Abstract
Ambrosia beetles (Coleoptera: Curculionidae: Scolytinae) are among the most successful invaders of trees on a global scale. Exotic species can establish large populations within forested habitats and disperse into tree nurseries and orchards with the potential for substantial economic losses. Our objective was to assess the seasonal dominance of exotic Scolytinae compared to native species by characterizing their flight phenology, abundance and species diversity. Weekly sampling using ethanol-baited traps was conducted within deciduous and coniferous woodlots in Ohio, USA from March/April to September/October in 2014, 2015, 2016, and 2019. Over the course of the study, 16 native and 11 exotic species of Scolytinae were identified. No difference was detected in the number of exotic Scolytinae species or their abundance, Shannon’s index (H), and evenness (Eh) between the coniferous vs. deciduous woodlots. On average, initial flight occurred at 188 degree days (DD) for exotic species compared to 273 DD for native species. Seasonal flight duration of exotic species averaged 49 days compared to 10 days for native species. Of the 145,882 total Scolytinae captured over the four years, only 622 were native beetles. Captures of exotic Scolytinae were 341-times greater than native species across the four trapping seasons, including captures of the exotic ambrosia beetle Xylosandrus germanus being 450-times greater than the most common native species Xyloborinus politus. These results provide insight into the invasion success of ambrosia beetles and will aid in predicting and monitoring key species.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Bark and ambrosia beetles (Coleoptera: Curculionidae: Scolytinae, Platypodinae) are wood-boring beetles that excavate brood galleries within woody plant tissue. Within our study, we aimed to capture and quantify ambrosia beetles, which are distinguished by xylomycetophagy, or fungus-farming, within these galleries; bark beetles are defined from ambrosia beetles because they often ingest wood as well as fungus (Kirkendall et al. 2015). We’ve quantified both ambrosia and bark beetle captures from our ethanol-baited traps and therefore use the term Scolytinae to encompass both within this paper. Scolytinae in the tribe Xyleborini are among the most successful invaders of novel habitats due their ability to infest a large variety of host tree species, elusive nature, haplodiploidy, and mutualism with nutritional fungal symbionts (Weber and McPherson 1983a; Atkinson et al. 1990; Kirkendall et al. 1993; Normark et al. 1999; Oliver and Mannion 2001; Brockerhoff et al. 2010; Dole et al. 2010; Ranger et al. 2015; Werle et al. 2015; Hulcr and Stelinski 2017; Gugliuzzo et al. 2021). Many ambrosia beetle introductions are through packing materials, ports of entry, or imported lumber (Haack 2001, 2006; Rassati et al. 2016a; Olenici et al. 2022). In addition to being forestry pests (Gossner et al. 2019; Økland et al. 2011), exotic Xyleborini beetles disperse into nurseries and orchards from adjacent woodlots and infest horticultural trees (Werle et al. 2015; Ranger et al. 2016; Agnello et al. 2017). Adult females bore tunnels and brood chambers in trees and cultivate their nutritional fungal symbiont that serves as a food source for the brood (Weber and McPherson 1983b, 1984; Werle et al. 2015; Hulcr and Stelinski 2017).
A select few exotic species of ambrosia beetles are problematic as tree pests because attacks can result in high economic loss in ornamental nurseries and orchards due to tree death, branch dieback, and negative impacts on growth and aesthetics (Ranger et al. 2016; Brockerhoff and Liebhold 2017; Gugliuzzo et al. 2020, 2021). Xylosandrus germanus (Blandford) has been established in the U.S. for decades after it was detected in the 1930’s in New York state (Felt 1932). Xylosandrus germanus has shown rapid population increases in the eastern United States (Rassati et al. 2016b) and Europe over just a few years (Henin and Versteirt 2004; Galko et al. 2018; Gossner et al. 2019; Olenici et al. 2022; Gugliuzzo et al. 2021). Xylosandrus crassiusculus (Motchulsky), which is also well-established in the eastern U.S., was found on peach trees in South Carolina in the 1970’s (Anderson 1974) and is also currently established in Europe and other parts of the world (CABI 2021). Anisandrus maiche (Stark) is becoming an increasing concern in the U.S. since its detection in Ohio, Pennsylvania, and West Virginia as early as 2005 (Rabaglia et al. 2009). These three species exhibit a broad range of host trees with additional host species being updated as these pests expand their distribution and host range (Weber and McPherson 1983a, b; Reding et al. 2015; Ranger et al. 2016, 2021; CABI 2021; Ruzzier et al. 2021; Gugliuzzo et al. 2021).
The ability of exotic ambrosia beetles and their fungal mutualists to colonize a wide range of genera in the absence of co-evolutionary history likely plays an important role in their invasion success. Different ambrosia beetles may be attracted to or prefer specific tree species (Gossner et al. 2019; Rassati et al. 2016a), as well as experience limitations with how well their symbiotic fungi grow within different host species (Castrillo et al. 2012). Furthermore, flight patterns and establishment of prominent Scolytinae pests may be influenced by beetles’ attraction to ethanol emitted by stressed trees, particularly trees that are flood stressed (Ranger et al. 2015) or freeze stressed (La Spina et al. 2013); attacks are more prevalent on stressed trees emitting ethanol due to higher rates of ethanol accumulation in tree tissues (Ranger et al. 2015; Ruzzier et al. 2021). A monoterpene, α-pinene, common in pine trees, has been shown to attract bark beetle species, and to attract some species of ambrosia beetle species and repel others when combined with ethanol (Miller and Rabaglia 2009). The chemical profile of trees can be complex and may vary between different stands of trees (Olenici et al. 2022), ultimately altering which Scolytinae species are better able to thrive in coniferous and deciduous woodlots. Although native species of ambrosia beetles are found throughout North America, they are more geographically isolated by trends in forest vegetation compared to exotic species. Ambrosia beetle species are less selective in host tree preference and climate limitations than their bark beetle counterparts (Rassati et al. 2016b). Although still in greater abundance in broadleaf forests, X. germanus thrived in pine forests in association with high management intensity (Gossner et al. 2019). Trap captures of X. germanus also occurred in conifer stands in Romania but were higher in broadleaf and mixed stands with a majority (> 50%) of beech trees (Olenici et al. 2022). Thus, the ability of exotic ambrosia beetles and their fungal symbionts to utilize a wide range of host tree species could contribute to a few exotic species increasing to much greater populations than native species (Rassati et al. 2016b).
The seasonal flight phenology of exotic ambrosia beetles compared to native species may also contribute to their invasion success. The influence of temperature on initiating earlier spring flight after overwintering and longer duration of flight activity of exotic ambrosia beetles compared to native species could allow exotic species to be prime invaders, either by earlier access to more vulnerable stressed trees, increasing range and rates of infestation, or potential for additional generations. Exotic species such as Xylosandrus saxesenii (Ratzeburg) and X. germanus have been shown to have flight in early April and as late as October in Missouri (Reed and Muzika 2010) and other exotic species, such as Xylosandrus compactus (Eichhoff), have been shown to have multiple peaks of flight activity occurring as early as April and as late as September in Italy (Gugliuzzo et al. 2019, 2020), indicating exotic ambrosia beetle species could have earlier and longer flight phenology. Seasonal flight patterns may not be fully discernable if trapping only focuses on peak flight. Many studies have limited the duration of trapping to shorter periods aimed at capturing peak flight rather than understanding season-long flight patterns (Maeto et al. 1999; Hulcr et al. 2008; Sittichaya et al. 2012). However, studies that consisted of seasonal trapping show a typical peak population in early spring and potential for a second, smaller peak in late summer (Hudson and Mizell 1999; Oliver and Mannion 2001; Reding et al. 2010; Werle et al. 2012, 2015).
To better understand why exotic ambrosia beetle species are prime invaders and able to obtain higher populations than native species, we monitored weekly captures of ambrosia beetles in coniferous and deciduous stands to evaluate seasonal flight patterns. We hypothesized exotic ambrosia beetles would dominate in both coniferous and deciduous habitats; that exotic ambrosia beetles would require fewer degree days (DD) until first trap detection and exhibit a longer duration of flight activity compared to native species. Ultimately, by better understanding when pest species first begin flying and are at their peak flight, management of these species will become more attainable.
Materials and methods
Area of study and experimental design
Experiments were set up over 4 years (2014, 2015, 2016, and 2019) at six locations in Wayne County, Ohio (Table 1). Three locations were coniferous woodlots comprised of Pinus spp., three were deciduous woodlots comprised of Acer spp., Quercus spp. and other hardwoods (Table 1). All woodlots were well-established with trees planted 30–50 years ago (Fig. 1A–C). Woodlots were not maintained with fire or pesticides, but had disturbance from collection of firewood and selective logging every 5–10 years (Stuart Courtney, personal communication).
Traps and monitoring
To trap ambrosia beetles, we used bottle traps baited with ethanol lures as described by Ranger et al. (2010). Traps were designed from a 1 L plastic bottle hung upside down with two windows cut out (11 cm × 7 cm) and the ethanol lure suspended within the open bottle with a twist tie (Ranger et al. 2010; Reding et al. 2010). A 0.5 L bottle with ~ 28 mL killing solution (1:1 propylene glycol: tap water; Sierra Antifreeze/Coolant; Old World Industries, Inc., Northbrook, Illinois) was connected to the 1 L bottle via a Tornado Tube® (Steve Spangler Science, Englewood, Colorado). Traps were suspended vertically 0.6 m above ground level by securing the inverted 1 L bottle to a metal rod. We used low-release lures in 2014 and 2019 (10 ml of 95% ethanol released at 65 mg/d at 30 °C; AgBio, Westminster, Colorado, USA) and made low release lures in other years by adding 8.5 mm of 95% ethanol and heat sealing. Lures were replaced if damaged, leaking or the ethanol was depleted.
Within each woodlot, four traps were deployed (n = 24 each year) in March/April and checked weekly through September/November (Table 1). Traps were placed 10 m apart in a diamond configuration with each trap placed at a cardinal direction to have a consistent pattern across all location (Fig. 1D). The collected bottles with beetles were brought back to the laboratory where beetles were rinsed and stored in 70% ethanol until they were sorted and identified to species (Gomez et al. 2018). We focused on damaging pests and therefore excluded Hypothenemus spp. from our analyses (Monterrosa et al. 2022).
Data analyses
Weather data was downloaded from “Daymet: Daily Surface Weather Data on a 1-km Grid for North America, Version 4 R1” for each year using the longitude and latitude for each location (Thornton et al. 2022). We calculated cumulative degree days (DD) starting on January 1 by adding the maximum and minimum temperature each day, dividing the sum by two and subtracting the base temperature of 10 °C. Longitude and latitude coordinates were determined using Google Earth Pro (Version 7.3.6.934; 2022 Google LLC.).
Mean beetle capture was the average of the four traps per site. Total trap captures were averaged for each site within each year. Number of captured beetles for each species was used to calculate a diversity index (H, Shannon’s Index) for each site within each year (n = 24). Shannon’s Index (H) for diversity is as follows:
where pi is the proportion of individuals for i-th species within a community (Shannon 1948; Shannon and Weaver 1949). Evenness (Eh) was calculated from Shannon’s Index and total number of species captured. Evenness (Eh) was calculated as:
where H is Shannon’s Index calculated in (Eq. 1) and k is the number of total species within a community (Shannon 1948; Shannon and Weaver 1949).
To compare flight patterns among species, we used a negative binomial generalized linear model (package ‘MASS’) to account for non-normal count data for DD for initial capture, duration of capture and number of captured beetles in R Statistical Software (R Core Team 2021). Our model included year, site, stand type, and species with stand type being excluded when considering beetles captured in either coniferous or deciduous woodlots. We tested Shannon’s Index (H) and evenness (Eh) using a generalized linear model. The model was initially tested with year, site, stand type and all their interactions, but because no interactions were significant, these were pooled. The package ‘agricolae’ was used for Tukey’s HSD mean comparison test with ⍺ = 0.05.
Results
Seasonal flight phenology of native and exotic Scolytinae
Initial flight, measured by cumulative degree days (DD), was not affected by year (χ2 = 0.29; df = 1; P = 0.5891) or stand type (χ2 = 3.77; df = 1; P = 0.0522), but was affected by site (χ2 = 16.46; df = 2; P = 0.0003) and species (χ2 = 485.72; df = 26; P < 0.0001; Table 2). Native species had a later average initial flight at 273 DD compared to only 188 DD for exotic species (P < 0.05). One of the earliest beetles to begin flying in spring was X. germanus at 41.1 DD, but with slightly earlier initial flight each year; mid-May in 2014 and 2015 and the last week of April in 2016 and 2019, respectively (Fig. 2), corresponding to a slight increase in degree days each year. Interestingly, A. maiche and X. crassiusculus were later flyers. X. crassiusculus typically began flying mid- to late-May (Fig. 3), around 423.5 DD (Table 2), but the variability was relatively high due to some location-years having low overall captures. A. maiche initial flight occurred consistently within the first week of June (Fig. 4) or 309.9 DD (Table 2).
Flight duration was affected by year (χ2 = 4.41; df = 1; P = 0.0357), site (χ2 = 41.01; df = 2; P < 0.0001) and species (χ2 = 628.31; df = 26; P < 0.0001; Table 2), but not by stand type (χ2 = 0.59; df = 1; P = 0.4418). Exotic Scolytinae species had an average fight duration of 49 d compared to only 10 d flight duration for native beetles (P < 0.05). Anisandrus sayi and X. politus had the longest flight duration for native species at 55.4 and 33.3 d respectively. In comparison, exotic species X. germanus had the longest flight duration of 167.8 d, followed by X. saxesenii at 86.2 d and A. maiche at 81.5 d (P < 0.05; Table 2).
Peak flight of native species occurred around 40‒170 DD with a smaller, secondary peak around 690‒790 DD (Fig. 5A). Generally, two flight peaks were observed for X. germanus–late April to late May or 50–360 DD and then a smaller peak in mid-July or 680–970 DD (Fig. 5B). X. crassiusculus peak flight is difficult to pin-point because overall numbers were quite low in 2014 and 2015, but the largest flight peak was 550–710 DD and smaller peaks occurred as early as 120 DD and as late as 1250 DD (Figs. 5C, 3). Captures of X. crassiusculus in 2016 were much higher and a peak can be clearly observed at the end of June. In comparison, peak flight occurs in mid-August in 2019, but this could be explained by a warmer spring in 2016 compared to 2019. Peak flight duration of A. maiche was much longer—from approximately 250–1100 DD (Fig. 5D), occurring mid-July in 2014, but with two peak flights observed in 2015 and 2016, from mid- or late-June and again in the first week of August. An earlier peak flight was observed in 2019 during the last week in June, with no large second peak (Fig. 4).
Abundance of native vs. exotic Scolytinae
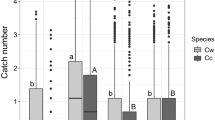
Of the 145,882 total Scolytinae beetles captured across the four trapping years, 622 (0.43%) were native beetles, which was significantly fewer than the 145,260 (99.57%) exotic beetles captured (χ2 = 12,489; df = 1; P < 0.0001). There were ~ 340 × more cumulative exotic species captured per trap each year (138.1 beetles) than native beetles (0.4 beetles; P < 0.05). Within each year, significantly more exotic vs. native cumulative beetles were caught in both the coniferous (χ2 = 4383; df = 1; P < 0.0001) and deciduous habitats (χ2 = 8216; df = 1; P < 0.0001; Fig. 6).
Abundance of native and exotic Scolytinae, Xylosandrus germanus (X.g.), Anisandrus maiche (A.m.) and Xylosandrus crassiusuculus (X.c.) captured in ethanol-baited traps within deciduous and coniferous woodlots are shown. Uppercase letters indicate significant differences between native and exotic Scolytinae; lowercase letters indicate significant differences among three exotic species of ambrosia beetles (general linear model and least squares means; α = 0.05). Mean values and percent of total captures are provided
Total beetles captured per year was significantly affected by year (χ2 = 4.6; df = 1; P = 0.0325), site (χ2 = 25.5; df = 2; P < 0.0001), beetle species (χ2 = 7325.1; df = 26; P < 0.0001), but not by stand type (χ2 = 0.1; df = 1; P = 0.8152). Considering just coniferous woodlots, total beetle captures were affected by year (χ2 = 6.28; df = 1; P = 0.0122), site (χ2 = 17.98; df = 2; P = 0.0001) and species (χ2 = 2252; df = 26; P < 0.0001; Table 2). In deciduous woodlots, beetle captures were not affected by year (χ2 = 0.6; df = 1; P = 0.4386), but were affected site (χ2 = 12.7; df = 2; P = 0.0018) and species (χ2 = 5885; df = 26; P < 0.0001; Table 2). Of the total native beetles collected, 61.1% were captured in coniferous woodlots compared to 38.9% captured in deciduous woodlots (χ2 = 4.053; df = 1; P = 0.0441). Notably, the native bark beetles Cnesinus strigicollis, Conophthorus coniperda, Ips grandicollis, Micracis suturalis, and Pityogenes hopkinsi, and the native ambrosia beetle Gnathotrichus materiarius, were only captured in the coniferous woodlots (Table 2). In contrast, the native ambrosia beetles Anisandrus obesus, Monarthrum fasciatum, Xyleborus pubescens and the exotic bark beetle Hylastes opacus were only captured in the deciduous woodlots (Table 2). Of the total exotic beetles collected, 40.6% were captured in the coniferous woodlots compared to 59.4% in the deciduous woodlots (χ2 = 2.18; df = 1; P = 0.1402).
Overall flight patterns of all native ambrosia beetle species combined showed low numbers of captured beetles, typically under 1 beetle per day with one peak of almost 4 beetles per day (Fig. 5A). The two most abundant native species in both coniferous and deciduous woodlots (P < 0.05), Xyleborinus politus and Anisandrus sayi, represented 3.0 and 1.9 cumulative individuals captured per trap per year, respectively; whereas, the two most abundant exotic species in the coniferous and deciduous habitats, X. germanus and A. maiche, represented 1347.8 and 132.2 cumulative individuals per trap per year, respectively. X. germanus had significantly higher cumulative captures per site per year than any other Scolytinae species in either deciduous or coniferous woodlots (P < 0.0001); with up to 238 beetles per day (Fig. 5B). Across the four trapping years, X. germanus represented a mean of 82.8% and 82.4% of the cumulative beetle captures in the coniferous and deciduous woodlots, respectively (Fig. 6) but were inconsistently higher in deciduous and coniferous woodlots across years (Fig. 2). Number of captured X. crassiusculus were up to 59 beetles per day with similar captures in coniferous and deciduous woodlots, albeit, with very low overall numbers, in 2014 and 2015 and slightly more captures in deciduous woodlots in 2016 and in coniferous woodlots in 2019. It is notable that the Metz location captured zero X. crassiusculus in 2014, 0.25 beetles per trap in 2015 (and much later in the season) and up to just 1 beetle per trap in 2016, then 20 beetles per trap in 2019. However, these numbers were similar in both the coniferous and deciduous Metz woodlots indicating X. crassiusculus likely was introduced into this area sometime between around 2015 and was initially slow to increase in population. Anisandrus maiche represented a mean of 14.6% and 14.3% of the cumulative beetle captures in the coniferous and deciduous woodlots, respectively (Fig. 6). Deciduous woodlots yielded slightly higher captures of A. maiche, with a few exceptions in 2015 and 2019 (Table 2; Fig. 4).
Scolytinae community diversity
Over the course of the study, 16 native species and 11 exotic species of Scolytinae were captured (Table 2). Eight of the 27 species were bark beetles of which seven species were native and one species (Hylastes opacus E.) was exotic (Table 2). Of the 19 species of ambrosia beetles, nine were native and 10 were exotic (Table 2).
No significant difference was detected in the total number of native vs. exotic Scolytinae species collected in the coniferous habitats within each trapping year (i.e., 2014, 2015, 2016, 2019; P > 0.05); however, significantly more exotic vs. native species were collected in the coniferous habitats when pooled across the four sampling years (χ2 = 4.72; df = 1; P = 0.03; Table 3). Significantly more exotic than native Scolytinae were collected in the deciduous habitats in 2015 (χ2 = 6.85; df = 1; P = 0.01) and 2019 (χ2 = 7.16; df = 1; P = 0.01), but not 2014 (χ2 = 3.31; df = 1; P = 0.07) and 2016 (χ2 = 1.81; df = 1; P = 0.18); significantly more exotic vs. native species were collected in the deciduous habitats when pooled across the four years (χ2 = 17.23; df = 1; P < 0.001; Table 3). No significant difference in Shannon’s Index (H) or Evenness (Eh) was detected between the coniferous and deciduous habitats within each of the four sampling years or when pooled across years (P > 0.05; Table 3).
Discussion
Our aim was to analyze flight patterns of native and established exotic ambrosia beetles in coniferous and deciduous woodlots to better understand flight phenology. Identifying initial and peak flight patterns and woodlot preferences allows for better monitoring and trapping to mitigate attacks. As we hypothesized, exotic Scolytinae exhibited earlier flight, longer flight duration and much higher total beetle captures in both coniferous and deciduous woodlots.
Seasonal flight phenology of native and exotic Scolytinae
The early initial flight and longer flight duration exhibited by exotic beetles in our study is likely another contributing characteristic to the invasion success of exotic Scolytinae. Initial flight for exotic species occurred around 188 DD compared to 273 DD for native species, which may allow for exotic species to better target stressed trees with early spring flood events as well as select for damaged trees if a spring frost occurs (La Spina et al. 2013; Ranger et al. 2015). Flight duration of native species was much shorter, about 10 days, compared to 49 days of flight for exotic species. The longer flight duration may allow exotic species to thrive because they have greater opportunity to infest further distances, attack at greater rates, have better selection of vulnerable trees and more adaptability timing their flight to changes in climate patterns.
Native ambrosia beetles had low peak captures, up to 4 beetles per day, around 40–170 DD with a second, smaller peak around 690–790 DD. In comparison, X. germanus flight had two distinct peaks, a larger initial peak from 50 to 350 DD with up to 235 beetles captured per day and a second smaller peak with up to 89 beetles captured per day from 680 to 970 DD, indicating potential for a second generation as has been observed as early as March/April in warmer climates within the US (Hudson and Mizell 1999; Oliver and Mannion 2001; Reding et al. 2010; Werle et al. 2012, 2015; Viloria et al 2021). Peak flight of X. crassiusculus was observed much later, at 550–710 DD, with up to 59 beetles captured per day. Similar to our study, only one generation was observed for X. crassiusculus in Ohio and Virginia (Reding et al. 2010), but a second peak, and potentially a second generation occurred in Tennessee (Reding et al. 2010; Viloria et al. 2021). Fight patterns of A. maiche sustained high numbers throughout a longer period—from 250 to 1100 DD. This sustained duration of comparatively high flight activity also makes it difficult to determine if later flight could be the result of a second generation of A. maiche or a long period of emergence from overwintering. Furthermore, because A. maiche is a more recent introduction, fewer studies have shown flight patterns of this species, so a second generation could also be possible in warmer climates. Compared to native species, X. germanus had an earlier and longer seasonal flight pattern and A. maiche had a later and longer seasonal flight pattern, which may help explain why competition is minimal and why these two species are so successful in Ohio. Although there is overlap in flight activity with A. maiche and X. crassiusculus, the windows of flight do seem to allow for some stratification between these species, allowing them to coexist rather than compete.
Abundance of native vs. exotic Scolytinae
Although the number of native species captured (16) was higher than that of exotic species (11), the difference of actual beetle captures over the four years of this study was alarmingly much higher for exotic (145,260) than native beetles (622). The proportion of exotic beetles captured in our study was higher (99.6%) compared to other studies which have shown as few as 60% adventive beetles captured (Gandhi et al. 2010), 86.9% exotic beetles captured (Reed and Muzika 2010) and up to 89.3–97.2% adventive beetles captured (Miller et al. 2015). The differences could be due to different combinations of trapping lures in other studies (compared to just ethanol in our study) or due to the longer trapping duration in our study. Furthermore, climate may affect the range of Scolytinae; temperature has been shown to limit the geographic range of non-native ambrosia beetle species within the US and rainfall is more limiting to ambrosia beetles than bark beetles when colonizing new areas (Rassati et al. 2016b). It is likely exotic beetles captured in our study are already established and have a lower species replacement component, meaning they are likely to be found across a larger geographic range, have lower host tree preference, and may be able to overcome changes in temperature or precipitation (Rassati et al. 2016a, b). This, coupled with their high numbers can cause severe damage and economic loss to trees and exhibit their increasing potential as a severe tree pest. Conversely, native bark beetle species have been shown to be more harmful than neonative or alien species, and are more limited in establishing in a non-native range by both biotic and abiotic conditions their environment (Forgione et al. 2022). However, if the environment is adequate, ambrosia beetles are less selective of their host compared to bark beetles (Rassati et al. 2016b). Indeed, studies have shown regional differences in ambrosia beetle species composition (Reding et al. 2010; Miller et al. 2018)—likely indicative of their preferred climates rather than host type or host range.
Our study quantified much higher numbers of exotic Scolytinae—341-times more total captured beetles per species than native beetles. Furthermore, we were able to illustrate how prominent X. germanus has become, with 1348 beetles captured per trap and show that A. maiche populations have increased in recent years, becoming a growing concern as a tree pest. As of 2013 and 2014, 19 and 9 specimens of A. maiche were collected in three counties within Ohio (Reding et al. 2015) but within a few years, A. maiche has increased to alarming numbers; our study showed an average capture of 132 beetles each year with a minimum capture of 37 beetles at one location in 2014 and a maximum of 1642 at one location in 2016. Although it is not currently found in large numbers in Europe, A. maiche has been reported in Ukraine in 2007 and Russia in 2009, and more recently in the Veneto Region of Italy in 2021 (Colombari et al. 2022). Based on DNA sequencing, the specimen collected is a closer match to the population in the Eastern U.S. and Canada than the specimen found in Ukraine, indicating a potential introduction from shipments from the US rather than expansion from Eastern Europe (Colombari et al. 2022). However, as we show in this current study, its population growth has occurred much more rapidly in North America than it has in Europe and should be continued to be monitored to prevent further population growth.
Ambrosia beetle community
There was no difference in community diversity (Shannon’s Index) nor was there a difference in evenness between exotic and native species. However, both Shannon’s Index and evenness decreased each year and Shannon’s Index varied by site. This indicates that, although the community diversity and evenness is decreasing each year– likely due to increasing populations of a few species, there is no indication of competition or displacement of native species from the highly successful exotic species. Indeed, although infrequently (i.e., 3.3% of caged galleries), multiple species have been shown to emerge from the same galleries (Oliver and Mannion 2001).
There are numerous studies on alien species colonization in new habitats (Zach et al. 2001; Økland et al. 2011; Rassati et al. 2016c; Olenici et al. 2022) and evidence of increasing rates of alien introductions (Kirkendall and Faccoli 2010). Elton’s diversity-invasion hypothesis predicts species to be less successful colonizers when introduced to more diverse communities (ie, deciduous woodlots) because there are fewer unoccupied niches available (Elton 1958). Although initial flight, duration of flight, total captured beetles and ambrosia community diversity and evenness were similar in both coniferous and deciduous woodlots in our study, other studies have shown a more diverse woodlot to have greater ambrosia beetles because of increased availability of niches, more shrubbery and canopy stratification and potential for reduced windspeed, and therefore, greater flight ability (Pasek 1988; Mahroof et al. 2010). Werle et al. (2015) attributed differences in flight between adjacent woodlots and nurseries to woodlots having less wind and allowing for easier flight. Alternatively, availability of breeding substrate (Gossner et al. 2019) could have been similar within sites in our study and produced similar progeny and therefore no statistical differences in trap captures. Stratification of flight patterns over a season may alter the Scolytinae community by allowing species to coexist, but at different times rather than compete for the same resources, especially if stressed trees are of a lower abundance in any given year. Another hypothesis may be that host trees are not as efficient at defending against exotic Scolytinae—that there is simply a lack of or delay of host tree defenses against these beetles (Forgione et al. 2022). Additionally, the resource allocation hypothesis poses that exotic beetles may be better able to shift their energy into reproduction rather than enemy defenses (Blossey and Nötzold 1995; Doorduin and Vrieling 2011; Forgione et al. 2022) and Darwin’s naturalization hypothesis suggests less competition for exotic species in the U.S. compared to their native range (Darwin 1859; Forgione et al. 2022).
Conclusion
Our study documented initial flight, duration of flight, number of captured beetles, community diversity and evenness to be similar in both coniferous and deciduous woodlots. By comparing exotic species to native species, we were able to show minimal competition, and stratification of flight phenology that allow these species to coexist. Results we obtained show that initial flight begins mid- to late-April and peaks in May and July for X. germanus and in June for A. maiche in Ohio, which allows us to better predict, detect and manage for these pest to mitigate tree damage in nurseries and orchards. Results also showed that exotic Scolytinae fly for a longer duration, which in conjunction with earlier flight phenology is at least partially responsible for their thriving populations compared to native species.
Data availability
All of the presented data associated with this study are freely available upon request to the corresponding author.
References
Agnello AM, Breth DI, Tee EM, Cox KD, Villani SM, Ayer KM, Wallis AE, Donahue DJ, Combs DB, Davis AE, Neal JA (2017) Xylosandrus germanus (Coleoptera: Curculionidae: Scolytinae) occurrence, fungal associations, and management trials in New York apple orchards. J Econ Entomol 110(5):2149–2164
Anderson DM (1974) First record of Xylebrous semiopacus in the continental United States (Coleoptera, Scolytidae). Cooperative Econ Insect Rep 24(45/48):863–864
Atkinson TH, Rabaglia RJ, Bright DE (1990) Newly detected exotic species of Xyleborus (Coleoptera: Scolytidae) with a revised key to species in eastern North America. Can Entomol 122(1):93–104
Blossey B, Nötzold R (1995) Evolution of increased competitive ability in invasive nonindigenous plants: a hypothesis. J Ecol 83(5):887–889
Brockerhoff EG, Liebhold AM (2017) Ecology of forest insect invasions. Biol Invasions 19:3141–3159
Brockerhoff EG, Liebhold AM, Richardson B, Suckling DM (2010) Eradication of invasive forest insects: concepts, method, costs and benefits. N Z J for Sci 40(suppl):S117–S135
CABI (2021) Xylosandrus crassiusculus (Asian ambrosia beetle). CABI compendium. 16 Nov 2021. https://doi.org/10.1079/cabicompendium.57235
Castrillo LA, Griggs MH, Vandenberg JD (2012) Brood production by Xylosandrus germanus (Coleoptera: Curculionidae) and growth of its fungal symbiont on artificial diet based on sawdust of different tree species. Environ Entomol 4:822–827
Colombari F, Martinez-Sañudo I, Battisti A (2022) First report of the alien ambrosia beetle Cnestus mutilates and further finding of Anisandrus maiche in the European part of the EPPO region (Coleoptera: Curculionidae: Scolytinae: Xyleborini). EPPO Bull 52:446–450
Darwin C (1859) On the origin of species by means of natural selection, or the preservation of favoured races in the struggle for life. Murray, London
Dole SA, Jordal BH, Cognato AI (2010) Polyphyly of Xylosandrus Reitter inferred from nuclear and mitochondrial genes (Coleoptera: Curculionidae: Scolytinae). Mol Phylogenet Evol 54:773–782
Doorduin LJ, Vrieling K (2011) A review of the phytochemical support for the shifting defence hypothesis. Phytochem Rev 10(1):99–106
Elton CS (1958) The ecology of invasions by plants and animals. Methuen, London
Felt EP (1932) A new pest in greenhouse grown grape stems. J Econ Entomol 25(2):418
Forgione L, Bacher S, Vimercati G (2022) Are species more harmful in their native, neonative or alien range? Insights from a global analysis of bark beetles. Divers Distrib 28:1832–1849
Galko J, Dzurenko M, Ranger CM, Kulfan J, Kula E, Nikolov C, Zúbrik M, Zach P (2018) Distribution, habitat preference, and management of the invasive ambrosia beetle Xylosandrus germanus (Coleoptera: Curculionidae, Scolytinae) in European forests with an emphasis on the West Carpathians. Forests 10(10):1–18
Gandhi KJK, Cognato AI, Lightle DM, Mosley BJ, Nielsen DG, Herms DA (2010) Species composition, seasonal activity, and semiochemical response of native and exotic bark and ambrosia beetles (Coleoptera: Curculionidae: Scolytinae) in northeastern Ohio. J Econ Entomol 103(4):1187–1195
Gomez DF, Rabaglia RJ, Fairbanks KEO, Hulcr J (2018) North American Xyleborini north of Mexico: a review and key to genera and species (Coleoptera, Curculionidae, Scolytinae). ZooKeys 768:19–68. https://doi.org/10.3897/zookeys.768.24697
Gossner MM, Falck K, Weisser WW (2019) Effects of management on ambrosia beetles and their antagonists in European beech forests. For Ecol Manage 437:126–133
Gugliuzzo A, Criscione G, Siscaro G, Russo A, Tropea Garzia G (2019) First data on the flight activity and distribution of the ambrosia beetle Xylosandrus compactus (Eichhoff) on carob trees in Sicily. EPPO Bull 49(2):340–351
Gugliuzzo A, Criscione G, Biondi A, Aiello D, Vitale A, Polizzi G, Tropea Garzia G (2020) Seasonal changes in population structure of the ambrosia beetle Xylosandrus compactus and its associated fungi in a southern Mediterranean environment. PLoS ONE 15(9):e0239011
Gugliuzzo A, Biedermann PHW, Carrillo D, Castrillo LA, Egonyu JP, Gallego D, Haddi K, Hulcr J, Jactel H, Kajimura H, Kamata N, Meurisse N, Li Y, Oliver JB, Ranger CM, Rassati D, Stelinksi LL, Sutherland R, Garzia GT, Wright MG, Biondi A (2021) Recent advance toward the sustainable management of invasive Xylosandrus ambrosia beetles. J Pest Sci 94:615–637
Haack RA (2001) Intercepted Scolytidae (Coleoptera) at U.S. ports of entry: 1985–2000. Integ Pest Manag Rev 6:253–282
Haack RA (2006) Non-native bark- and wood-boring Coleoptera in the United States: recent establishments and interceptions. Can J for Res 35:269–288
Henin JM, Versteirt V (2004) Abundance and distribution of Xylosandrus germanus (Blandford 1894) (Coleoptera Scolytidae) in Belgium: new observations and an attempt to outline its range. J Pest Sci 77:57–63
Hudson W, Mizell R (1999) Management of Asian ambrosia beetle, Xylosandrus crassiusculus, in nurseries. Proc Southern Nursery Assoc Res Conf 44:198–201
Hulcr J, Stelinski LL (2017) The ambrosia symbiosis: from evolutionary ecology to practical management. Annu Rev Entomol 62:285–303
Hulcr J, Beaver RA, Puranasakul W, Dole SA, Sontichai S (2008) A comparison of bark and ambrosia beetle communities in two forest types in Northern Thailand (Coleoptera: Curculionidae: Scolytinae and Platypodinae). Environ Entomol 37:1461–1470
Kirkendall LR, Faccoli M (2010) Bark beetles and pinhole borers (Curculionidae, Scolytinae, Platypodinae) alien to Europe. ZooKeys 56:227–251.
Kirkendall LR, Wrensch DL, Ebbert MA (1993) Ecology and evolution of biased sex ratios in bark and ambrosia beetles. In: Wrensch DL, Ebbert MA (eds) Evolution and diversity of sex ratio in insects and mites. Chapman & Hall, New York, NY, pp 235–345
Kirkendall LR, Biedermann PHW, Jordal BH (2015) Evolution and diversity of bark and ambrosia beetles. In: Vega FE, Hofstetter RW (eds) Bark beetles: biology and ecology of native and invasive species. Elsevier, Amsterdam, pp 85–156
La Spina S, De Cannière C, Dekri A, Grégoire JC (2013) Frost increases beech susceptibility to scolytine ambrosia beetles. Agric for Entomol 15:157–167
Maeto K, Fukuyama K, Kirton LG (1999) Edge effects on ambrosia beetle assemblages in a lowland forest, bordering oil palm plantations in peninsular Malaysia. J Trop Forest Sci 11:537–547
Mahroof RM, Edde PA, Robertson B, Puckette JA, Phillips TW (2010) Dispersal of Rhyzopertha dominica (Coleroptera: Bostrichidae) in different habitats. Environ Entomol 39:930–938
Miller DR, Rabaglia RJ (2009) Ethanol and (−)-α-pinene: Attractant kairomones for bark and ambrosia beetles in the southeastern US. J Chem Ecol 35:435–448
Miller DR, Dodds KJ, Hoebeke ER, Poland TM, Willhite EA (2015) Variation in effects of conophthorin on catches of ambrosia beetles (Coleoptera: Curculionidae: Scolytinae) in ethanol-baited traps in the United States. J Econ Entomol 108(1):183–191
Miller DR, Crowe CM, Ginzel MD, Ranger CM, Schultz PB (2018) Comparison of baited bottle and multiple-funnel traps for ambrosia beetles (Coleoptera: Curculionidae: Scolytinae) in Eastern United States. J Entomol Sci 53(3):347–360
Monterrosa A, Joseph SV, Blaauw B, Hudson W, Acebes-Doria AL (2022) Ambrosia beetle occurrence and phenology of Xylosandrus spp. (Coleoptera: Curculionidae: Scolytinae) in ornamental nurseries, tree fruit, and pecan orchards in Georgia. Environ Entom 51(5):998–1009
Normark BB, Jordal BH, Farrell BD (1999) Origin of a haplodiploid beetle lineage. Proc R Soc Lond B Biol Sci 266:2253–2259
Økland B, Erbilgin N, Skarpaas O, Christiansen E, Långström B (2011) Inter-species interactions and ecosystem effects of non-indigenous invasive and native tree-killing bark beetles. Biol Invasions 13:1151–1164
Olenici N, Duduman M, Popa I, Isaia G, Paraschiv M (2022) Geographical distribution of three forest invasive beetle species in Romania. Insects 13(7):621
Oliver JB, Mannion CM (2001) Ambrosia beetle (Coleoptera: Scolytidae) species attacking chestnut and captured in ethanol-baited traps in middle Tennessee. Environ Entomol 30:909–918
Pasek JE (1988) Influence of wind and windbreaks on local dispersal of insects. Agr Ecosyst Environ 22:539–554
R Core Team (2021) R: a language and environment for statistical computing. R Foundation for Statistical Computing. Vienna, Austria. https://www.R-project.org/
Rabaglia RJ, Vandenberg NJ, Acciavatti RE (2009) First records of Anisandrus maiche Stark (Coleoptera: Curculionida: Scoytinae) from North America. Zootaxa 2137:23–28
Ranger CM, Reding ME, Persad AB, Herms DA (2010) Ability of stress-related volatiles to attract and induce attacks by Xylosandrus germanus and other ambrosia beetles. Agric for Entomol 12(2):177–185
Ranger CM, Schultz PB, Frank SD, Chong JH, Reding ME (2015) Non-native ambrosia beetles as opportunistic exploiters of living but weakened trees. PLoS ONE 10:e0131496
Ranger CM, Reding ME, Shultz PB, Oliver JB, Frank SD, Addesso KM, Chong JH, Sampson B, Werle C, Gill S, Krause C (2016) Biology, ecology, and management of nonnative ambrosia beetles (Coleoptera: Curculionidae: Scolytinae) in ornamental plant nurseries. J Integr Pest Manag 7(1):1–23
Ranger CM, Reding ME, Addesso K, Ginzel M, Rassati D (2021) Semiochemical-mediated host selection by Xylosandrus spp. ambrosia beetles (Coleoptera: Curculionidae) attacking horticultural tree crops: a review of basic and applied science. The Canadian Entomol. 153: 103-120.
Rassati D, Faccoli M, Battisi A, Marini L (2016a) Habitat and climate preferences drive invasions of non-native ambrosia beetles in deciduous temperate forests. Biol Invasions 18:2809–2821
Rassati D, Faccoli M, Haack RA, Rabaglia RJ, Toffolo EP, Battisi A, Marini L (2016b) Bark and ambrosia beetles show different invasion patters in the USA. PLoS ONE. https://doi.org/10.1371/journal.pone.0158519
Rassati D, Lieutier F, Faccoli M (2016c) Alien wood-boring beetles in Mediterranean regions. In: Paine T, Lieutier F (eds) Insects and diseases of mediterranean forest systems. Springer, Cham. https://doi.org/10.1007/978-3-319-24744-1_11
Reding ME, Oliver JB, Schultz PB, Ranger CM (2010) Monitoring flight activity of ambrosia beetles in ornamental nurseries with ethanol-baited traps; influence of trap height on captures. J Environ Hortic 28:85–90
Reding ME, Ranger CM, Sampson BJ, Werle CT, Oliver JB, Schultz PB (2015) Movement of of Xylosandrus germanus (Coleoptera: Curculionidae) in Ornamental Nurseries and Surrounding Habitats. J Econ Entomol 108(4):1947–1953.
Reed SE, Muzika RM (2010) The influence of forest stand and site characteristics on the composition of exotic dominated ambrosia beetle communities (Coleoptera: Curculionidae: Scolytinae). Environ Entomol 39(5):1482–1491
Ruzzier E, Prazaru SC, Faccoli M, Duso C (2021) Xylosandrus germanus (Blandford, 1894) on grapevines in Italy with a compilation of world Scolytine weevils developing on Vitaceae. Insects 12(869):1–18
Shannon CE (1948) A mathematical theory of communication. Bell Syst Tech J 27:379–423
Shannon CE, Weaver W (1949) The mathematical theory of communication. University of Illinois Press, Urbana
Sittichaya W, Permkam S, Cognato AI (2012) Species composition and flight pattern of Xyleborini ambrosia beetles (Col. Curculionidae: Scolytinae) from agricultural areas in southern Thailand. Environ Entomol 41(4):776–784
Thornton MM, Shrestha R, Wei Y, Thornton PE, Kao S-C, Wilson BE (2022) Daymet: daily surface weather data on a 1-km grid for North America, Version 4 R1. ORNL DAAC, Oak Ridge, Tennessee, USA.https://doi.org/10.3334/ORNLDAAC/2129
Viloria Z, Villanueva RT, Bessin R, O’Neal P, Ranger CM, Dunwell W (2021) Scolytinae in nursery and fruit crops of Western Kentucky and seasonal population patterns of four invasive ambrosia beetles. J Entomol Sci 56(3):374–386
Weber BC, McPherson JE (1983a) World list of host plants of Xylosandrus germanus (Blandford) (Coleoptera: Scolytidae). Coleopt Bull 37(2):114–134
Weber BC, McPherson JE (1983b) Life history of the ambrosia beetle Xylosandrus germanus (Coleoptera: Scolytidae). Ann Entomol Soc Am 76(3):455–462
Weber BC, McPherson JE (1984) Attack on black walnut trees by the ambrosia beetle Xylosandrus germanus (Coleoptera: Scolytidae). For Sci 30(4):864–870
Werle CT, Sampson JJ, Oliver JB (2012) Diversity, abundance and seasonality of ambrosia beetles (Coleoptera: Curculionidae) in Southern Mississippi. Mid-South Entomol 5:1–5
Werle CT, Chong JH, Sampson BJ, Reding ME, Adamczyk JJ (2015) Seasonal and spatial dispersal patterns of select ambrosia beetles (Coleoptera: Curculionidae) from forest habitats into production nurseries. Fla Entomol 98(3):884–891
Zach P, Topp W, Kulfan J, Simon M (2001) Colonization of two alien ambrosia beetles (Coleoptera, Scolytidae) on debarked spruce logs. Biologia (bratislava) 56(2):175–181
Acknowledgements
We thank the reviewers for taking the time to give helpful feedback on this manuscript.
Funding
This research was supported by USDA-NIFA-SCRI award #2021-51181-35863, USDA-Floriculture and Nursery Research Initiative, and base funds associated with USDA-ARS National Program 305 Project #5082-21000-018-00D. The funders had no role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
JAB: statistical analysis, writing. JB: methodology, execution of experiments, data collection, writing—review and editing. MER: conceptualization, project administration, writing—review and editing. CMR: conceptualization, methodology, supervision, project administration, writing—review and editing.
Corresponding authors
Ethics declarations
Competing of interest
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Baniszewski, J.A., Barnett, J., Reding, M.E. et al. Seasonal dominance of exotic ambrosia beetles compared to native species within deciduous and coniferous woodlots. Biol Invasions 26, 1651–1668 (2024). https://doi.org/10.1007/s10530-024-03255-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10530-024-03255-z