Abstract
Predicting the spread of invasive species in a warming world calls for a better understanding of the mechanisms behind the relative performance of both invasive and indigenous species as recipient communities can offer biotic resistance. A particularly important functional trait in this context is reproductive potential. The Mediterranean mussel Mytilus galloprovincialis is invasive world-wide and threatens the native brown mussel Perna perna on South African rocky shores. Where they co-exist, Mytilus dominates the high and Perna the low mussel zone, overlapping in the middle. Mytilus generally exhibits high individual reproductive output, but feeding time is reduced, physical conditions are stressful and adult densities are lower on the higher shore. We compared overall reproductive potential of populations of the two species using a 1-year data set for three sites, accounting for within-shore distribution and abundances. Despite the restriction of Mytilus to the more stressful upper-shore, its aggregate reproductive potential was comparable to that of Perna. We then used Dynamic Energy Budget modelling to test the effects of changing conditions by calculating aggregate reproductive potential under temperature conditions predicted for the end of the century by the International Panel on Climate Change. The results suggest increased aggregate reproductive potential for both species, but the effect was stronger for the native species at two of three sites, implying increased biotic resistance by Perna to the further spread of Mytilus. Combining ecological context and physiological performance elucidates how interactions between native and invasive species may alter. Such nuances are fundamental to anticipating winners or losers in a world where distribution shifts are increasingly common.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Invasive species are a global threat to biodiversity that can negatively affect the structure and function of natural and managed communities (García Molinos et al. 2015). They do, however, face two primary problems: inoculation of suitable new habitats and biotic resistance (Mellin et al. 2016). While many studies have confirmed the relevance of species interactions in determining the ecological success of invasive species, predictions concerning the further spread of established invasives are difficult in part because of uncertainties associated with temporal and spatial variability in the mediating role of positive and negative interactions that take place within the recipient communities (Bruno et al. 2003; Mellin et al. 2016; Nicastro et al. 2012). Climate change further complicates the issue through its effects on biological interactions as both native and invasive species face unprecedented conditions that may either favour or hamper their physiological performance (Bates et al. 2013), potentially leading to alterations to competitive hierarchies and cryptic indirect effects (Russell et al. 2011).
The interactive effects of species interactions and a changing climate necessarily manifest as non-linear dynamics, which can only be resolved using mechanistic frameworks (Helmuth et al. 2005; Monaco and Helmuth 2011; Olabarria et al. 2016). A focus on species-specific physiological responses to environmental variability provides a means to disentangle the underlying processes leading to such nonlinearities, anticipate which species may be winners or losers in a rapidly changing world, and better predict ecological tipping points (Harley et al. 2017; Pfister et al. 2014; Somero 2011).
The ecological success of invasive species in colonizing new regions is often attributed to their higher rates of growth and reproduction, relative to functionally equivalent native species in the receiving communities. The relative difference in performance between local and invasive species is also strongly dependent on the prevailing environmental conditions (Branch and Steffani 2004). Several comparative studies have examined the relative physiological performance of interacting native and invasive species across geographic-scale environmental gradients (e.g. Bates et al. 2013). However, environmental drivers often show greater variability at local than regional scales due to non-climatic factors like habitat complexity or, in aquatic systems, small scale hydrodynamics. Less is known about how fine-scale local aspects can mediate the importance of physiological traits to the performance of invasive species, although a growing number of studies are demonstrating the relevance of local ecological processes to the establishment of invasive species (Schneider et al. 2010; Shinen and Morgan 2009; Suchanek 1981). The body temperature of ectothermic organisms is a prime example of this. Intertidal mussels and limpets distributed across large biogeographic scales exhibit thermal mosaics instead of the expected gradual increase in body temperatures towards the equator due to factors like the timing of low tide (Helmuth et al. 2006) or the aspect of the rocks they occupy (Seabra et al. 2011). While habitat-level factors are important in driving realized physical conditions of individuals, their influence on physiological performance is ultimately modulated by the organism’s biology. For instance, material properties of ectotherms can result in different body temperatures between co-ocurring species, such as predatory seastars and their mussel prey (Broitman et al. 2009). At even finer scales, epibiosis or changes in shell albedo due to parasitic epilithic cyanobacteria can directly affect the thermal conditions experienced at the individual level (Wahl 2008; Zardi et al. 2016). Finally, contrasting thermal sensitivities between species suggest that, even at the same realized body temperature, organisms may be experiencing the environment differently (Freitas et al. 2007; Monaco et al. 2016). Thus, predicting relative performance of interacting species, such as invasive and indigenous organisms, requires careful consideration of the local context, both in terms of the physical conditions as actually experienced and the sensitivity of the species to those drivers (Harley et al. 2017; Monaco and Helmuth 2011).
Here we examine differences in aggregate reproductive potential (Phillips 2007) (ARP) between two rocky shore mussel species presently found along the southern coast of South Africa: the globally invasive Mediterranean mussel, Mytilus galloprovincialis, (hereafter Mytilus) and the native brown mussel, Perna perna (hereafter Perna) (McQuaid et al. 2015). Previous studies showed that under equal conditions, individual energy investment into growth and reproduction is significantly greater for Mytilus than Perna (Van Erkom Schurink and Griffiths 1991; Van Erkom and Griffiths 1992; Zardi et al. 2007), granting the former a presumed invasive reproductive advantage. The two species co-exist on the south coast of South Africa, but the steep, tidally-driven gradients in physical stressors on rocky shores contribute directly and indirectly to partial vertical segregation of the two (Bownes and McQuaid 2006). The better tolerance to desiccation of Mytilus (Nicastro et al. 2010b) and the stronger byssal attachment of Perna (Zardi et al. 2006) lead to complex interactions of facilitation and competition (Rius and McQuaid 2006, 2009) that, in combination with differential zone-dependent recruitment success (Bownes and McQuaid 2009), result in domination of the high-mussel zone by Mytilus and of the low mussel zone by Perna with overlap and co-existence in the middle (Bownes and McQuaid 2009; Zardi et al. 2006). This segregation is disadvantageous for individuals of the invasive species, as higher shore distribution means shorter feeding times and higher thermal/desiccation stress (Dehnel 1956; Roberts et al. 1997; Widdows and Shick 1985). However, higher individual investment in reproduction by Mytilus should yield higher ARP. We use analyses of present day ARP data collected in situ, and simulations computed using Dynamic Energy Budget (DEB) models (Kooijman 2010), specifically built to account for local-scale environmental variability, to examine how the warmer temperatures predicted under climate change will affect ARP of the two species and thus the strength of biotic resistance offered by Perna to the future spread of Mytilus.
We first hypothesized that, despite the suboptimal environmental conditions experienced by Mytilus on the higher shore, its high abundance and intrinsically greater energy investment into reproduction would yield higher ARP than Perna. We tested this using data on individual reproductive output, complemented with observations of size-frequency and percent cover of mussels at three shore-levels and three sites. Second, using DEB models parameterized for local populations of both species (Monaco and McQuaid 2018), we tested the hypothesis that increases in air and sea-surface temperature due to climate change would exacerbate the observed reproductive advantage of the invasive species.
Materials and methods
Field sites
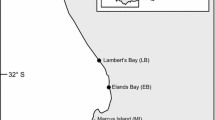
Both Perna and Mytilus can be found on exposed rocky shores along the south coast of South Africa (Fig. 1), but because of local topography, mussel vertical segregation is not always clear. Here we targeted sites that exhibited clear partial vertical segregation, with Mytilus and Perna dominating the high- and low-shore respectively, and both sharing the mid-shore (Bownes and McQuaid 2006). The three study sites were Brenton-on-sea (34°04′31.39″ S 23°01′25.32″ E), Plettenberg Bay (34°03′41.72″ S 23°22′47.16″ E), and Keurboomstrand (34°00′17.87″ S 23°27′19.16″ E) (Fig. 1). All sites experience moderate wave-exposure, however, Plettenberg Bay is in the lee of a headland and is likely to experience lower wave action than Brenton-on-sea and Keurboomstrand (von der Meden et al. 2008). As for most rocky outcrops on this coast, sandy beaches surround the three sites, with seasonal dynamics in sand accretion often disturbing mussel assemblages (Zardi et al. 2007).
Reproductive potential
We calculated ARP using data on individual reproductive output, and population data on percent cover and size-frequency distribution following Phillips (2007).
Individual reproductive output To calculate individual reproductive output we collected 30 Mytilus from the high-shore, 30 Mytilus from the mid-shore, 30 Perna from the mid-shore, and 30 Perna from the low-shore. We targeted mussels in the size range between 30 mm shell length (i.e. reproductively mature) and the largest individual found on that level, trying to maintain an even size-frequency distribution. Individuals were preserved in 70% ethanol for later analysis. In the laboratory we measured mussel shell length (0.01 mm resolution) and dissected gonadic/somatic tissues. Tissues were dried for 48 h at 60 °C, and weighed to the nearest 0.01 mg (Metler Toledo MS105DU, OH, USA). For each species and shore level we established linear relationships between (loge-transformed) gonad dry weight and shell length. This protocol was repeated for 8 sampling dates per site between October 2015 and October 2016 (Table S1).
Size-frequency distribution To quantify size-frequency distribution we collected all mussels within three 0.15 × 0.15 m quadrats placed at each shore level semi-randomly (i.e. randomly within patches of 100% mussel cover), and recorded the shell length (0.01 mm resolution) of all animals ≥ 30 mm. This was done only once (October 2015) during the sampling period because, unless catastrophic events take place (e.g. strong storms or sand inundation), the size-frequencies of mature mussels are not expected to vary markedly over a year (e.g., Reaugh-Flower et al. 2011). While this assumption cannot be generalized across all sites (Griffiths 1981) or over longer time periods (Reaugh-Flower et al. 2011), it allowed us to minimize damage to mussel beds, and to simplify the interpretation of ARP results. Temporal variability in mussel population size was assessed in terms of percent cover, which in this region is surprisingly stable over periods of 1–5 years (Reaugh-Flower et al. 2011).
Mussel percent cover We used a digital camera (Panasonic, DMC-TS20) to photograph 20 quadrats (0.2 × 0.2 m) per shore-level, placed non-randomly 1 m apart parallel to the coastline. Using ImageJ (Schneider et al. 2012), we overlaid a grid with 49 equidistant points, under which presence/absence was determined. For each shore level and site, percent mussel cover was calculated three times over the course of the study: October 2015, June 2016, and October 2016. Because mussel beds along this entire coast, including all our sites, are mono-layered, our calculations of percent cover represent a relevant proxy for mussel biomass.
Aggregate reproductive potential calculations We calculated ARP for Perna and Mytilus across shore levels and sites. First, all adult mussels found in the 3 size frequency distribution quadrats were grouped together by species. Second, the reproductive output linear models were applied to each of these individuals to calculate ARP for 100% cover mussel beds. This was done for each of the sampling dates for individual reproductive output, giving a total of 8 times (Fig. S1, Table S1). Data from all sampling dates were added to obtain an annual estimate of ARP (g of gonad dry weight) for each species, site, and shore level. Thirdly, we divided these data by the area of our size-frequency distribution quadrats to express the results per m2. Percent cover data allowed us to normalise the reproductive potential by the area actually utilized by mussels. The cover data collected in Oct-2015 were used to normalise the reproductive potential data between Oct-2015 and Jan-2016; cover data from Jun-2016 were used for reproductive data between Mar-2016 and Jul-2016; and Oct-2016 cover data for reproductive data between Sept-2016 and Oct-2016. The 20 mussel cover images taken served as replicates to test for the effects of species and shore level on ARP statistically.
Dynamic energy budget (DEB) model simulations
To explore possible effects of climate change on the physiological performance of individual mussels, we used DEB models as these can estimate quantitatively the energy budget dynamics of individuals experiencing fluctuating environmental conditions. For detailed descriptions of DEB theory and application see Kooijman (2010) and Monaco et al. (2014). Here we used previously parameterized DEB models, which can incorporate the effects of alternating periods of aerial exposure and submergence on physiological responses of Mytilus and Perna (Monaco and McQuaid 2018). While DEB models can address the effects of multiple environmental drivers, including temperature (Sarà et al. 2011), food availability (Saraiva et al. 2011), or ocean acidification (Jager et al. 2016), here we only focused on the influence of changes in body temperature on the ARP of mussels, as the evidence suggests this variable exerts the greatest physiological effects on mussels experiencing climate change (Gazeau et al. 2014; Kroeker et al. 2014). Additionally, we are still uncertain about the accuracy in climate change projections of ocean acidification and productivity, particularly in coastal waters with complex biogeochemical processes (Duarte et al. 2013). We ran simulations for populations at all three sites, assuming their size-frequency distributions and percent cover would be maintained to the end of this century.
To run model simulations under current conditions we used data on temperature, food, and tide height (available in Monaco and McQuaid 2018). Briefly outlined here, mussel body temperature data were collected with biomimetic temperature loggers, or robomussels (Fitzhenry et al. 2004; Lathlean et al. 2017), set to log every 30 min at 0.06 °C resolution. Species-specific robomussels were deployed on the shore according to natural habitat partitioning observed at those sites (von der Meden et al. 2008): i.e. Perna-low, Perna-mid, Mytilus-mid, and Mytilus-high. Thus, DEB simulations were conducted for each species only at the shore levels where they predominantly occur. Periods of submergence and aerial exposure were traced using our robomussel temperature data and tidal height predictions retrieved from XTide software (http://www.flaterco.com/xtide/) (Harley and Helmuth 2003; Lathlean et al. 2011; Monaco and McQuaid 2018). During submergence, chlorophyll-a concentration was considered to represent mussel food. As it was logistically impossible to collect these data in situ, estimates were obtained from daily Aqua/MODIS (Moderate Resolution Imaging Spectroradiametor) satellite images using Ocean Color’s web interface. We extracted 9 pixels of 4 km2, centred 15 km offshore from each study site.
To elucidate possible effects of climate change on intertidal mussels, we ran three additional DEB model simulations considering predicted increases in (1) air, (2) seawater, and (3) air and seawater temperatures. Based on IPCC predictions for this region, by the end of this century a business as usual emissions scenario (Representative Concentration Pathway 8.5, RPC8.5) would lead to increases in average air and seawater temperatures of 3.5 and 2.1 °C, respectively (IPCC 2014).
Data analyses
All data were processed and analysed using R version 3.3.2 (R Core Team 2016). Data on individual reproductive output (i.e. loge[x + 1] gonad dry weight as a function of shell length, Table S1) were analysed using a series of analyses of covariance (ANCOVA). First, for each site we examined whether date of collection influenced overall reproductive output. This was done using linear models testing the effects of species, collection date, and the co-variable shell length on gonad dry weight. Given that gonad dry weight almost invariably scaled with shell length (Table S1), a significant interaction between length and collection date would indicate a statistical effect of time on reproductive output. Secondly, for each site and sampling date we tested the effect of the fixed factor species/shore level on reproductive output. We combined species and shore level into a single factor because the natural vertical distribution of mussels does not allow for an orthogonal design.
Differences in size-frequency distribution between species were assessed via two-sample Kolmogorov–Smirnov tests, using the ks.test function in R. To compare species composition across shore levels and sites, we calculated the percentage of all mature mussels (i.e. > 30 mm shell length) formed by each species. Patterns in mussel percent cover among sites and shore levels, as well as variability across time, were examined based on mean ± SE. Because ARPs were calculated for Mytilus and Perna from different shore levels, the sampling design was non-orthogonal and the data exceedingly non-parametric. Thus, for each site we used Mann–Whitney Wilcoxon tests to compare ARPs between species (fixed factor), and Kruskal–Wallis tests to further examine differences between species/shore levels (fixed factor). Post hoc multiple comparisons were done with a Conover-Iman test using the R package conover.test (Dinno 2017).
To explore a possible effect of climate change on the gonad output of both species, we first computed the expected changes in ARP relative to predictions under current conditions. We then contrasted this relative change between Mytilus and Perna to determine which species would be more affected by the warmer temperatures predicted under climate change. The script for the DEB models used here, including environmental drivers data, is available at: github.com/cristianmonaco/DEB-model-application-Mussel-zonation-and-reproductive-potential.
Results
Individual reproductive output
Gonad dry weight increased with shell length (Fig. 2, Fig. S1, Table S1), the relationship fluctuating over time at every site (shell length*collection date interaction; Brenton-on-sea: F(7881) = 7.66, MSE = 1.99, p < 0.001; Plettenberg Bay: F(7896) = 3.90, MSE = 1.13, p < 0.001; Keurboomstrand: F(7896) = 3.25, MSE = 0.79, p = 0.002). The dynamics in reproductive output differed depending on shore level and species, likely as a consequence of cyclic accumulation of gametes and spawning events. Mid-shore Mytilus invariably exhibited the highest size-specific reproductive output. High-shore Mytilus usually followed, with mid- and low-shore Perna showing lower gonad mass (Fig. 2a). Because mussel size range differed between shore levels and species, these results are valid at the individual level, but ignore aspects of population structure that can only be captured using data on mussel cover and size-frequency distribution.
Linear regressions of individual reproductive output (gonad dry weight, mg) as a function of shell length (mm) estimated for each site and shore level. Site-specific data from all collection dates are collapsed (for extended version see Fig. S1). Shaded areas denote standard errors. Variables are natural logarithm + 1 transformed
Population structure
Size-frequency distribution: At every site, the mussels were larger towards the low-shore (Fig. 3). Mytilus was more abundant than Perna on the high-shore, where mussel densities were higher than at mid- and low-shore levels; however, few of these individuals were of reproductive size. Perna dominated the low-shore; except at Keurboomstrand where the proportion of low-shore Mytilus was higher than at the other sites (Fig. 3g–i). High-shore Keurboomstrand showed many Perna; however, very few of these were adults (Fig. 3c). Both species shared the mid-shore at every site, with Perna slightly more prevalent at Brenton-on-sea and Keurboomstrand and Mytilus more numerous at Plettenberg Bay (Fig. 3d–f).
Size-frequency distributions of Mytilus and Perna sampled across the shore (high-, mid-, and low-shore levels) and study sites (Brenotn-on-sea, Plettenberg Bay, and Keurboomstrand). Data include animals collected from three 15 × 15 cm quadrats, sampled in October 2015. Histogram bins are 2 mm wide. Vertical dashed lines mark the size at maturity, 30 mm shell length. On each panel, we show the percentage of Mytilus and Perna bigger than 30 mm, and the Kolmogorov–Smirnov p value comparing their distributions
Mussel cover: Percent mussel cover varied with shore level, sampling date and site (Fig. 4). Brenton-on-sea showed temporal variability, mostly driven by dynamics on the mid- and high-shore. Cover at Plettenberg Bay was more stable across time and shore levels. Keurboomstrand exhibited relatively high initial mussel cover, which declined towards winter (Fig. 4), following mortality due to sand inundation (Monaco, personal observation).
Aggregate reproductive potential
We found no significant differences in mean ARPs between species at any site (Mann–Whitney Wilcoxon test: Brenton-on-sea, W = 754, p = 0.66; Plettenberg Bay, W = 648, p = 0.15; Keurboomstrand, W = 744, p = 0.60) (Fig. 5). However, when arranged by species/shore level, significant effects were detected, with both species showing a decrease from lower to higher zones (Kruskal–Wallis test: p < 0.001 at every site). Most of the variability was explained by Mytilus, which consistently showed the highest and lowest ARP, for mid- and high-shore animals, respectively. Perna exhibited higher ARP on the low- than the mid-shore. Perna low-shore ARP was lower than Mytilus mid-shore at Brenton-on-sea and Keurboomstrand, but comparable at Plettenberg Bay. This was due to reduced ARP for mid-shore Mytilus from Plettenberg Bay relative to the other two sites (Fig. 5).
Aggregate reproductive potential (ARP) (g gonad dry weight m−2) calculated for low- and mid-shore Perna, and mid- and high-shore Mytilus at each study site (Brenton-on-sea, Plettenberg Bay, and Keurboomstrand). Boxes show the 25th and 75th percentiles, and horizontal lines the medians. The whiskers illustrate the extreme values that are within 1.5 times the interquartile range. Points represent outliers. Different letters above the boxplots indicate significant differences among species/shore levels based on Kruskal–Wallis test and Conover-Iman post hoc analysis
Model predictions under current and climate change conditions
DEB model predictions of mussel gonad annual production varied with site, species, shore level, and climate change scenario (Fig. 6). Under current conditions, the models matched our observed ARP partially. Like our observations, the models suggested that Perna ARP decreases from low- to mid-shore, and that of Mytilus decreases from mid- to high-shore (Figs. 5, 6). Contrary to our empirical observations, however, the models indicated that low-shore Perna, not mid-shore Mytilus, produced the most gonad material. Furthermore, the models predicted an overall higher ARP for Perna than Mytilus (Fig. 6). The relative difference in ARP between species differed across sites, being greatest at Brenton-on-sea, followed by Plettenberg Bay, and Keurboomstrand (Fig. 6a–c). On the mid-shore, Mytilus showed higher ARP than Perna at Brenton-on-sea and Keurboomstrand, but not Plettenberg Bay (Fig. 6)
Dynamic Energy Budget model estimated dynamics in aggregate reproductive potential (ARP) for Mytilus and Perna for low- and mid-shore Perna, and mid- and high-shore Mytilus at each study site (Brenton-on-sea, Plettenberg Bay, and Keurboomstrand). Abrupt changes in the accumulation of aggregate reproductive potential are explained by variability in percent mussel cover (Fig. 4). RCP = Representative Concentration Pathway (IPCC 2014)
In general, the models indicated that climate warming would favour increased annual ARP for both species. This tendency is mostly explained by the effect of warmer seawater temperatures. The effect of warmer air temperature alone leads to an annual gonad production that is practically indistinguishable from current conditions (Fig. 6). Because of the predominant influence of seawater temperature on mussel reproduction, and given that Perna spends more time submerged than Mytilus, the relative increase in annual ARP is expected to benefit the former slightly more than the later at Plettenberg Bay (Perna = 36.21 vs. Mytilus = 35.31%) and Keurboomstrand (Perna = 36.97 vs. Mytilus = 32.00%), but not Brenton-on-sea (Perna = 34.14 vs. Mytilus = 34.94%).
Climate change may also decrease annual gonad production, albeit marginally. Specifically, mid-shore Mytilus at all sites showed a slightly higher ARP under current conditions than under expected warmer air temperatures. This occurs when body temperatures exceed the peak of their thermal performance curves (Fig. S2), which is especially likely during warm low-tide periods.
Discussion
The establishment and spread of invasive species ultimately depend on the match between their physiological limits and environmental conditions (Mellin et al. 2016; Van Erkom and Griffiths 1992), though this can be strongly mediated by both biotic interactions and ongoing climate change (Bruno et al. 2003; Levine et al. 2004; Mellin et al. 2016; Nicastro et al. 2012). To more accurately forecast the ecological condition of natural systems in the future, conservation planners and resource managers require mechanistic models that can predict the fitness not only of the invasive organism (Helmuth et al. 2005; Monaco and Helmuth 2011), but also of the species with which it interacts. Here we show that despite the superior reproductive capacity of a globally invasive species, the interaction between local biotic resistance and physical drivers can limit its spread under current and future climate change scenarios.
Reproductive potential
High reproductive output is a characteristic attribute of many successful invaders, including Mytilus galloprovincialis (Van Erkom Schurink and Griffiths 1991) which has become a conspicuous rocky shore resident on every continent except Antarctica. On the south coast of South Africa, however, a complex interplay between sensitivity to physical drivers (i.e. wave action, temperature, desiccation) and competition with the native mussel Perna perna (Rius and McQuaid 2006, 2009; Zardi et al. 2006), has constrained Mytilus to a subset of its fundamental niche space (Bownes and McQuaid 2009). Our assessments confirm that individual Mytilus are more fecund than Perna (Van Erkom Schurink and Griffiths 1991; Van Erkom and Griffiths 1992; Zardi et al. 2007). This individual reproductive advantage does not necessarily manifest at the population level, however, because Mytilus is constrained to higher shore levels. In fact, calculations of ARP, which consider observed mussel abundance and distribution (Phillips 2007), indicated no reproductive advantage for Mytilus at the three sites studied. Most research comparing the responses of native and invasive species to intertidal gradients (e.g. Kennedy 1976; Petes et al. 2007; Suchanek 1981) has focused on standardized individuals (i.e. same body size and habitat), thus ignoring both the realized conditions that organisms actually experience in a heterogeneous environment and, critically, the population level parameters that contribute to the relative performance of interacting species.
Large scale habitat heterogeneity is responsible for important variability in the reproductive output of benthic marine invertebrates (Leslie et al. 2005; Lester et al. 2007; Phillips 2007; Sanford and Menge 2007) and, in mussels, reproductive output is linked to variability in primary production and wave exposure, which affect them directly and mediate the interaction strength between species (Dahlhoff and Menge 1996; Nicastro et al. 2010a; Petes et al. 2007). These effects have also been evaluated at smaller within-shore scales, where food availability and wave action decrease predictably from low- to high-shore (Widdows and Shick 1985) and experimental alterations to hydrodynamics have important effects even at centimetre scales (McQuaid and Mostert 2010). Consistent with previous studies comparing growth between these species (Rius and McQuaid 2009), we found that within-shore environmental gradients favour Perna over Mytilus, to a degree where the reproductive advantage of Mytilus is overridden and their ARPs are comparable.
Mytilus dominates the entire rocky intertidal zone in the cool temperate bioregion on the west coast where it has outcompeted the indigenous mussels Choromytilus meridionalis and Aulocomya ater largely through its higher reproductive output (Hockey and van Erkom 1992; Robinson et al. 2005; Van Erkom Schurink and Griffiths 1991; Van Erkom and Griffiths 1992). Perna does not naturally occur on the west coast, but is native to the warm temperate south coast where it constrains Mytilus to higher shore levels through competition for space (Robinson et al. 2005; Rius and McQuaid 2006, 2009). Our observations indicate that the higher reproductive performance of Perna than the west coast species may be an important mechanism of invasion resistance on the south coast (see also Bownes and McQuaid 2009). Lagrangian simulations indicate that Mytilus larvae could disperse further to the north-east than its present distributional limit (Assis et al. 2015), indicating that this is set by other factors. Thus, predicting the potential for invasive spread of species requires estimates of both the capacity for dispersal and biotic resistance (Connolly and Roughgarden 1999; McQuaid and Phillips 2000; Shinen and Morgan 2009).
Predictions under current and predicted conditions
Our models indicate that both Mytilus and Perna will increase their reproductive potential under climate change, which would increase the abundances of native and invasive mussels in the region. This was expected given the typically positive relationship between gonad output and temperature in marine invertebrates (Gosling 2003; Shephard et al. 2010; Valdizan et al. 2011), but we could also disentangle the effects of increased air and seawater temperatures. Accounting for the different responses of animals to air and water such as metabolic depression and suspension of feeding in air (Marshall et al. 2011; Storey and Storey 1990; Tagliarolo and McQuaid 2015) allows better characterization of physiological performance in dynamic models (Monaco and McQuaid 2018; Sarà et al. 2011; Seabra et al. 2016). Importantly, we found that seawater temperature had a much stronger influence on reproductive output than air temperature. This is in line with the finding that low seawater temperatures inhibit reproduction of Perna in the Benguela upwelling system, despite high food availability there (Tagliarolo et al. 2016).
As ecological generalists, invasive species are expected to cope with a wider range of temperatures than indigenous species (Bates et al. 2013). Indeed, comparative studies indicate that rising temperatures favour the reproduction and growth of invasive species, often to the detriment of indigenous species (Lockwood and Somero 2011; Walther et al. 2009), as has been suggested for Mytilus in South Africa (Van Erkom Schurink and Griffiths 1991). Our models do not support this general prediction, as increased temperatures are expected to increase the ARP of Perna more than Mytilus, possibly because of their different thermal sensitivities as this ultimately controls the effects of temperature on energy allocation (Monaco and McQuaid 2018; Sarà et al. 2011). The main difference in thermal sensitivity emerges during aerial exposure because Mytilus undergoes metabolic depression in air more readily than Perna (Tagliarolo and McQuaid 2015), as incorporated into our models (see Fig. S2 and Monaco and McQuaid 2018).
Our approach to predicting responses to environmental change differs from earlier work which often used direct manipulations in the field or laboratory to test hypotheses concerning inter- and intra-specific competition, and the role of environmental stressors (e.g., Rius and McQuaid 2006, 2009; Petes et al. 2007). Logistical constraints often limit the scope and resolution of such studies, while individual based models that quantify physiological performance as a function of environmental drivers can circumvent some of these limitations (Monaco et al. 2016; Porter et al. 1975). DEB is a particularly powerful alternative as it can estimate various fitness-related traits, including not only reproduction but also maintenance and growth (Kooijman 2010; Monaco and McQuaid 2018; Monaco et al. 2014).
Assuming constant effectiveness of larval delivery and post-settlement mortality rates, our results suggest that the propagule pressure of both Perna and Mytilus will be enhanced by climate change, so that we could predict that they will provide increased mussel biogenic habitat by the end of this century, especially as IPCC temperature predictions for the region are not expected to exceed the physiological limits of these species (Tagliarolo and McQuaid 2015). Intertidal mussels can buffer extreme temperatures (Jurgens and Gaylord 2018; Suchanek 1992), so we expect that their role as thermal refugia that maintain rich infaunal communities (Lathlean and McQuaid 2017) will be enhanced.
Our analysis ignores potential sources of error that could lead to disagreements between observations and predictions, depending on ecological context. For example, our models predicted higher ARP for mid-shore Perna than Mytilus, contradicting our observations from field-collected animals. This can probably be explained by the models’ inability to account for factors such as species interaction, both negative (competition and parasitism) and positive (facilitation), which can influence energy allocation (Nicastro et al. 2012; Zardi et al. 2007). The models also cannot accommodate changes to reproductive biology. For instance, mussels can switch from annual to semi-annual spawning, depending on temperature (Oyarzún et al. 2018). Finally, thermal stress can affect not only gonad production, but also the quality of gametes (Boni et al. 2016). Here we have ignored these nuances to simplify model runs and because the mechanisms are still poorly resolved (Monaco and McQuaid 2018), but they are certainly areas that need further attention.
Conclusions
Understanding the interplay between physiological responses to environmental drivers and species interactions is paramount to correctly anticipating community responses to climate change. This is made more complex because the reshuffling of the distributional ranges of an increasing number of species across latitudinal and altitudinal gradients (García Molinos et al. 2015; Pecl et al. 2017) will result in novel combinations of species and interactions. Our findings suggest that species-specific sensitivity can influence reproductive success and thus indirectly the outcome of species interactions in a warmer climate. Energy budget models that can account for sublethal effects on species fitness are powerful candidates for anticipating of the ecological consequences of climate change.
References
Assis J, Zupan M, Nicastro KR et al (2015) Oceanographic conditions limit the spread of a marine invader along Southern African shores. PLoS ONE 10:e0128124
Bates AE, McKelvie CM, Sorte CJB et al (2013) Geographical range, heat tolerance and invasion success in aquatic species. Proc R Soc B Biol Sci. https://doi.org/10.1098/rspb.2013.1958
Boni R, Gallo A, Montanino M et al (2016) Dynamic changes in the sperm quality of Mytilus galloprovincialis under continuous thermal stress. Mol Reprod Dev 83:162–173
Bownes SJ, McQuaid CD (2006) Will the invasive mussel Mytilus galloprovincialis Lamarck replace the indigenous Perna perna L. on the south coast of South Africa? J Exp Mar Biol Ecol 338:140–151
Bownes SJ, McQuaid CD (2009) Mechanisms of habitat segregation between an invasive and an indigenous mussel: settlement, post-settlement mortality and recruitment. Mar Biol 156:991–1006
Branch GM, Steffani CN (2004) Can we predict the effects of alien species? A case-history of the invasion of South Africa by Mytilus galloprovincialis (Lamarck). J Exp Mar Biol Ecol 300:189–215
Broitman BR, Szathmary PL, Mislan KAS et al (2009) Predator-prey interactions under climate change: the importance of habitat vs body temperature. Oikos 118:219–224
Bruno JF, Stachowicz JJ, Bertness MD (2003) Inclusion of facilitation into ecological theory. Trends Ecol Evol 18:119–125
Connolly SR, Roughgarden J (1999) Theory of marine communities: competition, predation, and recruitment-dependent interaction strength. Ecol Monogr 69:277–296
Dahlhoff EP, Menge BA (1996) Influence of phytoplankton concentration and wave exposure on the ecophysiology of Mytilus californianus. Mar Ecol Prog Ser 144:97–107
Dehnel PA (1956) Growth rates in latitudinally and vertically separated populations of Mytilus californianus. Biol Bull 110:43–53
Dinno A (2017) Conover.test: Conover-Iman test of multiple comparisons using rank sums. R package version 1.1.5 edn.
Duarte CM, Hendriks IE, Moore TS et al (2013) Is ocean acidification an open-ocean syndrome? Understanding anthropogenic impacts on seawater pH. Estuar Coasts 36:221–236
Fitzhenry T, Halpin PM, Helmuth B (2004) Testing the effects of wave exposure, site, and behavior on intertidal mussel body temperatures: applications and limits of temperature logger design. Mar Biol 145:339–349
Freitas V, Campos J, Fonds M et al (2007) Potential impact of temperature change on epibenthic predator-bivalve prey interactions in temperate estuaries. J Therm Biol 32:328–340
García Molinos J, Halpern Benjamin S, Schoeman David S et al (2015) Climate velocity and the future global redistribution of marine biodiversity. Nat Clim Chang 6:83
Gazeau F, Alliouane S, Bock C et al (2014) Impact of ocean acidification and warming on the Mediterranean mussel (Mytilus galloprovincialis). Front Mar Sci 1:62. https://doi.org/10.3389/fmars.2014.00062
Gosling E (2003) Bivalve molluscs: biology, ecology and culture. Wiley, Hoboken
Griffiths RJ (1981) Population dynamics and growth of the bivalve Choromytilus meridionalis (Kr.) at different tidal levels. Estuar Coast Shelf Sci 12:101–118
Harley CDG, Helmuth BST (2003) Local- and regional-scale effects of wave exposure, thermal stress, and absolute versus effective shore level on patterns of intertidal zonation. Limnol Oceanogr 48:1498–1508
Harley CDG, Connell SD, Doubleday ZA et al (2017) Conceptualizing ecosystem tipping points within a physiological framework. Ecol Evol 7:6035–6045
Helmuth B, Kingsolver JG, Carrington E (2005) Biophysics, physiological ecology, and climate change: does mechanism matter? Annu Rev Physiol 67:177–201
Helmuth B, Broitman BR, Blanchette C et al (2006) Mosaic patterns of thermal stress in the rocky intertidal zone: implications for climate change. Ecol Monogr 76:461–479
Hockey PAR, van Erkom Schurink C (1992) The invasive biology of mussel Mytilus galloprovincialis on the southern african coast. Trans R Soc S Afr 48:123–139
IPCC (2014) Climate Change 2014—impacts, adaptation and vulnerability: regional aspects. Working Group II contribution to the IPCC 5th assessment report. Cambridge University Press
Jager T, Ravagnan E, Dupont S (2016) Near-future ocean acidification impacts maintenance costs in sea-urchin larvae: identification of stress factors and tipping points using a DEB modelling approach. J Exp Mar Biol Ecol 474:11–17
Jurgens LJ, Gaylord B (2018) Physical effects of habitat-forming species override latitudinal trends in temperature. Ecol Lett 21:190–196
Kennedy VS (1976) Desiccation, higher temperatures and upper intertidal limits of three species of sea mussels (Mollusca: Bivalvia) in New Zealand. Mar Biol 35:127–137
Kooijman SALM (2010) Dynamic energy budget theory for metabolic organization. Cambridge University Press, Cambridge
Kroeker KJ, Gaylord B, Hill TM et al (2014) The role of temperature in determining species’ vulnerability to ocean acidification: a case study using Mytilus galloprovincialis. PLoS ONE 9:e100353
Lathlean JA, McQuaid CD (2017) Biogeographic variability in the value of mussel beds as ecosystem engineers on South African rocky shores. Ecosystems 20:568–582
Lathlean JA, Ayre DJ, Minchinton TE (2011) Rocky intertidal temperature variability along the southeast coast of Australia: comparing data from in situ loggers, satellite-derived SST and terrestrial weather stations. Mar Ecol Prog Ser 439:83–95
Lathlean JA, McWilliam RA, Pankhurst J et al (2017) Altering species interactions outweighs the effects of experimental warming in structuring a rocky shore community. J Exp Mar Biol Ecol 496:22–28
Leslie HM, Breck EN, Chan F et al (2005) Barnacle reproductive hotspots linked to nearshore ocean conditions. Proc Natl Acad Sci U S A 102:10534–10539
Lester SE, Gaines SD, Kinlan BP (2007) Reproduction on the edge: large-scale patterns of individual performance in a marine invertebrate. Ecology 88:2229–2239
Levine JM, Adler PB, Yelenik SG (2004) A meta-analysis of biotic resistance to exotic plant invasions. Ecol Lett 7:975–989
Lockwood BL, Somero GN (2011) Invasive and native blue mussels (genus Mytilus) on the California coast: the role of physiology in a biological invasion. J Exp Mar Biol Ecol 400:167–174
Marshall DJ, Dong YW, McQuaid CD et al (2011) Thermal adaptation in the intertidal snail Echinolittorina malaccana contradicts current theory by revealing the crucial roles of resting metabolism. J Exp Biol 214:3649–3657
McQuaid CD, Mostert BP (2010) The effects of within-shore water movement on growth of the intertidal mussel Perna perna: an experimental field test of bottom-up control at centimetre scales. J Exp Mar Biol Ecol 384:119–123
McQuaid CD, Phillips TE (2000) Limited wind-driven dispersal of intertidal mussel larvae in situ evidence from the plankton and the spread of the invasive species Mytilus galloprovincialis in South Africa. Mar Ecol Prog Ser 201:211–220
McQuaid CD, Porri F, Nicastro KR et al (2015) Simple, scale-dependent patterns emerge from very complex effects: an example from the intertidal mussels Mytilus galloprovincialis and Perna perna. In: Hughes RN, Hughes DJ, Smith IP, Dale AC (eds) Oceanography and marine biology: an annual review. CRC Press, Boca Raton
Mellin C, Lurgi M, Matthews S et al (2016) Forecasting marine invasions under climate change: biotic interactions and demographic processes matter. Biol Conserv 204:459–467
Monaco CJ, Helmuth B (2011) Tipping points, thresholds and the keystone role of physiology in marine climate change research. Adv Mar Biol 60:124–154
Monaco CJ, McQuaid CD (2018) Applicability of dynamic energy budget (DEB) models across steep environmental gradients. Sci Rep 8:16384
Monaco CJ, Wethey DS, Helmuth B (2014) A dynamic energy budget (DEB) model for the keystone predator Pisaster ochraceus. PLoS ONE 9:e104658
Monaco CJ, Wethey DS, Helmuth B (2016) Thermal sensitivity and the role of behavior in driving an intertidal predator–prey interaction. Ecol Monogr 86:429–447
Nicastro KR, Zardi GI, McQuaid CD (2010a) Differential reproductive investment, attachment strength and mortality of invasive and indigenous mussels across heterogeneous environments. Biol Invasions 12:2165–2177
Nicastro KR, Zardi GI, McQuaid CD et al (2010b) The role of gaping behaviour in habitat partitioning between coexisting intertidal mussels. BMC Ecol 10:1–11
Nicastro KR, Zardi GI, McQuaid CD et al (2012) Love thy neighbour: group properties of gaping behaviour in mussel aggregations. PLoS ONE 7:e47382
Olabarria C, Gestoso I, Lima FP et al (2016) Response of two Mytilids to a heatwave: the complex interplay of physiology, behaviour and ecological interactions. PLoS ONE 11:e0164330
Oyarzún P, Toro J, Garcés-Vargas J et al (2018) Reproductive patterns of mussel Perumytilus purpuratus (Bivalvia: Mytilidae), along the Chilean coast: effects caused by climate change? J Mar Biol Assoc UK 98:375–385
Pecl GT, Araújo MB, Bell JD et al (2017) Biodiversity redistribution under climate change: impacts on ecosystems and human well-being. Science 355:eaai9214
Petes LE, Menge BA, Murphy GD (2007) Environmental stress decreases survival, growth, and reproduction in New Zealand mussels. J Exp Mar Biol Ecol 351:83–91
Pfister CA, Esbaugh AJ, Frieder CA et al (2014) Detecting the unexpected: a research framework for ocean acidification. Environ Sci Technol 48:9982–9994
Phillips NE (2007) A spatial gradient in the potential reproductive output of the sea mussel Mytilus californianus. Mar Biol 151:1543–1550
Porter W, Mitchell J, Beckman W et al (1975) Environmental constraints on some predator-prey interactions. In: Gates D, Schmerl R (eds) Perspectives of biophysical ecology. Springer, New York, pp 347–364
R Core Team (2016) R: A language and environment for statistical computing. R foundation for statistical computing, Vienna, Austria
Reaugh-Flower K, Branch GM, Harris JM et al (2011) Scale-dependent patterns and processes of intertidal mussel recruitment around southern Africa. Mar Ecol Prog Ser 434:101–119
Rius M, McQuaid CD (2006) Wave action and competitive interaction between the invasive mussel Mytilus galloprovincialis and the indigenous Perna perna in South Africa. Mar Biol 150:69–78
Rius M, McQuaid CD (2009) Facilitation and competition between invasive and indigenous mussels over a gradient of physical stress. Basic Appl Ecol 10:607–613
Roberts DA, Hofmann GE, Somero GN (1997) Heat-shock protein expression in Mytilus californianus: acclimatization (seasonal and tidal-height comparisons) and acclimation effects. Biol Bull 192:309–320
Robinson TB, Griffiths CL, McQuaid CD et al (2005) Marine alien species of South Africa—status and impacts. Afr J Mar Sci 27:297–306
Russell BD, Harley CDG, Wernberg T et al (2011) Predicting ecosystem shifts requires new approaches that integrate the effects of climate change across entire systems. Biol Lett. https://doi.org/10.1098/rsbl.2011.0779
Sanford E, Menge BA (2007) Reproductive output and consistency of source populations in the sea star Pisaster ochraceus. Mar Ecol Prog Ser 349:1–12
Sarà G, Kearney M, Helmuth B (2011) Combining heat-transfer and energy budget models to predict thermal stress in Mediterranean intertidal mussels. Chem Ecol 27:135–145
Saraiva S, van der Meer J, Kooijman SALM et al (2011) Modelling feeding processes in bivalves: a mechanistic approach. Ecol Model 222:514–523
Schneider KR, Van Thiel LE, Helmuth B (2010) Interactive effects of food availability and aerial body temperature on the survival of two intertidal Mytilus species. J Therm Biol 35:161–166
Schneider CA, Rasband WS, Eliceiri KW (2012) NIH Image to ImageJ: 25 years of image analysis. Nat Methods 9:671–675
Seabra R, Wethey DS, Santos AM et al (2011) Side matters: microhabitat influence on intertidal heat stress over a large geographical scale. J Exp Mar Biol Ecol 400:200–208
Seabra R, Wethey DS, Santos AM et al (2016) Equatorial range limits of an intertidal ectotherm are more linked to water than air temperature. Glob Chang Biol 22:3320–3331
Shephard S, Beukers-Stewart B, Hiddink JG et al (2010) Strengthening recruitment of exploited scallops Pecten maximus with ocean warming. Mar Biol 157:91–97
Shinen JS, Morgan SG (2009) Mechanisms of invasion resistance: competition among intertidal mussels promotes establishment of invasive species and displacement of native species. Mar Ecol Prog Ser 383:187–197
Somero G (2011) Comparative physiology: a “crystal ball” for predicting consequences of global change. Am J Physiol Regul Integr Comp Physiol 301:R1–R14
Storey KB, Storey JM (1990) Metabolic rate depression and biochemical adaptation in anaerobiosis, hibernation and estivation. Q Rev Biol 65:145–174
Suchanek TH (1981) The role of disturbance in the evolution of life history strategies in the intertidal mussels Mytilus edulis and Mytilus californianus. Oecologia 50:143–152
Suchanek T (1992) Extreme biodiversity in the marine environment: mussel bed communities of Mytilus californianus. Northwest Environ J 8:150–152
Tagliarolo M, McQuaid C (2015) Sub-lethal and sub-specific temperature effects are better predictors of mussel distribution than thermal tolerance. Mar Ecol Prog Ser 535:145–159
Tagliarolo M, Montalto V, Sarà G et al (2016) Low temperature trumps high food availability to determine the distribution of intertidal mussels Perna perna in South Africa. Mar Ecol Prog Ser 558:51–63
Valdizan A, Beninger PG, Decottignies P et al (2011) Evidence that rising coastal seawater temperatures increase reproductive output of the invasive gastropod Crepidula fornicata. Mar Ecol Prog Ser 438:153–165
Van Erkom Schurink C, Griffiths CL (1992) Physiological energetics of four South African mussel species in relation to body size, ration and temperature. Comp Biochem Physiol A Physiol 101:779–789
Van Erkom Schurink C, Griffiths CL (1991) A comparison of reproductive cycles and reproductive output in four southern African mussel species. Mar Ecol Prog Ser 76:123–134
von der Meden C, Porri F, Erlandsson J et al (2008) Coastline topography affects the distribution of indigenous and invasive mussels. Mar Ecol Prog Ser 372:135–145
Wahl M (2008) Ecological lever and interface ecology: epibiosis modulates the interactions between host and environment. Biofouling 24:427–438
Walther G-R, Roques A, Hulme PE et al (2009) Alien species in a warmer world: risks and opportunities. Trends Ecol Evol 24:686–693
Widdows J, Shick JM (1985) Physiological responses of Mytilus edulis and Cardium edule to aerial exposure. Mar Biol 85:217–232
Zardi GI, Nicastro KR, McQuaid CD et al (2006) Hydrodynamic stress and habitat partitioning between indigenous (Perna perna) and invasive (Mytilus galloprovincialis) mussels: constraints of an evolutionary strategy. Mar Biol 150:79–88
Zardi GI, McQuaid CD, Nicastro KR (2007) Balancing survival and reproduction: seasonality of wave action, attachment strength and reproductive output in indigenous Perna perna and invasive Mytilus galloprovincialis mussels. Mar Ecol Prog Ser 334:155–163
Zardi GI, Nicastro KR, McQuaid CD et al (2016) Enemies with benefits: parasitic endoliths protect mussels against heat stress. Sci Rep 6:31413
Acknowledgements
This research was funded by the South African Research Chairs Initiative of the Department of Science and Technology and the National Research Foundation to CDM. CJM was supported by a Rhodes University post-doctoral fellowship. We are grateful to Carlota Fernández-Muñiz, Aldwin Ndhlovu, Diane Smith, and Jaqui Trassierra for assistance during laboratory and field work.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Monaco, C.J., McQuaid, C.D. Climate warming reduces the reproductive advantage of a globally invasive intertidal mussel. Biol Invasions 21, 2503–2516 (2019). https://doi.org/10.1007/s10530-019-01990-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10530-019-01990-2