Abstract
We studied the effect of mountain grassland afforestation with conifer trees (Pinus sylvestris, Picea abies and Pinus cembra) on soil organic matter (SOM) cycling and carbon (C) isotopic composition in two contrasting climate areas using a regional approach. Seventeen paired sites (each containing at least 40 years prior afforested and grassland plots) were investigated in the mountains of Central Spain and Western Austria. Topsoil CO2 effluxes were monitored under standardized conditions for six months as a proxy for soil organic carbon (SOC) mineralisation. The bulk C and nitrogen (N) concentrations and their isotopic composition in the soil and in the plants were assessed. The soil C:N ratio was consistently greater after afforestation in both regions, which in Spain was caused by a significant decrease in N concentration. No consistent effect was found on mineralisation rates due to vegetation change. Afforestation produced a more consistent soil 13C enrichment in the Spanish than in the Austrian sites. Our work strongly suggests that increasing altitude in Mediterranean mountain grasslands alleviates water limitation, favouring both plant growth and SOM decomposition, and ultimately accelerating C cycling. In contrast, temperate grassland areas at high altitudes were associated with severe temperature limitations, which constrained SOM transformation processes. In spite of the impact of afforestation on soil biogeochemical processes, C concentrations were marginally affected. We therefore conclude that grassland conversion to coniferous forests does not enhanced C sequestration in the mineral soil, for at least 40 years after land-use change.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Soils store twice as much organic carbon (C) as the atmosphere (Ciais et al. 2013), and represent the largest reservoir of C in terrestrial ecosystems. In this context, land use and land-use changes are key factors in the balance between soil organic C (SOC) stocks and atmospheric CO2 (Houghton et al. 2012). Afforestation is one of the major land-use changes currently taking place in Europe, mainly driven by the promotion of afforestation by environmental policies for containing rural depopulation (MacDonald et al. 2000; Marey-Pérez and Rodríguez-Vicente 2009). Afforestation may contribute to mitigating atmospheric CO2 concentration due to the increase in C stored in the vegetation (Powlson et al. 1998; Smith et al. 2014), but its total effect at the ecosystem level also depends on the net effect on SOC (Poeplau et al. 2011), since even minor changes in the equilibrium between soil C inputs and outputs may imply huge emissions into the atmosphere.
Vegetation shifts may affect above- and belowground C dynamics in different ways. Whereas an encroachment of woody species in grasslands usually increases aboveground C pools (Risch et al. 2008; Pérez-Cruzado et al. 2014), its effect on belowground C is still unclear. The capacity of soils to store organic C is simultaneously driven by environmental and biotic factors, which together with the dynamics between soil organic matter (SOM), mineral surfaces and aggregates, modulate rates of SOM mineralisation and degrees of protection (Lehmann and Kleber 2015). There have been reports of increases (Liao et al. 2006; Wang et al. 2013a), losses (Berthrong et al. 2009; Hiltbrunner et al. 2013; Guidi et al. 2014) and no changes (McCarron et al. 2003; Risch et al. 2008; Poeplau and Don 2013) in SOC stocks in mineral soils following the afforestation of former grasslands. This is likely the result of the huge variation in site-specific characteristics such as climate, vegetation type and composition, plant traits (e.g. roots) and soil (Post and Kwon 2000; Jackson et al. 2002; Poeplau et al. 2011). The search for common patterns of SOC changes following afforestation is today a pressing issue. Forest expansion with coniferous species has been associated with changes in above- and belowground litter quality and quantity (Pérez-Cruzado et al. 2014), which represents the substrate for microbial decomposition in the soil. The invasion of grass-dominated ecosystems by woody plants has been related to the slower decomposition rates of the litter originated from trees than from pasture lands (Hiltbrunner et al. 2013), as a result of the higher lignin content of conifer-litter (Berg 2000). However, recent approaches suggest that the chemical quality of C inputs plays a secondary role compared to environmental conditions, biotic controls and, stabilisation processes (Lehmann and Kleber 2015). Further, the observed decrease in total microbial biomass under forest soils (Macdonald et al. 2009) may lead to lower SOM decomposition rates than pasture lands (Ross et al. 2002; Hiltbrunner et al. 2013). Finally, afforestation with conifers may also imply a reduction in soil nitrogen (N) contents and exchangeable cations caused by the greater plant demand due to the increased biomass production compared to grasslands (Berthrong et al. 2009).
Afforestation may cause changes in the mineralisation rates of more accessible and easily decomposable SOM fractions, whereas more protected fractions may be better preserved. A good approach for quantifying the easily decomposable SOM pool is by measuring SOM mineralisation rates using laboratory incubations (Paul et al. 2001). Studies of natural isotopic abundance in the soil have also been applied to enhance the understanding of the processes involved in SOM decomposition. This research uses the fractionation processes involved in the biological mineralisation of vegetation residues –known as kinetic fractionation– which occur when soil microbes discriminate isotopically during the mineralisation of organic matter and release 13C-depleted CO2, enriching the remaining substrate (Mariotti et al. 1981; Högberg et al. 2005). A higher abundance of heavy stable isotopes in the soil has been associated with enhanced SOM decomposition (Tiunov 2007; Werth and Kuzyakov 2010). The isotopic signature of N in SOM has also been found to be positively correlated to the degree of SOM mineralisation (Nadelhoffer et al. 1996; Liao et al. 2006). Unlike the dynamic of soil C stable isotopes, several of the soil N transformation processes—nitrification, denitrification and ammonia volatilisation—discriminate in favour of the lightest isotope (Högberg 1997); consequently, 15N variations after afforestation may be related to N losses (Högberg and Johhannisson 1993), open N cycle (Vitousek et al. 1989) and soil N saturation (Emmett et al. 1998). Changes in microbial decomposition processes may also be related to the presence of a canopy structure due to afforestation, modifying microclimatic conditions by lengthening the periods of snow cover (Köck et al. 2002) that buffer harsh winter conditions (Kammer et al. 2009), softening annual soil temperatures (Hiltbrunner et al. 2013), and varying soil moisture regimes due to interception (Le Maitre et al. 1999). All these changes in microbial decomposition conditions may ultimately be reflected in the isotopic composition of the SOM after afforestation.
In recent decades, afforestation of mountain grasslands has occurred on a global scale (Jackson et al. 2002; Berthrong et al. 2009). This is particularly the case in Spanish (Poyatos et al. 2003; Camarero and Gutiérrez 2004) and Austrian mountain regions (Dullinger et al. 2004; Tasser et al. 2007), following the intensification of livestock production and economic constraints which have driven smallholders’ farmers in mountainous regions out of business, together with the afforestation programmes promoted by European governments. The growing body of information on the effect of forest expansion on SOM has produced contrasting and highly site-dependent results (Guo and Gifford 2002). The aim of this work is to determine common patterns caused by afforestation of grasslands on SOM features in two European mountainous areas with contrasting climates (Mediterranean and temperate). We sampled soil from 17 paired sites, all containing grassland and afforested plots. We determined bulk C and N concentrations and their stable isotope composition from soil and vegetation, and estimated SOC decomposition rates under standardised conditions in the laboratory for six months. We hypothesised that afforestation leads to changes (1) in SOC and total N concentrations, (2) in SOC mineralisation rates, and (3) in the signatures of soil 13C and 15N in the soil; and (4) that these changes are climate-dependent.
Materials and methods
Site description and experimental design
Two different mountain regions were selected: the Sistema Central range in Spain, and the Central Alps in Austria (Supplementary Fig. 1). The Sistema Central range is characterised by a sub-Mediterranean climate (Walter 1985) with warm dry summers and high year-round temperature fluctuations, although areas of high elevation are snow-covered throughout the winter (García Romero and Muñoz Jiménez 2010). The Central Alps region has a temperate mountain climate (Walter 1985) with cool summers, frequent rainfall and long periods of snow cover.
Sites were set up in locations where afforestation of former grasslands had occurred at least 40 years prior to soil sampling, and grasslands were still present in the vicinity and under the same soil type and position within the landscape. This time span is sufficient to assure that the short-term effects caused directly by forest operations on SOM dynamics have already passed (Poeplau and Don 2013), allowing us to evaluate changes due to the vegetation shift in the longer run. In most cases, afforestation was man-made and conducted between 1950 and 1970 in small patches of private forest, with the aim of increasing forest cover share and wood production while keeping grasslands present to some extent. Ten sites were selected in Spain and seven in Austria (Supplementary Fig. 1) (Table 1). Three replicated paired plots were established on each site, separated by 25–50 m. Each paired plot (20 × 20 m) consisted of an afforested plot with a closed tree canopy and an adjacent grassland plot. Spanish sites were located on granite and gneiss bedrock (Table 1) with soil pH values of the mineral A-horizon between 4.0 and 5.8. Soil texture ranged from loamy sand to sandy clay loam (Supplementary Table 1) and soils were classified as Cambisols and Umbrisols (Table 1, IUSS Working Group WRB 2014). The tree species in the Spanish forests was Scots pine (Pinus sylvestris L.), and grasslands were mainly formed by Festuca curvifolia (Lag. ex Lange) and Nardus stricta L. Grazing in the Spanish grasslands is almost non-existent and occurs only in summer—fewer than 0.25 livestock units per ha. Austrian sites were located on different soil substrates (Table 1) with A-horizon pH ranging from 3.4 to 6.4. The textures were sandy loam and sandy clay (Supplementary Table 1); soils were classified as Podzols, Cambisols and Leptosols (Table 1, IUSS Working Group WRB 2014). Austrian afforested plots included Norway spruce (Picea abies (L) Karst), European larch (Larix decidua Mill.) and Swiss pine (Pinus cembra L.); dominant grassland species were Carex sempervirens Vill. and Nardus stricta L. The Austrian grasslands are moderately grazed by cattle—0.5 to 1 livestock units per ha—for a period of 6–10 weeks during summer. The main features of the study sites are shown in Table 1.
Soil and plant material sampling
We sampled soil material from the mineral A-horizon from all the plots. We used genetic horizons instead of fixed soil depth sampling, as the genetic horizon system offers better comparability of the processes involved in SOC dynamics (Grüneberg et al. 2010), and the A-horizon stores most of the soil C (Jobbágy and Jackson 2000; Chiti et al. 2012). Mean A-horizon depth was 24.7 ± 9.8 and 23.3 ± 6.8 cm for forests and grasslands in Spain respectively, and 11.4 ± 4.0 and 10.6 ± 4.3 cm for forests and grasslands in Austria. Organic layer was present in afforested plots only, and was not considered in this study; therefore, it was completely removed before soil sampling. A composite soil sample was taken from the A-horizon from six randomly distributed spots in each vegetation plot. A small soil pit (20 × 20 × 30 cm) was dug at each spot, and a soil sub-sample was taken from the entire depth of the A-horizon with a garden shovel. Soil samples were transferred to the laboratory, roots were removed by hand and the soil was sieved (2 mm). Fresh material from the standing vegetation (needles from coniferous species and leaves from herbaceous species) was collected in each plot in the soil sampling location in Spain, and a composite sample for each site in Austria.
Estimation of potential SOC mineralisation efflux
CO2 efflux from soil samples was monitored in the laboratory as a measure of mineralised C. 20 g fresh weight of homogenised A-horizon soil from each plot (n = 102) was placed inside 150 ml glass bottles. The soil samples were set at 60% of water holding capacity and pre-incubated during two weeks at 10 °C in a thermostatic cabinet (Lovibond®, Amesbury, United Kingdom). This pre-incubation period permitted the equilibration of soil samples and avoided abrupt CO2 production pulses following soil disturbance caused by soil sampling and sieving (Schindlbacher et al. 2010). After the equilibration phase, the temperature inside the cabinet was increased to 20 °C and maintained throughout the whole duration of the experiment in order to achieve maximum respiration rates and highlight differences between the sampling sites (Schinner et al. 1993).
The bottles with the soil samples were kept continuously inside the incubation cabinets and covered with Parafilm®. This allowed outgassing of CO2 from the soil atmosphere with no significant desiccation of the soil samples. An air circulation system prevented CO2 from accumulating in the headspace, which could otherwise interfere in the soil-headspace CO2 diffusion process. The moisture levels of the soil samples were maintained by periodic addition of a standard rain mixture (Schierl 1991). To determine the soil-headspace CO2 efflux, a non-dispersive infrared sensor (MSH-P-CO2, Dynament Ltd., Derbyshire, UK) was placed inside each bottle and closed tightly. The sensor produced a voltage readout every 10 s that was transformed into CO2 concentrations. Following the non-flow-through non-steady-state technique (Livingston and Hutchinson 1995), the change in CO2 concentration over time was used to infer the soil CO2 production rate [mg CO2–C h–1 kg–1 soil dry weight (sdw)]. The sensors were calibrated weekly with four different calibration mixtures (400, 580, 1000 and 2000 ppm CO2, AirLiquide, Germany).
SOC mineralisation rates were monitored periodically for six months, with a higher temporal resolution at the beginning of the experiment, which was then lowered as the SOC mineralisation rate started to level off. Cumulative SOC mineralisation effluxes for the whole incubation period were estimated by linear interpolation of consecutive measurements. An estimation of SOC decomposability (mg CO2–C g–1 SOC, Díaz-Pinés et al. 2014) was obtained by normalising total CO2 by SOC content in the soil.
C and N concentration and stable isotope analysis (δ13C and δ15N)
Soil (n = 102) and plant (n = 74) samples were oven-dried (40 °C) and ground (<100 µm) prior to analysis. Samples with pH higher than 6 were acid-treated to check for carbonate presence, and no traces of carbonates were found for any of the sites. SOC and total N concentration and C and N isotopic composition of plant and soil samples were determined using continuous-flow isotope ratio mass spectrometry in a Costech ECS 4010 Elemental Analyzer (Costech Analytical Technologies, Inc., Valencia, CA, USA) fitted with a zero-blank auto-sampler coupled via a ConFloIII to a ThermoFinnigan DeltaPlus-XL (Thermo Scientific, Waltham, MA, USA). Isotopic compositions were expressed in δ notation, representing the ‰ variation from the standard reference material:
where R is the ratio 13C/12C or 15N/14N. The standards used were Vienna Pee Dee Belemnite and atmospheric air-N2 respectively. Precision (in terms of standard deviation) in internal standards for elemental C and N abundances were over ± 0.3 and 0.1‰ respectively.
The enrichment of heavy isotopes in soils compared to plant was used as an indicator of isotopic discrimination caused by SOM decomposition (Michener and Lajtha 2007).
where v refers to the plant material and s to the soil. We considered that values from fresh leaves could avoid biases due to possible discrimination that may already be occurring in the leaf fall caused by partial decomposition when the leaves are on the forest floor (Garten et al. 2000); we also assumed that isotopic fractionation due to translocation of nutrients prior to leaf senescence and fall was marginal (Balesdent et al. 1993). A negative Δ value indicates enrichment in the isotopic composition of the SOM compared to the vegetation.
We also calculated the difference in the composition of the δ13C stable isotope between afforested and grassland soils in each paired plot as a proxy for the change in the isotopic composition due to forest encroachment:
Statistical analyses
The soil variables studied were tested for differences between land-use types (afforested, grassland) at the regional scale using general linear mixed models (nlme package, Pinheiro et al. (2016)), with vegetation as a fixed factor and site as a random factor, to include within-site correlations in the model. Normality and homogeneity of the variance of the residuals were checked by the Shapiro–Wilk (stats package, R Core Team (2015)) and Levene test (car package, Fox and Weisberg (2011)) respectively. Correlations and linear regression were performed between the variables of interest and the site altitude (stats package, R Core Team (2015)). If normality of residuals was met, Pearson product-moment correlation coefficient was used (r); otherwise, correlations were explored with Spearman’s rank correlation coefficient (ρ). Significance level was 0.05 for all tests. Results are expressed as mean ±1 SD. All statistical analyses were carried out in R [version 3.2.3, R Core Team (2015)].
Results
Relationships between altitude, mean annual temperature and mean annual precipitation
In Spain, altitude showed a close negative correlation (\({\text{R}}_{\text{adj}}^{2}\) = 0.98) with mean annual temperature (MAT), and a positive correlation (\({\text{R}}_{\text{adj}}^{2}\) = 0.35) with mean annual precipitation (MAP) (Supplementary Fig. 2). In Austria, the same negative trend was found between altitude and MAT (\({\text{R}}_{\text{adj}}^{2}\) = 0.11); but a negative correlation was found between MAP and altitude (\({\text{R}}_{\text{adj}}^{2}\) = 0.26) (Supplementary Fig. 2).
SOC concentration, total N concentration and soil C:N ratio
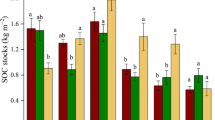
There was no significant effect of afforestation on the SOC concentration in Spanish plots (76 ± 34 and 75 ± 28 g C kg−1 for afforested and grassland respectively) or in Austrian plots (111 ± 46 g kg−1 for afforested and 91 ± 29 g kg−1 for grassland) (Fig. 1a). Total N in grassland plots was significantly higher than in afforested ones (5.8 ± 2.2 vs. 4.6 ± 2.5 g N kg−1; p < 0.01) (Fig. 1b) in the Spanish sites. In Austria, total N concentration was similar in grassland and afforested plots (6.1 ± 2.1 vs. 5.7 ± 1.9 g N kg−1) (Fig. 1b). Soil C:N ratio was significantly higher in afforested plots (18.2 ± 5.7) than in grassland plots (13.0 ± 1.3) in the Spanish sites (p < 0.001, Fig. 1c). Austrian sites showed a similar trend (19.6 ± 5.1 vs 15.5 ± 3.3, p < 0.01) (Fig. 1c). Supplementary Table 1 shows the soil features of individual sites. Weak positive relationships between altitude and SOC concentrations (\({\text{R}}_{\text{adj}}^{2}\) = 0.13; p < 0.05), and total N (\({\text{R}}_{\text{adj}}^{2}\) = 0.13; p < 0.05) were found in Spanish grasslands. Negative relationships were observed in Austrian grasslands between altitude and SOC concentration (\({\text{R}}_{\text{adj}}^{2}\) = 0.39; p < 0.01) and total N (\({\text{R}}_{\text{adj}}^{2}\) = 0.69; p < 0.001). In Austria, altitude was positively correlated with C:N ratio in both the grassland (\({\text{R}}_{\text{adj}}^{2}\) = 0.64; p < 0.001) and the afforested sites (\({\text{R}}_{\text{adj}}^{2}\) = 0.26; p < 0.01). No significant correlations between MAT or MAP and SOC, N and C:N were found across the dataset for both grassland and afforested areas in Spain and Austria.
Mean values (±1 SD) of soil organic carbon (SOC) concentration (a), Soil total nitrogen (N) concentration (b), C:N ratio (c) in the Sistema Central in Spain and the Central Alps in Austria. Different letters denote significant differences (p < 0.05) between grasslands and afforested plots in each region
Potential SOC mineralisation efflux
On average, SOC mineralisation efflux rates in Spain were 0.18 ± 0.12 mg CO2−C h−1 kg−1 sdw for afforested plots, and 0.14 ± 0.10 mg CO2–C h−1 kg−1 sdw for grassland plots (n.s.). The mean SOC mineralisation efflux rates in Austrian afforested and grassland plots were 0.35 ± 0.21 and 0.33 ± 0.26 mg CO2–C h−1 kg−1 sdw respectively. Despite some individual significant differences in SOC mineralisation efflux rates at the site level (Supplementary Table 2), there was no significant effect of afforestation on SOC mineralisation efflux.
Mineralised C throughout the whole incubation period (Fig. 2) was not significantly different between afforested and grassland plots in Spain (724 ± 371 and 586 ± 308 mg CO2−C kg−1 sdw for afforested and grassland respectively) and Austria (1408 ± 644 and 1316 ± 795 mg CO2−C kg−1 sdw for forests and grasslands respectively). By the end of the incubation period, Austrian soils had emitted approximately twice as much CO2 as Spanish soils.
The values of cumulative SOC decomposability throughout the whole incubation period were 11.3 ± 7.0 and 8.4 ± 4.0 mg CO2–C g−1 SOC for Spanish afforested and grassland plots respectively (n.s.). Decomposability was significantly correlated with MAT (ρ = 0.57; p < 0.01), MAP (ρ = −0.75; p < 0.001), and with C:N ratio (ρ = 0.60; p < 0.01), but not with C or N concentrations. In the Austrian soils, these values were 14.3 ± 7.0 and 14.4 ± 6.7 mg CO2–C g−1 SOC for afforested and grassland plots respectively (n.s.). However, in Austria there was no correlation between cumulative SOC decomposability and any other variable.
Stable isotopic composition in soil and plant material
In Spain, mean δ13C values were more negative in pine needles than in grasses (−28.2 ± 1.4 vs. −27.3 ± 0.7‰ respectively). In Austrian sites there were not differences between afforested and grassland plots (−29.3 ± 0.9 vs. −28.4 ± 0.7‰ for needles and grasses respectively). Mean soil δ13C was significantly less negative (p < 0.05) in afforested than in grassland plots in Spanish sites (Fig. 3) but not in Austrian sites. Mean soil δ15N values were significantly higher (p < 0.001) in grasslands than in afforested plots in Spain (5.3 ± 1.5 and 3.3 ± 1.0) (Fig. 3) but not in Austria (2.7 ± 1.4 and 2.5 ± 1.1‰).
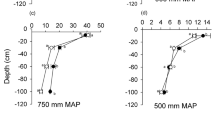
Soil δ13C showed a significant positive relationship with altitude in Spain in both afforested and grassland plots (Fig. 4). In Austria, soil δ13C only showed a significant positive relationship with altitude in grasslands (Fig. 4). Plant material δ13C was significantly correlated with altitude, positively in Spanish grasslands and afforested plots and in Austrian grasslands, but negatively in Austrian afforested plots (Fig. 4). In Spain, soil δ13C was correlated with MAP (ρ = 0.59; p < 0.01) and MAT (r = −0.71; p < 0.001); and plant δ13C correlated to MAT (ρ = −0.47; p < 0.05) in Spain. Plant material δ13C was positively correlated with the δ13C in mineral soil in Spanish and Austrian grasslands, and in Austrian afforested plots (Fig. 5).
Mean Δ13C values were significantly less negative in grasslands than in afforested plots (−1.4 ± 0.5 vs. −2.8 ± 1.4‰ Spain and −1.9 ± 0.6 vs. −2.8 ± 0.5‰ Austria). Mean Δ15N values in Spain showed significant differences (p < 0.01) between afforested (−5.3 ± 1.3‰) and grassland plots (−4.3 ± 2.3‰). Δ15N values in Austria were highly variable between sites and no significant differences were found between afforested (−5.1 ± 2.0‰) and grassland plots (−6.0 ± 2.8‰). Lower altitudes led to an enrichment of soil δ13C following afforestation, whereas high altitude led to depletion, as shown in the relationship between ΔAf−G and altitude (Fig. 6).
Discussion
Our experimental approach focused on the effect of the afforestation of mountain grasslands on SOM dynamics at the regional level. The study sites encompassed a wide range of environmental conditions, including differences in altitude, species composition and—in the case of Austria—bedrock material (Table 1). It was therefore within our expectations that the variability between the sites would influence SOM dynamics in addition to the afforestation itself (Guo and Gifford 2002; von Lützow et al. 2006; Schindlbacher et al. 2010). However, our aim was to identify common patterns in SOM changes due to afforestation, which would outweigh differences due to specific site conditions.
In agreement with previous findings (Risch et al. 2008; Wang et al. 2013b), we observed no consistent effect of converting grassland to forest on mineral SOC concentrations. SOC content is the net result of C inputs and outputs in the system, both of which may be altered following afforestation. Changes in C inputs are mainly caused by alterations in the quantity of above- and belowground litterfall (Pérez-Cruzado et al. 2012), whereas C losses due to mineralisation tend to be affected by litterfall quality, microclimatic conditions and the associated microbial community (Aerts 1997; Macdonald et al. 2009; Cleveland et al. 2014; Nazaries et al. 2015) which in combination may ultimately modify SOC mineralisation processes. Our results showed a (non-significant) trend of higher C concentrations in Austrian forests, likely due to higher C inputs in a closed-canopy forest compared to grasslands. Together with the presence of an organic layer in the forest (storing additional 10–30 Mg C ha−1, unpublished data), this suggests a higher C storage rate in the topsoil following the change in land use from grassland to forest. Afforestation very consistently increased soil C:N ratios in both climate areas; this generalisation has not been confirmed at a regional scale so far and—to our knowledge—was only observed locally (Berthrong et al. 2009). Litter from coniferous species has a wide C:N ratio and a chemically complex structure (Berg 2000; Thuille and Schulze 2006; Pérez-Cruzado et al. 2014), usually leading to low relative N availability for microbial nutrition and a high biochemical recalcitrance of SOC. Enhanced N demand by trees (Smal and Olszewska 2008) compared to pasture may have decreased soil N concentrations. Overall, our hypothesis that afforestation implies changes in SOC and N concentrations was consistently confirmed with regard to N concentrations, whereas SOC results were somehow less clear.
Oppositely to our expectation that SOC mineralisation rates change after afforestation, we found no effect, as previously reported elsewhere (Hiltbrunner et al. 2013), and only a marginally higher decomposability of SOC at forest sites. Further, effects of climatic features on decomposability were observed at the Spanish sites, only. Our experimental approach used standardised conditions in the laboratory for the mineralisation of SOC, in pursuit of a mechanistic understanding, and as a result may have lost some power of extrapolation and representativeness, as suggested from the observed decoupling between decomposition rates and SOC concentrations. We cannot therefore rule out the possibility that the heterotrophic soil CO2 efflux under field conditions may differ between afforested and grassland plots due to altered soil temperature and snow-cover patterns caused by canopy effects (Köck et al. 2002; Kammer et al. 2009) and changed soil moisture regimes (Le Maitre et al. 1999). The markedly higher CO2 emissions and decomposition rates observed in Austrian soils may be due to temperature constraints on the site, which preserve higher amounts of labile C in the Alpine temperate region compared to Mediterranean mountain areas.
The natural abundance of stable isotopes has been used to estimate the extent to which SOM dynamics are affected by different land uses (e.g. Michener and Lajtha 2007), and there is usually a correspondence between plant and soil isotopic signatures (Bird et al. 1994; Boutton et al. 1998). With regard to our hypothesis that afforestation leads to changes in the soil isotopic signature, we observed changes in soil δ13C values following afforestation in most of the sites (Supplementary Table 3), suggesting a gradual replacement of grass-origin SOC by tree-origin SOC (Hiltbrunner et al. 2013), although the shift is also interactively affected by climate conditions. The subtle differences between 13C signatures in grasses and needles prevents a clear differentiation, as would be the case with a clear transition from C3 to C4 plants. It is also likely that a substantial proportion of the SOC in the afforested plots is still from grass origins, and only a small fraction of coniferous litter has actually contributed to the new SOC pool 40 years after afforestation (Richter et al. 1999).
The relative depletion of 15N in Mediterranean soils after afforestation may be related to recent organic matter inputs to the soil (Liao et al. 2006) and a progressive biogeochemical degradation (Tiunov 2007) of N-forms in grasslands, leading to a loss of 15N-depleted forms (e.g. Pörtl et al. 2007). The identification of the specific biogeochemical processes responsible for the different δ15N is highly speculative for two main reasons: (i) the multiple processes discriminating against 15N at different rates (Michener and Lajtha 2007); and (ii) the fact that we only analysed the δ15N of the bulk soil, strongly limiting our explanatory power (Pörtl et al. 2007; Craine et al. 2015). High soil δ15N values such as those found in grasslands have been associated with a high availability of N (Martinelli et al. 1999) and open N cycles (Eshetu 2004; Awiti et al. 2008). This is similar to the pattern seen when Spanish and Austrian soil δ15N values were compared.
We had hypothesized that climate would affect SOM changes following afforestation. Contrary to our expectations, MAT or MAP had a poor explanatory power with regard to the effects of land use change on soil features. On the contrary, altitude was found to be a key factor influencing SOM dynamics. Altitude jointly modulates temperature and precipitation in each geographic region, and these are important parameters in the production of above- and belowground organic matter (Hitz et al. 2001), its decomposition (Rodeghiero and Cescatti 2005) and its final accumulation (Post et al. 1982). Increasing altitude led to a significant rise in SOC and total N concentrations in Spanish grasslands (estimated as 5 g C kg−1 100 m−1 and 0.4 g N kg−1 100 m−1), but a significant decrease in Austrian grasslands (estimated as 9 g C kg−1 100 m−1 and 0.8 g N kg−1 100 m−1). This contrasting effect of altitude on C and N concentrations has already been identified, including both increases (Leifeld et al. 2009; Schindlbacher et al. 2010) and decreases (Garcia-Pausas et al. 2007; Gutiérrez-Girón et al. 2015). High-altitude areas in Spain were associated to high MAP, and were therefore less water-limited during the growing season compared to low-lying areas, which had less total precipitation and higher evapotranspiration rates. This therefore produces a gradient of higher summer plant productivity in Mediterranean mountains as the altitude increases. Microbial decomposition at high altitudes is also constrained by low temperatures outside the growing period (Simmons et al. 1996), which explains the greater C and N contents found in the highest Mediterranean areas studied. Opposing patterns have been detected in Mediterranean areas but at altitudes well above those in our study (Gutiérrez-Girón et al. 2015). In the Alpine region the complex topography and regional effects on climate were probably responsible for the negative relationship between altitude and MAP. Summer drought is not a crucial limiting factor in temperate mountain regions, and plant productivity is mainly regulated by temperature, leading to shorter growing seasons at high altitudes (Djukic et al. 2010). This therefore produces a downward gradient of plant productivity in temperate mountains as the altitude increases.
As expected, altitude had a clear effect on C isotopic signatures. It is well documented that increasing altitude diminishes the ratio of internal to external partial pressure of CO2 in leaves, which in turn causes lower plant 13C discrimination (e.g. Körner et al. 1991). Low temperatures in high altitudinal areas may also influence plant 13C discrimination due to physiological effects on the plants that cause an increase in leaf thickness and higher carboxylation efficiency (Körner 1989; Körner et al. 1991). Nonetheless, the investigation of the variety of factors (e.g. water availability, light radiation, physiological and metabolic processes) that interactively determine isotopic composition in plant tissues (Brüggemann et al. 2011) is beyond the scope of this work, which is primarily focused in the mechanisms that cause changes in the stable isotopic signature of the soil. The change in soil δ13C after afforestation is determined by the rate of incorporation of litter-C from the trees into the soil, the isotopic composition of the tree litter, and the change in the discrimination during the mineralisation of this C debris (Liao et al. 2006; Werth and Kuzyakov 2010). Enhanced mineralisation rates in afforestation would lead to higher discrimination and thus to a positive change in soil δ13C due to afforestation, provided all other parameters remain unchanged. Figure 6 shows that the SOC becomes (slightly) 13C enriched after afforestation at lower altitudes, and is isotopically depleted at higher altitudes. This suggests that SOC mineralisation—inferred from greater soil δ13C—is higher in forests at low altitudes and in pastures at high altitudes. It can therefore be speculated that the mechanisms of C replacement and stabilisation after afforestation may vary with climate conditions (as defined by altitude), with the watershed point located at lower altitudes in the Alpine region (ca. 1775 m a.s.l.) than in Mediterranean mountains (ca. 1900 m a.s.l.). Our contention relies on very small (but highly significant) differences in δ13C and on a number of assumptions; further, we were not able to corroborate this pattern with our decomposability results, or even SOC concentrations. Nevertheless, the relationship between the soil isotopic pattern and elevation offers a promising working hypothesis whereby biogeochemical pathways may be differently affected by land-use change depending on altitude and its associated climatic conditions. These thresholds suggest the idea of an optimal altitude where the plant communities in the study and their associated microorganisms undergo a change in their habitat conditions—mainly climate-related—which impedes normal development, although they may potentially be found above or below these altitudes. In Spain, the threshold for mineralisation and stabilisation is in line with the current altitudinal treeline for Scots pine in the Sistema Central (Cañellas et al. 2000), and we therefore recommend further studies to address this issue.
Conclusions
Afforestation of former grassland areas in Mediterranean and Alpine mountains consistently led to a greater soil C:N ratio, suggesting changes in the SOM quality of the topsoil, although potential mineralisation was not influenced by afforestation. Soil isotopic signatures were more markedly influenced by afforestation in the Mediterranean mountains. Increasing altitude likely stimulated biogeochemical processes in the Mediterranean sites due to the alleviation of water constraints; conversely, temperate sites showed indicators of functioning constrains at higher altitudes due to low temperatures. Since the effects of afforestation on total SOC concentrations were negligible, we conclude that afforestation of mountain grasslands does not enhance C sequestration in the mineral soil, in both Mediterranean and temperate areas, for at least the first 40 years after the land use change. However, more research is required to test whether this trend persists over time and whether expected climatic changes will affect the SOM dynamics after afforestation.
References
Aerts R (1997) Climate, leaf litter chemistry and leaf litter decomposition in terrestrial ecosystems: a triangular relationship. Oikos 79:439. doi:10.2307/3546886
Awiti AO, Walsh MG, Kinyamario J (2008) Dynamics of topsoil carbon and nitrogen along a tropical forest-cropland chronosequence: evidence from stable isotope analysis and spectroscopy. Agric Ecosyst Environ 127:265–272. doi:10.1016/j.agee.2008.04.012
Balesdent J, Giradin C, Mariotti A (1993) Site-related δ 13C of tree leaves and soil organic matter in a temperate forest. Ecology 74:1713–1721
Berg B (2000) Litter decomposition and organic matter turnover in northern forest soils. For Ecol Manag 133:13–22. doi:10.1016/S0378-1127(99)00294-7
Berthrong ST, Jobbágy EG, Jackson RB (2009) A global meta-analysis of soil exchangeable cations, pH, carbon and nitrogen with afforestation. Ecol Appl 19:2228–2241. doi:10.1890/08-1730.1
Bird MI, Haberle SG, Chivas AR (1994) Effect of altitude on the carbon-isotope composition of forest and grassland soils from Papua New Guinea. Glob Biogeochem Cycles 8:13–22
Boutton TW, Archer SR, Midwood AJ et al (1998) δ13C values of soil organic carbon and their use in documenting vegetation change in a subtropical savanna ecosystem. Geoderma 82:5–41. doi:10.1016/S0016-7061(97)00095-5
Brüggemann N, Gessler A, Kayler ZE et al (2011) Carbon allocation and carbon isotope fluxes in the plant-soil-atmosphere continuum: a review. Biogeosciences 8:3457–3489. doi:10.5194/bg-8-3457-2011
Camarero JJ, Gutiérrez E (2004) Pace and pattern of recent treeline dynamics: response of ecotones to climatic variability in the Spanish Pyrenees. Clim Chang 63:181–200. doi:10.1023/B:CLIM.0000018507.71343.46
Cañellas I, García FM, Montero G (2000) Silviculture and dynamics of Pinus sylvestris L. stands in Spain. Investig Agrar Sist Y Recur For 1:233–253
Chiti T, Díaz-Pinés E, Rubio A (2012) Soil organic carbon stocks of conifers, broadleaf and evergreen broadleaf forests of Spain. Biol Fertil Soils 48:817–826. doi:10.1007/s00374-012-0676-3
Ciais P, Sabine C, Bala G, et al (2013) Carbon and Other Biogeochemical Cycles. In: Stocker TF, Qin D, Plattner G-K et al. (eds) Climate change 2013: The physical science basis. Contribution of Working Group I to the Fifth Assessment Report of the Intergovernmental Panel. Cambridge University, Cambridge pp 465–570
Cleveland CC, Reed SC, Keller AB et al (2014) Litter quality versus soil microbial community controls over decomposition: a quantitative analysis. Oecologia 174:283–294. doi:10.1007/s00442-013-2758-9
Craine JM, Brookshire ENJ, Cramer MD et al (2015) Ecological interpretations of nitrogen isotope ratios of terrestrial plants and soils. Plant Soil 396:1–26. doi:10.1007/s11104-015-2542-1
Díaz-Pinés E, Schindlbacher A, Godino M et al (2014) Effects of tree species composition on the CO2 and N2O efflux of a Mediterranean mountain forest soil. Plant Soil 384:243–257. doi:10.1007/s11104-014-2200-z
Djukic I, Zehetner F, Tatzber M, Gerzabek MH (2010) Soil organic-matter stocks and characteristics along an Alpine elevation gradient. J Plant Nutr Soil Sci 173:30–38. doi:10.1002/jpln.200900027
Dullinger S, Dirnböck T, Grabherr G (2004) Modelling climate change-driven treeline shifts: relative effects of temperature increase, dispersal and invasibility. J Ecol 92:241–252. doi:10.1111/j.0022-0477.2004.00872.x
Emmett BA, Kjønaas OJ, Gundersen P et al (1998) Natural abundance of 15N in forests across a nitrogen deposition gradient. For Ecol Manag 101:9–18. doi:10.1016/S0378-1127(97)00121-7
Eshetu Z (2004) Natural 15N abundance in soils under young-growth forests in Ethiopia. For Ecol Manag 187:139–147. doi:10.1016/S0378-1127(03)00315-3
Fox J, Weisberg S (2011) Nonlinear regression and nonlinear least squares in R. An R companion to applied regression. Sage, Thousand Oaks, pp 1–20
García Romero A, Muñoz Jiménez J (2010) Modificaciones recientes de la cubierta nival y evolución de la vegetación supraforestal en la Sierra de Guadarrama, España: el Puerto de los Neveros. Cuad Investig Geográfica 36:109–143
Garcia-Pausas J, Casals P, Camarero L et al (2007) Soil organic carbon storage in mountain grasslands of the Pyrenees: effects of climate and topography. Biogeochemistry 82:279–289. doi:10.1007/s10533-007-9071-9
Garten CT, Cooper LW, Post WM, Hanson PJ (2000) Climate controls on forest soil C isotope ratios in the Southern Appalachian Mountains. Ecology 81:1108–1119. doi:10.1890/0012-9658(2000)081[1108:CCOFSC]2.0.CO;2
Grüneberg E, Schöning I, Kalko EKV, Weisser WW (2010) Regional organic carbon stock variability: a comparison between depth increments and soil horizons. Geoderma 155:426–433. doi:10.1016/j.geoderma.2010.01.002
Guidi C, Vesterdal L, Gianelle D, Rodeghiero M (2014) Changes in soil organic carbon and nitrogen following forest expansion on grassland in the Southern Alps. For Ecol Manag 328:103–116. doi:10.1016/j.foreco.2014.05.025
Guo LB, Gifford RM (2002) Soil carbon stocks and land use change: a meta analysis. Glob Chang Biol 8:345–360. doi:10.1046/j.1354-1013.2002.00486.x
Gutiérrez-Girón A, Díaz-Pinés E, Rubio A, Gavilán RG (2015) Both altitude and vegetation affect temperature sensitivity of soil organic matter decomposition in Mediterranean high mountain soils. Geoderma 237–238:1–8. doi:10.1016/j.geoderma.2014.08.005
Hasenauer H, Merganicova K, Petritsch R, Pietsch SA (2003) Validating daily climate interpolations over complex terrain in Austria. Agric For Meteorol 119:87–107
Hiltbrunner D, Zimmermann S, Hagedorn F (2013) Afforestation with Norway spruce on a subalpine pasture alters carbon dynamics but only moderately affects soil carbon storage. Biogeochemistry 115:251–266. doi:10.1007/s10533-013-9832-6
Hitz C, Egli M, Fitze P (2001) Below-ground and above-ground production of vegetational organic matter along a climosequence in alpine grasslands. J Plant Nutr Soil Sci 164:389–397. doi:10.1002/1522-2624(200108)164:4<389:AID-JPLN389>3.0.CO;2-A
Högberg P (1997) 15N natural abundance in soil-plant systems. New Phytol 137:179–203
Högberg P, Johhannisson C (1993) l5N abundance of forests is correlated with losses of nitrogen. Plant Soil 157:147–150
Högberg P, Ekblad A, Nordgren A (2005) Factors determining the 13C abundance of soil-respired CO2 in Boreal forests. Stable isotopes and biosphere-atmosphere interactions: processes and biological controls. Elsevier, Amsterdam, pp 47–68
Houghton RA, House JI, Pongratz J et al (2012) Carbon emissions from land use and land-cover change. Biogeosciences 9:5125–5142. doi:10.5194/bg-9-5125-2012
IUSS Working Group WRB (2014) World reference base for soil resources. International soil classification system for naming soils and creating legends for soil maps. World Soil Resources Reports 106:191
Jackson RB, Banner JL, Jobbágy EG et al (2002) Ecosystem carbon loss with woody plant invasion of grasslands. Nature 418:623–626. doi:10.1038/nature00910
Jobbágy EG, Jackson RB (2000) The vertical distribution of soil organic carbon and its relation to climate and vegetation. Ecol Appl 10:423–436. doi:10.1890/1051-0761(2000)010[0423:TVDOSO]2.0.CO;2
Kammer A, Hagedorn F, Shevchenko I et al (2009) Treeline shifts in the Ural mountains affect soil organic matter dynamics. Glob Chang Biol 15:1570–1583. doi:10.1111/j.1365-2486.2009.01856.x
Köck R, Härtel E, Holtermann C (2002) Wechselwirkungen zwischen Vegetation und Bodenfeuchtigkeits-Dynamik in der subalpinen Stufe der Nordöstlichen Kalkalpen Österreichs. Cent für das gesamte Forstwes 119:297–306
Körner C (1989) The nutritional status of plants from high altitudes. Oecologia 81:379–391. doi:10.1007/BF00377088
Körner C, Farquhar GD, Wong SC (1991) Carbon isotope discrimination by plants follows latitudinal and altitudinal trends. Oecologia 88:30–40
Le Maitre DC, Scott DF, Colvin C (1999) A review of information on interactions between vegetation and groundwater. Water SA 25: 137–152. doi: http://www.wrc.org.za
Lehmann J, Kleber M (2015) The contentious nature of soil organic matter. Nature 528:60–68. doi:10.1038/nature16069
Leifeld J, Zimmermann M, Fuhrer J, Conen F (2009) Storage and turnover of carbon in grassland soils along an elevation gradient in the Swiss Alps. Glob Chang Biol 15:668–679. doi:10.1111/j.1365-2486.2008.01782.x
Liao JD, Boutton TW, Jastrow JD (2006) Organic matter turnover in soil physical fractions following woody plant invasion of grassland: evidence from natural 13C and 15N. Soil Biol Biochem 38:3197–3210. doi:10.1016/j.soilbio.2006.04.004
Livingston GP, Hutchinson GL (1995) Enclosure-based measurement of trace gas exchange: applications and sources of error. In: Matson PA, Harriss RC (eds) Biogenic trace gases: measuring emissions from soil and water. Blackwell Scientific Publications, Oxford, pp 11–51
MacDonald D, Crabtree JR, Wiesinger G et al (2000) Agricultural abandonment in mountain areas of Europe: environmental consequences and policy response. J Environ Manag 59:47–69. doi:10.1006/jema.1999.0335
Macdonald CA, Thomas N, Robinson L et al (2009) Physiological, biochemical and molecular responses of the soil microbial community after afforestation of pastures with Pinus radiata. Soil Biol Biochem 41:1642–1651. doi:10.1016/j.soilbio.2009.05.003
Marey-Pérez MF, Rodríguez-Vicente V (2009) Forest transition in Northern Spain: local responses on large-scale programmes of field-afforestation. Land Use Policy 26:139–156. doi:10.1016/j.landusepol.2008.02.004
Mariotti A, Germon JC, Hubert P et al (1981) Experimental determination of nitrogen kinetic frationation: some principles; illustration for the denitrification and nitrification process. Plant Soil 62:413–430
Martinelli LA, Piccolo MC, Townsend AR et al (1999) Nitrogen stable isotopic composition of leaves and soil: tropical versus temperate forests. Biogeochemistry 46:45–65. doi:10.1007/BF01007573
McCarron JK, Knapp AK, Blair JM (2003) Soil C and N responses to woody plant expansion in a mesic grassland. Plant Soil 257:183–192. doi:10.1023/A:1026255214393
Michener R, Lajtha K (2007) Stable isotopes in ecology and environmental science. Blackwell, Oxford. doi:10.1899/0887-3593-028.002.0516
Nadelhoffer KJ, Shaver G, Fry B et al (1996) 15N natural abundances and N use by tundra plants. Oecologia 107:386–394
Nazaries L, Tottey W, Robinson L et al (2015) Shifts in the microbial community structure explain the response of soil respiration to land-use change but not to climate warming. Soil Biol Biochem 89:123–134. doi:10.1016/j.soilbio.2015.06.027
Paul EA, Morris SJ, Bohm S (2001) The determination of soil C pool sizes and turnover rates: Biophysical fractionation and tracers. In: Kimble JM, Follett RF, Stewart BA, Lal R (eds) Assessment methods for soil carbon. Lewis Publisher, Boca Raton, pp 193–206
Pérez-Cruzado C, Mansilla-Salinero P, Rodríguez-Soalleiro R, Merino A (2012) Influence of tree species on carbon sequestration in afforested pastures in a humid temperate region. Plant Soil 353:333–353. doi:10.1007/s11104-011-1035-0
Pérez-Cruzado C, Sande B, Omil B et al (2014) Organic matter properties in soils afforested with Pinus radiata. Plant Soil 374:381–398. doi:10.1007/s11104-013-1896-5
Pinheiro JC, Bates DM, DebRoy S, R Core Team (2016) nlme: Linear and Nonlinear Mixed Effects Models
Poeplau C, Don A (2013) Sensitivity of soil organic carbon stocks and fractions to different land-use changes across Europe. Geoderma 192:189–201. doi:10.1016/j.geoderma.2012.08.003
Poeplau C, Don A, Vesterdal L et al (2011) Temporal dynamics of soil organic carbon after land-use change in the temperate zone—carbon response functions as a model approach. Glob Chang Biol 17:2415–2427. doi:10.1111/j.1365-2486.2011.02408.x
Pörtl K, Zechmeister-Boltenstern S, Wanek W et al (2007) Natural 15N abundance of soil N pools and N2O reflect the nitrogen dynamics of forest soils. Plant Soil 295:79–94. doi:10.1007/s11104-007-9264-y
Post WM, Kwon KC (2000) Soil carbon sequestration and land-use change: processes and potential. Glob Chang Biol 6:317–327. doi:10.1046/j.1365-2486.2000.00308.x
Post WM, Emanuel WR, Zinke PJ, Stangenberger AG (1982) Soil carbon pools and world life zones. Nature 298:156–159. doi:10.1038/298156a0
Powlson DS, Smith P, Coleman K et al (1998) A European network of long-term sites for studies on soil organic matter. Soil Tillage Res 47:263–274. doi:10.1016/S0167-1987(98)00115-9
Poyatos R, Latron J, Llorens P (2003) Land use and land cover change after agricultural abandonment. Mt Res Dev 23:362–368. doi:10.1659/0276-4741(2003)023[0362:LUALCC]2.0.CO;2
R Core Team (2015) R: a language and environment for statistical computing. Vienna
Richter DD, Markewitz D, Trumbore SE, Wells CG (1999) Rapid accumulation and turnover of soil carbon in a re-establishing forest. Nature 400:56–58. doi:10.1038/21867
Risch AC, Jurgensen MF, Page-Dumroese DS et al (2008) Long-term development of above- and below-ground carbon stocks following land-use change in subalpine ecosystems of the Swiss National Park. Can J For Res 38:1590–1602. doi:10.1139/X08-014
Rodeghiero M, Cescatti A (2005) Main determinants of forest soil respiration along an elevation/temperature gradient in the Italian Alps. Glob Chang Biol 11:1024–1041. doi:10.1111/j.1365-2486.2005.00963.x
Ross DJ, Tate KR, Scott NA et al (2002) Afforestation of pastures with Pinus radiata influences soil carbon and nitrogen. Aust J Soil Res 40:1303–1318
Sánchez-Palomares O, Sánchez F, Carretero MP (1999) Modelos y cartografía de estimaciones climáticas termopluviométricas para la España peninsular. Instituto Nacional de Investigación y Tecnología Agraria y Alimentaria, Ministerio de Agricultura, Pesca y Alimentación. 192 p. Madrid
Schierl R (1991) Saure Perkolation von Bodenproben aus dem Höglwald. Ökosystemforsch. Höglwald Beiträge zur Auswirkung von saurer Beregnung und Kalkung einem Fichtenaltbestand
Schindlbacher A, de Gonzalo C, Díaz-Pinés E et al (2010) Temperature sensitivity of forest soil organic matter decomposition along two elevation gradients. J Geophys Res 115:G03018. doi:10.1029/2009JG001191
Schinner F, Öhlinger R, Beck T et al (1993) Kohlenstof. In: Schinner F, Öhlinger R, Kandeler E, Margesin R (eds) Bodenbiol. Arbeitsmethoden. Springer, Berlin, p 344
Simmons JA, Fernández IJ, Briggs RD, Delaney MT (1996) Forest floor carbon pools and fluxes along a regional climate gradient in Maine, USA. For Ecol Manag 84:81–95
Smal H, Olszewska M (2008) The effect of afforestation with Scots pine (Pinus silvestris L.) of sandy post-arable soils on their selected properties. II. Reaction, carbon, nitrogen and phosphorus. Plant Soil 305:171–187. doi:10.1007/s11104-008-9538-z
Smith P, Bustamante M, Ahammad H, et al. (2014) Agriculture, Forestry and Other Land Use (AFOLU). In: Ofer O, Pichs-Madruga R, Sokona Y, et al. (eds) Climate Change in 2014 Mitig. Clim. Chang. Contrib. Work. Gr. III to Fifth Assess. Rep. Intergov. Panel Clim. Chang. Cambridge University Press, Cambridge pp 811–922
Tasser E, Walde J, Tappeiner U et al (2007) Land-use changes and natural reforestation in the Eastern Central Alps. Agric Ecosyst Environ 118:115–129. doi:10.1016/j.agee.2006.05.004
Thuille A, Schulze E-D (2006) Carbon dynamics in successional and afforested spruce stands in Thuringia and the Alps. Glob Chang Biol 12:325–342. doi:10.1111/j.1365-2486.2005.01078.x
Tiunov AV (2007) Stable isotopes of carbon and nitrogen in soil ecological studies. Biol Bull 34:395–407. doi:10.1134/S1062359007040127
Vitousek PM, Shearer G, Kohl DH (1989) Foliar 15N natural abundance in Hawaiian rainforest: patterns and possible mechanisms. Oecologia 78:383–388. doi:10.1007/BF00379113
von Lützow M, Kögel-Knabner I, Ekschmitt K et al (2006) Stabilization of organic matter in temperate soils: mechanisms and their relevance under different soil conditions—a review. Eur J Soil Sci 57:426–445. doi:10.1111/j.1365-2389.2006.00809.x
Walter H (1985) Vegetation of the earth and ecological systems of the geobiosphere, vol 3. Springer, Berlin
Wang S, Fan J, Song M et al (2013a) Patterns of SOC and soil 13C and their relations to climatic factors and soil characteristics on the Qinghai–Tibetan Plateau. Plant Soil 363:243–255. doi:10.1007/s11104-012-1304-6
Wang W, Zeng W, Chen W et al (2013b) Soil respiration and organic carbon dynamics with grassland conversions to woodlands in temperate China. PLoS ONE 8:1–10. doi:10.1371/journal.pone.0071986
Werth M, Kuzyakov Y (2010) 13C fractionation at the root–microorganisms–soil interface: a review and outlook for partitioning studies. Soil Biol Biochem 42:1372–1384. doi:10.1016/j.soilbio.2010.04.009
Acknowledgements
This study was partially funded by a grant from the government of the Madrid Region (ref. REMEDINAL3-CM MAE-2719). The Ministry of Education, Culture and Sport funded CO’s PhD studies thorough the FPU programme. We thank the Centre for Stable Isotopes at Garmisch-Partenkirchen and Dr Gustavo Saiz for laboratory support; Bernadette Sotier and Klaus Suntinger for their generous help with soil sampling in the Austrian Alps; Dr Hubert Hasenauer and the Department of Soil Science at the University of Natural Resources and Applied Life Sciences in Vienna for providing Austrian meteorological data; Ms Pru Brooke-Turner for her linguistic assistance; and two anonymous reviewers for their constructive comments.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Edward Brzostek.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ortiz, C., Vázquez, E., Rubio, A. et al. Soil organic matter dynamics after afforestation of mountain grasslands in both a Mediterranean and a temperate climate. Biogeochemistry 131, 267–280 (2016). https://doi.org/10.1007/s10533-016-0278-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10533-016-0278-5