Abstract
Pentachlorophenol (PCP) has been widely used as a pesticide in paddy fields and has imposed negative ecological effect on agricultural soil systems, which are in typically anaerobic conditions. In this study, we investigated the effect of repeated additions of PCP to paddy soil on the microbial communities under anoxic conditions. Acetate was added as the carbon source to induce and accelerate cycles of the PCP degradation. A maximum degradation rate occurred at the 11th cycle, which completely transformed 32.3 μM (8.6 mg L−1) PCP in 5 days. Illumina high throughput sequencing of 16S rRNA gene was used to profile the diversity and abundance of microbial communities at each interval and the results showed that the phyla of Bacteroidates, Firmicutes, Proteobacteria, and Euryarchaeota had a dominant presence in the PCP-dechlorinating cultures. Methanosarcina, Syntrophobotulus, Anaeromusa, Zoogloea, Treponema, W22 (family of Cloacamonaceae), and unclassified Cloacamonales were found to be the dominant genera during PCP dechlorination with acetate. The microbial community structure became relatively stable as cycles increased. Treponema, W22, and unclassified Cloacamonales were firstly observed to be associated with PCP dechlorination in the present study. Methanosarcina that have been isolated or identified in PCP dechlorination cultures previously was apparently enriched in the PCP dechlorination cultures. Additionally, the iron-cycling bacteria Syntrophobotulus, Anaeromusa, and Zoogloea were enriched in the PCP dechlorination cultures indicated they were likely to play an important role in PCP dechlorination. These findings increase our understanding for the microbial and geochemical interactions inherent in the transformation of organic contaminants from iron rich soil, and further extend our knowledge of the PCP-transforming microbial communities in anaerobic soil conditions.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Pentachlorophenol (PCP) is a highly toxic and persistent chemical that is classified as a priority pollutant by the US Environment Protection Agency (Keith and Telliard 1979). Since the 1980s, PCP has been widely used as a pesticide in Chinese paddy fields, which has negatively influenced the agricultural soil ecosystems (Augustijn-Beckers et al. 1994). Although banned as an insecticide in China by 1997, PCP residue in soils and groundwater still remains a widespread environmental concern because of its high toxicity and molecular stability (Augustijn-Beckers et al. 1994). As a result, the transformation of PCP in the environment has been the focus of attention for a considerable time. It is well known that PCP can be biodegraded under both aerobic and anaerobic conditions in paddy soils (Li et al. 2013). In the flooded paddy soils, PCP was observed to dechlorinate to less chlorinated phenols (Kuwatsuka and Igarashi 1975; Yoshida et al. 2007). According to those reports, PCP is expected to be dechlorinated by microorganisms under anaerobic conditions like flooded paddy soils.
Anaerobic bacteria capable of dechlorination were isolated from enrichment cultures, and exhibited excellent tolerance and rapidly dechlorinated PCP. For example, Desulfitobacterium frappieri PCP-1, isolated from a methanogenic consortium, can not only dechlorinate high concentrations of PCP, but also dechlorinate several different chlorophenols at ortho, meta, and para positions (Bouchard et al. 1996). Due to limitation of the culture-dependent methods, only a small proportion of the dechlorinating microbiota has been successfully isolated and cultivated from dechlorination cultures. Therefore, exploring potential functional diversity of the microbial community that is responsible for dechlorination of PCP is important part of bioremediation research. Anaerobic dechlorination of PCP forming lesser chlorinated phenols has been observed in sediments and soils environment (Kennes et al. 1996; Xu et al. 2015; Kranzioch-Seipel et al. 2016). Yoshida et al. (2007) reported that an anaerobic microbial community could dechlorinate PCP to phenol in paddy soils supplemented with lactate, thereby leading to the physiological characterization of novel bacteria in the Firmicutes phylum. In the subsequent studies about this enriched microbial community, Zhang and Katayama (2012) and Zhang et al. (2014) founded that the humic substances in the soils were essential factor for sustaining the dechlorination activity and functioned as redox mediator in PCP dechlorinating cultures. In the iron rich paddy soil, the generated Fe(II) species by iron-reducing microorganisms can act as reactive reductants in the biodegradation of PCP, which the possible dechlorination mechanism by microbial community was coupled with the biochemical electron transfer processes between iron redox cycling and reductive dechlorination (Chen et al. 2012; Xu et al. 2015).
Essentially, the anaerobic dechlorination of PCP is a redox reaction involving microbial respiration that dechlorinates PCP by using low molecular weight organic acids (such as acetate, lactate, and glucose) as electron donors in soils (Chang et al. 1996; Zhang et al. 2010; Payne et al. 2011). Various ionic species in soils such as Fe(III), NO3 −, and SO4 2− act as competitive electron acceptors for microorganisms with dechlorinating respiration, which influences the abundance and activity of dechlorinating bacteria (Adriaens et al. 1996; Kotik et al. 2013). During dechlorinated respiration, the soils of different soil types with the different contents of low molecular weight organic acids and ionic species impacted the microbial community structure, further affecting the reduction of PCP (Kuwatsuka and Igarashi 1975; Chen et al. 2014). In paddy soils, acetate is the most important metabolite of the anaerobic food chain (Hori et al. 2010) and dechlorination of PCP has been observed previously (Yoshida et al. 2007). However, there is poor information about the microbial community associated with the acetotrophic PCP dechlorination in iron rich paddy soil.
In this study, the microbial community responsible for PCP dechlorination was enriched through the serial transferred incubations in medium containing acetate, PCP in iron-rich soil. To further explore the succession of microbial communities at different intervals and the main metabolic processes of microorganisms during the PCP dechlorination, the members of this bacterial consortium were further characterized with high throughput sequencing of 16S rRNA. In addition, specific detection of dechlorinating bacteria, such as Dehalobacter spp. and Desulfitobacterium spp. were conducted by quantitative PCR (qPCR) using a set of the genus-specific primers.
Materials and methods
Soil sampling and chemicals
The soil sample was collected 15 cm beneath the surface of paddy soil in Sijiu Village of Jiangmen City, China (22o12′50.90″N, 112o53′4.03″E), in August 2012. After collection, the samples were quickly extruded into glass tubes, stoppered, and immediately transported back to the laboratory and stored in an anaerobic chamber to maintain anoxic conditions for one day before the experiments. No chlorinated phenols were detected in soil and our previous studies have found the chlorinated compounds can be degraded by the microbial communities in adjacent soil (Tong et al. 2014, 2015). The basic physicochemical properties of the soil were analyzed with a previously described method (Pansu and Gautheyrou 2007), and the results were as follows: 17.5% Al2O3, 63.6% SiO2, 20.3 g kg−1 total organic carbon, 20.4 g kg−1 total Fe, and 44% moisture content. The contents (weight/weight) of clay, sand, and silt in the soil were 33, 38, and 29%, therefore, the soil can be classified as clay loam, based on the USDA soil textural triangle standard (Staff 2014). Piperazine-1,4-bis (2-ethanesulfonic acid) (PIPES, 99%) and sodium acetate (99%) were purchased from Sigma Aldrich (USA). All other chemicals of analytical grade were obtained from Guangzhou Chemical Co. (Guangzhou, China). Deionized water (18.2 MΩ cm) was prepared using an ultrapure water system (EasyPure II RF/UV, ThermoScientific, USA) and used in all experiments.
Incubation experiments
To activate the soil microorganisms and reduce a small amount of electron acceptors such as available sulfate and nitrate, the soil was pre-incubated with 65% water content in the airtight bottles at 25 °C for several days before use (Yoshida et al. 2007; Hori et al. 2010). For the preparation of the dechlorination culture, ~10 g soils (wet weight) were introduced into 100 mL serum bottles containing 25 mL medium and were flushed with nitrogen gas (0.1 MPa, 99.99%) for 30 min, and then sealed with butyl rubber stoppers and aluminum crimp seals. The vitamin and mineral solution (Holliger et al. 1998) were added to the medium through a 0.22 μm membrane filter. The medium consisted of (per liter) 30 mM (9.1 g L−1) PIPES buffer (pH 7.0), 1 mL vitamin solution, and 1 mL mineral solution. Sterile, anoxic solutions of PCP and acetate were added to final concentrations of 37.6 μM (10 mg L−1) and 10 mM (600 mg L−1) using sterilized syringe. Each bottle was wrapped twice with aluminum foil to prevent the effect of light on PCP degradation. And this process (the soil transferred from pre-incubated culture) was identified as 1st cycle. After 13 days (upon the depletion of PCP) of incubation, 2.5 mL of the homogenized slurry was transferred to 22.5 mL of a new medium, which contained 30 mM PIPES, 37.6 μM acetate and 10 mM PCP. This transfer was equivalent to 2rd cycle. The serially transferred was performed by using 10% (vol/vol) inocula when PCP was completely removed. Acetate and PCP were added to each cycle using sterilized syringe. The experiments were conducted in duplicate. After flushed with nitrogen, the preparation of all microcosms and serially transferred were done in an anoxic glovebox (100% N2 atmosphere) in order to maintain an anaerobic condition for PCP dechlorination, and the experimental reactors were incubated at 25 °C in an anaerobic chamber without shaking (Shellab, Sheldon Manufacturing Inc., Cornelius, OR).
Chemical analytical methods
During the incubation period, triplicate bottles were taken out for chemical analysis at regular intervals with each chemical determination of each bottle performed without technical replicate. To determine acetate, an aliquot of sample was filtered using a 0.45-μm syringe filter. The concentration of acetate was determined in the filtrate by a Dionex ICS-90 ion chromatograph with an ion column (Ionpac AS14A 4 × 250 mm). Samples were eluted with a 1.8 mM carbonate and 1.7 mM bicarbonate eluent at a flow rate of 2 mL min−1. Dilute sulfuric acid (13.6 mM) was used as a regenerant (Coby et al. 2011). The HCl extractable Fe(II) in the reaction suspension was extracted using 0.5 M HCl for 1.5 h, and analyzed using the 1,10-phenanthroline colorimetric method at 510 nm (Fredrickson and Gorby 1996). To determine PCP, 2 mL soil suspension from the bottle was extracted with 2 mL of water/ethanol (1:1 in volume) by shaking on a horizontal shaker at 180 rpm for 1 h (Khodadoust et al. 1999). The water–ethanol–soil mixture was filtered through the 0.45-mm filter. The filtrate was collected for HPLC analysis of PCP concentration by high performance liquid chromatography (HPLC) using a Waters Alliance 1527-2487 HPLC system fitted with a Symmetry C18 column (5 μm, 4.6 × 250 mm, Waters, USA) (Tong et al. 2014). The PCP transformation intermediates in the suspension were extracted with hexane and identified with Gas Chromatography/Mass Spectrometry (GC/MS) on a Thermo Trace-DSQ-2000 with electron ionization and an Agilent silicon capillary column (0.25 mm × 30 m) (Tong et al. 2014).
DNA extraction, 16S rRNA gene amplification, qPCR and high throughput sequencing
The sample suspensions were centrifuged and ~0.25 g soil was collected. The DNA in the soil was extracted using a PowerSoil™ DNA isolation kit (MO BIO Laboratories, Inc., Carlsbad, CA, USA) according to the manufacturer’s instructions. The DNA was quantified with a Qubit 2.0 fluorometer DNA (Invitrogen, NY, USA). The abundance of dechlorinating bacteria, such as Desulfitobacterium spp. and Dehalobacter spp. in the suspension cultures were determined by quantitative polymerase chain reaction (qPCR) on a MyiQ™ 2 Optics Module (BIO-RAD, USA), and the procedures were performed according to that of Tong et al. (2014). The total genomic DNA extracts were submitted for high-throughput amplicon sequencing at Magigen Biotechnology (Shenzhen, China) following the protocol described elsewhere (Caporaso et al. 2011, 2012). PCR amplification of 16S rRNA gene fragments was performed using primers F515 (5′-GTGCCAGCMGCCGCGGTAA-3′) and R806 (5′-GGACTACVSGGGTATCTAAT-3′), with a sample-specific 12-bp barcode added to the reverse primer (Caporaso et al. 2011). Each DNA sample was amplified in 30 μL reaction mixtures in triplicate using the following PCR program: at 95 °C for 3 min; 35 cycles at 94 °C for 30 s, annealing at 50 °C for 1 min, and extension at 72 °C for 1 min; and a final extension of 10 min at 72 °C. Illumina specific fusion primers were used to amplify the V3–V4 region of the 16S ribosomal RNA (rRNA) gene, and then unique barcodes were added into samples in each well to enable pooling and sequencing. The bioinformatics analysis was processed using Mothur and QIIME (Schloss et al. 2009; Caporaso et al. 2011). The chimeric and low quality sequences were identified and removed, whereas the 12-bp barcode was examined in order to assign sequences to individual samples. Operational taxonomic units (OTUs) were identified at the 97% sequence similarity level using UCLUST (Edgar 2010), and a representative sequence from each phylotype was selected using PyNAST (DeSantis et al. 2006). The taxonomic classification of each phylotype was determined using the Ribosomal Database Project (RDP) at the 80% threshold. Relative abundance (%) of individual taxa within each community was estimated by comparing the number of sequences assigned to a specific taxon versus the number of total sequences obtained for that sample.
Results and discussion
PCP dechlorination culture
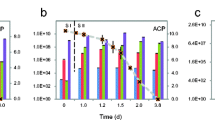
PCP and acetate were consumed simultaneously in the PCP-dechlorinating culture at a circumneutral pH in paddy soil (Fig. 1). At the beginning and the end of the incubation of each cycle, pH values did not changed markedly (data not shown), and similar relative stability of pH has been also observed in other PCP dehclorination cultures (Yoshida et al. 2007; Chen et al. 2016). Acetate concentration was measured in the first four cycles (C1–C4, C1 was equivalent to 1st cycle), and some acetate persisted throughout the experiments durations, whereas PCP was transformed by day 10–13. The paddy soil used in this study is iron-rich soil with a high content of total iron (20.4 g kg−1). Adsorbed Fe(II) species are critical to accelerating the reductive process of organochlorine pollutants (Chen et al. 2012), and 0.5 M HCl-extractable Fe(II) has been shown to be effective in extracting produced Fe(II). The concentrations of 0.5 M HCl-extractable Fe(II) were 12.5 and 10.7 μM in the 1st and 2nd cycle, respectively. These results indicate that the culture-induced dechlorination occurred coupled with acetate metabolism and iron reduction. This was consistent with the predominance of phylotypes related to known Firmicutes and Proteobacteria taxa in the microbial community analysis data (Fig. 2). During the microbial degradation of PCP, several intermediates were detected by GC–MS, including 2,3,4,5-tetrachlorophenol (2,3,4,5-TeCP), 3,4,5-trichlorophenol (3,4,5-TCP), and 4-chlorophenol (4-CP), in line with other reports with adjacent sampling points (Tong et al. 2014). The dechlorination pathway of this study is also consistent with the microbial reductive of PCP in river sediments by Kranzioch-Seipel et al. (2016). The dechlorination pathway of the enriched microcosm indicated the preferable dechlorination in ortho-position following to meta-positions.
A total of eleven cycles were carried out with fresh PCP (37.6 μM) and acetate (10 mM) added to the cultures at each time interval (Fig. 1). As each cycle progressed, the rate of PCP dechlorination increased and the dechlorination cycle time decreased. The maximum dechlorination rate was obtained at the 11th cycle, in which 8.6 mg L−1 of PCP was completely degraded within 5 days. This could be the result of the lag time necessary for microorganism activation (Lendvay et al. 2003). The microbial community structure and PCP dechlorination rate became relatively stable after 8th transfer (Figs. 1, 4). When the certain time for microbial activation, both abiotic and biotic reactions contributed to PCP removal in the cultures. In the first cycle, the indigenous organic matter can act as an electron donor for microorganism respiration. Added electron donors, such as acetate, formate, pyruvate, and lactate, can further support and stimulate the biotic dechlorination processes (Brahushi et al. 2004). Acetate is a favorable electron donor for the respiration of a wide range of microorganisms in the processes of contaminant transformation (Chang et al. 1996). More specifically, the dechlorinating and iron-reducing bacteria can utilize acetate as the electron donor to accelerate the reductive dechlorination of PCP (He et al. 2002; Chen et al. 2012). In the previous report, acetate was absolutely required for PCB-dechlorinating bacteria growth and dechlorinating activity (Cutter et al. 2001), and transformed to carbon dioxide and methane (Kittelmann and Friedrich 2008). In the present culture, acetate served as the electron donor for dehalorespiring bacteria, while acetate oxidation coupled to PCP reduction was a highly exergonic process for dehalorespiring bacterial growth (Kittelmann and Friedrich 2008), e.g. as presented in Eq. (1)
Microbial community diversity and composition during incubation
After adding PCP and acetate during the reaction period, the cultured microbial community acclimated and changed over time. Approximately 578,791 quality sequences for the 12 analyzed samples were generated with the Illumina high throughput sequencing, with sequences frequencies for individual samples ranging from 11,775 to 39,116 (Table 1). In total, 11,292 operational taxonomic units (OTUs) were identified in the complete data set, with an average of 3432 OTUs per sample. Over 99% of the OTUs were assigned to a taxonomic group (phylum), and over 80% were identified at the order level.
The dominant phyla in the original soil sample without any amendment (T0) were the following: Firmicutes, Nitrospirae, Proteobacteria, Crenarchaeota, Chloroflexi, Bacteroidetes, and Acidobacteria. A relative abundance >4% for each phylum accounted for >85% of the total sequences (Fig. 2; Table S1), with dominant taxa being roughly consistent with those reported in the previous study on soil bacterial community compositions (Li et al. 2016). The dominant phyla in the treatment without PCP were similar with the original soil sample after 1st cycle, and the date was provided in supporting information (Figure S1). However, during the PCP and acetate treatment, the dominant phyla changed to Euryarchaeota, Bacteroidetes, Firmicutes, Proteobacteria, Chloroflexi, and WWE1, which were largely different with that in T0. In the previous study, the microbial taxonomic composition in the paddy soil amended with PCP after 40 days exhibited significant differences with the original soil and the treatment without PCP (Chen et al. 2016). The relative abundance of the different phyla varied considerably. During the 1st–4th cycle of PCP dechlorination, the relative abundance of Firmicutes decreased from 36.0 to 11.6%, and then increased to 31.5% during the 5th–11th cycle, whereas that of Proteobacteria increased from 9.9 to 29.9% during the 1st–4th cycle, and then markedly decreased to 7.9% after the 6th cycle. The relative abundance of Chloroflexi increased to 16.6% during the first two cycle, and then maintained an average value of 4.3% during the 7th–11th cycle. The relative abundance of Bacteroidetes and WWE1 increased from 4.7 to 14.1%, and 0.1 to 12.3% during the 1st–11th cycle, respectively. For the archaea, the majority of sequences were affiliated with Euryarchaeota, which the relative abundance increased markedly from 1.5 to 27.2% at end of 7th cycle, and then maintained an average value of 12.0% during the 8th–11th cycle.
At the genus level, the composition of the microbial community was obviously different and displayed a distinct succession between the initial and following cycles (Fig. 3; Table S2). The dominant genera in the original soil sample were Clostridium, GOUTA19, unclassified Nitrospirales, unclassified Anaerolineae, Anaeromyxobacter, Bacillus, unclassified Bacteroidales, unclassified Crenarchaeota, and Methanobacterium (relative abundance >1%). In the treatment without PCP after 1st cycle, besides the dominant genera mentioned in the original soil, Anaeromusa was also enriched in the culture (Figure S2). However, the relative abundance of Anaeromusa (3.2%) in the treatment without PCP was lower than that in the treatment with PCP (9.3%) indicated Anaeromusa was likely to play an important role in PCP dechlorination coupled with acetate oxidation. With the amendment of PCP and acetate, the relative abundances of Methanosarcina and Anaeromusa evidently increased after the 2nd cycle. The relative abundance of Methanosarcina achieved an average 7.7% during the 2nd–7th cycle, and then decreased to an average value of 2.5% after the 8th cycle. The relative abundance of Anaeromusa dramatically decreased to 3.7% at the 4th cycle, and then steadily increased to 21.2% at the 11th cycle. The relative abundance of Zoogloea dramatically increased to 21.6% during 2nd–4th cycle, but decreased to approximately 1% at the 11th cycle. Syntrophobotulus slightly increased over throughout the whole process, and the relative abundance maintained at an average value of 1.6% during the 8th–11th cycle. The relative abundances of Treponema, W22, and unclassified SHA-116, however, increased continuously to 13.7, 5.0, and 4.6%, respectively, at the 11th cycle. The changes in these cycle communities were analyzed using a principal component analysis (PCA), and the results are presented in Fig. 4. The results show that C6–C11 group relatively close together, which demonstrates that microbial communities stability increased as cycles increased (Li et al. 2016). In the enrichment culture, PCP dechlorinating microbial community structure by serial transfers from a paddy soil become stable in dechlorination in different cultures (Yoshida et al. 2007; Zhang and Katayama 2012). To further explore dechlorinating bacteria, such as Desulfitobacterium spp. and Dehalobacter spp. in the cycles of PCP incubation, 16S rRNA gene copy numbers of the dechlorinating bacteria were measured by qPCR. However, the copy numbers of these dechlorinating bacteria were very low, even not observed (Figure S3). To date, only few PCP respiring isolates are known to use acetate as a direct electron donor (Field and Sierra-Alvarez 2008). The dominant genera in the enrichment culture were seldom associated with PCP dechlorination coupled with acetate oxidation. In our study, the extent of direct acetotrophic PCP dechlorination by soil microorganisms remains elusive.
Methanosarcina has been reported as a reductive dechlorinating microorganism under anaerobic conditions (Wang et al. 2000). The isolation of Methanosarcina from methanogenic and sulfate-reducing consortium was able to dechlorinate high concentrations of tetrachloroethylene, 2,4-dichlorophenol and 2,4,6-trichlorophenol (Wang et al. 2000; Sponza and Cigal 2008). Previous work with fluorescence in situ hybridization (FISH) results indicated that Methanosarcina-like was the dominant methanogenic archaea present during PCP degradation experiments (Heimann et al. 2006). Under methanogenic culture, PCP was dechlorinated by acetoclastic methanogens of Methanosarcina, oxidizing acetate into methane and carbon dioxide (Stuart et al. 1999; Montenegro et al. 2003), suggesting that Methanosarcina was directly associated with PCP dechlorination.
Notably, besides the archaea, the bacteria also seem to play important roles in PCP dechlorination in the transferred soil/acetate suspensions. Anaeromusa and Syntrophobotulus are in the Clostridia class and associated with the Veillonellaceae and Peptococcaceae family, respectively, which contain many PCP-dechlorinating bacteria (Ruckdeschel et al. 1987; Tartakovsky et al. 2001; Chen et al. 2012). In a previous study, Veillonellaceae and Peptococcaceae bacteria have been disclosed the function for iron reduction coupling PCP dechlorination (Tong et al. 2014). Veillonellaceae are strictly anaerobic gram-negative cocci, which contain many iron reducing species (Li et al. 2011), whereas Peptococcaceae contains the well-known reductive dechlorinating bacterial genera Dehalobacter which can reduce PCP in soil suspensions with biochar and lactate (Tong et al. 2014). In our study, the enriched microcosm was preferable dechlorination in ortho-position following to meta- then para-positions, which was consistent with the pathway of dechlorination by some dechlorinating bacteria belonged to Peptococcaceae family (Christiansen and Ahring 1996; Utkin et al. 1995). The iron reducing microorganisms may be capable of using PCP as electron acceptors directly or they may first donate electrons to Fe(III), and then PCP was dechlorinated by Fe(II) generated from Fe(III) reduction (McCormick and Adriaens 2004). The aforementioned discussions suggested the important roles of Syntrophobotulus and Anaeromusa in iron reduction coupling PCP dechlorination. In our experiment, the soil contained 20.4 g kg−1 total Fe, and the content of Fe gradually declined with increased cycle transferred. Thus, a lower amount of Fe was supplied for microorganism respiration as cycles increased, which was concordant to the change of relative abundance of Veillonellaceae (genera Anaeromusa, Fig. 3). Also, a similar tendency existed in the variation of relative abundance of Anaeromusa and the PCP dechlorination rate (Figure S4), which indicated Anaeromusa directly involved in PCP dechlorination.
The relative abundance of Zoogloea, belonging to the Rhodocyclaceae family of β-Proteobacteria, achieved maximum value of 21.5% at the 4th cycle. Rhodocyclaceae strains have been described as having the high reduction abilities, not only for dechlorinating chlorinated compounds (Bae et al. 2007; Guan et al. 2013), but also in NO3 − reduction and organic contaminant degradation (Li et al. 2005; Shao et al. 2009). In the recent study, Rhodocyclaceae was identified as PCP degraders with lactate as carbon source, and the dechlorination pathway was consistent with our present study (Tong et al. 2015), indicated that Zoogloea may directly participate in dechlorination in ortho-position (2,3,4,5-TeCP was detected in our experiments). In addition, Zoogloea species isolated from contaminated groundwater were able to degrade 2,3,4,6-TeCP and PCP (Männistö et al. 1999). At circumneutral pH, Zoogloea has been reported as anaerobic Fe(II) oxidizer in iron redox cycling cultures (Coby et al. 2011; Li et al. 2016). Because Zoogloea was rarely found in the initial soil sample but became increasingly enriched in the treatments during PCP dechlorination in different cycles, it most likely acclimated to the transferred cycle cultures and involved in iron cycle and PCP degradation.
The relative abundances of Treponema, W22, and unclassified Cloacamonales continuously increased with the increasing cycle. Treponema, belonged to the Spirochaetaceae family, has been detected in many dechlorination cultures (Duhamel and Edwards 2007; Miura et al. 2015). However, it should be noted that none direct evidence has been ever provided to link the function of Treponema with PCP dechlorination. In dechlorination cultures, Treponema acts as a homoacetogen that is able to ferment carbon sources to hydrogen, carbon dioxide, and formate, which are essential for other dechlorinating bacteria (Graber et al. 2004; Miura et al. 2015). In the experiments, a significant positive liner correlation was observed between the relative abundance of Treponema and PCP dechlorination rate (R 2 = 0.59) (Figure S5). The dechlorination rate of PCP increased when with a higher relative abundance of Treponema. Thus, the fast PCP dechlorination and increased of relative abundance of Treponema during transferred cycles, suggest that Treponema plays important roles in the PCP dechlorination and may be a key syntrophy required for dechlorinating bacteria to dechlorinate PCP. Compared to the proportion of Treponema, the relative abundances of W22 and unclassified SHA-116, belonged to the Cloacamonales order, occupied a lower percentage of the culture. Cloacamonales has been previously described as a new bacterial phylum, WWE1 branching, and has been widely observed in anaerobic ecosystems (Riviere et al. 2009; Lucas et al. 2015), while not being reported to be able to use chlorophenols as the terminal electron acceptor. In the anaerobic sludge digester, Cloacamonales plays important roles in organic waste and wastewater treatment (Chouari et al. 2005; Narihiro and Sekiguchi 2007). The enrichment of Cloacamonales in the dechlorination cultures indicated that Cloacamonales may participate in syntrophic acetate-oxidization coupled with the dechlorination of PCP.
Based on the above mentioned PCP dechlorination, input consumption, and the microbial communities involved, an overall hypothesis can be proposed as Fig. 5. In the incubation cultures, Syntrophobotulus, Anaeromusa, and Zoogloea play dominant roles in iron cycle coupled to PCP dechlorination, whereas, Methanosarcina may be the dominant genera for direct PCP dechlorination coupled to organic matter oxidation to carbon dioxide and methane. With the input of organic carbon (acetate), Treponema, W22, and unclassified Cloacamonales became the dominant genera for direct or syntrophic function of PCP dechlorination. Acetate is the most important metabolite of the anaerobic food chain in rice field soils and thermodynamically feasible to oxidize to CO2 when it is coupled to iron reduction or syntrophic acetate-oxidizing consortia (Hori et al. 2010). In addition, the iron cycle process with high iron contents from southern China coupled with acetate oxidation can be strongly influenced the reductive dechlorination (Chen et al. 2012; Xu et al. 2015). Our results obtained from the microcosm experiments only revealed the dominant PCP-respiring microorganisms collected in the cycle culture. Further studies are needed to explore the function of acetate and the mechanism of direct acetotrophic PCP dechloriantion, which would be help for the bioremediation of iron rich paddy soil with organic chloride pollutants. However, a highly enriched culture or a pure culture is require, and efforts are made in our lab in this direction (e.g. isolation and additional testing).
Conclusion
Our experiments show that a microbial system undergoing repeated cultivations in the input of organic substrate and PCP, develop a community structure capable of PCP dechlorination under anaerobic conditions. Anaeromusa, Syntrophobotulus, and Zoogloea were found to be the dominant genera in an iron cycle-associated PCP dechlorination. The Methanosarcina associated with humic substances and organic substrate, was enriched in the PCP dechlorination culture at different cycles. Treponema, W22, and unclassified Cloacamonales were dominant in the subsequent input of organic substrate, which expanded the diversity of microbial communities associated with PCP dechlorination. The more cycles of PCP dechlorination, the more stable the microbial communities became. The results of this study have important implications for shelling insight on understanding of microbial and geochemical interactions in the transformation of organic contaminants in iron rich soil.
References
Adriaens P, Chang P, Barkovskii A (1996) Dechlorination of PCDD/F by organic and inorganic electron transfer molecules in reduced environments. Chemosphere 32:433–441
Augustijn-Beckers P, Hornsby A, Wauchope R (1994) The SCS/ARS/CES pesticide properties database for environmental decision-making. II. Additional compounds, reviews of environmental contamination and toxicology. Springer, New York, pp 1–82
Bae HS, Rash BA, Rainey FA, Nobre MF, Tiago I, Costa MS, Moe WM (2007) Description of Azospira restricta sp. nov., a nitrogen-fixing bacterium isolated from groundwater. Int J Syst Evol Microbiol 57:1521–1526
Bouchard B, Beaudet R, Villemur R, McSween G, Lepine F, Bisaillon JG (1996) Isolation and characterization of Desulfitobacterium frappieri sp. nov., an anaerobic bacterium which reductively dechlorinates pentachlorophenol to 3-chlorophenol. Int J Syst Evol Microbiol 46:1010–1015
Brahushi F, Dörfler U, Schroll R, Munch JC (2004) Stimulation of reductive dechlorination of hexachlorobenzene in soil by inducing the native microbial activity. Chemosphere 55:1477–1484
Caporaso JG, Lauber CL, Walters WA, Berg-Lyons D, Lozupone CA, Turnbaugh PJ, Fierer N, Knight R (2011) Global patterns of 16S rRNA diversity at a depth of millions of sequences per sample. Proc Natl Acad Sci USA 108:4516–4522
Caporaso JG, Lauber CL, Walters WA, Berg-Lyons D, Huntley J, Fierer N, Owens SM, Betley J, Fraser L, Bauer M (2012) Ultra-high-throughput microbial community analysis on the Illumina HiSeq and MiSeq platforms. ISME J 6:1621–1624
Chang BV, Zheng JX, Yuan SY (1996) Effects of alternative electron donors, acceptors and inhibitors on pentachlorophenol dechlorination in soil. Chemosphere 33:313–320
Chen M, Shih K, Hu M, Li F, Liu C, Wu W, Tong H (2012) Biostimulation of indigenous microbial communities for anaerobic transformation of pentachlorophenol in paddy soils of southern China. J Agric Food Chem 60:2967–2975
Chen M, Tao L, Li F, Lan Q (2014) Reductions of Fe(III) and pentachlorophenol linked with geochemical properties of soils from Pearl River Delta. Geoderma 217–218:201–211
Chen Y, Tao L, Wu K, Wang Y (2016) Shifts in indigenous microbial communities during the anaerobic degradation of pentachlorophenol in upland and paddy soils from southern China. Environ Sci Pollut Res 23:23184–23194
Chouari R, Le Paslier D, Dauga C, Daegelen P, Weissenbach J, Sghir A (2005) Novel major bacterial candidate division within a municipal anaerobic sludge digester. Appl Environ Microbiol 71:2145–2153
Christiansen N, Ahring BK (1996) Desulfitobacterium hafniense sp. nov., an anaerobic, reductively dechlorinating bacterium. Int J Syst Bacteriol 46:442–448
Coby AJ, Picardal F, Shelobolina E, Xu H, Roden EE (2011) Repeated anaerobic microbial redox cycling of iron. Appl Environ Microbiol 77:6036–6042
Cutter LA, Watts JE, Sowers KR, May HD (2001) Identification of a microorganism that links its growth to the reductive dechlorination of 2,3,5,6-chlorobiphenyl. Environ Microbiol 3:699–709
DeSantis TZ, Hugenholtz P, Keller K, Brodie EL, Larsen N, Piceno YM, Phan R, Andersen GL (2006) NAST: a multiple sequence alignment server for comparative analysis of 16S rRNA genes. Nucleic Acids Res 34:W394–W399
Duhamel M, Edwards EA (2007) Growth and yields of dechlorinators, acetogens, and methanogens during reductive dechlorination of chlorinated ethenes and dihaloelimination of 1,2-dichloroethane. Environ Sci Technol 41:2303–2310
Edgar RC (2010) Search and clustering orders of magnitude faster than BLAST. Bioinformatics 26:2460–2461
Field JA, Sierra-Alvarez R (2008) Microbial degradation of chlorinated phenols. Rev Environ Sci Biotechnol 7:211–241
Fredrickson JK, Gorby YA (1996) Environmental processes mediated by iron-reducing bacteria. Curr Opin Biotechnol 7:287–294
Graber JR, Leadbetter JR, Breznak JA (2004) Description of Treponema azotonutricium sp. nov. and Treponema primitia sp. nov., the first spirochetes isolated from termite guts. Appl Environ Microbiol 70:1315–1320
Guan X, Liu F, Xie Y, Zhu L, Han B (2013) Microbiota associated with the migration and transformation of chlorinated aliphatic hydrocarbons in groundwater. Environ Geochem Health 35:535–549
He J, Sung Y, Dollhopf ME, Fathepure BZ, Tiedje JM, Löffler FE (2002) Acetate versus hydrogen as direct electron donors to stimulate the microbial reductive dechlorination process at chloroethene-contaminated sites. Environ Sci Technol 36:3945–3952
Heimann AC, Batstone DJ, Jakobsen R (2006) Methanosarcina spp. drive vinyl chloride dechlorination via interspecies hydrogen transfer. Appl Environ Microbiol 72:2942–2949
Holliger CH, Harmsen H, Ludwig W, Schumacher W, Tindall B, Vazquez F, Weiss N, Zehnder AJB (1998) Dehalobacter restrictus gen. nov. and sp. nov., a strictly anaerobic bacterium that reductively dechlorinates tetra- and trichloroethene in an anaerobic respiration. Arch Microbiol 169:313–321
Hori T, Muller A, Igarashi Y, Conrad R, Friendrich MW (2010) Identification of iron-reducing microorganisms in anoxic rice paddy soil by 13C-acetate probing. ISME J 4:267–278
Keith L, Telliard W (1979) ES&T special report: priority pollutants: Ia perspective view. Environ Sci Technol 13:416–423
Kennes C, Wu WM, Bhatnagar L, Zeikus J (1996) Anaerobic dechlorination and mineralization of pentachlorophenol and 2,4,6-trichlorophenol by methanogenic pentachlorophenol-degrading granules. Appl Microbiol Biotechnol 44:801–806
Khodadoust AP, Suidan MT, Acheson CM, Brenner RC (1999) Solvent extraction of pentachlorophenol from contaminated soils using water-ethanol mixtures. Chemosphere 38:2681–2693
Kittelmann S, Friedrich MW (2008) Identification of novel perchloroethene-respiring microorganisms in anoxic river sediment by RNA-based stable isotope probing. Environ Microbiol 10:31–46
Kotik M, Davidová A, Voříšková J, Baldrian P (2013) Bacterial communities in tetrachloroethene-polluted groundwaters: a case study. Sci Total Environ 454:517–527
Kranzioch-Seipel I, Beckert U, Shen C, Yin D, Tiehm A (2016) Microbial dechlorination of HCB, PCP, PCB180, HCH and PCE in a Yangtze Three Gorges Reservoir enrichment culture, China. Environ Earth Sci 75(10):1–9
Kuwatsuka S, Igarashi M (1975) Degradation of PCP in soils: II. The relationship between the degradation of PCP and the properties of soils, and the identification of the degradation products of PCP. Soil Sci Plant Nutr 21:405–414
Lendvay J, Löffler FE, Dollhopf M, Aiello M, Daniels G, Fathepure B, Gebhard M, Heine R, Helton R, Shi J (2003) Bioreactive barriers: a comparison of bioaugmentation and biostimulation for chlorinated solvent remediation. Environ Sci Technol 37:1422–1431
Li P, Wang X, Stagnitti F, Li L, Gong Z, Zhang H, Xiong X, Austin C (2005) Degradation of phenanthrene and pyrene in soil slurry reactors with immobilized bacteria Zoogloea sp. Environ Eng Sci 22:390–399
Li H, Peng J, Weber KA, Zhu Y (2011) Phylogenetic diversity of Fe(III)-reducing microorganisms in rice paddy soil: enrichment cultures with different short-chain fatty acids as electron donors. J Soils Sediment 11:1234–1242
Li Z, Inoue Y, Suzuki D, Ye L, Katayama A (2013) Long-term anaerobic mineralization of pentachlorophenol in a continuous-flow system using only lactate as an external nutrient. Environ Sci Technol 47:1534–1541
Li X, Zhang W, Liu T, Chen L, Chen P, Li F (2016) Changes in the composition and diversity of microbial communities during anaerobic nitrate reduction and Fe(II) oxidation at circumneutral pH in paddy soil. Soil Biol Biochem 94:70–79
Lucas R, Kuchenbuch A, Fetzer I, Harms H, Kleinsteuber S (2015) Long-term monitoring reveals stable and remarkably similar microbial communities in parallel full-scale biogas reactors digesting energy crops. FEMS Microbiol Ecol 91:fiv004
Männistö MK, Tiirola MA, Salkinoja-Salonen MS, Kulomaa MS, Puhakka JA (1999) Diversity of chlorophenol-degrading bacteria isolated from contaminated boreal groundwater. Arch Microbiol 171:189–197
McCormick ML, Adriaens P (2004) Carbon tetrachloride transformation on the surface of nanoscale biogenic magnetite particles. Environ Sci Technol 38:1045–1053
Miura T, Yamazoe A, Ito M, Ohji S, Hosoyama A, Takahata Y, Fujita N (2015) The impact of injections of different nutrients on the bacterial community and its dechlorination activity in chloroethene-contaminated groundwater. Microbes Environ 30:164
Montenegro MA, Araujo JC, Vazoller RF (2003) Microbial community evaluation of anaerobic granular sludge from a hybrid reactor treating pentachlorophenol by using fluorescence in situ hybridization. Water Sci Technol 48:65–73
Narihiro T, Sekiguchi Y (2007) Microbial communities in anaerobic digestion processes for waste and wastewater treatment: a microbiological update. Curr Opin Biotechnol 18:273–278
Pansu M, Gautheyrou J (2007) Handbook of soil analysis: mineralogical, organic and inorganic methods. Springer, Berlin, pp 289–367
Payne RB, May HD, Sowers KR (2011) Enhanced reductive dechlorination of polychlorinated biphenyl impacted sediment by bioaugmentation with a dehalorespiring bacterium. Environ Sci Technol 45:8772–8779
Riviere D, Desvignes V, Pelletier E, Chaussonnerie S, Guermazi S, Weissenbach J, Li T, Camacho P, Sghir A (2009) Towards the definition of a core of microorganisms involved in anaerobic digestion of sludge. ISME J 3:700–714
Ruckdeschel G, Renner G, Schwarz K (1987) Effects of pentachlorophenol and some of its known and possible metabolites on different species of bacteria. Appl Environ Microbiol 53:2689–2692
Schloss PD, Westcott SL, Ryabin T, Hall JR, Hartmann M, Hollister EB, Lesniewski RA, Oakley BB, Parks DH, Robinson CJ (2009) Introducing mothur: open-source, platform-independent, community-supported software for describing and comparing microbial communities. Appl Environ Microbiol 75:7537–7541
Shao Y, Chung BS, Lee SS, Park W, Lee SS, Jeon CO (2009) Zoogloea caeni sp. nov., a floc-forming bacterium isolated from activated sludge. Int J Syst Evol Microbiol 59:526–530
Soil Survey Staff (2014) Keys to soil Taxonomy. In: 12th USDA, p 179
Sponza DT, Cigal C (2008) Relationships between anaerobic consortia and removal efficiencies in an UASB reactor degrading 2,4 dichlorophenol (DCP). J Environ Manag 87:177–192
Stuart S, Woods S, Lemmon T, Ingle J (1999) The effect of redox potential changes on reductive dechlorination of pentachlorophenol and the degradation of acetate by a mixed, methanogenic culture. Biotechnol Bioeng 63:69–78
Tartakovsky B, Manuel MF, Beaumier D, Greer C, Guiot S (2001) Enhanced selection of an anaerobic pentachlorophenol-degrading consortium. Biotechnol Bioeng 73:476–483
Tong H, Hu M, Li F, Liu C, Chen M (2014) Biochar enhances the microbial and chemical transformation of pentachlorophenol in paddy soil. Soil Biol Biochem 70:142–150
Tong H, Liu C, Li F, Luo C, Chen M, Hu M (2015) The key microorganisms for anaerobic degradation of pentachlorophenol in paddy soil as revealed by stable isotope probing. J Hazard Mater 298:252–260
Utkin I, Dalton DD, Wiegel J (1995) Specificity of reductive dehalogenation of substituted ortho-chlorophenols by Desulfitobacterium dehalogenans JW/IU-DC1. Appl Environ Microbiol 61:346–351
Wang CC, Lee CM, Lu CJ, Chuang MS, Huang CZ (2000) Biodegradation of 2,4,6-trichlorophenol in the presence of primary substrate by immobilized pure culture bacteria. Chemosphere 41:1873–1879
Xu Y, He Y, Zhang Q, Xu J, Crowley D (2015) Coupling between pentachlorophenol dechlorination and soil redox as revealed by stable carbon isotope, microbial community structure, and biogeochemical data. Environ Sci Technol 49:5425–5433
Yoshida N, Yoshida Y, Handa Y, Kim HK, Ichihara S, Katayama A (2007) Polyphasic characterization of a PCP-to-phenol dechlorinating microbial community enriched from paddy soil. Sci Total Environ 381:233–242
Zhang C, Katayama A (2012) Humin as an electron mediator for microbial reductive dehalogenation. Environ Sci Technol 46:6575–6583
Zhang H, Zivel M, Rittmann BE, Krajmalnik-Brown R (2010) Effect of dechlorination and sulfate reduction on the microbial community structure in denitrifying membrane-biofilm reactors. Environ Sci Technol 44:5159–5164
Zhang C, Zhang D, Li Z, Akatsuka T, Yang S, Suzuki D, Katayama A (2014) Insoluble Fe-humic acid complex as a solid-phase electron mediator for microbial reductive dechlorination. Environ Sci Technol 48:6318–6325
Acknowledgment
This work was funded by the National Natural Science Foundation of China (41603127, 41420104007, and 41673135), the Guangdong Natural Science Foundation of China (2016A030313780, 2016B020242006 and S2013050014266), Special Fund for Agro-Scientific Research in the Public Interest of China (201503107-4), the Scientific Platform and Innovation Capability Construction Program of GDAS (2016GDASPT-0212), and One Hundred Talents Programme of the Chinese Academy of Sciences.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing financial interests.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Tong, H., Chen, M., Li, F. et al. Changes in the microbial community during repeated anaerobic microbial dechlorination of pentachlorophenol. Biodegradation 28, 219–230 (2017). https://doi.org/10.1007/s10532-017-9791-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10532-017-9791-z