Abstract
We used automatic sound recorders to study spontaneous vocalizations of wild wolves during the pup-rearing season around rendezvous sites from 24 wolf packs in six study areas across North America, Asia, and Europe. Between 2018 and 2021, for a total of 1225 pack-days, we recorded 605 spontaneous wolf chorus howls and 224 solo-howl series. Howling occurrence varied across areas, from 12.50 to 94.12% days with howling. Daily howling ranged from 0.00 to 3.47 solo howls/day and 0.13 to 5.29 chorus howls/day. Generally, spontaneous chorus howls peaked between sunset and sunrise. Howling rate depended on area, pack size, and density of people living nearby, being greater where fewer people lived. High rates in Yellowstone National Park, (800,000+ visitors during the study) could reflect accommodation to human activities such as wolf watching. One to six automatic recorders per site within 1000 m of rendezvous sites needed 4–15 days to detect the pack (average 9.5) and 5–21 days (average 11.3) to detect pups, both with a probability of 95%. Our results may guide wolf-monitoring programs using automatic sound recorders, a promising method offering advantages over howling surveys, especially in human-dominated landscapes.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Howls are the most conspicuous gray wolf (Canis lupus) vocalizations, allowing communication over long distances and the coordination of group movements. Howls can be territorial displays (inter-pack communication), conveying information about pack location, and minimizing contact between packs (Harrington and Mech 1979). Other functions include communication among pack members, for instance, to reunite separated pack mates (Mech 1970; Harrington and Asa 2003). Due to the elusive behavior of wolves and the extent of their territories, studying wolf vocalizations is challenging. Therefore, most such studies have been done with captives or with responses of wild wolves to simulated howling. Only a few have dealt with spontaneous vocalizations of wild wolves (Harrington and Mech 1978, 1979; Nowak et al. 2007; McIntyre et al. 2017; Suter et al. 2017; Kershenbaum et al. 2019).
Spontaneous wolf howling follows a seasonal pattern (Harrington and Mech 1979). Daytime howling in Yellowstone National Park peaked during pre-breeding and breeding seasons (winter) and increased through summer and fall and rose to a lower peak in October (McIntyre et al. 2017). In Poland, Nowak et al. (2007) found a spontaneous howling peak from July to October (between 18:00 and 00:00 h).
Spontaneous howling of captive wolves and the factors affecting wild-wolf response to simulated howling have been studied in some detail. Environmental factors such as wind or time of day influence both (Harrington and Mech 1982; Ausband et al. 2020; Palacios et al. 2021). Small packs reply less to simulated howls (Harrington and Mech 1982), but larger packs take longer to respond (Ausband et al. 2020). Pup presence increases reply rates, and pups over 16–18 weeks old are more likely to respond to howls (Ausband et al. 2020). Social factors such as relationship quality between pack members can influence howling in captive wolves (Mazzini et al. 2013), but the factors that influence spontaneous howling in wild wolves remain unknown.
Recently, the use of automatic recording units (ARUs) has increased in wildlife studies (Ramsier et al. 2012; Hill et al. 2017; Lynch and Lynch 2017). ARUs represent a useful, non-invasive method to detect, monitor, or identify species that emit sounds (Hill et al. 2017). They have been used, for instance, to (1) detect maned wolf, Chrysocyon brachyurus, (Rocha et al. 2015) and gray wolf vocalizations (Suter et al. 2017; Kershenbaum et al. 2019; Barber-Meyer et al. 2020); (2) to study vocalization rates or temporal patterns of male impala, Aepyceros melampus, vocal activity (Volodina et al. 2021); and (3) spontaneous howling of captive wolves (Palacios et al. 2021). ARUs have been proposed as to monitor wild wolf populations (Suter et al. 2017; Barber-Meyer et al. 2020) and for monitoring the dynamics of gray wolf recolonization (Papin et al. 2018), although Cozzi et al. (2021) found camera trapping and scat dog detection more effective. ARUs can also be used to identify individual wolves (Larsen et al. 2022) and to track wolves via acoustic multilateration (Kershenbaum et al. 2019).
We used ARUs to study spontaneous vocalizations of wild wolves at rendezvous sites and ways to improve the effectiveness of wolf monitoring. We assessed (1) spontaneous howling rates, (2) environmental factors influencing howling occurrence, and (3) the minimum time to achieve a significant probability of detecting pups. We predicted that larger packs would vocalize more and that wolves inhabiting human-dominated landscapes would howl less, possibly to minimize detection by humans.
Methods
We studied populations of wolves across six areas with different environmental conditions and varying human-landscape domination (Fig. 1):
-
(1)
Yellowstone National Park, USA (USYNP). Elevations range from 1500 to 3800 m.a.s.l. Vegetation varies from grassland to alpine, with high mountain meadows and treeless plateaus. Forests are common (detailed in Despain 1990), and sagebrush is abundant in non-forested areas to about 2500 m. Temperatures in August–September 2019 (during the study) varied from − 5 to 26 °C, and average 24-h temperatures, between 1 and 16 °C. Among our study areas, USYNP hosted the fewest humans (0.08 people/km2 -obtained in http://www.city-data.com/city/Yellowstone-National-Park-Wyoming.html), although park visitors during August 2019 exceeded 800,000 (https://www.yellowstone.co/stats.htm). USYNP wolves belong to the subspecies C. l. occidentalis and were reintroduced in 1995–1996, the only reintroduced population of our study (Smith et al. 2020a). Inside USYNP no hunting of wolves is allowed.
-
(2)
Superior National Forest, Minnesota, USA (USSNF). Elevations range from 325 to 700 m.a.s.l., including swamps, uneven upland, and rocky ridges (Mech 2009). The vegetation is comprised mostly of conifers interspersed with birch and aspen due to logging and fires (see Heinselman 1993 for details). Temperatures in mid-July–mid October 2019 (study period) ranged from − 3 to 30 °C, and average 24-h temperatures, 4 to 22 °C. Human density is < 8 habitants/km2 (Department of Natural Resources 2001). Tourist activity included 1.49 million of visitors to the wider 16,000-km2 national forest in 2016 (USDA Forest Service 2016). USSNF wolves belong to the subspecies C. l. nubilus (Nowak 2009 but see Mech et al. 2011) and were legally protected there during the study.
-
(3)
Sierra de la Culebra Regional Game Reserve in northwest Spain (SSC). Elevations range from 800 to 1707 m.a.s.l., and vegetation is dominated by scrubland, oak forests, and conifer plantations. Temperatures in mid-July–late October 2019 and September 2021 varied from − 1 to 36 °C, and average 24-h temperatures, between 6 and 25 °C. Human densities are 2–6 habitants/km2 (municipal register of inhabitants, Statistics National Institute, obtained in https://www.ine.es). SSC wolves are C. l. lupus and during the study were a game species (Consejería de Fomento y Medio Ambiente 2016). SSC is one of the best-known wolf-watching destinations in Europe (Martínez and Pastor-Alfonso 2019). No official tourist data exist, but tourism levels are much lower than USYNP or USSNF (e.g. the Felix Rodríguez de la Fuente Wolf Center in the area received ~ 21,000 visitors/year since 2015, https://www.laopiniondezamora.es/comarcas/2022/05/04/centro-lobo-supera-200-000-65707631.html).
-
(4)
Cordillera Cantábrica, northern Spain (SCC), the rugged region of Asturias and a small mountainous area within Galicia. The studied packs occupy western Cordillera Cantábrica, with elevations from sea level to 1800 m.a.s.l. Vegetation is mainly scrublands, woodlands, and grasslands (pastures) used by free-ranging cattle and horses. Woodlands are dominated by beech, oaks, birch, and anthropogenic chestnut trees. Scrublands predominate, and forest fragmentation is high. Temperatures during the study (July–October 2018, 2019, and 2020) ranged from 6 to 31 °C, and average 24-h temperatures, between 13 and 24 °C. Human densities are 5–47 habitants/km2 (municipal register of inhabitants, Statistics National Institute, obtained in https://www.ine.es). Wolves in SCC are C. l. lupus. During the study, some lethal control was conducted by rangers (annual quota) based on conflict level (Consejería de Agroganadería y Recursos Autóctonos 2015).
-
(5)
Tierra de Campos, northwestern Spain (STC). The area comprises flat, almost treeless agricultural land, with cereal and maize fields providing cover for wolves during certain seasons. Remnant forests cover 7% of the area (Blanco and Cortés 2007). Temperatures in early August–late September 2019 and 2021 ranged from 7 to 34 °C, and average 24-h temperatures, 15 and 25 °C. Human densities are 2–7 habitants/km2 (municipal register of inhabitants, Statistics National Institute, obtained in https://www.ine.es). STC wolves are C. l. lupus and were a game species during the study (Consejeria de Fomento y Medio Ambiente 2016).
-
(6)
Maharashtra, India (INM), a drought-prone area of the Deccan Biogeographic Zone. Major habitat types comprise pure grassland, grassland dotted with shrubs and trees, grazing land, and crop fields. Grassland is distributed in fragmented patches, forming a mosaic of grazing land, agricultural land, and human settlements. Temperatures in early January–late March 2020 ranged from 10 to 37 °C, and average 24-h temperatures, 17 and 29 °C. INM wolves are C. l. pallipes and are legally protected in the study area (Wildlife Protection Act 1972). INM has a human density of 224 inhabitants/km2 in the wolf area (Singh and Kumara 2006).
Areas where spontaneous vocalizations of wild wolves around rendezvous sites were assessed: USYNP (U.S. Yellowstone National Park), USSNF (U.S. Superior National Forest), SSC (Spain Sierra de la Culebra), SCC (Spain Cordillera Cantábrica), STC (Spain Tierra de Campos), and INM (India Maharastra). Base map layer obtained in https://www.worldpop.org/
Overall, we studied 24 wolf packs during the pup-rearing season (3 in USYNP, 2 in USSNF, 5 in SSC, 5 in SCC, 2 in STC, and 7 in INM, all of them with pups (Table 6 in Appendix), obtaining 1225 wolf-pack days of recordings using ARUs (Audiomoth, version 1.1.0 and 1.2.0, Hill et al. 2017). Fieldwork was conducted during summer–fall of 2018, 2019, 2020, and 2021, when pups remain in rendezvous sites (RS). In India, because births occur in winter (Dave and Raval 2019), recordings were obtained during January–March 2020. During the RS season, pack activity is focused on rearing the pups, the only time when one can reliably locate all pack members in one place repeatedly so ARUs can be stationed (Ausband et al. 2016; Gable et al. 2018). We detected pups and located RS by searching for wolf signs and howling surveys (Jiménez et al. 2016), camera traps (Galaverni et al. 2012), wolf sightings or data from collared wolves. We installed 1–6 ARUs per RS, having up to 3 units recording simultaneously in 75% of packs (Table 6 in Appendix). Most ARUs (68%) were installed at 500–1000 m from the RS, ranging from 200 m to 1.6 km, and recorded 24 h/day. In six packs studied in Spain, we did not record from sunrise till sunset (Table 6 in Appendix). To ensure that RS were active we used data from collared wolves (17 packs) or checked for pups via observation stations (5 packs) or simulated howling every 5 to 10 days (2 packs) (Ausband et al. 2010; Gable et al. 2018). We considered a RS period the time since ARU installation until the last pup presence detected. We discarded data from nights with simulated howling.
We used the same AudioMoth configuration settings as in Barber-Meyer et al. (2020) and Kaleidoscope Pro 5.1.9 g (Wildlife Acoustics 2017) to select audiofiles of wolf vocalizations (signal-detection parameters: frequency range 300–1000 Hz, length of detection = 3–20 s, maximum inter-syllable gap = 1 s). We then examined the audiofiles to audibly distinguish howls by a single individual (solo) and choruses (> 1 individual vocalizing simultaneously). We determined that choruses included pups when comprised vocal signals consistent with descriptions of pup vocalizations (Palacios et al. 2016). We arbitrarily grouped all consecutive vocalizations with silence intervals < 60 s as one event, as 60 s is approximately the mean chorus-howl duration for different wolf populations (Martí-Domken et al. 2022). Howling events included solo howl series (one to several howls by one individual) and chorus howls (events that also could include solo howls when the interval between the vocalizations was < 60 s).
Howling rates
We calculated daily vocal rates (solo-howl rates and chorus-howl rates) of wolf packs for RS periods (number of vocalization events/number of sampling days), considering only days with all the scheduled hours recorded. Central daylight hours generally correspond to wolf inactivity (Packard 2003). Thus, to estimate daily vocal rates, we defined “days” as 24 h beginning at noon local time (wolves commonly begin activity at dusk and end at dawn). We compared daily vocal rates obtained for solo howls and chorus howls via a Welch two-sample t-test. For each area, we assessed the hourly variation of howling activity as the number of howling events each hour/number of sampling days.
We did not calculate vocalization rates in two cases: the 8-Mile pack at USYNP and the 7268 pack at USSNF. The 8-Mile pack RS was used by only part of the 8-Mile pack, a subordinate breeding female and a few others. The main 8-Mile wolves had another RS concurrently about 15-km away. With the 7268 pack the RS was moved, so we had to move the ARUs. Although some vocalizations were recorded, we could not be confident that our recordings included all the howling activity of the pack around the RS during an RS period.
Factors influencing howling behavior
We used daily weather data to estimate the weather conditions during spontaneous howls (mean wind speed; mean, minimum and maximum temperature; and daily precipitation). Weather data for the United States and India were gathered by https://www.wunderground.com/history/ and, for Spain, from AEMET, https://opendata.aemet.es/, from the closest weather station to the specific study area. We also tested if moon phase influenced spontaneous howling, considering the percentage of moon illuminated by the sun at midnight, obtained from https://phasesmoon.com/.
For each pack, we selected a 25-km2 square with the RS at the center and estimated the human density living there (HAB, data obtained in https://www.worldpop.org/). As an index of topography ruggedness (MEANSLOPE), we used Google Earth (Google Earth Pro 7.3, https://www.google.com/earth/, base map data from Google, Landsat 2015) to plot circles for each RS (radius = 2 km with the RS at the center) and measured the mean slope of the resulting circumference.
We used winter wolf densities as an index of abundance: 50–70/1000 km2 in northern Yellowstone (Smith et al. 2020a), 16/1000 km2 in the Superior National Forest (2060-km2 study area—Barber-Meyer 2022), < 10/1000 km2 in India (Jhala et al. 2022). In Spain, where no such data were available, we used the number of packs estimated in each area (Consejería de Medio Rural y Cohesión Territorial 2021, and Consejería de Fomento y Medio Ambiente 2019). We multiplied the number of packs by 4.2 wolves (Iberian wolf mean pack size in winter (Fernández-Gil et al. 2020)) and divided by the area to estimate density (non-pack wolves were not included). We also used size of the studied packs to assess the influence of wolf numbers on wolf-howling behavior. We used the number of days since the recordings began to estimate pup growth/age.
We used generalized linear mixed models (GLMMs) with binomial error distribution and logit link to assess which factors influenced presence/absence of (1) solo howls in a day, and (2) chorus howls in a day. We removed highly correlated variables (Pearson correlation coefficient ≥ 0.5) to avoid multicollinearity and assessed leverage and Cook’s Distance (values < 0.2 indicated acceptable influence) to examine the effects of potential outliers on the regression. Finally, we included AREA (study area), ARUS (the number of ARUs used), MINDIS (distance between the RS and the closest ARU), WIND (daily-mean-wind speed); TEMP (daily mean temperature); PREC (daily precipitation), MOON (percentage of visible moon), HAB (human population around the RS), MEANSLOPE, and AGE (number of days since recordings began) as fixed factors, and pack and study area were included as a nested random factor. We built a set of competing GLMMs considering all the possible combinations using the selected variables (including the null model; i.e. the intercept-only model). We used Akaike’s Information Criterion (AIC) to rank models, selecting the model with the lowest AIC (Burnham and Anderson 2002). Models within two AIC units were considered to be of similar support, and cumulative AIC weights were calculated to evaluate the strength of each model. We estimated the variance explained by the best model calculating marginal R2 (variance explained by fixed factors) and conditional R2 (variance explained by both fixed and random factors (Nakagawa and Schielzeth 2013)). We did not include in the models pack size (unavailable for all the packs) or wolf abundance (collinearity with AREA) and computed Pearson correlation coefficients to assess the relationship between wolf numbers (pack size and wolf abundance) and howling rates. GLMMs were fitted in R software (R Development Core Team 2016) using the “lme4” package (Bates et al. 2015).
Detection of wolf packs and the presence of pups
We calculated the average cumulative probability of howl detection by area to estimate the mean effort needed (in days of recording) to record vocal activity (including solo and chorus howls), pack vocal activity (chorus howls), and pup vocal activity with 80 and 95% probability.
Results
We recorded 605 wolf chorus howls and 224 solo-howl series emitted spontaneously by 24 wolf packs around RS. We recorded pups in 88% of packs. The percentage of days with spontaneous vocal activity ranged from 12.50% in Spain (SCC, RS period = 16 days) to 94.12% in the United States (USYNP, RS period = 17 days, Table 7 in Appendix). Spontaneous solo-howl series recorded in a day varied from 1 to 14, but during most days with solo howling we recorded one (52% of days), two (23%), or three (11%) solo howls. Chorus howls recorded in a day ranged from 1 to 14, most days recording 1–3 chorus howls (1: 52%, 2: 27%, and 3: 8% of days).
Howling rates
Howling recorded near RS mainly involved packs. Daily solo rates were fewer than chorus rates (Welch two sample t-test = 2.16; df = 39; p = 0.037), ranging from 0 in four RS periods (corresponding to SSC and SCC packs) to 3.47 solo howls/day in Junction Butte pack, USYNP (Table 7 in Appendix). Daily chorus rates varied from 0 (Carondio pack, SCC, and Nannaj pack, INNM) to 5.29 chorus/day in Junction Butte pack, USYNP (Table 7 in Appendix).
Howling occurred primarily at night in most areas (Fig. 2). The highest hourly rates were found in USYNP where the Junction Butte pack vocalized by far the most of all the packs we studied. Howling activity showed two peaks in all the studied areas. Chorus howls occurred the most after sunset at USSNF, SSC, SCC, STC, and INM, and after sunrise at USYNP. Almost no vocalizing was recorded before sunshine and after sunrise at USSNF, SSC, SCC, and STC. Indian wolves also howled before sunset and after sunrise and howling peaked after sunrise at USYNP.
Hourly distribution of vocal activity recorded in summer around rendezvous sites (winter 2020 in INM). Hourly rates: number of howling events each hour/number of sampling days. Local sunset (ss) and sunrise (sr) times are plotted with vertical lines (local ss and sr at the middle of the study for each area)
Factors influencing howling behavior
Solo howls decreased with wind speed and increased with the number of ARUs used and the distance of the closest ARU to the RS (Table 1). Considering all the competing models with AIC ≤ 2, only wind speed significantly affected solo howling (Tables 2, 3). Chorus howls decreased with wind speed and human density around RS and was influenced by study area (Table 1). Tukey post-hoc analyses revealed wolves howled more in USYNP than in the STC and SCC (p = 0.019 and p = 0.001 respectively); SSC wolves howled more than those in SCC (p = 0.036), and those in INM more than SCC wolves (p = 0.036). Considering all models with AIC ≤ 2, human density around RS and wind speed significantly affected chorus howling (Tables 4, 5).
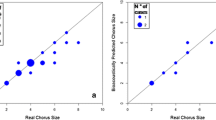
We found no relationship between vocal rates and wolf abundance (solo howl rates: Spearman’s rank correlation, rho = 0.137, p = 0.514; chorus howl rates: Spearman’s rank correlation, rho = 0.322, p = 0.116). However, chorus rates increased with pack size and the number of adults/subadults per pack (Spearman’s rank correlation between pack size and chorus rates: rho = 0.758, p = 0.018; number of adults/subadults and chorus rates: rho = 0.785, p = 0.012; number of pups and chorus rates: rho = 0.651, p = 0.054).
Detection of wolf packs and the presence of pups
On average, we found an 80% probability of detecting vocalizations, choruses, and pups in 3.73, 4.45, and 5.15 days respectively; and a 95% probability in 7.7, 9.5, and 11.3 days (Fig. 3). The highest cumulative probability of howling detection occurred in USYNP and SSC, and the lowest in SCC (Fig. 3).
Discussion
We studied spontaneous howling in wild wolves at RS in six areas across the wolf´s world range, recording 829 howling events, most of them chorus howls with pups. Some workers propose that howling helps coordinate events such as departures and movements around homesites and serves as long-range intrapack communication as pups become more mobile (Harrington and Mech 1978). Our results include important benchmark values useful for studies of the rates and temporal distribution of vocal behavior in wolves. Excepting the Junction Butte pack at USYNP, with 5.29 chorus howls/day and 3.47 solo howls/day, the howling rates we obtained are lower than those of captive wolves of the same subspecies (Palacios et al. 2021). Rates of captive wolves should be higher than those of wild wolves because all vocalizations of captives are easily recorded, and captives have no need to limit howling to minimize detection by humans or other wolf packs that could endanger them (e.g., infanticide; Smith et al. 2015). We found 31 and 42% of days with vocal activity in two RS periods for the USSNF 7282 pack, compared with the 21–27% of days at two other wolf homesites in our study area (Harrington and Mech 1978). However, hourly distribution of chorus howls was similar in both studies, peaking at 11 p.m. and a lower peak before sunrise.
Wind influences wolf response rate to simulated howls (Joslin 1967; Ausband et al. 2020), including away from homesites (Harrington and Mech 1982). Factors such as wind speed also affect spontaneous howling in captivity (Palacios et al. 2021). Our results accord with those findings, with more vocalizations (both solo and chorus howls) with low wind. This result could reflect that ARUs have higher detectability with low wind or winds that carry the vocalizations to the detector (Barber-Meyer et al. 2020). However, because in most cases we used several ARUs at each site, this would minimize that problem. In any case, our models showed that weather had low explanatory power, perhaps due to the low variability among weather conditions during our study.
The influence of lunar cycles on animal behavior is well documented. Elephants (Loxodonta africana), for instance, decrease crop-raiding during the full moon, apparently to avoid the height of human activity (Gunn et al. 2014). Sánchez-Ferrer et al. (2016) reported decreased movement during new and full moon in captive Mexican wolves, dependent on the age of the individuals, and chorus howling is negatively related to increasing moonlight in coyotes, Canis latrans (Bender et al. 1996). In our study, however, moonlight had no effect on spontaneous vocalizations, similar to wild-wolf responses to simulated howls (Joslin 1967; Harrington and Mech 1982).
We found differences in the factors influencing solo and chorus howling. Solo howls increased with the numbers of ARUs used and with the distance of the closest ARU to the RS, factors that did not appear relevant in the chorus-howl models. This could be due to the fact that, near RS, solo howls trigger the quick response of other pack members, thus becoming a chorus howl. The fact that solo howls are harder to detect than chorus howls (we expect to lose 63% of chorus howls and 91% of solo howls emitted at 0.54 km from the ARUs, (Barber-Meyer et al. 2020)) might explain the low solo howls detected and why the more ARUs used, the easier to detect solo howls.
Our results indicate that chorus howling in wild wolves depends on the area, pack size, and density of people living near RS. Differences in study area could reflect subspecies differences. Howling differs among subspecies in acoustic structure (Kershenbaum et al. 2016) and chorus-howl length, with Mexican wolves’ howls shorter than those of other North American wolves (Servín 2000). Also, captive Iberian wolves howl less than North American wolves (Palacios et al. 2021). However, our unbalanced sample, with most of our packs of one subspecies, prevents assessment of a subspecies effect on howling. Differences in area reflect the low chorus howling at SCC compared to USYNP, SSC, and INM, which could be because only one recorder was used for most Cantabrian packs or because wolves living in the humanized Western part of Cordillera Cantábrica howl less than other wolves, a subject that warrants further research.
Larger packs howled more often, similar to previous studies and probably because there simply were more wolves (Harrington and Mech 1978; Harrington 1989). That was not true for wolf abundance. Acoustic territorial displays increase with the number of competitors (Eckenweber and Knörnschild 2013), but assuming that most howling around RS represents intrapack communication, this result could be expected because smaller packs would howl less around RS even where wolf density is high. However, a lack of relationship between howling and wolf density could also be because our measurement and accuracy of wolf abundance varied considerably.
Packs vocalized more where fewer people were living near RS, suggesting that wolves adjust their vocal behavior when living near humans. Many animals adapt their vocal behavior to anthropogenic sounds, such as changing their temporal patterns or the frequency and amplitude of their vocalizations (Francis and Barber 2013). Others adjust their vocal activity to their perceived predation risk (Abbey-Lee et al. 2016). If reduced vocal activity increases survival of wolves living near humans, we would expect those persisting in highly human-dominated landscapes and near urban areas to howl less. Our lowest vocal rates occurred in packs with greatest human densities around RS in INM, SCC, and STC. Notably, the most howling and the highest cumulative probabilities of wolf detection happened in the USYNP Junction Butte pack, where the fewest people live around RS but where > 800,000 people visited during the study. Yellowstone National Park is the world’s most iconic wildlife-watching place, and wildlife is fully protected. In addition, in Spain the most vocalizations transpired in SSC where, despite wolves having been a game species until September 2021, wolf watching is an important activity, and attitudes toward wolves are positive compared to other areas of the Iberian Peninsula (Pettersson et al. 2021). These findings suggest that neutral or positive human activities and attitudes might counteract the negative effect of human presence on wolves’ vocal behavior.
Besides decreasing overall vocalization rates, another adaptation of vocalization behavior to perceived risk could be a change in the temporal distribution of vocal activity because the most howling before sunset and after sunrise occurred in USYNP and INM, where wolves are fully protected. Human activity influences wolf resting-site selection (Llaneza et al. 2016) and overall habitat selection near road corridors and during higher human activity (Anton et al. 2020). Wolves minimize the risk of exposure at pup-rearing sites by avoiding human structures, selecting shelter in vegetation and avoiding agricultural lands (Sazatornil et al. 2016, but see Thiel et al. 1998). Our results suggest that wolves howl less and during darkness probably because of perceived risk from humans, a finding that merits further research.
Passive automatic recorders constitute a useful method to detect wolf presence (Suter et al. 2017; Barber-Meyer et al. 2020). Here, we show that pups commonly participate in spontaneous chorus howls around the RS across packs of different wolf subspecies in different areas with different habitat and human pressure. Hence, using ARUs can confirm wolf reproduction by detecting spontaneous chorus howls with pups. Although simulated howling surveys can be very effective (Harrington and Mech 1982; Llaneza et al. 2005) and are extensively used to study the status of wolf populations (Kaczensky et al. 2013), ARUs could solve the main problems associated with eliciting responses to simulated howls: possible effect on wolf behavior, potential increase of vulnerability to humans, low reliability of estimates from wolf responses (Palacios et al. 2017), and uncertainty from lack of response.
Simulated howls may affect wolves’ movements. Wolves often approach the human “howler” (Murie 1944; Harrington and Mech 1979) or the howl box (Brennan et al. 2013) howling stations and other pack members after simulated howls (Leblond et al. 2017). Leblond et al (2017) suggested that simulated howling was unlikely to have strong negative effects on wolf movement but encouraged further evaluation. This suggestion warrants consideration especially in the design of intensive wolf-monitoring programs. For example, in a national wolf-population monitoring in Slovenia during 2016, 178 surveyors howled two consecutive nights in 436 3 × 3-km grid cells (Ražen et al. 2020). Simulated howling in human-dominated landscapes may increase pack vulnerability if not properly planned, especially if the low vocal rates we found (except in USYNP) are an adaptation to minimize risk by humans. Other problems with active howling surveys are related to the low reliability of aural estimates of pup presence and pack size from wolf responses (Palacios et al. 2016, 2017) and the response rates. Radiocollared wolves in the Montmorency Forest (Quebec, Canada) only responded to 5% of simulated howls (Leblond et al. 2017) and wolf-response rates may range from 0.1 to 0.56 (see review in Leblond et al. 2017).
Using ARUs 500–1000 m from an occupied RS, we recorded multiple chorus howls including pups in 88% of the packs without altering wolf behavior or increasing detectability by humans. Depending on the area, 4–15 days were needed to detect the pack and 5–21 days to detect pups, with a probability of 95%, with the lowest cumulative probabilities of wolf detection found at SCC. Most of the packs (76%) were studied using three ARUs within 1000 m, but at SCC we used only one ARU per RS, which could have affected the results. Although number of ARUs and distance from RS were not included in the chorus-howl model, we are aware that SCC results could be underestimated. However, the information obtained can be used as a benchmark in the design of wolf-monitoring programs.
Besides demonstrating the potential of ARUs to study wolf acoustic behavior, our results show that ARUs could be useful to monitor wolf populations as complementary or even alternative to howling surveys. In the present study we usually deployed three AudioMoths (~ US$115/device) around a RS, changed batteries every 10 days (all ARUs at each RS could be checked in one day), and spent one day searching a month of recordings. When RS locations are unknown, and the aim is to detect reproduction, installing the ARUs in zones with concentration of wolf signs (Llaneza et al. 2005) could be useful. This method might not be cost-effective compared to camera traps or scat-detection dogs to detect wolf presence in areas with low wolf densities (Cozzi et al. 2021), or might not be practical in large roadless areas, but it can be very useful in human-dominated landscapes or areas with high conflict, when other methods might be less suitable.
References
Abbey-Lee RN, Kaiser A, Mouchet A, Dingemanse NJ (2016) Immediate and carry-over effects of perceived predation risk on communication behavior in wild birds. Behav Ecol 27:708–716. https://doi.org/10.1093/beheco/arv210
Anton CB, Smith DW, Suraci JP, Stahler DR, Duane TP, Wilmers CC (2020) Gray wolf habitat use in response to visitor activity along roadways in Yellowstone National Park. Ecosphere 11(6):e03164. https://doi.org/10.1002/ecs2.3164
Ausband DE, Mitchell MS, Doherty K et al (2010) Surveying predicted rendezvous sites to monitor gray wolf populations. J Wildl Manag 74:1043–1049. https://doi.org/10.2193/2009-303
Ausband DE, Mitchell MS, Bassing SB et al (2016) Dog days of summer: influences on decision of wolves to move pups. J Mammal 97:1282–1287. https://doi.org/10.1093/jmammal/gyw114
Ausband DE, Bassing SB, Mitchell MS (2020) Environmental and social factors influencing wolf (Canis lupus) howling behavior. Ethology 126:890–899. https://doi.org/10.1111/eth.13041
Barber-Meyer SM (2022) Can non-invasive methods replace radiocollar-based winter counts in a 50-year wolf study? Lessons learned from a three-winter trial. Wildl Res. https://doi.org/10.1071/WR22001
Barber-Meyer SM, Palacios V, Marti-Domken B, Schmidt LJ (2020) Testing a new passive acoustic recording unit to monitor wolves. Wildl Soc Bull 44:590–598. https://doi.org/10.1002/wsb.1117
Bates D, Mächler M, Bolker B, Walker S (2015) Fitting linear mixed-effects models using lme4. J Stat Softw 67:1–48. https://doi.org/10.18637/jss.v067.i01
Bender DJ, Bayne EM, Brigham RM (1996) Lunar condition influences coyote (Canis latrans) howling. Am Midl Nat 136:413–417
Blanco JC, Cortés Y (2007) Dispersal patterns, social structure and mortality of wolves living in agricultural habitats in Spain. J Zool 273:114–124. https://doi.org/10.1111/j.1469-7998.2007.00305.x
Brennan A, Cross PC, Ausband DE et al (2013) Testing automated howling devices in a wintertime wolf survey. Wildl Soc Bull 37:389–393. https://doi.org/10.1002/wsb.269
Burnham KP, Anderson DR (2002) Model selection and multimodel inference: a practical information theoretic approach. Springer, New York
Consejería de Agroganadería y Recursos Autóctonos (2015) Plan de Gestión del Lobo en el Principado de Asturias. Boletín Oficial del Principado de Asturias 1–9
Consejeria de Fomento y Medio Ambiente (2016) Plan de Conservación y Gestión del Lobo en Castilla y León
Consejería de Fomento y Medio Ambiente (2019) Plan de aprovechamientos comarcales de lobo en los terrenos cinegéticos situados al norte del Río Duero en Castilla y León para las temporadas 2019/2020, 2020/2021 y 2021/2022.
Consejería de Medio Rural y Cohesión Territorial (2021) Plan Anual de Actuaciones de Control del Lobo 2021–2022
Cozzi G, Hollerbach L, Suter SM et al (2021) Eyes, ears, or nose? Comparison of three non-invasive methods to survey wolf recolonisation. Mamm Biol 101:881–893. https://doi.org/10.1007/s42991-021-00167-6
Dave MS, Raval J (2019) The behaviour of Indian gray wolf (Canis lupus pallipes) in captivity at Sakkarbaug Zoo Junagadh, Gujarat, India. Int J Environ Ecol Fam Urban Stud 9:55–68. https://doi.org/10.24247/ijeefusapr20195
Department of Natural Resources M (2001) Minnesota wolf management plan. St Paul, Minnesota
Despain DG (1990) Yellowstone vegetation: consequences of environment and history in a natural setting. Roberts Rinehart Publishers, Boulder
Eckenweber M, Knörnschild M (2013) Social influences on territorial signaling in male greater sac-winged bats. Behav Ecol Sociobiol 67(639):648. https://doi.org/10.1007/s00265-013-1483-z
Fernández-Gil A, Quevedo M, Barrientos LM, Nuño A, Naves J, de Gabriel M, Ordiz A, Revilla E (2020) Pack size in humanized landscapes: the Iberian wolf population. Wildl Biol. https://doi.org/10.2981/wlb.00594
Francis CD, Barber JR (2013) A framework for understanding noise impacts on wildlife: an urgent conservation priority. Front Ecol Environ 11:305–313. https://doi.org/10.1890/120183
Gable TD, Windels SK, Bump JK (2018) Finding wolf homesites: improving the efficacy of howl surveys to study wolves. PeerJ 6:e5629. https://doi.org/10.7717/peerj.5629
Galaverni M, Palumbo D, Fabbri E et al (2012) Monitoring wolves (Canis lupus) by non-invasive genetics and camera trapping: a small-scale pilot study. Eur J Wildl Res 58:47–58. https://doi.org/10.1007/s10344-011-0539-5
Gunn J, Hawkins D, Barnes RFW et al (2014) The influence of lunar cycles on crop-raiding elephants; evidence for risk avoidance. Afr J Ecol 52:129–137. https://doi.org/10.1111/aje.12091
Harrington FH (1989) Chorus howling by wolves: acoustic structure, pack size and the beau geste effect. Bioacoustics 2:117–136. https://doi.org/10.1080/09524622.1989.9753122
Harrington FH, Asa CS (2003) Wolf communication. In: Mech LD, Boitani L (eds) Wolves: behavior, ecology, and conservation. University of Chicago Press, Chicago, pp 66–103
Harrington FH, Mech LD (1978) Howling at two Minnesota wolf pack summer homesites. Can J Zool 56:2024–2028
Harrington FH, Mech LD (1979) Wolf howling and its role in territory maintenance. Behaviour 68:207–249
Harrington FH, Mech LD (1982) An analysis of howling response parameters useful for wolf pack censusing. J Wildl Manag 46:686–693
Heinselman M (1993) The boundary waters wilderness ecosystem. University of Minnesota Press, Minneapolis
Hill AP, Prince P, Covarrubias EP et al (2017) AudioMoth: evaluation of a smart open acoustic device for monitoring biodiversity and the environment. Methods Ecol Evol 12:3218–3221. https://doi.org/10.1111/ijlh.12426
Jhala Y, Saini S, Kumar S, Qureshi Q (2022) Distribution, status, and conservation of the Indian Peninsular wolf. Front Ecol Evol. https://doi.org/10.3389/fevo.2022.814966
Jiménez J, García EJ, Llaneza L et al (2016) Multimethod, multistate Bayesian hierarchical modeling approach for use in regional monitoring of wolves. Conserv Biol. https://doi.org/10.1111/cobi.12685
Joslin PWB (1967) Movements and home sites of timber wolves in Algonquin Park. Am Zool 7:279–288
Kaczensky P, Chapron G, von Arx M et al (2013) Status, management and distribution of large carnivores—bear , lynx , wolf & wolverine—in Europe. IUCN/SSC Large Carnivore Initiative for Europe
Kershenbaum A, Root-Gutteridge H, Habib B et al (2016) Disentangling canid howls across multiple species and subspecies: structure in a complex communication channel. Behav Process 124:149–157. https://doi.org/10.1016/j.beproc.2016.01.006
Kershenbaum A, Owens JL, Waller S (2019) Tracking cryptic animals using acoustic multilateration: a system for long-range wolf detection. J Acoust Soc Am 145:1619–1628. https://doi.org/10.1121/1.5092973
Larsen HL, Pertoldi C, Madsen N et al (2022) Bioacoustic detection of wolves: identifying subspecies and individuals by howls. Animals 12:631. https://doi.org/10.3390/ani12050631
Leblond M, Dussault C, St-Laurent M-H (2017) Space use by gray wolves (Canis lupus) in response to simulated howling: a case study and a call for further investigation. Can J Zool 95:221–226. https://doi.org/10.1139/cjz-2016-0191
Llaneza L, Ordiz A, Palacios V, Uzal A (2005) Monitoring wolf populations using howling points combined with sign survey transects. Wildl Biol Pract 1:108–117. https://doi.org/10.2461/wbp.2005.1.13
Llaneza L, García EJ, Palacios V et al (2016) Resting in risky environments: the importance of cover for wolves to cope with exposure risk in human-dominated landscapes. Biodivers Conserv 25:1515–1528. https://doi.org/10.1007/s10531-016-1134-6
Lynch MA, Lynch HJ (2017) Variation in the ecstatic display call of the Gentoo Penguin (Pygoscelis papua) across regional geographic scales. Auk 134:894–902. https://doi.org/10.1642/AUK-17-4.1
Martí-Domken B, Palacios V, Monzón A (2022) Pack members shape the acoustic structure of a wolf chorus. Acta Ethol 25(1):3. https://doi.org/10.1007/s10211-021-00388-5
Martínez FA, Pastor-Alfonso MJ (2019) Wolf watching tourism in the Culebra Mountain Range (Spain). Rev Lider 35:137–160
Mazzini F, Townsend SW, Virányi Z, Range F (2013) Wolf howling is mediated by relationship quality rather than underlying emotional stress. Curr Biol 23(17):1–4. https://doi.org/10.1016/j.cub.2013.06.066
McIntyre R, Theberge JB, Theberge MT, Smith DW (2017) Behavioral and ecological implications of seasonal variation in the frequency of daytime howling by Yellowstone wolves. J Mammal 98:827–834. https://doi.org/10.1093/jmammal/gyx034
Mech LD (1970) The wolf: the ecology and behavior of an endangered species. The Natural History Press, Garden City
Mech LD (2009) Long-term research on wolves in the Superior National Forest. In: Wydeven AP, Van Deelen TR, Heske EJ (eds) Recovery of Gray Wolves in the Great Lakes Region of the United States. Springer, New York, pp 15–34
Mech LD, Nowak RM, Weisberg D (2011) Use of cranial characters in the taxonomy of the Minnesota Wolf (Canis sp.). Can J Zool 89:1188–1194. https://doi.org/10.1139/Z11-097
Murie A (1944) The wolves of Mount McKinley. Fauna of the National Parks of the United States. Fauna series no 5
Nakagawa S, Schielzeth H (2013) A general and simple method for obtaining R2 from generalized linear mixed-effects models. Methods Ecol Evol 4:133–142. https://doi.org/10.1111/j.2041-210x.2012.00261.x
Nowak RM (2009) Taxonomy, morphology, and genetics of wolves in the Great Lakes region. In: Wydeven AP, Van Deelen TR, Heske EJ (eds) Recovery of Gray Wolves in the Great Lakes Region of the United States: an endangered species success story. Springer, New York, pp 233–250
Nowak S, Jedrzejewski W, Schmidt K et al (2007) Howling activity of free-ranging wolves (Canis lupus) in the Białowieza Primeval Forest and the Western Beskidy Mountains. J Ethol 25:231–237. https://doi.org/10.1007/s10164-006-0015-y
Packard JM (2003) Wolf behavior: reproductive, social, and intelligent. In: Mech LD, Boitani L (eds) Wolves: behavior, ecology, and conservation. University of Chicago Press, Chicago, pp 35–65
Palacios V, López-Bao JV, Llaneza L et al (2016) Decoding group vocalizations: the acoustic energy distribution of chorus howls is useful to determine wolf reproduction. PLoS ONE 11:e0153858. https://doi.org/10.1371/journal.pone.0153858
Palacios V, Font E, García EJ et al (2017) Reliability of human estimates of the presence of pups and the number of wolves vocalizing in chorus howls: implications for decision-making processes. Eur J Wildl Res 63:59. https://doi.org/10.1007/s10344-017-1115-4
Palacios V, Barber-Meyer SM, Martí-Domken B, Schmidt LJ (2021) Assessing spontaneous howling rates in captive wolves using automatic passive recorders. Bioacoustics. https://doi.org/10.1080/09524622.2021.2006083
Papin M, Pichenot J, Guérold F, Germain E (2018) Acoustic localization at large scales: a promising method for grey wolf monitoring. Front in Zool 15:1–10. https://doi.org/10.1186/s12983-018-0260-2
Pettersson HL, Quinn CH, Holmes G et al (2021) Welcoming wolves? Governing the return of large carnivores in traditional pastoral landscapes. Front Conserv Sci 2:1–19. https://doi.org/10.3389/fcosc.2021.710218
R Development Core Team (2016) R: A language and environment for statistical computing. Austria, Vienna
Ramsier MA, Cunningham AJ, Moritz GL et al (2012) Primate communication in the pure ultrasound. Biol Lett 8:508–511. https://doi.org/10.1098/rsbl.2011.1149
Ražen N, Kuralt FU et al (2020) Citizen science contribution to national wolf population monitoring: what have we learned? Eur J Wildl Res. https://doi.org/10.1007/s10344-020-01383-0
Rocha LHS, Ferreira LS, Paula BC et al (2015) An evaluation of manual and automated methods for detecting sounds of maned wolves (Chrysocyon brachyurus Illiger 1815). Bioacoustics. https://doi.org/10.1080/09524622.2015.1019361
Sánchez-Ferrer JC, Servín J, Muñoz-Delgado J et al (2016) Variations in the locomotor activity of the Mexican wolf (Canis lupus baileyi) respect to moon periodicity: a study in an outdoor enclosure. Biol Rhythm Res 47:539–551. https://doi.org/10.1080/09291016.2016.1164281
Sazatornil V, Rodríguez A, Klaczek M et al (2016) The role of human-related risk in breeding site selection by wolves. Biol Conserv 201:103–110. https://doi.org/10.1016/j.biocon.2016.06.022
Servín J (2000) Duration and frequency of chorus howling of the Mexican wolf (Canis lupus baileyi). Acta Zool Mex 80:223–231. https://doi.org/10.21829/azm.2000.80801902
Singh M, Kumara HN (2006) Distribution, status and conservation of Indian gray wolf (Canis lupus pallipes) in Karnataka, India. J Zool 270:164–169. https://doi.org/10.1111/j.1469-7998.2006.00103.x
Smith DW, Metz MC, Cassidy KA et al (2015) Infanticide in wolves: seasonality of mortalities and attacks at dens support evolution of territoriality. J Mammal 96:1174–1183. https://doi.org/10.1093/jmammal/gyv125
Smith DW. Stahler DR, MacNulty DR (eds) (2020a) Yellowstone Wolves. Science and Discovery in the World’s First National Park. The University of Chicago Press, Chicago
Smith DW, Stahler DR, Cassidy KA et al (2020b) Yellowstone National Park Wolf Project Annual Report 2019. Yellowstone National Park, WY, USA, YCR-2020-01
Suter SM, Giordano M, Nietlispach S et al (2017) Non-invasive acoustic detection of wolves. Bioacoustics 26:237–248. https://doi.org/10.1080/09524622.2016.1260052
Thiel RP, Merrill S, Mech LD (1998) Tolerance by denning wolves, Canis lupus, to human distrubance. Can Field Nat 112:340–342
USDA Forest Service (2016) National visitor use monitoring data collected FY 2016. https://apps.fs.usda.gov/nvum/results/A09009.aspx/FY2016
Volodina EV, Volodin IA, Frey R (2021) Male impala (Aepyceros melampus) vocal activity throughout the rutting period in Namibia: daily and hourly patterns. Afr J Ecol 60:95–99. https://doi.org/10.1111/aje.12923
Wildlife Acoustics (2017) Kaleidoscope analysis software. https://www.wildlifeacoustics.com/products/kaleidoscope-pro. Accessed 4 Aug 2020
Wildlife Protection Act (1972) Wildlife Protection Act number 53 of 1972. Ministry of Law Justice and Company, New Delhi
Acknowledgements
Funding was provided by the National Geographic Society and the U.S. National Park Service. We thank numerous field technicians for collecting the data, and S. H. Fritts and three anonymous reviewers. Also, the staff of the Regional Government of Asturias and Pedro Luis Ramos from Junta de Castilla y León, and the rangers of Asturias and Sierra de la Culebra Regional Hunting Preserve, for their support. JVLB was supported by the Spanish Ministry of Economy, Industry and Competitiveness (RYC-2015-18932, CGL2017-87528-R AEI/FEDER EU), by the Regional Government of Asturias, and by a GRUPIN research grant from the Regional Government of Asturias (IDI/2021/000075). Emma Narotzky and Francisco Javier Lema helped in Yellowstone National Park. Any use of trade, firm, or product names is for descriptive purposes only and does not imply endorsement by the U.S. Government.
Funding
Funding was provided by the National Geographic Society (Grant Number NGS-59729R-19) and the U. S. National Park Service. JVLB was supported by the Spanish Ministry of Economy, Industry and Competitiveness (RYC-2015-18932, CGL2017-87528-R AEI/FEDER EU), by the Regional Government of Asturias, and by a GRUPIN research grant from the Regional Government of Asturias (IDI/2021/000075).
Author information
Authors and Affiliations
Contributions
VP, SB, BH, JVL, DWS, DS, and LDM study design. VP, BM, SB, BH, JVL, EG field work. VP, BM, SB, VS data analysis. DWS and DS field work support. VP manuscript writing. VP, BM, SB, JVL, DS, VS, and LDM manuscript revision. VP and VS prepared the figures.
Corresponding author
Ethics declarations
Competing interests
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Communicated by Sandro Lovari.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Palacios, V., Martí-Domken, B., Barber-Meyer, S.M. et al. Automatic recorders monitor wolves at rendezvous sites: do wolves adjust howling to live near humans?. Biodivers Conserv 32, 363–383 (2023). https://doi.org/10.1007/s10531-022-02506-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10531-022-02506-6