Abstract
To determine the prognostic value of a new, ultrafast, low dose myocardial perfusion SPECT (MPS) protocol in a cadmium-zinc telluride (CZT) camera. CZT cameras have introduced significant progress in MPS imaging, offering high-quality images despite lower doses and scan time. Yet, it is unknown if, with such protocol changes, the prognostic value of MPS is preserved. Patients had a 1-day 99 m-Tc-sestamibi protocol, starting with the rest (185–222 MBq) followed by stress (666–740 MBq). Acquisition times were 6 and 3 min, respectively. MPS were classified as normal or abnormal perfusion scans and summed scores of stress, rest, and difference (SSS, SRS and SDS), calculated. Patients were followed with 6-month phone calls. Hard events were defined as death or nonfatal myocardial infarction. Late revascularization was that occurring after 60 days of MPS. 2930 patients (age 64.0 ± 12.1 years, 53.3% male) were followed for 30.7 ± 7.5 months. Mean dosimetry was 6 mSv and mean total study time, 48 ± 13 min. The annual hard event and late revascularization rate were higher in patients with greater extension of defect and ischemia. SSS was higher in patients with hard events compared to those without events (2.6 ± 4.9 vs. 5.0 ± 6.3, p < 0.001), as well as the SDS (0.7 ± 1.9 vs. 1.7 ± 3.4, p < 0.00). The same was true for patients with or without late revascularization (SSS: 2.5 ± 4.7 vs. 6.6 ± 7.1; SDS: 0.6 ± 1.7 vs. 2.9 ± 3.8, p < 0.01). A new, faster, low-radiation, MPS protocol in a CZT camera maintain the ability to stratify patients with increased risk of events, showing that, in the presence of greater extension of defect or ischemia, patients presented higher rates of hard events and late revascularization.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Over the past 30 years, successive technical innovations- including, but not limited to, SPECT acquisition, ECG gating, among others- have granted myocardial perfusion imaging the status of a reliable, widely applicable, and increasingly useful technique [1]. The excellent diagnostic and prognostic values of myocardial perfusion SPECT (MPS) have led it to be extensively employed to evaluate patients with suspected or known coronary artery disease (CAD) [2].
Nonetheless, traditional MPS has two important limitations: Prolonged image acquisition time, leading to long procedural times, and relatively large radiation doses. The available literature demonstrates the possibility of high-speed cameras to reduce acquisition times, improving patient’s tolerance to the test, and reducing radiation dose [3]. These new cameras rely on a pinhole collimation design and multiple cadmium zinc telluride (CZT) crystal arrays. Compared to the traditional SPECT camera, this type of collimation provides a three- to five-fold increase in photon sensitivity, thereby reducing imaging times significantly, while providing a 1.7 to 2.5-fold increase in spatial resolution. This makes shorter scans or lower doses (or even both) a reality, without the loss of image quality [3,4,5].
However, the prognostic value of MPS imaging with this new protocol is still unknown. This study therefore sought to assess the prognostic value of a new faster, low-radiation MPS protocol performed in a CZT gamma camera (CZT-GC).
Materials and methods
Consecutive patients who underwent CZT MPS for the assessment of suspected or known CAD at a single laboratory in Rio de Janeiro, Brazil, between November 2011 and December 2012 were prospectively enrolled and followed by 6-month phone calls.
Those who underwent myocardial revascularization (either by coronary angioplasty or coronary artery by pass grafting surgery) <60 days after MPS were later excluded.
Prior to scanning, patient´s medical history and physical examination data were collected by a team of experienced cardiologists.
All procedures performed were in accordance with the ethical standards of the institutional research committee and with the 1964 Declaration of Helsinki and its later amendments. Informed consent was obtained from all individual participants included in the study.
Study protocol
The patients were instructed to abstain from any products containing caffeine for 24 h before the test. Beta-blockers, calcium-channel antagonists, and nitrates were terminated 48 h before testing. A 1-day protocol was employed, with 185–222 MBq of 99 m-Tc-sestamibi used for the resting phase and 666–740 MBq for stress. Initially, to determine the best duration for the acquisition of the MPS scan, 24 patients (13 men) were selected for a pilot study in which scan acquisition was performed for 6 min in list mode. The scans were then processed using 1–6 min of the total scan time for reconstruction, and images were analyzed by two experienced readers unaware of the time range used for reconstruction, who had their readings evaluated for agreement. The study protocol was then defined according to best combination of reading agreements for stress and rest MPS studies among time ranges.
All patients underwent a 1-day, gated, rest/stress 99 m-Tc-sestamibi protocol. 10 min after tracer injection, image acquisition was performed in the supine position. The second phase was the stress study, in which either symptom-limited exercise treadmill test using the standard Bruce protocol with 13-lead electrocardiographic or pharmacologic stress were performed. Upon 5 min of stress phase completion, patients underwent image acquisition in the supine and prone positions [6, 7]. The CZT-GC (Discovery NM 530c, GE Healthcare, Haifa, Israel) was equipped with a multiple pinhole collimator and 19 stationary cadmium-zinc-telluride detectors simultaneously imaging 19 cardiac views. Each detector contained 32 × 32 pixelated 5-mm thick (2.46 × 2.46 mm) elements. The system design enabled high-quality imaging of a three-dimensional volume by all detectors (quality field-of-view), where the patient’s heart should be positioned. Once acquisition was initiated, no detector or collimator motion occurred.
Image analysis
All images were interpreted by a consensus of two experienced readers. Image processing was performed using Evolution for Cardiac® software. Images were reconstructed without scatter or attenuation correction. Short-axis, vertical and horizontal long-axis tomograms, as well as polar maps, were generated and analyzed. The image reconstruction method used allows extra-cardiac activity to be isolated more easily. Still, two readers analyzed the image before the patient was removed from the camera. The repetition rate of the images was less than 5%. A semi quantitative 17-segment visual interpretation of the gated myocardial perfusion images was performed [8]. Each segment was scored by consensus of the two observers using a standard five-point scoring system [9] (0 = normal, 1 = equivocal, 2 = moderate, 3 = severe reduction of uptake, and 4 = absence of detectable tracer uptake). Summed stress scores (SSS) were obtained by adding the scores of the 17 segments of the stress images. Summed rest scores (SRS) were obtained by adding scores of the 17 segments of the rest images and a summed difference score (SDS) was calculated by segmental subtraction (SSS-SRS). For evaluating the SSS and the SDS as predictors of events, we performed separate analyses with different cut points for each perfusion variable. We created four groups o SSS and SDS with the purpose of evaluating the prognostic value and the stratification power of this new protocol on a CZT-GC based not only in positive or negative MPI results, but specially, based on the extension of defect and ischemia, which is widely stablished to be achieved by using this type of classification [10, 11]. A normal study was considered when SSS < 3 and SDS < 1.
Post-stress eight frames gated short-axis images were processed using quantitative gated SPECT software (Cedars-Sinai Medical Center, Los Angeles, California), and left ventricular ejection fraction (LVEF), end-systolic and end-diastolic volumes (ESV and EDV, respectively) were automatically calculated.
Follow-up
Follow-up was performed by telephone interview every 6 months after MPI. All-cause death, nonfatal myocardial infarction, or late revascularization (>60 days after MPS) were registered. Evaluation of hospital records and/or review of civil registries confirmed these events. Nonfatal myocardial infarction was defined based on the criteria of typical chest pain, elevated cardiac enzyme levels and typical alterations of the electrocardiogram [12]. Death or nonfatal myocardial infarction were classified as hard events. Late revascularization was studied separately.
Statistical analyses
Categorical variables are presented as frequencies and continuous variables as mean ± SD. The annual event rate was calculated as the % of events divided by person-years, and was compared among groups using the log-rank test. Kaplan–Meier curves were generated to visually assess survival in different groups. A Cox proportional hazards analysis was done to evaluate predictors of hard events and late revascularization, using variables with p value <0.05 in univariable analysis or clinical relevance.
Analyses were performed with SPSS software, version 17.0. A p value <0.05 was considered significant.
Results
Among 3265 patients, 235 were excluded due to early revascularization, and 100 were lost to follow-up, leaving 2930 patients who were followed for 30.7 ± 7.5 months. Mean age was 64.0 ± 12.1 years and 53.5% were male. Among these patients, 2072 (70.7%) were asymptomatic. The most frequent indications in asymptomatic patients were a previous treadmill test with intermediate-high risk Duke score, pre-op risk stratification and a previous calcium score >100. The most prevalent risk factor for CAD was hypertension (61.6%), followed by hypercholesterolemia (52.2%), smoking (36.4%) and family history of CAD (31.2%). Diabetes was present in 22.7% and previous myocardial infarction in 12.5%. From the 2930 patients, 501 (17.1%) had already been submitted to coronary angioplasty and 222 (7.6%) to coronary artery bypass grafting (CABG). About regularly medications, 37.9% was in use of ACE inhibitors, 28.6% was using beta-blocker and 10.9% Calcium receptor blockers. Mean perfusion scores were overall low and mean LVEF was normal with only 6% of patients with a LVEF <40%. These characteristics are summarized in Table 1.
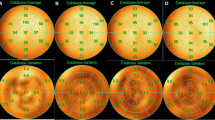
For the definition of optimal scan time, acquired scans were processed using 1, 2, 3, 4, 5 or 6 min of the total list-mode acquisition previously performed for the pilot study. Intra and inter-observer agreement rates of MPS readings among time ranges are shown in Table 2. Based on those, the best combination, which was chosen for the subsequent MPS studies, was 6 min for the rest acquisition, 3 min for post-stress supine acquisition and 1.5 min for post-stress prone acquisition. Figure 1 shows an example of six post-stress images processed using a 6, 5, 4, 3, 2 and 1 min acquisition with a progressive degradation of image quality as we reduced the acquisition time. Considering all MPS studies performed thereafter, mean radiation dose was 6 mSv and mean scan time was 48 ± 13 min.
During follow-up, there were 61 deaths, 29 nonfatal infarctions (90 hard events), 148 coronary angioplasty procedures and 22 bypass surgeries (170 late revascularization procedures). Table 3 shows the comparison between patients with or without hard events. The former were older, more frequently male, with prior myocardial infarction or prior CABG and less frequently able to perform exercise stress. Perfusion scores were higher, as well as left ventricular volumes, and LVEF was lower in patients with hard events. In Table 4, comparisons between patients with or without late revascularization are shown. Similarly to patients with hard events, those with late revascularization were older, more frequently male, with cardiovascular risk factors, known coronary artery disease, and less frequently able to perform exercise stress, with higher perfusion scores, left ventricular volumes, and lower LVEF.
Based on the SSS, 4 patient groups were created: group 1 with SSS values from 0 to 2 [n = 2098 (71.6%)]; group 2, SSS values from 3 to 5 [n = 430 (14.6%)]; group 3, SSS values from 6 to 11 [n = 220 (7.5%)] and group 4, SSS ≥ 12 [n = 182 (6.2%)]. Kaplan–Meier survival curves (Fig. 2a, b) showed that the highest the SSS, the lowest the survival free of hard events (p < 0.001) or late revascularization (p < 0.001), respectively. Table 5 shows the annualized hard events and late revascularization rates for individual groups of SSS, with highest rates in the group of highest SSS values. Four groups were also created according to SDS values: group 1 with SDS = 0 [n = 2334 (79.6.0%)]; group 2, SDS values from 1 to 2 [n = 305 (10.4%)]; group 3, SDS from 3 to 5 [n = 171 (5.8%)] and group 4, SDS ≥ 6 [n = 120 (4.0%)]. Kaplan–Meier survival curves also showed that, with increasing SDS, event-free survival was reduced, both for hard events (Fig. 3a) (p < 0.001) or late revascularization (Fig. 3b) (p < 0.001). Also, the annualized events rates, both for hard events or late revascularization, showed the same behavior presented on SSS groups for SDS groups, with higher rates in groups with higher values of SDS (Table 6). For all Kaplan Meier survival curves, p values were demonstrated in a pairwise comparison, below the graphs, to express the statistical differences in events rates between individual groups.
Finally, in the Cox analysis, male gender (hazard ratio = 2.26 [1.33–3.8], p = 0.03), age (hazard ratio = 1.03 [1.01–1.06], p = 0.001), diabetes (hazard ratio = 1.69 [1.09–2.62], p = 0.02), pharmacologic stress (hazard ratio = 2.91 [1.75–4.8], p < 0.001) and the SSS (hazard ratio = 1.36 [1.07–1.73], p = 0.01). were independently associated with hard events. SDS, when substituting SSS in the cox analysis, also showed to be an independent predictor of hard events (hazard ratio = 1.09 [1.02–1.17], p = 0.01) and maintained the results of the other variables. Table 3 shows all the variables used in the cox regression for hard events, with their respective hazard ratio and confidence interval.
For late revascularization, diabetes (hazard ratio = 1.94 [1.41–2.66], p < 0.001), prior percutaneous coronary intervention (hazard ratio = 2.54 [1.76–3.68], p < 0.001), and the SSS (hazard ratio = 1.06 [1.04–1.08], p < 0.001) were the independent predictors. Again, SDS also showed to be an independent predictor of late revascularization (hazard ratio = 1.22 [1.17–1.26], p < 0.001) when analyzed substituting the SSS with no modification in the other variables. Table 4 shows the variables used for this cox regression, with their respective hazard ratio and confidence interval.
Discussion
Previous studies have shown that CZT cameras are able to perform ultrafast and low-dose MPS studies, with even higher sensitivity and image quality when compared to traditional cameras [13]. However, there is still incomplete evidence supporting the prognostic value of MPS performed in CZT cameras. This study shows, in a large patient population, that the prognostic value of a new MPS protocol in a high-speed CZT-GC could be comparable to what literature has shown about the prognostic value traditionally provided by conventional MPS [9]. Our group had already demonstrated the prognostic value of MPS with a new reconstruction algorithm [11] in traditional Anger cameras, which also allowed faster scans. However, with the advent of CZT technology, it became imperative to define if these new cameras would provide MPS studies with reliable prognostic value, which might be reliably used to manage patients with suspected or known CAD.
Dolan et al. demonstrated the prognostic value of MPS in a CZT camera, but as these authors recognized, they used the conventional dose of radiotracers [14]. The radiation dose used in this study was considerably lower than standard dose used in traditional protocols, and we initially tested different acquisition times to obtain the best possible images with low radiation. Total procedure time was reduced to less than one hour, with imaging time of 6 min for rest and 3 min for stress phase.
After establishing these parameters, we then studied the prognostic value of this protocol. Male gender, increasing age, the use of pharmacologic stress were significant predictors of hard events, as previously described [15, 16]. Diabetes was independently associated both with hard events or late revascularization. Of note, LVEF was not associated with events, what might be explained by the overall normal left ventricular function of the study population. Importantly, the extent and intensity of myocardial ischemia, as expressed by the SDS, was significantly associated with outcomes, what supports the prognostic value of this new protocol.
It is worth noting the characteristics of the study population, composed of outpatients, most asymptomatic (performing MPS as part of a preoperative evaluation or general screening due to cardiac risk factors) with normal left ventricular function. Nonetheless, the prevalence of diabetes was >20%, and over 10% had a history of myocardial infarction, what increases overall risk and may improve the generalizability of these results. Therefore, we believe that CZT MPS may be reliably used to evaluate patients for CAD, with the advantages of reduced imaging time and lower radiation dose.
We recognize, as a limitation, that the best method to establish the prognostic value of this new protocol in a CZT-GC would be a comparison between new and traditional cameras, with each patient being studied in both cameras and being control for themselves. However, it assumes that the same protocol would be used for both cameras [14]. Since the new low radiation dose and acquisition time protocol could not be used for traditional cameras, the study would not verify the protocol that this study aim to establish.
Conclusion
A new, faster, low-radiation, MPS protocol in a CZT camera was able to maintain the ability of stratifying patients with increased risk of events, showing that, in the presence of greater extension of defect or ischemia, patients presented higher rates of hard events and late revascularization.
References
Verberne HJ, Acampa W, Anagnostopoulos C, Ballinger J, Bengel F, De Bondt P et al (2015) EANM procedural guidelines for radionuclide myocardial perfusion imaging with SPECT and SPECT/CT: 2015 revision. Eur J Nucl Med Mol Imaging 42(12):1929–1940
Hansen CL, Goldstein RA, Akinboboye OO, Berman DS, Botvinick EH, Churchwell KB et al (2007) Myocardial perfusion and function: single photon emission computed tomography. J Nucl Cardiol 14(6):e39–e60
Oddstig J, Hedeer F, Jögi J, Carlsson M, Hindorf C, Engblom H (2013) Reduced administered activity, reduced acquisition time, and preserved image quality for the new CZT camera. J Nucl Cardiol 20(1):38–44
Duvall WL, Sweeny JM, Croft LB, Ginsberg E, Guma KA, Henzlova MJ (2012) Reduced stress dose with rapid acquisition CZT SPECT MPI in a non-obese clinical population: comparison to coronary angiography. J Nucl Cardiol 19(1):19–27
Mouden M, Timmer JR, Ottervanger JP, Reiffers S, Oostdijk AH, Knollema S et al (2012) Impact of a new ultrafast CZT SPECT camera for myocardial perfusion imaging: fewer equivocal results and lower radiation dose. Eur J Nucl Med Mol Imaging 39(6):1048–1055
Hayes SW, De Lorenzo A, Hachamovitch R, Dhar SC, Hsu P, Cohen I et al (2003) Prognostic implications of combined prone and supine acquisitions in patients with equivocal or abnormal supine myocardial perfusion SPECT. J Nucl Med 44(10):1633–1640
Burrell S, MacDonald A (2006) Artifacts and pitfalls in myocardial perfusion imaging. J Nucl Med Technol 34(4):193–211 (quiz 2–4)
Berman DS, Kang X, Van Train KF, Lewin HC, Cohen I, Areeda J et al (1998) Comparative prognostic value of automatic quantitative analysis versus semiquantitative visual analysis of exercise myocardial perfusion single-photon emission computed tomography. J Am Coll Cardiol 32(7):1987–1995
Berman DS, Hachamovitch R, Kiat H, Cohen I, Cabico JA, Wang FP et al (1995) Incremental value of prognostic testing in patients with known or suspected ischemic heart disease: a basis for optimal utilization of exercise technetium-99m sestamibi myocardial perfusion single-photon emission computed tomography. J Am Coll Cardiol 26(3):639–647
Hachamovitch R, Berman DS, Shaw LJ, Kiat H, Cohen I, Cabico JA et al (1998) Incremental prognostic value of myocardial perfusion single photon emission computed tomography for the prediction of cardiac death: differential stratification for risk of cardiac death and myocardial infarction. Circulation 97(6):535–543
Lima R, Ronaldo L, De Lorenzo A, Andrea DL, Camargo G, Gabriel C et al (2014) Prognostic value of myocardium perfusion imaging with a new reconstruction algorithm. J Nucl Cardiol 21(1):149–157
Thygesen K, Alpert JS, White HD, Joint ESC/ACCF/AHA/WHF Task Force for the Redefinition of Myocardial Infarction (2007) Universal definition of myocardial infarction. J Am Coll Cardiol 50(22):2173–2195
Duvall WL, Croft LB, Ginsberg ES, Einstein AJ, Guma KA, George T et al (2011) Reduced isotope dose and imaging time with a high-efficiency CZT SPECT camera. J Nucl Cardiol 18(5):847–857
Oldan JD, Shaw LK, Hofmann P, Phelan M, Nelson J, Pagnanelli R et al (2016) Prognostic value of the cadmium-zinc-telluride camera: a comparison with a conventional (Anger) camera. J Nucl Cardiol 23(6):1280–1287
Hachamovitch R, Berman DS, Kiat H, Bairey CN, Cohen I, Cabico A et al (1996) Effective risk stratification using exercise myocardial perfusion SPECT in women: gender-related differences in prognostic nuclear testing. J Am Coll Cardiol 28(1):34–44
Navare SM, Mather JF, Shaw LJ, Fowler MS, Heller GV (2004) Comparison of risk stratification with pharmacologic and exercise stress myocardial perfusion imaging: a meta-analysis. J Nucl Cardiol 11(5):551–561
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Lima, R.S.L., Peclat, T.R., Souza, A.C.A.H. et al. Prognostic value of a faster, low-radiation myocardial perfusion SPECT protocol in a CZT camera. Int J Cardiovasc Imaging 33, 2049–2056 (2017). https://doi.org/10.1007/s10554-017-1202-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10554-017-1202-3