Abstract
Peacock basses (Cichla spp.) are native to the Amazon basin but introduced to different parts of the world. Almost thirty years ago, Cichla kelberi was introduced in an impoundment of the São João River, a coastal system in southeastern Brazil. Recently, this cichlid invaded the estuarine section of the basin. This study aims to analyze spatial and temporal variations in catch of C. kelberi and fish assemblage structure along the estuarine stretch of this river and how abiotic variables affect their distribution. Sampling was performed in four segments downstream of the dam. Principal component analysis revealed that abiotic variables displayed temporal and spatial variation, in part due to the salinity gradient, that were more pronounced in the dry season. Cichla kelberi occurred in all segments, but mainly in shallow and vegetated habitats of the middle course and barely in the most downstream. Eighty-one fish species were recorded, nine of which were non-native, representing 33.4% of total catch. A redundancy analysis indicated that the fish assemblages showed marked spatial variation, mostly related to the salinity gradient. The lowermost segment of the river was dominated by marine species, the only locality where non-native species summed less than 40% of the catches. In the upstream segments, higher oxygen levels and lower temperatures influenced the occurrence of most species. Higher salinity of the estuary seems to limit the spread of C. kelberi, but the invader may reach adjacent basins in years of exceptional floods. The eurihalinity and piscivory of C. kelberi partly explain its invasive success.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Habitat loss and biological invasions are major threats to global biodiversity (Mack et al. 2000; Simberloff et al. 2013). In freshwater ecosystems, habitat loss and degradation (e.g., pollution, channelization and regulation) are usually followed by species invasions (Agostinho et al. 2005, 2008; Pelicice et al. 2017). For example, several non-native fish species have been introduced in reservoirs to compensate for the negative impacts of damming on fisheries (i.e., decline of commercially important species; see Hoeinghaus et al. 2009), causing additive or multiplicative effects on fish diversity. In fact, reservoirs have facilitated the invasion and further establishment of non-native species, working as sources for their dispersal towards adjacent areas (Havel et al. 2005; Johnson et al. 2008; Petesse and Petrere Jr 2012). In addition, fish assemblages in impounded rivers are impoverished, so they are more susceptible to invasions (Petesse and Petrere Jr 2012; Vitule et al. 2012; Daga et al. 2015; Pelicice et al. 2018).
In South America, species of peacock basses (genus Cichla) have been introduced into several reservoirs outside their native range, i.e., tributaries of the Amazon system (Agostinho et al. 2007; Franco et al. 2018). Once established in the reservoir, these voracious predators are able to successfully invade adjacent areas, such as riverine sections downstream from the dam (Poff et al. 2007; Agostinho et al. 2008; Espínola et al. 2010). However, no study has evaluated the invasion of Cichla in estuarine ecosystems. The lower course of coastal rivers in eastern South America is generally characterized by meandering channels that flow into the estuary, subjected to daily variations in salinity due to the tidal cycle. However, several coastal rivers, particularly in southeastern Brazil, experienced drastic alterations during the twentieth century, mostly related to damming, excavation of secondary narrow channels, channelization and bank clearing to drain floodplains and to replace the original Atlantic Forest with pasture for livestock ranching (Sofiati 2010; Marçal et al. 2017). These alterations may affect the native fish fauna and create opportunity for invasions. This aspect of invasion biology has been less investigated, although Cichla species have been introduced in rivers and impoundments close to estuaries.
The yellow peacock bass Cichla kelberi (Kullander and Ferreira 2006) is currently introduced in several non-Amazonian reservoirs and rivers. This species was introduced decades ago in the Juturnaíba Reservoir, São João River, a costal system in southeastern Brazil. However, this fish was recently recorded in the estuarine section located downstream, and no study has investigated C. kelberi populations and the structure of fish assemblages in this recently invaded area. In this study, we sampled fish assemblages along the estuarine course of the São João River to investigate whether the non-native C. kelberi succeed in invading assemblages irrespective of the increasing salinity gradient imposed by the tidal cycle and flow regulation. Our main goals were to analyze spatial and temporal variations in total catch of C. kelberi and fish assemblage structure, and their relationship with abiotic conditions. We hypothesized that the estuarine conditions mainly related to gradients in water salinity would explain fish assemblage composition and restrain the distribution of the C. kelberi to riverine reaches that resemble its native range (Winemiller 2001; Pelicice et al. 2015), i.e., natural channels that are structurally complex, freshwater and with high temperatures.
Materials and methods
Study area
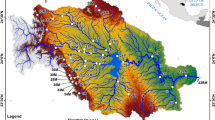
The São João River basin covers approximately 2190 km2 of Atlantic rainforest (22° 30′–22° 41′ S and 41° 59′–42° 20′ W), in the Rio de Janeiro State, Brazil. The area is moderately occupied by forestry and livestock farming. The river originates at 800 m of elevation and runs about 120 km west, flowing into the Atlantic Ocean; in the lower reach, it runs along a sinuous mangrove-dominated estuary (Fig. 1). Annual mean air temperature range between 22 and 24 °C, and maximum annual precipitation reaches 1000 mm. Rainfall is concentrated between October and March (warm months, hereafter rainy season), and lower precipitation between April to September (cold months, hereafter dry season) (Alvares et al. 2013). During the study, peaks of rainfall and river flow occurred respectively in January and February 2016, with the lowest values in April 2016 (Online Resource 1).
Location of the São João River basin, Rio de Janeiro State, Brazil, and the four 10 km long segments downstream Juturnaíba Reservoir (S1: channelized segment, just below the dam, S2: meandering segment, on the abandoned channel, S3: channelized segment on the longest channelized stretch, and S4: meandering segment along the mangrove area)
The Juturnaíba Reservoir is located about 40 km upstream from the river mouth, where several low order tributaries of the São João River formerly drained into a wetland known as Juturnaíba lagoon. The reservoir was constructed between 1978 and 1984, inundated an area of 43 km2 and since then supplies fresh water to nearly 600,000 inhabitants of the nearby coastal municipalities. Other hydraulic modifications of the main channel and tributaries involved channelization and loss of meandering courses along the stretch (ca. 25 km) below the dam, which includes the estuarine area. In this downstream segment, most of the natural riparian forest was cleared and replaced by pasture with non-native grasses; forest remnants are still found in some areas, especially in the remaining meanders of the river, which run parallel and maintain hydrological connectivity with the channelized main course (Fig. 1).
Sampling and laboratory procedures
Fish assemblages were sampled downstream from the dam in four segments along the São João River (Fig. 1). These segments show different degrees of hydrological alteration, influenced by dam operation and tidal cycle. They also cover the entire river section between the dam and the ocean, representing a strong salinity gradient. “Segment 1” (S1; about 15 km long) is the straight channel of the São João River, extending from the dam to the confluence of the uppermost abandoned meander; this area is under greater influence of flow regulation. Downstream from Segment 1, the São João River splits in two branches, where segments 2 and 3 run parallel. “Segment 2” (S2) is about 15 km long and encompasses the longest abandoned meandering channel, which is deep, densely colonized by aquatic macrophytes and covered by forests. “Segment 3” (S3) is about 10 km long and consists of the channelized section, with banks dominated by grasses and shallow areas colonized by the widgeon grass Ruppia maritima. “Segment 4” (S4) is the lowermost stretch, about 10 km long, and runs sinuously along a mangrove area subjected to higher marine influence.
Fish sampling and measurement of abiotic variables were performed at three fixed equidistant sites in each segment between August 2014 and November 2016, divided in two campaigns: the first one (from August 2014 to August 2015) designed to catch the piscivorous fish, and the second one (from January to November 2016) designed to sample fish assemblages. In the first campaign, we monthly sampled piscivorous fish with standardized angling effort, which consisted of two anglers using line and hook with artificial baits for one hour at each sampling site. We used this methodology because some piscivorous are poorly captured by traditional sampling techniques, particularly Cichla species. In the second campaign, fish assemblages were sampled bimonthly under neap tides with different gears: gillnets (mesh sizes: 15, 20, 25, 30, 35, 40, 45, 60, and 80 mm between opposing knots), trammel nets (mesh sizes: 35 and 110 mm), cast nets (mesh sizes: 10, 20 and 25 mm), seines (mesh size: 5 mm), sieves and dip nets. Sampling effort was standardized among sites and dates. Trammel and gill nets were set between 16:00 and 08:00 and checked at 21:00. Fish abundance (catch) was expressed as the number of individuals captured corrected by sampling effort (catch per unit effort, CPUE). On each sampling occasion, we recorded the following abiotic variables: salinity, conductivity, dissolved oxygen, and water temperature with a multi-parameter probe (YSI Pro2030), pH (pH 1–14 Merck™ test strips), water transparency (Secchi disk), and depth (a scaled rope).
Captured specimens were stored in ice in the field, immersed in a 10% formalin solution and later transferred to a 70% alcohol solution. In the laboratory, total length (TL, to the nearest mm) of each specimen was measured; identification of the specimens employed taxonomic guides and keys (e.g., Figueiredo and Menezes 1978, 1980, 2000; Menezes and Figueiredo 1980, 1985; Kullander and Ferreira 2006). Native and non-native species were identified (Eschmeyer and Fricke 2016). Voucher specimens were deposited in the Fish Collection of the Instituto de Biodiversidade e Sustentabilidade, Universidade Federal do Rio de Janeiro (NUPEM/UFRJ).
Statistical analyses
Data on conductivity, dissolved oxygen, depth, salinity and CPUE of C. kelberi were log10 transformed prior to analyses [log10 (x + 0.01) for salinity and log10 (x + 1) for CPUE]. We ran a principal component analyses (PCA) to summarize the temporal and spatial variation in abiotic variables (first campaign: 12 sampling sites in 12 monthly surveys; second campaign: 12 sampling sites in six bimonthly surveys). Only PCA eigenvalues higher than those produced by a broken-stick model, available in the “PCAsignificance” function of the “BiodiversityR” package (Kindt and Coe 2005) in R (R Core Team 2020), were retained for interpretation (Jackson 1993).
Two (multiple) zero-inflated negative binomial regressions (NBR), using the ‘‘zeroinfl’’ function in package “pscl” (Jackman 2020) in R, were employed to assess the variation of CPUE of C. kelberi in the first campaign among seasons (rainy and dry seasons) and segments (P1 to P4) and to test for the role of abiotic variables. For this analysis, conductivity was removed because it was strongly correlated to salinity (r = 0.97); the rest of predictors did not show collinearity (all Variance inflation factors < 1.8). NBR were used because conventional generalized linear models with Poisson errors showed overdispersion, and a zero-inflation NBR was preferable to these models and also a conventional NBR (Akaike weight = 0.95), following R scripts provided by Magellan and García-Berthou (2015).
A redundancy analysis (RDA) was applied to explore the relationship between the fish species catch matrix and abiotic variables. Since the use of seines was unfeasible at some sites, species uniquely caught with this gear were excluded from analyses (13.8% of the species recorded). A Hellinger transformation was used for the species catch matrix, since it provides low weights to species with low counts and many zeros (Legendre and Gallagher 2001). A forward model selection was performed with function “ordiR2step” (200 permutations; Blanchet et al. 2008) in ‘vegan’ (Oksanen et al. 2019), in order to select a parsimonious RDA model with the highest adjusted R2. All ordination analyses were carried out with the ‘vegan’ package in R.
Results
Limnological features
Salinity and conductivity varied markedly among the four segments and related negatively with the distance to river mouth. Segments 1, 2 and 3 were generally freshwater (< 0.5 ppt), but Segments 2 and 3 were sometimes oligohaline; Segment 4 varied more strongly from freshwater to polyhaline conditions, i.e., mixohaline/brackish waters (Table 1). Brackish waters (up to 30.1 ppt) were recorded in Segment 4 especially during the dry season, whereas salinity levels dropped to half during the rainy season (Table 1; Fig. 2). Temperature was lower in the dry season (22 to 25.6 °C) compared to the rainy season (25.2 to 29.8 °C). In general, the channelized segments (S1 and S3) were shallower than the meandering segments (S2 and S4) (Table 1).
Principal Component Analysis of the abiotic variables recorded at four segments of the São João River during the first campaign (August 2014 to August 2015) and the second campaign (January to November 2016). 95% data ellipses for the rainy (October to March) and dry (April to September) seasons are shown (symbols in blue and red, respectively). Different symbols for the four segments are also used
The first two axes of the PCA were significant (broken-stick model) and explained most of the variation (51.3%) in water properties, which varied particularly between the two seasons (Fig. 2). Samples from the dry season presented higher PC1 and PC2 scores and were associated to higher values of Secchi depth, water depth, oxygen concentration, pH and lower values of temperature. Spatial variation was less clear and depended on season: Segment 1 presented less variability than the others; Segment 3 had higher Secchi depth and water depth in the dry season; and Segment 4 had the highest pH, salinity, and oxygen concentration in the dry season but low in the rainy season (Fig. 2).
Catches of piscivorous fish
Three piscivorous species were recorded in the first campaign, the marine/estuarine Centropomus parallelus and the freshwater Hoplias malabaricus and Cichla kelberi. The latter was the most captured, representing 73.3% of the catches of piscivorous fishes (Fig. 3), whereas the others were recorded in low numbers, with higher catches in the rainy season. Cichla kelberi catches by angling varied significantly among segments (NBR, P < 0.001) and seasons (P = 0.002), but with no significant segment × season interaction (P = 0.987). Most C. kelberi specimens (58% of 162) were captured in Segment 3, whereas no individual was recorded in Segment 4 by angling. Most catches of C. kelberi were at shallow sites of the channelized vegetated course (Segment 3) with grass-dominated riverbanks (Fig. 3, Online Resource 2). The multiple zero-inflated NBR explained 60% of the total variation in catches (Table 2). Among the abiotic predictors, only salinity was significantly related to peacock bass catches (P = 0.001) (Table 2). C. kelberi catches by angling were highest in Segment 3, where they increased with salinity, and null in the estuarine Segment 4 (Online Resource 2).
Fish assemblages
A total of 2,551 fish specimens belonging to 80 species, 36 families and 18 orders were captured in the second campaign (Online Resource 3). Most specimens (54.7% of total catch) belonged to the families Callichthyidae, Cichlidae, Serrasalmidae, Carangidae and Mugilidae. Twelve species were recorded in all four segments, four non-native species, including C. kelberi. Total species richness ranged from 25 to 62 species, with highest richness in Segment 4, mostly influenced by the presence of marine fishes (Fig. 4); freshwater species richness was low in this segment. We observed low variation in non-native richness among segments (Fig. 4). Total catches ranged between 281 and 957, with higher values in Segment 3 (Fig. 4), dominated by native and non-native freshwater fishes. The vast majority of C. kelberi catches (76%) occurred in Segment 3, but only four peacock basses were captured there by sampling gears other than line and hook.
Non-native species dominated segments 1 to 3 (Fig. 5a–c), where Hoplosternum littorale and Metynnis lippincottianus presented the highest relative catches. Considering the nine most common species, non-native species accounted for 42, 55 and 50% of total catch in segments 1, 2 and 3, respectively. The main freshwater species in these segments were Trachelyopterus striatulus, Geophagus brasiliensis and Hoplias malabaricus. Marine species prevailed in Segment 4 (Fig. 5d), although the non-native H. littorale was among the main species (Online Resource 3).
The first two axes of the RDA explained 8% of the variation in species composition (Fig. 6). The “ordiR2step” function selected salinity and dissolved oxygen as the best predictors of the composition and catch of the assemblages (permutation tests, P < 0.05). Sampling sites at Segment 4, characterized by higher salinity and temperature, had higher relative catch of estuarine species such as Genidens genidens, Lycengraulis grossidens and Anchovia clupeoides (Fig. 6). Higher catches of three non-native species (H. littorale, M. lippincottianus and C. kelberi) and the native G. brasiliensis occurred in Segment 3 and 2, which had lower salinity and temperature and higher dissolved oxygen.
Discussion
Cichla. kelberi was barely captured in the estuarine Segment 4, with higher levels of salinity during the dry season, when the influence of seawater upstream increases and saline intrusion reaches upper sites of the São João River. Peacock basses have historically invaded lentic environments (e.g. reservoirs), but our results revealed that this fish successfully invaded the lotic stretch below Juturnaíba Dam, which includes an estuarine area. As expected, water salinity restricted the spread of C. kelberi to the lowermost stretch of the estuary and affected the structure of fish assemblages in respect to species origin (marine or freshwater; Online Resource 1). Moreover, our results showed that the invader and fish assemblages experience marked spatial and temporal variation, mostly associated to climatic seasonality, flow regulation and influence of the tides. Among the nine non-native species recorded in the São João River, C. kelberi is the only essentially piscivorous. By considering its voracious habits (e.g. Pelicice et al. 2015) that includes high rates of cannibalism (Mendonça et al. 2018), its presence may negatively affect fish diversity in the estuary. Future studies must investigate if the non-native predator affected assemblage structure, especially because other studies have reported strong predatory effects in reservoirs (Pelicice and Agostinho 2009).
Environmental influences on Cichla kelberi distribution and catch
The non-native C. kelberi successfully invaded the area downstream Juturnaíba and reached the estuarine stretch under greatest influence of salinity. The highest catches of C. kelberi in Segment 3 were associated with lower depth, a consequence of erosional processes in the channelized course, enhanced by deforested banks, tidal regime and reduced river flow. In its native range, Cichla species inhabit structured lentic environments with clear waters (Winemiller 2001), but in the São João River, the highest catches of the peacock bass occurred in the shallow and vegetated waters of lotic environments subjected to different human impacts and abiotic pressures. In reservoirs from the Upper Paraná River Basin, the occurrence of C. kelberi was associated with transparent waters and higher water temperatures (Espínola et al. 2010). We suspect that transparency was not limiting factor in our studied system, since it ranged between 0.2 to 5.4 m and on average was > 1.0 m in each segment. The higher water transparency may facilitate the success of Cichla species (Espínola et al. 2010), because they are visual piscivores, and their foraging rates should benefit from greater Secchi depth (Winemiller 2001).
Although present in the estuary, the catch of C. kelberi in Segment 4 was negligible (restricted to only two individuals). In coastal environments, salinity is the most relevant abiotic factor limiting the permanence and spread of many native and non-native freshwater fishes (Schofield et al. 2011; Vitule et al. 2013). For freshwater invaders, salinity represents an abiotic filter to the invasion process (Freire et al. 2008; Beatty and Morgan 2013; Ricciardi et al. 2013; Gutierre et al. 2014). However, cichlids are notably euryhaline; as fishes from the secondary freshwater division (sensu Myers 1937), they share an evolutionary history of marine ancestry, which may play a role in their dispersion in the study area. Among other non-native cichlids, catches were extremely low (Cichlasoma orientale) or mostly restricted to one segment (Oreochromis niloticus). Compared to these species and the other six non-natives which belong to the primary freshwater division, C. kelberi seems to be the most tolerant to salinity and therefore, able to cope with salinity for some period. Gutierre et al. (2017) named as “salt bridges” the strategy of using saline or brackish waters for limited time to invade new freshwater areas. There are few studies addressing experimentally salt tolerance in cichlids (but see Gutierre et al. 2017), but similarly to C. kelberi in the São João River, Oreochromis niloticus has successfully established in areas under salinity influence of South (São Francisco River; Brito and Magalhães 2017) and North America (coastal Mississippi watersheds; Peterson et al. 2004). Cichla kelberi may find environmental constraints to colonize Segment 4, but it may explore the area during periods of decreased salinity (i.e., rainy season, higher river flow). In estuaries with multiple drainage systems, such as the São João River, it may create a window of opportunity to reach and invade adjacent drainages.
Environmental influences on fish distribution
We observed a clear environmental gradient along the river, affected mainly by seasonality and dam regulation. Lower depth and higher water transparency characterized samples of the dry season, an expected consequence of water storage in the reservoir and thus decreased flow downstream (Alber 2002). Besides the effect of seasonality, the most downstream site, which is under direct marine influence, was segregated due to higher values of salinity. The major influence of salinity occurred in samplings in the dry season, mostly in Segment 4 and to a lesser extent in Segment 3. Even though characterized as a freshwater environment, the channelized Segment 3 is under greater influence of saline intrusion during the dry season, because flow is reduced. During the wet season, however, high rainfall prevents a major intrusion of the salt wedge even in the lowermost segment, decreasing the salinity gradient along the river.
These dynamic limnological conditions affected the spatial and temporal distribution of fish diversity. From the dam to the river mouth, zonation of fish species along the São João River was mainly influenced by salinity, due to the replacement of freshwater fishes by marine species. Especially in estuarine systems, salinity is the main factor affecting the distribution and composition of fish (Blaber 2000; Elliot et al. 2007), which creates turnover patterns along the salinity gradient (Garcia et al. 2010). Fishes living in estuaries must be able to cope with wide salinity fluctuations, being such capacity highly variable among species (Blaber 2000). Therefore, species with wide distribution in estuaries are typically those that tolerate wide variations in salinity (Elliot et al. 2007).
Fish zonation was less evident during the rainy season (Online Resource 1). The increased freshwater inflow decreased the longitudinal gradient of salinity and allowed the dispersion of freshwater species to the segment close to the sea. Hoplosternum littorale and G. brasiliensis, which belong respectively to families of the primary and secondary freshwater divisions (sensu Myers 1937), were relatively abundant in Segment 4. Despite the low catch, C. kelberi reached the proximities of the river mouth in the rainy season. Therefore, the greater amounts of freshwater discharges during the tropical rainy season may aid the dispersal of freshwater non-native species, such as the peacock bass, to adjacent basins that are connected by artificial channels, such as the Ostras and Una Rivers in the study area. Similar records of downstream expansion of freshwater species in estuaries following freshwater masses are reported elsewhere. For example, in the Patos Lagoon estuary, southern Brazil, extraordinary high freshwater flows associated to ENSO events reduce strongly the conspicuous salinity gradient of the system, allowing freshwater species to reach the estuary mouth (Garcia et al. 2004).
Catches of invasive species
The estuarine stretch of the São João River presented a relatively high species richness (80), in spite of river regulation, channelization, deforestation and the occurrence of non-native species. Tropical and subtropical estuaries are characterized by high species diversity, c.a. 100–200 species (Barletta et al. 2005; Andrade-Tubino et al. 2008; Neves et al. 2010). However, in terms of numerical representativeness, non-native species were important, corresponding to 33.4% of the total catch. Half of this amount was recorded in the channelized Segment 3. This segment was characterized by low salinity and dissolved oxygen, and higher catches of the non-natives M. lippincottianus and C. kelberi. In general, habitats disturbed by human activities are more vulnerable to non-native species (Moyle and Light 1996; Pelicice et al. 2018). In this sense, channelization and reduced flow seems to play a major role in the success of non-native species (Corbacho and Sanchéz 2001), mainly for C. kelberi, which is pre-adapted to semi-lentic to lentic environments. A similar result was found below a dam in the São Francisco River, Northeastern Brazil, where the decrease flow facilitated the establishment of invaders such as O. niloticus, Astronotus ocellatus, Cichla spp. and M. lippiconttianus (Assis et al. 2017). Considering that most non-native species recorded in the São João River were absent in the few studies previously carried out in the basin (Bizerril 1995; Jaramillo-Villa 2010; Mendonça et al. 2018), it seems reasonable to suppose that these introductions occurred in the last 20 years. In the survey conducted by Bizerril (1995), which included the stretch of São João River below the dam, no invasive species was recorded. In the Juturnaíba Reservoir, the first records of C. kelberi, O. niloticus and C. gariepinus date back 12 years (Mendonça et al. 2018). New introductions are ongoing in the basin, considering that two species (Nannostomus cf. beckfordi and Aequidens sp.) were recorded by Jaramillo-Villa (2010) in the riverine stretch upstream from Juturnaíba, although they were absent in the downstream area (this study).
The diversity and catch of non-native fish species, together with the wide distribution of the piscivorous C. kelberi and its higher catches in shallow vegetated habitats, represent additional threats to the native fishes of the São João River. These threats, together with damming, river channelization and deforestation, have caused systemic and considerable damage to the São João River ecosystem. While threats are increasingly unabated, information gathered herein might be useful to guide policies toward the conservation of native fishes in the São João River basin. Furthermore, non-native cichlids such as C. kelberi are sufficiently robust to survive and disperse across shallow mesohaline habitats where tides prevent high flows and currents to reach adjacent basins (Gutierre et al. 2014). Our results indicate that Cichla species are powerful invaders, able to colonize distinct ecosystems ranging from reservoirs to estuarine areas but are strongly influenced by salinity.
References
Agostinho A, Thomaz SM, Gomes LC (2005) Conservation of the biodiversity of Brazil’s inland waters. Conserv Biol 19(3):646–652
Agostinho AA, Gomes LC, Pelicice FM (2007) Ecologia e manejo de recursos pesqueiros em reservatórios do Brasil. Eduem, Maringá
Agostinho AA, Pelicice FM, Gomes LC (2008) Dams and the fish fauna of the Neotropical region: impacts and management related to diversity and fisheries. Braz J Biol 68(4):1119–1132
Alber MA (2002) A conceptual model of estuarine freshwater inflow management. Estuaries 25(6B):1246–1261
Alvares CA, Stape JL, Sentelhas PC, Gonçalves JLM, Sparovek G (2013) Köppen’s climate classification map for Brazil. Meteorol Z 22(6):711–728. https://doi.org/10.1127/0941-2948/2013/0507
Andrade-Tubino MF, Ribeiro ALR, Vianna M (2008) Organização espaço-temporal das ictiocenoses demersais nos ecossistemas estuarinos brasileiros: uma síntese. Oecolog Bras 12(4):640–661
Assis DAS, Dias-Filho VA, Magalhães ALB, Brito MFG (2017) Establishment of the non-native fish Metynnis lippincottianus (Cope 1870) (Characiformes: Serrasalmidae) in lower São Francisco River, northeastern Brazil. Stud Neotrop Fauna E 52(3):2–11
Barletta M, Barletta-Bergan AB, Saint-Paul U, Hubold G (2005) The role of salinity in structuring the fish assemblages in a tropical estuary. J Fish Biol 66:45–72. https://doi.org/10.1590/S1679-62252012000100011
Beatty SJ, Morgan DL (2013) Introduced freshwater fishes in a global endemic hotspot and implications of habitat and climatic change. BioInvasions Rec 2(1):1–9
Bizerril CSRF (1995) Análise da distribuição espacial da ictiofauna de uma bacia hidrográfica do Leste brasileiro. Arq Biol Tecnol 38(2):477–499
Blaber SJM (2000) Tropical estuarine fishes. Ecology, exploitation and conservation. Blackwell Science, Oxford
Blanchet FG, Legendre P, Borcard D (2008) Forward selection of explanatory variables. Ecology 89:2623–2632
Brito MFG, Magalhães ALB (2017) Brazil’s development turns river into sea. Science 358:179
Corbacho C, Sanchéz JM (2001) Patterns of species richness and introduced species in native freshwater fish faunas of a Mediterrenean-type Basin: the Guadiana River (southwest Iberian Peninsula). Regul Rivers Res Manag 17:699–707. https://doi.org/10.1002/rrr.631
Daga VD, Skóra F, Padial AA, Abilhoa V, Gubiani EA, Vitule JRS (2015) Homogenization dynamics of the fish assemblages in Neotropical reservoirs: comparing the roles of introduced species and their vectors. Hydrobiologia 746:327–347. https://doi.org/10.1007/s10750-014-2032-0
Elliot M, Whitfield AK, Potter IC, Blaber SJM, Cyrus DP, Nordlie FG, Harrison TD (2007) The guild approach to categorizing estuarine fish assemblages: a global review. Fish Fish 8:241–268
Eschmeyer WN, Fricke R (2016) Catalog of fishes: genera, species, references. https://researcharchive.calacademy.org/research/ichthyology/catalog/fishcatmain.asp). Electronic version Accessed 12 Feb 2016
Espínola LA, Minte-Vera CV, Júlio HF Jr (2010) Invasibility of reservoirs in the Paraná Basin, Brazil, to Cichla kelberi Kullander and Ferreira 2006. Biol Invas 12:1889–1889
Figueiredo JL, Menezes NA (1978) Manual de Peixes Marinhos do Sudeste do Brasil. II. Teleostei (1). Museu de Zoologia da Universidade de São Paulo, São Paulo
Figueiredo JL, Menezes NA (1980) Manual de Peixes Marinhos do Sudeste do Brasil. III. Teleostei (2). Museu de Zoologia da Universidade de São Paulo, São Paulo
Figueiredo JL, Menezes NA (2000) Manual de Peixes Marinhos do Sudeste do Brasil. VI. Teleostei (5) Museu de Zoologia da Universidade de São Paulo, São Paulo
Franco ACS, Santos LN, Petry AC, García-Berthou E (2018) Abundance of invasive peacock bass increases with water residence time of reservoirs in southeastern Brazil. Hydrobiologia 817:155–166. https://doi.org/10.1007/s10750-017-3467-x
Freire CA, Amado EM, Souza LR, Veiga MPT, Vitule JRS, Souza MM, Prodocimo V (2008) Muscle water control in crustaceans and fishes as a function of habitat, osmoregulatory capacity, and degree of euryhalinity. Comp Biochem Physiol 149:435–446. https://doi.org/10.1016/J.CBPA.2008.02.003
Garcia AM, Vieira JP, Winemiller KO (2004) Grimm AM. Comparison of 1982–1983 and 1997–1998 El Niño effects on the shallow-water fish assemblage of the Patos Lagoon Estuary (Brazil). Estuaries 27:905–914
Garcia AM, Jaureguizar AJ, Protogino LC (2010) From fresh water to the slope: fish community ecology in the Rio de la Plata and the sea beyond. Lat Am J Aquat Res 38:81–94
Gutierre SMM, Vitule JRS, Freire CA, Prodocimo V (2014) Physiological tools to predict invasiveness and spread via estuarine bridges: tolerance of Brazilian native and worldwide introduced freshwater fishes to increased salinity. Mar Freshw Res 65:425–436. https://doi.org/10.1071/MF13161
Gutierre SMM, Schulte JM, Schofield PJ, Prodocimo V (2017) Osmorregulation and muscle water control in vitro facing salinity stress of the Amazon fish Oscar Astronotus ocellatus (Cichlidae). Mar Freshw Behav Physol 50(4):303–311. https://doi.org/10.1080/10236244.2017.1387480
Havel JE, Lee CE, Vander Zanden MJ (2005) Do reservoirs facilitate invasions into landscapes? Bioscience 55:515–525
Hoeinghaus DJ, Agostinho AA, Gomes LC, Pelicice FM et al (2009) Effects of river impoundment on ecosystem services of large Tropical rivers: Embodied energy and market value of artisanal fisheries. Conserv Biol 23:1222–1231
Jackson DA (1993) Stopping rules in principal component analysis: a comparison of heuristical and statiscal approaches. Ecology 74:2204–2214
Jackman S (2020) pscl: Classes and Methods for R Developed in the Political Science Computational Laboratory. United States Studies Centre, University of Sydney, Sydney, New South Wales, Australia. R package version 1.5.5, https://github.com/atahk/pscl/
Jaramillo-Villa U (2010) Efeito da Retificação de Rios sobre as Taxocenoses de Peixes: Estudo em Rios Costeiros de Mata Atlântica no Estado do Rio de Janeiro, Brasil. Master Dissertation, Rio de Janeiro: Universidade Federal do Rio de Janeiro
Johnson PTJ, Olden JD, Vander Zanden MJ (2008) Dam invaders: impoundments facilitate biological invasions into freshwaters. Front Ecol Environ 6:357–363
Kindt R, Coe R (2005) Tree diversity analysis. A manual and software for common statistical methods for ecological and biodiversity studies. World Agroforestry Centre (ICRAF), Nairobi. ISBN 92-9059-179-X
Kullander SO, Ferreira EJG (2006) A review of the South American cichlid genus Cichla, with descriptions of nine new species (Teleostei: Cichlidae). Ichthyol Explor Freshw 17(4):289–398
Legendre P, Gallagher ED (2001) Ecologically meaningful transformations for ordination of species data. Oecologia 129(2):271–280. https://doi.org/10.1007/s004420100716
Mack RN, Simberloff D, Lonsdale WM, Evans H, Clout M, Bazzaz FA (2000) Biotic invasions: causes, epidemiology, global consequences, and control. Ecol Appl 10(3):689–710
Magellan K, García-Berthou E (2015) Influences of size and sex on invasive species aggression and native species vulnerability: a case for modern regression techniques. Rev Fish Biol Fish 25(3):537–549
Marçal M, Brierley G, Lima R (2017) Using geomorphic understanding of catchment-scale process relationships to support the management of river futures: Macaé Basin, Brazil. Appl Geogr 84:23–41
Mendonça HS, Santos ACA, Martins MM, Araújo FG (2018) Size-related and seasonal changes in the diet of the non-native Cichla kelberi Kullander & Ferreira, 2006 in a lowland reservoir in the southeastern Brazil. Biota Neotrop 18(3):e20170493. https://doi.org/10.1590/10.1590/1676V0611VBNV2017V0493&
Menezes NA, Figueiredo JL (1980) Manual de Peixes Marinhos do Sudeste do Brasil. IV. Teleostei (3). Museu de Zoologia da Universidade de São Paulo, São Paulo
Menezes NA, Figueiredo JL (1985) Manual de Peixes Marinhos do Sudeste do Brasil. V. Teleostei (4). Museu de Zoologia da Universidade de São Paulo, São Paulo
Moyle PB, Light T (1996) Fish invasions in California: do abiotic factors determine success. Ecology 77:1666–1670
Myers GS (1937) Fresh-water fishes and West indian zoogeography. Rep Smithsonian Instn 92:339–364
Neves LM, Teixeira TP, Araújo FG (2010) Structure and dynamics of distinct fish assemblages in three reaches (upper, middle and lower) of an open tropical estuary in Brazil. Mar Ecol 32:115–131. https://doi.org/10.1111/j.1439-0485.2010.00407.x
Oksanen JF, Blanchet G, Friendly M, Kindt R et al 2019. vegan: Community Ecology Package. R package version 2.5-6. Available from: https://CRAN.R-project.org/package=vegan
Pelicice FM, Agostinho AA (2009) Fish fauna destruction after the introduction of a non-native predator (Cichla kelberi) in a Neotropical reservoir. Biol Invasions 11:1789–1801
Pelicice FM, Latini JD, Agostinho AA (2015) Fish fauna disassembly after the introduction of a voracious predator: main drivers and the role of the invader’s demography. Hydrobiologia 746:271–283. https://doi.org/10.1007/s10750-014-1911-8
Pelicice FM, Azevedo-Santos VM, Vitule JRS, Orsi ML et al (2017) Neotropical freshwater fishes imperilled by unsustainable policies. Fish Fish 18:1119–1133
Pelicice FM, Azevedo-Santos VM, Esguícero ALH, Agostinho AA, Arcifa MS (2018) Fish diversity in the cascade of reservoirs along the Paranapanema River, southeast Brazil. Neotrop Ichthyol 16(2):e170150
Peterson MS, Slack WT, Brown-Peterson N (2004) McDonald JL (2004) Reproduction in nonnative environments: establishment of Nile tilapia, Oreochromis niloticus, in Coastal Mississipi Watersheds. Copeia 4:842–849
Petesse ML, Petrere M Jr (2012) Tendency towards homogenization in fish assemblages in the cascade reservoir system of the Tietê river basin, Brazil. Ecol Eng 48:109–116
Poff NL, Olden JD, Merritt DM, Pepin DM (2007) Homogenization of regional river dynamics by dams and global biodiversity implications. PNAS 104(14):5732–5737
R Core Team (2020) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/
Ricciardi A, Hoopes MF, Marchetti MP, Lockwood JL (2013) Progress toward understanding the ecological impacts of nonnative species. Ecol Monog 83:263–282. https://doi.org/10.1890/13-0183.1
Schofield PJ, Peterson MS, Lowe MR, Brown-Peterson NJ, Slack WT (2011) Survival, growth and reproduction of nonindigenous Nile tilapia, Oreochromis niloticus (Linnaeus 1758). I. Physiological capabilities in various temperatures and salinities. Mar Freshw Res 62:439–449. https://doi.org/10.1071/MF10207
Simberloff D, Martin J-L, Genovesi P, Maris V, Wardle DA, Aronson J, Courchamp F, Galil B, García-Berthou E, Pascal M, Pyšek P, Sousa R, Tabacchi E, Vilà M (2013) Impacts of biological invasions—what’s what and the way forward. Trends Ecol Evol 28:58–66
Sofiati A (2010) Macaé em quatro tempos. Herculano S, Correa HD (org) Oficina sobre Impactos Sociais, Ambientais e Urbanos das Atividades Petrolíferas: o caso de Macaé (RJ). UFF, Niterói, pp 130–148
Vitule JRS, Skóra F, Abilhoa V (2012) Homogenization of freshwater fish faunas after the elimination of a natural barrier by a dam in neotropics. Divers Distrib 18:111–120
Vitule JRS, Gazola da Silva FF, Bornatowski H, Abilhoa V (2013) Feeding ecology of fish in a coastal river of the Atlantic Rain Forest. Environ Biol Fish 96:1029–1044. https://doi.org/10.1007/S10641-012-0101-7
Winemiller KO (2001) Ecology of peacock cichlids (Cichla spp.) in Venezuela. J Aquaric Aquat Sci 9:93–112
Acknowledgements
The authors are grateful for the partnership made in the field work with Erica Pellegrini and Vagner dos Santos. We also thank students of the Laboratório de Ecologia de Peixes and Laboratório Integrado de Zoologia of the Universidade Federal do Rio de Janeiro for help in the samplings and laboratory work. The authors thank Arthur Bauer for the map and two anonymous reviewers for helpful comments. This study was financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior—Brasil (CAPES)—Finance Code 001. P. A. Catelani also thanks CAPES for the sandwich doctorate scholarship (CSF-PVE-S 88887.127845/2016-00). Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) provided research grants for F. M. Pelicice. E.G.B. was financially supported by the Spanish Ministry of Science and Innovation (projects CGL2016‐80820-R, PCIN-2016-168, and RED2018‐102571‐T) and the Government of Catalonia (ref. 2017 SGR 548). The comments and suggestions made by the Reviewers and the Editor helped to improve earlier versions of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Catelani, P.A., Petry, A.C., Pelicice, F.M. et al. When a freshwater invader meets the estuary: the peacock bass and fish assemblages in the São João River, Brazil. Biol Invasions 23, 167–179 (2021). https://doi.org/10.1007/s10530-020-02363-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10530-020-02363-w