Abstract
The conservation value of forest fragments remains controversial. An extensive inventory of rainforest trees in post-logging regrowth forest in the southern Philippines provided a rare opportunity to compare stem density, species richness, diversity and biotic similarity between two types of post-logging forests: broken-canopy forest fragments and adjacent tracts of closed-canopy ‘contiguous’ forest. Tree density was much lower in the fragments, but rarefied species richness was higher. ‘Hill’ numbers, computed as the exponential of Shannon’s diversity index and the inverse of Simpson’s diversity index, indicated that fragments have higher numbers of typical and dominant species compared to contiguous forest. Beta diversity (based on species incidence) and the exponential of Shannon’s diversity index was higher in fragmented forest, indicating higher spatial species turnover than in contiguous forest samples. Lower mean values of the Chao-Jaccard index in fragmented forest compared to contiguous forest also indicated a lower probability of shared species across fragments. The high species richness of contiguous forest showed that an earlier single logging event had not caused biodiversity to be degraded leaving mostly generalist species. Fragmentation and further low-level utilisation by local farmers has also not caused acute degradation. Post-logging regrowth forest fragments present a window of opportunity for conservation that may disappear in a few years as edge effects become more apparent. For the conservation of trees in forests in south-east Asia generally, our findings also suggest that while conservation of remaining primary forest may be preferable, the conservation value of post-logging regrowth forests can also be high.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Land use change is the main driver of global biodiversity loss (Leemans 2005; Arroyo-Rodriguez et al. 2013). In many developing countries, once-extensive stands of forest are increasingly being reduced to small and isolated fragments (e.g. see Kupfer and Franklin 2009; Lopez et al. 2009; Lôbo et al. 2011). The extent of fragmentation has focused increasing attention on the value of forest fragments for biodiversity conservation. Debate has emerged from suggestions that small fragments provide a ‘safety net’ for species conservation (Turner 1996), a counter view that primary forests are irreplaceable for sustaining biodiversity (see Gibson et al. 2011), to recognition that global increases in deforestation make habitat conservation and restoration increasingly dependent on forest fragments which are functionally viable, i.e. remnants which have not suffered an irreversible decline in diversity (see Kupfer and Franklin 2009; Schleuning et al. 2011; Arroyo-Rodriguez et al. 2013, 2015; Villard and Metzger 2014).
The deleterious effects of fragmenting forest into ‘islands’ are widely reported, e.g. the vulnerability of fragments to logging, fire and further clearing, increased edge effects and consequent reductions in species diversity (see Pimm 1998; Kupfer and Franklin 2009; for species generally, Bruna et al. 2005; for ants and Cayuela et al. 2006; Echeverría et al. 2007; Schleuning et al. 2011; Lôbo et al. 2011 for trees). Small fragments become ‘all-edge’ (Bunyan et al. 2012), with increased radiation transmittance elevating air and soil temperature, reducing air humidity, and increasing rates of nutrient cycling. Over time, fragmented communities become increasingly dominated by ‘generalist’ species which occupy a wide ecological range, disturbance-adapted opportunists and species with small area requirements (Laurance et al. 2002; Lopez et al. 2009; Lôbo et al. 2011). Cleared areas within fragmented landscapes are increasingly susceptible to soil erosion and landslides (Kräuchi et al. 2000).
Our understanding of ecological processes within forest fragments is still evolving. The known deleterious effects of human-induced disturbance (e.g. logging, see Appiah 2013; Cayuela et al. 2006) are countered with evidence that moderate selective logging may not negatively affect the functional viability of forest fragments (Schleuning et al. 2011). McNamara et al. (2012) also found little reduction in the occurrence of primary forest tree species between frequently and infrequently disturbed secondary forests in Laos. The dilemma is that the biological and physical effects of forest fragmentation are poorly understood and a lack of empirical data is hampering researchers’ ability to inform land-management policy makers at a time when forest fragmentation is increasing (Streubig et al. 2008; Gardner et al. 2009; Lopez et al. 2009; Bierregaard et al. 1992). Hence, to facilitate how conservation resources should be allocated to ecological communities within a region, Jost et al. (2010) suggested that conservationists need to know how diversity is partitioned, whether communities are homogenous or distinct and how much each community contributes to regional diversity. For forest fragments, the key question is whether they are a nested sub-set of an overall forested region, or are sufficiently different to warrant specific conservation measures.
Here, we compare tree stem density, species richness, diversity and biotic similarity between two types of post-logging regrowth tropical forest in the Tampakan Highlands of southern Mindanao; closed-canopy ‘contiguous’ forest and broken-canopy forest fragments. After primary forests in the Highlands were heavily logged in the 1970s and 1980s, sawmilling collapsed and logging roads became closed. Since then, regrowth forest has been disturbed by clearing for swidden agriculture and (within fragments), by low levels of utilisation by local people. The fragments have a broken canopy of upper-level trees. These regrowth forests provided an opportunity to compare the biodiversity of fragments with the larger contiguous tracts of forest. First, we use aerial photography to investigate the pattern of deforestation and regrowth in the Tampakan Highlands. Second, we use the results of a field inventory to calculate key diversity metrics and compare them between contiguous and fragmented forest. Given the socio-economic situation in the Tampakan Highlands, we also suggest likely trends in tree species diversity in succeeding years. Finally, we also suggest a best-practice approach to conserving the ecological integrity of native forest in the Highlands.
Methods: design of the inventory and data collection
The study area and the local people
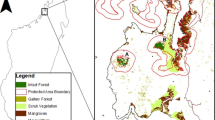
The Philippine Archipelago is recognized as one of the world’s major hotspots of biodiversity and endemism; ~3200 flowering plants are unique to the Philippines, with many of them endemic to just one or several islands (Warburton 2009). The Tampakan highlands are situated ~40 km to the north of General Santos City on the island of Mindanao (Fig. 1a) and cover ~20,000 ha of land ranging from 500 m above sea level (‘masl’) to peaks mostly below 1500 masl (Fig. 1b). Almost all land below 500 m elevation has been cleared for agriculture. At higher elevations, surviving forest is now represented by three blocks of regrowth forests, each of several thousand ha in area, interspersed with numerous smaller fragments. Two forest types are present, i.e. tropical lowland evergreen rainforest which typically occurs between 500 and 1000 masl and tropical lower montane forest which typically occurs above 1000 masl (Fernando et al. 2008).
The highlands are inhabited by members of the B’laan tribe who live in semi-isolation from lowlanders in individual houses or small villages or ‘sitios’. They practice shifting cultivation, with consequent deforestation, erosion and loss of soil fertility. Small-scale clearing, mostly of scrub (i.e. a sparse cover of small trees and bushes) often occurs to an ‘edge’, i.e. a physical boundary such as steep land. Although logging has ceased and many logging roads have fallen into disuse, the B’laan occasionally cut trees for domestic uses, individual trees being fallen and sawn into lumber at the stump.
Designing and undertaking the field inventory: mapping the pattern of deforestation and regrowth in the Tampakan Highlands
As detailed cadastral or soils maps of the area do not exist, we prepared a base map using ArcGIS software and high-quality aerial photographs taken in 1995, 2008 and 2010. The base map depicted:
-
Three stands of closed-canopy, post-logging regrowth ‘contiguous’ forest;
-
Fragments of post-logging regrowth forest, (almost always with a visually observable broken canopy);
-
Scrub (having a sparse cover of small trees and bushes);
-
Cleared land.
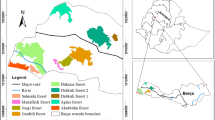
Because a limited area was covered by all three sets of photographs, two polygons of land were selected for detailed study. The first area (area ‘A’) is situated on the western side of the highlands and covers 1230 ha with an elevation range of 700–1435 masl. The second area (area ‘B’) is situated on the eastern side of the highlands and covers 1390 ha with an elevation range of 640–820 masl.
Security concerns precluded inventory of forest north of Mt Magolo and adjacent to Mt Bolomolot. Consequently, inventory strips were located on area A and area B (Fig. 2), i.e. high-elevation land (~1000 to 1500 masl) on the western side of the area and low-elevation land (~500 to 1000 masl) on the eastern side of the area, respectively.
The overall inventory was 51.80 ha of the 643.3 ha (i.e. an 8 % sample) of trees in contiguous forest which was all situated in area A; and 15 ha of the 351.6 ha of fragmented forest (i.e. a 4.3 % sample) in areas A and B. Twenty-five fragments were sampled. The fragments ranged in area from 0.5 to 18.3 ha and had been separated from contiguous forest since 1995. Hence, these fragments have been subject to influences that may potentially degrade their structure and species richness for at least 15 years.
In both contiguous forest and fragments, the samples consisted of sample-based incidence and abundance data, where the presence of each species was recorded for each sampling unit (plot). The plots were 0.2 ha in area and were measured as sequential continuous strips, each plot being 20 m wide and 100 m long. The inventory was undertaken in 2009 and 2010 according to Department of Environment and Natural Resources (DENR) timber inventory guidelines. Transects were run from convenient baselines (e.g. roads), with inventory data collected in contiguous plots, 100 m long and 20 m wide, along each strip.
For each plot, the species of each tree with a diameter at breast height (DBH) of 15 cm or more was recorded. Subplots (5 × 5 m), situated at the front left hand corner of each plot were used to inventory sapling regeneration with a DBH range of 6–14 cm. Within these subplots, smaller 2 × 2 m subplots were used to count and identify seedlings with a maximum DBH of 5 cm.
If species identification in the field proved difficult, leaves were collected with the assistance of a tree climber, dried and pressed and subsequently identified by a local or a Visayas State University (VSU) botanist. Based on their advice, trees were classified as ‘rare’, ‘endangered’, ‘vulnerable’, ‘other threatened species’ or ‘other wildlife species’ as per the IUCN Red List of Threatened Species (2012) and DENR Departmental Administrative Order (DAO) 2007-01. Tree species were classified into ecological species groups (i.e. ‘guilds’) as defined by Swaine and Whitmore (1988), i.e. ‘pioneer’ species which regenerate after clearing, ‘secondary’ and ‘climax’ species.
Inventory data were collated from field sheets, input into a Microsoft Access database, a Geographic Information System (ArcGIS®) and the EstimateS statistical package (Colwell 2013) for analysis. Plot data included plot coordinates and confirmation that plot location was either in a contiguous stand or a fragment of remnant forest (nominally <20 ha in area); elevation above sea level and terrain slope, in five classes (slope <8, 9–18, 19–30, 30–50 and >50 %). For the 25 forest fragments, we calculated the steepness of the terrain on which fragments were situated. We also calculated the elevation range at which species occurred.
Species richness and diversity
We present a brief explanation of the background to calculating metrics of diversity in Appendix 1. For contiguous forest and forest fragments, we used EstimateS software to calculate tree species richness accumulation curves. To assist comparison of the smaller sample inventoried in fragmented forest with the larger sample inventoried in contiguous forest, we extrapolated the species accumulation curves of the fragmented forest to the maximum recommended by Colwell et al. 2012; Gotelli and Chao (2013), i.e. double the size of the original sample. EstimateS software was also used to calculate the three key Hill numbers, i.e. 0D (species richness), 1D (the exponential value of Shannon’s diversity index) and 2D (the inverse of Simpson’s diversity index) and the number of singleton, doubleton and unique species. To estimate compositional similarity, i.e. tree species turnover for contiguous and fragmented forest, we calculated alpha, gamma and hence beta diversity using the equations presented in Appendix 1 (see Jost et al. 2011). We also used EstimateS software to calculate the Chao-Jaccard index as an estimate of the biotic similarity between contiguous and fragmented forest (Chao et al. 2005).
Results: the pattern of deforestation and regrowth and characteristics of contiguous forest and fragments
The 1995 aerial photographs showed that the logging operation had been very severe across area A and B. An extensive network of logging roads and log dumps indicated that virtually no area of forest had been left unlogged. Extensive and uniformly similar forest regrowth was also visually evident. Given its uniform appearance, the regrowth would appear to be the same age, i.e. ~30 years old. Mapping forest cover using the 1995, 2008 and 2010 aerial photographs revealed two clear patterns of subsequent land-cover change. In remote areas not served by a local village (e.g. area A), forest that had been heavily logged in the 1980s recovered to form contiguous tracts of closed-canopy forest. In more populated areas, recent clearing for agriculture has resulted in fragmentation and in some places, the reduction in forest cover over time is severe, as high as 85 % in area B in 2010 (Table 1; Fig. 3a, b, c). In area B, clearing was well advanced by 1995 and has accelerated in recent years. In particular, the area of scrub decreased dramatically, from 18.5 to 6.4 % between 2008 and 2010, indicating increased rates of land clearing.
Species composition
Overall, 150 native tree species were identified (Appendix 2 in Table 4) with 121 species found in contiguous forest and 102 species in fragments. Fifty species were found only in contiguous forest and 22 were found only in fragments. Two species Lithocarpus llanosii, and Cinnamomum mercadoi constituted 30.4 % of the total of 8959 trees recorded. Many species were recorded in very low numbers, 53 species being recorded at a frequency of 0.05 % or less.
The average stem density of native trees greater than 15 cm DBH in the contiguous forest was 153 trees per ha, falling to 68 trees per ha in forest fragments. Similarly, the mean DBH of trees in the contiguous forest was higher at 38.0 cm compared to 34.5 cm in the fragments. The mean elevation of plots in the contiguous stands was 1230 masl while the mean elevation of plots in forest fragments was 810 masl, reflecting the propensity of the B’laan to clear lower elevation forest, (often closer to towns and less steep) in preference to more remote higher elevations. Similarly, fragments are typically situated on steep land, 67 % of inventory plots being situated on slopes between 30 and 50 %.
The number of species found in the understorey seedling bank declined from 48 in contiguous forest to 21 in fragmented forest. Seedling stocking also declined from ~5410 to 2560 seedlings per ha, respectively. Species found as saplings declined from 45 to 21 and stocking declined from 510 to 220 stems per ha in contiguous forest and fragments, respectively. For both contiguous and fragmented forest the proportion of seedlings of pioneer species was high, being 54 and 81 % respectively. Similarly, the proportion of saplings of pioneer species in contiguous and fragmented forest respectively was also high, being 58 and 67 % respectively. In short, the fragments have lower seedling and sapling densities than the contiguous forests and this regeneration is dominated by pioneer species.
Presence of endangered tree species
Of the nine species described as ‘critically endangered’ or ‘endangered’ under the IUCN classification or DAO 2007-01, 44 specimens were recorded in 12 fragments. Twenty-six specimens of Shorea contorta and a single specimen of Shorea Guiso (both listed as critically endangered by the IUCN) were exclusively recorded in fragments.
Twenty-four species listed as being threatened by IUCN or DAO 2007-01 were recorded, with 384 individuals of Toona calantas found mostly in high-elevation contiguous forest. This species is listed in the DAO as being critically endangered. In addition 390 specimens of Palaquium luzionense, listed in the DAO as being vulnerable were also found, mostly in high-elevation contiguous forest.
Species richness, diversity and biotic similarity
Fragmented forest has lower tree abundance than contiguous forest, but for equivalent numbers of trees inventoried, higher species accumulation (Table 2; Fig. 4a, b). Fragmented forest also showed higher numbers of singleton, doubleton and unique species (Table 2).
a Relationship of species richness (‘S’) with increasing number of plots sampled for contiguous forest (259 plots) and fragmented forest (75 measured plots, data extrapolated by a factor of two, i.e. to 150 plots). b Relationship of species richness (‘S’) with increasing number of trees sampled for contiguous forest (7941 trees) and fragmented forest (1018 measured trees, extrapolated by a factor or two, i.e. to 2036 trees)
The exponential of Shannon’s diversity index (1D) and the inverse of Simpson’s diversity index (2D) are sensitive to the number of samples. When data from contiguous forest were scaled back to the 75 plots inventoried in forest fragments, both the exponential of Shannon’s diversity index and the inverse of Simpson’s diversity index are higher for fragmented forest than for contiguous forest (see Table 3). This infers that fragmented forest has both more typical and common species than contiguous forest.
Βeta (β) diversity of contiguous and fragmented forest
For 259 plots in contiguous forest and 75 plots in fragmented forest, multiplicative beta diversity for species richness (q = 0) was 10.78 and 15.59 for contiguous and fragmented forest, respectively. Similarly, multiplicative beta diversity calculated for the Shannon entropy index (q = 1) was 3.10 and 9.30 for contiguous and fragmented forest respectively. Species turnover is therefore higher in fragments compared to contiguous forest for both incidence- and abundance-based measures.
Biotic similarity
The mean of the pairwise Chao-Jaccard index, for 259 plots in contiguous forest and 75 plots in fragmented forest was 0.45 and 0.12, respectively, indicating higher similarity of species composition between plots in contiguous forest than in fragments.
Discussion and conclusion: implications for a hypothetical conservation program
Our results confirm that for trees, both fragmented and continuous post-logging forests in the southern Philippines retain a high conservation value. Our results may not be applicable to other groups of organisms, however for tree species, the fragmented forests are surprisingly diverse, even though smaller fragments are effectively ‘all-edge’. Consequently, our findings do not corroborate results from other studies (e.g. see Pimm 1998; Streubig et al. 2008) that small fragments tend to be depauperate in species, particularly rare species. At Tampakan, fragments have stem density which is lower than contiguous forest but higher species accumulation with comparable numbers of stems inventoried. This result may be because (as also noted by Cayuela et al. (2006), the time since fragmentation has been too short for tree populations to reach new equilibrium levels. Other studies have shown that over time, fragments become reduced in size and forest structure is irreversibly changed (e.g. see Laurance et al. 1997 and Lopez et al. 2009, in Brazil; Lele et al. 2008, in India), particularly if disturbance is on-going (see Botzag et al. 2015); although Gardner et al. (2009) noted that the lack of a wide base of empirical evidence makes generalising difficult. Our evidence suggests that at Tampaken, fragments present a time-window of opportunity for conservation that may disappear as edge effects become more apparent. This is supported by the lower levels of sapling and seedling regeneration within the fragments.
Considering the small geographic range of the highlands, and the severity of the initial logging—more akin to clear cutting than selective logging—the high number of species found in both the contiguous forest and the fragments is surprising. Using terminology from Ashton et al. (2001) the Tampakan forests have undergone ‘acute’ degradation from severe industrial logging followed, in fragments, by ‘chronic’ continuing degradation from low-level utilisation. In Borneo, Cannon et al. (1998) found that a conservative logging regime reduced stem density, but species richness for an equivalent number of trees was similar to unlogged forest. Unfortunately, we were unable to compare the diversity of logged with unlogged forest at Tampaken. However, the high diversity of both the contiguous forest and the fragments suggest that these forests are resilient to an acute disturbance event. We suggest that the condition of forest at Tampakan may be typical of many Philippine and south-east Asian forests that were heavily logged in the 1970s and 1980s (see Edwards et al. 2010). In the absence of further acute degradation, the conservation value of these forests is high.
The high beta diversity and the low biotic similarity of fragments compared to contiguous forest indicate a high species turnover between and even within fragments. This suggests that regrowth post-logging fragments have a high ecological resilience in terms of their ability to retain species variation across the landscape. From a conservation perspective, individual fragments are not a nested subset of the overall forest.
Comparing the two assemblages is confounded by differences in elevation. However, the evidence from the inventory and the aerial photographs is that once the entire primary forest was severely logged, widespread regrowth occurred and the main source of further degradation has come from clearing for swidden agriculture and low levels of utilisation (e.g. building temporary houses). Unfortunately, security issues may preclude conservation activities in the high elevation forests. Hence, conservation or restoration measures may be best directed at the forest fragments which occur at lower elevations. Fragments or contiguous forest may be useful as ‘springboards’ for species to recolonise adjacent land. In a similar situation in Sri Lanka, Ashton et al. (2001), recommended enrichment planting and grass control to link fragments into a mosaic of regenerating forest. Known locally as ‘assisted natural regeneration’, widely spaced sun-tolerant colonising species achieve dominance over grass and allow remnant canopy to expand (FAO 2011). For those species which exist in very low numbers, an alternative method recommended by DENR (2010) may be to use individual trees as a source of germplasm, either as seedlings or wildlings. In the Philippines these parent trees are known as ‘mother trees’. Although many dipterocarps have an irregular seeding habit, the longevity of wildlings for several years after germination offers an alternative to seed collection. As is usual with forest conservation programs in developing countries, the cooperation of the B’laan is likely to be contingent on the simultaneous development of alternative or additional livelihood opportunities.
At Tampakan, overall tree biodiversity of secondary contiguous forest and fragments is still high. Unfortunately, renewed clearing activities may eliminate the forest in the foreseeable future, with poor prospects for seedling and sapling regeneration. The implication for conservation measures is that the timeframe for action is short to protect the forests of this region from large-scale clearance.
References
Appiah M (2013) Tree population inventory, diversity and degradation analysis of a tropical dry deciduous forest in Afram plains, Ghana. For Ecol Manag 295:145–154
Arroyo-Rodríguez V, Melo FPL, Martínez-Ramos M, Bongers F, Chazdon RL, Meave JA, Norden N, Santos B, Leal IR, Tabarelli M (2015) Multiple successional pathways in human-modified tropical landscapes: new insights from forest succession, forest fragmentation and landscape ecology research. Biol Rev. doi:10.1111/brv.12231
Arroyo-Rodŕiguez V, Rös M, Escobar F, Melo FPL, Santos BA, Tabarelli M, Chazdon R (2013) Plant β-diversity in fragmented rain forests: testing floristic homogenisation and differentiation hypotheses. J Ecol. doi:10.1111/1365.2475.12153
Ashton MS, Gunatilleke CVS, Singakumara BMP, Gunatilleke IAUN (2001) Restoration pathways for rain forest in southwest Sri Lanka: a review of concepts and models. For Ecol Manag 154:409–430
Bierregaard RO, Lovejoy TE, Kapos V, dos Santos AA, Hutchings RW (1992) The biological dynamics of tropical rainforest fragments. Bioscience 42(11):859–866
Botzag A, Fischer L, Farwig N (2015) Regeneration potential in South African forest fragments: extinction debt paid off or hampered by contemporary matrix modification? Plant Ecol 216:535–551
Bruna EM, Vascondelos HL, Heredia S (2005) The effect of habitat fragmentation on communities of mutualists: Amazonian ants and their host plants. Biol Conserv 124:209–216
Bunyan M, Shibu J, Fletcher R (2012) Edge effects in small forest fragments: why more is better. Am J Plant Sci 3(7):869–878
Cannon CH, Peart DR, Leighton M (1998) Tree species diversity in commercially logged Bornean rainforest. Science 281:1366–1388
Cayuela L, Golicher DJ, Benayas JMR, González-Espinosa M, Ramírez-Marcial N (2006) Fragmentation, disturbance and tree diversity conservation in tropical montane forests. J Appl Ecol 43(6):1172–1181
Chao A, Chazdon RL, Colwell RK, Shen TJ (2005) A new statistical approach for assessing similarity of species composition with incidence and abundance data. Ecol Lett 8:148–159
Chao A, Chiu CH, Jost L (2010) Phylogenetic diversity metrics based on Hill numbers. Philos Trans R Soc 365:3599–3609
Chiu C-H, Chao A (2014) Distance-based functional diversity measures and their decomposition: a framework based on hill numbers. PLoS One 9(7):e100014. doi:10.1371/journal.pone.0100014
Colwell RK (2013) EstimateS: statistical estimation of species richness and shared species from samples. Version 9. Users guide and application. http://purl.oclc.org/estimates. Accessed 28 March 2015
Colwell RK, Chao A, Gotelli NJ, Lin S, Mao CX, Chazdon RL, Longino JT (2012) Models and estimators linking individual-based and sample-based rarefaction, extrapolation and comparison of assemblages. J Plant Ecol 5(1):3–21
DENR (2010) Mass propagation and nursery management of dipterocarps. DENR recommends: volume 6 http://erdb.denr.gov.ph/publications/denr/denr_v6.pdf. Accessed 21 Jan 2013
Echeverría C, Newton AC, Lara A, Benayas JMR, Coomes DA (2007) Impacts of forest fragmentation on species composition and forest structure in the temperate landscape of southern Chile. Glob Ecol Biogeogr. doi:10.1111/j.1466-8238.2007.00311.x1
Edwards DP, Larsen TH, Docherty TDS, Ansell FA, Hsu WW, Derhé MA, Hamer KC, Wilcove DS (2010) Degraded lands worth protecting: the biological importance of Southeast Asia’s repeatedly logged forests. Proc R Soc B 2011:278. doi:10.1098/rspb.2010.1062
FAO (2011) Assisted natural regeneration of forests. http://www.fao.org/forestry/anr/en/. Accessed 21 Jan 2013
Fernando E, Suh M, Lee J, Lee D (2008) Forest formations of the Philippines. ASEAN-Korea Environmental Cooperation Unit (AKECU), Seoul, Korea
Gardner TA, Barlow J, Chazdon R, Ewers RM, Harvey CA, Peres CA, Sodhi NS (2009) Prospects for tropical forest biodiversity in a human-modified world. Ecol Lett 12:561–582. doi:10.1111/j.1461-0248.2009.01294
Gibson L, Lee TM, Koh LP, Brook BW, Gardner TA, Barlow J, Peres CA, Bradshaw CJA, Laurance WF, Lovejoy TE, Sodhi NS (2011) Primary forests are irreplaceable for sustaining forest biodiversity. Nature 478:378–381. doi:10.1093/jpe/rtr044
Gotelli NJ, Chao A (2013) Measuring and estimating species richness. Species diversity and biotic similarity from sampling data. In: Levin SA (ed) Encyclopedia of biodiversity, vol 5, 2nd edn. Academic Press, Waltham, pp 195–211
Hill MO (1973) Diversity and evenness: a unifying notation and its consequences. Ecology 54:427–431
IUCN (International Union for Conservation of Nature and Natural Resource) (2012). http://www.iucnredlist.org/. Accessed 15 June 2012
Jost L (2006) Entropy and diversity. Oikos 113(2):363–375
Jost L (2007) Partitioning diversity into independent alpha and beta components. Ecology 88(10):2427–2439
Jost L, DeVries P, Walla T, Greeney H, Chao A, Ricotta C (2010) Partitioning diversity for conservation analyses. Divers Distrib 16:65–76
Jost L, Chao A, Chazdon RL (2011) Chapter 6, compositional similarity and β (beta) diversity. In: Magurran AE, McGill BJ (eds) Biological diversity: frontiers in measurement and assessment. Oxford University Press, Oxford, pp 66–84
Kräuchi NA, Brang P, Schönenberger W (2000) Forests of mountainous regions: gaps in knowledge and research needs. For Ecol Manag 132:73–82
Kupfer JA, Franklin SB (2009) Linking spatial pattern and ecological responses in human-modified landscapes: the effects of deforestation and forest fragmentation on biodiversity. Geogr Compass 3(4):1331–1355. doi:10.1111/j.1749-198.2009.00245.x
Laurance WF, Laurance SG, Ferreira LV, Rankin de Merona JM, Gascon C, Lovejoy TE (1997) Biomass collapse in Amazonian forest fragments. Science 278:1117–1118
Laurance WF, Lovejoy TE, Vascolcelos HL, Bruna EM, Didham RK, Stouffer PC, Gascon C, Bierregard RO, Laurance SG, Sampaio E (2002) Ecosystem decay of forest fragments: a 22 year investigation. Conserv Biol 16(3):605–618
Leemans R (2005) The millennium ecosystem assessment: securing interactions between ecosystems, ecosystem services and human well-being. In: Chapter 3, facing global environmental change: environmental, human, energy, food, health and water security concepts. Springer, Berlin, pp 53–61
Lele N, Joshi PK, Agrawal SP (2008) Assessing forest fragmentation in northeastern region (NER) of India using landscape matrices. Ecol Ind 8:657–663
Lôbo D, Leão T, Melo FPL, Santos AMM, Tabarelli M (2011) Forest fragmentation drives Atlantic forest of northeastern Brazil to homogenisation. Divers Distrib 17:287–296
Lopez VA, Girão LC, Santos BA, Peres CA, Rabarelli M (2009) Long-term erosion of tree reproductive trait diversity in edge-dominated Atlantic forest fragments. Biol Conserv 142:1154–1165
MacArthur RH (1965) patterns of species diversity. Biol Rev 40:510–533. doi:10.1111/j.1469-185x.1965.rb00815.x
Magurran AE, McGill BJ (2011) Biological diversity: frontiers in measurement and assessment. Oxford University Press, Oxford. 345
Marcon E, Scotti I, Hérault B, Rossi V, Lang G (2014) Generalisation of the partitioning of Shannon diversity. PLoS One 9(3):1–8
Maurer BA, McGill BJ (2011) Measurement of species diversity. Chapter 5, compositional similarity and β (beta) diversity. In: Magurran AE, McGill BJ (eds) Biological diversity: frontiers in measurement and assessment. Oxford University Press, Oxford, pp 55–66
McNamara S, Erskine PD, Lamb D, Chantalangsy L, Boyle S (2012) Primary tree species diversity in secondary fallow forests of Laos. For Ecol Manag 281:93–99
Pimm SL (1998) Ecology: the forest fragment classic. Nature 393:23–24. doi:10.1038/29892
Schleuning M, Farwig N, Peters MK, Bergsdorf T, Bleher B, Brandl R, Dalitz H, Fischer G, Freund W, Gikungu MW, Hagen M, Garcia FH, Kagezi GH, Kaib M, Kraemer M, Lung T, Naumann CM, Schaab G, Templin T, Uster D, Wägele W, Böhning-Gaese K (2011) Forest fragmentation and selective logging have inconsistent effects on multiple animal-mediated ecosystem processes in a tropical forest. PLoS One 6(11):e27785
Streubig MJ, Kingston T, Zubaid A, Mohd-Adnan A (2008) Conservation value of forest fragments to Palaeotropical bats. Biol Conserv 141:2112–2126
Swaine MD, Whitmore TC (1988) On the definition of ecological species groups in tropical rain forests. Vegetation 75:81–86
Turner IM (1996) Species loss in fragments of tropical rain forest: a review of evidence. J Appl Ecol 33:200–209
Villard MA, Metzger JP (2014) Beyond the fragmentation debate: a conceptual model to predict when habitat configuration really matters. J Appl Ecol 51:309–318. doi:10.1111/1365-2664.12190
Warburton T (2009) The Philippine owl conservation programme: why is it needed? Ardea 97(4):429–438
Acknowledgments
The authors would like to acknowledge our use of EstimateS software provided as a free internet download by Rober K. Colwell of the University of Connecticut. The assistance of Mr. Nelson Quilario in identifying tree species is also gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Peter Ashton.
Appendices
Appendix 1: Background to estimating metrics of diversity
Metrics of species diversity have been extensively discussed in the recent literature, (e.g. see Jost 2006, 2007; Marcon et al. 2014; Chao et al. 2010; Magurran and McGill (2011); Chiu and Chao 2014). In particular, Jost et al. (2011) and Gotelli and Chao (2013) provided a comprehensive description of how diversity metrics are calculated. We present a brief description of the key metrics used in this research.
Species richness
Species richness is the total number of different species in an assemblage and is thus a key ecological attribute of an assemblage. However, unless the entire assemblage is inventoried, observed species richness is always an underestimate of true species richness, because increasing the number of samples increases the likelihood of adding new rare species. Species richness does not take into account levels of species abundance; thus rare and common species are not distinguished.
Within an assemblage, the observed numbers of species depend upon the area sampled and the number of individuals sampled. For sample-based data, species richness is often depicted in a species accumulation curve which shows the cumulative number of individuals or sampling units on the x axis and cumulative species richness on the y axis). Typical species accumulation curves rise rapidly at first, as common species are encountered and then rise more slowly as rare species are eventually inventoried, the curve eventually becoming asymptotic. The curves are often calculated using rarefaction, a resampling and interpolation technique, in which individuals or sampling units are selected at random from the entire sampling assemblage until all individuals and species have all been accumulated. The resampling process is repeated many times and mean species richness and other diversity indices are calculated for each level of accumulation. This results in a smoothed species accumulation curve which may be extrapolated. Extrapolation should not be extended more than double the reference sample’s size because variance may become very large (Colwell et al. 2012; Gotelli and Chao 2013). Alternatively, species accumulation curves of two (or more) datasets of different size, may be compared by scaling back the cumulative number of individuals in the larger dataset to the same number of individuals or sampling units of the smaller dataset.
Species diversity indices
Species diversity is a function of species richness and the relative abundance (evenness) of species in an assemblage (Maurer and McGill 2011). Two popular diversity indices are Shannon’s entropy (H) and the Gini-Simpson (D) diversity indices, calculated as:
where ‘s’ is the number of species in the assemblage and ‘p i ’ is the population frequency of the ith species.
Shannon entropy quantifies the uncertainty of the species identity of a randomly chosen individual in the assemblage and the Gini–Simpson index measures the probability that two randomly chosen individuals (selected with replacement) belong to two different species. In their ‘raw’ form, these diversity indices calculate entropy i.e. the uncertainty of the species identity of a sample, not diversity itself and they are non-linear with respect to increasing diversity (Jost 2006; Chao et al. 2010). In certain circumstances, the indices can be misleading as they do not provide a measure based on numbers of species (e.g. see Chao et al. 2010). Hence, following the work of MacArthur (1965) and Hill (1973) these raw diversity metrics are often converted into ‘Hill’ numbers, which provide a measure of the equivalent number of equally abundant species.
Hill numbers (qD) are calculated according to their order, ‘q’ which determines their sensitivity to species relative abundance. When q = 0, 0D is equal to species richness, when q = 1, the Hill number 1D is the exponential form of Shannon entropy which weights species according to their frequency and can be approximately expressed as the number of typical species in the assemblage. When q = 2, the Hill number 2D is the inverse of the Gini-Simpson entropy and is heavily weighted towards very common or dominant species in the assemblage.
Decomposition into alpha (α), beta (β) and gamma (γ) diversity components
Spatially, tropical forests typically have high rates of species turnover, i.e. compositional similarity is low (Arroyo-Rodriguez et al. 2013). Hence, ecologists often wish to compare the diversity of a sub-assemblage to the diversity of the overall assemblage. If weighted appropriately, sample data may be pooled and diversity may be calculated as an overall assemblage diversity (gamma, γ), within or sub-assemblage diversity (alpha, α), and between-group (beta, β) diversity.
Recent debate concerning the most appropriate way to partition gamma diversity into alpha and beta diversity has resulted in consensus that when considering diversity in terms of Hill numbers, the relationship between gamma, alpha and beta diversity is multiplicative, rather than additive i.e., gamma = beta × alpha or Dγ = Dβ × Dα (see Jost 2007; Jost et al. 2010; Marcon et al. 2014). Beta diversity is then calculated as the ratio of gamma to mean alpha diversity and may be interpreted as the effective number of distinct communities in an assemblage (Jost 2007). In general, high beta diversity implies that alpha is much less than gamma diversity and that species turnover between the sub-assemblage and the assemblage is high.
Alpha, gamma and hence beta diversity for q = 0 (species richness) and q = 1 (exponential form of Shannon entropy) may be calculated using equations presented by Jost et al. (2011), i.e.
Biotic similarity
Whereas beta diversity describes the turnover of species from a sub-assemblage to the overall assemblage, indices of biotic similarity enable comparison of species shared by two assemblages. The comparison may be between two different assemblages or between a sub-assemblage and the combined assemblage. For this purpose the Jaccard index compares the number of shared species in an assemblage (i.e. a sample) to the total number of species in the combined assemblages. The Chao-Jaccard index considers the probability that two individuals (one each drawn from both assemblages) belong to any shared species, not necessarily the same shared species. The index has a maximum value of one when all species are shared and a minimum value of zero when no species are shared (Jost et al. 2011; Gotelli and Chao 2013).
Appendix 2
See Table 4.
Rights and permissions
About this article
Cite this article
Baynes, J., Herbohn, J., Chazdon, R.L. et al. Effects of fragmentation and landscape variation on tree diversity in post-logging regrowth forests of the Southern Philippines. Biodivers Conserv 25, 923–941 (2016). https://doi.org/10.1007/s10531-016-1098-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10531-016-1098-6