-2015
Acknowledgements
The present study was supported by the grant of Capital Health Development and Scientific Research Projects (Grant No. 2016-2-4036), CAMS Initiative for Innovative Medical (Grant No. 2016-I2 M-3-006) and PUMC Innovation fund for graduate students (Grant No. 2018-1002-01-18).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Liu, BY., Wu, WC., Zeng, QX. et al. The value of three-dimensional echocardiography in risk stratification in pulmonary arterial hypertension: a cross-sectional study. Int J Cardiovasc Imaging 36, 577–584 (2020). https://doi.org/10.1007/s10554-019-01743-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10554-019-01743-1