Abstract
Purpose
There is limited information about the dietary habits associated with stomach adenocarcinoma in the Brazilian population, so our purpose is to analyze the consumption of processed and ultra-processed foods by patients with stomach adenocarcinoma in Brazil.
Methods
A multicentric hospital-based case–control study was conducted in São Paulo (southeastern region) and Belém (Amazon region) of Brazil with 1,045 individuals, both sexes, between 18 and 75 years old. In São Paulo, there were 214 cases with stomach adenocarcinoma and 150 controls patients submitted to stomach endoscopy named as Group I (without any pre-malignant gastric disease) and the Healthy Controls (Group 2) comprised 401 individuals matched by age and sex from the prevention unit at A.C .Camargo Cancer Center. In Belém, it has two groups one are cases 140 and second 140 hospital controls, recruited in outpatient clinics. Lifestyle and food frequency questionnaires (FFQ) were administered in cases and controls in both places. Univariate and multivariable binomial logistic regression analyses were performed.
Results
In São Paulo, cases reported two times greater consumption of processed meat (adjusted OR 2.56, 95% CI 1.32–4.96) and of sweets (≥ 80 g/day) than Group 1 (endoscopic controls) (adjusted OR 2.25, 95% CI 1.21–4.18). Compared with Group 2, processed food consumption (≥ 44 g/day) as well as ≥ 44 g/day of salted bread increased the odds of having stomach adenocarcinoma (adjusted OR 2.96, 95% CI 1.82–4.81 and adjusted OR 2.03, 95% CI 1.30–3.18), respectively. In Belém, individuals who reported consuming ≥ 166 g/day of fried and roasted meat and fish were more likely to have stomach adenocarcinoma (adjusted OR 2.21, 95% CI 1.13–4.30).
Conclusions
In both cities, consumption of processed and ultra-processed foods, especially salted bread, yellow cheese, fried and roasted meats, fish fried, processed meat, and sweets, was independently associated with the chance of having stomach adenocarcinoma.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Stomach cancer is the fifth most common type of cancer in the world and the fourth leading cause of cancer-related death [1]. In Brazil, the estimated number of new stomach cancer cases for each year of the triennium 2020–2022 is 21,230, with age-standardized incidence rates of 12.8/100,000 for men and 7.3/100,000 for women [1, 2]. Although the stomach cancer mortality rate in Brazil decreased from 1996 to 2012, it remains high and differs among geographic regions of the country. The incidence rate has followed the global decreasing trend in São Paulo, whereas, in Belém, it was stable in men and increased in women in the period 1996–2010 [3,4,5,6].
Various risk factors, including Helicobacter pylori infection and Epstein–Barr virus positivity, have been associated with the presence of stomach adenocarcinoma. Food habits such as high consumption of salt and ultra-processed foods, consumption of > 35–40 g/day alcohol, and low fruit and vegetable intake have also been identified as risk factors, while high consumption of fruit has been shown to reduce this risk [7].
Food habits changed between 1987 and 2009 throughout the world, including in Brazil, with fresh and minimally processed foods of vegetable origin (e.g., rice, beans, cassava, potatoes, and vegetables) being replaced by industrialized (processed and ultra-processed) ready-to-eat products, leading to excessive daily calorie intake [8]. Monteiro et al. [9] described an increase in the consumption of ultra-processed foods from outside the home, which contributed to chronic disease development, in the Brazilian population. The NOVA food classification system has identified increasing trends in the production and consumption of ultra-processed food and drink products throughout the world [10].
At present, there is limited information about the association of food habits with the development of stomach adenocarcinoma in the Brazilian population. The objective of this case–control study was to analyze the consumption of processed and ultra-processed food in patients with stomach adenocarcinoma, in two cities in the southeast and Amazon regions of Brazil.
Methods
Study population
This study was part of a hospital-based multicentric case–control study on stomach adenocarcinoma “Genomics and epidemiology for gastric adenocarcinomas in Brazil” (FAPESP grant no. 2014/26897–0) conducted at the A.C.Camargo Cancer Center, São Paulo (southeast region of Brazil), and the Hospital Ophir Loyola and General Hospital Universitário João de Barros Barreto, Belém (Amazon region of Brazil). Recruitment was conducted from April 2016 to February 2019 in São Paulo and from July 2017 to April 2019 in Belém.
The study was approved by the Committee on Ethics in Human Research of the Antônio Prudente Foundation Cancer Hospital of A. C. Camargo Cancer Center (no. 1.480.537), and the Hospital Ophir Loyola and General Hospital Universitário João de Barros Barreto (no. 2.395.946). All participants provided written informed consent.
The sample size calculation used a study power (1 – β) of 80%, alpha error rate of 5%, and odds ratio (OR) of 2 for a two-tailed hypothesis and indicated that a minimum of 133 cases and 133 controls were needed. The cases were patients who were treated at the participating centers with histologically confirmed stomach adenocarcinoma, classified according to the International Classification of Diseases for Oncology, third edition (ICD-O3). In São Paulo, the controls were recruited in two groups at the A.C.Camargo Cancer Center, as follows: Group 1 was comprised of patients with negative results for malignancy and preneoplastic lesions, who underwent upper-digestive endoscopy; Group 2 was composed of asymptomatic individuals participating in a cancer prevention campaign. In Belém, the control group were individuals recruited in outpatient clinics (physiotherapy, dentistry, nutrition, and psychology) of the Hospital Universitário João de Barros Barreto. The difference of recruitment between control groups was because in São Paulo there was a prevention clinic where healthy controls (Group 2) were recruited. While in Belém, the controls were hospitals from clinics where patients had no diagnosis of gastric cancer and no clinical suspicion. Cases and controls were matched by frequency according to sex and age (18–75 years, in 5-year groups). All controls were interviewed in the same period of case recruitment.
The exclusion criteria for participation in the study were previous malignancy except non-melanoma skin cancer, preneoplastic lesions (such as intestinal metaplasia), and no physical conditions such as patients with impaired mobility due to illness, or mental and cognition condition precluding to understand the questions made by the interviewers.
Procedures and instruments
Nutritionists trained for interview administered questionnaires and conducted face-to-face interviews with the study participants.
A sociodemographic questionnaire was used to collect information on participants’ sex, age, marital status, education level, and skin color. The lifestyle factors evaluated were alcohol and tobacco consumption and weight status that was available in the medical record. Participants were classified according to their body mass index (BMI) as underweight, eutrophic, overweight, and obese [11]. A Food Frequency Questionnaire (FFQ) validated for the Brazilian population with cancer [12] was adapted with inclusion regional Belém foods. For each food, the participants indicated the frequency, the number of times consumed (1–10) per day, week, month, or year. The size of the ingested portion was presented as small, medium, or large represented in slices, spoons, and transformed into grams according to each type of food. The consumption (grams/day) was calculated as follows: ((frequency × portion)/days)) × grams (e.g., 2 portions of 50 g of bread, consumed 7 times/week). The consumption values, foods, and food groups were stratified into terciles.
The food consumption was presented for each participant at the centers. For the assessment, the foods consumed were classified as processed or ultra-processed (Table 1). Processed foods included culinary or by industrial products group prepared food for meals served in homes and restaurants, and ultra-processed foods group included formulated foods to reduce microbial deterioration (“long shelf life”), which are mostly highly palatable and easy to prepare and/or consume [10].
Some food groups described in Table 1 had low consumption and, to enable the analysis, the following were grouped: (1) Sweets in general (Cakes with or without filling, whipped cream, confectionery sweets/ Cookies with or without filling/ Made sweets (cornflakes, chocolate, gelatin, candies, chocolates, ice cream)); (2) Pizza, fried, and baked snacks/ Popcorn and snacks; and (3) Seasonings (salt, soy sauce, mayonnaise for sauce, and vinaigrette)/ Other industrialized spices (ketchup / mustard and industrialized pepper sauce).
Statistical analyses
The chi-squared test was used to examine differences in demographic and lifestyle characteristics between cases and controls. Univariate and multivariable binomial regression analyses with logistic link function were performed separately for each region to obtain the exponential β to identify the odds ratios (OR) with 95% confidence intervals (CIs) for the outcome of stomach adenocarcinoma. The multiple regression models were adjusted using the Hosmer–Lemeshow test which was built with the following assumptions: improvement in accuracy, as reflected by 95% CIs which measure the stability of OR and total degrees of freedom allowed for each outcome variable (overfitting control) [13, 14]. The Wald test was calculated to determine statistical significance.
“The multiple regression models were adjusted for confounding variables tobacco and alcohol consumption, BMI, and social variables, education level and marital status.”
The data were entered into the Research Electronic Data Capture (REDcap) platform (Vanderbilt University, Tennessee, USA) and statistical analyses were performed using the Statistical Package for Social Science SPSS (version 23.0 for Windows).
Results
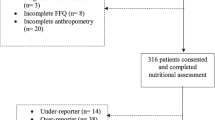
This multicentric case–control study consisted of 214 and 140 cases with stomach adenocarcinoma in São Paulo and Belém, respectively, while there were 150 controls in Group 1 (upper-digestive endoscopy) and 401 controls in Group 2 (healthy controls) in São Paulo, and 140 hospital controls in Belém (Fig. 1).
The majority of cases occurred in males aged > 60 years in São Paulo and Belém. In São Paulo, about 17.5% of the cases were smokers, 42% were ex-smokers, and 48.6% consumed alcohol. In Belém, low weight was observed in 36.7% (n = 51) of the cases, 65.7% (n = 92) were ex-smokers, and 60.7% (n = 85) consumed alcohol (Table 2).
In São Paulo, between cases and endoscopic controls, significant differences were observed for education level, BMI, tobacco consumption, and number of cigarettes smoked/day, while between cases and healthy controls there were differences for skin color, education level, BMI, tobacco consumption, the number of cigarettes smoked/day, and alcohol consumption. In Belém, between cases and controls, differences were observed for skin color, education level, BMI, and tobacco and alcohol consumption (Table 2).
Univariate regression analysis was performed to identify the OR, according to foods or groups (Supplementary Tables S1 and S2). In São Paulo, when comparing cases versus endoscopic controls, it was observed that patients with salted bread consumption between 16 – 43 g/day and ≥ 44 g/day had a chance to present stomach adenocarcinoma (OR = 2.70, 95% CI 1.30 – 5.59 and OR = 1.66, 95% CI 1.02 – 2.73, respectively) when comparing cases and healthy controls who had consumption between 16 – 43 g/day and ≥ 44 g/day of bread were more likely to have stomach adenocarcinoma (OR = 1.82, 95% CI 1.08 – 3.08) and OR = 2.45, 95% CI 1.63 – 3.67, respectively).
Endoscopic controls with white rice consumption ≥ 125 g/day were twice as likely to have stomach adenocarcinoma (OR = 2.23, 95% CI 1.01–4.93). Endoscopic and healthy control groups had an increased chance of having stomach adenocarcinoma when leguminous consumption was ≥ 102 g/day (OR = 2.30, 95% CI 1.35–3.94 and OR = 1.55, 95% CI 1.02–2.34, respectively). Likewise, processed meat, sausage, cold cuts and “tropeiro beans” (sausage, bacon, canned and dried meat, and nuggets) consumption was associated with stomach adenocarcinoma for both control groups, with a two to three times increased chance of having stomach adenocarcinoma. Pizza, fried and baked snacks, popcorn, and snack consumption ≥ 37 g/day was associated with stomach cancer in healthy controls (OR = 1.91, CI 95% 1.27–2.87). In relation to sugar drinks and sweets in general, individuals who had consumption in the last tercile a higher chance of stomach adenocarcinoma (Supplementary Table S1).
Whole grain bread consumption ≥ 6.7 g/day was a protective factor of stomach adenocarcinoma for endoscopic (OR = 0.48, 95% CI 0.30–0.76) and healthy controls (OR = 0.62, 95% CI 0.43–0.90). Pasta consumption was also a protective factor healthy controls (OR = 0.53, 95% CI 0.35–0.80) (Supplementary Table S1).
In Belém, salted bread consumption was a risk factor of stomach adenocarcinoma when intake was 49–100 g/day (OR = 3.31, 95% CI 1.78–6.17) and ≥ 101 g/day (OR = 4.44, 95% CI 2.33–8.47). Individuals who consumed fried and roasted meat ≥ 166 g/day showed a chance of 2.45 (95% CI 1.36–4.42), while sugary drink consumption between 9 and 76.5 g/day increased the chance almost three times (OR = 2.75, 95% CI 1.50–5.06) and ≥ 76.6 g/day increased the chance of stomach adenocarcinoma by about five times (OR = 4.73, 95% CI 2.51–8.89) (Supplementary Table S2).
In Belém, it was observed that yellow cheese consumption ≥ 8.7 g/day showed an 84% decreased chance of stomach cancer (OR = 0.16, 95% CI 0.08–0.29), also individuals who consumed > 1.7 g/day whole grain bread were less likely to have stomach cancer (OR = 0.08, 95% CI 0.02–0.36). Similarly, pasta consumption ≥ 54 g/day showed a decreased chance of stomach cancer (OR = 0.48, 95% CI 0.02–0.36). In addition, French fries were protective factors (> 5 g/day, OR = 0.29, 95% CI 0.17–0.50), as well as 8.3–32.6 g/day (OR = 0.28, 95% CI 0.15–0.52) and ≥ 32.7 g/day (OR = 0.25, 95% CI 0.14–0.47) sweets consumption (Supplementary Table S2).
Total consumption of processed and ultra-processed food ≥ 1448 g/day increased the chance of having stomach adenocarcinoma greater than two times in São Paulo and six times in Belém when processed and ultra-processed food were consumed ≥ 913 g/day (Supplementary Tables S1 and S2).
For the cases as compared with the endoscopic controls from São Paulo, the chance of having stomach adenocarcinoma was increased by the consumption of processed meat, sausage, cold cuts, and “tropeiro beans” (sausage, bacon, canned and dried meat, and nuggets) between 17 and 43 g/day (adjustedOR 1.89, 95% CI 1.01–3.53) and ≥ 44 g/day (adjustedOR 2.56, 95% CI 1.32–4.96), as well as ≥ 80 g/day sweets in general (adjustedOR 2.25, 95% CI 1.21–4.18) (Table 3).
Regarding cases compared to healthy controls in São Paulo, it was observed an increased chance by the consumption of processed meat, sausage, cold cuts, and “tropeiro beans” (sausage, bacon, canned and dried meat, and nuggets) between 17 and 43 g/day (adjustedOR 1.98, 95% CI 1.24–3.18) and ≥ 44 g/day (adjustedOR 2.96, 95% CI 1.82–4.81), as well as of salted bread consumption ≥ 44 g/day (adjustedOR 2.03, 95% CI 1.30–3.18) (Table 3). In Belém, consumption of ≥ 166 g/day fried and roasted meats and fried fish increased the chance of having stomach adenocarcinoma (adjustedOR 2.21, 95% CI 1.13–4.30) (Table 4).
Discussion
We observed that the consumption of processed and ultra-processed foods, mainly salted bread, yellow cheese, processed meat, and sweets increased the chance of having stomach adenocarcinoma in São Paulo and Belém (Brazil). These types of food may contribute to cancer development due to their higher total and saturated fat, as well as added sugar and salt contents, along with lower fiber and vitamin density and low protein [15,16,17,18,19,20,21]. Fiolet et al. [22] reported that ultra-processed food consumption was associated with a 10% increase in general cancer risk.
Monteiro et al. [23] described, in Brazil, an increase in the consumption of ultra-processed foods, which may have contributed to the increased incidence of chronic diseases. Costa Louzada et al. [24] reported that the Brazilian diet had been changing, with traditional meals based on natural or minimally processed foods being replaced with ultra-processed foods. The consumption of added sugar and meat has been shown to be high in Brazil [25, 26]. In São Paulo, sugar consumption increased from 100 to 113 g/day between 2003 and 2008. In addition, the red meat and processed meat intakes were 138 g/day for men and 81 g/day for women, and about 81% of men and 58% of women consumed more meat than recommended [27].
According to the 2018 World Cancer Research Fund report [7], there is sufficient evidence to support that foods preserved in salt contribute to the development of stomach adenocarcinoma; evidence for the contributions of sausages and potatoes remains limited [28, 29]. In these results, patients with stomach adenocarcinoma were characterized by high consumption of ultra-processed foods, increasing the chance of stomach adenocarcinoma by more than two times.
A previous study conducted in São Paulo showed that healthy dietary habits were more frequent among older adults than young adults (36.9% versus 15.4%), and that the Revised Brazilian Healthy Eating Index (BHEI-R) scores improved gradually, and most markedly among older adults, over a 12-year period [30]. The overall population showed increases in total fruit, whole fruit, whole grain, oil, and sodium intakes. The main contributor to socioeconomic inequality in diet quality shifted from ethnicity in 2003 to per-capita household income in 2008 and 2015; age was a persistent factor related to inequality. Concentration indices indicated that individuals with lower incomes had higher BHEI-R scores in 2003, a shift in favor of individuals with higher incomes was noted in 2008 and 2015 [30].
Regarding stomach adenocarcinoma, evidence that the consumption of citrus fruits reduces the risk of gastric cardia adenocarcinoma is also limited [7]. In Brazil, the attributable dietary fractions of low vegetable consumption, low fruit consumption, > 10 g/day salt intake, processed meat consumption, and > 70 g/day red meat intake were 24% for non-cardia stomach cancer and 55% for cardia cancer in women; these percentages were 20% and 55%, respectively, in men [31]. These findings confirm the importance of healthy diets for reducing the risk of stomach adenocarcinoma.
In Belém, higher consumption of salty foods (e.g., jerky, canned meat, shrimp, and salted fish) and carbohydrate-rich foods such as manioc and its derivatives (e.g., flour and tucupi) were identified. According to Baiao et al. [32], processing to produce cassava flour can affect the nutritional value of cassava root through molecular changes and nutrient loss. In addition, cassava flour often contains added aniline-based dyes, which are sources of NH2 and NO2 radicals, and thus, could act as substrates for the endogenous formation of nitrosamines, which are carcinogens associated with the pathogenesis of stomach adenocarcinoma [33]. Family budget surveys conducted in 2002–2003, 2008–2009, and 2017–2018 have documented reductions in the ingestion of fresh or minimally processed foods and processed culinary ingredients, as well as increases in the intake of processed and ultra-processed foods, in all Brazilian regions, including the Amazon [34].
After adjusted for multiple regression models, our results corroborate that consumption of processed and ultra-processed foods such as meat, sausage, fried and roasted meat, and fried fish is associated with stomach adenocarcinoma in São Paulo and Belém. Meta-analysis studies have shown that the consumption of processed foods, such as processed meat and salt-preserved foods, was associated with the risk of stomach cancer development [35,36,37,38,39,40].
The associations observed in this study are according to the literature. The consumption of red meat, in which cooking at high temperatures results in the formation of heterocyclic amines and polycyclic aromatic hydrocarbons, has mutagenic potential and has been associated with the development of cancer in experimental studies [33]. In the same way, poor diets characterized as processed and ultra-processed foods rich in refined starches, including products made with white flour such as bread, pasta, and pizza, as well as processed foods high in fat, starches, or sugars which include cakes, candies, and cookies was observed in these studies may be associated to cancer. As a result, these types of diets favor the accumulation of body fat, which can lead to hormonal changes and a chronic inflammatory state. These conditions stimulate cell proliferation, inhibit programmed cell death, and contribute to the formation and progression of several types of cancer, such as stomach adenocarcinoma [7, 9].
This case–control study is subject to limitations. One limitation is associated with the use of the FFQ, which is long and detailed; regarding other questionnaires, interviewees’ responses may have been affected by memory bias. Interviewer bias may also affect this type of study, although trained nutritionists interviewed our cases and controls. Despite this potential limitation, the FFQ showed good validity and reproducibility in the estimation of usual food consumption among residents of São Paulo [36]. Moreover, we did not classify the cases as cardia and non-cardia to verify the differences associated with diet, and we opted to not adjust for energy intake according to Smith et al. 2013 [41].
Finally, the type of the food (although processed and ultra-processed) has local and cultural food habits in the Amazon region which are quite different from in São Paulo. However, consumption of processed and ultra-processed foods, especially bread (salted), yellow cheese, fried and roasted meats, fried fish, processed meat, and sweets, were associated independently with the chance of having stomach adenocarcinoma in both cities.
Data availability
Data will be made available on reasonable request.
Code availability
Not applicable.
References
Ferlay J, Ervik M, Lam F, Colombet M, Mery L, Piñeros M, Znaor A, Soerjomataram I, Bray F (2020). Global Cancer Observatory: Cancer Today. Lyon, France: International Agency for Research on Cancer. Available from: https://gco.iarc.fr/today (accessed 09 April 2021).
Ministry of Health of Brazil (2021) José Alencar Gomes da Silva National Cancer Institute. Estimate 2020–2022: Cancer incidence in Brazil. Rio de Janeiro. INCA
Curado MP, Silva DRME, Oliveira MM, Soares F, Begnami MD, Coimbra FJF, Assumpção PP, de Sant’Ana RO, Demachki S, Dias-Neto E (2019) Disparities in Epidemiological Profile of Gastric Adenocarcinoma in Selected Cities of Brazil. Asian Pac J Cancer Prev 20:2253–2258. https://doi.org/10.31557/APJCP.2019.20.8.2253
Guimarães RM, Muzi CD (2012) Trend of mortality rates for gastric cancer in Brazil and regions in the period of 30 years (1980–2009). Arq Gastroenterol 49:184–188. https://doi.org/10.1590/S0004-28032012000300003
Giusti ACBS, Salvador PTO, Dos Santos J, Meira KC, Camacho AR, Guimarães RM, Souza DL (2016) Trends and predictions for cancer mortality in Brazil. World J Gastroenterol 22:6527–6538. https://doi.org/10.3748/wjg.v22.i28.6527
Guerra MR, Bustamante-Teixeira MT, Corrêa CSL, Abreu DMX, Curado MP, Mooney M, Naghavi M, Teixeira R, França EB, Malta DC (2017) Magnitude and variation of the burden of cancer mortality in Brazil and Federation Units, 1990 and 2015. Rev Bras Epidemiol 20:102–115. https://doi.org/10.1590/1980-5497201700050009
World Cancer Research Fund/American Institute for Cancer Research. Continuous Update Project Expert Report 2018. Diet, nutrition, physical activity and stomach cancer. Available from: dietandcancerreport.org (accessed 10 Abril 2020).
Ministry of Health of Brazil. Health Care Secretariat. Department of Primary Care. Food guide for the Brazilian population. Brasília: Ministry of Health of Brazil, 2014. Available from: http://www.fao.org/nutrition/education/food-based-dietary guidelines/regions/countries/brazil/en/ (accessed 13 March 2020).
Monteiro C, Levy R, Claro R, De Castro I, Cannon G (2010) Increasing consumption of ultra-processed foods and likely impact on human health: Evidence from Brazil. Public Health Nutr 14:5–13. https://doi.org/10.1017/S1368980010003241
Monteiro CA, Cannon G, Moubarac J-C et al (2018) The UN Decade of Nutrition, the NOVA food classification and the trouble with ultra-processing. Public Health Nutr 21:5–17
[WHO] World Health Organization. Health topics: cancer. 2018 Avaliable from: <URL:http://www.who.int/topics/cancer/en/> (accessed 20 April 2020)
Lameza MMS. Validação de questionário de frequência alimentar para pacientes tratados de câncer colorretal. São Paulo; 2010. [Master's Dissertation-Antônio Prudente Foundation]
Bagley SC, White H, Golomb BA (2001) Logistic regression in the medical literature: standards for use and reporting, with particular attention to one medical domain. J Clin Epidemiol 54(10):979–985. https://doi.org/10.1016/s0895-4356(01)00372-9 (PMID: 11576808)
Kumar R, Chhabra P (2014) Cautions required during planning, analysis and reporting of multivariable logistic regression. Current Medicine Research and Practice 4(1):31–39. https://doi.org/10.1016/j.cmrp.2014.01.004
Luiten CM, Steenhuis IH, Eyles H, Ni Mhurchu C, Waterlander WE (2016) Ultra-processed foods have the worst nutrient profile, yet they are the most available packaged products in a sample of New Zealand supermarkets–CORRIGENDUM. Public Health Nutr 19:539. https://doi.org/10.1017/S1368980015002840pmid:26419699
Adams J, White M (2015) Characterisation of UK diets according to degree of food processing and associations with socio-demographics and obesity: cross-sectional analysis of UK National Diet and Nutrition Survey (2008–12). Int J Behav Nutr Phys Act 12:160. https://doi.org/10.1186/s12966-015-0317-ypmid:26684833
Cediel G, Reyes M, da Costa Louzada ML et al (2018) Ultra-processed foods and added sugars in the Chilean diet (2010). Public Health Nutr 21:125–133. https://doi.org/10.1017/S1368980017001161pmid:28625223
Martínez E, Baraldi LG, Louzada ML, Moubarac JC, Mozaffarian D, Monteiro CA (2016) Ultra-processed foods and added sugars in the US diet evidence from a nationally representative cross-sectional study. BMJ Open. https://doi.org/10.1136/bmjopen-2015-009892pmid:26962035
Moubarac JC, Martins AP, Claro RM, Levy RB, Cannon G, Monteiro CA (2013) Evidence from Canada: consumption of ultra-processed foods and likely impact on human health. Public Health Nutr 16:2240. https://doi.org/10.1017/S1368980012005009pmid:23171687
Moubarac JC, Batal M, Louzada ML, Martinez Steele E (2017) Monteiro CA Consumption of ultra-processed foods predicts diet quality in Canada. Appetite 108:512–520. https://doi.org/10.1016/j.appet.2016.11.006pmid:27825941
Rauber F, Steele EM, Louzada MLC, Millet C, Monteiro CA, Levy RB (2020) Ultra-processed food consumption and indicators of obesity in the United Kingdom population (2008–2016). Plos Ones. 15(5):232676. https://doi.org/10.1371/journal.pone.0232676
Fiolet T, Srour B, Sellem L et al (2018) Consumption of ultra-processed foods and cancer risk: results from NutriNet-Santé prospective cohort. BMJ 360:322. https://doi.org/10.1136/bmj.k322
Monteiro C, Cannon G, Levy R, Moubarac J, Louzada M, Rauber F, Jaime P (2019) Ultra-processed foods: What they are and how to identify them. Public Health Nutr 22:936–941. https://doi.org/10.1017/S1368980018003762
Costa Louzada ML, Martins AP, Canella DS et al (2015) Ultra-processed foods and the nutritional dietary profile in Brazil. Rev Saude Publica 49:38. https://doi.org/10.1590/S0034-8910.2015049006132
Ferguson LR (2010) Meat and cancer. Meat Sci 84:308–313. https://doi.org/10.1016/j.meatsci.2009.06.032
Fisberg M, Kovalskys I, Gómez G, Rigotti A, Sanabria LY, García MC, Torres RG, Herrera-Cuenca M, Zimberg IZ, Koletzko B, Pratt M (2018) Total and added sugar intake: assessment in eight Latin American countries. Nutrients 10:389. https://doi.org/10.3390/nu10040389
Carvalho AM, César CL, Fisberg RM, Marchioni DM (2014) Meat consumption in São Paulo-Brazil: trend in the last decade. PLoS ONE. https://doi.org/10.1371/journal.pone.0096667
Carvalho AM, César CL, Fisberg RM, Marchioni DM (2013) Excessive meat consumption in Brazil: diet quality and environmental impacts. Public Health Nutr 16:1893–1899. https://doi.org/10.1017/S1368980012003916
Bomfim NS, Dias EP, Sbeghen MR, Sbeghen MR (2014) A atuação do nutricionista em pacientes com câncer gástrico. Unoesc & Ciência – ACBS. 5:129–134
Ferrari TK, Cesar CL, Alves MC, Barros MB, Goldbaum M, Fisberg RM (2017) Estilo de vida saudável em São Paulo. Brasil Cadernos de Saúde Pública. https://doi.org/10.1590/0102-311x00188015
Silva AE, G, de Moura L, Curado MP, Gomes Fda S, Otero U, Rezende LF, Daumas RP, Guimarães RM, Meira KC, Leite Ida C, Valente JG, Moreira RI, Koifman R, Malta DC, Mello MS, Guedes TW, Boffetta P, (2016) The fraction of cancer attributable to ways of life, infections, occupation, and environmental agents in Brazil in 2020. PLoS ONE. https://doi.org/10.1371/journal.pone.0148761
Baiao DDS, de Freitas CS, Gomes LP, da Silva D, Correa A, Pereira PR, Aguila EMD, Paschoalin VMF (2017) Polyphenols from root, tubercles and grains cropped in Brazil: Chemical and nutritional characterization and their effects on human health and diseases. Nutrients 9:1044. https://doi.org/10.3390/nu9091044
Campolina AG et al (2012) Multimorbidade e Expressão Gênica Perspectivas Nutrigenômicas e Nutrigenética para Abordagem das Doenças Crônicas. Kur´yt`yba. São Paulo 4:19–42
Resende ALS, Mattos IE, Koifman S (2006) Dieta e câncer gástrico: aspectos históricos associados ao padrão de consumo alimentar no Estado do Pará. Rev Nutr 19:511–519. https://doi.org/10.1590/S1415-52732006000400010
Search for family budgets: 2017–2018: avaliação nutricional da disponibilidade domiciliar de alimentos no Brasil / IBGE, Coordenação de Trabalho e Rendimento. - Local: Rio de JaneiroEditor: IBGE. 56
Selem SS, Carvalho AM, Verly-Junior E, Carlos JV, Teixeira JA, Marchioni DM, Fisberg RM (2014) Validity and reproducibility of a food frequency questionnaire for adults of São Paulo. Brazil Rev Bras Epidemiol 17:852–859. https://doi.org/10.1590/1809-4503201400040005
Kim SR, Kim K, Lee SA, Kwon SO, Lee JK, Keum N, Park SM (2019) Effect of red, processed, and white meat consumption on the risk of stomach cancer: an overall and dose response meta-analysis. Nutrients 11(4):826. https://doi.org/10.3390/nu11040826
Song P, Wu L, Guan W (2015) Dietary nitrates, nitrites, and nitrosamines intake and the risk of stomach cancer: a meta-analysis. Nutrients 7(12):9872–9895
Poorolajal J, Moradi L, Mohammadi Y, Cheraghi Z, Gohari-Ensaf F (2020) Risk factors for stomach cancer: a systematic review and meta-analysis. Epidemiol Health 42:e2020004. https://doi.org/10.4178/epih.e2020004]
Wang Z, Graham DY, Khan A, Balakrishnan M, Abrams HR, El-Serag HB, Thrift AP (2018) Incidence of stomach cancer in the USA during 1999 to 2013: a 50-state analysis. Int J Epidemiol 47(3):966–975. https://doi.org/10.1093/ije/dyy055
Smith AD, Emmett PM, Newby PK, Northstone K (2013) Dietary patterns obtained through principal components analysis: the effect of input variable quantification. Br J Nutr 109(10):1881–1891. https://doi.org/10.1017/S0007114512003868 (Epub 2012 Sep 6 PMID: 22950853)
Acknowledgments
We thank the interviewers from São Paulo (Iara Monique, Camila Gatti, Ana Carolina Cantelli, and Graziela Baladão) and from Belém (Tayana Vago, Ana Carla da Silva, Anna Byatriz Lopes, and Lucas Trindade) who helped with data collection; the study participants, whose generosity made this research possible; and Emmanuel Dias-Neto, the principal investigator of the Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP) project.
Funding
This study was financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior – Brazil (CAPES) – Finance Code 001. This research received funding of Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP) Grant 2014/26897–0.
Author information
Authors and Affiliations
Contributions
M.P.C., F.J.F.C., P.P.A., M.S.A., and A.G.P. designed the research; M.A.F. and J.J.N.A. conducted the research; S.V.P. analyzed the data; D.R.M.S., J.J.N.A., S.V.P., M.A.F., and M.P.C. wrote the paper; M.P.C. and S.V.P. had primary responsibility for the final content. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest to declare. All authors have seen and agree with the contents of the manuscript. We certify that the submission is original work and is not under review by any other publication.
Ethical approval
Data described in the manuscript, the codebook, and the analytical codes will be made available upon request and pending approval.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Peres, S.V., Silva, D.R.M., Coimbra, F.J.F. et al. Consumption of processed and ultra-processed foods by patients with stomach adenocarcinoma: a multicentric case–control study in the Amazon and southeast regions of Brazil. Cancer Causes Control 33, 889–898 (2022). https://doi.org/10.1007/s10552-022-01567-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10552-022-01567-w