Abstract
Purpose
To examine trends in the surgical treatment of breast cancer by age, rurality, and among Black women in a populous, racially diverse, state in the Southeastern United States of America.
Methods
We identified women diagnosed with localized or regional breast cancer between 2003 and 2016 in the North Carolina Central Cancer Registry (n = 86,776). Using Joinpoint regression we evaluated the average annual percentage change in proportion of women treated with mastectomy versus breast-conserving surgery overall, by age group, among Black women, and for women residing in rural areas.
Results
Overall, the rate of mastectomy usage in the population declined 2.5% per year between 2003 and 2016 (95% CI − 3.2, − 1.7). Over this same time interval, breast-conserving surgery increased by 1.6% per year (95% CI 0.9, 2.2). These temporal trends in surgery were also observed among Black women and rural residing women. Trends in surgery type varied by age group: mastectomy declined over time among women > 50 years, but not among women aged 18–49 at diagnosis.
Discussion
In contrast to national studies that reported increasing use of mastectomy, we found declining mastectomy rates in the early 2000s in a Southern US state with a racially and geographically diverse population. These decreasing trends were consistent among key subgroups affected by cancer inequities, including Black and White rural women.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Surgical treatment is an integral part of care for early-stage breast cancer with more than 90% of patients undergoing some form of surgical resection of their cancer [1]. Since 1991, in the USA, the National Institutes of Health consensus guidelines for women with early-stage breast cancer have described an equivalence in long-term outcomes between mastectomy and breast-conserving surgery (BCS) with adjuvant radiation therapy [2]. The existence of multiple possible surgical treatment options for breast cancer denotes preference-sensitive care where decisions are likely to be made in a shared decision-making framework between the patient and care team [3]. Specifically, the decision to opt for mastectomy over BCS involves clinical, cost, or quality of life tradeoffs for patients [4,5,6]. Rate of breast-conserving surgery among eligible patients is a Commission on Cancer (CoC) quality measure among accredited facilities [7]. Given documented barriers to high-quality care for Black women and White rural women in the United States of America, particularly within Southeastern states, monitoring trends in surgical type is a critical step for evaluating racial and geographic equity in delivering optimal cancer care [8,9,10].
Evidence is mixed regarding BCS and mastectomy trends in the past 30 years in the USA. Since the early 2000s some studies have described an increase in the use of mastectomy, while others described an initial decrease followed by an increase and still others described no change in mastectomy rates [11,12,13,14,15,16,17]. For example, studies conducted in Kentucky, Florida, and using US national Surveillance Epidemiology and End Results data have separately described a decrease in rate of mastectomy among women with early-stage breast cancer from 2000 to 2004 and then a change in trend indicating an increase in mastectomy rate from 2005 to 2008 when follow-up ended for these studies [18,19,20].
Of these studies, several noted temporal increases in contralateral prophylactic mastectomies over time. Data from the Agency for Healthcare Research and Quality suggest that the US national uptick in overall mastectomy rates between 2003 and 2013 has been driven by an increase in contralateral mastectomy [21]. Consistent across these studies is the under-representation of patients of color with breast cancer, patients residing in rural areas, and specifically patients of color living in rural areas who are at increased risk for experiencing inequities in care delivery [22]. This is consequential, as rural cancer patients and specifically Black rural cancer patients in the USA experience profound inequities in cancer treatment and outcomes [23].
Large population-based studies of cancer care delivery often undersample Black patients and those that do have a sizeable Black population have additional population restrictions. For instance, some studies that included a large sample of Black breast cancer patients have primarily focused on women who had inpatient rather than outpatient surgeries [24, 25]. This restriction to inpatient procedures is particularly limiting, given that many BCS procedures happen in the outpatient setting, and the number of outpatient mastectomies has nearly doubled from 2003 to 2013 [21].
Other large studies have been restricted to a certain age range or insurance payers dominated by certain age groups, such as Medicare beneficiaries. These studies of selected populations may yield incomplete insights about the overall breast cancer patient population [15]. For example, restriction to Medicare populations provides an incomplete picture, as patterns in surgical care differ for older patients with recent movement toward the de-escalation of care among the oldest patients [26]. Additionally, restriction to Medicare-insured populations leaves an important gap for understanding surgical trends in women younger than 65. On the other hand, large database studies of surgical trends commonly underrepresent rural patients due to higher rates of non-insurance and being less likely to be treated at a CoC-accredited facility [27]. No prior study has explicitly addressed how surgical treatment differs among Black women by their age group and the rurality of where they live.
The main objective of this study is to examine temporal trends from 2003 to 2016 in the prevalence and type of surgical treatment of breast cancer for over 80,0000 adult women living in North Carolina. North Carolina is in the Southeastern United States with substantial Black rural populations; in fact 23% of the Black population resides in a rural county [28]. In this investigation, using the total North Carolina breast cancer population as a referent, we evaluate the within-group treatment variation for Black women by age and rurality.
Methods
Data and study sample
This study used a retrospective cohort design of all first primary, unilateral cases of loco-regional breast cancer diagnosed in North Carolina between 2003 and 2016. The primary source of data was the North Carolina Central Cancer Registry, a statewide population-based registry accredited by the North American Association of Central Cancer Registries [29]. We identified all records for women over the age of 18 diagnosed with the Surveillance, Epidemiology, and End Results (SEER) cancer registry defined Summary Stage localized or regional breast cancer [30]. Unlike other staging criteria, the SEER program criteria were a required data element across the entire time period, enabling us to maximize inclusion in the cohort. Women were excluded if they were missing SEER Summary stage at diagnosis (n = 3140), diagnosed with in situ cancer (n = 20,368), or diagnosed with distant cancer (n = 5624). Patients were also excluded if they had unknown surgery status (n = 74), Fig. 1.
Definition of study variables
The primary outcome of interest was the type of first course of surgical treatment a patient received as recorded in the North Carolina Central Cancer Registry. Prior research using these registry data has demonstrated high agreement for the surgical treatment information contained in the North Carolina Central Cancer Registry when compared to insurance claims [31]. Surgery types were determined by the North American Association of Central Cancer Registry cancer-specific treatment codes contained in the cancer registry (Item #670). Type of surgery was categorized as follows: breast-conserving surgery (partial mastectomy or lumpectomy, codes 20–24) and mastectomy (modified radical mastectomy, total mastectomy, or simple mastectomy, codes 30–80). This variable included both unilateral and bilateral procedures.
There were several key sub-populations of interest. Black women, all women residing in rural areas, and specifically Black women residing in rural areas. These groups were selected as populations of interest because of previously documented breast cancer disparities and under-representation in previous investigations of secular trends in breast cancer surgery [32,33,34]. Race was categorized into Black and non-Black categories based on the abstracted data from health systems included in the cancer registry. Rurality was operationalized using the United States Department of Agriculture Rural–Urban Commuting Area (RUCA) codes based on census tract of residence at the time of diagnosis, an ordinal measure of rurality [35]. Every census tract is assigned a score of 1 to 10 with 1 being the most urban and 10 being the most rural. As recommended by the Federal Office of Rural Health Policy we dichotomized rurality: RUCA codes 1–3 were classified as urban and census tracts with RUCA codes 4–10 we classified as rural [36]. We were also interested in assessing differences in surgical trends by age at diagnosis. Age at diagnosis was categorized into the following groupings: 18–49 years, 50–64 years, and 65 years and older. Insurance type was defined by the cancer registry as the type of insurance at the time of diagnosis and was categorized into commercial, Medicaid, Medicare, Medicaid/Medicare dual enrolled, Uninsured, and Insured with unknown type.
Statistical analysis
Demographic and clinical characteristics of women who had mastectomy compared to breast-conserving surgery were calculated stratified by key subgroup, Black women, rural women, and Black women in rural areas. To detect secular trends in the proportion of procedures that were mastectomy compared to BCS we used Joinpoint Regression. Joinpoint Regression uses a series of permutation tests to identify the inflection point at which there was a significant change in the annual proportion of procedures of a given surgery type (APC) [37]. These models tested for up to 3 joinpoints or inflection points where the slope changes, during 2003 to 2016 with the p-value for each permutation test being determined by Monte Carlo methods [38]. We also calculated the overall average annual percentage change (AAPC) to describe the net change in type of procedures over the whole time period where the null hypothesis is that there is no change in the proportion of a given procedure. Stratified analyses were conducted by race, age group, and rurality of residence.
Given that SEER staging is primarily used for research while TNM staging is used in clinical practice and may be more meaningful in assessing who is BCS eligible, we conducted a sensitivity analysis to evaluate the missingness of TNM staging to evaluate the consistency of results when restricting to patients with non-missing TNM stage.
Statistical analyses were performed using SAS version 9.4 and the Joinpoint regression program, version 4.0.4, from the Surveillance Research Program of the National Cancer Institute [37, 38]. This investigation was approved by the Institutional Review Board (IRB) at the University of North Carolina at Chapel Hill (IRB # 18–3552).
Results
Cohort characteristics
Overall, there were 116,019 women diagnosed with breast cancer between 2003 and 2016, of which 86,776 women met inclusion criteria, Fig. 1. Of these women, 67.4% had localized disease and 32.6% had regional disease (Table 1). Demographically, 19.4% of women were Black, 21.6% lived in rural areas, and 3.8% of patients overall were Black and lived in rural areas.
Change in procedure type
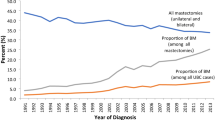
At the start of the study period in 2003, the proportions of mastectomy and BCS were relatively equal at 49.1% and 48.4%, respectively, with 2.5% of women not having any surgery. At the end of the study period in 2016, BCS was the more common procedure at 60.8%, while the proportion of mastectomy dropped to 33.7%. In 2016, 5.5% of women had no record of surgery. In Joinpoint regression analyses, there was one detected change in slope for the proportion of mastectomies between 2003 and 2016. Between 2003 and 2010 there was no significant change in mastectomy (APC − 0.4%; 95% CI − 1.5%, 0.7%), culminating with a notable decrease from 2010 to 2016 (APC − 4.8%, 95% CI − 6.2%, − 3.3%) per year, Fig. 2A. The proportion of BCS also remained flat (APC 0.2; 95% CI − 0.9, 1.2) between 2003 and 2010 and then increased sharply from 2010 onward (APC 3.2%; 95% CI 2.2%, 4.3%). Trends were similar among Black women. Among Black women, there was no net change in proportion of mastectomy from 2003 to 2011 (APC − 0.9%; 95% CI − 2.3%, 0.6%) or breast-conserving surgery from 2003 to 2010 (APC 0.3%; 95% CI − 1.1%, 1.7%), (Fig. 2B). After 2010 and 2011, respectively, among Black women, the proportion of mastectomy decreased (APC − 6.5%, 95% CI − 9.5%, − 3.4%) while BCS increased (APC 3.5%; 95% CI 2.1%, 4.9%), until 2016.
Temporal trends in surgical treatment of early-stage breast cancer 2003–2016. A Total North Carolina Population. B Among Black Women. APC indicates Annual Percent Change which calculates the annual percentage rate change in a model specified time interval. AAPC is Average Annual Percent Change which calculates the average annual percentage rate change in a model specified time interval over the entire time interval
There were several notable differences in the distribution of surgery type by age group. In contrast to what was observed in the overall population, among women aged 18–49 years, mastectomy was the most common procedure type. Among these younger women there was a modest increase in proportion of mastectomy from 2003 to 2016, (AAPC 0.7, 95% CI 0.1, 1.4), Fig. 3A, while BCS decreased overtime (AAPC − 1.1%, 95% CI − 1.7%, − 0.6%), data not shown. However, among Black women aged 18–49, breast-conserving surgery remained the more common procedure type, and there was no net change in proportion for either surgery over the study period. Trends among women aged 50–64, and older than 65, resembled those seen in the overall population, with BCS increasing and mastectomy decreasing particularly after 2010, Fig. 3B and C. Among Black women aged 50 to 64 and those older than 65 years, the proportion of surgeries that were mastectomy steadily decreased and BCS procedures increased over the entire time interval (Fig. 3E, F.
Average Annual Percent Change (AAPC) for use of mastectomy in the treatment of early-stage breast cancer 2003–2016 stratified by age group. A–C Total North Carolina Population. D–F Among Black Women. AAPC is Average Annual Percent Change which calculates the average annual percentage rate change in a model specified time interval over the entire time interval. AAPC is presented for mastectomy only
With regard to patients living in rural areas, mastectomy was the more prevalent procedure type at the start of the time interval. In 2011 for rural women overall, BCS became the more prevalent procedure type (Fig. 4A). Similar to the overall North Carolina population, among rural women, BCS proportion remained flat between 2003 and 2010 (APC 1.1%; 95% CI − 0.2%, 2.3%) and then increased from 2010 onward (APC 3.4%; 95% CI 2.0%, 4.7%). Accordingly, among rural patients overall the proportion of mastectomy decreased steadily from 2003 to 2011 (AAPC − 1.5, 95% CI − 2.4, − 0.6) and then decreased more sharply after 2011 (AAPC − 5.8, 95% CI − 8.0, − 3.6). Specifically, among Black women in rural areas, the observed crossover in most prevalent procedure type happened a slightly earlier, in 2009, Fig. 4B. Among Black rural women there were no detected inflections in slope for procedure type. In this population, the proportion of BCS increased (APC 2.3%; 95% CI 1.6, 3.0) and mastectomy decreased (AAPC − 3.0, 95% CI − 4.2, − 1.7) between 2003 and 2016 with no changes in slope during this time interval.
Stage at diagnosis sensitivity analyses
TNM stage at diagnosis was missing for 35.7% of records and therefore was not used in the primary analysis. Of the 30,964 patients missing stage, (19%) were Black which is a comparable proportion to the proportion of Black patients included in the study using SEER staging inclusion criteria. However, 42.6% of rural residing women were missing TNM stage at diagnosis, compared to 34.3% overall (Supplemental Table 1). Further, missingness of TNM stage was associated with year of diagnosis. Cumulatively, years 2003 through 2008 made up 62.4% of TNM stage missingness (Supplemental Table 2).
In sensitivity analyses restricted to non-missing TNM Stage (n = 67,731) our interpretation of trends in surgery practices remained unchanged across age, race, and rurality. Among the total NC population among those with non-missing TNM staging, the change in proportion of mastectomy for the time interval 2003 to 2016 yielded an AAPC of − 4.5% (95% CI − 4.3%, − 0.9%) per year. While the proportion of procedures that were BCS increased steadily with an AAPC of 2.2% (95% CI 1.8%, 2.6%) between 2003 and 2016.
Discussion
In this investigation, we observed a considerable decrease in the use of mastectomy among women eligible for either BCS or mastectomy in North Carolina between 2011 and 2016. This overall finding differs from previously reported national trends assessed using the National Cancer Database and other data sources that described an increase in the proportion of patients receiving mastectomy overall [1, 18,19,20]. Our finding was consistent by race and rurality but differed by age, where BCS decreased for women 18–49 years old between 2003 and 2016. Characteristics of our study populations differ from prior investigations in several important ways. There were a higher proportion of Black and rural patients represented and patients from all insurance types, including the uninsured, and all treating facility types were included. Importantly study includes data through 2016, representing an update on previous trends. Our study is one of the few investigations to evaluate within-group variation in the Black patient surgical treatment experience by age group and rurality.
Temporal trends in surgical treatment have previously been shown to vary by age. A national study conducted among women older than 65 found a consistent decline in mastectomy rate between 2000 and 2008 [15]. This decline in mastectomy for women older than 65 was consistent with what we observed in our investigation in the North Carolina overall population and our key sub-populations of interest. The prevalence of mastectomy across the time period was in general higher for women younger than 65 than women older than 65 potentially reflecting a trend to de-escalate breast cancer therapy in older women or a preference for a surgery perceived as “more aggressive” among younger women [39, 40]. Temporal trends in breast cancer surgery differed for Black women younger than 50 relative to North Carolina as a whole. Among younger Black women, breast-conserving surgery remained the most common procedure type in contrast to mastectomy being the more common procedure across the time interval overall in the North Carolina population. This difference may represent cultural differences in procedure preference or could possibly be a gap in shared decision-making around surgery.
Our finding of overall decreasing rates of mastectomy for the treatment of early-stage breast cancer was surprising given the prior literature. North Carolina, is relatively unique with the density of three National Cancer Institute designated comprehensive cancer centers that in total, cover the entire state within the catchment area [41, 42]. This potential high level of access to academic medical centers with comprehensive cancer centers may influence the patterns of surgical treatment that we observed. Further, nationally and internationally, there has been increased attention on reducing the treatment burden of breast cancer care. This has been observed in the move from axillary lymph node dissection to sentinel lymph node biopsies where indicated, shorter courses of radiation therapy, and greater use of neoadjuvant therapy [43,44,45]. The observed decline of mastectomies in our study may be in alignment with the broader state of breast cancer care delivery, especially given updated data on long-term outcomes for patients treated with BCS.
Presently, studies have revisited questions related to potential differences in long-term survival among women diagnosed with early-stage breast cancer by surgery type using large-scale observational and real-world data studies highlighting the ongoing need for population-level surveillance of surgical treatment trends [46]. A single-state cancer registry study in Louisiana found that women treated with breast-conserving surgery had higher overall survival after 10 years than women treated with mastectomy, after controlling for sociodemographic and clinical characteristics [47]. Similarly, in the National Cancer Database, researchers found higher overall and breast cancer-specific survival for women treated with BCS than those with mastectomy [48]. Globally, a recent prospective cohort study conducted in Sweden found greater 5-year overall survival for patients treated with BCS and radiation than those treated with mastectomy with or without radiation [49]. As the relative benefits of each surgery type continue to be evaluated, it is imperative to monitor temporal trends in treatment to ensure all groups are receiving treatment that is based on contemporary standards of evidence.
There are important demographic and methodologic considerations in studying temporal trends of care delivery over long windows of time, namely the change in rurality designation that can occur. In the USA, since the year 2000, the proportion of people living in rural areas is decreasing, while the proportion of people residing in suburban or urban areas is increasing [50]. For this investigation, specifically in North Carolina, the urban population increased from 60% of residents to 66% of residents, largely attributable to in-migration to already urban areas from other states [51,52,53]. While there were no substantial shifts in rural designation in North Carolina during the study period, the changing demography of the USA remains an important factor to consider in the evaluation of temporal trends in cancer treatment.
Our study has several strengths. Using surgical information contained in the cancer registry we were able to include all women across the state of North Carolina including uninsured women who are not represented in insurance claims-based research and also those treated outside of CoC-accredited facilities that report to the NCDB. We were able to conduct specific subgroup analyses among women from populations affected by health disparities in breast cancer outcomes, such as Black and rural women, populations often underrepresented in investigations of surgical management of breast cancer. Additionally, the recency of our data through 2016 permits a more recent update on surgical trends than was previously available. Our study is not without limitations. This is an investigation that was limited to a single state and it is unknown how results from this investigation generalize to other US states or global trends in breast cancer surgery. Additionally, while our previous investigation demonstrated high validity for the surgical treatment information contained in the registry, there remains the potential for surgery misclassifications particularly for patients who had multiple procedures or re-excisions. While cancer registrars can update a treatment record following a second procedure, it is unknown how commonly this occurs. Further, the surgical landscape of breast cancer is constantly changing, with oncoplastic techniques becoming increasingly common for BCS. In the current national surgical data standards for cancer registries, including North Carolina, information on oncoplastic techniques is not currently reported and as such we are not able to evaluate this in our analyses, masking the potential population-level heterogeneity that may exist for type BCS received.
The frequent scientific and clinical updates in the treatment of cancer necessitate a thorough understanding of population-level trends in cancer care delivery and how those trends may differ across geographic and demographic lines. In our investigation, we observed that in a Southern state with a large Black population that mastectomy rates decreased overall in contrast to prior national estimates of increasing rates. This decrease was consistent among marginalized populations of Black women and rural White women. Our work underscores the need for ongoing population-level surveillance of cancer treatment trends that is inclusive of populations who are most likely to experience cancer care inequities.
Data availability
The data that support the findings of this study are available from the North Carolina Central Cancer Registry but restrictions apply to the availability of these data, which were used under license for the current study and so are not publicly available.
References
Kummerow KL, Du L, Penson DF et al (2015) Nationwide trends in mastectomy for early-stage breast cancer. JAMA Surg 150:9. https://doi.org/10.1001/jamasurg.2014.2895
(1991) NIH consensus conference. Treatment of early-stage breast cancer. JAMA 265:391–5
Mitchell S, Gass J, Hanna M (2018) How well informed do patients feel about their breast cancer surgery options? Findings from a nationwide survey of women after lumpectomy and/or mastectomy. J Am Coll Surg 226:134-146.e3. https://doi.org/10.1016/J.JAMCOLLSURG.2017.10.022
Fisher ES, Wennberg JE (2003) Health care quality, geographic variations, and the challenge of supply-sensitive care. Perspect Biol Med 46:69–79. https://doi.org/10.1353/pbm.2003.0004
Lee CN, Chang Y, Adimorah N et al (2012) Decision making about surgery for early-stage breast cancer. J Am Coll Surg 214:1–10. https://doi.org/10.1016/j.jamcollsurg.2011.09.017
Elwyn G, Frosch D, Rollnick S (2009) Dual equipoise shared decision making: definitions for decision and behaviour support interventions. Implementation science : IS 4:75. https://doi.org/10.1186/1748-5908-4-75
CoC Quality of Care Measures 2020 Surveys. https://www.facs.org/quality-programs/cancer/ncdb/qualitymeasurescocweb. Accessed 10 Nov 2021
Vines AI, Carpenter WR, Chen RC, et al (2017) Responding to a community’s concern: a comparison of breast cancer characteristics and initial treatment in three selected North Carolina Counties. North Carolina Med J 78:357–365. https://doi.org/10.18043/ncm.78.6.357
Meilleur A, Subramanian SV, Plascak JJ et al (2013) Rural residence and cancer outcomes in the United States: issues and challenges. Cancer Epidemiol Biomark Prev 22:1657–1667. https://doi.org/10.1158/1055-9965.EPI-13-0404
McLaughlin JM, Anderson RT, Ferketich AK et al (2012) Effect on survival of longer intervals between confirmed diagnosis and treatment initiation among low-income women with breast cancer. J Clin Oncol 30:4493. https://doi.org/10.1200/JCO.2012.39.7695
Vaz-Luis I, Hughes ME, Cronin A et al (2016) Trends in the use of mastectomy in women with small node-negative breast cancer treated at US academic centers. Breast Cancer Res Treat 155:569–578. https://doi.org/10.1007/s10549-016-3707-1
Showalter SL, Grover S, Sharma S et al (2013) Factors influencing surgical and adjuvant therapy in stage I breast cancer: a SEER 18 database analysis. Ann Surg Oncol 20:1287–1294. https://doi.org/10.1245/s10434-012-2693-8
Portschy PR, Tuttle TM (2013) Increasing mastectomy rates among all age groups for early stage breast cancer: a 10-year study of surgical choice. Breast Dis 24:62–63. https://doi.org/10.1016/j.breastdis.2013.01.028
Covelli AM, Baxter NN, Fitch MI, Wright FC (2014) Increasing mastectomy rates—the effect of environmental factors on the choice for mastectomy: a comparative analysis between Canada and the United States. Ann Surg Oncol 21:3173–3184. https://doi.org/10.1245/s10434-014-3955-4
Shirvani SM, Jiang J, Likhacheva A et al (2016) Trends in local therapy utilization and cost for early-stage breast cancer in older women: implications for payment and policy reform. Int J Radiat Oncol Biol Phys 95:605–616. https://doi.org/10.1016/j.ijrobp.2016.01.059
Freedman RA, Kouri EM, West DW et al (2016) Higher stage of disease is associated with bilateral mastectomy among patients with breast cancer: a population-based survey. Clin Breast Cancer 16:105–112. https://doi.org/10.1016/j.clbc.2015.08.004
Nagar H, Yan W, Christos P et al (2017) Older patients with early-stage breast cancer. Am J Clin Oncol 40:300–305. https://doi.org/10.1097/COC.0000000000000144
Dragun AE, Pan J, Riley EC et al (2013) Increasing use of elective mastectomy and contralateral prophylactic surgery among breast conservation candidates. Am J Clin Oncol 36:375–380. https://doi.org/10.1097/COC.0b013e318248da47
Mahmood U, Hanlon AL, Koshy M et al (2013) Increasing national mastectomy rates for the treatment of early stage breast cancer. Ann Surg Oncol 20:1436–1443. https://doi.org/10.1245/s10434-012-2732-5
McGuire KP, Santillan AA, Kaur P et al (2009) Are mastectomies on the rise? A 13-year trend analysis of the selection of mastectomy versus breast conservation therapy in 5865 patients. Ann Surg Oncol 16:2682–2690. https://doi.org/10.1245/s10434-009-0635-x
Steiner CA, Weiss AJ, Barrett ML, et al (2006) Trends in bilateral and unilateral mastectomies in hospital inpatient and ambulatory settings, 2005–2013: statistical brief #201
Caldwell JT, Ford CL, Wallace SP et al (2016) Intersection of living in a rural versus urban area and race/ethnicity in explaining access to health care in the United States. Am J Public Health 106:1463–1469. https://doi.org/10.2105/AJPH.2016.303212
Moore JX, Royston KJ, Langston ME et al (2018) Mapping hot spots of breast cancer mortality in the United States: place matters for Blacks and Hispanics. Cancer Causes Control 29:737–750. https://doi.org/10.1007/s10552-018-1051-y
Akinyemiju T, Sakhuja S, Vin-Raviv N (2016) Racial and socio-economic disparities in breast cancer hospitalization outcomes by insurance status. Cancer Epidemiol 43:63–69. https://doi.org/10.1016/j.canep.2016.06.011
Dehal A, Abbas A, Johna S (2013) Racial disparities in clinical presentation, surgical treatment and in-hospital outcomes of women with breast cancer: analysis of nationwide inpatient sample database. Breast Cancer Res Treat 139:561–569. https://doi.org/10.1007/s10549-013-2567-1
Mandelblatt JS, Kerner JF, Hadley J et al (2002) Variations in breast carcinoma treatment in older medicare beneficiaries. Cancer 95:1401–1414. https://doi.org/10.1002/cncr.10825
Zahnd WE, Askelson N, Vanderpool RC et al (2019) Challenges of using nationally representative, population-based surveys to assess rural cancer disparities. Prev Med. https://doi.org/10.1016/J.YPMED.2019.105812
Catchment Area - UNC Lineberger. https://unclineberger.org/community-outreach/catchment/. Accessed 15 Nov 2021
NC SCHS: Central Cancer Registry. https://schs.dph.ncdhhs.gov/units/ccr/. Accessed 18 Oct 2021
Adamo MB, Johnson CH, Ruhl JL D LA (2010) Seer program coding and staging manual 2010
Roberson ML, Nichols HB, Wheeler SB et al (2021) Validity of breast cancer surgery treatment information in a state-based cancer registry. Cancer Causes Control 2021:1–9. https://doi.org/10.1007/S10552-021-01520-3
Mets EJ, Chouairi FK, Gabrick KS et al (2019) Persistent disparities in breast cancer surgical outcomes among hispanic and African American patients. Eur J Surg Oncol 45:584–590. https://doi.org/10.1016/J.EJSO.2019.01.016
Sposto R, Keegan THM, Vigen C et al (2016) The effect of patient and contextual characteristics on racial/ethnic disparity in breast cancer mortality. Cancer Epidemiol Biomark Prev 25:1064–1072. https://doi.org/10.1158/1055-9965.EPI-15-1326
Ojinnaka CO, Luo W, Ory MG et al (2017) Disparities in surgical treatment of early-stage breast cancer among female residents of Texas: the role of racial residential segregation. Clin Breast Cancer 17:e43–e52. https://doi.org/10.1016/J.CLBC.2016.10.006
USDA ERS - Rural-Urban Commuting Area Codes. https://www.ers.usda.gov/data-products/rural-urban-commuting-area-codes.aspx. Accessed 21 Oct 2021
Hall SA, Kaufman JS, Ricketts TC (2006) Defining urban and rural areas in U.S. epidemiologic studies. J Urban Health 83:162–175
Kim H-J, Fay MP, Feuer EJ, Midthune DN (2000) Permutation tests for joinpoint regression with applications to cancer rates. Stat Med 19:335–351. https://doi.org/10.1002/(SICI)1097-0258(20000215)19:3%3c335::AID-SIM336%3e3.0.CO;2-Z
Kim H-J, Luo J, Kim J et al (2014) Clustering of trend data using joinpoint regression models. Stat Med 33:4087–4103. https://doi.org/10.1002/sim.6221
Downs-Canner S, Zabor EC, Wind T et al (2019) Radiation therapy after breast-conserving surgery in women 70 years of age and older: how wisely do we choose? Ann Surg Oncol 26:969–975. https://doi.org/10.1245/s10434-018-07151-4
Ruddy KJ, Ganz PA (2019) Treatment of nonmetastatic breast cancer. JAMA 321:1716. https://doi.org/10.1001/jama.2019.3927
Geographic Information Systems & Science—NCI Catchment Areas. https://gis.cancer.gov/ncicatchment/app/. Accessed 15 Feb 2022
Delnero PF, Buller ID A National Map of NCI-Designated Cancer Center Catchment Areas 1 on the 50 th Anniversary of the Cancer Centers Program. 276–6558. https://doi.org/10.1158/1055-9965.EPI-21-1230/3034778/epi-21-1230.pdf
Curigliano G, Burstein HJ, Winer EP, et al (2017) De-escalating and escalating treatments for early-stage breast cancer: the St. Gallen International Expert Consensus Conference on the Primary Therapy of Early Breast Cancer 2017. Ann Oncols 28:1700–1712. https://doi.org/10.1093/ANNONC/MDX308
Heil J, Pfob A, Morrow M (2021) De-escalation of breast and axillary surgery in exceptional responders to neoadjuvant systemic treatment. Lancet Oncol 22:435–436. https://doi.org/10.1016/S1470-2045(21)00057-7
Morrow M, Winer EP (2020) De-escalating breast cancer surgery-where is the tipping point? JAMA Oncol 6:183–184. https://doi.org/10.1001/jamaoncol.2019.4849
Newman LA (2021) Safety of breast-conserving surgery in breast cancer and risk of overtreatment vs undertreatment. JAMA Surg 156:638–638. https://doi.org/10.1001/JAMASURG.2021.1450
Chu QD, Hsieh MC, Lyons JM, Wu XC (2021) 10-year survival after breast-conserving surgery compared with mastectomy in Louisiana women with early-stage breast cancer: a population-based study. J Am Coll Surg 232:607–621. https://doi.org/10.1016/J.JAMCOLLSURG.2020.11.011
Wrubel E, Natwick R, Wright GP (2021) Breast-Conserving Therapy is Associated with Improved Survival Compared with Mastectomy for Early-Stage Breast Cancer: A Propensity Score Matched Comparison Using the National Cancer Database. Ann Surg Oncol 28(2):914–919. https://doi.org/10.1245/s10434-020-08829-4
de Boniface J, Szulkin R, Johansson ALV (2021) Survival after breast conservation vs mastectomy adjusted for comorbidity and socioeconomic status: a Swedish national 6-year follow-up of 48 986 women. JAMA Surg 156:628–637. https://doi.org/10.1001/JAMASURG.2021.1438
Demographic and economic trends in urban, suburban and rural communities|Pew Research Center. https://www.pewresearch.org/social-trends/2018/05/22/demographic-and-economic-trends-in-urban-suburban-and-rural-communities/. Accessed 14 Feb 2022
Is North Carolina Rural or Urban?|NC OSBM. https://www.osbm.nc.gov/blog/2020/11/19/north-carolina-rural-or-urban. Accessed 14 Feb 2022
Urbanization Trends|Carolina Demography. https://www.ncdemography.org/2015/01/05/urbanization-trends/. Accessed 14 Feb 2022
NC in Focus: Shifting from small towns to larger cities|Carolina Demography. https://www.ncdemography.org/2016/03/10/nc-in-focus-shifting-from-small-towns-to-larger-cities/. Accessed 14 Feb 2022
Funding
ML Roberson was supported by a Robert Wood Johnson Foundation Health Policy Research Scholars Award and an Agency for Healthcare Research dissertation research award R36HS027299. The database infrastructure used for this project was supported through the UNC Lineberger Comprehensive Cancer Center, University Cancer Research Fund via the State of North Carolina.
Author information
Authors and Affiliations
Contributions
MLR contributed to study design; data acquisition, analysis, and interpretation; and writing of the original draft. HBN contributed to study design; data interpretation; and writing, reviewing, and editing of the manuscript. SBW contributed to data acquisition and interpretation and writing, reviewing, and editing of the manuscript. KERH contributed to data acquisition and interpretation and writing, reviewing, and editing of the manuscript. AFO contributed to data acquisition and interpretation and writing, reviewing, and editing of the manuscript. WRR contributed to study design; data interpretation; and writing of the original draft.
Corresponding author
Ethics declarations
Conflict of interest
Drs. Wheeler and Reeder-Hayes received unrelated grant funding from Pfizer paid to their institution in the past 3 years. The other authors declare that they have no conflict of interest.
Ethical approval
This investigation was approved by the Institutional Review Board (IRB) at the University of North Carolina at Chapel Hill (IRB # 18-3552).
Consent to participate
We received a waiver of informed consent through the University of North Carolina at Chapel Hill IRB. This waiver was received because this is an investigation of secondary data originally collected for other than research purposes. This secondary data analysis represented no more than minimal risk to participants, the waiver did not adversely affect the rights and welfare of participants, and the research could not practicably be carried out without the waiver. There was no direct communication or contact with the participants in this investigation.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Roberson, M.L., Nichols, H.B., Olshan, A.F. et al. Trends in surgical treatment of early-stage breast cancer reveal decreasing mastectomy use between 2003 and 2016 by age, race, and rurality. Breast Cancer Res Treat 193, 445–454 (2022). https://doi.org/10.1007/s10549-022-06564-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-022-06564-w